Approximately 1.1 million people in the United States currently are living with HIV (CDC, 2012d; Hall et al., 2012). Of these individuals, only about 82 percent have been diagnosed (CDC, 2012d,f; Hall et al., 2012)1; fewer still are receiving clinical care for HIV, including antiretroviral therapy (ART); and only 19 to 25 percent have achieved viral suppression (Burns et al., 2010; CDC, 2012d; Gardner et al., 2011; Hall et al., 2012). In July 2010, the White House Office of National AIDS Policy (ONAP) released its National HIV/AIDS Strategy (NHAS), the primary goals of which are to (1) reduce the number of people who become infected with HIV, (2) increase access to care and optimize health outcomes for people with HIV, and (3) reduce HIV-related health disparities (ONAP, 2010). The NHAS identifies action steps for each of the three primary goals and sets quantitative targets to be achieved by 2015 (see Box 1-1).
In September 2010, ONAP commissioned the Institute of Medicine (IOM) to establish a committee of experts to review public and private data systems that capture information on the care of people with HIV and to recommend ways to utilize and supplement existing data to track the impact of the NHAS and the Patient Protection and Affordable Care Act (ACA) (P.L. 111-148) on improving HIV care in the United States. In response to this charge, the IOM convened the 17-member Committee to Review Data Systems for Monitoring HIV Care composed of experts in clinical HIV care, mental health, health services research, private health insurance, health
_________________
1Eighteen percent undiagnosed in 2009 is an improvement over the 20 percent of people living with undiagnosed HIV in 2006 (CDC, 2012f).
BOX 1-1
National HIV/AIDS Strategy Action Steps and Targets
Reducing New HIV Infections
Action Steps
- Intensify HIV prevention efforts in communities where HIV is most heavily concentrated.
- Expand targeted efforts to prevent HIV infection using a combination of effective, evidence-based approaches.
- Educate all Americans about the threat of HIV and how to prevent it.
Targets By 2015,
- lower the annual number of new infections by 25 percent.
- reduce the HIV transmission rate, which is a measure of annual transmissions in relation to the number of people living with HIV, by 30 percent.
- increase from 79 to 90 percent the percentage of people living with HIV who know their serostatus.
Increasing Access to Care and Improving Health Outcomes for People Living with HIV
Action Steps
- Establish a seamless system to immediately link people to continuous and coordinated quality care when they are diagnosed with HIV.
- Take deliberate steps to increase the number and diversity of available
- providers of clinical care and related services for people living with HIV. • Support people living with HIV with co-occurring health conditions and those who have challenges meeting their basic needs, such as housing.
Targets By 2015,
- increase the proportion of newly diagnosed patients linked to clinical care within 3 months of their HIV diagnosis from 65 to 85 percent.
policy, housing policy, the Ryan White HIV/AIDS Program, biostatistics, epidemiology, health disparities, and biomedical informatics (see the Appendix, Biographical Sketches of Committee Members).
The committee, which was given a two-part statement of task (Box 1-2), released its first report, Monitoring HIV Care in the United States: Indicators and Data Systems, in March 2012 (IOM, 2012). Responding to the first part of the committee’s statement of task, the report identifies core and additional indicators related to continuous clinical HIV care and access
- increase the proportion of Ryan White HIV/AIDS Program clients who are in care (at least two visits for routine HIV medical care in 12 months at least 3 months apart) from 73 to 80 percent.
- increase the percentage of Ryan White HIV/AIDS Program clients with permanent housing from 82 to 86 percent.
Reducing HIV-Related Health Disparities and Health Inequities
Action Steps
- Reduce HIV-related mortality in communities at high risk for HIV infection.
- Adopt community-level approaches to reduce HIV infection in high-risk communities.
- Reduce stigma and discrimination against people living with HIV.
Targets By 2015,
- increase the proportion of HIV diagnosed gay and bisexual men with undetectable viral load by 20 percent.
- increase the proportion of HIV diagnosed Black Americans with undetectable viral load by 20 percent.
- increase the proportion of HIV diagnosed Latinos with undetectable viral load by 20 percent.
Achieving a More Coordinated National Response to the HIV Epidemic in the United States
- Increase the coordination of HIV programs across the federal government and between federal agencies and state, territorial, local, and tribal governments.
- Develop improved mechanisms to monitor and report on progress toward achieving national goals.
to supportive services, such as housing, food and nutrition, and transportation, as well as 12 public and private data collection efforts the committee concluded would be the most useful for estimating the indicators in order to monitor the effect of the NHAS and the ACA on access to and provision of quality HIV care.
The report discusses how data can be used to estimate the committee’s recommended indicators and describes potential barriers to and the role of health information technology in the collection and linkage of data pertain-
The White House Office of National AIDS Policy has requested that the Institute of Medicine (IOM) convene a committee of experts to assess available public and private data systems that capture information about HIV care to investigate ways to maximize their usefulness and recommend approaches for supplementing current data sources and to identify and provide recommendations for the most critical data and indicators to gauge the impact of the National HIV/AIDS Strategy and the Patient Protection and Affordable Care Act in improving HIV/AIDS care.
The committee will address the following questions in its first consensus report:
1. What are the best sources of data (and which data elements should be used) from public and private HIV care databases to assess core indicators related to continuous care and access to supportive services, such as housing, for people living with HIV?
a. What data collection items need to be revised or reconsidered in existing databases of care and services provided to people living with HIV and in demographic data about populations receiving these services? Are there proposed changes that can provide necessary data without adding additional burden to data collection?
b. What is the difference between claims data and clinical data found in medical records and do these differences encompass gaps in measures for HIV care?
2. What similar data collection or standardization efforts are currently under way by public agencies or private industry that should be tapped?
3. How do we regularly obtain data (core indicators) that capture the care experiences of people living with HIV without substantial new investments?
4. What situations may impose barriers to the collection of core indicators?
a. What policies, reimbursement issues or reporting issues need to be addressed to collect necessary data?
ing to HIV care, as well as the possibilities and challenges of combining data from different data systems to estimate the indicators identified by the committee.
The current report, which responds to the remaining portion of the committee’s charge, addresses how to monitor the changes in health care coverage, service utilization, and quality of care for people with HIV that are anticipated under the ACA. Almost 30 percent of people with HIV in the United States have no source of health care coverage and only 17
b. How can data be collected in a way that will not significantly increase provider burden?
5. How can federal agencies efficiently analyze care indicators and disseminate data to improve HIV care quality?
6. What models or best practices in data system integration can be gleaned from public agencies or private industry to make existing data systems and core indicators interoperable?
a. Which among these models or combination of models would be most cost effective?
7. How should health information technology (including electronic medical records) be utilized and configured in order to improve the collection of comprehensive data describing the care experiences of people living with HIV?
In a second consensus report, the committee will address the following question:
1. How do we obtain national estimates that characterize the health care of people living with HIV in public and private settings?
a. How can we obtain data from a nationally representative sample of HIV positive individuals in the United States to establish a baseline for health insurance and health care access status prior to 2014?
b. If it is not possible to obtain a nationally representative sample of people living with HIV are there other alternatives (including using multiple existing data sources or requiring a complete accounting of all positive persons in care) to obtain data on care and utilization beyond those individuals enrolled in Ryan White?
c. How do we continue to regularly obtain data from a large sample (nationally representative or otherwise) of HIV-positive individuals after 2014 to monitor the impact of the Affordable Care Act on health insurance and health care access?
percent have private insurance; the remaining 53 percent are covered by government programs such as Medicaid, Medicare, and the Ryan White HIV/AIDS Program (HHS, 2012a). Many aspects of health reform under the ACA, including expansion of Medicaid eligibility requirements, elimination of preexisting condition exclusions, and lifting of annual and lifetime dollar limits on care, should substantially decrease the number of people with HIV who have no health care coverage and possibly result in changes to sources of coverage for others.
THE COMMITTEE’S APPROACH TO ITS CHARGE
The committee’s two reports complement one another. In considering the statement of task for the current report, the committee thought that the first, overarching, question about how to obtain “national estimates that characterize the health care of people living with HIV in public and private settings” had been addressed already in its first report. One of the questions posed in the statement of task for the first report asks about how to “obtain data (core indicators) that capture the care experiences of people living with HIV without substantial new investments,” and the first report focuses primarily on the identification of indicators and data systems for and approaches to estimating the indicators and monitoring the quality of clinical HIV care and access to mental health, substance abuse, and supportive services for people with diagnosed HIV in the United States. Although the statement of task for the first report does not specifically mention “national” or “nationally representative” estimates, the committee was mindful of this goal in its approach to the first report, including its identification of public and private data collection efforts for estimating the recommended indicators.
The present report focuses on the collection of data on health coverage status and data systems needed to address the current statement of task subquestions (a) through (c) pertaining to the establishment of “a baseline for health insurance and health care access status prior to 2014” and to monitoring “the impact of the [ACA] on health insurance and health care access” following its implementation in 2014. The committee considered subquestions (a) and (b) to be linked. The first asks how to obtain a nationally representative sample of people living with HIV in the United States prior to 2014, and the second poses a conditional question about alternative ways, including the use of “multiple existing data sources or requiring a complete accounting of all HIV-positive persons in care,” to obtain “data on care and utilization beyond those individuals enrolled in the Ryan White HIV/AIDS Program” if it is not possible to obtain a nationally representative sample.
The committee interpreted this conditional question to apply to the acquisition of necessary data to establish a baseline for health care coverage and utilization status prior to 2014, if it concluded it were not currently possible to collect these data from a nationally representative sample of HIV-diagnosed individuals in the United States. The committee understood subquestion (c) to be a forward-looking opportunity to recommend an ongoing, dynamic strategy for capturing data from a nationally representative sample of HIV-diagnosed individuals in the United States. Given the challenges discussed in the committee’s first report of collecting and combining data from disparate systems to generate an overall picture of the care
experiences of people with HIV in the United States (IOM, 2012, Chapters 4-6), development of a unique mechanism for capturing relevant information would simplify the collection and analysis of data and provide more detailed and representative data than currently exist to monitor the impact of the ACA on health care coverage and utilization for people with HIV.
The committee recognized that the development and maintenance of a successful, ongoing, dynamic strategy for capturing data from a nationally representative sample of HIV-diagnosed individuals in the United States will require sufficient funding. However, it determined that a detailed cost analysis and budgetary recommendation was beyond the scope of its charge.
The committee’s two reports, although distinct, do overlap in certain ways. For example, as discussed in later chapters, it will be important to monitor care quality using indicators such as those recommended in the committee’s first report in addition to tracking the movement of individuals into and among different sources of health coverage, which is the purview of the present report. In addition, the first report examined how best to obtain data to estimate the indicators using existing data systems and “without substantial new investments.” The same ongoing mechanism recommended in the present report to capture data from a nationally representative sample of HIV-diagnosed individuals in the United States “to monitor the impact of the ACA on health insurance and health care access” potentially could also be used to simplify the collection and analysis of data to generate a national estimate of the indicators recommended in the first report.
Although the statement of task refers generally to “people living with HIV,” for the purpose of this report, the committee interpreted this phrase as referring to people living with a diagnosis of HIV in the United States, since there is no practical way to obtain a “large sample (nationally representative or otherwise)” of people living with HIV that includes individuals with undiagnosed infection.2 In addition, the present report limits the population under consideration to adults and adolescents (ages 13 and older). The use of ART to reduce or prevent perinatal transmission of the virus has resulted in a relatively small number of newly diagnosed pediatric HIV cases in the United States each year (CDC, 2012a, Table 1a). Although there
_________________
2The availability of rapid home-based HIV testing in the wake of the U.S. Food and Drug Administration’s approval of an over-the-counter rapid HIV test in July 2012 “has the potential to identify large numbers of previously undiagnosed HIV infections especially if used by those unlikely to use standard screening methods” (FDA, 2012). Availability of such testing may affect timely reporting of individuals newly diagnosed through home-based testing to public health authorities for surveillance purposes and timely linkage of those individuals to care, as well as related measures pertaining to HIV diagnosis and linkage to care. Speculation on specific ways in which home-based testing may affect these measures is premature and beyond the scope of the committee’s charge.
is a similarly low rate of new HIV diagnoses among 13- to 14-year-olds (CDC, 2012a, Table 1a), adolescents age 13 and older fall within the same HIV screening and treatment guidelines as adults (Branson et al., 2006; HHS, 2012b) and therefore are included with the adult population. For ease of reference, however, the report generally uses variations of “people with HIV” to refer to adults and adolescents living with a diagnosis of HIV. Where it is important to distinguish among diagnostic status and age groups (e.g., in the epidemiology section of this chapter), the committee took care to do so.
In considering the statement of task, the committee interpreted the term “health insurance” broadly to include all types of private and public plans or programs that cover health care and related services, including private health insurance; federal programs (e.g., Medicaid, Medicare, Ryan White HIV/AIDS Program, military health care, Indian Health Service); and individual state health plans. Since not all of the programs that cover care for people living with HIV (e.g., Ryan White HIV/AIDS Program, Veterans Health Administration) are insurance, this report preferentially uses the term health care “coverage.”
For its first report, the committee considered more than 30 data systems and data collection efforts, including some that are HIV specific and others that are not but capture information on people living with HIV. The committee then requested information from 29 public and private data systems on the types of data collected (e.g., data on demographics, access to care, need for supportive services) to identify best sources of data to estimate core indicators of HIV care. The committee revisited these data sources for this second report to identify those that capture data relevant to monitoring health care coverage and utilization for people with HIV within the context of the ACA. Data of particular interest include enrollment and demographic information; sources of health coverage; and utilization of care, preventive, and supportive services. The committee considered which data collection efforts, including convenience samples (e.g., North American AIDS Cohort Collaboration on Research and Design [NA-ACCORD], CFAR [Centers for AIDS Research] Network of Integrated Clinical Systems [CNICS], HIV Research Network [HIVRN]) and national health-related surveys (e.g., National Health Interview Survey [NHIS], National Health and Nutrition Examination Survey [NHANES], National Health Care Surveys), could best capture data on health care coverage and utilization for a nationally representative sample of people with HIV in the United States and which best capture these data at the state level. The committee also took into account the findings from its first report on the extent to which various data sources capture information to estimate indicators of care quality and outcomes for people with HIV. Care quality and outcomes are important to monitor as
the ACA is implemented because continuity of care may be disrupted and the range of benefits available to individuals may change as they move into and among sources of care coverage.
The remainder of Chapter 1 discusses the current epidemiology of the HIV epidemic, provides background on clinical HIV care and supportive services, and discusses the importance of an ongoing strategy for monitoring health care coverage and utilization for people with HIV in the United States. The chapter also highlights some examples of current and past broad-based data collection efforts. Chapter 2 gives a selective overview of the ACA; discusses ways in which implementation of the ACA is likely to affect people with HIV in the United States; and highlights some of the challenges of implementing the ACA. Chapter 3 provides background information to support the committee’s conclusions and recommendations for how to establish a baseline for health care coverage, utilization, and quality prior to the implementation of the ACA; how to obtain relevant data from a nationally representative sample of people with HIV in the United States; and how to continue to monitor the impact of the ACA on these outcomes. The chapter first provides background information on the limitations of national health-related surveys for collecting data on specific diseases such as HIV and the sampling design and methodology used in the HIV Cost and Services Utilization Study (HCSUS), the first nationally representative study of HIV-infected adults in care in the United States. Referring to the committee’s first report, it then notes the challenges and possibilities that attend establishing a baseline of health care coverage and utilization prior to 2014. The chapter next describes and discusses the strengths and limitations of the Medical Monitoring Project, a surveillance project conducted by the Centers for Disease Control and Prevention (CDC) that is currently designed to obtain nationally representative estimates of the clinical and behavioral characteristics of HIV-diagnosed individuals in care. The chapter also discusses how data from programs most apt to be affected by health reform, particularly Medicaid, Medicare, and the Ryan White HIV/AIDS Program, will be needed to monitor changes in health care coverage and utilization for people with HIV in the context of the ACA at the state and programmatic level. Chapter 4 contains the committee’s conclusions and recommendations for monitoring health care coverage, utilization, and outcomes for people with HIV prior to and following full implementation of the ACA in 2014.
Epidemiology of HIV
The epidemiology of the HIV epidemic is important for evaluating how representative different data collection efforts are of the national HIV-diagnosed population and for highlighting areas in which they may need to improve. As outlined in this section, differences in the racial and ethnic distribution of HIV-infected individuals among different regions of the country, differences in transmission category between men and women, and differences in prevalence trends among age groups illustrate the complexity of the HIV epidemic in the United States. Such variations, along with other factors such as differences in socioeconomic status, highlight the need for careful attention to sampling in any effort to obtain a nationally representative sample of people living with HIV in the United States.
There has been a significant shift in the HIV epidemic in the United States since the first cases were reported in the 1980s. In the early years of the epidemic, the virus that leads to AIDS had not yet been identified and treatments were limited, resulting in an epidemiologic focus on AIDS diagnoses and deaths. Within 11 years, the number of people diagnosed with AIDS grew rapidly from about 300 in 1981 to more than 75,000 in 1992, and the disease accounted for more than 50,000 deaths in 1995 (CDC, 2012c). In recent years, the annual rate of new AIDS diagnoses has decreased from 11.5 per 100,000 in 2007 to 10.8 per 100,000 in 2009 and 2010, while the death rate has held steady at 5.8 per 100,000 (CDC, 2012a, Tables 2b and 12a). Advances HIV diagnosis and treatment have contributed to the decrease in AIDS diagnoses as well as increased prevalence of non-AIDS HIV cases. With the advent of highly active antiretroviral therapy, HIV has become a chronic disease, and infected individuals are living longer, healthier lives. Although the number of newly HIV-infected individuals has stabilized at approximately 50,000 per year in the past few years (CDC, 2012c), the number of people estimated to be living with HIV in the United States has increased 8 percent from 2006 through 2009 (CDC, 2012f, Table 5b) because there are fewer deaths than new infections each year. In addition, increased testing and resultant diagnoses have contributed to a 9 percent increase in the number of people living with diagnosed HIV infection in the same time period. By the end of 2009, an estimated 1,148,200 people 13 years of age and older were living with HIV/AIDS in the United States, including an estimated 207,600 people with undiagnosed HIV/AIDS (CDC, 2012f, Table 5a). The increase in HIV prevalence has important resource implications for the diagnosis and care of people living with HIV.
Geographic Variations
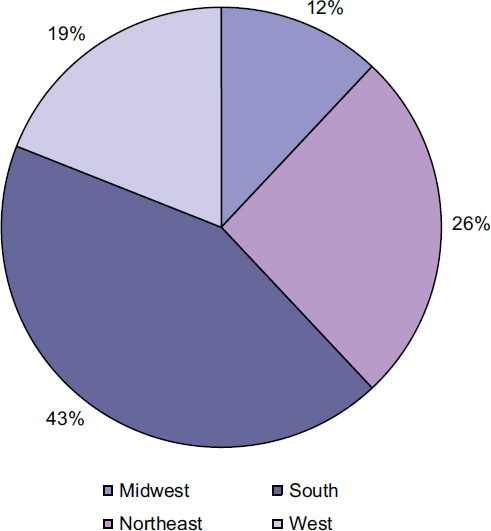
The prevalence of individuals living with diagnosed HIV varies among U.S. regions and among racial and ethnic groups within the regions (Table 1-1).3 In addition, there have been geographical shifts in the distribution of HIV in the United States. The burden of the epidemic, which was initially concentrated in major metropolitan areas such as San Francisco and New York City, has expanded over time to include more rural areas and the Southeast. In 2009, the southern region had the highest percentage of reported individuals with diagnosed HIV; 43 percent of people living with diagnosed HIV in the United States were living in the South (CDC, 2012a, Table 21; see Figure 1-1). In addition, the South had the highest percentage (45 percent) and the second highest rate (13 per 100,000) of newly diagnosed AIDS cases in the United States in 2010 (CDC, 2012f, Table 4a). The Northeast has the next highest percentage of people living with diagnosed HIV (26 percent) and of new AIDS diagnoses (24 percent), but the highest rate of newly diagnosed AIDS cases (14.2 per 100,000) (CDC, 2012a, Table 21; 2012f, Table 4a). The West accounts for 19 percent of people living with diagnosed HIV and of new AIDS cases, and the Midwest accounts for 12 percent and 13 percent of each respectively (CDC, 2012a, Table 21; 2012f, Table 4a).
The CDC also reports data for the U.S. dependent areas of American Samoa, Guam, Northern Mariana Islands, Puerto Rico, Republic of Palau, and U.S. Virgin Islands (CDC, 2012a, Table 21). The U.S. Virgin Islands and Puerto Rico have the highest rates of adolescents and adults living with diagnosed HIV (approximately 633 and 556 per 100,000 respectively).
Racial and Ethnic Variations
Overall, racial and ethnic minorities are disproportionately affected by HIV (Figure 1-2). Although blacks/African Americans accounted for 14 percent of the U.S. population in 2009, they represented 43 percent of people living with diagnosed HIV in 2009 and 46 percent of new HIV diagnoses in 2010 (CDC, 2011a, 2012a, Tables 1a and 15a). The rate of
_________________
3The CDC (2012a, p. 14) divides the country into four geographic regions—Northeast, South, Midwest, and West. States that comprise the Northeast are Connecticut, Maine, Massachusetts, New Hampshire, New Jersey, New York, Pennsylvania, Rhode Island, and Vermont. Areas of residence that comprise the South are Alabama, Arkansas, Delaware, District of Columbia, Florida, Georgia, Kentucky, Louisiana, Maryland, Mississippi, North Carolina, Oklahoma, South Carolina, Tennessee, Texas, Virginia, and West Virginia. States that comprise the Midwest are Illinois, Indiana, Iowa, Kansas, Michigan, Minnesota, Missouri, Nebraska, North Dakota, Ohio, South Dakota, and Wisconsin. States that comprise the West are Alaska, Arizona, California, Colorado, Hawaii, Idaho, Montana, Nevada, New Mexico, Oregon, Utah, Washington, and Wyoming.
| Region | Race/Ethnicity | Number | Rate per 100,000a |
| Northeast | American Indian/Alaska Native | 216 | 101.5 |
| Asian | 2,121 | 69.3 | |
| Black/African American | 98,296 | 1,500.7 | |
| Hispanic/Latino | 60,655 | 867.5 | |
| Native Hawaiian/Other Pacific | 69 | 335.7 | |
| Islander | |||
| White | 58,587 | 154.1 | |
| Multiple race | 5,285 | 487.5 | |
| Regional | 209,600 | 384.3 | |
| South | American Indian/Alaska Native | 862 | 93.3 |
| Asian | 1,509 | 47 | |
| Black/African American | 205,786 | 935.2 | |
| Hispanic/Latino | 43,464 | 238.5 | |
| Native Hawaiian/Other Pacific Islander | 163 | 196.6 | |
| White | 106 627 | 155.2 | |
| Multiple race | 3,926 | 184.0 | |
| Regional | 358,411 | 314.3 | |
aThe rate per 100,000 was for this table was determined by dividing the reported number of HIV-infected individuals (CDC, 2012a) by the total ethnic/racial populations for each of the regions (Ennis et al., 2011; Hixson et al., 2011, 2012; Hoeffel et al., 2012; Norris et al, 2012; Rastogi et al., 2011; U.S. Census, 2012a) and multiplied by 100,000.
blacks/African Americans living with diagnosed HIV is seven times that of whites (951.9 compare to 143.9 per 100,000) (CDC, 2012a, Table 15a). Hispanics/Latinos accounted for 16 percent of the U.S. population but 19 percent of people living with diagnosed HIV in 2009 and 20 percent of new HIV diagnoses in 2010 (CDC, 2011b, 2012a, Tables 1a and 15a). The rate of Hispanics/Latinos living with diagnosed HIV is 319.9 per 100,000, more than two times that of whites (CDC, 2012a, Table 15a). Multiracial individuals also are more likely to be living with an HIV diagnosis than whites (286.4 compared to 143.9 per 100,000) (CDC, 2012a, Table 15a).
Even though the rates of American Indians/Alaska Natives and Native Hawaiians/Other Pacific Islanders living with a diagnosis of HIV (130.8
| Region | Race/Ethnicity | Number | Rate per 100,000a |
| Midwest | American Indian/Alaska Native | 420 | 91.6 |
| Asian | 821 | 47.5 | |
| Black/African American | 44,467 | 639.7 | |
| Hispanic/Latino | 9,161 | 196.5 | |
| Native Hawaiian/Other Pacific | 56 | 189.5 | |
| Islander | |||
| White | 43,138 | 82.8 | |
| Multiple race | 1,494 | 130.1 | |
| Regional | 98,063 | 148.4 | |
| West | American Indian/Alaska Native | 1,655 | 123.8 |
| Asian | 4,476 | 67.1 | |
| Black/African American | 26,100 | 762.5 | |
| Hispanic/Latino | 42,766 | 207.6 | |
| Native Hawaiian/Other Pacific | 554 | 136.1 | |
| Islander White | 82 762 | 217.8 | |
| Multiple race | 1,414 | 53.8 | |
| Regional | 158,313 | 219.8 | |
SOURCE: Based on CDC, 2012a, Table 21; Ennis et al., 2011, Table 2; Hixson et al., 2011, Table 4, 2012, Table 2; Hoeffel et al., 2012, Table 2; Norris et al., 2012, Table 2; Rastogi et al., 2011, Table 5; U.S. Census, 2012a (following the guidelines in U.S. Census, 2012b).
and 184.5 per 100,000, respectively) are comparable to that of whites (CDC, 2012a, Table 15a), their rates of HIV and AIDS diagnoses in 2010 were significantly greater. Although American Indians/Alaska Natives are 10 percent more likely than non-Hispanic whites to have ever been tested for HIV (Schiller et al., 2012, Table 41), they were 30 percent and 60 percent more likely than whites to be diagnosed with HIV and AIDS (CDC, 2012a, Tables 3a and 4a). Native Hawaiians/Other Pacific Islanders were 260 percent and 220 percent more likely than whites to have been diagnosed with HIV and AIDS (CDC, 2012a, Tables 3a and 4a) yet were 20 percent less likely to have ever been tested (Schiller et al., 2012, Table 41). In addition, both groups were estimated to have a higher percentage of
people living with undiagnosed HIV infection in 2009 compared to whites (CDC, 2012f, Table 5b).
Asian Americans were 10 percent less likely than non-Hispanic whites to have ever been tested for HIV (Schiller et al., 2012, Table 41). In addition, an estimated 27.4 percent of Asian Americans living with HIV in 2009 were undiagnosed, the highest percentage of undiagnosed individuals among all racial and ethnic groups (CDC, 2012f, Table 5b).
Variations by Sex
The proportion of people living with HIV in the United States also varies by sex. Approximately 75 percent of people living with diagnosed HIV in the United States are male (CDC, 2012a, Table 15a). In addition, there
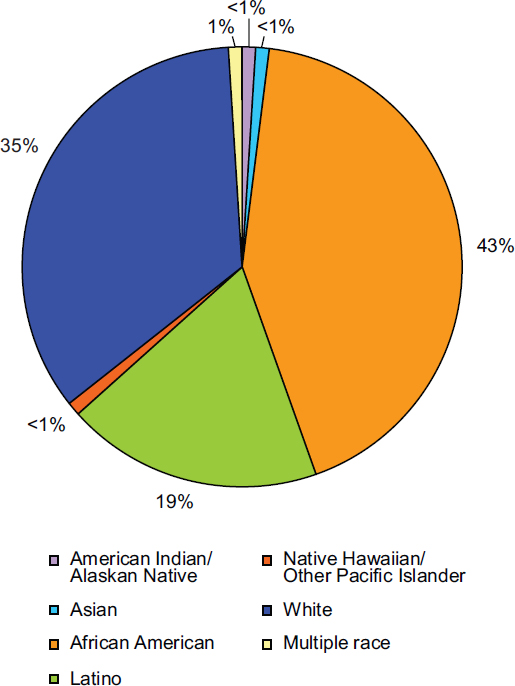
FIGURE 1-2 Percentage of racial and ethnic populations living with a diagnosis of HIV, year-end 2009.
SOURCE: CDC, 2012a, Table 15a.
are differences between males and females in the distribution of HIV transmission categories as well as race and ethnicity (see Table 1-2; Figure 1-3).
Sixty-eight percent of diagnosed HIV infections among men are acquired through male-to-male sexual contact (CDC, 2012a, p. 9, Table 15a), even though men who have sex with men (MSM) account for just 5.2 percent of sexually experienced men 18 to 59 years of age in the United
| Race or Ethnicity | Female Estimated | Male Estimated | |||||
| Number | Rate (per 100,000) | Percent of HIV-Diagnosed People | Number | Rate (per 100,000) | Percent of HIV-Diagnosed People | Total Estimated Number | |
| American Indian/Alaska Native | 782 | 83 | <1 | 2,218 | 245 | <1 | 3,000 |
| Asian | 1,457 | 27 | <1 | 6,816 | 138 | 1 | 8,273 |
| Black/African American | 114,721 | 764 | 61 | 214,464 | 1,612 | 37 | 329,185 |
| Hispanic/Latino | 31,753 | 190 | 17 | 117,101 | 647 | 20 | 148,854 |
| Native Hawaiian/Other Pacific Islander | 108 | 83 | <1 | 498 | 383 | <1 | 606 |
| White | 36,587 | 44 | 19 | 235,610 | 297 | 40 | 272,197 |
| Multiple race | 3,083 | 225 | 2 | 7,908 | 617 | 1 | 10,991 |
| Totala | 188,688 | 154 | 24 | 585,197 | 496 | 76 | 773,885 |
aBecause column totals include persons of unknown race and ethnicity, the values in each column may not sum to the column total.
SOURCE: CDC, 2012a, Table 17a.
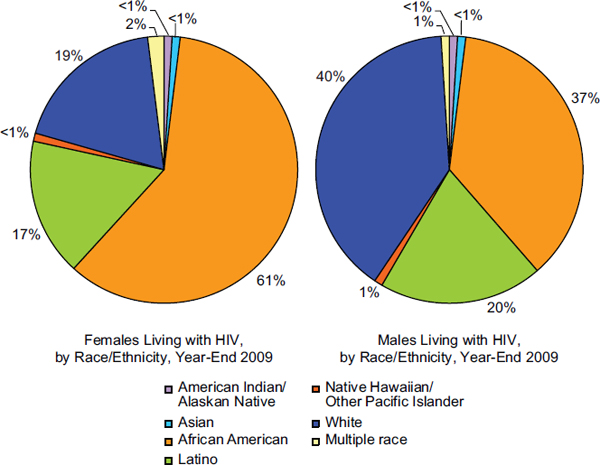
FIGURE 1-3 Percentage of females and males living with a diagnosis of HIV in the United States by racial or ethnic group, year-end 2009.
SOURCE: Based on CDC, 2012a, Table 17a.
States (Xu et al., 2010). MSM accounted for 51 percent of people living with HIV in the United States in 2009 and an estimated 61 percent of new HIV diagnoses in 2010 (CDC, 2012a, Tables 1a and 15a). Young black/African American MSM (13-29 years of age) had a 48 percent increase of new HIV infections from 2006 to 2009, making it the only risk group to have a statistically significant increase in new HIV infections during that period (Prejean et al., 2011).The remainder of diagnosed infections among males can be attributed to injection drug use (13 percent), heterosexual contact (11 percent), and combined MSM contact and injection drug use (8 percent) (CDC, 2012a, p. 9, Table 15a). The racial and ethnic distribution of males diagnosed with HIV in 2009 is similar to that of the entire population of diagnosed individuals in the United States (see Figures 1-2 and 1-3).
In contrast to the male population, 74 percent of women living with diagnosed HIV in 2009 were infected through heterosexual contact with a person known to have or to be at high risk for HIV infection (CDC, 2012a, p. 9, Table 15a). There is also a significantly different racial distribution among women with diagnosed HIV compared to HIV-diagnosed men. Black/African American females comprise 61 percent of females in the United States with an HIV diagnosis, whereas black/African American males account for 37 percent of males living with an HIV diagnosis (CDC, 2012a, Table 17a; see Figure 1-3). White females account for only 19 percent of females diagnosed with HIV in the United States, while white males represent 40 percent of the diagnosed male population (CDC, 2012a, Table 17a; see Figure 1-3).
Age Variations
The proportion of individuals living with HIV varies by age group, and there has been a change in the age distribution over time (Table 1-3). The prevalence of HIV among children under 13 years of age, and among adolescents 13 to 14 years of age, has declined significantly due to routine HIV testing of pregnant women and administration of ART to HIV-infected women. Between 2007 and 2009, the number of individuals living with a diagnosis of HIV in each age group decreased by 21 and 17 percent respectively (Table 1-3; CDC, 2102a, Table 15a). In addition, there is a significant increase in the number of people living with diagnosed HIV between the 13- to 19-year-old and the 20- to 29-year-old age groups (Table 1-3; CDC, 2102a, Table 15a).
The age groups that had the largest increase in the number of people living with an HIV diagnosis between 2007 and 2009 were the 50-to-59 and the 60-and-older groups due to increased survival among people in care, as well as to new infections. From 2007 to 2009, the number of HIV-diagnosed individuals in those age groups increased 20 and 31 percent
TABLE 1-3 People Living with a Diagnosis of HIV in the United States by Age Group, 2007-2009
| Age at End of Year | 2007 Estimated | 2008 Estimated | 2009 Estimated | |||
| Number | Rate | Number | Rate | Number | Rate | |
| <13 | 3,729 | 7.4 | 3,318 | 6.5 | 2,945 | 5.7 |
| 13-14 | 1,459 | 18.4 | 1,362 | 17.4 | 1,218 | 15.7 |
| 15-19 | 6,611 | 32.2 | 6,947 | 33.7 | 6,983 | 34.1 |
| 20-24 | 21,047 | 104.1 | 23,374 | 115.1 | 25,866 | 126.1 |
| 25-29 | 42,992 | 214.7 | 44,701 | 218.8 | 46,394 | 224.7 |
| 30-34 | 61,326 | 332.8 | 62,330 | 335.4 | 63,541 | 335.6 |
| 35-39 | 101,426 | 508.4 | 96,619 | 487.3 | 91,523 | 468.2 |
| 40-44 | 145,078 | 700.0 | 140,427 | 692.1 | 134,215 | 673.8 |
| 45-49 | 139,947 | 647.2 | 148,956 | 688.4 | 155,757 | 719.0 |
| 50-54 | 100,497 | 505.0 | 108,914 | 535.2 | 117,290 | 567.7 |
| 55-59 | 59,313 | 343.1 | 66,005 | 374.9 | 73,759 | 409.3 |
| 60-64 | 27,935 | 203.5 | 32,581 | 227.5 | 37,066 | 246.9 |
| ≥65 | 21,688 | 60.2 | 24,816 | 67.3 | 28,143 | 74.8 |
SOURCE: Based on CDC, 2012a, Table 15a.
respectively.4 In contrast, the number of people aged 35 to 44 years living with diagnosed HIV decreased by 8 percent in the same time period, and the number of 30- to 34-year-olds increased by only 3 percent (Table 1-3; CDC, 2012a, Table 15a).
Clinical HIV Care and Supportive Services
The advent of highly active antiretroviral therapy in the mid-1990s changed the face of the HIV epidemic. HIV care has been transformed from acute and palliative end-of-life care into chronic disease management. With appropriate treatment, infected individuals on ART can live long and relatively healthy lives. Studies have shown that the life expectancy of people with HIV who are receiving ART is similar to that of uninfected individuals who engage in unhealthy behaviors such as the heavy use of alcohol or cigarettes (ART-CC, 2009; May et al., 2011; Nakagawa et al., 2012), although life expectancy is lower among injection drug users and individuals who are diagnosed with HIV at later stages of the disease or who delay treatment (ART-CC, 2008; May et al., 2011; Nakagawa et al., 2012). Appropriate use of ART also reduces morbidity among people with HIV, as indicated by a reduction in the incidence of opportunistic infections and AIDS-defining illnesses (Iwuji et al., 2011; Mocroft et al., 1999; Moore and Chaisson, 1999; Palella et al., 1999).
In addition, awareness of one’s HIV-infected status can lead to behavior changes that reduce the risk of transmitting the virus to others, and appropriate use of ART also reduces infectivity. Initial results from the recent HIV Prevention Trials Network HPTN 052 clinical trial, funded by the National Institutes of Health, indicate that early initiation of ART reduces sexual transmission of HIV in serostatus-discordant couples by 96 percent (Cohen et al., 2011). The U.S. Department of Health and Human Services (HHS) Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents issued in March 2012 recommend the initiation of ART for all HIV-infected individuals, although the strength of the recommendation varies based on the individual’s CD4+ T-cell count (HHS, 2012b, p. E-1). In practice, the movement in the United States toward universal treatment for people with HIV had begun prior to the release of the current HHS Guidelines. In April 2010, the San Francisco and, in December 2011, the New York City departments of health already were recommending
_________________
4Individuals age 50 years and older now comprise more than 30 percent of the HIV-diagnosed population in the United States (Table 1-3; CDC, 2012a, Table 15a). Reflecting the increasing number of older adults with diagnosed HIV and the special concerns that attend HIV infection in that population, the U.S. Department of Health and Human Services added a new section on “HIV and the Older Patient” to the March 27, 2012, revision of its treatment guidelines (HHS, 2012b, pp. I-27-I-32).

FIGURE 1-4 Continuum of engagement in care.
SOURCE: Cheever, 2007.
initiation of ART regardless of individuals’ CD4 count (Charlebois et al., 2011; NYC DOHMH, 2011).
Despite the importance of appropriate antiretroviral use for reducing HIV-related morbidity and mortality and viral transmission, many people with HIV are not on ART. The goal of ART is to reduce individuals’ viral load below the level of detection. According to estimates for the United States, although approximately 77 to 80 percent of individuals being treated with ART have an undetectable viral load, only 24 to 33 percent of people with HIV are receiving ART and only 19 to 25 percent of people with HIV overall have an undetectable viral load (CDC, 2012d; Gardner et al., 2011). Primary barriers to optimal outcomes for people with HIV include late diagnosis, delayed linkage to care, poor retention in care, delayed initiation of ART, and poor adherence to ART (i.e., discontinuing or intermittent ART), as well as untreated non-HIV comorbidities and unmet basic needs (Castilla et al., 2002; Gardner et al., 2011; Justice, 2006; Lo et al., 2002). Figure 1-4 shows the continuum of engagement in HIV care and Figure 1-5 shows the number and percentage of people lost to care at various points along the care continuum.
A number of factors, including mental health symptoms and disorders, substance abuse, and difficulty securing and maintaining basic needs of housing, food, and access and transportation to medical care and supportive services, have been shown to have a significant, negative impact on the health status and outcomes of people with HIV (see, e.g., Conviser and Pounds, 2002a,b; Gaynes et al., 2007; Kidder et al., 2007; Leaver et al., 2007; Lo et al., 2002; Pence, 2009; Royal et al., 2009; Stall et al., 2003; Weiser et al., 2009a,b).
Furthermore, significant disparities exist among racial, ethnic, and sexual minorities with respect to access to and continuity of care and treatment (Espinoza et al., 2008; Gebo et al., 2005; Hall et al., 2008; Mays et al., 2011; Prejean et al., 2008, 2011; Robison et al., 2008). Several stud-
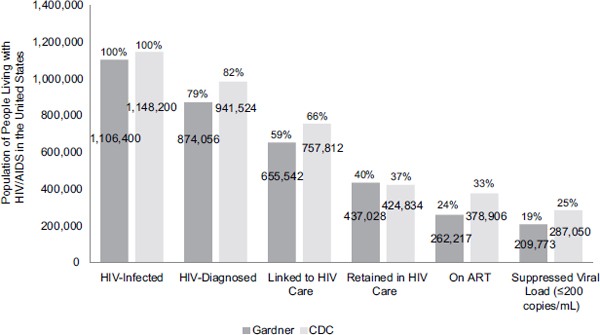
FIGURE 1-5 Engagement in HIV care cascade.
NOTE: The numbers shown are estimates and in some cases are extrapolated from single time points. For example, the CDC linkage to care estimate is based on people who were diagnosed with HIV in 2009.
SOURCES: Adapted from CDC, 2012d; Gardner et al., 2011; Hall et al., 2012.
ies report differences in the care experiences of racial or ethnic minorities compared to whites, including differences in access to health care services due to lack of health coverage, differences in the presentation of health care information and advice as a function of patient–provider interactions, and perceived bias and discrimination, although the mechanisms of how these work are not well understood (Baicker et al., 2004; Cooper-Patrick et al., 1999; Doescher et al., 2001; Garland et al., 2010; Johnson et al., 2004a,b; Korthuis et al., 2008; Schneider et al., 2002). In addition to race and ethnicity, individuals’ country of origin and citizenship status are factors that may be related to HIV-related disparities (Chen et al., 2010; Garland et al., 2010). Age, sex, and socioeconomic and health coverage status are other demographic characteristics associated with disparities in HIV care (Agwu et al., 2011; Aziz and Smith, 2011; Meditz et al., 2011; Mugavero et al., 2007; Wohl et al., 2011).
Access to HIV care also varies based on geographic area of residence, in terms of rural, urban, and suburban populations and region of the country (South, Northeast, Midwest, West) (Krawczyk et al., 2006a,b; Moon et al., 2001; Qian et al., 2006; Reif et al., 2005). People living in rural areas face barriers to accessing quality HIV care, including greater stigma regarding
HIV infection; increased fear of HIV status being disclosed; reduced availability of local HIV-knowledgeable providers; and difficulty traveling to obtain HIV care elsewhere (Heckman et al., 1998; Krawczyk et al., 2006a,b; Mays et al., 2011; Meditz et al., 2011; Moon et al., 2001; Ohl et al., 2010; Qian et al., 2006; Reif et al., 2005, 2011; Schur et al., 2002; Vermund et al. 2010; Weis et al., 2010).
The vital role of appropriate HIV care (including medical and supportive services) and the use of ART in reducing HIV morbidity, mortality, and transmission—in conjunction with the number of people with HIV who either are never engaged in or are lost to HIV care and the significant disparities in HIV care and outcomes among different demographic groups—highlights the need to monitor health care coverage, service utilization, and quality of care for people with HIV in the United States.
In its first report, the committee recommended 14 core indicators of HIV care (9 for clinical HIV care; 5 for mental health, substance use, and supportive services) for monitoring HIV care in the United States (IOM, 2012, pp. 75-77; see Table 1-4). The committee also identified 15 additional indicators (10 for clinical HIV care; 5 for mental health, substance use, and supportive services) that provide a more comprehensive assessment of quality HIV care (IOM, 2012, Chapter 2). The committee used critical benchmarks along the HIV care continuum; NHAS targets (ONAP, 2010); and existing indicators (HHS, 2010; PEPFAR, 2009), quality measures (Horberg et al., 2010; NQF, 2011), and treatment standards (HHS, 2011)5 as a basis for the HIV care indicators. In addition, the committee took account of mental health, substance use, and the need for supportive services, such as housing, food, and transportation, as mediators of HIV care in its formulation of the relevant indicators. Figure 1-6 shows the core and additional HIV care indicators mapped to the continuum of HIV care.
MONITORING HIV IN THE UNITED STATES
Development of an ongoing strategy for monitoring HIV in the United States is important for a variety of reasons: (1) monitoring the incidence of new HIV infections; (2) acquiring data on the numbers and demographic characteristics of people lost to care at various points along the HIV care continuum; (3) tracking HIV-related disparities and health inequities; (4) helping to inform potential redistribution of resources to improve the
_________________
5In its first report, the committee followed the HHS panel’s then-current recommended CD4+ T-cell count threshold of 500 cells/mm3 in formulating a core indicator for the initiation of ART. The committee further noted that if future HHS guidelines were to recommend universal treatment for people with HIV (as they now do), the committee would support a similar revision of its ART-initiation indicator or an alternate indicator tracking the time from HIV diagnosis to ART initiation (IOM, 2012, pp. 47-48).
|
Core Indicators for HIV Clinical Care |
|
Proportion of people newly diagnosed with HIV with a CD4+ cell count >200 cells/mm3 and without a clinical diagnosis of AIDS |
|
Rationale: Improve health outcomes by reducing the number of people living with HIV/AIDS (PLWHA) with late diagnosis. |
|
Proportion of people newly diagnosed with HIV who are linked to clinical care for HIV within 3 months of diagnosis |
|
Rationale: Timely linkage to care improves individual health outcomes and reduces transmission of the virus to others. |
|
Proportion of people with diagnosed HIV infection who are in continuous care (two or more visits for routine HIV medical care in the preceding 12 months at least 3 months apart) |
|
Rationale: Continuous HIV care results in better outcomes, including decreased mortality, and reduced transmission of the virus to others. |
|
Proportion of people with diagnosed HIV infection who received two or more CD4 tests in the preceding 12 months |
|
Rationale: Regular CD4 testing permits providers to monitor individuals’ immune function, determine when to start antiretroviral therapy (ART), and assess the need for prophylaxis for opportunistic infections. |
|
Proportion of people with diagnosed HIV infection who received two or more viral load tests in the preceding 12 months |
|
Rationale: Regular viral load (plasma HIV RNA) testing is important for monitoring clinical progression of the disease and therapeutic response in individuals on ART. |
|
Proportion of people with diagnosed HIV infection in continuous care for 12 or more months and with a CD4+ cell count ≥350 cells/mm3 |
|
Rationale: Achieving and maintaining a CD4+ cell count ≥350 cells/mm3 reduces the risk of complicating opportunistic infections and cancers. |
|
Proportion of people with diagnosed HIV infection and a measured CD4+ cell count <500 cells/mm3 who are not on ART* |
|
Rationale: Appropriate initiation of ART improves individual health outcomes and reduces transmission of the virus to others. |
|
Proportion of people with diagnosed HIV infection who have been on ART for 12 or more months and have a viral load below the level of detection |
|
Rationale: The goal of ART is durable virologic suppression, which improves health outcomes and reduces transmission of the virus. |
|
All-cause mortality rate among people diagnosed with HIV infection* |
|
Rationale: Mortality rate is the ultimate outcome measure for people diagnosed with HIV infection. Mortality among PLWHA should be inversely related to the quality of overall care delivered. |
|
Core Indicators for Mental Health, Substance Abuse, and Supportive Services |
|
Proportion of people with diagnosed HIV infection and mental health disorder who are referred for mental health services and receive these services within 60 days** |
|
Rationale: Untreated mental health disorders can negatively affect maintenance in care, adherence to treatment, and health outcomes for PLWHA and may increase the risk of transmitting the virus to others. |
|
Proportion of people with diagnosed HIV infection and substance use disorder who are referred for substance abuse services and receive these services within 60 days** |
|
Rationale: Untreated substance use disorders can negatively affect maintenance in care, adherence to treatment, and health outcomes for PLWHA and may increase the risk of transmitting the virus to others. |
|
Proportion of people with diagnosed HIV infection who were homeless or temporarily or unstably housed at least once in the preceding 12 months* |
|
Rationale: Homelessness and housing instability negatively affect maintenance in care, adherence to treatment, and health outcomes for PLWHA and may increase the risk of transmitting the virus to others. |
|
Proportion of people with diagnosed HIV infection who experienced food or nutrition insecurity at least once in the preceding 12 months* |
|
Rationale: Food insecurity affects maintenance in care, adherence to treatment, and health outcomes for PLWHA and may increase the risk of transmitting the virus to others. Poor nutrition affects absorption of medications and can contribute to diet-sensitive comorbidities. |
|
Proportion of people with diagnosed HIV infection who had an unmet need for transportation services to facilitate access to medical care and related services at least once in the preceding 12 months* |
|
Rationale: Unmet need for transportation to access HIV health care and related services negatively affects treatment access, service utilization, and health outcomes for PLWHA and may increase the risk of transmitting the virus to others. |
|
|
*In contrast to the other indicators, the estimates for these indicators should decrease with improved access to care and supportive services.
**Receipt of care within 30 days would reflect optimal care, but 60 days is more realistic given the current limited capacity of many providers to see new patients within a shorter time frame. Urgent cases should be seen as soon as possible.
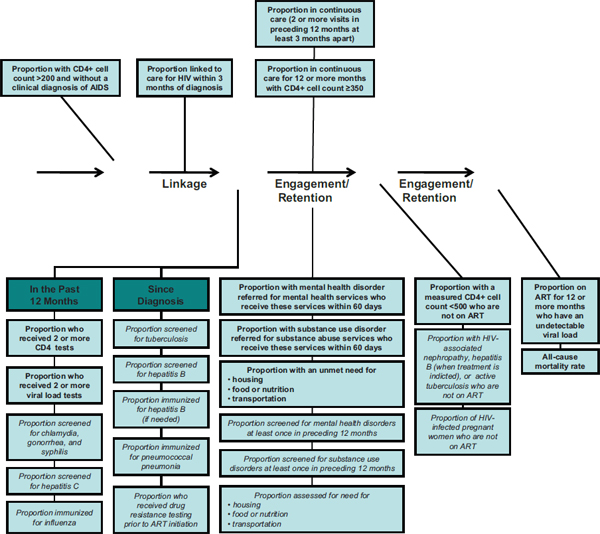
FIGURE 1-6 Continuum of HIV care arrow mapped to indicators for HIV care and supportive services.
NOTE: Indicators written in bold text correspond to the committee’s recommended core indicators; the indicators written in italics correspond to the additional indicators identified by the committee
SOURCE: Adapted from Das, 2011.
efficiency and quality of care and reduce health disparities; (5) permitting assessment of the impact of the NHAS and the ACA on health care coverage, utilization, and quality for people with HIV, facilitating identification of any difficulties encountered, and informing future planning; and (6) providing a real-time window into national health policy.
HIV is an expensive, chronic infectious disease, for which treatment of infected individuals has important public health consequences. Increased survival resulting from more effective treatments has led to a greater number of people with HIV in the United States. In addition, although the number of new HIV infections occurring annually has leveled off in recent years, an estimated 50,000 people in the United States were newly infected with HIV each year from 2006 through 2009 (Prejean et al., 2011). In addition to improving health outcomes for people with HIV who are treated, the movement toward universal treatment of HIV-infected individuals with ART may help to reduce the incidence of new infections. Reductions in the overall morbidity and mortality associated with new HIV infections in conjunction with decreases in HIV incidence could reduce HIV-related health care costs over time, although this benefit will not be realized for some time, given the lifespan of people being effectively treated for HIV. Each new HIV infection detected generates a responsibility to treat and monitor an additional patient, at an average cost of $19,912 per year in 2006 (Gebo et al., 2010) or $23,000 in 2010 dollars (CDC, 2012b). Based on a lifetime cost estimate discounted to the time of infection, the potential savings in HIV-related health care costs per HIV infection prevented is $303,100 in 2004 dollars for an adult initiating ART at a CD4+ cell count <350/mm3 (Schackman et al., 2006) or $379,668 in 2010 dollars (CDC, 2012b).
Earlier diagnosis and improved linkage to HIV care may help to improve health outcomes (ART-CC, 2008; Iwuji et al., 2011; May et al., 2011; Mocroft et al., 1999; Moore and Chaisson, 1999; Nakagawa et al., 2012; Palella et al., 1999), reduce new transmissions, and reduce the additional costs associated with late entry into care.6 In addition to late diagnosis and linkage to care, poor retention in care, delayed initiation of ART, and poor adherence to ART (i.e., discontinuing or intermittent ART) contribute to suboptimal outcomes for
_________________
6Studies have shown that HIV treatment costs are significantly higher for individuals who enter treatment with lower CD4+ cell counts (e.g., <350/mm3) than for those who enter treatment earlier in the course of their disease (e.g., CD4+ cell counts >500/mm3) (Fleishman et al., 2010; Krentz and Gill, 2012). Notably the cost differential remains significant even among those remaining in care for 7 to 8 years, despite improved CD4 counts (Fleishman et al., 2010; Krentz and Gill, 2012).
people with HIV. The fact that only 19 to 25 percent of people with HIV in the United States have an undetectable viral load in an era of effective treatment (CDC, 2012d; Gardner et al., 2011) is alarming. Monitoring health care coverage, utilization, and quality for people with HIV would provide data on the numbers and demographic characteristics of people who never enter care, are lost to care at various points along the care continuum, or receive suboptimal care. It also would permit the assessment of progress not only in improving health outcomes for all people with HIV, but also in reducing HIV-related disparities. These data could help inform potential redistribution of resources and identify progress and challenges associated with implementation of the ACA.
Although implementation of the ACA is expected to improve access to health care coverage and services and reduce health disparities among people with HIV, at least in states that choose to implement the Medicaid expansion provision, these outcomes are not guaranteed. For example, access may be reduced if new sources of health coverage limit the number of medications covered per month or reimbursement for substance abuse, mental health, and supportive services that were previously covered under the Ryan White HIV/AIDS Program. In particular, it will be important to assess how the care people receive under new public and private insurance and other sources of coverage compares to care they previously received under the Ryan White HIV/AIDS Program and other discretionary and entitlement programs. In this vein, monitoring the service utilization and quality of care for people with HIV in the wake of the ACA not only will permit assessment of the impact of the ACA on the amount and quality of care received by people with HIV, but also will facilitate identification of difficulties encountered during ACA implementation and inform planning, including future funding, workforce, or service needs.
In order to take maximum advantage from any strategy for monitoring health care coverage and utilization for people with HIV, it is important to ensure that the monitoring process is living and dynamic rather than static, allowing it to be modified as needed to collect the most useful and relevant data. Development of a strategy for monitoring HIV care coverage and utilization also may provide a window into national health care policy, such as interstate disparities. Furthermore, the system for monitoring health care coverage and utilization for HIV might facilitate the development of strategies for monitoring other chronic conditions, such as diabetes and heart disease.
Use of Nationally Representative Samples for Monitoring Health Care Coverage and Utilization Among People with HIV
In monitoring health care coverage and utilization for people with HIV in the United States, the use of nationally representative sampling is important to ensure that all subgroups within the U.S. HIV-infected population are included in the results. As discussed previously, HIV disproportionately
affects “vulnerable populations,”7 which contributes to the health disparities experienced by many people with HIV. In addition, a majority of people with HIV in the United States also must confront actual or perceived stigma and discrimination based on their sexual orientation or practices, further contributing to health disparities within this population.
The NHAS targets improvements in the care and health outcomes of individuals at the greatest risk for poor outcomes and unmet basic and health care needs, and the ACA is expected to significantly improve access to health care coverage and services for low-income people with HIV. Consequently, a monitoring system requires proportionate attention to groups that often are the most difficult to assess, including individuals with mental and substance use disorders, homeless individuals, those living in rural areas, and undocumented immigrants, which makes the task of developing a “representative national sample” particularly challenging.
As discussed in the committee’s first report, data relevant for monitoring progress toward meeting the goals of the NHAS and ACA currently are being collected by a number of public and private data systems, some specific to HIV and others not, each of which has limitations. The committee identified 12 data systems in particular that can serve as a collective platform for evaluating the use of continuous and high-quality care in all HIV-infected populations in the United States (Box 1-3; IOM, 2012, p. 167). As discussed in detail below, two of these data collection efforts, the National HIV Surveillance System (NHSS) and the Medical Monitoring Project (MMP), both under the auspices of the CDC, are either relatively comprehensive or designed to be nationally representative of people in the United States who have been diagnosed with HIV. Other data collection efforts, such as NA-ACCORD, CNICS, and HIVRN, draw from convenience samples. Although convenience samples may be demographically diverse, they are not necessarily nationally representative of the population of interest (Shapiro et al., 1999). NA-ACCORD, CNICS, and HIVRN, for example, draw data primarily from cohorts or sites in urban areas. Therefore, the data may not reflect the experiences of people with HIV in rural areas (IOM, 2012, Chapter 3). Data from health plans (e.g., Kaiser Permanente, Veterans Health Administration) or other sources of health care coverage (e.g., Medicaid, Medicare, Ryan White HIV/AIDS Program), including private insurers, also are not nationally representative, including data only from enrollees in their plans or programs. Data
_________________
7“Vulnerable populations are groups that are not well integrated into the health care system because of ethnic, cultural, economic, geographic, or health characteristics…. Commonly cited examples of vulnerable populations include racial and ethnic minorities, the rural and urban poor, undocumented immigrants, and people with disabilities or multiple chronic conditions” (Urban Institute, 2012).
BOX 1-3
Data Collection Activities for Monitoring HIV Care
HIV Care-Specific Data Systems
Public
- National HIV Surveillance System
- Medical Monitoring Project
- Ryan White Services Report
- Ryan White AIDS Drug Assistance Program Reports
Private
- North American AIDS Cohort Collaboration on Research and Design
- CFAR Network of Integrated Clinical Systems
- HIV Research Network
Data Systems with Information That Includes People with HIV
Public
- Medicaid Statistical Information System
- Medicare Chronic Condition Data Warehouse
- Clinical Case Registry: HIV
- National Vital Statistics Information System
Private
- Kaiser Permanente
from these sources, although useful in many ways, have other limitations as well (IOM, 2012, Chapter 3; Shapiro et al., 1999). National health-related surveys (e.g., NHIS, NHANES), which draw data from multistage probability-based samples of households, generate nationally representative data, but are neither apt to capture a statistically significant number of people with nor designed to capture detailed information about any given disease (Shapiro et al., 1999).
Examples of Broad-Based Data Collection for Monitoring Health Care Coverage and Utilization Among People with HIV
Population-based surveillance of the HIV epidemic in the United States began in 1981 when the first cases of opportunistic illnesses caused by what later would be identified as HIV were reported. Early in the epidemic, CDC, as well as state and local bodies, used AIDS case reporting to track the epidemic, rather than reports of newly diagnosed persons. The advent of
highly active antiretroviral therapies in the mid-1990s led to an increase in the time from HIV infection to AIDS diagnosis and thereby rendered AIDS case reporting insufficient to track the epidemic overall and to provide adequate information about prevention and care service needs (Fleming et al., 1999). In 1985, states began to institute HIV case reporting in addition to AIDS case reporting. In 1994, CDC integrated its AIDS and HIV reporting systems into a single, unified system, the NHSS (CDC, 2012e).8
Although surveillance data are vitally important for monitoring many aspects of the HIV epidemic, they traditionally have been relatively limited in scope. The NHSS, as a comprehensive national surveillance system, contains data on virtually all people diagnosed with HIV in the United States. However, surveillance data are not designed to describe HIV care and utilization in the United States as would be captured by the core indicators recommended by the committee in its first report (IOM, 2012, pp. 89-90, 110-118; see Table 1-4). Other large-scale HIV data collection efforts, such as the AIDS Cost and Services Utilization Survey (ACSUS), HIV Cost and Services Utilization Study, and MMP, have attempted to collect a broader range of data about health care coverage and utilization as well as supportive service needs, use, and experiences among people with HIV.
National HIV Surveillance System
The NHSS is a population-based census of all persons in the United States diagnosed with HIV infection and reported to CDC, including individuals receiving HIV care and those who are not in care, approximately 942,000 individuals (CDC, 2011d).9 CDC funds and assists state, territorial, and local health departments to collect HIV surveillance data and report them to CDC using Adult and Pediatric HIV Confidential Case Report forms. The state, territorial, and local HIV surveillance systems represent valuable additional sources of data pertinent to HIV care utilization.10 By
_________________
8Since 2008, CDC also conducts HIV Incidence Surveillance in 25 jurisdictions disproportionately affected by the disease and, since 2004, Variant, Atypical, and Resistance HIV Surveillance, to collect HIV genetic sequence data, in 11 jurisdictions (CDC, 2012c). Another CDC initiative, National HIV Behavioral Surveillance, collects data on “behavioral risks for HIV, HIV testing behaviors, access to and use of prevention services, and HIV testing results” in 20 AIDS-prevalent jurisdictions (CDC, 2012c).
9The national surveillance system is meeting its completeness standard of ≥85 percent for all diagnosed cases being reported to the system (CDC response to IOM request for information, April 4, 2011).
10These HIV surveillance systems may include additional data elements not captured on the CDC case report forms, as well as data from code-based reports initiated prior to name-based reporting and anonymous results that have not been name ascertained and hence are not included in the NHSS. The proportion of these uncounted cases can be calculated precisely by the reporting areas that have made the transition to name-based reporting.
April 2008, all 50 states and the District of Columbia, as well as American Samoa, Guam, Puerto Rico, the U.S. Virgin Islands, the Northern Mariana Islands, and the Republic of Palau—one of three freely associated states that report HIV surveillance data to CDC11—had implemented confidential, name-based HIV reporting (CDC, 2011c). Although the data for all reporting areas are included in CDC’s annual HIV Surveillance Report, 2012 marks the first year that all states have sufficiently mature reporting systems to permit CDC to statistically adjust the data for reporting delays and missing information (CDC, 2012e).12
In terms of data elements of interest for tracking the impact of the ACA, the NHSS includes date of HIV/AIDS diagnosis, information on CD4+ T-cell count and plasma HIV RNA (viral load) closest to diagnosis, and optional fields for HIV and substance abuse treatment referral, pregnancy status, and ART status at the time of reporting. Demographic data captured in the NHSS can be used to monitor health disparities among people with HIV with regard to race, ethnicity, sex, gender, age, geographic area, and country of birth.13 Most jurisdictions also report all CD4 count and viral load results,14 which permits the tracking of individuals’ health status over time.
AIDS Cost and Services Utilization Survey
Although not designed to be nationally representative of people with HIV, ACSUS (1991-1992) was the first large-scale and broadly representative effort, beyond surveillance, to collect data on HIV-infected individuals. Sponsored by the Agency for Health Care Policy and Research, now the Agency for Healthcare Research and Quality (AHRQ), ACSUS focused on people receiving HIV care and provided longitudinal data on health care services use, expenditures, and sources of payment for care of adults and children at various stages of HIV infection, and the ways in which those
_________________
11The Federated States of Micronesia and the Republic of the Marshall Islands are the other two freely associated states.
12The HIV Surveillance Report for 2012, to be issued in 2014, will be the first to include aggregate data from all 50 states (CDC, 2010).
13The full set of data elements captured in the NHSS can be obtained from the Adult and Pediatric Confidential HIV Case Report forms (available at http://www.odh.ohio.gov/pdf/IDCM/frm5042a.pdf; http://www.odh.ohio.gov/pdf/IDCM/frm5042b.pdf [accessed June 13, 2012]).
14As of June 15, 2010, 33 of 59 reporting areas (50 states, District of Columbia, 5 U.S. dependent areas, and 3 freely associated states) were reporting all CD4 and viral load test result, including 30 states, District of Columbia, Guam, and Puerto Rico (Personal communication, Amy Lansky, Centers for Disease Control and Prevention, October 6, 2011). More states are moving toward reporting all CD4 and viral load test results. Massachusetts, for example, mandated that all CD4 and all HIV viral load results be electronically reported by clinical and commercial laboratories as of January 2012 (Massachusetts Department of Public Health, 2012).
variables changed over the course of the illness (Berk et al., 1993; Fleishman et al., 1994).
The study collected data on 2,090 HIV-infected adults and children receiving clinical HIV care from spring 1991 through fall 1992. Participants (or their proxies) were interviewed in person quarterly for a total of six interviews (Berk et al., 1993; Hsia et al., 1995). Interview data were supplemented by medical, service utilization, billing, and other data obtained from the providers of inpatient, ambulatory, and home health care, as well as pharmacy data and Medicaid, and Health Care Financing Administration claims records (Berk et al., 1993). Data also were collected from nonmedical providers, including community-based organizations that offer supportive services, podiatrists, and alternative therapy providers.15
Interview data were gathered on participants’ age, sex, race and ethnicity, educational level, employment history, income sources, insurance type, stage of illness, route of exposure, and clinical trials participation. Information also was collected on participant’s living arrangements and social support network; experience of barriers to the receipt of services; functional status; and psychological affect and experience of pain (Berk et al., 1993; Hsia et al., 1995; Niemcryk et al., 1998). During each interview, participants were asked to give a full accounting of all providers from whom they had received services since the previous interview. With permission, those providers were contacted to obtain clinical and laboratory data, as well as data on service utilization, charges, and source of payment (Berk et al., 1993). Such data were collected on hospital admissions, emergency department use, outpatient services, drug and alcohol treatment, dental treatment, and counseling or support group services (Niemcryk et al., 1998), in addition to data on pharmacy and home health service utilization (Berk et al., 1993).
HIV Cost and Services Utilization Study
HCSUS (October 1994-September 2000) was the first major research effort to collect information on a nationally representative sample of adults receiving care for their HIV infection (AHRQ, 1998; RAND, 2011). Designed to collect and analyze data on medical and nonmedical cost and service utilization for adults with HIV, HCSUS was a cooperative effort conducted by a public-private consortium based out of the RAND Corporation and funded by a number of public agencies and private entities, through an agreement between AHRQ and RAND (RAND, 2011).
HCSUS was a prospective observational study that collected data
_________________
15Providers of dental services were not contacted directly due to concern about exposing patients’ HIV status.
through three serial interviews with a national probability sample of HIV-infected adults receiving regular medical care in the first 2 months of 1996 and interviews with providers and caregivers. Additional data were collected through health, dental, pharmacy, and billing record abstraction (Bozzette et al., 1998; Frankel et al., 1999; Shapiro et al., 1999). Participants were identified using a three-stage probability design with population-proportion-to-size sampling in which cases were randomly selected based on geographic location, provider, and patient (RAND, 2011). Of 4,042 eligible participants, 76 percent were interviewed (Andersen et al., 2000), yielding data on approximately 3,000 HIV-infected persons receiving care in hospitals, clinics, HMOs, or private practices who were living in urban areas or clusters of rural counties in the contiguous United States. The study population included racial and ethnic minorities, adult males and females with varying levels of education, routes of HIV infections, and health care service coverage (see Shapiro et al., 1999).
HCSUS consisted of a core study that explored the “cost, use, and quality of care; access to and unmet needs for care; quality of life; social support; knowledge of HIV; clinical outcomes; mental health; and the relationship of these variables to provider type and patient characteristics” and seven supplemental studies (RAND, 2011). In addition to interviews, participants’ medical, financial, and pharmaceutical records were abstracted, and a subset of participants had blood drawn to measure CD4+ T-cell count, viral load, and the presence of genotypic and phenotypic sequences associated with antiviral resistance (RAND, 2011).
Medical Monitoring Project
Initiated in 2005 in response to the IOM report Measuring What Matters: Allocation, Planning, and Quality Assessment for the Ryan White CARE Act (IOM, 2004), MMP is a CDC-sponsored population-based surveillance system currently designed to collect comprehensive clinical and behavioral services need, utilization, and outcomes data on a nationally representative sample of adults (≥18 years of age) living with HIV/AIDS who are receiving medical care from outpatient facilities in the United States and Puerto Rico (Blair et al., 2011). Approximately 480,000 of the 942,000 diagnosed HIV-infected individuals in the United States are retained in clinical HIV care (CDC, 2011d). It is this population from which MMP draws. MMP is the first project since HCSUS that is designed to obtain comprehensive information about HIV care from a nationally representative population of people with HIV who are in care. As discussed in greater detail in Chapter 3, MMP employs a probability proportional to size sampling design to obtain cross-sectional probability samples of its target population. Data are obtained from individual patient interviews and medical record
reviews. MMP also includes a minimum data set of core surveillance data from NHSS for all individuals sampled.
Limitations of Nationally Representative Studies
Although nationally representative studies hold promise for providing an overarching picture of health care coverage and utilization among people with HIV in the United States, they are generally not useful for generating comparisons among subgroups where stratification results in subgroups too small for meaningful analysis. For example, nationally representative studies such as MMP may not be sufficiently large to permit state-by-state analysis and comparisons of health care coverage and utilization.16 Yet, monitoring health care coverage and experiences at the state level is important because of state variations in implementation of the ACA (discussed in Chapter 2), Medicaid eligibility and benefits, and health care coverage requirements and available options, as well as state and regional disparities in access to health care by people with HIV. Similarly, it will be important to monitor changes in enrollment and in some cases benefits that are expected to occur in various sources of health coverage as the ACA is implemented.
All studies designed to be nationally representative face the difficulty inherent in including marginalized, hard-to-reach populations (e.g., individuals with mental and substance abuse disorders, those in correctional facilities, the homeless). The funding and labor necessary to ensure that all populations are sufficiently represented can be high. Adequate funding and careful attention to sampling and recruitment methods are necessary to ensure the collection of sufficient data for all subpopulations.
Agwu, A. L., J. A. Fleishman, P. T. Korthuis, G. K. Siberry, J. M. Ellen, A. H. Gaur, R. Rutstein, and K. A. Gebo. 2011. Disparities in antiretroviral treatment: A comparison of behaviorally-HIV-infected youth and adults in the HIV Research Network. Journal of Acquired Immune Deficiency Syndromes 58(1):100-107.
AHRQ (Agency for Healthcare Research and Quality). 1998. HCSUS Fact Sheet. HIV Cost and Services Utilization Study. http://www.ahrq.gov/data/hcsus.htm (accessed March 26, 2012).
Andersen, R., S. Bozzette, M. Shapiro, P. St Clair, S. Morton, S. Crystal, D. Goldman, N. Wenger, A. Gifford, A. Leibowitz, S. Asch, S. Berry, T. Nakazono, K. Heslin, W. Cunningham, and the HCSUS Consortium. 2000. Access of vulnerable groups to antiretroviral therapy among persons in care for HIV disease in the United States. Health Services Research 35(2):389-416.
_________________
16In addition, MMP data are limited to 17 states.
ART-CC (Antiretroviral Therapy Cohort Collaboration). 2008. Life expectancy of individuals on combination antiretroviral therapy in high-income countries: A collaborative analysis of 14 cohort studies. Lancet 372(9635):293-299.
ART-CC. 2009. Mortality of HIV-infected patients starting potent antiretroviral therapy: Comparison with the general population in nine industrialized countries. International Journal of Epidemiology 38(6):1624-1633.
Aziz, M., and K. Y. Smith. 2011. Challenges and successes in linking HIV-infected women to care in the United States. Clinical Infectious Diseases 52(Suppl 2):S231-S237.
Baicker, K., A. Chandra, J. S. Skinner, and J. E. Wennberg. 2004. Who you are and where you live: How race and geography affect the treatment of Medicare beneficiaries. Health Affairs (Suppl Variation, October):VAR33-44, doi: 10.1377/hlthaff.var.33.
Berk, M. L., C. Maffeo, and C. L. Schur. 1993. Research Design and Analysis Objectives. AIDS Cost and Services Utilization Survey (ACSUS) Reports, No. 1. AHCPR Publication No. 93-0019. Rockville, MD: Agency for Health Care Policy and Research.
Blair, J. M., A. D. McNaghten, E. L. Frazier, J. Skarbinski, P. Huang, and J. D. Heffelfinger. 2011. Clinical and behavioral characteristics of adults receiving medical care for HIV infection—Medical Monitoring Project, United States, 2007. Morbidity and Mortality Weekly Report Surveillance Summaries 60(11):1-20.
Bozzette, S. A., S. H. Berry, N. Duan, M. R. Frankel, A. A. Leibowitz, D. Lefkowitz, C. A. Emmons, J. W. Senterfitt, M. L. Berk, S. C. Morton, and M. F. Shapiro. 1998. The care of HIV-infected adults in the United States. HIV Cost and Services Utilization Study Consortium. New England Journal of Medicine 339(26):1897-1904.
Branson, B. M., H. H. Handsfield, M. A. Lampe, R. S. Janssen, A. W. Taylor, S. B. Lyss, and J. E. Clark. 2006. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. Morbidity and Mortality Weekly Report 55(RR14):1-17.
Burns, D. N., C. W. Dieffenbach, and S. H. Vermund. 2010. Rethinking prevention of HIV Type 1 infection. Clinical Infectious Diseases 51(6):725-731.
Castilla, J., P. Sobrino, L. De La Fuente, I. Noguer, L. Guerra, and F. Parras. 2002. Late diagnosis of HIV infection in the era of highly active antiretroviral therapy: Consequences for AIDS incidence. AIDS 16(14):1945-1951.
CDC (Centers for Disease Control and Prevention). 2010. Summary of Changes to the National HIV Surveillance Report. National Center for HIV/AIDS, Hepatitis, STD, and TB Prevention. http://www.cdc.gov/hiv/topics/surveillance/resources/factsheets/pdf/summary_changes.pdf (accessed October 20, 2011).
CDC. 2011a. HIV among African Americans. November. http://www.cdc.gov/hiv/topics/aa/PDF/aa.pdf (accessed July 3, 2012).
CDC. 2011b. HIV among Latinos. November. http://www.cdc.gov/hiv/resources/factsheets/pdf/latino.pdf (accessed July 3, 2012).
CDC. 2011c. HIV Infection Reporting. http://www.cdc.gov/hiv/topics/surveillance/reporting.htm (accessed July 2, 2012).
CDC. 2011d. Vital signs: HIV prevention through care and treatment—United States. Morbidity and Mortality Weekly Report 60(47):1618-1623.
CDC. 2012a. Diagnoses of HIV infection and AIDS in the United States and dependent areas, 2010. HIV Surveillance Report. Volume 22. March. http://www.cdc.gov/hiv/surveillance/resources/reports/2010report/index.htm (accessed July 3, 2012).
CDC. 2012b. HIV Cost-Effectiveness. http://www.cdc.gov/hiv/topics/preventionprograms/ce/index.htm (accessed May 10, 2012).
CDC. 2012c. HIV in the United States: At a Glance. http://www.cdc.gov/hiv/resources/factsheets/PDF/HIV_at_a_glance.pdf (accessed July 3, 2012).
CDC. 2012d. HIV in the United States: The Stages of Care. http://www.cdc.gov/nchhstp/newsroom/docs/2012/Stages-of-CareFactSheet-508.pdf (accessed September 10, 2012).
CDC. 2012e. HIV Surveillance Supported by the Division of HIV/AIDS Prevention. http://www.cdc.gov/hiv/topics/surveillance/resources/factsheets/pdf/HIV_surveillance.pdf (accessed June 11, 2012).
CDC. 2012f. Monitoring selected national HIV prevention and care objectives by using HIV surveillance data—United States and 6 U.S. dependent areas—2010. HIV Surveillance Supplemental Report. Volume 17, Number 3 (Part A). http://www.cdc.gov/hiv/surveillance/resources/reports/2010supp_vol17no3/index.htm (accessed July 31, 2012).
Charlebois, E. D., M. Das, T. C. Porco, and D. V. Havlir. 2011. The effect of expanded antiretroviral treatment strategies on the HIV epidemic among men who have sex with men in San Francisco. Clinical Infectious Diseases 52(8):1046-1049.
Cheever, L. W. 2007. Engaging HIV-infected patients in care: Their lives depend on it. Clinical Infectious Diseases 44(11):1500-1502.
Chen, N., E. Erbelding, H. C. Yeh, and K. Page. 2010. Predictors of HIV testing among Latinos in Baltimore City. Journal of Immigrant and Minority Health 12(6):867-874.
Cohen, M. S., Y. Q. Chen, M. McCauley, T. Gamble, M. C. Hosseinipour, N. Kumarasamy, J. G. Hakim, J. Kumwenda, B. Grinsztein, J. H. S. Pilotto, S. V. Godbole, S. Mehendale, S. Charjvalertsak, B. R. Santos, K. H. Mayer, I. F. Hoffman, S. H. Eshelman, E. PiwowarManning, L. Wang, J. Makhema, L. A. Mills, G. de Bruyn, I. Sanne, J. Eron, J. Gallant, D. Havlir, S. Swindells, H. Ribaudo, V. Elharrar, D. Burns, T. E. Taha, K. Nielsen-Saines, D. Celentano, M. Essex, and T. R. Fleming. 2011. Prevention of HIV-1 infection with early antiretroviral therapy. New England Journal of Medicine 365(6):493-505.
Conviser, R., and M. B. Pounds. 2002a. Background for the studies on ancillary services and primary care use. AIDS Care 14(Suppl 1):S7-S14.
Conviser, R., and M. B. Pounds. 2002b. The role of ancillary services in client-centred systems of care. AIDS Care 14(Suppl 1):S119-S131.
Cooper-Patrick, L., J. J. Gallo, J. J. Gonzales, H. T. Vu, N. R. Powe, C. Nelson, and D. E. Ford. 1999. Race, gender, and partnership in the patient-physician relationship. Journal of the American Medical Association 282(6):583-589.
Das, M. 2011. Intensifying HIV Prevention in the Communities Where HIV Is Most Heavily Concentrated. PowerPoint presentation at the 2011 National HIV Prevention Conference, Atlanta, GA, August 15.
Doescher, M. P., B. G. Saver, K. Fiscella, and P. Franks. 2001. Racial/ethnic inequities in continuity and site of care: Location, location, location. Health Services Research 36(6 Pt 2):78-89.
Ennis, S. R., M. Ríos-Vargas, and N. G. Albert. 2011. The Hispanic Population: 2010. Economics and Statistics Administration. http://www.census.gov/prod/cen2010/briefs/c2010br-04.pdf (accessed September 12, 2012).
Espinoza, L., H. I. Hall, R. M. Selik, and X. Hu. 2008. Characteristics of HIV infection among Hispanics, United States, 2003-2006. Journal of Acquired Immune Deficiency Syndromes 49(1):94-101.
FDA (U.S. Food and Drug Administration). 2012. FDA Approves First Over-the-Counter Home-Use Rapid HIV Test. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm310542.htm (accessed September 10, 2012).
Fleishman, J. A., D. C. Hsia, and F. J. Hellinger. 1994. Correlates of medical service utilization among people with HIV infection. Health Services Research 29(5):527-548.
Fleishman, J. A., B. R. Yehia, R. D. Moore, K. A. Gebo, and HIV Research Network. 2010. The economic burden of late entry into medical care for patients with HIV infection. Medical Care 48(12):1071-1079.
Fleming, P. L., J. W. Ward, R. S. Janssen, K. M. DeCock, R. O. Valdiserri, H. D. Gayle, in collaboration with J. L. Jones, J. S. Lehman, M. L. Lindegren, A. K. Nakashima, P. S. Sullivan, P. A. Sweeney, P. M. Wortley, E. M. Seiler, and H. W. Jaffe. 1999. Guidelines for National Human Immunodeficiency Virus Case Surveillance, including monitoring for human immunodeficiency virus infection and acquired immunodeficiency syndrome. Morbidity and Mortality Weekly Reports 48(RR13):1-28.
Frankel, M. R., M. F. Shapiro, N. Duan, S. C. Morton, S. H. Berry, J. A. Brown, M. A. Burnam, S. E. Cohn, D. P. Goldman, D. F. McCaffrey, S. M. Smith, P. A. St. Clair, J. F. Tebow, and S. A. Bozzette. 1999. National probability samples in studies of low-prevalence diseases. Part II: Designing and implementing the HIV Cost and Services Utilization Study sample. Health Services Research 34(5, Part 1):969-992.
Gardner, E. M., M. P. McLees, J. F. Steiner, C. Del Rio, and W. J. Burman. 2011. The spectrum of engagement in HIV care and its relevance to test-and-treat strategies for prevention of HIV infection. Clinical Infectious Diseases 52(6):793-800.
Garland, J. M., A. S. Andrade, and K. R. Page. 2010. Unique aspects of the care of HIV-positive Latino patients living in the United States. Current HIV/AIDS Reports 7(3):107-116.
Gaynes, B. N., A. J. Rush, M. H. Trivedi, S. R. Wisniewski, G. K. Balasubramani, D. C. Spencer, T. Petersen, M. Klinkman, D. Warden, L. Nicholas, and M. Fava. 2007. Major depression symptoms in primary care and psychiatric care settings: A cross-sectional analysis. Annals of Family Medicine 5(2):126-134.
Gebo, K. A., J. A. Fleishman, R. Conviser, E. D. Reilly, P. T. Korthuis, R. D. Moore, J. Hellinger, P. Keiser, H. R. Rubin, L. Crane, F. J. Hellinger, and W. C. Mathews. 2005. Racial and gender disparities in receipt of highly active antiretroviral therapy persist in a multistate sample of HIV patients in 2001. Journal of Acquired Immune Deficiency Syndromes 38(1):96-103.
Gebo, K. A., J. A. Fleishman, R. Conviser, J. Hellinger, F. J. Hellinger, J. S. Josephs, P. Keiser, P. Gaist, and R. D. Moore. 2010. Contemporary costs of HIV healthcare in the HAART era. AIDS 24(17):2705-2715.
Hall, H. I., R. Song, P. Rhodes, J. Prejean, Q. An, L. M. Lee, J. Karon, R. Brookmeyer, E. H. Kaplan, M. T. McKenna, and R. S. Janssen. 2008. Estimation of HIV incidence in the United States. Journal of the American Medical Association 300(5):520-529.
Hall, H. I., E. L. Frazier, P. Rhodes, D. R. Holtgrave, C. Furlow-Parmley, T. Tang, K. M. Gray, S. M. Cohen, and J. Skarbinski. 2012. Continuum of HIV care: Differences in Care and Treatment by Sex and Race/Ethnicity in the United States. Oral abstract presented by H. I. Hall, XIX International AIDS Conference, Washington, DC, July 27, 2102.
Heckman, T. G., A. M. Somlai, J. Peters, J. Walker, L. Otto-Salaj, C. A. Galdabini, and J. A. Kelly. 1998. Barriers to care among persons living with HIV/AIDS in urban and rural areas. AIDS Care 10(3):365-375.
HHS (U.S. Department of Health and Human Services). 2010. Healthy People 2020 Summary of Objectives. http://www.healthypeople.gov/2020/topicsobjectives2020/pdfs/HIV.pdf (accessed April 23, 2012).
HHS. 2011. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. October 14. http://aidsinfo.nih.gov/contentfiles/AdultandAdolescentGL002534.pdf (accessed July 2, 2012).
HHS. 2012a. How Does the Affordable Care Act Impact People Living with HIV/AIDS. http://aids.gov/federal-resources/policies/health-care-reform/ (accessed July 16, 2012).
HHS. 2012b. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. March 27. http://aidsinfo.nih.gov/contentfiles/lvguidelines/adultandadolescentgl.pdf (accessed July 2, 2012).
Hixson, L., B. B. Hepler, and M. Ouk Kim. 2011. The White Population: 2010. Economics and Statistics Administration. http://www.census.gov/prod/cen2010/briefs/c2010br-05.pdf (accessed September 12, 2012).
Hixson, L., B. B. Hepler, and M. Ouk Kim. 2012. The Native Hawaiian and Other Pacific Islander Population: 2010. Economics and Statistics Administration. http://www.census.gov/prod/cen2010/briefs/c2010br-12.pdf (accessed September 12, 2012).
Hoeffel, E. M., S. Rastogi, M. Ouk Kim, and H. Shahid. 2012. The Asian Population: 2010. Economics and Statistics Administration. http://www.census.gov/prod/cen2010/briefs/c2010br-11.pdf (accessed September 11, 2012).
Horberg, M. A., J. A. Aberg, L. W. Cheever, P. Renner, E. O’Brien Kaleba, and S. M. Asch. 2010. Development of national and multiagency HIV care quality measures. Clinical Infectious Diseases 51(6):732-738.
Hsia, D. C., J. A. Fleishman, J. A. East, and F. J. Hellinger. 1995. Pediatric human immunodeficiency virus infection: Recent evidence on the utilization and costs of health services. Archives of Pediatric and Adolescent Medicine 149:489-496.
IOM (Institute of Medicine). 2004. Measuring What Matters: Allocation, Planning and Quality Assessment for the Ryan White CARE Act. Washington, DC: The National Academies Press.
IOM. 2012. Monitoring HIV Care in the United States: Indicators and Data Systems. Washington, DC: The National Academies Press.
Iwuji, C. C., B. N. Mayanja, H. A. Weiss, E. Atuhumuza, P. Hughes, D. Maher, and H. Grosskurth. 2011. Morbidity in HIV-1-infected individuals before and after the introduction of antiretroviral therapy: A longitudinal study of a populations-based cohort in Uganda. HIV Medicine 12(9):553-561.
Johnson, R. L., D. Roter, N. R. Powe, and L. A. Cooper. 2004a. Patient race/ethnicity and quality of patient-physician communication during medical visits. American Journal of Public Health 94(12):2084-2090.
Johnson, R. L., S. Saha, J. J. Arbelaez, M. C. Beach, and L. A. Cooper. 2004b. Racial and ethnic differences in patient perceptions of bias and cultural competence in health care. Journal of General Internal Medicine 19(2):101-110.
Justice, A. C. 2006. Prioritizing primary care in HIV: Comorbidity, toxicity, and demography. Topics in HIV Medicine 14(5):159-163.
Kidder, D. P., R. J. Wolitski, S. Royal, A. Aidala, C. Courtenay-Quirk, D. R. Holtgrave, D. Harre, E. Sumartojo, and R. Stall. 2007. Access to housing as a structural intervention for homeless and unstably housed people living with HIV: Rationale, methods, and implementation of the housing and health study. AIDS and Behavior 11(2 Suppl):149-161.
Korthuis, P. T., S. Saha, J. A. Fleishman, M. M. McGrath, J. S. Josephs, R. D. Moore, K. A. Gebo, J. Hellinger, and M. C. Beach. 2008. Impact of patient race on patient experiences of access and communication in HIV care. Journal of General Internal Medicine 23(12):2046-2052.
Krawczyk, C. S., E. Funkhouser, J. M. Kilby, and S. H. Vermund. 2006a. Delayed access to HIV diagnosis and care: Special concerns for the southern United States. AIDS Care 18(Suppl 1):S35-S44.
Krawczyk, C. S., E. Funkhouser, J. M. Kilby, R. A. Kaslow, A. K. Bey, and S. H. Vermund. 2006b. Factors associated with delayed initiation of HIV medical care among infected persons attending a southern HIV/AIDS clinic. Southern Medical Journal 99(5):472-481.
Krentz, H. B., and M. J. Gill. 2012. The direct medical costs of late presentation (<350/mm3) of HIV infections over a 15-year period. AIDS Research and Treatment 2012:757135.
Leaver, C. A., G. Bargh, J. R. Dunn, and S. W. Hwang. 2007. The effects of housing status on health-related outcomes in people living with HIV: A systematic review of the literature. AIDS and Behavior 11(2 Suppl):85-100.
Lo, W., T. MacGovern, and J. Bradford. 2002. Association of ancillary services with primary care utilization and retention for patients with HIV/AIDS. AIDS Care 14(Suppl 1):S45-S57.
Massachusetts Department of Public Health. 2012. Letter to clinical laboratories. February 28.
May, M., M. Gompels, V. Delpech, K. Porter, F. Post, M. Johnson, D. Dunn, A. Palfreeman, R. Gilson, B. Gazzard, T. Hill, J. Walsh, M. Fisher, C. Orkin, J. Ainsworth, L. Bansi, A. Phillips, C. Leen, M. Nelson, J. Anderson, and C. Sabin. 2011. Impact of late diagnosis and treatment on life expectancy in people with HIV-1: UK Collaborative HIV Cohort (UK-CHIC) Study. British Medical Journal 343:d6016.
Mays, V. M., R. Maas, J. Ricks, and S. D. Cochran. 2011. HIV and African American women in the South: Employing a population-level HIV prevention and interventions. In Handbook of Health Psychology, Second Edition, edited by A. Baum, T. Revenson, and J. Singer. London: Psychology Press.
Meditz, A. L., S. MaWhinney, A. Allshouse, W. Feser, M. Markowitz, S. Little, R. Hecht, E. S. Daar, A. C. Collier, J. Margolick, J. M. Kilby, J. P. Routy, B. Conway, J. Kaldor, J. Levy, R. Schooley, D. A. Cooper, M. Altfeld, D. Richman, and E. Connick. 2011. Sex, race, and geographic region influence clinical outcomes following primary HIV-1 infection. Journal of Infectious Diseases 203(4):442-451.
Mocroft, A., C. A. Sabin, M. Youle, S. Madge, M. Tyrer, H. Devereux, J. Deayton, A. Dykhoff, M. C. I. Lipman, A. N. Phillips, and M. A. Johnson. 1999. Changes in AIDS-defining illnesses in a London clinic, 1987-1998. Journal of Acquired Immune Deficiency Syndromes 21(5):401-407.
Moon, T. D., S. H. Vermund, T. C. Tong, and S. D. Holmberg. 2001. Opportunities to improve preventions and services for HIV-infected women in nonurban Alabama and Mississippi. Journal of Acquired Immune Deficiency Syndromes 28(3):279-281.
Moore, R. D., and R. E. Chaisson. 1999. Natural history of HIV infection in the era of combined antiretroviral therapy. AIDS 13(14):1933-1942.
Mugavero, M. J., C. Castellano, D. Edelman, and C. Hicks. 2007. Late diagnosis of HIV infection: The role of age and sex. American Journal of Medicine 120(4):370-373.
Nakagawa, F., R. K. Lodwick, C. J, Smith, R. Smith, V. Cambiano, J. Lundgren, V. Delpech, and A. N. Phillips. 2012. Projected life expectancy of people with HIV according to time of diagnosis. AIDS 26(3):335-343.
Niemcryk, S. J., A. Bedros, K. M. Marconi, and J. F. O’Neill. 1998. Consistency in maintaining contact with HIV-related service providers: An analysis of the AIDS Cost and Services Utilization Study (ACSUS). Journal of Community Health 23(2):137-152.
Norris, T., P. L. Vines, and E. M. Hoeffel. 2012. American Indian and Alaskan Native population: 2010. Economics and Statistics Administration. http://www.census.gov/prod/cen2010/briefs/c2010br-10.pdf (accessed September 11, 2012).
NQF (National Quality Forum). 2011. NQF—Endorsed Standards. http://www.qualityforum.org/Measures_List.aspx?#k=HIV&p=2&s=n&so=a (accessed April 23, 2012).
NYC DOHMH (New York City Department of Health and Mental Hygiene). 2011. 2011 Advisory #27: Health Department Releases New HIV Treatment Recommendations. http://home2.nyc.gov/html/doh/downloads/pdf/cd/2011/11md27.pdf (accessed December 19, 2011).
Ohl, M., J. Tate, M. Duggal, M. Skanderson, M. Scotch, P. Kaboli, M. Vaughan-Sarrazin, and A. Justice. 2010. Rural residence is associated with delayed care entry and increased mortality among veterans with human immunodeficiency virus infection. Medical Care 48(12):1064-1070.
ONAP (Office of National AIDS Policy). 2010. National HIV/AIDS Strategy for the United States. http://www.whitehouse.gov/sites/default/files/uploads/NHAS.pdf (accessed January 6, 2012).
Palella, F. J., K. M. Delaney, A. C. Moorman, M. O. Loveless, J. Fuhrer, G. A. Satten, D. J. Aschman, A. D. Holmberg, and the HIV Outpatient Study Investigators. 1999. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. New England Journal of Medicine 338(13):853-860.
Pence, B. W. 2009. The impact of mental health and traumatic life experiences on antiretroviral treatment outcomes for people living with HIV/AIDS. Journal of Antimicrobial Chemotherapy 63(4):636-640.
PEPFAR (President’s Emergency Plan for AIDS Relief). 2009. Next Generation Indicators Reference Guide. http://www.pepfar.gov/documents/organization/81097.pdf (accessed April 23, 2012).
Prejean, J., R. Song, Q. An, and H. I. Hall. 2008. Subpopulation estimates from the HIV incidence surveillance system—United States, 2006. Morbidity and Mortality Weekly Report 57(36):985-989. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5736a1.htm (accessed December 20, 2011).
Prejean, J., R. Song, A. Hernandez, R. Ziebell, T. Green, F. Walker, L. S. Lin, Q. An, J. Mermin, A. Lansky, and H. I. Hall. 2011. Estimated HIV incidence in the United States, 2006-2009. PLoS One 6(8):e17502.
Qian, H. Z., R. D. Taylor, H. J. Fawal, and S. H. Vermund. 2006. Increasing AIDS case reports in the South: U.S. trends from 1981-2004. AIDS Care 18(Suppl 1):S6-S9.
RAND. 2011. HIV Cost and Services Utilization Study (HCSUS). http://www.rand.org/health/projects/hcsus.html (accessed March 26, 2012).
Rastogi, S., T. B. Johnson, E. M. Hoeffel, and M. P. Drewery. 2011. The Black Population: 2010. http://www.census.gov/prod/cen2010/briefs/c2010br-06.pdf (accessed September 11, 2012).
Reif, S., C. E. Golin, and S. R. Smith. 2005. Barriers to accessing HIV/AIDS care in North Carolina: Rural and urban differences. AIDS Care 17(5):558-565.
Reif, S., K. Whetten, E. Wilson, and W. Gong. 2011. HIV/AIDS Epidemic in the South Reaches Crisis Proportions in Last Decade. Durham, NC: Duke Center for Health Policy and Inequalities Research (CHPIR), Duke University. http://globalhealth.duke.edu/news/2011/ResearchReportFinal3-12.pdf (accessed May 8, 2012).
Robison, L. S., A. O. Westfall, M. J. Mugavero, M. C. Kempf, S. R. Cole, J. J. Allison, J. H. Willig, J. L. Raper, C. M. Wilcox, and M. S. Saag. 2008. Short-term discontinuation of HAART regimens more common in vulnerable patient populations. AIDS Research and Human Retroviruses 24(11):1347-1355.
Royal, S. W., D. P. Kidder, S. Patrabansh, R. J. Wolitski, D. R. Holtgrave, A. Aidala, S. Pals, and R. Stalle. 2009. Factors associated with adherence to highly active antiretroviral therapy in homeless or unstably housed adults living with HIV. AIDS Care 21(4):448-455.
Schackman, B. R., K. A. Gebo, R. P. Walensky, E. Losina, T. Muccio, P. E. Sax, M. C. Weinstein, G. R. Seage III, R. D. Moore, and K. A. Freedberg. 2006. The lifetime cost of current human immunodeficiency virus care in the United States. Medical Care 44(11):990-997.
Schiller, J. S., J. W. Lucas, B. W. Ward, and J. A. Peregoy. 2012. Summary health statistics for U.S. adults: National Health Interview Survey, 2010. National Center for Health Statistics. Vital Health Statistics 10(252).
Schneider, E. C., A. M. Zaslavsky, and A. M. Epstein. 2002. Racial disparities in the quality of care for enrollees in Medicare managed care. Journal of the American Medical Association 287(10):1288-1294.
Schur, C. L., M. L. Berk, J. R. Dunbar, M. F. Shapiro, S. E. Cohn, and S. A. Bozzette. 2002. Where to seek care: An examination of people in rural areas with HIV/AIDS. Journal of Rural Health 18(2):337-347.
Shapiro, M. F., M. L. Berk, S. H. Berry, C-A Emmons, L. A. Athey, D. C. Hsia, A. A. Leibowitcz, C. A. Maida, M. Marcus, J. F. Perlman, C. L. Schur, M. A. Schuster, W. Senterfitt, and S. A. Bozzette. 1999. National probability samples in studies of low-prevalence diseases. Part 1: Perspectives and lessons learned from the HIV Cost and Services Utilization Study. Health Services Research 34(5, Part 1):951-968.
Stall, R., T. C. Mills, J. Williamson, T. Hart, G. Greenwood, J. Paul, L. Pollack, D. Binson, D. Osmond, and J. A. Catania. 2003. Association of co-occurring psychosocial health problems and increased vulnerability to HIV/AIDS among urban men who have sex with men. American Journal of Public Health 93(6):939-942.
Urban Institute. Health Policy Center. 2012. Vulnerable Populations. http://www.urban.org/health_policy/vulnerable_populations/index.cfm (accessed May 11, 2012).
U.S. Census Bureau. 2012a. State by age, sex, race, and Hispanic origin: 6 race groups—5 race alone groups and one multiple race group. Economics and Statistics Administration. http://www.census.gov/popest/data/state/asrh/2011/files/SC-EST2011-alldata6-ALL.csv (accessed September 12, 2012).
U.S. Census Bureau. 2012b. State by age, sex, race, and Hispanic origin: 6 race groups—5 race alone groups and one multiple race group (file layout). Economic and Statistics Administration. http://www.census.gov/popest/data/state/asrh/2011/files/SC-EST2011ALLDATA6.pdf (accessed September 12, 2012).
Vermund, S. H., S. L. Hodder, J. E. Justman, B. A. Koblin, T. D. Mastro, K. H. Mayer, D. P. Wheeler, and W. M. El-Sadr. 2010. Addressing research priorities for prevention of HIV infection in the United States. Clinical Infectious Diseases 50(Suppl 3):S149-S155.
Weis, K. E., A. D. Liese, J. Hussey, J. J. Gibson, and W. A. Duffus. 2010. Associations of rural residence with timing of HIV diagnosis and stage of disease at diagnosis, South Carolina 2001-2005. Journal of Rural Health 26(2):105-112.
Weiser, S. D., D. R. Bangsberg, S. Kegeles, K. Ragland, M. B. Kushel, and E. A. Frongillo. 2009a. Food insecurity among homeless and marginally housed individuals living with HIV/AIDS in San Francisco. AIDS and Behavior 13(5):841-848.
Weiser, S. D., E. A. Frongillo, K. Ragland, R. S. Hogg, E. D. Riley, and D. R. Bangsberg. 2009b. Food insecurity is associated with incomplete HIV RNA suppression among homeless and marginally housed HIV-infected individuals in San Francisco. Journal of General Internal Medicine 24(1):14-20.
Wohl, A. R., W. H. Garland, J. Wu, C. W. Au, A. Boger, R. Dierst-Davies, J. Carter, F. Carpio, and W. Jordan. 2011. A youth-focused case management intervention to engage and retain young gay men of color in HIV care. AIDS Care 23(8):988-997.
Xu, F., M. R. Sternberg, and L. E. Markowitz. 2010. Men who have sex with men in the United States: Demographic and behavioral characteristics and prevalence of HIV and HSV-2 infection. Sexually Transmitted Diseases 37(6):399-405.