Nuclear medicine is a specialty that involves the use of radiopharmaceuticals (a radionuclide either by itself or attached to a molecule) in conjunction with highly specialized imaging instrumentation to detect the radionuclide emissions in the body after oral, inhalation, or intravenous administration. Radiopharmaceuticals may be used to assess normal physiologic processes, diagnose and treat diseases, measure the distribution of drugs, and monitor treatment effectiveness.
Fostered by unique partnerships between national laboratories, academia, and industry, the field of nuclear medicine has evolved over the past 55 years through advances in imaging instrumentation, radionuclide production, and radiopharmaceutical development. Nuclear reactors and particle accelerators have been developed to produce a wide array of radionuclides for diagnostic and therapeutic applications; innovative chemistry and automated synthesis devices have been designed to produce a multitude of new radiopharmaceuticals for imaging and treatment; and high-resolution and high-sensitivity instrumentation has been advanced for detection of radiopharmaceutical distributions in living systems, from small animal models to humans.
Radiochemistry is used in nuclear medicine to combine elemental radionuclides with biologically active chemical compounds to form radiopharmaceuticals. These agents are designed to trace specific metabolic or biologic pathways and localize to specific organs or sites of disease. Instruments with external detectors—such as gamma cameras, single photon emission computed tomography (SPECT), or positron emission tomography (PET) scanners—then produce an image of the distribution of radioactivity in the living system. Radiopharmaceuticals have been developed to study a wide range of normal processes and disease states, including normal brain function, aging, neurodegenerative diseases, cardiovascular disease, and cancer.
The field of nuclear medicine is highly diverse and multidisciplinary, but nuclear and radiochemistry are the core disciplines because radiopharmaceuticals are integral to every nuclear medicine study. The workforce for the field of nuclear medicine consists of personnel at all levels of education (B.S., M.S., Ph.D., Phar.M.D., and M.D.) in academia, industry, and government laboratories. In academia, nuclear and radiochemistry expertise involving nuclear medicine is mainly found in radiology departments, not in chemistry departments.
Those performing nuclear and radiochemistry in the field of nuclear medicine are trained in a wide variety of disciplines and may receive on-the-job training. The field of nuclear medicine is growing rapidly, and properly trained workers will be essential for continued success in this important area of modern health care.
A BRIEF HISTORY OF RADIOPHARMACEUTICAL DEVELOPMENT1
The use of radioactivity in medicine started with Wilhelm Röntgen, who discovered x-rays in 1895. A week after his discovery, Röntgen took an x-ray of his wife’s hand, clearly revealing her wedding ring and bones. In 1901 he was awarded the Nobel Prize in Physics for his innovation.
In 1934, building on the work of the Pierre and Marie Curie, their daughter Irène and her husband, Frédéric Joliot, created radioactive elements by irradiating stable isotopes with alpha particles. At the time there was significant interest in the use of radioactive materials in medicine and this discovery allowed for the quick, economic creation of radioactive materials in larger quantities. Based on these discoveries, Irène and Frédéric Joliot-Curie won the Nobel Prize in Chemistry in 1935. The important research of the Joliot-Curies is in many ways the foundation of modern nuclear medicine and radiopharmaceutical research, as the production of radionuclides by bombarding stable isotopes with various types of particles is the key method of production of many of the most widely used radionuclides for nuclear medicine imaging and therapy.
George de Hevesy followed up on the work by the Joliot-Curies with his Nobel Prize–winning research on the use of radionuclides as tracers in the study of chemical processes, which paved the way for the development of radiopharmaceuticals that trace biochemical and physiological processes in vivo but do not produce any pharmacological effects.
The invention of the cyclotron in the early 1930s by Ernest Lawrence paved the way for the discovery of many biologically relevant artificially
_______________
1 For more information, see www.accessexcellence.org/AE/AEC/CC/historical_background.php [accessed July 5, 2012].
produced isotopes (e.g., iron-59, iodine-131, and technetium-99m) that have become invaluable nuclides for nuclear molecular imaging and therapy. John H. Lawrence, a physician, used his brother Ernest’s radioisotopes in humans, treating a leukemia patient in 1937. John was also one of the early presidents of the Society of Nuclear Medicine (1966-1967). A colleague of the Lawrence brothers, Joseph G. Hamilton, coined the term “nuclear medicine” after observing John’s treatments of people with radionuclides. In the late 1930s, Hamilton asked Nobel Laureate Glenn Seaborg if he could create a radioactive isotope of iodine with a half-life of about a week for studying thyroid metabolism, and Seaborg promptly produced radioiodine (iodine-133 or 131I), which is still used for imaging and therapy of thyroid diseases.
After World War II there was enormous growth in the field of nuclear medicine. In 1946, a New York internist, Dr. Samuel M. Seidlin, together with colleagues Leo Marinelli and Eleanor Oshry at Montefiore Medical Center in New York City, treated and cured a patient with thyroid cancer using 131I obtained from Oak Ridge National Laboratory. This work was published in the Journal of the American Medical Association (Seidlin et al. 1946) and produced a flurry of publicity. After this, there were almost yearly discoveries in the new field of nuclear medicine, in both chemistry and physics. The development of instruments to detect the various decays of radionuclides went hand in hand with new discoveries in radiopharmaceuticals.
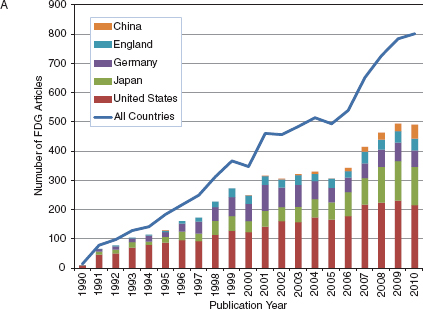
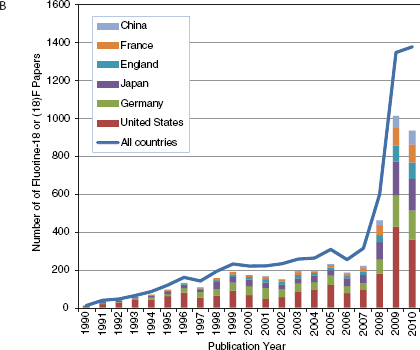
No one could have predicted how valuable the cyclotron would become to modern molecular imaging for the production of a variety of radionuclides, especially the short-lived positron-emitting isotopes of carbon, nitrogen, oxygen, and fluorine. The availability of both small academic- and hospital-based cyclotrons spurred growth of the field and now regional cyclotron facilities have increased the availability of PET tracers, mostly through the production and distribution of 2-deoxy-2-[18F]fluoro-D-glucose ([18F]fluorodeoxyglucose, FDG), the most widely produced and indispensable molecular imaging agent. Figure 4-1A shows the international growth in publications about FDG since 1990. Figures 4-1B and 4-1C show the growth in publications for newer areas of nuclear medicine involving Fluorine-18 and Gallium-68. While the growth for both of these new areas is dominated by German- and U.S.-authored papers, more recently many other countries mostly in Europe and Asia are now contributing to the steep growth in numbers of publications in these areas.
There are three major sources for the production of radionuclides for nuclear medicine applications—particle accelerators (linear and cyclotrons),
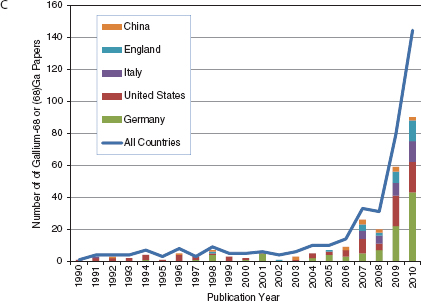
FIGURE 4-1 Keyword search of journal articles by country for nuclear medicine related keyword, 1978-2010. (A) fluorodeoxyglucose (FDG), (B) Fluorine-18 or (18)F, (C) Gallium-68 or (68)Gallium.
SOURCE: Web of Science keyword search, 2011.
nuclear reactors, and radioisotope generators. Most radioisotopes used for radiodiagnostics and radiotherapeutics are produced by cyclotrons. Figure 4-2 shows the distribution of cyclotron facilities across the United States. As of October 2011, there are over 150 cyclotrons in the United States that are operated by commercial entities, universities, or hospitals producing radiopharmaceuticals for PET centers (B. Clarke, SNM, personal communication, 2011). In addition to cyclotron-produced radioisotopes, nuclear reactors produce medical radioisotopes by either separation of isotopes from the fission materials (for example, 125/131I and 99Mo) or through neutron activation of stable isotopes (for example, 64Cu from 64Zn, or natural Zn targets). A listing of the common PET, SPECT, and radiotherapeutic isotopes used in nuclear medicine is provided in Table 4-1.
There are many aspects of the field of radiopharmaceutical chemistry, including radionuclide production, organic chemistry, inorganic chemistry,
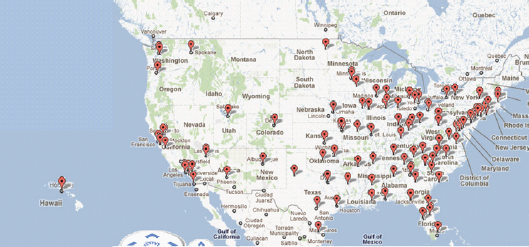
FIGURE 4-2 Map of U.S. commercial PET radiopharmacies using small, self-shielded cyclotrons that supply PET imaging probes for molecular imaging diagnostics used in patient care and research (does not include cyclotrons at medical schools).
SOURCE: B. Clarke, Society of Nuclear Medicine, personal communication, 2011.
biological chemistry, radiochemistry, automation (engineering), and regulatory science. The evaluation of novel radiopharmaceuticals in biological assays and animal models is vital to the successful translation of new agents into human studies. Prior to human studies, one must have knowledge of the production of radiopharmaceuticals for human use, which includes understanding federal regulations regarding production of the cold substrate, radionuclide, and radiopharmaceutical under good manufacturing practice guidelines. One must also have knowledge and proficiency in the safe handling of radioactivity—that is, radiation safety. Box 4-1 describes the various steps in the preparation of a radiopharmaceutical.
Radiopharmaceutical Research and Development
The development of radionuclide production requires extensive knowledge of nuclear reactions by bombardment of particles onto targets on a biomedical cyclotron or bombardment of neutrons on targets in a nuclear reactor. It is essential to apply nuclear and radiochemistry principles to maximize yields and to set the energies of the bombarding particles to optimize yields of the desired radionuclide and minimize production of longer-lived
TABLE 4-1 Widely Used Positron Emission Tomography, Single Photon Emission Computed Tomography, and Therapeutic Radionuclides for Imaging and Radiopharmaceutical Preparation
| Isotope | Production Method Parent/Stable Isotope |
Half-life |
| Positron Emission Tomography Radionuclides | ||
| Fluorine-18 | cyclotron (oxygen-18) | 110 min |
| Carbon-11 | cyclotron (nitrogen-14) | 20 min |
| Nitrogen-13 | cyclotron (oxygen-16) | 10 min |
| Oxygen-15 | cyclotron (nitrogen-14/15) | 122 s |
| Copper-64 | reactor (zinc-64) | 12.7 h |
| cyclotron (nickel-64) | ||
| Gallium-68 | generator (germanium-68) | 68 min |
| Rubidium-82 | generator (strontium-82) | 75 s |
| Zirconium-89 | cyclotron (yttrium-89) | 3.3 d |
| Iodine-124 | cyclotron (tellurium-124) | 4.2 d |
| Single Photon Emission Computed Tomography Radionuclides | ||
| Gallium-67 | cyclotron (zinc-68) | 78 h |
| Technetium-99m | generator (molybdenum-99) | 6 h |
| Indium-111 | cyclotron (cadmium-111) | 2.8 d |
| Iodine-123 | cyclotron (xenon-124) | 13.2 h |
| Iodine-131 | reactor (tellurium-130) | 8 d |
| Thallium-201 | cyclotron (thallium-203) | 3.1 d |
| Therapeutic Radionuclides | ||
| Yttrium-90 | reactor (strontium-90) | 2.7 d |
| Iodine-131 | reactor (tellurium-130) | 8 d |
| Lutetium-177 | reactor (ytterbium-176) | 6.7 d |
| Rhenium-186 | reactor (rhenium-185) | 3.7 d |
| Strontium-89 | reactor (strontium-88) | 50.5 d |
| Samarium-153 | reactor (samarium-152) | 46.3 h |
SOURCES: Cyclotron (2010); IAEA (2003, 2009); Unterweger et al. (2010).
byproduct radionuclides. A strong knowledge of targetry and separation chemistry is essential in order to produce high purity products.
A solid working knowledge of organic chemistry combined with radiochemistry is required for the design of radiopharmaceuticals labeled with radiohalogens (for example, 18F and radioiodines) and 11C. In the past 40 or more years, there have been a large number of 18F- and 11C-labeled small molecules designed for imaging of cancer, cardiovascular disease, and neurological diseases. While there are a substantial and growing number of PET radiopharmaceuticals that are FDA approved under an Investigational New Drug (IND), only FDG has been approved by FDA for use in patient care under a New Drug Application (NDA). Examples of PET radiopharmaceuticals under FDA INDs include probes for imaging Alzheimer’s plaques (Nelissen et al. 2009; Rowe et al. 2008; Wong et al. 2010), cellular prolif-
BOX 4-1 PREPARATION OF A RADIOPHARMACEUTICAL
There are a number of components that comprise the preparation of radiopharmaceuticals for clinical and research applications. Using fluorine-18 (18F)-labeled fluorodeoxyglucose (FDG) as an example, the production process is highlighted below along with the radiochemistry emphasis areas that are associated with each part of the process.
Radionuclide Production
• Areas of expertise involved in this stage: cyclotron engineer and targetry chemist

Particles are accelerated by an accelerator (cyclotron, linear accelerator) or nuclear reactor react with the stable isotope nucleus to give an excited compound nucleus that emits a particle yielding the radioactive isotope.
Source: Image credits: The Crump Institute for Biological Imaging, Department of Pharmacology, University of California at Los Angeles. Brain & Mind. 2008. The cyclotron and PET [online]. Available: http://www.cerebromente.org.br/n01/pet/petcyclo.htm [accessed March 7, 2012].
Radiopharmaceutical Chemistry
• Areas of expertise involved in this stage: radiochemist and nuclear pharmacist.
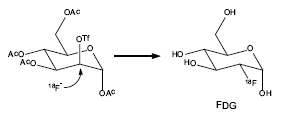
SOURCE: Henry VanBrocklin.
Fluorine-18 fluoride ion from the cyclotron is reacted with the 2-deoxymannosetriflate precursor. The 18F-fluorodeoxyglucose tetraacetate intermediate is deprotected to give FDG. Automated chemistry synthesis units have been developed to provide reliable and reproducible batches of the radiopharmaceuticals and enhance chemist radiation protection.

SOURCE: Henry VanBrocklin.
Quality Control
• Areas of expertise involved in this stage: radiochemist and nuclear pharmacist.

SOURCE: Henry VanBrocklin.
The quality of every dose preparation must be assessed prior to injection into the patient. A series of tests are conducted to assure that the product is sterile and free of contaminants that might be harmful to the patient.
Patient Injection and PET Imaging
• Areas of expertise involved in this stage: nuclear medicine technologist, nuclear medicine physician, and medical physicist.
SOURCE: A.D.A.M., Inc.
Ten to twenty millicuries (mCi; 370-740 MBq) of FDG are injected intravenously 30 minutes prior to PET imaging
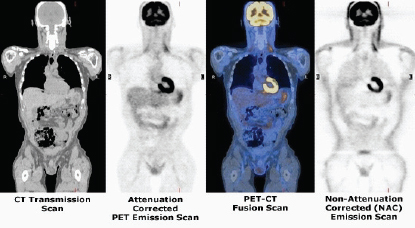
SOURCE: Henry VanBrocklin
PET and CT scans following the injection of FDG. Prominent FDG uptake is seen in the heart and brain because these are areas of high glucose metabolism. The fused PET/CT image combines the functional and anatomical scans in one image.
eration (Bading and Shields 2008) and hypoxia in cancer (Chitneni et al. 2011), and blood flow for cardiovascular disease (Maddahi et al. 2011) (see the Clinical Applications section later in this chapter).
Expertise in inorganic chemistry and radiochemistry has produced a burgeoning development of radiopharmaceuticals labeled with metal radionuclides, such as 68Ga, 64Cu, and 89Zr. A critical component of developing these radiometal-labeled agents is the design of chelators that form stable complexes of the radiometals in vivo. This occurs as the release of radiometals causes a high uptake in radiation-sensitive tissues such as bone marrow, and results in poor target-tissue contrast due to high accumulation of the radiometals in blood and liver (Wadas et al. 2010). Many of the current clinical SPECT radiopharmaceuticals are 99mTc chelates. Altering the chelate backbone structure has led to a variety of imaging probes for bone, tumor, and heart and for brain blood flow.
Throughout the field of radiopharmaceutical chemistry is the theme of interweaving nuclear and radiochemistry with an understanding and practical knowledge of biochemistry, cell and molecular biology, and medicine. When modifying a known biological targeting molecule with either a radiohalogen (for example, 18F or 123I) or a radiometal (for example, 68Ga, 64Cu, 89Zr, or 99mTc) chelate, one must first determine that the biological activity of the new compound is not significantly altered from that of the parent molecule. The biodistribution in normal rodents is performed to demonstrate that the agents do not accumulate in non-target tissues. This is followed by evaluation of the radiopharmaceutical uptake in target or diseased tissues. Validation that the radiopharmaceutical is specifically accumulating in its target tissue often requires use of microscopic techniques such as immunohistochemistry. Many iterations of optimizing the overall chemistry and specific radiochemistry followed by a biological evaluation is often required prior to choosing an agent for initial testing in humans. Moving agents into the clinic often requires collaborations of nuclear and radiochemists with cancer biologists, neuroscientists, and clinicians of various specialties. However, while it is important that nuclear and radiochemists embrace the many disciplines within the field of radiopharmaceutical sciences, the primary expertise of nuclear and radiochemistry is critical for success of this field.
Clinical Applications
For nuclear medicine imaging, a radiopharmaceutical is administered to the patient (typically intravenously) and a scanner that detects radioactivity in the patient is used to show the uptake of the tracer. Nuclear medicine images provide quantitative data on the biochemistry of normal tissues and disease conditions in living subjects in contrast to the anatomical imaging
modalities (for example, computed tomography, ultrasound, and Magnetic Resonance Imaging), which primarily provide structural information. According to the Society of Nuclear Medicine’s (SNM’s) annual report for 2010, more than 20 million patients in the United States undergo nuclear medicine procedures for the diagnosis and treatment of a wide variety of diseases, including cancer, heart disease, and neurological diseases such as Alzheimer’s disease (Center for Molecular Imaging, 2011).
A list of commercially available radiopharmaceuticals is provided in Appendix G. The majority of the tracers listed were approved prior to 1995. This is a direct result of the economics—that is, the cost of development vs. the potential market for the approved radiopharmaceutical—of bringing new entities through the full drug development and regulatory approval processes. While little has changed with FDA approval since 1995, there has been increasing pressure to develop new PET radiopharmaceuticals to augment the four currently approved PET radiopharmaceuticals (rubidium-82, 13N-ammonia, sodium 18F-fluoride, and 18F-FDG). A small pipeline of PET and SPECT radiopharmaceuticals that are nearing approval has built up over the last 5 to 10 years. Several late-development stage radiopharmaceuticals and commercially available radiotherapeutics are also shown in Appendix G. The development and ultimate marketing of these new diagnostic and therapeutic radiopharmaceuticals is directly linked to the demand for trained radiopharmaceutical scientists at all skill levels.
Nuclear and radiochemists in the area of nuclear medicine are mainly employed in academia and industry (Box 4-2). Within these two sectors, there are many sub-sectors. Academia entails scientists working in the basic science departments—that is, chemistry, biology, engineering—and scientists working in medical schools and in departments such as radiology or medicine. Hospitals affiliated with universities may have PET centers that provide FDG and other radiopharmaceuticals for clinical diagnostic imaging and clinical research. The industry sector is subdivided among companies that provide equipment and instrumentation for the nuclear medicine field (for example, cyclotrons, scanners, hot cells, and generators), companies that develop PET imaging probes, manufacture pharmaceuticals, and companies that develop and distribute radiopharmaceuticals. Many large pharmaceuticals developers such as Merck and Genentech, among others, now possess radiochemistry and nuclear imaging capabilities. This sector represents a growth area for radiochemistry where the existing and growing demand far exceeds the supply of radiochemists.
BOX 4-2 CAREERS IN NUCLEAR MEDICINE
Nuclear medicine is a highly multi-disciplinary field that involves many types of scientists and clinicians. Physicists and engineers develop and optimize charged particle accelerators and nuclear reactors for the production of radionuclides, as well as develop imaging instrumentation for the diagnosis of disease. Chemists design, synthesize, and radiolabel agents for diagnostic imaging and targeted radiotherapy of disease while biologists are involved in evaluating these agents using bioassays, cells grown in culture, and animal models of disease. Many scientists have a focus on one of these areas, but perform or lead research that involves multiple disciplines. Clinicians are typically board certified in radiology, nuclear medicine, radiation oncology, or internal medicine, although many have dual certifications. Regardless of the field of study in science, engineering, or medicine, the most critical and rate limiting disciplines in of nuclear medicine is nuclear and radiochemistry. Below is a sampling of the careers that are available to radiochemists who choose to enter the field of nuclear medicine.
Nuclear Pharmacist. According to the Board of Pharmacy Specialties, a nuclear pharmacist (also called a radiopharmacist) is “a member of a nuclear medicine team who specializes in the procurement, compounding, quality control testing, dispensing, distribution, and monitoring of radiopharmaceuticals” (BPS 2012). Nuclear pharmacists are employed by hospitals, academic medical centers, or centralized radiopharmacies. These individuals typically have either a doctorate in pharmacy or are registered pharmacists. After obtaining their degree, they pursue continuing education in the form of certification in nuclear pharmacy (for example, through the certification program at Purdue University) or radiopharmacy (for example, through the certification program at the University of New Mexico).
Industrial careers. Positions in the nuclear medicine industry for radiochemists include nuclear medicine imaging, both pre-clinical and clinical, which is a key component of drug development. Pharmaceutical companies employ radiochemists to synthesize and radiolabel drugs of interest to determine their pharmacokinetics and target tissue uptake. These companies also perform clinical trials that incorporate PET and SPECT imaging to monitor whether their drug is effective at an earlier stage rather than following disease progression or remission. There are also companies that produce radiopharmaceuticals at centralized pharmacies, as well as companies that develop novel radiopharmaceuticals for PET and SPECT imaging.
Academic careers. There are many career opportunities in academia for radiochemists in the nuclear medicine field at the B.S., M.S., and Ph.D. levels. The
primary goals of Ph.D. radiochemists in academia are in the areas of research and education. Many of these positions are as tenure track faculty members and involve having a research group that includes undergraduate students, graduate students, postdoctoral trainees, and technicians. These faculty members may also teach courses in the area of imaging and nuclear medicine. Faculty members who are engaged in nuclear medicine research also either work at or direct cyclotron/PET facilities that produce radiopharmaceuticals for routine clinical studies and pre-clinical and clinical research. Aside from being a faculty member of a university, other career opportunities are as support staff that supervise or work in the PET/cyclotron facilities and also perform research in the labs of the faculty members. Nuclear pharmacists are also employed by academic universities and medical centers.
Government careers. The DOE national labs and the National Institutes of Health (NIH) employ nuclear and radiochemists involved in nuclear medicine research. NIH currently has at least six groups that are located in the National Institute of Biomedical Imaging and Bioengineering, the National Cancer Institute, the National Institute of Mental Health, the National Heart Lung and Blood Institute, and the NIH Clinical Center. These groups employ nuclear and radiochemists for developing radiopharmaceuticals for various diseases. These positions are typically either technical support or Ph.D.-level jobs that are research oriented. The focus of the national labs has shifted in the past several years away from biomedical research into the area of using radiotracers for energy-related research.
To determine the number of nuclear and radiochemists involved in nuclear medicine, the committee initially looked at a report on the nuclear medicine scientist workforce completed in 2006 by the SNM (Center for Health Workforce Studies, 2007). The report included survey data on physicians, nuclear pharmacists, physicists, and chemists. Out of the 898 survey respondents who indicated they were active in nuclear medicine science, 122 participants identified themselves as chemists. Those participants were asked to provide further information about their sub-specialization in chemistry, and, 36.9 percent selected radiochemistry as their major area of interest while an additional 21.3 percent indicated organic chemistry as their subspecialty.
To follow up on the 2006 survey and obtain more recent data, this committee received input (Table 4-2) from the radiochemist and nuclear pharmacist members of the SNM Radiopharmaceutical Sciences Council (RPSC) and
TABLE 4-2 Results of a Questionnaire Sent to Nuclear Medicine Radiochemists by the Society of Radiopharmaceutical Sciences/Radiopharmaceutical Sciences Council of the Society of Nuclear Medicine
| No. of Yes Responses |
% Yes Responses |
|
| U.S. Respondents (110) | ||
| Use radiotracers as part of job | 107 | 97% |
| Highest degree Ph.D. | 78 | 72% |
|
Primary discipline (nuclear, chemistry, radiochemistry, |
36 | 34% |
| Employed in | ||
| academia | 58 | 55% |
| industry | 22 | 21% |
| medical facilities | 9 | 9% |
| Hold faculty appointment | 73 | 69% |
| in a radiology dept | 39 | 58% |
| in a chemistry dept | 4 | 5% |
SOURCE: See Appendix D for questionnaire.
the members of the Society of Radiopharmaceutical Sciences (SRS).2 There were a total of 110 responses received from U.S. members (110/425 or 25.9 percent response rate). The average age of the respondents was 52 (range: 26-83; median: 52). It was found that 34 percent of the respondents received their degrees in nuclear chemistry, radiochemistry, or radiopharmacy, which corresponds well with the 37 percent found in the earlier 2006 SNM survey (Center for Health Workforce Studies 2007).
Positions at the Bachelor’s and Master’s Degree Level
There are many positions requiring entry level B.S. and M.S. radiochemists in industry. For example, input to the committee from a sampling of industries that employ radiochemists in the nuclear medicine field (Table 4-3) show that 44 percent of the chemists employed have B.S. degrees, 30 percent have Ph.D.s, 20 percent have MS degrees, and 4 percent have A.A.S. degrees. Over the next 5 years, a 38 percent increase in the number of positions at the B.S./M.S. level at these companies is expected. This indicates the need for increasing training opportunities at the undergraduate level. Many, if not most, of the employees entering the nuclear medicine industrial workforce do not have any training in nuclear and radiochemistry
_______________
2 As of July 28, 2011, there were 760 unique members (425 from the United States) of the RPSC and SRS (Jennifer Mills, Society of Nuclear Medicine, personal Communication, July 28, 2011). See Appendix D for questionnaire.
TABLE 4-3 Estimated Number of Employees Currently Employed in Industry and a 5-year Projection of the Number of Employees Needed in Industry, Stratified by Highest Degree of Employee
| Current Number of Employees |
5-year Projection of Required Employees |
|
| Associate’s degree | 7 | 27 |
| Bachelor’s degree | 84 | 110 |
| Master’s degree | 38 | 58 |
| Doctorate degree | 60 | 106 |
| Total employees | 189 | 301 |
SOURCE: Data kindly provided by the following companies: Abbott, ABT, Covidien, Eckert Ziegler, GE, Genentech, IBA, Immunomedics, Merck, NorthstarTM, Siemens, Sofie Biosciences, Pfizer, and UPPI.
and companies must rely on on-the-job training to educate their employees. A knowledgeable workforce will better serve this community.
Academic Sector Demand
Responses to the SRS/RPSC questionnaire also provided information on the ages of nuclear and radiochemists currently working in the academic sector of nuclear medicine. The average age of the academic survey respondents was 55 years, and 24 percent of the respondents were over the age of 60, suggesting that approximately 24 percent of the respondents will be of retirement age by 2016. This data suggests that a large component of the academic workforce will need to be replaced in the next 5-10 years. Much of the demand in academia is dependent on the federal funding climate. As of this writing, future federal funding from NIH and DOE for nuclear medicine based research is uncertain (see Figures 4-3 and 4-4 below for recent funding data). If federal funding dollars increase, the demand in academia will likely increase beyond that of replacing retired faculty and staff at universities.
Industrial Sector Demand
In order to determine the demand in the industrial sector, representatives from companies that are involved in the nuclear medicine industry that employ nuclear and radiochemists were contacted and asked for numbers of current employees and estimated demand over the coming 5 years. These companies were selected based upon their known activities in the area of nuclear and radiochemistry for the field of nuclear medicine. Table 4-5 summarizes the data obtained from 14 companies, with a response rate of
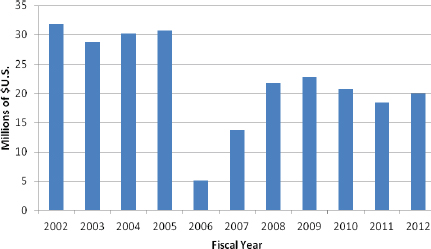
FIGURE 4-3 US Department of Energy funding for Radiochemistry and Imaging Instrumentation.
NOTE: Since 2010, the program name has been Radiochemistry and Imaging Instrumentation. Previously, other names were used: Radiopharmaceutical Design and Synthesis in FY 2005, Medical Applications in FY 2006 and FY 2007, Radiopharmaceuticals and Imaging in FY 2008, and Radiochemistry and Instrumentation in FY 2009.
SOURCE: DOE 2011.
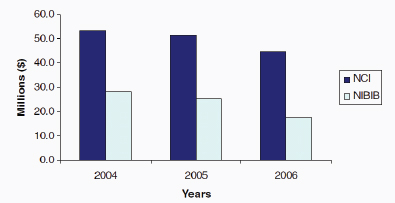
FIGURE 4-4 NIH Extramural funding for nuclear medicine research, 2004-2006. NCI, National Cancer Institute; NIBIB, National Institute of Biomedical Imaging and Bioengineering.
SOURCE: NRC/IOM 2007. Data originally provided by NCI and NIBIB.
88 percent. These data are not entirely representative, and there may be additional companies from which the committee neglected to obtain data. The data from the 14 companies suggest that there will likely be a large demand for trained nuclear and radiochemists in the next 5 years, with the projection being an approximately 60 percent increase in the nuclear medicine industrial workforce. In addition to nuclear and radiochemists being employed directly by the companies listed in the footnote of Table 4-3, many major companies outsource positions to academia, and an additional 40 full-time employees were reported from companies who responded to this question.
Government Sector Demand
U.S. national laboratories, NIH, and other agencies, including FDA and the National Institute of Standards and Technology, employ radiochemists and nuclear pharmacists involved in nuclear medicine research and regulatory activities. At the FDA, there are about a dozen employees who work in the Radioactive Drug Research Committee program in the Division of Medical Imaging Products.
There are at least six groups at NIH that employ radiochemists involved in nuclear medicine-related projects. A breakdown of the NIH radiochemists by degree is given in Table 4-4. The groups that employ radiochemists include the Laboratory of Molecular Imaging and Nanomedicine in the National Institute of Biomedical Imaging and Bioengineering, the Radioimmune Inorganic Chemistry Section of the Radiation Oncology branch at the National Cancer Institute, the NIH Clinical Center, the Imaging Probe Development Center at the National Heart Lung and Blood Institute, and two groups at the National Institute of Mental Health. Although there are projected budget cuts in the intramural program at NIH, most of these laboratories are projecting stable numbers of staff and trainees over the next 5 years.
TABLE 4-4 Estimated Number of Radiochemists Currently Employed at the National Institutes of Health, Stratified by Highest Degree
| Current Number of Employees | |
| Pharmacy degree | 4 |
| Bachelor’s degree | 5 |
| Master’s degree | 5 |
| Doctorate degree | 30 |
| Total employees | 44 |
SOURCE: Data were kindly provided by the following individuals: Martin Brechbiel (NCI); Gary Griffiths (NHLBI); Peter Herscovitch (NIH Clinical Center); Robert Innis (NIMH); Dale Kieswetter (NIBIB); and Victor Pike (NIMH).
The PET, SPECT, and Therapeutic Radiopharmaceutical Market in the United States
Recent market analyses indicate continued growth for nuclear medicine in both imaging and radiopharmaceutical development, which indicates the demand for nuclear and radiochemistry expertise will also continue. One report by Global Industry Analysts, Inc. (PR Web News Wire 2010), states that “nuclear medicine is one of the most promising and rapidly growing segments of the medical imaging industry.” It says the global market for nuclear medicine is predicted to reach the US$1.69 billion by 2015, and attributes the growth to improvements in the development of molecular imaging-based diagnostics and treatments, along with an increased demand from the aging U.S. population. Another market report by Bio-tech Systems, Inc. (BTSI), states that the U.S. sales of SPECT and PET radiopharmaceuticals reached $1.16 billion in 2009, $1.20 billion in 2010, and are expected to rise to $6 billion by 2018 (BTSI 2006, 2010, 2011; PR Web News Wire 2009). BTSI’s reports also detail how PET procedures grew 9 percent in 2009 to about 2 million, and grew to 2.1 million in 2010. In addition, sales of 18F-labeled 2-FDG were increased from $276 million in 2009 to $299 million in 2010, consistent with the increased numbers of procedures.
As new agents are developed and approved by FDA and new products are introduced to consumers, total PET radiopharmaceutical sales are predicted to rise to $4.3 billion by 2018 (BTSI 2011), of which the majority of the increase will be from sales of new products other than FDG. For example, an FDA review panel unanimously recommended approval of a new PET agent (florbetapir) for imaging amyloid plaque associated with Alzheimer’s disease, while two other similar agents (florbetabane and flumetamol) and an 18F-labeled cardiac blood flow agent (flurpiridaz) are in phase 3 clinical trials. SPECT sales were down 9 percent in 2010, primarily due to reductions in pricing of perfusion agents, increased use of generic Sestamibi,3 and the shortage of 99Mo for 99mTc radiopharmaceuticals (BTSI 2011). The 99Mo shortage was stabilized by the end of 2010 with two reactors back on line. The outlook for SPECT agents is improving, with a new agent, DaTscan (Ioflupane), recently approved for assisting in the evaluation of Parkinsonian syndromes.
In addition to the market for diagnostics, therapeutic radiopharmaceuticals include both current and emerging products for lymphoma, myeloma,
_______________
3 Sestamibi is a generic kit for the preparation of Technetium (Tc-99m).
and cancers of the breast, prostate, brain, liver, pancreas, and other types of cancer that are resistant to traditional therapies. The U.S. sales of therapeutic radiopharmaceuticals were $71 million in 2005, with rapid growth anticipated (BTSI 2006). There has been increased research activity in the area of therapeutic radiopharmaceuticals with a variety of molecular targeting strategies and therapeutic radionuclides for a wide range of tumor types. Although there are currently no approved alpha-emitting products, there is a strong push in research and development of agents labeled with 212Bi, 213Bi, 211At, 223Ra, and 210Po. One agent, Alpharadin (223Radium Chloride), recently closed successful phase III clinical trials and a 2012 filing for FDA approval is anticipated. This one agent alone may bring annual U.S. therapeutic radiopharmaceutical sales close to $1 billion by 2015 (PMLiVE 2012).
Research Funding
One of the underlying factors in creating the supply of expertise to meet the future demand is funding for basic research and education in nuclear medicine. Two of the key sources of funding over the years for basic and applied radiochemistry research with relevance to nuclear medicine have been the U.S. Department of Energy (DOE) Office of Science and Office of Biological and Environmental Research and NIH (Figures 4-3 and 4-4).
Drastic funding cuts, nearly eliminating the DOE Medical Applications program in FY2006, and the desire to focus research activities on DOE mission-driven programs, including biofuels production and climate change research and the DOE Office of Biological and Environmental Research, funding for the Radiochemistry and Imaging Instrumentation (RII; formerly called Medical Applications and Measurement Science Research subprogram) significantly altered the DOE nuclear medicine research landscape. The cuts included funding for “molecular nuclear medicine research, research and technology development activities in imaging gene expression, magnetoencephalography, biosensors, PET instrumentation for human clinical applications, MRI and neuroscience research, radiation dosimetry for therapeutic dose estimation, and targeted molecular radionuclide therapy” (DOE 2005, p. 258).
In 2008, Congress restored funding for the RII program, albeit at a reduced level from the FY 2005 allocation. The RII had funding opportunity announcements in FY2008 and FY2010 to support basic radiochemistry and imaging instrumentation research. In FY2009 the RII funded the DOE Integrated Radiochemistry Research Programs of Excellence, a training program that was recommended in the 2007 report Advancing Nuclear Medicine Through Innovation (NRC/IOM 2007).
These changes have been a significant programmatic shift for the RII program away from specific nuclear medicine applications to DOE mission centric applications of radiochemistry. While there are still opportunities for radiochemistry and imaging instrumentation, it is difficult for scientists in radiology departments and medical institutions to apply for these funds, given that they and their collaborators are not studying plant physiology and biofuel production. Overall, the reduction in congressional funding allocations to this program and the shift in research emphasis resulted in the loss of this valuable source of funding, which has had a negative impact on the nuclear medicine training pipeline for the future workforce and on valuable technology development resources that represented a significant return for the DOE investment.
NIH funding has supported many radiochemistry and imaging technology developments related to nuclear medicine (Figure 4-4).4 The focus, however, has been largely on the applications of the radiotracers and translation into the clinic rather than on the underlying radiochemistry technology and new radiochemistry reactions. Since more recent NIH funding data for nuclear medicine were not available to the committee, it is not clear if NIH has been able to make up the difference created by the reduction in DOE funding, especially for nuclear medicine projects that are more basic science in nature. Funding increases from both DOE and NIH as well as other sources will likely be necessary to support the projected need for trained scientists and sustained future growth.
Health Care Regulations
Utilization of the technology in medical procedures also drives the supply and demand of the nuclear medicine workforce. The cost associated with a complete scan, including the radiotracer, professional fees, and scanner costs, all factor into the utilization of the technology. There is ongoing research in the area of cost effectiveness to determine the extent of health care savings offered by early diagnosis and staging, as well as the ability to determine patients’ response to therapy prior to treatment or sooner after treatment. The magnitude of the importance of this is clear from the fact that across all diseases and all drugs, on average, only ~20% of patients treated have a measurable positive response, while ~80% take the risk with no benefit and enormous resources are lost to healthcare. Research demonstrating that the benefits of nuclear medicine outweigh the costs will have a
_______________
4 Note: The committee requested, but did not obtain more recent NIH nuclear medicine funding data.
positive impact on the demand for more nuclear and radiochemists in this field. Cost reimbursement by insurance and Centers for Medicare and Medicaid Services (CMS) will also dictate the utilization and growth of imaging technology, and thus employment of radiochemists. Flat or declining reimbursement will reduce growth. On the other hand, as the U.S. population ages, the number of imaging studies based on molecular diagnostics of the biology of disease is likely to increase. Since the leading molecular imaging technique is PET, the need for radiochemists will also increase.
The extent of the regulatory requirements, such as Medicare reimbursement and FDA oversight of radiopharmaceutical approval, on the industry may factor into the demand for radiochemists. Reimbursement for clinical nuclear medicine procedures dictated by CMS will determine the direction of growth or shrinkage in the field. Increased reimbursement equals growth in the number of procedures. Likewise, FDA regulations of radiopharmaceutical approval will determine the demand for radiochemists. Since 1995, while there are a substantial and growing number of radiopharmaceuticals that are FDA approved under an IND, there have only been seven radiopharmaceuticals or radiotherapeutics approved by the FDA with NDA for patient care (see Appendix G). Current regulations treat radiopharmaceuticals like therapeutics, not accounting for the major administered mass differences between tracer molecules and therapeutic drugs. Establishment of a new regulatory paradigm or regulatory discretion may lead to an increase of new tracers, supporting the need for more radiochemists
Finally, isotope availability is an important factor for the field. The lack of an adequate national supply of medical radioisotopes, especially 99Mo, creates a reliance on foreign sources. Fluctuation in foreign supply streams creates an uncertain future for 99mTc radiopharmaceuticals. Development of a national facility for long-lived isotope production would reduce the foreign dependence and create more demand for radiochemists.
The nuclear and radiochemistry workforce within the field of nuclear medicine is a vital and important component. Radiochemists and related disciplines support the infrastructure that is needed to prepare the imaging agents and radiotherapeutics for research and patient care. According to market reports and predictions from several company representatives, the field is growing and a significant increase in the number of trained personnel will be needed at every level of education (including B.S., M.S., Ph.D., and Phar.M.D.).
Bading, J. R., and A. F. Shields. 2008. Imaging of cell proliferation: Status and prospects. J Nucl Med 49 Suppl 2:64S-80S.
BPS (Board of Pharmacy Specialties). 2012. Nuclear Pharmacy [online]. Available: http://www.bpsweb.org/specialties/nuclear.cfm [accessed March 7, 2012].
BTSI (Bio-Tech System, Inc.). 2006. The World Market for Therapeutic Radiopharmaceuticals. Report 230. Bio-Tech System, Inc. [online]. Available: http://www.biotechsystems.com/downloads/BTSI230.pdf [accessed November 8, 2011].
BTSI. 2010. The Market for PET Radiopharmaceuticals and PET Imaging. Report 320. Bio-Tech System, Inc. [online]. Available: http://www.biotechsystems.com/downloads/BTSI320.pdf [accessed November 8, 2011].
BTSI. 2011. The US Market for SPECT and PET Radiopharmaceuticals. Report 330. Bio-Tech System, Inc. [online]. Available: http://www.biotechsystems.com/downloads/BTSI330.pdf [accessed November 7, 2011].
Center for Health Workforce Studies. 2007. Nuclear Medicine Scientists: Findings and Recommendations Based on a 2006 Survey. Center for Health Workforce Studies, School of Public Health, University at Albany, Rensselaer, NY [online]. Available: http://www.snm.org/docs/NM_ScientistReport070212.pdf [accessed October 4, 2011].
Center for Molecular Imaging. 2011. Beneficial Medical Uses of Radiation [online]. Available: http://www.molecularimagingcenter.org/index.cfm?PageID=7083 [accessed October 4, 2011].
Chitneni, S. K., G. M. Palmer, M. R. Zalutsky, and M. W. Dewhirst. 2011. Molecular imaging of hypoxia. J Nucl Med 52(2):165-168.
Cyclotron Co. 2010. 68Ga–generator. Cyclotron Radio Isotope Production [online]. Available: http://www.cyclotronzao.ru/?page=generator-galiya-68&lng=eng&design=pages [accessed November 10, 2011].
DOE (U.S. Department of Energy). 2005. FY 2006 Congressional Budget Request, Vol. 4. DOE/ME-0049. Office of Management, Budget and Evaluation/CFO. February 2005 [online]. Available: http://www.mbe.doe.gov/budget/06budget/Content/Volumes/Vol_4_SC_DA.pdf [accessed November 10. 2011].
DOE. 2011. BER Budgets [online]. Available: http://science.energy.gov/ber/about/ber-budget/ [accessed November 10, 2011].
IAEA (International Atomic Energy Agency). 2003. Manual for Reactor Produced Radioisotopes. IAEA-TECDOC-1340. Vienna: IAEA [online]. Available: http://www.isotopes.gov/outreach/reports/Reactor_Isotopes.pdf [accessed November 10, 2011].
IAEA. 2009. Cyclotron Produced Radionuclides: Physical Characteristics and Production Methods. Technical Report No. 468. Vienna: IAEA [online]. Available: http://www-pub.iaea.org/MTCD/publications/PDF/trs468_web.pdf [accessed November 10, 2011].
Maddahi, J., J. Czernin, J. Lazewatsky, S. -C. Huang, M. Dahlbom, H. Schelbert, R. Sparks, A. Ehlgen, P. Crane, Q. Zhu, M. Devine, and M. Phelps. 2011. Phase I, first-in-human study of BMS747158, a novel 18F-labeled tracer for myocardial perfusion PET: dosimetry, biodistribution, safety, and imaging characteristics after a single injection at rest. J Nucl Med 52(9):1490-1498.
Nelissen, N., K. Van Laere, L. Thurfjell, R. Owenius, M. Vandenbulcke, M. Koole, G. Bormans, D. J. Brooks, and R. Vandenberghe. 2009. Phase 1 study of the Pittsburgh compound B derivative 18F-flutemetamol in healthy volunteers and patients with probable Alzheimer disease. J Nucl Med 50(8):1251-1499.
NRC/IOM (National Research Council and Institute of Medicine). 2007. Advancing Nuclear Medicine through Innovation. Washington, DC: The National Academies Press.
PMLiVE. 2012. Bayer on Track to File Prostate Cancer Drug in 2012. PMLiVE, February 13, 2012 [online]. Available: http://www.pmlive.com/pharma_news/bayer_on_track_to_file_prostate_cancer_drug_in_2012_370132 [accessed March 7, 2012].
PRWeb News Wire. 2009. Radiopharmaceuticals Market in US and Europe to reach $5.4 Billion by 2015, According to New Report by Global Industry Analysts. PRWeb News Wire, February 18, 2009 [online]. Available: http://www.prweb.com/releases/radiopharmaceuticals/diagnostic_therapeutic/prweb2080594.htm [accessed November 7, 2011].
PRWeb News Wire. 2010. Global Nuclear Medicine Market to Reach US $1.69 Billion by 2015, According to New Report by Global Industry Analysts, Inc. PRWeb News Wire, October 21, 2010 [online]. Available: http://www.prweb.com/releases/nuclear_medicine_PET/scanners_gamma_cameras/prweb4677724.htm [accessed November 7, 2011].
Rowe, C. C., U. Ackerman, W. Browne, R. Mulligan, K. L. Pike, G. O’Keefe, H. Tochon-Danguy, G. Chan, S. U. Berlangieri, G. Jones, K. L. Dickinson-Rowe, H. P. Kung, W. Zhang, M. P. Kung, D. Skovronsky, T. Dyrks, G. Holl, S. Krause, M. Friebe, L. Lehman, S. Lindemann, L. M. Dinkelborg, C. L. Masters, and V. L. Villemagne. 2008. Imaging of amyloid beta in Alzheimer’s disease with 18F-BAY94-9172, a novel PET tracer: Proof of mechanism. Lancet Neurol 7(2):129-135.
Seidlin, S., L. D. Marinelli, and E. Oshry. 1946. Radioactive iodine therapy: Effect on functioning metastases of adenocarcinoma of the thyroid. JAMA 132(14):838-847.
Unterweger, M. P., D. D. Hoppes, F. J. Schima, and J. S. Coursey. 2010. Radionuclide Half-Life Measurements. The National Institute of Standards and Technology [online]. Available: http://www.nist.gov/pml/data/halflife-html.cfm [accessed November 10, 2011].
Wadas, T. J., E. H. Wong, G. R. Weisman, and C. J. Anderson. 2010. Coordinating radiometals of copper, gallium, indium, yttrium and zirconium for PET and SPECT imaging of disease. Chem. Rev. 110(5):2858-2902.
Wong, D. F., P. B. Rosenberg, Y. Zhou, A. Kumar, V. Raymont, H. T. Ravert, R. F. Dannals, A. Nandi, J. R. Brasiæ, W. Ye, J. Hilton, C. Lyketsos, H. F. Kung, A. D. Joshi, D. M. Skovronsky, and M. J. Pontecorvo. 2010. In vivo imaging of amyloid deposition in Alzheimer disease using the radioligand 18F-AV-45 (florbetapir [corrected] F 18). J Nucl Med 51(6):913-920.