Confronting Challenges to the Supply Chain for Second-Line Drugs
Key Messages
- The global marketplace for second-line drugs to treat drug-resistant TB is not operating efficiently or effectively.
- The cost of stockpiling or failing to use second-line drugs demands careful attention because it creates issues with the supply chain.
- Harmonizing quality standards and treatment regimens could attract additional suppliers of second-line drugs, create competition, and reduce prices.
- It could be beneficial to quantify the risks associated with supplying second-line drugs through more effective forecasting, aggregated to reduce country-by-country exposure of suppliers to risk and shared among countries to create a transnational market for these drugs.
MDR TB cannot be controlled without an adequate, uninterrupted supply of second-line drugs. But these drugs are expensive, which means they must be used effectively. Moreover, the markets for these drugs are relatively small and uncertain, making suppliers reluctant to commit resources to their manufacture. The resulting high prices exacerbate shortages and strain national TB programs.
One session at the workshop focused specifically on challenges to delivering quality-assured second-line drugs to patients. Presenters delineated the specific challenges at each step in the supply chain; considered
current methods of drug procurement in India and the implementation of the country’s national MDR TB program; described lessons learned from experience with HIV/AIDS initiatives with respect to increasing the number of suppliers and reducing the prices of second-line anti-TB drugs; and explained the need to quantify, aggregate, and share risks to improve the marketplace for second-line drugs.
CHALLENGES IN DRUG SUPPLY CHAIN LOGISTICS1
DOTS programs have been focused on reducing stockouts, in which drugs become unavailable. Less risk is associated with overstocking first-line than second-line drugs because the former are relatively inexpensive, noted Prashant Yadav, Senior Research Fellow, and Director of Healthcare Research, William Davidson Institute, University of Michigan. For second-line drugs, overstocking can waste scarce financial resources that could otherwise be used for expanding MDR TB programs. At the same time, an uninterrupted supply of second-line drugs is imperative if an MDR TB program is to be successful. MDR TB programs therefore must maintain a fine balance between effectiveness and efficiency.
Yadav highlighted six challenges that arise in the supply chain for second-line drugs, from grant disbursement to distribution. The descriptions below correspond to the challenges numbered 1 through 6 in Figure 8-1.
Uncertainties in the Timing of Grant Disbursement
The flow of funds, whether from a ministry or a Global Fund grant, is often uncertain. When financing is uncertain, procurement planning is difficult. For example, stringent procedures must be followed before Global Fund grants can be disbursed, which often leads to delays in disbursement, said Yadav. Similarly, Ministry of Health budgets often are subject to delays in the release of funds.
Unpublished data show that among the categories of drugs purchased using Global Fund grants, the time gap between planning a purchase and receiving a shipment is greatest for TB drugs. According to Yadav, this gap also is higher in India than in the surrounding region and compared with the global average.
If it is difficult to predict when funds will become available, procurement
____________________
1 This section is based on the presentation of Prashant Yadav, Senior Research Fellow, and Director of Healthcare Research, William Davidson Institute, University of Michigan. (At the time of the workshop, Dr. Yadav was Professor of Supply Chain Management, MIT-Zaragoza International Logistics Program.)
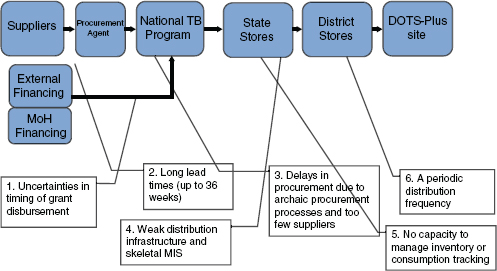
FIGURE 8-1 A schematic of the typical drug supply chain structure, which may not hold for all countries. Challenges in the supply chain for MDR TB drugs extend from grant disbursement to distribution.
NOTE: DOTS = Directly Observed Treatment-Short course; MIS = management information systems; MoH = Ministry of Health.
SOURCE: Yadav, 2011.
planning is problematic because the funds must be available before the first steps in the procurement process can occur. This uncertainty is one of the leading causes of national-level stockouts.
Innovative bridge financing arrangements can help alleviate this problem. With such financing, procurement and planning processes need not change because of financial uncertainty. For example, the United Nations Foundation recently initiated a program called the Pledge Guarantee for Health. If a country anticipates a short-term gap in financing that will lead to a stockout in the national program, it can secure bridge financing until the funds become available.
Long Lead Times and Delays in Procurement
Most second-line drug manufacturers employ make-to-order production—they do not start production until final purchase orders are received. Further, some manufacturers procure the active pharmaceutical ingredient (API) for a drug only after receiving a purchase order, and there are very few sources of these ingredients for many second-line drugs. As a result of
these and other factors, the procurement of second-line drugs can take up to 36 weeks after an order has been placed.
Manufacturers cite poor forecasts and small markets as reasons for requiring long lead times and employing make-to-order production. In a market that is uncertain and small—as is the case for many second-line drugs—the manufacturer is unlikely to keep stock on hand.
If national TB programs could provide advance information about their needs to global agencies, lead times would be reduced not just for those programs but for all programs. In this way, sharing advance order information would create a global public good, said Yadav. Countries or international organizations should not be in the business of holding inventory on behalf of manufacturers. The manufacturer could hold a larger inventory of finished product, and short-term imbalances in supply and demand could stabilize. Manufacturers could draw on stockpiles to meet the needs of country programs that were close to stockouts. Inventories should be held strategically to rectify short-term imbalances in high-risk areas.
In addition, delays in procurement can arise from archaic procurement processes and the lack of an adequate number of suppliers. For example, tender regulations often require purchasers to provide additional justification if a fully competitive market does not exist. In this case, having few suppliers not only increases prices but also makes the procurement process more challenging.
Weak Distribution Infrastructure
The distribution infrastructure for second-line drugs often is skeletal, and logistics management information systems (LMIS) beyond the district or state level often are weak. Yadav suggested that programs must learn to distinguish between forecasts and targets. Forecasts frequently are made by specifying coverage targets and then forecasting what quantity of drugs is needed. But going from a target to a forecast also requires considering such factors as consumption data and progress toward achieving a target. Forecasts typically are poor unless they are supplemented by field consumption data. Collecting these data requires synchronizing LMIS and surveillance data, which are generally separate. Especially for MDR TB, these data gathering mechanisms must be carefully coordinated, said Yadav.
Lack of Capacity for Inventory Management or Consumption Tracking
Yadav observed that it is unnecessary for the structure of the second-line drug supply chain to mimic the government’s administrative structure. Just because both a state and a district exist does not require that both the state and the district hold stock. Stockholding could be only at the national
level, with no state stores, or it could be at the state level, with no district stores. The key point for consideration is determination of the optimal stocking pattern.
Yadav cited two examples from the retail industry that could hold potential for improving the efficiency and effectiveness of the second-line drug supply chain. First, consumption data from the point of sale are embedded into the planning process at every level of the retail supply chain. This synchronization of data ensures that stock availability remains high. With MDR TB, the equivalent challenge is to collect consumption data at the facility level, preferably through use of user-friendly computerized systems. One system, for example, uses pictures of stock control cards at a facility to compile data, which allows for planning at each stage of the supply system and for high service levels. Second, the retail industry has a high frequency of deliveries. The counterpart for MDR TB would be more frequent deliveries than in the past, which also would counter some of the uncertainties of the market.
INDIA’S SECOND-LINE DRUG SUPPLY CHAIN2
Since the DOTS-Plus program began in India in 2007, guidelines for the diagnosis and treatment of MDR TB and for drug logistics have been established, noted Pradeep Saxena, Director, Central Bureau of Health Intelligence, Directorate General of Health Services, Government of India, and Head, WHO Collaborating Center for the Family of International Classifications, India. The program in India has, however, faced many challenges in providing patients with second-line drugs. Fewer patients than expected have been initiated on MDR TB treatment during the early phase of the program, which has posed the risk of having an excess of second-line drugs. Accurate forecasting of the need for drugs has been difficult, and delays in the provision of drugs have occurred. Ensuring the quality of drugs has been difficult as well, as has been preventing stockouts. Also, second-line drugs have a shorter shelf life than first-line drugs—just 2–3 years as opposed to 5 years.
The RNTCP has responded to these challenges in several ways. It has streamlined identification and referral of suspected cases of MDR TB and introduced rapid diagnostic methods, such as the LPA, which reduce delays in diagnosis and patient attrition. The program also has improved planning
____________________
2 This section is based on the presentation of Pradeep Saxena, Director, Central Bureau of Health Intelligence, Directorate General of Health Services, Government of India, and Head, WHO Collaborating Center for the Family of International Classifications, India. (At the time of the workshop, Dr. Saxena was Chief Medical Officer, Central TB Division, Government of India.)
for future treatment through data-driven state-level micro-planning that incorporates all aspects of the DOTS-Plus program, including geographic and time-based expansion of services.
In 2010, the RNTCP planned to detect 8,000 MDR TB cases, and drugs for 8,000 patients were procured (Figure 8-2). For 2011, the plan called for detecting 15,000 cases, with a steady increase in future years. The need for second-line drugs can be expected to increase accordingly. Funding for second-line drugs in India has come from the Global Fund Rolling Continuation Channel (RCC) and Round 9, UNITAID, and the World Bank, with the World Bank providing credit for the purchase of drugs.
The Central TB Division is forecasting the need for second-line drugs with technical assistance from partners of the RNTCP, including WHO and the Clinton Foundation. Requirements are being developed for all three weight bands based on past experience, assumptions of weight gain, and an expected prolongation of the intensive phase in many patients. Regular validation of the data is performed, and available stock balances are taken into account in the forecasting. With these techniques, forecasting for future drug requirements has become much more accurate.
To ensure patients’ adherence to 2 years of treatment, a large number of community DOTS providers have been trained. Detailed guidelines for the logistics management of second-line anti-TB drugs have been established.
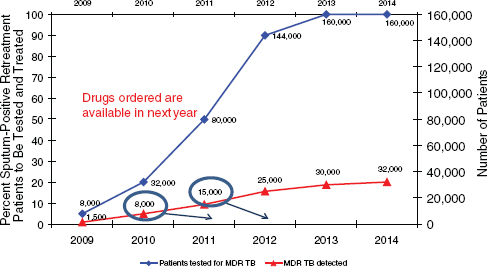
FIGURE 8-2 The Revised National TB Control Program (RNTCP) goals for MDR TB diagnosis call for increasing the number of sputum-positive retreatment patients to be tested and treated in future years. SOURCE: Saxena, 2011.
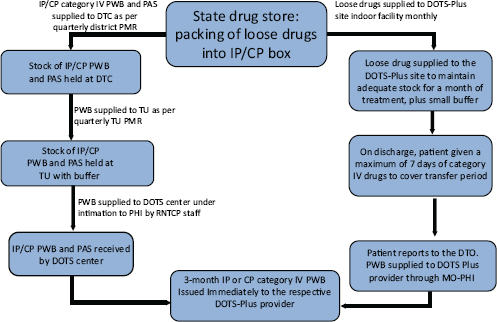
FIGURE 8-3 Second-line drugs move from state drug stores to DOTS-Plus providers through a series of steps.
NOTE: CP = continuation phase; DOTS = Directly Observed Treatment-Short course; DTC = District TB Center; DTO = District TB Officer; IP = intensive phase; MO-PHI = Medical Officer-Peripheral Health Institute; PAS = P-aminosalicylic acid; PMR = Program Management Report; PWB = Patient-Wise Box; RNTCP = Revised National TB Control Program; TU = TB unit.
SOURCE: Saxena, 2011.
And treatment adherence plans have been implemented for NGOs and private practitioners.
Loose drugs are now packed at the state warehouse into 3-month boxes for both the intensive and continuation phases under the guidance of medical officers. Each box is divided into three 1-month segments. A label on the box gives the itemized names of the drugs, the quantities in the box, the batch numbers of the drugs, the expiration dates of the drugs, and an identification number to help track every box. Drugs are then supplied to the DOTS-Plus site and to patients. Figure 8-3 depicts the movement of second-line drugs from the state drug store to patients.
To counter delays in the provision of second-line drugs, the RNTCP procures drugs through two independent sources. For states funded by the Global Fund, the drugs are procured through the GDF by the IDA Foundation after approval from the GLC. For World Bank-funded states, drugs are procured by the procurement agency of the Ministry of Health. Interstate
transfer of drugs also takes place to ensure optimum stock levels in all implementing states at any given time.
Steps are taken to ensure the quality of drugs both at the time of and after procurement. For states funded by the Global Fund, drugs are procured only from suppliers prequalified by WHO, so that high quality standards are maintained. For states funded by the World Bank, drugs are procured through international competitive bidding, with procurement restricted to suppliers compliant with WHO-Good Manufacturing Practice (GMP) guidelines. A joint inspection team under the Drug Controller General of India verifies the WHO-GMP certificates and performs predispatch inspection of all batches. After procurement, an independent International Organisation for Standardization (ISO) 17025-certified laboratory conducts quality testing of drugs. In addition, storage guidelines ensure maintenance of proper temperature and humidity.
To prevent stockouts, monthly reports from implementing states and quarterly reports from states and districts allow for regular monitoring of drug stocks. An information system for second-line drugs also is in place, with reports moving from the subdistrict to the district to the state level and on to the Central Division.
Finally, to address the shorter shelf life of second-line drugs, drugs come in three tranches instead of the usual two to ensure their full use. The Central Division also regularly monitors second-line drug stocks in state drug stores.
Taken together, these steps have had many positive benefits, said Saxena. They have improved planning for the future numbers of MDR TB patients to be diagnosed and placed on treatment. Having more than one source of second-line drugs ensures uninterrupted supplies. Drug logistics guidelines are dynamic and are revised according to new experiences from the field. Stock status is reported regularly to higher levels by subdistrict, district, and state drug stores. As a result of these steps, the RNTCP has been able to prevent stockouts in the concerned states.
Taking the steps described above has provided valuable lessons for program managers and others, said Saxena. The lessons learned from India’s experience in responding to the challenges of supplying second-line drugs include the following:
- Plan for the number of MDR TB patients to be diagnosed and treated in the future.
- Have more than one source of second-line drug supplies if possible.
- Implement drug logistics guidelines.
- Monitor drug stocks regularly.
- Provide training for field staff.
- Conduct supervisory visits and frequent consultations with state and district officials.
IMPROVING THE AVAILABILITY AND REDUCING
THE COST OF MDR TB DRUGS3
The availability of second-line drugs in India is problematic, said Inder Singh, Executive Vice President of Access Programs, Clinton Health Access Initiative. Five of nine second-line drugs could not be awarded in India’s 2009-2010 World Bank tender. The fact that only about 3,600 of 5,400 diagnosed MDR TB patients actually started treatment was due partly to the lack of second-line drugs. And the drugs are expensive, costing $4,400 or more per patient as compared with $19 for first-line drugs.
Coordinated actions can improve the availability and dramatically reduce the price of WHO-prequalified drugs for MDR TB in the near to mid-term, according to I. Singh. Today, the demand for certain drugs is below a key threshold, leading to a natural monopoly, he said. Exceeding this threshold would enable the sustainable entry of additional suppliers and the application of market dynamics.
To improve availability and reduce prices, demand must grow, said I. Singh. One way to create new demand is to invest in case finding for MDR TB, and considerable work is being done in this area. A second way to increase demand is to harmonize quality standards across key buyers. I. Singh noted that virtually no work is being done in this second area.
I. Singh described several case studies drawn from HIV/AIDS initiatives that could be applied to MDR TB. Price remains a barrier to access to antiretroviral therapy for HIV/AIDS. Yet, the price of one important drug for HIV/AIDS, tenofovir, dropped by more than 58 percent between 2006 and 2010. The price reduction was achieved through a variety of strategies, including finding new suppliers, using less expensive inputs, and developing new processes.
In a second case study, advanced procurement practices expanded the supply base and reduced supply risks, thereby reducing prices. An important HIV/AIDS drug had been selling at $500 for 10 years, and the developer of that drug had said no other company in the world could produce it according to quality standards. After 30–50 percent of the global volume of demand for this drug was allocated to any second supplier that could meet quality standards, regardless of price, a generic supplier entered at a price higher than that of the developer. Over the course of several years, however, the second supplier reduced the price of the drug dramatically, and the developer has now, in turn, reduced the price to be competitive. As a result of this approach, three generic manufacturers now meet WHO prequalification standards for the drug.
Finally, I. Singh cited a case in which the Clinton Health Access Initiative
____________________
3 This section is based on the presentation of Inder Singh, Executive Vice President of Access Programs, Clinton Health Access Initiative.
worked with suppliers to avoid supply disruptions. In the first half of 2010, an impending global supply shortage of tenofovir loomed. By providing suppliers with data on the impending shortage and emphasizing both the business and the humanitarian opportunities, the foundation persuaded three suppliers to expand their production capacity significantly.
In the area of MDR TB, growth in demand is essential before these approaches can be applied to bring new suppliers into the marketplace. Demand must grow by factors of 2 to 10 for several second-line drugs to support the entry of additional suppliers, create competition, and reduce prices, said I. Singh. Today, different quality standards effectively create distinct markets for the same drug. Similarly, different treatment regimens fragment the market. Harmonizing both quality standards and treatment regimens could boost demand, said I. Singh, creating opportunities for dramatic price reductions for two or even three of the second-line drugs.
MOVING TOWARD A FUNCTIONAL MARKET
FOR SECOND-LINE TB DRUGS4
Owen Robinson, Partnerships Manager, Mirebalais National Teaching Hospital, Partners In Health, discussed the relationship between management of MDR TB drugs and a strong, quality-assured drug marketplace. A strong drug management strategy can manage risk, lower price, and increase availability, but such a strategy must be global in scope, he said.
A healthy marketplace for MDR TB drugs has several cycles. As case finding takes place more quickly, patients are enrolled more quickly, and drug orders increase. Also, as higher volumes of drugs are needed, iterative negotiations can lower prices. These two processes can reinforce each other, with a larger market potential resulting from higher enrollment and higher enrollment capacity resulting from price reductions.
However, the marketplace for second-line TB drugs is largely stalled at present. Case finding is limited by low drug stocks, and drug orders are limited by low case finding. Suppliers opt not to scale up because of historically low order volumes, and orders remain low because of high prices. The supply side is averse to the risk of making drugs that will not be purchased, while the demand side is averse to the risk of purchasing drugs that will not be used.
The application of three principles can help manage risk in the MDR TB drug marketplace, said Robinson. First, risks need to be quantified, which allows everyone to understand what the risks are. Second, risks
____________________
4 This section is based on the presentation of Owen Robinson, Partnerships Manager, Mirebalais National Teaching Hospital, Partners In Health.
need to be aggregated, at the global level if possible. Third, risks need to be shared among the different entities involved in the transaction.
In the past, risk has been quantified by summing target-based forecasts, such as Global Fund targets for quality-assured procurement. However, these forecasts often fail to materialize because of barriers within countries and at the global level. As a result, suppliers become unwilling to risk their money on the basis of these forecasts. To change this dynamic, countries must consider such risk factors as funding availability, case finding rates, drug sensitivity trends, and programmatic hurdles to arrive at risk-adjusted analyses. Internally, these analyses can allow for optimal ordering processes. Externally, they enable market transparency by supplying providers with information about likely demand.
Once risk has been quantified and placed in the context of risk factors, it must be aggregated internationally, not just country by country. This aggregation can smooth out fluctuations in overall demand, much as investing in mutual funds can smooth out returns by masking the ups and downs of individual stocks. Key requirements for aggregating risk include a centralized aggregating entity; a credible global forecasting mechanism marked by two-way information flows; and a virtual rotating stockpile, in which one country can shift its supplies to another if it does not meet its projections for drug consumption. This approach relies on coordination among countries rather than centralized control of drug procurement. It is complementary to the market leverage associated with aggregating orders across countries and provides greater ability to negotiate lower prices.
Finally, when risk has been quantified through more effective forecasting and aggregated to reduce country-by-country exposure to risk for suppliers, it must be shared between buyer and seller. One way to accomplish this is through payment structures that use such mechanisms as initial up-front deposits, regardless of whether orders materialize; sales guarantees; or the option to purchase later at a certain price. Another approach is to negotiate around lead times using such levers as predictable cyclical orders instead of piecemeal ordering and capacity reservation. Contract terms can provide incentives for new entrants or rely on specified time horizons. Other incentives include reducing barriers to entry and helping with registration issues in different countries.
Robinson posed several important questions to be considered in efforts to improve the second-line drug supply chain:
- Who is involved in these processes, both within each country and globally?
- What is the right role for procurement mechanisms within individual countries?
- What is the right role for the procurement agencies that are involved today?
- What is the right balance between allowing countries to place their own orders and being coordinated enough to mitigate risk factors?
- What informational, human, and organizational resources are required?
During the discussion period, Salmaan Keshavjee, Harvard Medical School, talked about the factors that lead to incorrect forecasts of demand. The estimate of 440,000 people with MDR TB is one factor. Countries know they have large numbers of MDR TB patients but are unable to implement programs to reach them. Some people have seen the GLC as a bottleneck. As of the end of 2009, however, the GLC had approved treatment for 80,000 patients, yet countries had implemented only 22,000 treatments. The capacity of countries to implement MDR TB scale-up, a complex health intervention, has been lacking, said Keshavjee.
Part of the solution is to help create demand, Keshavjee continued. Countries with weak health systems need assistance in implementing programs to treat patients. In the past, a lack of rapid and inexpensive diagnostics made it difficult to increase demand, but successful efforts to build laboratory capacity have helped alleviate this problem. Now better systems are needed to procure drugs for and treat diagnosed patients.
Robinson noted that in some countries, the necessary level of transparency has been difficult to achieve for cultural reasons. These countries may have had difficulty with inviting international partners to work in the country, or they may have been reluctant to change past practices. Robinson suggested that there is now an increasing sense of transparency around the issues, as well as greater willingness to work with partners to strengthen forecasting and think strategically.
Puneet Dewan, WHO, remarked that five countries—Brazil, China, India, Russia, and South Africa—account for a large percentage of the world’s MDR TB patients. Yet several of these countries operate at least partially outside the system for procuring Global Fund-supported prequalified products. Thus these countries’ demand is fragmented into separate procurement mechanisms driven by in-country sources. Unless these countries consolidate their demand, volumes will remain so low that the ultimate goals of stable supply, stable manufacturers, and price reductions are unlikely to be achieved.
Robinson pointed to an analysis of first-line drugs conducted by the Clinton Health Access Initiative that found that countries procured prequalified drugs only when they were forced to work through the GDF mechanism
as a condition of the funding they were receiving. When they were using their own funding or were large enough to make their own demands, they opted for alternative ways of procuring first-line drugs. The result was what Robinson characterized as a missed opportunity for both quality control and aggregation of demand. The situation is similar for second-line drugs, he said. If countries are forced to channel their orders through a centralized mechanism as a condition of funding, they will do so; otherwise, they will do what is most convenient for them. Robinson suggested that the latter countries must have incentives for aggregation of demand. To that end, it is important to meet their needs in a way they find acceptable, as well as to explain the benefits of aggregation.
Saxena noted that national governments need to consider not only the quality and pricing of drugs but also availability and delays. A 4-month delay in accurate diagnosis can result in the loss of 40 percent of identified patients, which contributed to India’s being able to initiate only 3,600 of 5,400 identified patients on treatment. If drugs are not available for 12 or 24 months after diagnosis, the losses will be even greater. Consolidating demand around a single quality standard would integrate the market, said Saxena, which should improve lead times dramatically.
Nigorsulton Muzafarova, GDF, WHO Regional Office for Southeast Asia, cited the general scarcity of APIs for second-line drugs discussed by Yadav in his presentation—a major issue for producers of second-line drugs. Muzafarova noted that a drug manufacturer’s decision to seek WHO prequalification is complicated by underlying concern about the possibility that one of these critical ingredients is lacking.
Yadav noted that the industry relies on heavy investments, and the individual treatment price does not matter as much as aggregated volumes. Manufacturing, from the point of acquiring raw materials to the point of delivery, tends to be complex and to require a long time. Given the risk involved, moreover, few drug manufacturers are willing to initiate the process until they have an order in hand, and they tend to wait until they have an adequate number of orders collected to begin making one batch. Reducing risk and providing manufacturers with accurate forecasts would probably reduce the time to delivery significantly, said Yadav. I. Singh added that manufacturers are more likely to produce drugs when they can do so on a continuous basis, not for a product that is made in one or two batches per year.
POTENTIAL INNOVATIONS AND ACTION ITEMS
Through the presentations provided in this session and the subsequent discussions, individual workshop speakers and participants noted key innovations and action items. They include the following:
- Given delays in payment, the production and shipment of second-line drugs are often delayed. Bridge financing can offer a solution in the short term. Initial up-front deposits, guaranteed order volumes, options pricing, and the ability to purchase later are all innovative ideas for addressing current challenges in supply chain financing.
- Supply chains can be improved by forecasting future utilization through data-driven state-level micro-planning; incorporating DOTS-Plus geographic, time-based expansion of services; and increasing laboratory capacity.
- Maintaining transparency of information and data with suppliers can drive down costs.
- The harmonization of quality standards to create a homogeneous marketplace has the potential to create demand at a sufficient level of quality.
- Aggregating risk by global ordering, rather than country-by-country ordering, would improve the supply chain as part of a global architecture that would innovate drug procurement procedures.
- A virtual rotating stockpile may help prevent stockouts.
- Effective forecasting would help quantify risk.














