The workshop summarized in this volume was the third international meeting in a series sponsored by the Forum on Drug Discovery, Development, and Translation of the Institute of Medicine (IOM) to gather information from experts around the world on the threat of drug-resistant tuberculosis (TB) and how it can be addressed. The workshop was held April 18–19 and 21, 2011, in New Delhi, India, in collaboration with the Indian National Science Academy (INSA) and the Indian Council of Medical Research (ICMR).
The Forum held a foundational workshop in Washington, DC, in 2008. The summary of that workshop, Addressing the Threat of Drug-Resistant Tuberculosis: A Realistic Assessment of the Challenge: Workshop Summary (IOM, 2009), and the accompanying white paper (Keshavjee and Seung, 2008) provided background for and informed the development of four subsequent workshops in countries with a high burden of drug-resistant TB. The first international workshop in the series was held in Pretoria, South Africa, on March 3–4, 2010 (IOM, 2011a). The second international workshop was held in Moscow, Russia, on May 26–27, 2010 (IOM, 2011b). The final workshop in the series is being planned for China. Box 1-1 summarizes
____________________
1 The planning committee’s role was limited to planning the workshop, and the workshop summary has been prepared by the workshop rapporteurs as a factual summary of what occurred at the workshop. Statements, recommendations, and opinions expressed are those of individual presenters and participants, and are not necessarily endorsed or verified by the Forum or the Institute of Medicine (IOM) and they should not be construed as reflecting any group consensus.
Key Viewpoints from Previous Workshops
To set the stage for the workshop in India, Gail Cassell, Visiting Professor, Harvard Medical School, and Vice President of TB Drug Discovery, Infectious Disease Research Institute, provided an overview of selected key messages from the first three workshops held by the Forum in Washington, DC; Pretoria, South Africa; and Moscow, Russia (IOM, 2009, 2011a,b).
Global Surveillance of Drug-Resistant TB
According to Cassell, a clear message that emerged from these meetings is that the actual number of multidrug-resistant (MDR) TB cases is certain to exceed the 440,000 (range of 390,000 to 510,000) new cases estimated by the World Health Organization (WHO) to have occurred in 2008 (WHO, 2010b). Quality data on the incidence and prevalence of MDR TB are not always available for a country or region. Data from many countries are based on statistical modeling results rather than laboratory-based surveillance, often because the laboratories in countries with a high burden of MDR TB lack the capacity to test for susceptibility to second-line drugs.b
Pediatric Drug-Resistant TB
Existing MDR TB surveys rarely include children. Cassell noted that even when children are included, they generally are lumped together into broad age groups, a practice that obscures the profile of pediatric MDR TB. If South Africa is an indication of the situation in other countries, Cassell said, MDR TB in children is a significant problem. According to a 2008 study of 148 children who underwent drug susceptibility testing (DST) while being treated for TB at two hospitals in Johannesburg, 8.8 percent, or 13 children, had MDR TB (Fairlie et al., 2011). Of those 13 children, 53.9 percent were HIV-coinfected, and 10 children received appropriate treatment. Four children with MDR TB died within 0.1 to 4.0 months after the date of TB investigation. In other studies presented at the Moscow meeting, data for Argentina and Peru indicated that MDR TB represented 15.4 percent of 136 previously treated TB cases in children in Argentina and 23.6 percent of 360 previously treated TB cases in children in Peru (IOM, 2011b; Llerena et al., 2010; Wright et al., 2009).
The microbiological diagnosis of drug-resistant TB in children is a challenge as children often have paucibacillary disease (few bacilli in sputum for testing), and specimens for DST are difficult to obtain. Cassell
suggested that to measure infection in the pediatric population accurately, the presence of the organism in other types of specimens must be detectable in a more sensitive way.
Transmission of MDR TB
Cassell noted that another strong message from the South Africa workshop was that human-to-human transmission of drug-resistant strains of TB is much more common than previously appreciated. In the past, infection control has been overlooked because there was a belief that drug-resistant strains are not spread as easily from person to person as susceptible strains. Whereas in the 1970s and 1980s, most MDR TB appeared to result from a lack of patient compliance with treatment or sequential treatment regimens, transmission of MDR and extensively drug-resistant (XDR) TB strains appears to dominate today, as evidenced by experience in Shanghai, South Africa, Tomsk, and Lima (IOM, 2011a,b).
Transmission of drug-resistant strains among children also is occurring in South Africa. In the 2008 South African study noted above, only 4 of the 13 children diagnosed with MDR TB had known exposure to an adult with TB, and none of these adult contacts had MDR TB (Fairlie et al., 2011). “Spread in the pediatric population is an important public health issue,” said Cassell. Similarly, data presented at the Moscow workshop described 128 culture-confirmed pediatric cases in Colombia, South America. Almost all of these cases had never been treated, and most had no history of adult MDR TB contacts.
Diagnosis and Treatment of MDR TB
As discussed in a white paper prepared for the Washington, DC, workshop (Keshavjee and Seung, 2008), the number of patients receiving treatment for TB worldwide is small, and in many cases the treatment they are receiving is ineffective because it is not based on DST. Rather, patients have failed treatment with first-line drugs and therefore have been put on second-line drugs without the susceptibility of their TB strain to those drugs being known. In 2010, only 16 percent of global MDR TB cases estimated to exist among reported TB cases were actually enrolled in MDR TB treatment regimens (WHO, 2011a). It is also estimated that as of 2010, fewer than 5 percent of TB patients were being tested for MDR TB in most parts of the world (WHO, 2011a).
Cassell cited the views expressed by some speakers at previous workshops that while enhancing laboratory capacity might improve surveillance, it would be unlikely to affect individual patient treatment and thus would fail to affect the spread of drug-resistant strains. It is
unrealistic to think that in countries that currently have fewer than 1 laboratory per 10 million population, which is the case in most high-burden countries, sufficient resources and time would be available to scale up capacity quickly enough to have a major impact on rapid diagnosis and treatment, especially given that most patients are in remote settings. Countries need one laboratory per 5 million population to perform culture and DST, according to standards developed by WHO (2011a). Of 27 countries with a high burden of MDR TB, however, just 13 meet both of these standards (Armenia, Azerbaijan, Bulgaria, Estonia, Georgia, Kazakhstan, Kyrgyzstan, Latvia, Lithuania, Republic of Moldova, Russian Federation, South Africa, and Ukraine).
Recently introduced diagnostics and technologies in late-stage development increase the speed and sensitivity of diagnosis. GeneXpert, for example, is an impressive advance. But a diagnostic still is needed that can determine antimicrobial susceptibility quickly at the point of care so that patients can be managed appropriately. Also, new technologies still require laboratory infrastructure and have limited capability to detect MDR genes or to detect infection other than in sputum.
The three previous workshops also emphasized the importance of the procurement and distribution of high-quality drugs. Critical issues include the need for better data on drug quality, quality enforcement, quality strategies, and accurate demand forecasting.
One of the most urgent needs is to obtain accurate data on the existence of totally drug-resistant (TDR) TB, said Cassell, because only then will the rest of the world take notice of the problem and policy makers increase funding for its control. Striking new data from KwaZulu-Natal reveal the magnitude of the problem: in the studied population, 88 percent of cases identified as XDR TB were actually TDR.c Even under the best of circumstances—as has been the case in Tomsk (Keshavjee et al., 2008) and in Peru (Mitnick et al., 2008)—only 48 percent and 60 percent, respectively, of XDR TB cases are treatable, which means that 52 and 40 percent, respectively, are untreatable. Currently there are no consistent policies for dealing with patients whose TB is untreatable. Proof that the disease in these patients is untreatable may take months, during which time they may spread their resistant organisms to family members and others in the community, including health care workers.
Development of New Antibiotics
Successfully treating these patients will require not just one new antibiotic in the regimen but a combination of three to four new classes of antibiotics simultaneously. This represents an enormous financial and technical challenge requiring massive cooperation. Today the failure rate from the time of target identification to regulatory approval of a new drug is 90 percent. Half of drugs fail even in phase III clinical trials. The average cost of developing a new drug is more than $1.5 billion, and the average time for drug discovery and development from target identification to approval is 10 to 14 years. Both of these figures would probably be higher for TB drugs given the lack of infrastructure and point-of-care diagnostics in high-burden countries. Yet in 2010, the world was investing only $226.8 million in TB drug research and development from all sources (Treatment Action Group, 2011).
According to Cassell, the public perception is that TB remains a problem but that drugs are available to treat it. The reality is that MDR and XDR TB are increasing at a rapid rate. As noted, current estimates are that 440,000 new cases of MDR TB are occurring each year, which is not a large number compared with other unmet medical needs. However, the reality is that while the number of patients diagnosed with and treated for MDR TB is increasing globally, the majority of MDR TB patients are not diagnosed and not receiving treatment. Only 16 percent of the TB patients estimated to have MDR TB in 2010 were diagnosed and given appropriate treatment (WHO, 2011a,b; Zignol et al., 2012).
____________
a This box is based on the presentation of Gail Cassell, Visiting Professor, Harvard Medical School, and Vice President of TB Drug Discovery, Infectious Disease Research Institute.
b A report from WHO (2011a) released after the workshop indicates that 60 percent of countries currently have at least one direct and representative measurement of drug resistance among their TB patients. Despite overall global increases in the coverage of data on drug resistance, however, considerable uncertainty remains as to the actual levels of MDR TB among TB patients.
c Data provided via personal communication, June 22, 2011, with Kristina Wallengren, Acting Clinical Core Manager, KwaZulu-Natal Research Institute for Tuberculosis and HIV (K-RITH), Nelson R. Mandela School of Medicine, University of KwaZulu-Natal.
key viewpoints and findings from the workshops held previously in Washington, Pretoria, and Moscow.
The workshop in India brought together about 100 disease experts, community leaders, policy makers, and patient advocates from India, the United States, and other countries for 2 days of intensive discussions. While the workshop was specifically designed to address the current status of drug-resistant TB in India, the presentations and discussions were anchored in a framework reflective of the global experience with MDR TB. The aim of the workshop was to highlight key challenges to controlling the spread of drug-resistant strains of TB and to discuss innovative strategies for advancing and harmonizing local and international efforts to prevent and treat drug-resistant TB.2
HISTORY AND DIMENSIONS OF THE PROBLEM3
Evidence indicates that TB has plagued mankind since ancient times, said Prakash N. Tandon, Emeritus Professor, INSA, in his opening remarks at the workshop. A human skeleton from a Neolithic cemetery near Heidelberg, Germany, dating to 5000 BCE shows evidence of spinal TB. Of interest, said Tandon, is evidence in this skeleton of healing in the absence of any drugs. Egyptian skeletons dating back to 3500 BCE likewise show evidence of TB. Hymns in the Rigveda and Yajurveda indicate that the early Indo-Aryans were familiar with the disease in the second millennium BCE.
Today, an estimated 2 billion people, one-third of the global population, are infected with Mycobacterium tuberculosis (M.tb.), the bacterium that causes TB (Keshavjee and Seung, 2008). Spread through the air, this infectious disease killed 1.7 million people in 2009, or approximately 4,700 people each day (WHO, 2010a).
Although antibiotics developed in the 1950s are effective against a large
____________________
2 The National Institute of Allergy and Infectious Diseases (NIAID), U.S. National Institutes of Health (NIH), held a meeting focused on exploring opportunities for collaboration in TB drug discovery research on April 20–21, 2011, the 2 days following the IOM workshop, also in New Delhi. The NIAID meeting was cosponsored by the Department of Biotechnology, Ministry of Science and Technology, Government of India, and ICMR. Meeting objectives included sharing the latest scientific information on drug discovery research focused on combating MDR and XDR TB, discussing TB drug development needs and the ways in which biomedical research can contribute, and identifying partnership opportunities to advance and accelerate new drug discovery efforts in order to simplify and improve therapeutic options for drug-resistant TB. Topics and meeting participants overlapped between the NIAID and IOM meetings in India, creating synergies and connections for future collaborations in the areas of TB research and policy. Appendix B of this report includes a summary of the NIAID meeting.
3 This section and the two that follow are based on the welcoming remarks of Prakash N. Tandon, Emeritus Professor, INSA; Krishan Lal, President, INSA; and Vishwa Mohan Katoch, Director General, ICMR.
percentage of TB cases, resistance to these first-line therapies has developed over the years, resulting in the growing emergence of MDR and XDR TB (see Box 1-2 for definitions). Diagnosing and effectively treating MDR and XDR TB patients requires increasingly complex public health interventions. MDR TB, for example, is resistant to first-line drugs and must be treated with second-line drugs that are more expensive and more toxic, often require injection, and involve longer treatment regimens (2 years or more to treat MDR TB compared with 6–9 months to treat drug-susceptible TB). As drug resistance develops, the challenge is to stop the transmission or spread of MDR TB and identify MDR TB cases early; treatment should include efforts to preserve the effectiveness of current drugs and create new treatment regimens to combat drug-resistant strains as they emerge.
THE BURDEN OF DRUG-RESISTANT TB
According to data from WHO on global drug resistance, an estimated 3.6 percent of global incident (new) TB cases, or a total of 440,000 cases, were MDR TB in 2008 (95 percent confidence interval, 390,000-510,000) (WHO, 2010c).4 The available data on drug-resistant TB are inadequate, however, and lead to an underestimation of the true global burden of MDR TB. In many developing countries where the MDR TB burden is likely to be significant, surveillance systems do not exist or lack the capacity to generate reliable data. Even the most recent global surveillance data on MDR TB do not include 79 countries—41 percent of all countries in the world (WHO, 2010c, p. 6).
The burden of XDR TB is even less well known because many countries lack the laboratory and infrastructure capacity necessary to test MDR TB patients routinely for susceptibility of their infection to second-line drugs. The provision of optimal patient care for MDR and XDR TB patients is based on DST, and many countries are ill equipped to conduct such tests. It is through such testing that physicians determine which drugs are likely to be effective against a particular drug resistance profile. The vast majority of MDR and XDR TB cases are undetected and thus untreated with appropriate second-line drugs. Of those patients who are treated with second-line drugs, many are not taking the right drugs to treat their drug resistance profile effectively.
____________________
4 Instead of providing a global estimate of incident MDR TB cases each year, an updated WHO (2011a) report on TB control, released after the workshop, estimates the prevalence of MDR TB (number of cases) globally. According to that report, an estimated 650,000 MDR TB cases existed among the world’s 12 million cases of TB in 2010. (Prevalence measures the level of a disease in a population at a particular point in time, while incidence measures the occurrence of new cases of a disease in a population.)
Definitions
Multidrug-resistant (MDR) TB is caused by bacteria resistant to isoniazid and rifampicin, the two most effective first-line anti-TB drugs, originally developed and introduced in the 1950s and 1960s.
Extensively drug-resistant (XDR) TB is resistant to the same drugs as MDR TB (isoniazid and rifampicin), as well as any fluoroquinolone (levofloxacin, moxifloxacin, or ofloxacin) and at least one second-line injectable drug (kanamycin, amikacin, or capreomycin).
Totally drug-resistant (TDR) TB is TB for which no effective treatments are available.
Pathways for Infection
MDR/XDR TB results from either primary infection with a drug-resistant strain of TB (i.e., transmitted by person-to-person contact) or acquired infection with such a strain that occurs in the course of a patient’s treatment, resulting, for example, from failure to ensure regular treatment with high-quality existing drugs. Amplified resistance, or the enhancement of existing drug resistance as a result of initiating an inappropriate drug regimen at the beginning of care, is a significant challenge
The diagnosis of TB is no longer a death warrant, said Krishan Lal, President, INSA, but the existence of treatments raises sociological and psychological issues. Patients may take a treatment just until they feel well, which can foster the development of resistance and lead to the spread of the disease. In addition, many health problems other than TB, such as diabetes and high blood pressure, occur in India, which can complicate treatment. The lack of quick, accurate, and inexpensive tests for drug-resistant TB hampers treatment, said Tandon. Drug-resistant TB needs to be diagnosed earlier and with greater specificity than is currently the case, especially given the much greater costs of treating drug-resistant TB.
created by providing an incorrect combination of drugs. For example, a patient might display resistance to streptomycin and isoniazid at the beginning of treatment and subsequently become resistant to streptomycin, isoniazid, and rifampicin during the course of treatment. Even when an empirically appropriate drug regimen is selected at the beginning of treatment, by the time drug susceptibility information is available, resistance may be amplified.
WHO and the International Union Against Tuberculosis and Lung Disease (IUATLD) have urged replacement of the term “primary resistance” with “drug resistance among new cases” and the term “acquired resistance” with “drug resistance among previously treated cases.”
Treatment
MDR/XDR TB treatment requires 2 years or more of daily, directly observed treatment (DOT) with drugs that are less potent, more toxic, and much more expensive than those used to treat drug-susceptible TB. Despite the challenges, aggressive treatment with second-line drugs has produced positive outcomes in MDR/XDR TB patients. However, TDR TB is a growing threat. The spread of TDR TB is especially ominous as it would return the globe to the pre-antibiotic era (Keshavjee and Seung, 2008).
____________
a The information in this box was originally presented at the Forum’s 2008 workshop on drug-resistant TB (IOM, 2009).
India has in the past had great success in tackling major health problems such as leprosy, observed Vishwa Mohan Katoch, Director General, ICMR. The country has instituted a massive program to deliver drugs to TB patients, but the disease also needs to be monitored and managed very carefully. Comprehensive approaches, such as those reflected in the agenda of this workshop, are essential, V. M. Katoch said.
Tandon, Lal, and V. M. Katoch praised the extent of international collaboration in responding to drug-resistant TB, especially the collaboration between India and the United States. Both countries are members of the Global Network of Academies, Lal observed, and this organization also has worked with the InterAcademy Medical Panel. Such collaborations will be essential, he said, for evolving strategies to fight TB.
OVERVIEW OF TB AND MDR TB IN INDIA5
In his opening keynote address, K. Srinath Reddy, President, Public Health Foundation of India, provided a broad overview of TB in India and the nation’s response to the disease. (Chapter 2 covers these topics in greater detail.)
India accounts for approximately one-fifth of the global incidence of TB (RNTCP Status Report, 2011). Fully 40 percent of the country’s population is infected with the tubercule bacillus. Each year the country sees 2 million new cases (the global incidence is 9.4 million), which lead to 280,000 deaths annually, although the prevalence of HIV among new cases in India is just 6.4 percent compared with a global average of 12 percent. TB is one of the leading causes of death among adults in India, and it also takes a large toll on the country’s younger generation, which makes up a significant proportion of the total population.6 TB also takes a disproportionately large toll among young females: more than 50 percent of TB cases among females occur before age 34, and an estimated 100,000 women are rejected by their families every year because they have the disease. Some workshop participants noted that national-level, all-India studies evaluating the effect of a TB diagnosis on family dynamics could provide more specific data and have an impact on understanding and preventing the rejection of TB patients by their families.
TB also disproportionately affects the poorest and most marginalized populations in India, as well as people in their most productive ages—70 percent of TB patients are aged 15–54. People with TB incur an average potential loss of 20–30 percent of their annual household income as a result of 3–4 months of lost work time. In India, about 14 million people fall into poverty each year because they experience unaffordable health care costs, and TB is a major cause of health-related impoverishment.
Drug-Resistant TB in India
Reddy noted that, based on 2008 data, MDR TB represents an estimated 2.3 percent of new TB cases in India (compared with 3.3 percent
____________________
5 This section is based on the presentation of K. Srinath Reddy, President, Public Health Foundation of India.
6 According to the 2001 Indian census, the country has a large proportion of young people—35 percent of the population is aged 14 and younger (Government of India, 2001). Provisional population totals from the 2011 Indian census reveal a total population of 1.21 billion people, reflecting an additional 181 million people since 2001. The United Nations has estimated that the world population grew at an annual rate of 1.23 percent from 2000 to 2010. Over this decade, China’s population grew at an annual rate of 0.53 percent and India’s at an annual rate of 1.64 percent (Government of India, 2011).
worldwide) and 17 percent of retreatment cases. These figures represent about 99,000 MDR TB cases in the country.
XDR TB has been reported in India. However, its magnitude remains undetermined because of a lack of laboratories capable of conducting quality-assured second-line DST.7
The Revised National TB Control Program
The United Nations’ Millennium Development Goals call for halting and beginning to reverse the incidence of TB by 2015. The STOP TB Partnership has established the target of reducing the global burden of TB (defined by per capita prevalence and death rates) by 50 percent relative to 1990 levels by 2015 and the long-term goal of reducing the global incidence of active TB to less than 1 case per million population per year by 2050.
India’s strategy for working toward these goals is embodied in its Revised National TB Control Program (RNTCP). This program is structured around five elements:
1. political and administrative commitment;
2. good-quality diagnosis, primarily by sputum smear microscopy;
3. an uninterrupted supply of quality drugs;
4. DOT; and
5. systematic monitoring and accountability.
A massive expansion of the program began in 1998, so that by 2006, Directly Observed Treatment-Short course (DOTS) coverage had been extended to 632 districts and more than 1.1 billion people.
In 2010, DOTS-Plus services were introduced in some states of India to treat MDR TB. By 2012, these services will have been extended to all smear-positive retreatment cases and to new cases that have failed an initial first-line drug treatment. By 2015, services are to be made available to all smear-positive pulmonary TB cases registered under the program. By 2012–2013, the program’s goal is to treat at least 30,000 MDR TB cases annually. Providing DOTS-Plus for MDR TB requires giving special attention to several key factors in program design and delivery:
____________________
7 The emergence of what has been described as TDR TB was reported in January 2012 (Udwadia et al., 2012) at Hinduja Hospital in Mumbai when four patients were found to be resistant to all first- and second-line drugs tested. India’s Revised National TB Control Program (RNTCP) has issued a response to the report and provided information on the program’s approach to combating all forms of drug-resistant TB. For more information, visit http://tbcindia.nic.in/pdfs/RNTCP%20Response%20DR%20TB%20in%20India%20-%20Jan%202012%20update.pdf (accessed April 17, 2012).
- quality-assured laboratory capacity for smear, culture, and drug sensitivity testing;
- treatment design;
- adherence to difficult-to-take regimens for long periods of time;
- management of side effects;
- drug procurement;
- recording and reporting; and
- human and financial resource constraints.
As of the end of 2010, MDR TB treatment had been scaled up to cover 287 million people in 139 districts across 12 states of India. Since the inception of services, more than 19,000 suspected MDR TB cases have been examined for diagnosis, more than 5,000 cases have been confirmed, and more than 3,500 cases have been initiated on category IV treatment through 20 DOTS-Plus sites.
Reddy noted that in general, India has a strong national program for basic TB control with a high treatment success rate. The country has made provisions for the participation of all health care providers, including private practitioners. Efforts also have been made to control the dual epidemics of TB and HIV (see the discussion of this topic in Chapter 6). Extensive laboratory expansion is planned in collaboration with the private sector, which has to date been a largely untapped resource in the development of laboratory capacity. (The expansion plans for the program are described in Chapter 2 and public–private participation in Chapter 7.)
Challenges in the Management of MDR TB in India
Several challenges are faced in the management of MDR TB in India, said Reddy. First is the limited supply of human resources to carry out training and assessments. Indeed, India is a country where in general, human resources in health care are limited and not well distributed. According to Reddy, the country’s public health workforce needs to be expanded, and those who are currently employed need training to augment their knowledge and skills.
Second, there is a lack of funding for the management of MDR TB, especially given the high cost of second-line drugs as treatment is scaled up. A high-level expert group recently recommended that a larger portion of the country’s health budget be allocated to providing drugs free of cost and that the national capacity to produce and supply low-cost drugs, including public procurement, be enhanced (Planning Commission of India, 2011). Other workshop participants noted that policy implementation delays will negatively affect MDR TB cases—diagnosed and undiagnosed.
Third, laboratory capacity for diagnosis and follow-up of MDR TB
patients and quality assurance is limited. Although expansion currently is taking place, India now has only 23 functional laboratories across the country, and according to Reddy, many more are needed. The availability of second-line drugs and DST also is limited. High-throughput diagnostics and a specimen transportation infrastructure are particular needs. An MDR TB surveillance or survey program still does not exist, and an infection control plan is lacking. Reddy stressed that all of these issues need to be addressed and that managerial capacity must be improved as well. India needs a strong vertical program that integrates all aspects of MDR TB control and care, including infection control, diagnosis, treatment, and follow-up. However, Reddy added, it is difficult to impose a strong vertical program on a weak national health system.
The Need for Action
The prevalence of MDR and XDR TB in India and globally raises the possibility that the current epidemic of mainly drug-susceptible TB will be replaced by a form of TB with severely restricted treatment options, Reddy observed. If so, plans to move toward a world where TB is no longer a public health problem will be derailed.
Reddy stressed that the basic TB program in India needs to be strengthened to reach out to unnotified and missed cases and to poor and highly vulnerable populations. More broadly, the social determinants of TB need to be addressed. The Public Health Foundation of India is currently working with the RNTCP to assess the barriers experienced by vulnerable groups in accessing services and determine how those barriers can be overcome.
In terms of surveillance, India’s Department of Health Research needs to supplement efforts currently in place, particularly for MDR and XDR TB, said Reddy. Laboratory networks need to be strengthened and expanded, human resources and financial management need to be enhanced, and the drug supply chain needs to be strengthened. Reddy also noted that the National Board of Examinations is currently connected to more than 700 hospitals that are distributed across the country and have postgraduate trainees who are expected to conduct research. According to Reddy, “If at least 100 of these hospitals can be linked up and their surveillance changed to regularly report on issues related to MDR TB and XDR TB—in terms of detection as well as management and outcomes—we can develop a centralized surveillance system across the country extending to medical colleges.” In this way, India could quickly build a cost-effective surveillance system that would be nationally representative.
Policies should ensure that all TB patients have equitable access to care and that their interests and rights are protected, said Reddy. Policies also should ensure that all relevant public and private health care providers are
engaged in managing TB according to national priorities. And the primary health care system should be strengthened to ensure early detection, effective treatment, and support for patients.
Reddy emphasized that TB medicines should be sold by prescription only and should be prescribed and dispensed by accredited public and private providers. He noted that in India today, drugs often are sold over the counter instead of through appropriate prescriptions, a situation that can foster misuse. Infection control policies are needed, and investments should be made to promote research, surveillance, molecular diagnostics, and drug development.
Reddy concluded by lauding the collective commitment to combating drug-resistant TB, “which now transcends geographical barriers and also brings scientific coalescence from multiple disciplines.” In this context, he suggested, “this workshop should be a landmark for initiating action against MDR TB and XDR TB.”
To set the stage for the remainder of the workshop, Salmaan Keshavjee, Assistant Professor, Harvard Medical School, provided a global overview of the challenges and potential solutions in confronting drug-resistant TB (discussed in detail in Chapter 3), with an emphasis on the slow pace of treatment scale-up and the consequences of inaction. He urged the participants “to be blunt with each other, because we are TB experts, and if we can’t be honest with each other, we are going to have big trouble as we move forward.” To this end, throughout the workshop, there was discussion of existing bottlenecks and challenges, as well as examples of incentives and disincentives from other locations or experiences. Experiences with both success and failure were shared.
In 2006, the STOP TB Partnership established a plan for combating the epidemic of drug-resistant TB. At that time, WHO was estimating that about 500,000 new cases of drug-resistant TB occurred each year, meaning that between 2006 and 2015 there would be 5 million cases. The goal of the 2006 plan was to treat 1.6 million cases, said Keshavjee, leaving the other 3.4 million without treatment; he referred to this target as “dismal.”
Since that plan was established, official efforts to treat MDR TB have failed to achieve even this target, Keshavjee stated. Of the 5 million cases estimated to have occurred between 2000 and 2009, only approximately 22,000 patients received treatment through programs approved by the Green Light Committee (GLC)—a multilateral coalition
____________________
8 This section is based on the presentation of Salmaan Keshavjee, Assistant Professor, Harvard Medical School.
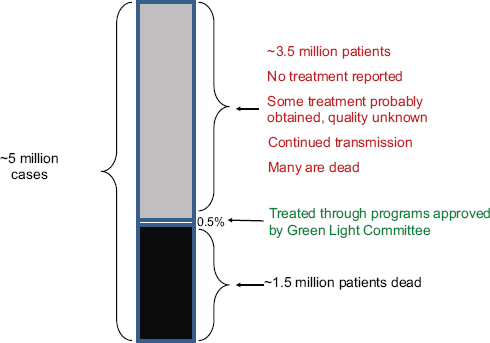
FIGURE 1-1 Of the estimated 5 million MDR TB cases that occurred between 2000 and 2009, only 0.5 percent were treated in programs approved by the Green Light Committee.
SOURCE: Keshavjee, 2011a.
created in 2000 (Figure 1-1). About 3.5 million of the 5 million patients received no reported treatment, although an unknown fraction undoubtedly received some treatment of unknown quality from private physicians, pharmacies, or other sources. Meanwhile, many continued to transmit the disease while alive. According to global statistics, 1.5 million of these 5 million people died. Keshavjee emphasized that this is an epidemic of profound proportions.
Scaling up treatment for drug-resistant TB is complex, Keshavjee acknowledged. But treating HIV in poor countries is also complex, and the global community has done a much better job of that. Between 2004 and 2008, the U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) provided antiretroviral therapy to more than 1.6 million people, including 367,000 patients who were coinfected with HIV and TB. The contrast between the responses to TB and HIV, said Keshavjee, calls for a rethinking of the global approach to TB.
This report summarizes the main points made at the workshop in India during both the formal presentations and the discussions among participants. In accordance with IOM and National Research Council policies, this report provides an accurate summary of the presentations and discussions held at the workshop; it does not contain any commentaries or views that were not presented at the workshop, and any supplementary viewpoints shared outside of the workshop context are not included in this summary. Observations and recommendations made by individual speakers and participants do not represent the formal positions of the planning committee, the Forum, the IOM, INSA, or ICMR; however, they have provided valuable input to the Forum and to the IOM and the workshop contributors as they deliberate on future initiatives. Presentations at the workshop addressed the following topics:
- TB and MDR TB in India, including local and national responses to the epidemic (Chapter 2);
- the global burden of TB and drug-resistant TB, including data from another high-burden country, China (Chapter 3);
- prevention of the transmission of drug-resistant TB in India (Chapter 4);
- rapid methods of detecting drug resistance and strengthening laboratory capacity (Chapter 5);
- approaches to reaching vulnerable populations affected by drug-resistant TB (Chapter 6);
- public–private engagement and innovative methods in combating drug-resistant TB (Chapter 7);
- the drug supply chain for second-line drugs (Chapter 8); and
- the major viewpoints expressed at the workshop and next steps suggested by workshop participants (Chapter 9).
Each of these chapters opens with a box listing the key messages emerging from the workshop presentations and discussions, as identified by the workshop rapporteurs.
















