2
The Structure and Function Of the Immune System And Mechanisms of Immunotoxicity
This chapter provides an overview of the structure and function of the immune system and the mechanisms of immunotoxicity.
Immunotoxicology is the study of injury to the immune system that can result from occupational, inadvertent, or therapeutic exposure to a variety of environmental chemicals or biologic materials. The field has two broad research areas: One involves studies of the suppression of immunity; the other concerns studies of enhanced or excessive immune response, such as allergy or autoimmunity. The immune system acts as a passive target for suppressive xenobiotics, and exposure to them can result in an increased incidence of infectious disease or neoplasia because of the host's inability to respond. When the immune system responds to other xenobiotics, a marked increase in a specific immune response can occur and adverse health consequences, such as respiratory tract allergies (e.g., asthma and rhinitis), allergic contact dermatitis, or autoimmune disease, can develop. Therefore, although other tissues can be affected after alterations in an immune response, the immune system is the primary site of action.
The field of immunotoxicology has grown considerably since the early 1970s (Luster et al., 1988). The discipline developed through studies that combined knowledge of immunology and toxicology to determine the effects of xenobiotics on the functioning of the immune system. The field now includes initial identification of suspected immunotoxic chemicals and the development of sensitive, quantitative animal assays to assess any chemically induced immunologic effects and determinations of the mechanisms by which xenobiotics can alter immune function. Various assays have been developed to characterize the immunotoxic properties of xenobiotics in animals (Dean et al., 1982). These immunotoxicologic assays measure effects on humoral immunity, cell-mediated immunity, macrophage function, natural-killer-cell cytotoxicity, and cytokine activity in animals. Recent developments in monoclonal antibody technology and the advances in cell-culture techniques and molecular biology allow immunotoxicologists to examine the molecular mechanisms of action of drugs and chemicals.
Certain chemical exposures and doses that do not affect particular organs can result in immune dysfunction. For example, lead, polychlorinated biphenyls, toxaphene, pentachlorophenol, and some organophosphate pesticides have been shown to affect significantly the immune response of mice and
rats at exposures that did not alter other commonly tested measures of toxicity, such as body weight and some blood chemistry measurements. The conclusion is that the immune system could be more sensitive to certain xenobiotics than other major organ systems are.
The immune system can be affected by a variety of conditions, substances, and agents. Those most commonly encountered are radiation, immunosuppressive drugs, bacteria, viruses, protozoa, other parasites, and some forms of cancer. There are a number of hereditary syndromes of immune deficiency. Occasionally, immune deficiencies result from other environmental causes. Stress, for example, from loud noise, electric shock, infant-mother separation, overcrowding, the death of a spouse, or severe emotional or mental dysfunction could be associated with immunologic abnormality. Hereditary and environmental factors must be evaluated carefully in the assessment of suspected chemical-induced immune dysfunction. The putative causes of immune modulation, particularly in humans, are diverse, and therefore, they must at least include emotional distress as a contributing factor.
DEVELOPMENT AND FUNCTION OF THE IMMUNE SYSTEM
The immune system is the body's main defense against foreign materials and biologic agents such as bacteria, viruses, chemicals, and foreign cells and tissues. The immune response includes specific action of lymphocytes (one type of white blood cell) and is facilitated by other white blood cells, including neutrophils, monocytes, macrophages, eosinophils, and basophils. The immune system can be viewed as a system controlled by negative feedback. The central component of the system is the lymphatic tissues, which include mature T (thymic) lymphocytes that have matured through development in the thymus and mature B lymphocytes that have matured in the bone marrow. Each of these cell groups is composed of subpopulations with varied functions, especially the T cells (Twomey, 1982; Golub, 1987; Mangan, 1987). Some T-cell populations act to enhance or suppress immune function; the B cells differentiate to varied immunoglobulin-secreting plasma cells after the immune system's exposure to the antigen (Paul, 1984; Miedema and Melief, 1985; Young and Geha, 1986).
Lymphocytes circulate throughout the body, moving in and out of tissues via the circulatory and lymphatic systems, where they meet foreign antigens. The immune response is called specific because each mature T or B lymphocyte has a specific antigen receptor on its surface. After an antigen is bound to the receptor, stimulation of a B cell produces antibodies (immunoglobulins) that specifically react with the stimulating antigen. The binding of antibodies to antigens can lead to the inactivation or removal of the foreign antigen (Twomey, 1982). Various T cells, including the T-helper and T-suppressor cells, regulate the immune response. There also are cytotoxic (killer) T cells, which destroy target cells. Target cells are virally infected or transformed cells whose elimination will prevent the progression of a virus infection or tumor development, respectively. Target cells have surface antigens that bind to antigen-specific receptors on the surfaces of killer T cells. About 5% of the circulating lymphocytes are called ''null" lymphocytes because they lack the specific cell-surface molecules characteristic of B cells and T cells. The cell-to-cell regulation of the immune system is mediated by locally acting hormones called cytokines, which are secreted by activated cells. Lymphokines are cytokines secreted by lymphocytes. Interleukin-2 (IL-2) is a lymphokine produced by helper T cells that acts as a growth factor for all T cells including the cells that make it. Helper T cells make other lymphokines, e.g., gamma interferon and IL-4.
The development of the human immune system begins late in the fetal period, is functioning at birth, and reaches maximum capacity around the time of puberty (Paul, 1984; Claman, 1987). In the human adult, the majority of circulating lymphocytes are T cells, and the remainder are B cells and NK cells. However, the production of B cells and T cells continues, albeit at a reduced rate, throughout life. The total lymphocyte count in the peripheral blood can be up to 3,000/mm 3 during childhood; in the adult, the average is about 2,500/mm3 with a low below 1,000/mm3 (Golub, 1987).
The sequence of events in the immune response is shown in Figure 2-1 and Figure 2-2. After exposure to an antigen (a molecule that stimulates a specific immune response), there is phagocytosis (ingestion) of the antigen by macrophages, during which the antigen undergoes intracellular breakdown through enzymatic hydrolysis. After hydrolysis, the fragments of the antigen move to the surface of the macrophage for reaction with specific T lymphocytes, called helper-inducer T cells. Activation of these T lymphocytes occurs only if the interacting lymphocyte has specific receptors that bind to a complex of antigen fragment and a special protein derived from the major histocompatibility complex (MHC) (Paul, 1984; Claman, 1987; Golub, 1987). The Class I MHC protein defines transplantation antigens normally recognized during graft rejection and present on all nucleated cells. The Class II MHC is a cluster of genes that encodes specific cell-surface molecules that are normally restricted to phagocytic cells (e.g., macrophages) and some lymphocytes (e.g., B cells). When a large number of the specific receptors on the individual T cell binds to the complementary complex of an antigen fragment and the MHC Class II protein on a macrophage, the T cell is stimulated into a proliferative response that leads to clonal expansion and secretion of lymphokines. If an activated helper T cell then binds to the antigen-MHC complex on a B cell, that B cell is stimulated to proliferate and differentiate into an antibody-secreting plasma cell. An activated killer T cell will bind and kill any target cell (e.g., an infected cell) if that cell carries on its surface the appropriate antigen-MHC complex. T cells that bear the CD8 or CD4 molecule will interact with target cells that bear Class I or Class II MHC protein, respectively.
The generation of antibody-producing
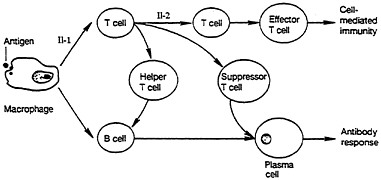
FIGURE 2-1 Cellular interactions involved in generation of immune response.
Antigen presentation leads to stimulation of T-cell or B-cell systems. Factors involved in T-cell system include interleukin-1, which stimulates T cells to acquire receptor for T-cell growth factor called interleukin-2 (Il-2); same subpopulation T cells can also secrete Il-2. Source: NRC, (1989b).
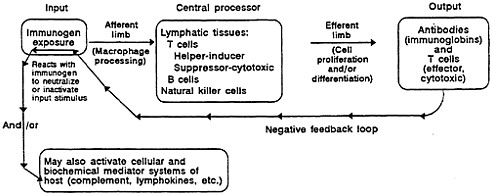
FIGURE 2-2 A model of the competent immune system depicting normal interrelations of the major components.
plasma cells (B cells) and cytotoxic T cells requires the presence of biochemical factors (lymphokines and cytokines) secreted by T cells and macrophages. As a result of the clonal expansion, the number of these specifically reactive T and B cells increases so that subsequent exposure to the same antigen leads to a rapid, specific immune response, the secondary response, which is characterized by increased secretion of antibody or proliferation of specific populations of effector T cells (Katz, 1977; Paul, 1984; Claman, 1987; Golub, 1987). As an immune response occurs, a decrease of the T cells is likely and negative feedback into the earlier phases prevents the excessive reaction. Thus, the specific antibody reacts with the offending antigen to cause neutralization or inactivation while effector T cells inactivate or destroy cellular targets.
The normal function of the immune system involves a complex sequence of cellular and biochemical events. Opportunities for dysfunction can occur at any point in the system and can result in a variety of immunologic effects from hypersensitivity to immunodeficiency (Twomey, 1982).
MECHANISMS OF CHEMICALLY INDUCED IMMUNE DISEASE
Exposure to immunotoxicants can cause immunologic suppression, resulting in altered host resistance. The outcome of immune suppression is influenced by the dose and mechanism of action of the immunotoxicant along with concomitant exposure to other agents, such as bacteria, viruses, parasites, or chemicals at levels so low they might normally be innocuous. In its suppressed condition, the immune system does not respond adequately to the hazardous agent. Adverse health consequences are those of severe disseminated infectious disease caused by a variety of agents that are not usually pathogenic. Age, poor nutrition, and stress (physiologic and psychologic) can exacerbate the development of such immunologic diseases.
Xenobiotics also can act as sensitizers to stimulate the immune system as antigens by provoking a substantial immune response that leads to hypersensitivity. Immunologic tissue damage can result from activation of cellular and biochemical systems in the host. The major immunologic tissue reactions are summarized in Table 2-1. The interaction of an antigen with a specific antibody or with
effector lymphocytes triggers the sequence of humoral and cellular events to produce the pathophysiologic effects that lead to tissue injury and disease (Samuelsson, 1983; Frank, 1987). Diseases that are immune-reaction mediated include rheumatoid arthritis, some types of diabetes, and myasthenia gravis. Each immune reaction is associated with different principal cells and biochemical mediator systems (Lachmann and Peters, 1982). This classification by the cells and immune function involved is used in Table 2-1. An older classification of immune reactions, developed by Gell and Coombs (Gell et al., 1975), is noted in Table 2-1 for comparison.
IgE-Mediated Hypersensitivity
IgE-mediated hypersensitivity involves antibodies that are produced in response to exposure to a wide variety of antigens, such as grass pollens, animal danders, foods, and commonly encountered environmental substances (Sheffer and Pennoyer, 1986). This is the most common type of hypersensitivity disorder, and it has been known variously as atopic, reaginic hypersensitivity; Prausnitz-Kustner (P-K) reactivity; or as the Type I reaction in the Gell-Coombs classification. The name used here is preferred.
A susceptible individual is initially sensitized by exposure to an antigen and subsequently develops a significant IgE response. The IgE then disseminates through the circulation and binds via specific receptors to the cell membranes of basophils in the circulation and to mast cells in the tissues. Those cells are then "sensitized," and the individual is susceptible to reacting on later exposure to the antigen.
Exposure of a sensitive individual leads to the activation of mast cells by binding of specific antigen with the IgE on the cell membranes. This interaction leads to the release of cytoplasmic granules and histamine, leukotrienes, and other vasoactive substances (Ishizaka, 1984; Sheffer and Pennoyer, 1986). These substances cause vascular dilatation and increased blood flow, increased permeability, and tissue edema along with smooth muscle contraction and hypersecretion of mucus. The local tissue reaction depends on the site of antigen exposure. Inhaled antigens cross the mucous membranes of the nose, sinuses, and lower respiratory tract to cause an increase in mucus secretion, congestion, and edema of the mucous membranes. Constriction of bronchi and bronchiolar smooth muscles limits pulmonary air exchange and causes wheezing. Sinusitis, rhinitis, and bronchial asthma can all result from IgE-mediated hypersensitivity.
Antigens ingested in food or medications can cause symptoms of intestinal hypermotility, dyspepsia, colicky pain, or a sensation of fullness and bloating. Those symptoms result from the local release of mediators in the mucosa of the gastrointestinal tract. Antigens that are absorbed in the tract enter the circulation and disseminate throughout the body, causing systemic reactions (generalized anaphylaxis) or local reactions in the skin and viscera. Skin reactions include acute edematous papules and large urticarial lesions. Chronic skin reactions, such as maculopapular rashes and eczema, also can occur. The injection of an antigen, such as a drug or vaccine, can cause disseminated systemic anaphylactic reactions, which can lead to vascular collapse, respiratory insufficiency, and death unless there is immediate treatment (Delage and Irey, 1972). Such systemic reactions result from the massive release of biochemical factors from circulating basophils.
Complement-Mediated and Immune-Complex-Mediated Injury
The complement system is made up of a series of serum proteins (C1 through C9) that can be activated by immune and nonimmune
TABLE 2-1 Immunologic Hypersensitivity Reaction—Types I-IV
|
Types of Reactiona |
Onset |
Antibody |
Principal Cell |
Site of Reaction |
Biochemical Reaction |
|
Antibody dependent: Anaphylactic (Atopic reaginic) (Type Ia) |
Rapid (seconds to minutes) |
IgE (homocytotropic) |
Basophil/mast cell |
Varies with antigen portal of entry |
Histamine, serotonin, platelet activating factor (PAF), leukotrienes (SRS-A), prostaglandins, thromboxanes |
|
Complement-mediated immune adherence (Phagocytic reaction) (Type IIa) |
Intermediate (minutes to hours) |
IgM/IgG |
R-E cells |
Vascular sinuses reticuloendothelial system |
Complement activation C3 to C3b |
|
Immuno-complex (or tissue reactive antibody) (Type IIIa) |
Intermediate (30 minutes to 2 hours) |
IgM/IgG |
Neutrophil |
Varies with tissue localization of complexes or site of reaction with tissue constituents |
Complementchemotactic factors Neutrophil lysosomal hydrolytic enzymes |
|
T-cell reactions: Granulomatous (Type IVa) |
Prolonged (delayed 18-24 hours) |
None |
Responder T cell and monocyte/macrophage |
Varies with tissue localization of antigen |
Soluble lymphokines from antigen stimulated T cell |
|
a Gell and Coombs (Gell et al., 1975) classification of Types I-IV. |
|||||
factors. Activation of complement causes chemotaxis, cell adherence, cell lysis, phagocytosis, and mast cell activation. Immune activation of complement occurs only with antigen-antibody reactions that involve IgG or IgM antibodies (Wiggins and Cochrane, 1981; Frank, 1987). Alternative pathway activation of complement can occur nonspecifically but still yield effects similar to those of immune activation. Complement-dependent immune reactions are usually localized to the tissue sites where antigen-antibody complexes form or lodge. They constitute the principal types of immunologic tissue injury.
The antigens in complement-mediated immune reactions arise from varied sources. Those from infections are derived from bacteria, fungi, or viruses. Others are from injections or inoculations (in a course of immunization), blood transfusions, or drug therapy. Sensitization occurs in an individual who was exposed to an antigen previously and has developed a specific IgG or IgM antibody response. Subsequent exposure to the specific antigen can result in immune-complex formation with complement activation, which yields molecular fragments that exhibit a variety of biologic activities. Some are potent chemoattractants for neutrophils and monocytes; others facilitate immune adherence and phagocytosis or cause release of mast cell mediators.
The sequence of events in immune-complex injury is as follows (Wiggins and Cochrane, 1981): soluble immune complexes form in the circulation of a sensitized individual. The complexes can become localized in tissue, via permeability of small venules, and produce tissue lesions. Fragments of activated complement promote chemotaxis of monocytes and neutrophils to the specific tissue site, where they release destructive hydrolytic enzymes that cause necrotic lesions. Examples of disseminated immune-complement disease in humans include systemic lupus erythematosus (SLE), various forms of glomerulonephritis, hypersensitivity pneumonitis, and polyarteritis nodosa.
Another form of complement-dependent cell destruction (Table 2-1) results from immune activation of complement on the surface of an antibody-coated target cell that generates abundant fragments of C3b complement (Fearon, 1984; Schifferli et al., 1986). The C3b molecules promote phagocytosis by attachment to the target cells and to cell-surface receptors on macrophages and reticuloendothelial cells and thus speed removal of the target cells from the circulation. The C3b-coated cells removed by phagocytosis are destroyed within the phagocyte. If erythrocytes are the target cells, chronic destruction leads to severe anemia. These reactions often occur when IgM or IgG antibody reacts with native cell-surface antigens, with drugs, or with infectious agents that have become bound to the cell surface. Autoimmune hemolytic anemias, neutropenias, and immune thrombocyto-penias are examples of this type of immune injury in humans.
T-Cell Reactions
Immune-system reactions that result in specific T-lymphocyte activation and response can cause T cells to act as autoimmune effector cells (Turk, 1980). Alternatively, some T-cell-dependent reactions might result in the formation of granulomas (Turk, 1980; Springer et al., 1987) (Table 2-1). Activated T cells produce and release lymphokines, biochemical factors that mediate T-cell reactions. Some lymphokines activate blood monocytes to cause their transformation to macrophages; others are chemotactic for monocytes and attract them into tissue sites. If the process is chronic (lasts for weeks or months), the attracted macrophages cluster and fuse to form giant cells in the tissue at the site of lymphocyte activation. Those cellular events produce a typical lesion called a granuloma. The
multinucleate giant cells (macrophages) are mixed with clusters of mononuclear cells (macrophages and lymphocytes). Granulomatous reactions are characteristic of chronic T-cell-dependent reactions (Table 2-1). They are found in infectious diseases in which the antigen persists with continued T-cell activation. Granulomatous reactions are common in tuberculosis and in fungal infections. They also can occur with exposure to particulate chemicals such as beryllium and talc.
Another T-cell-dependent tissue injury results from the binding of specific T-effector cells to a cell-bound antigen on target cells. Such effector cells are called killer T cells because they release chemicals that kill the target cell shortly after contact. Examples are found in a variety of viral infections, chemical contact sensitivity, and drug reactions. In chemical contact sensitivity, the chemical becomes adsorbed to a cell surface and becomes the target antigen for specific effector T cells that kill the cell linked to the chemical.
EFFECTS OF XENOBIOTICS ON THE IMMUNE SYSTEM
Some drugs and chemotherapeutic agents can suppress the immune system. These agents are used clinically to prevent rejection of transplanted organs and in the treatment of some autoimmune diseases. When the drugs are administered at doses that prevent organ rejection, patients are at increased risk of infection and neoplasia as a consequence of their reduced immune function. Other xenobiotics are suspected of causing immunosuppression in humans through environmental exposure, but definitive evidence is lacking.
Toxic agents might cause abnormalities in immune function at several points. These points are illustrated in Figure 2-3 superimposed on the model in Figure 2-2. There are four major sites at which toxic agents might affect immune function (Gibson et al., 1983; Luster et al., 1987). At point 1, a toxic material could lead to a specific immune response that produces tissue injury through
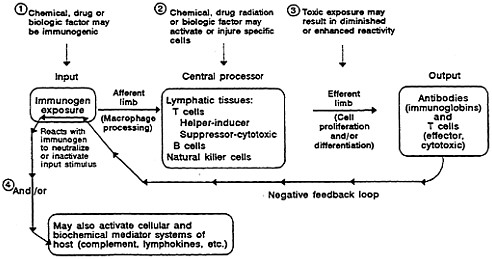
FIGURE 2-3 A model of the competent immune system depicting sites of potential effects on the major components by toxic factors.
any of the different types of immunologic injury (Twomey, 1982). At point 2, a toxic agent could alter the activity of specific lymphocytes. The result can be either a regulatory defect in the immune function, which could lead to excessive immune reactivity, or an autoimmune reaction (Twomey, 1982). A third point of potential toxic effect might be on lymphocyte proliferation or differentiation that could lead either to a defect or to an enhancement of specific reactivity. Finally, at point 4, immune reactivity could be excessive in widespread activation of other cells (macrophages, granulocytes, platelets, and mast cells), activation of potent plasma biochemical systems (complement, fibrinolytic, coagulation, and kinin), or secretion of biochemicals from stimulated lymphocytes.
Chemicals that suppress bone marrow function can affect the reserves of stem cells that are needed for cell replacement. Blood-cell lines are derived from pluripotent stem cells, which in adult humans are primarily in the bone marrow. Within the marrow microenvironment, these self-renewing cells mature into committed progenitor cells, which are in peripheral blood and tissues. The continued development of these cells is under the control of various growth factors, many of which originate in bone marrow stromal cells. Bone marrow stromal cells also provide a supporting matrix for development of hematopoietic cells. Various studies, including those on the use of long-term bone marrow cultures, have demonstrated the importance of the microenvironment in regulating myeloid and lymphoid development. Stem cells often appear to be sensitive targets for therapeutic and environmental toxicants, most likely because of their rapid proliferation. Myelotoxicity or bone marrow toxicity caused by xenobiotics or various drugs can result in profound immunosuppression due to loss of stem cells.
Xenobiotics can act as immunogens to stimulate the production of specific immunoglobulin as a part of an immune response. Specific immunoglobulins might be used as markers of exposure to specific xenobiotics.
Other biologic markers that could be applied to the human immune system are discussed in subsequent chapters. They are derived principally from or related to the varied biochemical and cellular factors discussed in the foregoing sections. They include the total and relative numbers of circulating lymphocytes and their subpopulations; the different classes of immunoglobulins (in addition to the specific antibodies already mentioned); lymphocyte proliferation stimulated by mitogens and specific antigens (xenobiotics); complement activation by specific xenobiotics; skin test response patterns to xenobiotics; in vitro lymphocyte and monocyte activation by xenobiotics, with measurement of lymphokine secretion; and other markers found by the techniques of cellular and molecular biology that can sensitively assess the structure, function, and complex interactions of the many components of the immune system. Animal models could prove useful in defining appropriate immune-system markers for xenobiotics in humans. The study and application of immune-system markers in humans to the assessment of toxic environmental exposures are now in the initial stages. Many questions require experimental and epidemiologic answers to ascertain the usefulness of particular markers. It is expected that this report will help stimulate the major research needed.










