1
Introduction
At the request of the U.S. Environmental Protection Agency (EPA), the National Institute of Environmental Health Sciences (NIEHS), and the Agency of Toxic Substances and Disease Registry (ATSDR), the Board on Environmental Studies and Toxicology in the National Research Council's Commission on Life Sciences convened the Committee on Biologic Markers to examine the use of biologic markers in environmental health research. Biologic markers are broadly defined as indicators of events in biologic systems; they can be variations in the number, structure, or function of cellular or biochemical components. Biologic markers are of interest as a means to identify early stages of disease and to understand the basic mechanisms of the effects of exposure and the biologic responses to substances found in the environment (Committee on Biological Markers of the National Research Council, 1987). Four specific biologic systems were chosen for study: the reproductive system (NRC, 1989a), the respiratory system (NRC, 1989b), the immune system, and the urinary system. This is the report of the Subcommittee on Immunotoxicology.
The immune system recognizes and defends against infectious micro-organisms and neoplastic cells. Many foreign materials are prevented from entering the body or are rapidly eliminated by nonspecific, nonimmune mechanisms (e.g., mucous secretions and phagocytosis by macrophages) and by immune mechanisms. With some substances, individuals may develop an immune response that is specific to the substance so that the body is able to react more quickly and effectively to a future attack by the substance. This adaptive immune system may be considered in simple terms to consist of three specific elements: the foreign substance, which is called the antigen; lymphocytes, which are cells of the blood and lymphoid system; and antibodies, the immunoglobulin (Ig) proteins formed by the immune system. Interactions among these three specific elements and other nonspecific cells (e.g., antigen-presenting cells) or other biologic systems (e.g., the immune-complement system) form the basis of the activity of the immune system. A response against an antigen that requires the local accumulation of lymphocytes is termed cell-mediated immunity and the lymphocytes involved are called T cells. Responses involving antibodies made at a distant site are referred to as humoral immunity and the lymphocytes producing the antibodies are called B cells. A generalized reduction in the capacity for
either type of response is known as immunosuppression and may result in an increased susceptibility to infection by micro-organisms or to the development of tumors, as seen, for example, in acquired immune deficiency syndrome (AIDS). A generalized increase in immune responsiveness is known as immunopotentiation. One manifestation is hypersensitivity (allergy). When the immune system responds to and attacks the proteins of its own tissue, autoimmune disease may occur. In Chapter 2, the function of the immune system is given with greater detail along with an explanation for how disease may evolve from disregulation of the immune system.
Immunology is primarily a science that began in the late nineteenth century. Special interest in chemicals from nonbiologic sources—xenobiotics—is of recent origin. Immunotoxicology formally emerged as a distinct discipline within toxicology during the 1970s (Descotes, 1988), prompted by animal studies that demonstrated the researcher's ability to measure the effects of chemicals on the immune system (Koller, 1980; Vos, 1980; Dean et al., 1982; Luster et al., 1982). Landsteiner in the mid-1930's demonstrated that the guinea pig could be used to determine the sensitization potential of chemicals (Landsteiner and Jacobs, 1935, 1936), and these techniques have been used over the years in the determination of the potential to produce hypersensitivity responses, particularly with cosmetics ingredients, consumer products, and drugs. The ''new" field was fully recognized in 1978 when an immunotoxicology program was established within the National Toxicology Program. The goal of the immunotoxicology program was to select, develop, and validate animal models for use in assessing immunomodulation induced by xenobiotic substances. The initial focus of investigations was the potential of nonbiologic chemicals to cause immunosuppression. More recently, active sensitization and hypersensitivity reactions to xenobiotics have received attention.
Immune responses are many and varied, and they produce markers that can indicate environmental exposure. Such markers include increases in specific antibodies, increases or decreases in total immunoglobulin, changes in the absolute or relative numbers of lymphocytes, and changes in the in vitro or in vivo reaction to antigens or mitogens. It can be difficult to distinguish responses that indicate toxicity, and are thus markers of effect, from those that are normal physiologic adaptations to the environment.
Continued integration of the work of toxicologists, pharmacologists, and other researchers has led to the use of a diversity of biologic markers. For this study, the committee, for the most part, deals with the potential of xenobiotics to produce an adverse effect on the immune system manifested by hypersensitivity, autoimmunity, or suppression of the immune system. The suppression of the immune system can lead to the development of an increased rate of infection or cancer.
Humans are often and unavoidably exposed to many hazardous environmental chemicals. The immune system functions to neutralize foreign materials and infectious agents by specifically responding to varied macromolecular factors in the environment. In addition to its response to molecules derived from bacteria and viruses, the immune system may respond to varied macromolecular components in ingested food, inhaled air, and anything that touches the skin. the responses of the immune system usually are beneficial, although some can be harmful, either because of an over-response that leads to hypersensitivity or autoimmunity or because of a suppression of the immune system that impairs the response to other environmental stimuli.
Immune responses can be specific or nonspecific. Specific responses usually involve a small fraction of highly specific lymphocytes, and the degree of response to different antigens can vary widely within an
individual. Animal experiments can demonstrate which substances are potential antigens in humans. However, animal results do not always predict the occurrence of hypersensitivity reactions in humans. For example, exposure to ragweed pollen induces a normal antibody response in mice, but it can cause severe allergy in some humans.
Many persons are afflicted with diseases, such as skin allergies and asthma, that are related to immune hypersensitivity. Immunologic (allergic) asthma is defined as variable airway obstruction that results from exposure to generally low-level concentrations of immune-reactive substances in the environment. Skin-sensitizing chemicals often are found in the industrial workplace, and 20-25% of all cases of occupational dermatitis are estimated to result from immune sensitization. Between 50,000 and 100,000 workers in the United States are regularly exposed to highly reactive compounds, such as diisocynates (Musk et al., 1988); many individuals will develop antigenspecific antibodies to toluene diisocyanate (TDI) after respiratory exposure. Five to ten percent of exposed workers have become hypersensitive to TDI. In such situations, the presence of specific antibody is a definitive marker of exposure, although not all workers hypersensitive to TDI have detectable TDI antibodies, nor do all workers with antibodies develop symptoms of hypersensitivity.
The Spanish toxic oil syndrome developed in approximately 20,000 people in Madrid in 1981 after they had ingested adulterated rapeseed cooking oil. Affected persons had symptoms of fever, rash, dyspnea, malaise, and gastrointestinal symptoms. Most made an uneventful recovery, but approximately 15% developed scleroderma-like illness that was suggestive of autoimmune disease.
Nonspecific effects of xenobiotics on the immune system can involve a variety of cells and similar actions are observed between individual and species. For example, exposure to whole-body radiation or cyclosporin A depresses the immune systems of mice and humans; each modality affects different cells, but they act in a similar immunosuppressive manner in both species.
Organ-transplant patients are treated with drugs that suppress the immune response to prevent organ rejection, but because their immune system is deficient, these patients frequently develop infections and some types of neoplasm. The relationship between the immune system and the development of cancer has been recognized for years. In adopting standards on 13 occupational carcinogens in 1974, the Occupational Safety and Health Administration required that "in all physical examinations, the examining physician shall consider whether there exist conditions of increased risk including reduced immunological competence. …"
Recent animal immunotoxicity studies suggest that some environmental substances induce immune-system suppression. Trichloroethylene (TCE) in the drinking water of mice has been found to suppress humoral and cell-mediated immunity. Neither the period of TCE exposure nor dose-response correlations have been established in human studies, but leukemia and increased infections have developed in some populations exposed to TCE as a result of contaminants in their drinking water. The Agency for Toxic Substances and Disease Registry (ATSDR) has recently established a registry of individuals exposed to TCE, which eventually could be helpful in establishing a relationship between the animal bioassay results and presumptive human health effects of TCE (Burg, 1990). In addition, registries for benzene and dioxin have also been established. These chemicals also suppress the immune system as part of their spectrum of toxicity.
BIOLOGIC MARKERS
In the broadest sense, biologic markers are measurements on biologic specimens
that will elucidate the relationship between environmental exposures and human diseases, so that such exposures and diseases can be prevented. Early detection that leads to prevention of disease and of disability is the ultimate goal and promise of the use of biologic markers.
The term "marker" is commonly used by immunologists for membrane proteins that "mark" different kinds of cells. Interest is growing in the use of biologic markers by researchers in clinical medicine, epidemiology, toxicology, and related biomedical fields to study the health effects of exposure to environmental toxicants. Clinicians can use markers for early detection of disease. Epidemiologists can use them as indicators of exposure to determine internal dose or health effects. Toxicologists can use them to develop estimates of dose-response relationships and to facilitate assessment of risk associated with small exposures. Biologic markers also can be helpful in clarifying the underlying mechanisms of chemically induced diseases. Recently, selected immune-system markers have been measured in epidemiologic studies of exposed populations to detect associations between exposure and disease. However, the complexity of the test populations and uncertainties in the measurement of markers of immunotoxicity have prevented meaningful evaluation.
New developments in molecular biology and biochemical approaches to medicine have elucidated sensitive markers for assessing exposure (NRC, 1991). They have also increased our knowledge of disease, improved our ability to predict the outcome of disease, and helped direct courses of treatment. There has been an explosive expansion in interest and increased activity in immunologic markers because of AIDS. Many diseases are defined not only by clinical signs and symptoms but also by the assessment of biologic markers at the subcellular and molecular levels. Diseases of the liver and kidney, for example, are often detected by measurement of enzymes in blood or proteins in urine; diabetes can be suspected if glucose is found in urine; and inborn errors of metabolism, such as phenylketonuria, are found by early biochemical analysis, rather than later as a result of clinical dysfunction.
The Committee on Biologic Markers of the National Research Council (1987) defined biologic markers as indicators of events or conditions in biologic systems or samples. As such, they are indicators of exposure, effect, or susceptibility. This classification is a useful theoretical scheme by which to characterize biologic markers of any organ system; however, it must be qualified somewhat for practical application. First, classification with respect to any of the three categories will depend on the particular definition of that event. Second, the three categories often are related and can be seen as descriptors of a continuum from environmental exposure to clinical disease (Figure 1-1).
The measurement of a chemical in a biologic specimen is a marker of exposure, and it could be more useful than measurements
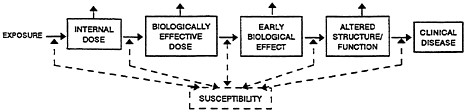
FIGURE 1-1 Simplified Flow Chart of Classes of Biologic Markers.
on air samples at a potential exposure site. Cellular or molecular changes associated with disease are biologic markers of adverse health effects. Biologic markers often are elucidated by clinical laboratory measurements or clinical tests used in the differential diagnosis of various diseases. The markers can serve as surrogates for other methods of detection in determining the molecular and cellular events in the development of health problems. If such markers could be detected before exposed persons became obviously ill, the disease process might be reversed in those affected or prevented in others. In addition, some cellular or molecular measurements can identify people who are more vulnerable to the effects of toxic exposure; these are markers of susceptibility. There also might be biologic markers that would indicate individual susceptibility to environmentally induced disease.
Markers of Exposure
A biologic marker of exposure is a xenobiotic chemical or its metabolite or the product of an interaction between the chemical and some target cell or biomolecule. Most commonly, the indicators of exposure are the concentration of the material in urine, blood, or other body tissue, including the hair or nails. The most definitive immune-system markers of exposure are antigen-specific antibodies or cellular responses to a particular xenobiotic. Immune-specific biologic markers of exposure include antibodies to toxicants (Pezzini et al., 1984), in vitro proliferative responses of immune cells upon exposure to toxicants or toxicants conjugated to proteins (Kapsenberg et al., 1988), and in vivo responses (e.g., as shown by skin tests) to toxicants (Baur et al., 1984).
Several problems limit the usefulness of antigen-specific markers of exposure. First, many environmental xenobiotics are small molecules that must act as haptens to elicit an immune response. Haptens alone cannot evoke an immune response; they must be covalently linked or strongly bound to "carrier" proteins in the tissues. For instance, isocyanates react chemically with tissue proteins, and nickel forms very strong tissue chelates. Antigen-specific immune markers can be detected for both of these agents. However, many environmental toxicants do not trigger antigen-recognition pathways and will not produce specific immune markers.
Even if a xenobiotic can elicit a specific immune-system response, the dose and route of exposure must be appropriate. Very small or very large doses of a substance can induce tolerance and even paralyze the immune system, ablating any indicators of exposure. The route of administration also can be critical.
A third problem with specific immune-system markers of exposure is that they decay with time after exposure ceases. This decay of responsiveness is the reason re-immunization is often required for maximum protection. A false-negative result can therefore be obtained if specific immune markers are sought long after exposure. The persistence of immune markers varies widely among the different humoral mediators and cellular components, and it can depend on conditions within the organism. Highly reactive humoral mediators that act locally (such as prostaglandin) are often inactivated within minutes of their formation, even though their inactivation products can circulate in the system much longer. Serum IgE is cleared more quickly than is serum IgG, but IgE bound to mast cells or basophils persists much longer than any serum protein does.
Markers of Effect
A marker of effect is a measurable cellular or biochemical alteration within an organism that, depending on magnitude, can be recognized as an established or potential health impairment or disease. A marker of
susceptibility is an indicator of an inherent or acquired limitation of an organism's ability to respond to the challenge of exposure to a specific xenobiotic substance.
Immunologic markers of effect include changes in the components of the immune system itself (such as shifts in the distribution of lymphocyte subpopulations) and changes in other tissues caused by immune-mediated dysfunction (such as signs of kidney failure caused by autoimmune kidney disease). As with all organ systems, the most critical aspect of defining immune-system markers of effect is to define the criteria for determining whether the effects have occurred (Radford, 1981). This point is especially important because changes in the immune system can reflect an extremely broad spectrum of biologic effects ranging from normal variation to sudden death (Table 1-1). Therefore, immune-system markers of effect must be defined in terms of the specific health effects for which they might serve as indicators.
When there are clinically apparent health effects caused by immune-system dysfunction, changes in immune-system markers are often evident and serve as helpful indicators of pathogenesis. Biologic markers of effect are usually less obvious during the inactive phases of immune-mediated disease. If immune-related effects occur below the clinical disease level, the altered state of the immune system cannot necessarily be "recognized as impairment or disease" (Committee on Biological Markers of the National Research Council, 1987). However, because of immune "memory," a silent effect can still have disease potential. Ultimately, the usefulness of immunologic markers of effect will be determined by the extent to which they indicate or predict actual disease. The greater the correlation between an immunologic marker change and an eventual problem with host resistance, hypersensitivity, or autoimmunity, the greater the value of the marker in determining public-health priorities.
Markers of Susceptibility
Susceptibility factors, also called effect modifiers, can act at any point along the exposure-disease continuum (Figure 1-1), and in some cases, these factors cause uniformly
TABLE 1-1 Examples of Health Effects Associated with Immune Dysfunction
|
Effect |
Hypersensitivity |
Immunosuppression |
|
Death |
Lethal allergic reaction |
Overwhelming sepsis |
|
Overt disease |
Chronic asthma |
Frequent recurring infections, some cancers |
|
Subclinical disease |
Early chronic beryllium lung disease |
Increased susceptibility to infection |
|
Subclinical dysfunction |
IgE hyperproduction |
Complement C5 deficiency |
|
Nonspecific complaints |
Arthritis, headache, fatigue |
Recurrent minor infections, longer infection duration |
|
Years potential life lost |
Contributions from all above |
Contributions from all above |
exposed populations to exhibit markedly different effects (Hulka and Wilcosky, 1988). However, in other cases, it can be difficult to distinguish between susceptible and nonsusceptible individuals, either because of multiple interactive influences and genetic polymorphisms or because of difficulties with measuring susceptibility factors.
Potential sources of normal variability in markers of immunotoxicity can be the result of genetic, environmental, or biorhythmic influences other than the specified events that are the object of study. Such influences can cause differences between populations, differences between individuals, and differences within individuals over time. Three of the most important sources of variability are genetic, age-related, and neuropsychologic factors.
Some genes code for the antigen recognition portions of antibodies and T-cell receptors, while other genes code for the type of antibody produced (such as IgG or IgE). In some cases, susceptibility genes code for proteins involved with other activities not directly related to immune-system function. For instance, the probability of developing autoimmune illness from exposure to procainamide is influenced by the genetically determined rate at which it is acetylated (Reidenberg and Drayer, 1986). Women are predisposed to autoimmune disease (Bias et al., 1986). The activation of some oncogenes (Haluska et al., 1986; Nowell and Croce, 1988) also could signal an increased susceptibility, especially toward immunoproliferative disorders (lymphocytic leukemias and lymphomas). Confounding factors can be controlled in epidemiologic study designs by restriction or matching and in analyses by stratification or multivariate methods. When genetic factors confound, the choice of inheritance models is important for interpreting results.
Immune function and markers of the immune system vary normally with age, and differences are especially notable in the very young and the elderly. In general, the variation of many immunologic factors, which is relatively small in young populations, increases with age (Hausman and Weksler, 1985); hence, there can be a wide range of variability in older populations. The developing fetus is unable to recognize and react to a wide range of foreign substances, and it is more susceptible to long-term immunotoxic effects than is the adult (Lewis et al., 1978; Bick, 1985; Hausman and Weksler, 1985).
Pregnancy produces profound changes that can have specific and nonspecific effects on the immune system. The influence of pregnancy on many immune-system markers is not well established, so interpretation of results from pregnant women can be difficult. The nonspecific effects of pregnancy on the immune system include those mediated by hormones such as estrogen, which is largely immunosuppressive. For instance, women with autoimmune inflammatory diseases such as lupus often undergo a profound remission of symptoms while pregnant, although the underlying disease often reappears shortly after the birth (Jansson et al., 1987). The specific effects of pregnancy involve the maternal immune response to paternally derived antigens of the fetus and placenta. Placental antigens (mostly from the major histocompatibility complexes) can evoke antibody, cellular, and morphologic responses by the maternal immune system (Gill, 1985). Humoral responses to antigens of the human fetoplacental unit include antibody to antigens of the ABO and Rh blood groups. Immunoglobulin G levels appear to decrease during gestation and could be inversely correlated with α-fetoprotein levels (Wajner et al., 1987). Some of the results reported for peripheral blood leukocyte and lymphocyte subset counts during different stages of pregnancy have been contradictory, but a decrease in peripheral blood lymphocytes in early pregnancy has been observed consistently (Siegel and Gleicher, 1981; Valdimarsson et al., 1983; Degenne et al., 1988; Iwatani et al., 1988; Castilla et al., 1989). A
relationship between reduced lymphocyte counts and normal birthweight has been suggested (Milns and Gardner, 1989). Pregnancy-related changes in mitogenicity also have been reported (Yoshida et al., 1989). Cytotoxic antibodies and lymphocytes to human leukocyte antigen (HLA) increase in prevalence as pregnancy progresses. The antibody response is detectable in the first month of gestation during the first pregnancy, and by the second month in subsequent pregnancies. The cell-mediated response begins at about 14 weeks of gestation and increases rapidly thereafter (Gill, 1985). Transplacental sensitization of the fetus also can occur by passage of molecules and cells from the mother. This transfer would be important in studies of cord blood, fetal tissues, or neonates.
Involution of the thymus gland, the site of T-cell lymphocyte maturation, and the decline of serum thymic hormone activity are changes of immune function found in all persons as they age (Hausman and Weksler, 1985). Results of T-cell changes in the elderly are contradictory, some showing small changes in total cell counts and their distributions among subclasses and others showing no differences (Antonaci et al., 1987). The total number of B cells in peripheral blood does not appear to change significantly with age (Antonaci et al., 1987). Enzyme activity in lymphocytes can change with age (Weksler and Hütteroth, 1974). Small, but statistically significant, changes in the concentration of immunoglobulins are found in serum of older persons compared with those who are younger. The concentration of IgA and IgG in human serum increases in older blood donors; serum IgM decreases (Buckley and Dorsey, 1970).
Older people have an increased frequency of autoantibodies to nucleic acids, smooth muscle, mitochondria, lymphocytes, gastric parietal cells, immunoglobulin, and thyroglobulin (Pandey et al., 1979; Hausman and Weksler, 1985). These antibodies might contribute to the deregulation of the immune system that accompanies aging. It is important in evaluating age-related immunotoxic effects to distinguish between an alteration in the immune response that causes shortened survival and an alteration in the immune response that results from factors that lead to reduced survival.
Neurogenic and psychogenic factors also can influence susceptibility to immune-system dysfunction. Neurogenic factors (for which useful markers are difficult to measure) are probably involved in the pathogenesis of atopic reactions, IgE-mediated hypersensitivity (Greene et al., 1988). Psychoneuroimmunology is a relatively new research area, but the clinical literature suggesting that the immune system can be altered by psychological means is old. Nonspecific effects of neurogenic factors on the immune system are well established, but specific effects are difficult to explain and must be viewed with some skepticism. Ader et al. (1990) recently reviewed the interactions between the brain and the immune system. The interactions are sometimes rather remarkable. For example, Braun (1983) reported that patients with multiple personality disorders can have distinct hypersensitivity-response profiles in each of their different personalities.
Studies in humans have implicated psychological elements in the susceptibility to and recovery from infections, bacterial, allergic, autoimmune, and neoplastic diseases. Stress of various types (loss of a spouse is frequently mentioned) can affect the immune system. Stress involves the endocrine, neurobehavioral, and immune systems. Changes in glucocorticoid levels are frequently cited as a result of this interaction. The influences of stress on the immune system are generally suppressive and can lead to decreases in qualitative and quantitative immune factors (Bartrop et al., 1977; Crary et al., 1983).
Interactions between the nervous system and the immune system can also be demonstrated in experimental animals. For
example, Haloperidol, an antipsychotic drug, reduces antibody responses in normal mice, but restores humoral immunity in mice stressed by crowding (Baranic et al., 1979). The cellular and molecular events that form the basis of this interaction are only now being elucidated. Immunologic cells can have a variety of receptors for hormones, including corticosteroids, insulin, and sex hormones. Conversely, some of the cytokines that transmit signals through the immune system also can affect endocrine and nerve tissues. For instance, interleukin-1 (IL-1) can trigger adrenal cortical trophic hormone (ACTH) and glucocorticoid synthesis by way of the pituitary-adrenal axis (Bateman et al., 1989) and could have direct neurologic effects (Fibbe et al., 1989).
Simon et al. (1990) suggested that association of unpleasant job-related conditions might be the underlying basis of some apparent hypersusceptibility responses in workers. Animal models show responses can be immunosuppressive (Dantzer and Kelley, 1989) or immunoreactive (Koehler, 1985). In experimental animals, immunosuppression can be conditioned by pairing a stimulus with an immune suppressive agent, such as cyclophosphamide phosphamide (Ader and Cohen, 1991). Modification of homeostasis in immune system organs or direct interaction of neurotransmitter such as epinephrine, serotonin endorphins, enkephalins, norepinephrine, and acetylcholine with the cells of the immune system can occur. Receptor sites for some neurotransmitters among subpopulations of lymphocytes have been demonstrated (Bishopric et al., 1980), as have binding sites on other constituents of the immune system (Root-Berstein and Westfall, 1984).
Although neuropsychologic, age-related, and hereditary factors can influence the functional integrity of the immune system, susceptibility to immune disorders also can be influenced by a variety of other conditions and behavioral and environmental factors, and the markers of immunotoxicity can be influenced by a variety of physiologic or pathologic processes, some of which are completely unrelated to toxicant exposure or immune function. These factors include exposure to toxicants acting either directly or indirectly on the immune system, concurrent disease and the associated medications, dietary factors, and even daily or seasonal variations in the exposure to light. Table 1-2
TABLE 1-2 Factors Influencing the Immune System and Associated Markers
|
Factors |
Results |
|
Hormonal, diurnal, seasonal, age |
Variability within a healthy individual |
|
Environmental, genetic, neuropsychologic |
Variability between healthy individuals |
|
Viral, rickettsial, bacterial, parasitic |
Infectious diseases |
|
Immunoproliferative diseases |
Myeloma, leukemia, lymphoma |
|
Chronic reactive diseases |
Autoimmune diseases |
|
Acute reactive diseases |
Allergies, asthma, death (anaphylaxis) |
|
Suppressive disorders |
Increased host susceptibilities, death (infection or cancer) |
indicates a variety of the factors that influence the immune system and their outcome. The importance of controlling for these factors in any particular study will depend on the study design and the populations involved.
Although previous exposure to an antigen resulting in sensitization is an obvious example of an exposure resulting in an effect on the immune system, atmospheric contaminants can have an effect on such immunologic markers as serum immune complement-inactivation products (Stiller-Winkler et al., 1989). Simultaneous exposure to low levels of two or more air contaminants can result in a synergistic effect on the immune system (Holt and Keast, 1977). Because of the geographic variability of air pollutants, immunotoxic differences should be considered in field studies that compare geographically disparate groups. There is extensive evidence from human and animal studies that exposure to environmental tobacco smoke results in changes in the immune system (Holt and Keast, 1977). Peripheral blood leukocytes from smokers exhibit impaired chemotactic responsiveness, and smokers invariably have increased numbers of all major classes of peripheral lymphocytes (Corre et al., 1971; Holt and Keast, 1977). An association has been demonstrated between smoking and serum IgE levels (Warren et al., 1982). Other immunoglobulins, such as IgA, are decreased in smokers. Smoking also could decrease levels of natural-killer-cell activity (Ferson et al., 1979).
Much of the knowledge of immunotoxicology is derived from evaluations of exposure to therapeutic drugs. Any study of the effects of environmental chemicals on the immune system must entail consideration of the drugs the subjects use. Descotes (1988) has provided a comprehensive review of immunotoxicologic effects of drugs. Almost all categories of therapeutic agents can have some effect on the immune system: antimicrobial and antiparasitic agents; drugs acting on the nervous, cardiovascular, gastrointestinal, and respiratory systems; hormones and hormone antagonists; anti-inflammatory, immunosuppressive, and immunoenhancing drugs; drugs that produce blood clotting and fibrinolysis; and vitamins and miscellaneous drugs. Nonprescription drugs can be important modulators of immune-system markers. Aspirin, for example, can have a wide range of immunologic effects, including possible inhibition of lymphocyte response to mitogens and depression of neutrophil function. Vitamins also are used commonly, and their individual or combined influence on most immune-system markers is not known. Likewise, the influence of oral contraceptives on the immune system is poorly established, although the well-known influence of sex hormones on the immune response should lead to the expectation that oral contraceptives exert similar immunologic effects. Other widely used drugs are generally believed to exert effects on the immune system. Glucocorticosteroids are directly lymphocytolytic. Other anti-inflammatory drugs, such as acetaminophen and ibuprofen, can be expected to exert various immunopharmacologic effects (Descotes, 1988) and could thereby influence immune-system markers. In addition to considering the effect of a single drug on the immune system and its markers, investigators also must be concerned about the effects of multiple-drug use. This area has received little attention and remains a potential source of confounding variables.
Frank malnourishment is unquestionably immunosuppressive and causes a decrease in many immune markers (Chandra, 1987) and in host resistance to infection (Chandra and Wadhwa, 1989). The effects are so pronounced that, theoretically, certain immune-system markers could be used in nutritional assessment; skin-test anergy and decreased peripheral blood lymphocyte counts are considered the most reliable of such markers (Dominioni and Dionigi, 1987). The effects
of marginal malnutrition on immune status are not clear, but T-cell functions might be particularly sensitive (Rogers and Newberne, 1987). Protein deficiency results in decreased IL-I release and in impaired tissue responses to it (Klasing, 1988). Deficiencies of certain amino acids, vitamins, and trace elements also are associated with decreased immune competence (Chandra, 1987). These effects can be compounded by other variables, most commonly by aging (Thompson et al., 1987) and alcoholism (Watson, 1988). Conversely, the excessive intake of polyunsaturated fatty acids, iron, and vitamin E can be immunosuppressive (Chandra, 1987).
Serum concentration of IgE may vary with the time of day. Seasonal variations in light may indirectly influence immune responsiveness by altering antigen exposure—e.g., increased pollen—or indirectly by psychologic mechanisms. Photoallergy is an important component of contact hypersensitivity. Light, particularly in the wavelengths of 315-280 nm (sometimes referred to as UVB), alters immune-system function. Immunologically mediated contact sensitivity reactions to drugs, cosmetics, and soaps occurring in conjunction with sunlight have significantly increased in the past two decades (Harber and Baer, 1972). Guinea pigs are most often used in testing chemicals for their potential to produce immunologically mediated contact photosensitivity.
Leenutaphong et al. (1989) propose two types of solar-induced skin response. The first is IgE-mediated hypersensitivity to specific photoallergens generated only in patients with solar urticaria. The second type is the result of IgE-mediated induction of hypersensitivity to a nonspecific allergen and is generated in patients with solar urticaria and in normal persons.
Schwarz (1988) has demonstrated that UVB exposure leads to local and systemic immunosuppression. Epidermal Langerhans cells are the main target of the local action, and they lose their antigen-presenting capacity after UVB exposure.
Photohypersensitivity is a major concern in immunotoxicology but was beyond the scope of this report. Only a few chemicals have been tested for photoactivation to produce a structure that elicits a contact hypersensitivity response. The role and mechanisms of chemical-induced photosensitization also require further investigation. In addition, because light might have a role as an immunosuppressant, gathering additional information about the role that the immune system might have on development of skin tumors seems warranted.
VALIDITY OF BIOLOGIC MARKERS
In the course of developing this document, the subcommittee noted that various disciplines used validity and sensitivity in different ways. The oversight committee discussed the validation of biologic markers for use in assessments of human health, particularly in epidemiologic studies. To validate the use of a biologic measurement as a marker, it is necessary to understand the relationship between the marker and the event or condition of interest, for example, the potential for health impairment or susceptibility to disease. Sensitivity and specificity are critical components in the process of validation (MacMahon and Pugh, 1970). Sensitivity is the quality of an epidemiologic test method that confers the ability to identify correctly persons who have the disease or condition of interest. A test is specific if it correctly identifies persons who do not have the disease or condition of interest. By extension, the ability of markers of exposure or effect to indicate true exposure or disease (sensitivity) and their ability to indicate lack of exposure or disease (specificity) must be validated. Analytical sensitivity refers to the ability to detect small amounts of the marker. The terms validated and sensitivity are
used in describing the usefulness of certain bioassays by immunotoxicologists, who speak of a sensitive assay as one that produces the end points indicating an adverse effect on the immune system at low doses. Low doses are those that do not otherwise overwhelm other systems of the body involved with the maintenance of homeostasis. Immunotoxicologists also speak of validated bioassays to indicate those that have been used in several laboratories and provide similar results for compounds that are known to alter immune function. These terms are used in this way in chapters 3 and 6 with regard to the animal tests discussed therein.
Specificity of markers is important in the consideration of analytic validity. For example, urinary concentrations of trichoroethanol and trichloroacetic acid, metabolites of TCE, are used as indicators of exposure, but they are not specific for TCE because they are also formed by the metabolism of perchloroethylene and chloral hydrate. In addition, because of their relatively short half-lives, they must be measured within short times after exposure. In a similar manner, immune system biologic markers of isocynate hypersensitivity may lack some of the specificity that one would normally expect because of cross-reactivity (Thorne et al., 1987a).
The variability between individuals and the influence of the factors affecting sensitivity greatly influence the ability of the markers to detect those individuals exposed. Some markers that are validated generally have been established only for obvious clinical events. For instance, serum levels of immunoglobulin isotypes (IgG, IgA, IgM, IgD, and IgE) can be measured with precision and accuracy, mainly because of standard reference ranges published by the International Union of Immunological Societies and the World Health Organization (Bentwich et al., 1988). Reference ranges for adult and pediatric populations have been established, so it is known that these levels show wide normal variation between individuals. Results that fall well outside of the ranges are highly predictive of obvious health effects, such as immunodeficiency, infection, or neoplasm. However, the significance of variability within the normal range is not well known. Some correlations have been observed between shifts in values within the normal range and toxicant exposures (e.g., to lead and cadmium). Correlations in susceptibility or health effects within the normal range have not been established.
Serum immunoglobulins are proteins for which reference standards can be developed and measurements validated. Many immune-system markers are cellular, and these are generally much more difficult to measure in a standard fashion. Functional markers, for example, those found from in vitro cell stimulation or in vivo skin testing, present even more difficulties in standardized measurement. And although cellular and functional measurements offer the greatest opportunities for more sensitive markers, until such assays are made standard and their predictive values determined, they cannot provide useful information in epidemiologic studies.
These analytic considerations are complicated by the intrinsic variability of the immune system within and between individuals, making the sensitivity and specificity of many tests for immune-system markers modest to poor. Optimizing the analytic specificity and sensitivity of current assays and developing new assays with better specificity and sensitivity also should be given the greatest possible attention.
A major purpose of markers in environmental health research is to identify exposed persons, so that risk can be predicted and disease prevented. Validation involves both forward and backward processes of association: from the marker back to exposure and from the marker forward to effect. Validation of markers as applied to their use in epidemiologic studies is discussed in Chapter 9.
UNCERTAINTY AND RISK
Because so many factors can affect the immune system, a review of uncertainty factors is necessary for a comprehensive assessment of immune function in the assessment of risk. When genetic factors confound, the choice of inheritance models is important for interpreting results. The variation in age groups is particularly important and can often be controlled for epidemiologic studies. The neuropsychologic-immune relationship must be considered an important variable in analyzing immune-system markers, especially in situations where the threat or effect of environmental exposure can subject study groups to greater stress than control groups experience. Under such conditions, assessment of stress should be an essential component of study design. The importance of controlling for these factors and others known to influence the immune system in any particular study will depend on the study design and the populations involved. However, confounding factors can influence the distribution of markers significantly.
Restricting analysis to a single factor would make it more easily subject to intervening or confounding influences that could be attributed mistakenly to a xenobiotic exposure. Conversely, such factors could mask a true effect. An important question is how such factors should be analyzed statistically. Choosing among multiple markers requires attention to the appropriate statistical approaches that assist in the determination of whether the markers are statistically independent of one another. The complex interactions of the immune system require that a judgment be made of whether immunologic markers are biologically independent and should be treated as statistically independent or statistically correlated. Confounding factors can be controlled in epidemiologic study designs by restriction or matching and in analyses by stratification or multivariate methods.
ETHICAL AND PRACTICAL ISSUES
The availability of highly sensitive assays that can identify dose and effects resulting from the interplay between low-level exposures and genetic or acquired susceptibility raises several thorny ethical and moral questions. The reader is referred to Ashford and Miller (1989), Committee on Biologic Markers of the National Research Council (1987), Schulte (1987), and Weiss (1989) for discussion of those issues. Primary among them is the use that is to be made of biologic marker information, Take, for example, the theoretical case of a biologic marker known to reflect susceptibility. Should a worker who tests positive or has an increased measurement be removed from the workplace? If so, should he or she be offered an equivalent job in the same industry? Or should the workplace be cleaned up to protect the most sensitive worker?
In reality, few, if any, biologic markers are established as predictors of individual risk associated with inborn traits, exposure, or a combination of the two. On the other hand, in regard to immunotoxicity, some markers might have this predictive potential. Therefore, it is important to inform research-study participants in advance that the results are interpretable only on the group level. Participants in such studies should be provided test results that are presented and discussed in context with available information (or lack thereof) on the variability within and between people in the normal (nonexposed) population, as well as that observed in the research-study group. Participants in epidemiologic studies may resist invasive techniques for obtaining markers, or they may resist providing markers obtained by techniques perceived to be not fully safe. For example, the use of urinary concentration as a marker may be more universally accepted than a marker from a blood sample, from bronchiolar lavage, or from imaging technique. In addition, if the marker technology is to be applied to relatively large populations, it must be economically feasible.
STRUCTURE OF THE REPORT
Because of the multidisciplinary nature of this report, certain concessions have been made in application of particular terms. In this report, the discussion of the impact of materials on the immune system is largely limited to the effects of xenobiotics. This is the major drive in immunotoxicology, although antigens from biologic sources are certainly the major cause of immune modulation. In this report, the term marker is used for indicators of exposure, effect, or susceptibility, not as an identifying feature of subpopulations of lymphocytes. The reader should, however, be alerted to the special use of validity, sensitivity, and specificity as these terms apply to analytic capabilities, immunotoxicologic bioassays, or epidemiologic investigations.
Chapter 2 provides information on the structure of the immune system and on the mechanisms of immunotoxicity. Chapter 3 deals with excessive responses of the immune system that result in hypersensitivity. Chapter 4 examines how autoimmune reactions might result from chemical exposure. Chapter 5 reviews the toxicity and disease that result from factors that suppress the immune system. Chapter 6 discusses the potential role of animal models in immunotoxicology, and Chapter 7 assesses strategies for applying biologic markers of immunotoxicity to humans. Chapter 8 gives an approach to the design of field studies to evaluate the effects of immunotoxic chemicals. Chapter 9 addresses the need for updating research on the use of biologic markers in controversial areas of environmental health. Chapter 10 is a summary of conclusions and recommendations.














