![]()
STEVEN A. FRANK*‡AND BERNARD J. CRESPI†
Evolutionary conflicts cause opponents to push increasingly hard and in opposite directions on the regulation of traits. One can see only the intermediate outcome from the balance of the exaggerated and opposed forces. Intermediate expression hides the underlying conflict, potentially misleading one to conclude that trait regulation is designed to achieve efficient and robust expression, rather than arising by the precarious resolution of conflict. Perturbation often reveals the underlying nature of evolutionary conflict. Upon mutation or knockout of one side in the conflict, the other previously hidden and exaggerated push on the trait may cause extreme, pathological expression. In this regard, pathology reveals hidden evolutionary design. We first review several evolutionary conflicts between males and females, including conflicts over mating, fertilization, and the growth rate of offspring. Perturbations of these conflicts lead to infertility, misregulated growth, cancer, behavioral abnormalities, and psychiatric diseases. We then turn to antagonism between the sexes over traits present in both males and females. For many traits, the different sexes favor different phenotypic values, and constraints prevent completely distinct expression in the sexes. In this case of sexual antagonism, we present a theory of conflict between X-linked genes and autosomal genes. We suggest that dysregulation of the exaggerated conflicting forces between the X chromosome and the autosomes may be associated with various pathologies caused by extreme expression along the male–female
______________
*Department of Ecology and Evolutionary Biology, University of California, Irvine, CA 92697-2525; and †Department of Biosciences, Simon Fraser University, Burnaby, BC, Canada V5A 1S6. ‡To whom correspondence should be addressed. E-mail: safrank@uci.edu.
axis. Rapid evolution of conflicting X-linked and autosomal genes may cause divergence between populations and speciation.
Pathologies often arise from perturbations of evolutionary conflict. In conflict between different components of the genome, the opposing genes push in opposite directions on a particular trait, such as sex ratio or offspring growth rate (Burt and Trivers, 2006). The regulation of such traits under conflict becomes dominated by a balance of opposing forces. This precarious regulatory balance contrasts with the typically supposed design of regulation to achieve efficient and robust expression (Foster, 2011; Werren, Chapter 10, this volume). Mutation or knockout of one side in the conflict leads to the other side dominating expression, often pushing the trait to an extreme in the absence of the opposing force. Extreme expression typically causes pathology.
In this chapter, we develop the idea of pathology arising from perturbations to evolutionary conflicts. We discuss several examples of evolutionary conflicts, the ways in which conflict may lead to exaggerated opposition of forces on a trait, and the occasional breakdown in the normal balance of opposing forces that leads to pathology. We also present a theory of evolutionary conflict between X-linked and autosomal genes over traits that differ in their consequences for male and female fitness. Perturbations to the X–autosome conflict may lead to pathologies of extreme expression along a male–female continuum in trait expression.
The first section develops the general concept of pathology arising from evolutionary conflict. Although the evolutionary dynamics and mechanistic constraints vary greatly between cases, pathology seems likely to increase with the difference between the optimal phenotypic values favored by the conflicting parties. The difference in conflicting fitnesses sets the potential instability of regulatory control built from opposing forces. The degree of pathology in particular cases also increases with the rarity of pathological expression, because rarity reduces the intensity of selection. Weaker selection allows greater exaggeration of opposing forces between conflicting parties, creating greater instability and pathology when the uneasy balance between strongly opposing forces does break down.
The second section analyzes the pathology of mammals derived from growth-related conflicts between paternal and maternal components of the genome (Haig, 2010). Several regulatory control networks of growth do appear to be a conflict between exaggerated paternal enhancers of growth and opposing maternal brakes on growth rate. We consider pathologies arising from imbalances between these strongly opposing
forces (Úbeda and Wilkins, 2008). Overly aggressive growth may lead to cancer.
The third section extends our discussion of growth-related pathologies in mammals by considering morphological and behavioral pathologies. Overexpression of normally paternally expressed factors in humans associates with characters such as a protruding tongue, a wide mouth, and excessive feeding solicitation behavior by offspring. By contrast, overexpression of normally maternally expressed factors associates with characters such as growth hormone deficiency, low birth weight, lack of appetite, and poor sucking ability (Eggermann et al., 2008). We also discuss psychiatric pathologies that associate the paternally expressed tendencies with autism and the maternally expressed tendencies with psychosis (Crespi and Badcock, 2008).
The fourth section reviews antagonism between the sexes (Rice and Holland, 1997). Distinct male and female characters interact in mating and fertilization. The sexes often conflict because, in a mating, males push to increase the chance of fertilization success, to increase current female investment in the male’s offspring, and to reduce future female mating. Females may push back by resisting male control over fertilization, future mating, and patterns of maternal resource investment in different offspring. Perturbations to these conflicts may lead to infertility.
A different sort of antagonism between the sexes occurs when the same trait is expressed in both males and females, such as aspects of metabolism, physiology, or structure (van Doorn, 2009). Often, males and females are favored to express this common trait in different ways. To the extent that the trait cannot be modulated completely to different expression in the two sexes, natural selection favors a balanced expression of the trait that averages the best trait value in each sex. In some cases, there is no conflict, but rather an intermediate outcome between the divergent characters favored in males and females.
The fifth section presents our theory of X versus autosome conflict. For a trait expressed in both sexes, the autosomes typically favor an intermediate expression that weights equally the best trait expression in males and females. By contrast, the X chromosome favors an intermediate value that weights the trait expression favored by females twice as much as the trait expression favored by males. This conflict between the X chromosome and the autosomes can lead to exaggeration of the opposing forces and to pathology when perturbations disrupt the conflict.
We conclude by reiterating the importance of pathology in the study of conflict. Normally, one cannot see the strongly opposed forces in a conflict, because the observed trait typically reflects an intermediate balance that might be expected in the absence of conflict. Perturbation
of the conflict often leads to extreme expression and pathology (Burt and Trivers, 2006), revealing the hidden nature of evolutionary design.
MODEL OF OPPOSING FORCES
In this section, we summarize conclusions from a model of conflict. The model describes how a particular balance of opposing forces leads to a particular level of pathology when the balance is perturbed. We give the conclusions here and present the details of the model in Appendix A.
Fig. 13.1 shows the main concepts. Two parties, A and B, are in conflict, each with different optima for some character. The observed character value arises as an outcome of the opposing forces: B pushing for higher values, and A pushing for lower values. The opposing forces may become exaggerated as each side pushes harder against the other, with little net change in the outcome. As long as the opposing forces continue to balance, one often cannot see the underlying opposition that leads to a particular character value, such as a particular growth rate. However, when the force imposed by one party is knocked out, for example, by mutation, then the exaggerated force imposed by the other party may push the character value beyond its own optima. Such exaggerated expression, now revealed by the lack of opposition, may lead to a pathological character that is so extreme that it is disadvantageous to all parties.
The model in Appendix A develops these ideas of exaggeration and pathology in a simple way. The conclusions from the model are as follows: (i) Between conflicting parties, the greater the divergence of favored trait values is, the greater the tendency for a trait to be the outcome of a precarious balance between strongly opposed forces. (ii) The less frequently perturbations occur, the weaker the penalty against the pathologies that result from perturbation. A weaker penalty allows evolution of more extreme exaggeration for the conflicting forces and thus greater pathology when the balance is perturbed. (iii) The weaker the fitness consequence is for perturbation to a particular opposition of conflicting forces, the greater the opposition of forces becomes. The opposing forces diverge toward an ever more precarious balance until the consequences of pathology or other costs of exaggeration outweigh the tendency for opponents to push oppositely on the trait.
GROWTH PATHOLOGIES: CANCER
The paternally derived genes of a mammal may do better by enhancing early childhood growth at the expense of maternal survival. The paternal push for growth arises because the fathers of particular offspring
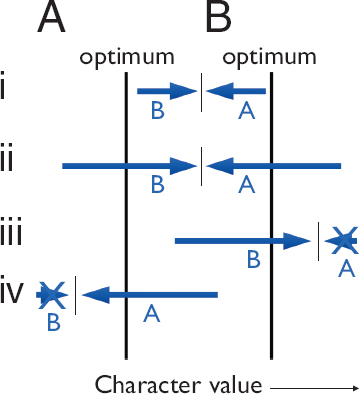
FIGURE 13.1 Pathology from evolutionary conflict. The conflict arises between two parties, A and B, which have distinct optima for some character. For example, A may be a mother and B a father, and the character value may be the growth rate of their child. In this case, the father favors a higher growth rate for the child than does the mother. (i) Party B pushes for higher character value, and party A opposes by pushing for lower character value. An observer often can see only the resolution measured as the character value that results from the hidden opposing forces. (ii) The resolution in i is not at either optimum, so B may push harder for an increase in character, which is then opposed by a stronger push by A in the other direction. This exaggeration of forces may be difficult to see, because the observed character value may be nearly unchanged under the stronger opposing forces that continue to balance at essentially the same level. (iii) The force imposed by A is knocked out. B’s force, now unopposed, may push the character value to a high level beyond B’s own optimum, causing a pathological outcome that is disadvantageous to all. (iv) A knockout of B, causing A’s unopposed force to push the character value too low, leading to pathology that is disadvantageous to both parties.
are frequently unrelated to other offspring produced by the same mother. By contrast, maternally derived genes may do better by slowing childhood growth to balance current offspring success against future maternal reproduction (Haig, 2010).
The opposition of parental interests can influence the regulatory networks that control growth. Several paternally derived genes exaggerate childhood growth rate; several maternally derived genes compensate by slowing growth (Haig, 2010). The net growth rate depends in part on how the conflict is resolved.
Epigenetic imprints of several growth-regulating genes appear to mediate the parental conflict over offspring growth (Fowden et al., 2011). The paternally derived allele may carry an imprint that silences expression, causing only the maternal allele to be expressed. Or the maternal allele may be imprinted and silenced, so that only the paternal allele is expressed.
The insulin growth factor gene IGF2 is maternally imprinted and paternally expressed. In mice, this gene is perhaps the most important stimulator of fetal growth and determinant of offspring size. The paternally imprinted and maternally expressed gene H19 produces a noncoding RNA associated with reduced expression of IGF2 and a lower rate of growth (Gabory et al., 2009). There appears to be a broad network of imprinted genes influencing growth in mice, in which the maternally expressed H19 acts to repress many growth-promoting components of the imprinted network (Gabory et al., 2009; Fowden et al., 2011). Several other imprinted loci affect growth. There is a tendency for growth stimulation to be associated with paternally expressed loci and growth repression to be associated with maternally expressed loci (Fowden et al., 2011).
The opposition of parental forces can lead evolutionarily to repeated enhancement of paternal pushing toward faster growth and repeated counterresponses of maternal pushing toward slower growth (Haig, 2010; Wilkins, 2011). To the extent that such opposition escalates over evolutionary history, the growth regulatory network becomes a precarious balance between strongly opposing forces that may be easily perturbed (Fig. 13.1). Such perturbations may lead to pathology (Úbeda and Wilkins, 2008; Haig, 2010).
Cancer is excessive growth. Thus, cancer may be a common pathology arising from perturbations to a precarious balance between strongly opposing growth promoters and growth repressors. Some evidence does connect perturbations of imprinted growth regulators to early stages of cancer progression (Lim and Maher, 2010; Monk, 2010).
Higher expression than normal of maternally silenced IGF2 or lower expression than normal of paternally silenced H19 or CDKN1C leads to a broad spectrum of overly rapid growth pathologies known as
Beckwith–Weidemann syndrome. The risk of certain childhood cancers, such as Wilms’ tumor and hepatoblastoma, is increased >100-fold in individuals with this rapid growth syndrome (DeBaun and Tucker, 1998). Other childhood cancers are also significantly increased in frequency (Rump et al., 2005), with a tissue distribution that closely matches that of typical sporadic childhood cancers. These excess, widely distributed cancers are consistent with the interpretation that an overly active IGF2 pathway exerts its growth effects broadly by stimulating cell replication in many tissues.
An indirect link between imprinting and childhood cancer comes from the association between higher birth weight, accelerated fetal growth, and higher rates of most of the major childhood cancers (Troisi et al., 2006; Milne et al., 2007; Laurvick et al., 2008; Callan and Milne, 2009; Samuelsen et al., 2009). To the extent that perturbations to imprinting can lead to misregulated growth, this association between growth and cancer may also link misregulated imprinting to cancer.
An inherited loss of the maternal IGF2 imprint causes a fivefold increase in human colorectal tumor risk (Cui et al., 2003). In a mouse study, knockout of the normal maternal IGF2 imprint led to expression of the maternal allele, increased IGF2 dosage, and higher sensitivity of the insulin growth factor signaling pathway (Kaneda et al., 2007). These growth-stimulatory changes in the IGF2 network may increase the number of intestinal progenitor cells at risk for progression or enhance the effects of other growth-promoting mutations (Kaneda and Feinberg, 2005). Somatic loss of imprinting for growth-promoting genes such as IGF2 has been associated with early stages in cancer progression (Feinberg et al., 2006).
The key question remains: How much of cancer pathology arises from perturbations to maternally and paternally opposed growth regulation? At present, the strongest hints come from the IGF2 network and from the fact that some other key cancer-related loci, such as RB1 associated with retinoblastoma and WT1 associated with Wilms’ tumor, are imprinted and are involved in growth (Dallosso et al., 2004; Buiting et al., 2010). These hints suggest that some fraction of cancer pathology may indeed come from growth-related conflicts. However, on the basis of the current evidence, the total cancer risk from growth conflict remains unclear.
The open problem concerns how deeply growth conflict and imprinting influence broad aspects of cellular proliferation. On the negative side, we have only a small number of known genes that fit. On the positive side, the number of genes that fit has increased steadily as data accumulate. It has been technically difficult to identify imprinted genes, leaving open the possibility that the known imprinted genes are just a small fraction of the total amount of imprinting.
With respect to the problem of identifying imprinted genes, Gregg et al.’s (2010) recent study is interesting. In their analysis of mouse brains, they estimated that >1,300 loci have the kind of parent-of-origin effects typical of imprinting. If widespread imprinting does in fact occur, then the conflicting interests of mothers and fathers over offspring growth may indeed lead to a growth regulation system precariously poised between strongly opposing forces. The pathologies from perturbations to a conflict-influenced regulatory design might contribute significantly to cancer risk.
GROWTH PATHOLOGIES: MORPHOLOGY AND BEHAVIOR
The previous section discussed how the mother–father conflict over offspring growth rate may lead to tissue-level pathologies and cancer. In this section, we follow the same conflict in relation to two syndrome pairs. We begin with the syndromes’ morphological and feeding-related pathologies. We then turn to psychiatric pathologies, which are more complex.
Morphology and Feeding-Related Behavior
The Beckwith–Weidemann syndrome (BWS) often associates with over-expression of the normally maternally silenced and paternally expressed IGF2 (Cohen, 2005). The opposing Silver–Russell syndrome (SRS) often arises by repression of IGF2 (Eggermann, 2010). Not all cases have a known direct association to IGF2. It is not clear whether those other cases derive primarily from different growth-related pathways or from unknown connections to regulation of IGF2 (Eggermann et al., 2008).
BWS individuals often have an enlarged tongue and high birth weight and height (Cohen, 2005). Other abnormalities, such as enlarged kidneys, may follow from a general tendency for rapid growth. Excess placental inclusions associated with rapid fetal growth occur. BWS individuals typically become adults of normal size and proportion, suggesting that the growth abnormalities are concentrated in the preweaning period associated with the primary demands on maternal resources. SRS individuals are small at birth and remain small through development, have significantly reduced subcutaneous fat, and have poor muscle tone (Eggermann, 2010). SRS babies typically lack interest in feeding and may have difficulty taking more than a small amount of food (Blissett et al., 2001). Growth hormone therapy is often an effective treatment.
The second pair of imprinted gene pathologies opposes Angelman syndrome (AS) and Prader–Willi syndrome (PWS). These syndromes associate with imprinted loci on the long arm of chromosome 15, although
other causes may be involved. AS typically associates with loss of the normally maternally expressed gene UBE3A of the ubiquitin pathway (Johnstone et al., 2006), whereas PWS individuals usually lose function of normally paternally expressed factors in the same chromosomal region (Haig and Wharton, 2003).
AS individuals often have a protruding tongue, a wide mouth, and excessive mouthing behavior (Dan, 2009). PWS individuals tend to have growth hormone deficiency and low birth weight (Haig and Wharton, 2003). Before the typical age of weaning at 2 or 3 years, they also lack appetite and have poor sucking ability, a weak cry, and a low activity level. After typical weaning age, they tend to overeat, perhaps associated with growth compensation derived from low size and weight at weaning age.
Overall, the two syndromes that are biased toward paternal expression, BWS and AS, have preweaning attributes associated with obtaining excess maternal resources. By contrast, the two syndromes that are biased toward maternal expression, SRS and PWS, have preweaning attributes associated with reduced acquisition of maternal resources.
The growth and feeding behavior of the two syndrome pairs fit well with the maternal–paternal conflict theory (Haig, 2010). By this theory, the design of regulatory control arises from opposition of forces rather than maximizing efficiency or enhancing robustness against perturbations. These syndromes may be the extreme expressions among numerous opposing forces in the regulation of preweaning growth and feeding behavior. If so, there may be a variety of potential perturbations leading to varying degrees of deviation from normal. Also, the breakdown of the normal paternal and maternal opposition of forces may lead to other pathologies besides mother–child resource transfer.
Psychiatric Pathologies
Crespi and Badcock (2008) suggested a continuum of psychiatric pathologies arising from the precarious balance between opposed maternal and paternal interests over maternal investment in each offspring. This theory of psychiatric pathology is more speculative than the growth-related pathologies, because complex mental aberrations are harder to quantify and are perhaps influenced by a broader spectrum of causes. In addition, severe pathologies can be difficult to relate to simple theories such as the interests of opposing parties in a conflict, because pathologies are by definition abnormal and maladaptive, favoring no clear interests with respect to design. Failure is always harder to parse than coherent design, because the logic that explains failure arises only from a full understanding of the forces that create normal design. In other words,
explaining the causes of pathology is hard. However, it is worth trying, because the causes of pathology lead back to the nature of design. And understanding cause is likely to be helpful in treatment.
To repeat: It is important to keep in mind that pathologies are abnormal and maladaptive. To give a simple example on the basis of the concepts illustrated in Fig. 13.1, suppose mother favors a trait associated with the quantity 10, and father favors 20. The mother might push toward the low end with a contribution that, by itself, causes a value of -15, and the father may respond with a push that, by itself, causes a value of 30. The opposing forces combine additively to a precarious compromise of 15, between the two favored values. However, a loss of the push by either side leads to a pathologically extreme outcome that is maladaptive for both parties.
Clearly, psychiatric pathologies do not sit along a single line of numbers. However, it is worthwhile to ask how much of pathology can be arrayed along an axis between the opposing forces of behavioral regulation favored by maternal and paternal interests.
The Crespi–Badcock (2008) theory defines a psychiatric pathology axis with autism at one end and psychotic disorders such as schizophrenia at the other end. By their theory, normal behavior arises from a balance between opposing forces. The balance arises mechanistically from the relative dominance between the “selfish” limbic and the “social” neocortical brain systems.
Paternally expressed genes tend to push for greater growth and enhanced demand on maternal resources associated with enhancement of placentation, growth factors, suckling, tongue, orofacial muscles, and engagement with mother in infancy. The paternally expressed push for relatively greater development may lead to excess limbic control, which motivates behavior underlying solicitation for food in infancy and, more generally, behaviors that may be regarded as primarily selfish or self-centered. Many paternally expressed genes influence the hypothalamus, a core component of the limbic system.
A paternal bias in imprinted gene expression most commonly arises from reduced expression of normally maternally expressed genes, as in AS (Dan, 2009). Paternal bias associates with relative dominance of limbic versus neocortical function, possibly causing overdevelopment of limbic self-centered behavior and underdevelopment of neocortical social aspects of behavior. Excess self-centered and reduced social behaviors associate with autistic spectrum pathologies. In addition, low IQ may arise because IQ develops in part from neocortical functions, which are relatively reduced when a paternal bias enhances limbic relative to neocortical control. Both AS and BWS associate with excess relative expression of
certain paternally expressed genes and an increased risk of characteristics associated with autistic behavior (Bonati et al., 2007; Kent et al., 2008).
A bias toward maternally expressed genes, as in PWS, may associate with increased dominance of the neocortex, enhancing social aspects of behavior sometimes to the extremes of pathology (Badcock, 2010). The definitions and delineations of those behaviors that are social or pathological remain somewhat vague at present, leading to difficulties of interpretation and controversy. According to Crespi and Badcock (2008), social hyperexpression associates with hyperdevelopment of language leading to auditory hallucinations, hyperdevelopment of self in a social context leading to megalomania, hyperdeveloped theory of mind leading to paranoia, amplification of social emotions of elation or depression, and other behaviors sometimes associated with psychosis, schizophrenia, bipolar disorder, and depression.
The example of PWS illustrates the connections between growth, offspring demand on maternal resources, and the mechanistic bases of psychiatric pathologies. In PWS, there is a great reduction in numbers of oxytocin-secreting neurons in the hypothalamus (Swaab et al., 1995; Muscatelli et al., 2000), apparently associated with reduced relative effects on brain development from paternal gene expression and greater relative effects from maternal gene expression. In adults, oxytocin has been called a natural “antipsychotic” (Caldwell et al., 2009) because it appears to connect people socially (Rosenfeld et al., 2010). PWS children do not bond normally with their mothers, and they are complacent and undemanding (Crespi, 2011). Mechanistically, the hypothesis is that a relative bias toward maternal gene expression caused by reduced paternal gene expression associates with lower oxytocin, weak attachment, relatively reduced limbic compared with neocortical functions, and dysregulation of social interactions and bonding. PWS associates with a greatly increased risk of psychosis, especially in cases caused by inheriting two copies of maternally derived chromosome 15 (Webb et al., 2008), presumably creating a maternal expression bias.
CONFLICT BETWEEN THE SEXES
The previous sections discussed conflict over offspring growth rate. In that case, the conflict occurs between maternally and paternally derived genes over the expression of traits within the offspring. In this section, we introduce two other types of conflict between the sexes, each type with its own structure of competing interests and expression of traits. This introduction reviews prior work on sexual conflict.
In the following section, we extend prior work with our own theory of conflict between the sexes. Our theory develops a conflict in
which X-linked and autosomal genes are favored to push in opposite directions on traits with different effects on male and female fitness.
Sexual Conflict: Sex-Limited Traits
Many traits arise from male–female interaction. Examples include the timing and frequency of mating and the processes of fertilization. These traits typically depend on the interaction between male and female characters, such as male courtship and female response to courtship. Each character involved in sexual interaction is often expressed only in one sex. Different male and female characters may be in conflict (Rice, 1984; Chapman, 2006).
For example, males express proteins in their seminal fluid that manipulate their mates’ reproductive physiology. A male can gain by pushing his mate to invest more in immediate reproduction associated with fertilization by that male’s sperm or by pushing his mate to reduce copulation frequency in the future with other males. Females may, in turn, gain by pushing against these male manipulations. That type of sexual conflict matches the structure of Fig. 13.1. The opposing male and female pushes on traits influencing mating and reproduction may become exaggerated. Pathology may occur when a perturbation blocks or alters expression by one of the opposing parties.
Numerous male and female characters conflict over mating and fertilization (Eberhard, 1996; Rice and Holland, 1997; Holland and Rice, 1998; Lew et al., 2006; Chapman, 2008). Infertility is perhaps the most likely type of pathology, arising from abnormalities in fertilization or mating (Lew et al., 2006). However, few studies have directly analyzed the role of conflicting, exaggerated sexual characters in pathology.
Sexual Antagonism: Traits Expressed in both Sexes
Many traits are expressed in both sexes, such as structural components and basic aspects of metabolism, physiology, and morphology. Although both sexes often express the same gene that influences a basic biochemical or structural function, the male and female optima for that trait will sometimes differ. Males may, for example, gain from diverting more resources to muscle growth; females may gain from diverting more resources to fat deposition.
Different male and female optima favor modulation of the trait separately in each sex, leading to sex-limited expression that moves the trait toward its distinct optimum in each sex (Lande, 1980; Rice, 1984). However, various constraints may prevent complete uncoupling of the trait expression between the sexes, or evolutionary dynamics may
take a long time to produce pure sex-limited expression. The degree of coupling, or correlation, between the sexes in the expression of the trait determines the degree of potential sexual antagonism (van Doorn, 2009; Rice and Chippindale, 2001).
In the case of a trait expressed in both sexes, current theory suggests that no conflict of interest occurs. Instead, for each individual gene influencing the trait, natural selection favors an averaging of the separate optima in males and females. This averaging of distinct optima is often called intralocus antagonism, to emphasize that the divergent selective pressures of male and female optima act simultaneously on the same locus (Rice and Chippindate, 2001; van Doorn, 2009).
From the perspective of a single locus, this averaging of distinct optima is like a situation in which individuals express the same trait in two distinct habitats. The favored trait value is an average of the trait values favored in each habitat. In the case of sexual antagonism, the gene lives alternately in the two distinct habitats of male and female bodies.
SEXUAL ANTAGONISM: A THEORY OF X
VERSUS AUTOSOME CONFLICT
The previous section reviewed the theory of sexual antagonism for a trait that is expressed in both sexes. In that case, each gene favors a trait that averages the distinct male and female optima. All genes on the autosomes favor an equal weighting of the male and female optima, because the reproductive value of those autosomal genes is the same in both sexes. By contrast, genes on the X chromosome favor weighting the female optimum twice as strongly as the male optimum, because X-linked genes in females have twice the reproductive value of X-linked genes in males.
The different weightings of male and female optima by autosomal and X-linked genes create a conflict of interest. Haig (2006a,b) briefly mentioned this conflict, but did not develop the consequences. To understand the consequences, consider that X-linked genes are selected to push more strongly toward the female optimum than are autosomal genes, and autosomal genes are selected to push more strongly toward the male optimum than are X-linked genes. With conflict, there is the potential for exaggeration, in which the conflicting parties push oppositely and increasingly hard on the trait. The resulting precarious balance may lead to pathology (Appendix B, notes on X inactivation and inbreeding).
More generally, conflicts driven by the different weightings of male and female fitness arise between various genomic subsets: Mitochondria favor the female optimum, X chromosomes favor a weighting of
two-thirds of the female optimum and one-third of the male optimum, autosomes favor equal weighting of the optima, and Y chromosomes favor the male optimum. Here, we develop the X–autosome conflict, but note that other genomic conflicts of this sort may also be important. For example, mitochondria push metabolic traits toward the female optimum and may therefore be opposed by other genomic components that push the regulation of metabolic traits toward the male optimum. Exaggeration and the potential for pathology may follow.
X versus autosome conflict has been discussed in a variety of situations, such as meiotic drive (Burt and Trivers, 2006). However, apart from Haig’s (2006a,b) brief comments, we could not find in the literature mention of the conflict between different genomic subsets, such as the X and the autosomes, over divergent male–female optima. Given the very simple logic of the conflict, it is not clear why the extensive discussions of sexual antagonism have not emphasized this particular aspect of X versus autosome conflict.
The evolutionary dynamics of sexual antagonism for a trait expressed in both sexes may explain the lack of discussion about X versus autosome conflict. The stable outcome, with the highest fitness, would be modulation of the trait to express differently in the two sexes. With sex-limited expression, each sex if favored to match the trait to its own optimum, and the conflict disappears.
The literature discusses extensively the evolutionary path to pure sex-limited expression and complete sexual dimorphism (Lande, 1980; Rice, 1984; van Doorn, 2009; Connallon and Clark, 2010). However, the data suggest that a significant correlation between the sexes remains for traits with divergent optima between the sexes (Chenoweth et al., 2008; Bonduriansky and Chenoweth, 2009; Poissant and Coltman, 2009; van Doorn, 2009; Poissant et al., 2010; Stewart et al., 2010). Such correlation can arise because constraints of regulation and expression prevent tuning of the traits separately in each sex. Alternatively, the constraints may slow the evolutionary path toward sex limitation sufficiently to maintain a balance between the rate at which sex-limited expression is enhanced and the rate at which new antagonisms arise. In any case, given the observed correlation between the sexes in traits for which sexual antagonism occurs, there is wide scope for X-linked versus autosomal conflict.
Any behavioral, metabolic, physiological, or structural trait with divergent male and female fitness will be subject to X–autosome conflict whenever traits are not completely tuned in each sex to achieve perfect sex-limited expression. To the extent that the conflict induces exaggerated and opposing forces by the X chromosome and autosomes, subsequent evolutionary change to enhance sex-limited expression may become more difficult to achieve. Thus, the conflict, once established, may
tend to be maintained because of the complexities in trait regulation induced by the conflict.
Observations have not previously been interpreted in light of this particular kind of X chromosome versus autosome conflict. The most obvious prediction is widespread interaction between X–linked and autosomal genes over sexually antagonistic traits, with the X–linked genes pushing toward the female optimum and the autosomal genes pushing toward the male optimum. However, it may be difficult to see those sorts of interactions in a particular population. If, for example, a particular pair of X-linked and autosomal genes interact as predicted, but lack polymorphism, their interaction would be hidden from observation.
Loss-of-function mutations or chromosomal duplications provide one type of perturbation that can lead to pathology and provide a window into the underlying genetic architecture of trait regulation. Our theory predicts a simple directionality along the male–female axis. X chromosomes push traits toward expression favored by females. Knockout of X-linked genes therefore tends to cause excess expression in the direction favored by males. Similarly, autosomes push traits toward expression favored by males. Knockout of autosomal genes therefore tends to cause excess expression in the direction favored by females.
The most interesting, and controversial, discussion of a male–female axis in the recent literature concerns differences in behavior. By that theory, extreme maleness associates with autistic characteristics (Baron-Cohen, 2009) and extreme femaleness associates with psychotic characteristics (Crespi and Badcock, 2008; M. Brosnan et al., 2010). Our theory predicts that X knockouts associate with extreme maleness. Thus, by the theory of a male–female behavioral axis, one would expect X-linked knockouts to be associated with autistic characteristics. To evaluate this hypothesis fully, one would have to estimate the relative number of genes influencing autism on the X chromosome and the autosomes and then show that the X carries a disproportionate share. Not enough data exist at present. Some intriguing hints of X-linked effects associated with autistic tendencies have been reported (Marco and Skuse, 2006). Other extremes along a male–female axis may also be evaluated with regard to our predictions about the alternative directions of pathology associated with X-linked and autosomal genes.
Hybridization between populations or species provides another sort of perturbation that can reveal the underlying genetic architecture of traits. Genes in conflict may tend to diverge relatively rapidly between populations (Frank, 1991; Hurst and Pomiankowski, 1991; Werren, Chapter 10, this volume). Upon hybridization, mismatched X-linked and autosomal genes may cause pathological expression of traits. Such pathologies in crosses between populations are referred to as hybrid incompatibilities.
Our theory predicts hybrid incompatibilities between X-linked and auto-somal loci. These X–autosome incompatibilities may be dispersed widely throughout the genome, because many traits may be subject to sexually antagonistic selection. Many observations suggest relatively rapid divergence of X chromosomes or widespread X–autosome incompatibilities in hybrids (Coyne and Orr, 2004; Carneiro et al., 2010; Lu et al., 2010).
CONCLUSIONS
Some traits are regulated by the opposition of conflicting forces. For example, early offspring growth in mammals balances the powerful opposing pushes of paternal enhancement and maternal slowing. These opposing forces appear to have become exaggerated by the conflict. Nonetheless, the typical outcome remains intermediate and apparently normal because the opposing forces come to a precarious balance. When a mutation or other block to one of the exaggerated forces occurs, the unopposed push in the opposite direction often causes a pathologically disrupted growth trajectory.
Increasing evidence supports this conflict interpretation for the regulation of early offspring growth in mammals. The interesting question is: How often is the evolutionary design of regulatory control dominated by the precarious balance of conflicting and exaggerated forces rather than by the efficiency and robustness of control? We do not know the answer to that question. In this paper, we reviewed theory for sexual conflicts that suggests opposing forces may be important for many characters. We also gave some examples of particular traits that may be regulated by conflict. Although those examples are preliminary with regard to empirical support, they do show the wide range of organismal characters and associated pathologies that may ultimately have to be understood in the light of evolutionary conflict.
From previous studies, conflicts have been invoked to explain childhood growth, excessive male-like or female-like characteristics, infertility from exaggeration of mating or fertilization traits, and psychiatric disorders of misregulated social behavior. Sexual differences are often the first kind of trait that can be studied with regard to strong contrasts, because male–female dimorphism can appear binary and relatively easy to identify. How many other traits follow the evolutionary path of exaggerated conflict and occasional pathology? Again, we do not know. However, it would certainly be worthwhile to consider the wide range of genomic conflicts and social conflicts that may be associated with pathologically disrupted genetic or social regulation. The normal and apparently cooperative working of genomes, insect societies, and other groups may be regulated in part by precariously balanced opposing forces.
How does conflict influence the design of regulatory control? Scant research has focused on that interesting question (Foster, 2011). Speculating briefly, genes that share common interests may be more cooperative when opposed by a group of genes with conflicting interests. For example, the paternally imprinted and maternally expressed genes TP73, RB1, and CDKN1C are all in the same regulatory pathway influencing the cell cycle (Boominathan, 2007; Buiting et al., 2010). In general, do genes with common interests often segregate into common pathways? And do genes with opposing interests tend to segregate into different pathways with opposing effects? Or, as with IGF2 versus IGF2R (Haig and Graham, 1991), do conflicting genes frequently interact directly within the same pathway, perhaps causing opposing tendencies in regulatory control?
In this paper, we also added to the theory of conflict. Previously, a variety of male–female conflicts were identified. For example, we reviewed the maternal–paternal conflict over offspring growth rate and the male–female conflicts over mating and fertilization. Our theory focused on the conflict between X chromosomes and autosomes. When a trait has different consequences for males and females, natural selection favors the sexes to express the trait differently. However, many traits of metabolism, physiology, and structure arise from a common genetic basis in the two sexes. Those traits may be difficult to tune perfectly to different expression in the sexes.
To the extent that expression is constrained to be correlated between the sexes, genes tend to favor an averaging of the best trait values in males in females. Our theory of conflict arises because autosomal genes tend to weight the sexes equally, whereas X-linked genes tend to weight females about twice as much as males. Once this sort of conflict occurs, the autosomal and X-linked genes may push in opposite directions on the trait, with the opposing forces becoming exaggerated. Once exaggerated, all of the tendencies for pathology and consequences of regulatory control arise that we have emphasized throughout. The X versus autosome conflict may be particularly important, because it applies to any trait with different optima in males and females. By contrast, the other sexual conflicts that we reviewed are usually confined to a particular type of trait, such as growth or mating. Thus, the X versus autosome conflict may be particularly associated with widely dispersed genetic interactions throughout the genome, providing another hypothesis for rapid evolution and hybrid incompatibilities between species involving the X chromosome.
In all cases of disrupted conflict, the particular disease pathologies are interesting in themselves. The ordering of the different human childhood overgrowth and undergrowth pathologies is the most obvious example. More speculatively, the ordering of psychosocial pathologies
such as autism and various psychoses may turn out to be an interesting component of psychiatric disease.
Beyond the explanation of particular diseases, pathologies are interesting because they reveal the underlying evolutionary design. In most individuals, the opposing forces precariously balance. One cannot see the underlying conflict. The conflict becomes apparent only upon perturbation and the observation of pathology. Once one recognizes the axis of conflict, it may be possible to order apparently different pathologies along that axis. The extreme pathologies at the opposite ends of the axis of conflict reflect the exaggerated pushes in opposing directions. Once we recognize the paired extremes and the underlying structure of normal regulation, we may begin to understand many graduations in the traits along the conflict axis. Pathology reveals design.
APPENDIX A: CONFLICT BETWEEN TWO INDIVIDUALS OVER
A TRAIT THAT INFLUENCES THE FITNESS OF BOTH PARTIES
We consider two parties in conflict over a trait (Fig. 13.1). To present the simplest case, suppose the final trait, x, is the sum of the contributions from the two parties, x = xA + xB. The first party has optimal trait value, mA, and the second party has optimal trait value, mB. The expected fitness of each party is given by
![]()
where i = A or B, allowing this single equation to describe the fitnesses of the two opposing parties.
The first two terms of the fitness equation describe a typical stabilizing selection function, in which the final trait x is favored to converge to the optimum mi, with fitness falling off quadratically from the optimum.
The last term of the fitness equation quantifies the penalty for opposition of forces acting on the trait. The penalty rises with the distance between the contributions of the two parties. That distance is weighted by a cost parameter, c, that scales the penalty for perturbation in relation to distance, and a probability parameter, p, that describes the probability that a perturbation occurs.
A perturbation may, for example, be the knockout of the contribution by one party, leaving the other party’s contribution as the sole determinant of the trait. Such a knockout affects fitness by moving the trait in relation to the optimum, mi, and by invoking the penalty that depends on the distance between the parties and the scaling, c.
Assuming no constraints on the traits, the optimum is
![]()
where m = (mA + mB)/2 is the midpoint between the opposing optima, j is the opposing party to i such that if i = A, then j = B, and vice versa, and b = a/4pc. The conclusions given in the main text follow.
APPENDIX B: CONFLICT BETWEEN X-LINKED AND
AUTOSOMAL GENES OVER A TRAIT WITH DIFFERENT
FITNESS CONSEQUENCES IN MALES AND FEMALES
Suppose, for a particular trait, that the fitness of a female is maximized at F*, and the fitness of a male is maximized at M*. Optimally, each sex would separately express its own maximal trait value in a sex-limited way. However, a certain fraction of trait expression may arise from genes that influence the trait in the same way in both sexes, creating a genetic correlation between trait values in males and females. If so, then divergent selection on these jointly expressed genes will pull in different directions in the two sexes. For the phenotypic contribution to trait expression shared by the two sexes and encoded by autosomal genes, natural selection typically favors the average of the optimal values in the two sexes. The simple averaging arises because the total reproductive value of autosomal genes is the same in the two sexes. In this case, there is no conflict of interest, because each autosomal gene weights the two sexes equally.
If both X-linked and autosomal genes influence the part of trait expression shared by the sexes, a conflict of interest occurs between the different components of the genome. The reproductive value of X-linked genes is twice as great in females as in males, compared with the equal reproductive value weighting of the two sexes by autosomal genes. Thus, X-linked genes pull toward the female optimum and, relative to the X, autosomal genes pull in the other direction toward the male optimum. Here, we present a simple model to illustrate this X versus autosome conflict. To keep things simple, we do not consider a full genetical model, but instead use a phenotypic model with reproductive value weightings. We also mention some interesting extensions with regard to X inactivation and inbreeding.
Let X and A be the contributions of X-linked and autosomal genes to the trait value. We assume that X makes the same contribution to male and female trait values independently of the fact that females have two X chromosomes and males have one. The ploidy normalization for the sexes may happen in various ways, and the particular mechanisms can have interesting consequences. Our initial description ignores those ploidy issues. Our discussion of X inactivation and inbreeding at the end
of this section hints at some important extensions to the theory that need to be studied further.
We start by writing the trait expressed in females as
![]()
where 1 - δ is the fraction of the trait that is not sex limited in expression and is controlled by a combination of X-linked and autosomal genes. The fraction that is sex limited, δ, is at the female optimum, F*. The distance between the actual trait expressed and the optimum is Tf – F*.
Quadratic Fitness
We write the fitness of a female as
![]()
where a = α(1 - δ)2, with α as the weighting of the fitness penalty for distance from the optimum trait value. The last term is a penalty for divergent contributions of the X and autosomal genes, as in Appendix A. The expression for male fitness, w M, is the same, replacing the female optimum F* by the male optimum M*.
The fitness of an autosomal gene is the average of the fitnesses of the females and males, wA = (1/2)w F + (1/2)wM, whereas the fitness of an X-linked gene is weighted twice as strongly toward females as males, wX= (2/3)wF + (1/3)wM.
We assume that the contributions of X and A are normalized with respect to ploidy differences, as mentioned above. With that assumption, we can find the evolutionarily stable strategy (ESS) values, X* and A*, by jointly maximizing the X-linked and autosomal fitnesses and solving for the ESS values. Without loss of generality, we can set F* = 0 and define d = a/5b and M’ = (5/24)M*, allowing us to write the ESS values as
![]()
These solutions show that the X-linked genes push toward the female optimum at F* = 0 and the autosomal genes push toward the male optimum at M*. As the relative cost of pushing on the trait, b, becomes small, d increases, causing exaggeration of the opposing forces.
Consequences of X Inactivation
If there is X inactivation of one X allele in females, then the situation is more complex. About 15% of genes on the human X chromosome escape inactivation, and another 10% of X-linked loci are variably expressed on inactive X chromosomes (Carrel and Willard, 2005). Thus, a significant number of X-linked loci may be expressed from both copies and may conflict with autosomes. Occasional diploid expression on the X is sufficient to create the conflict.
Among loci with complete X inactivation, different cells may inactivate different copies of the X. Thus, each cell may express only one of the X copies, but each individual female may express both copies. The consequences of inactivation for a particular phenotype depend on the particular tissue that controls the phenotype and the relative fraction of each X chromosome inactivated in that tissue. If there is sufficient mixture of expression of the two copies in the focal tissue, then the phenotypic consequences may in some cases be equivalent to diploid expression.
In certain cases, most of the focal tissue may express only one particular copy, or the phenotype may be dominated by one particular X copy. If so, we would need to account for three types of fitness classes for an X-linked gene: the copy of the gene in males, the expressed copy of the gene in females, and the silent copy of the gene in females. We have not done the full analysis of this model. Here are a few conjectures based on concepts from class-structured models (Taylor and Frank, 1996; Frank, 1998).
With no inbreeding, the conflict between X-linked and autosomal genes disappears with X inactivation, because, for each copy of an X linked gene, the probability that it is expressed in males or females is equal in each generation. In particular, there is a one-third chance of being in males and expressed, a one-third chance of being in females and expressed, and a one-third chance of being in females and unexpressed. With no inbreeding, an unexpressed allele has average fitness and so does not contribute to evolutionary change. (It is more accurate to say that the reproductive values of alleles in the two sexes are equal for autosomal loci and the reproductive value of alleles in females is twice that in males for X-linked loci, as above.)
If there is inbreeding, there will be a correlation between the expressed and latent trait values of the two X-linked copies in females. That correlation causes an unexpressed (inactivated or imprinted) X-linked copy to have its fitness associated with its own latent trait value, adding a further push toward the female optimum and creating once again a conflict between X-linked and autosomal genes, including X-linked loci subject to X inactivation.
Gaussian Fitness and Genetics
Many aspects of this preliminary phenotypic model deserve further study. We mention just two. First, the simple quadratic fitness function used here is a special case of a Gaussian fitness function, which becomes quadratic when the selective intensity is weak. For example, if we focus only on selection on the X chromosome by setting A = b = 0, and we rescale so that F* = 0 and M* = 1, then the expressions for Gaussian fitness functions are
![]()
where af and am are the selective intensities on females and males for deviations from each respective optimum. The ESS phenotype favored by the X chromosome maximizes w = (1/3)wM + (2/3)wF, which can be obtained by solving for X in
![]()
Similarly, the ESS phenotype favored by autosomes in the absence of contribution from the X chromosome maximizes w = (1/2)wM + (1/2)wF, which can be obtained by solving for A in
![]()
Typically, the X chromosome favors a phenotype relatively closer to the female optimum than that favored by the autosomes.
The second issue concerns the range of underlying genetic assumptions for which the ESS phenotypic model correctly expresses the key evolutionary forces. Such phenotypic models are generally accurate for alleles that contribute additively to phenotype, under the assumption of a continuous spectrum of mutational effects and when accounting for the possibility of alternative equilibria (Frank, 1998). By contrast, many genetic models of sexually antagonistic traits find significant complexities with respect to the dominance interaction patterns among alleles (Rice, 1984; Patten and Haig, 2009; Fry, 2010). Those genetical models did not analyze the X versus autosome conflict. So it remains an open question how the genetic complexities of dominance and polymorphism would play out in a model of interactions between X-linked and autosomal loci. Often, if one studies a polygenic model and allows a spectrum of allelic effect sizes and parameters of dominance and epistasis, the ESS phenotypic model captures reasonably well the long-term evolutionary forces of the polygenic model. However, the particular problem of X versus autosome conflict remains to be studied in full genetical detail.
ACKNOWLEDGMENTS
S.A.F.’s research is supported by National Science Foundation Grant EF-0822399, National Institute of General Medical Sciences Models of Infectious Disease Agent Study Program Grant U01-GM-76499, and a grant from the James S. McDonnell Foundation. B.J.C.’s research is supported by the Natural Sciences and Engineering Research Council.
This page intentionally left blank.
























