Highlight: The Fukushima Event—A Nuclear Detective Story
On March 11, 2011, a massive earthquake and tsunami struck Japan. The Fukushima Dai-ichi reactor complex suffered severe damage and experienced a series of explosions in subsequent days (Figure FUK 1). Radioactivity was released into the atmosphere and deposited on large areas of Japan (Figure FUK 2). In the United States there was concern that this radioactivity would cross the Pacific Ocean and present a health hazard in the United States as well. Physicists in the nuclear physics community working on neutrinoless double beta decay realized that the radioactivity might render the backgrounds too high for the success of their experiment. At the University of Washington an air-monitoring system was quickly set up to collect and measure airborne radioactivity attached to dust particles. Scientists at Berkeley prepared to monitor rainwater for fallout. Across the country, other university nuclear physics groups set up detectors and collection methods, and the universities became one of the main sources of detailed information about the radioactivity.
Radioactivity associated with the Fukushima reactors arrived at Seattle during the night of March 17 (PST). The strongest signal was from iodine-131, and gamma-ray lines from tellurium-132, cesium-134, and cesium-137 were seen as well. The 8-day iodine-131 and 3.2-day tellurium-132 provided clear evidence that the primary source of the radioactivity was a reactor that had recently been in operation, not spent fuel. However the 21-hour iodine-133 was not seen, giving assurance that the nuclear chain reaction had been successfully shut down at about the time of the earthquake (Figure FUK 3). The presence of cesium-134 is typical of reactor debris as distinct from nuclear weapons, because it is blocked by stable xenon-134 from being produced in fission and can only be produced by neutron capture. While superficially similar to the Chernobyl disaster, the Fukushima radioactivity inventory is very different, with only three isotopes present in abundance instead of the broad spectrum of fission products released at Chernobyl. One possible explanation for this is the release of contaminated steam instead of burning fuel elements, as happened at Chernobyl. In addition to gaining detailed insight into the unfolding disaster in Fukushima, the university groups were able to reassure the public that no dangerous levels of activity were reaching the United States.
The efforts at the University of Washington were only part of the efforts that nuclear scientists across the United States dedicated to helping assess and address the challenges in Japan as well as to communicate with the public. As an example, one of the NNSA’s radiological field monitoring team leaders was Daniel Blumenthal. The goal of his team was to provide timely information on environmental conditions so that fact-based protective action decisions could be made by decision makers that included the U.S. Armed Forces, the U.S. Embassy in Tokyo, and the Government of Japan. The members of this interdisciplinary team were prepared to perform “in extremis” science, where there is no time for lengthy deliberation or peer review yet accuracy during both collection and analysis is paramount. Dr. Blumenthal’s 33-member team was deployed to Japan via a dedicated U.S. military airlift on March 14 at the direction of the White House. They brought radiation detection systems to perform both aerial and ground-based radiological surveys. The aerial systems were mounted on Air Force aircraft and operated by the deployed scientists and technicians. The ground-based systems included equipment for basic exposure rate measurements; high-resolution in situ gamma-ray spectroscopy; and sampling of air and soil. In addition to the field team, an essential component of the response capability was a home team staffed by a similar array of nuclear scientists and other experts trained to analyze field data quickly and to communicate their results to leaders in the field and in Washington, D.C.
One of the biggest challenges during the Japan response was translating the measurements into actionable information. It was critical that the information be framed so that it made sense

FIGURE FUK 1 Aerial image of the Fukushima nuclear power plant complex after the initial explosions in March 2011. SOURCE: Copyright Air Photo Service Co. Ltd., Japan.
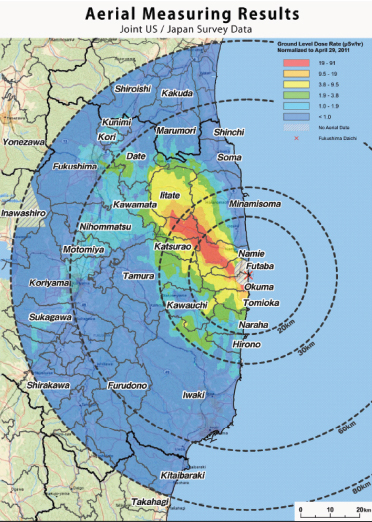
FIGURE FUK 2 Example of aerial measurements of the radiation plume in the region near the Fukushima nuclear power plants. SOURCE: Joint United States-Japan survey data provided by the U.S. Department of Energy, National Nuclear Security Administration (NNSA), Office of Emergency Response.
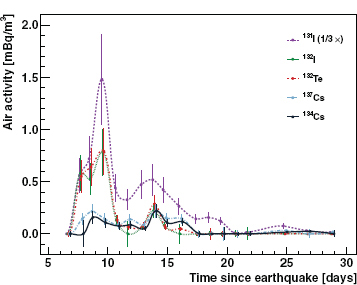
FIGURE FUK 3 Airborne activity observed in Seattle for radioactive isotopes of iodine, tellurium, and cesium in millibecquerels per cubic meter. As a reference, the average human body is exposed to 4,400 becquerels from the decay of potassium-40, a naturally occurring radioactive isotope of potassium. SOURCE: Reprinted from J. Diaz Leon, D.A. Jaffe, J. Kaspar, A. Knecht, M.L. Miller, R.G.H. Robertson, A.G. Schubert, 2011, Arrival time and magnitude of airborne fission products from the Fukushima, Japan, reactor incident as measured in Seattle, WA, USA, Journal of Environmental Radioactivity 102 (11): 032, Copyright 2011, with permission from Elsevier.
and that the decision makers would embrace the scientific process and have confidence in the information provided. In Japan, the success of the effort was endued by the fact that both civilian and military leaders requested that the technical experts remain in Japan as long as possible. Dr. Blumenthal communicated the results to a wide range of audiences, including to the general public on ABC News.1
1 Available at http://abcnews.go.com/International/video/tracking-japan-radiation-from-the-sky-13628048.




