Contact Lenses and Corneal Energy Metabolites in the Rabbit
Morris R. Lattimore, Jr.
The U.S. Army's increased interest in contact lenses for aviators is driven primarily by the development of the AH-64 (Apache) attack helicopter. Integral to this flight platform are a sophisticated electrooptical display device and a specialized environmental protection system. These have combined to create a spectacle compatibility problem that can restrict operational efficiency. Fifteen to 18 percent of active Army aviators are ametropic; this situation exists as a result of some waivers to existing standards and the development of late-onset maturational myopia in some individuals. Without an alternative means of optical correction, these aviators, many with advanced skills and superior performance abilities, may face nonretention on flight duty.
The potential use of contact lenses by Army aviators has stimulated organizational interest in three areas: flight performance and safety, ocular health, and physiological/biochemical effects. A preliminary study at the U.S. Army Aeromedical Research Laboratory (USAARL) (Bachman, 1988) documented certain aspects of the first two issues. A new study will attempt to examine the third issue of physiological/biochemical effects of contact lens wear in an animal model. The remainder of this discussion will highlight four issues concerning contact lens wear, summarize the methodology being used by this investigator, and provide normative energy metabolite data for the corneal epithelium of the pigmented rabbit.
PERTINENT ISSUES
The traditional concern pertaining to effects of contact lens wear has been the critical oxygen tension (COT) for supporting normal corneal function. The clinical measure for loss of normal corneal function is corneal edema
or swelling. The avoidance of corneal edema has been shown to depend on sufficient amounts of oxygen reaching the tissue (Fatt and St. Helen, 1971; Fatt and Linn, 1976; Polse, 1979). However, there is some debate as to what constitutes a sufficient amount of oxygen available to avoid corneal insult.
Early estimates of tolerable hypoxia used gas-infused goggles to create an exposure to artificially low oxygen levels for 1.5 hours (Polse and Mandell, 1970); below a critical level of 2.5 percent oxygen (partial pressure of 19 mmHg) the corneas of experimental subjects reacted with increased hydration and edema. A similar goggle study (Mandell and Farrell, 1980) established the minimum oxygen requirement for the avoidance of corneal swelling to be at least 3.02 percent (equivalent to a partial pressure of 23 mmHg). A later study, using another hypoxia-inducing goggle system (Holden et al. 1984), implied that the above values were insufficient for normal corneal function. Holden et al. (1984) indicated that the minimum precorneal oxygen tension to avoid corneal edema was at least 10.1 percent (74 mmHg).
More recent work (Weissman et al., 1988), using hydrophilic contact lenses of varying oxygen transmissibility and monitoring central corneal thickness, has suggested the corneal COT could be in excess of 20 to 40 mmHg but not necessarily as high as 70 mmHg. However, the monitoring of corneal oxygen uptake rates (Benjamin, 1986) has provided evidence that 18 percent oxygen (137 mmHg) represents the minimum value for normal corneal respiration, although corneal swelling is not evident well below 18 percent oxygen. This could be interpreted as an indication that the clinical method of assessing loss of normal corneal function (i.e., corneal thickness) is inadequate. Lastly, other investigators (Efron and Brennan, 1987) have suggested the critical oxygen requirement of the cornea is that which is normally available from the natural environment, 20.9 percent (159 mmHg).
Contact lens wear has been shown to adversely affect the corneal epithelium in two animal models. Hamano and Hori (1983) documented the suppression of basal cell mitosis in rabbit corneal epithelium associated with contact lens wear. In addition, a loss of desmosomes and superficial epithelial sloughing has been documented within 8 hours after contact lens wear in the rabbit (Francois, 1983). A similar contact-lens-induced epithelial thinning has been demonstrated in the owl monkey (Bergmanson et al., 1985). It has been suggested that contact lenses may cause a delay in epithelial cell turnover characterized by the presence of abnormal, large, senescent surface cells (Lemp and Gold, 1986); other findings included trapped surface debris, intraepithelial pseudocysts, and increased uptake of water-soluble dyes, suggesting increased permeability of these surface cells.
An additional issue has surfaced in the past 2 years: carbon dioxide expiration. Previously, it had been calculated that hydrogel contact lenses may provide a barrier to carbon dioxide efflux from the cornea, although at
the time it was considered to be an insignificant concern in terms of corneal physiology (Fatt et al., 1969). However, recent measurements of carbon dioxide calculation under hydrogel lenses (Holden et al., 1987), paired with the detection of a decrease in stromal pH following contact lens wear (Bonano and Polse, 1986), indicate CO2 to have the potential of being a significant factor in corneal physiology. While Holden et al. (1987) have tied the issues of carbon dioxide accumulation and tissue pH changes to the endothelial bleb response, it should be remembered that specific enzymatic activity (either by activation or inactivation) can be affected by pH changes. It is possible that an examination of corneal metabolic activity may answer pertinent questions regarding effects of contact lens wear.
Continuing along that line of thought, the typical corneal endothelial mosaic, consisting of cells of similar shape and size, may be altered by contact lens wear such that the normally uniform monolayer is transformed into a variety of cell shapes (pleomorphism) and a variety of cell sizes (polymegethism). Virtually every type of contact lens has been implicated in the induction of polymegethous changes (Snyder, 1982; Schoessler, 1983; Stocker and Schoessler, 1985; Holden et al., 1985). These polymegethous changes have been linked to the efficacy of the endothelial pump function, suggesting a cause and effect relationship (Rao et al., 1979, 1984; Holden et al., 1985; Sweeney et al., 1985; O'Neal and Polse, 1986). However, it may be possible that inhibition of the endothelial pump function is what alters cell shape and size, since some endothelial changes can be detected within minutes after placing contact lenses on the eyes of unadapted patients (Zantos and Holden, 1977; Barr and Schoessler, 1980; Kamiya, 1982). An immediate response could be indicative of a metabolic shift that occurs as a direct result of contact lens wear.
The application of a biochemical/metabolic approach is not a new one. Many studies have evaluated contact lens effects on corneal metabolism (Uniacke and Hill, 1972; Thoft and Friend, 1972, 1975). However, most of that work used techniques that involved traumatic separation of the corneal layers and/or bulk tissue extraction processes. As a result, multiple assays from the same cornea were not obtainable. In addition, results from such studies have yielded a variety of data with units based on wet weight, dry weight, or per milligrams of protein. Consequently, data comparison has been difficult, hampering the formation of a coordinated representation.
A microfluorometric energy metabolite assay technique (Lowry and Passonneau, 1972) has routinely been employed in the evaluation of regional brain metabolism (McCandless and Schartzenburg, 1982; McCandless, 1985; McCandless and Abel, 1985). Moreover, this technique can readily be applied to the cornea (Lattimore, 1988). Advantages of this methodology include liquid nitrogen freezing of the entire globe in order to immediately suspend metabolic activity; cryosectioning and cross-sectioning with freeze
drying of tissue samples to ensure stabilized metabolite levels; vacuum thawing to prevent condensation-stimulated enzyme activity; and microdissection of the sample, permitting a regional corneal metabolite analysis by dry weight. This method will permit a detailed investigation into the corneal metabolic response to induced stress. Data obtained for all metabolites will be in comparable units, establishing a standardized unit format for future reference. Thus far this technique has been applied to the corneal epithelium in the pigmented rabbit in order to provide initial normative data.
METHODOLOGY
Experimental Animals
Healthy, adult, Dutch-belted, pigmented rabbits were used as the experimental animals. The animals are housed in quarters approved by the National Institutes of Health under controlled artificial lighting conditions. The animals are maintained and the experiments conducted in accordance with procedures outlined in the Guide for Laboratory Animals Facilities and Care of the National Research Council (1965). Appropriately sized contact lenses are worn on one eye only for a predetermined period of time. Then, prior to sacrifice, the animals are anesthetized with intramuscular injections of Ketamine (30 milligrams/kilogram) and Rompun (7 milligrams/kilogram) and are sacrificed by cervical dislocation. The contact lens is removed, and the eye is excised immediately and immersed in liquid nitrogen to prevent significant change in metabolite levels. The contact lens is retained for protein analysis and histological sectioning.
Processing Procedure
The rabbit eyes are transferred from the liquid nitrogen container into a −80°C freezer for storage until tissue processing can be accomplished. The cornea is removed from the globe by dissection under −80°C conditions in a Lehrer cryostat. The isolated cornea is cut into halves, which are mounted on a sectioning button by immersion in a dry ice-cooled hexane solution. The corneal button mount is then transferred to a cryostatic microsome where tissue sectioning is performed. The resulting central cornea cross sections are approximately 20 micrometers in thickness. The sectioned tissue samples are placed in a metal tissue holder, covered with glass slides, and inserted into a vacuum tube. The tube is placed in a −20°C freezer and attached to a vacuum pump. The tissue is then freeze dried for a 24-hour period. After the freeze-drying process is completed, the tissue can be kept at −20°C until it is assayed.
Samples needed for assay are thawed under vacuum for 1 hour to prevent
condensation-stimulated enzyme action. The different layers of the cornea are clearly defined, which permits easy isolation of the corneal epithelium under a 3x binocular dissecting microscope. Tissue size is determined by dry weight, rather than by tissue section dimensions, which permits the analysis of very small and irregularly shaped specimens. The tissue samples are immediately weighed on a quartz fiber fishpole balance possessing µg sensitivity. After weighing and recovery, the samples are placed in an oil well rack for specific metabolite assay.
Underlying Principles
The cycling system contains several enzymes that catalyze specific interrelated reactions yielding a “net reaction.” A by-product of this multistep reaction is reduced nicotinamide adenine dinucleotide phosphate (NADPH), which fluoresces light of 460 nm wavelength when excited with ultraviolet radiation of 340 nm wavelength. By measuring the amount of this reduced pyridine nucleotide fluorescence, the original concentration of the assayed metabolite can be inferred by calculation (Lowry and Passonneau, 1972). Appropriate blanks and standards are employed to monitor the reliability of the assays. Individual enzymatic cycling and incubating techniques permit isolation of the specific metabolite being analyzed.
RESULTS
Each assay result is based on 25 to 32 separate tissue samples from three eyes, one each from three different rabbits. Metabolite concentration units are nanomoles per microgram dry weight. For the specific assay values refer to Table 1
TABLE 1 Normative Corneal Epithelial Energy Metabolite Concentrations (nanomoles per microgram dry weight)
|
Glucose |
Glycogen |
ATP |
PCr |
|
|
Mean concentration |
5.45 |
38.23 |
12.09 |
8.06 |
|
Standard deviation |
1.22 |
10.30 |
2.24 |
2.13 |
|
SOURCE: Data were obtained from three eyes, one each from three differentrabbits. |
||||
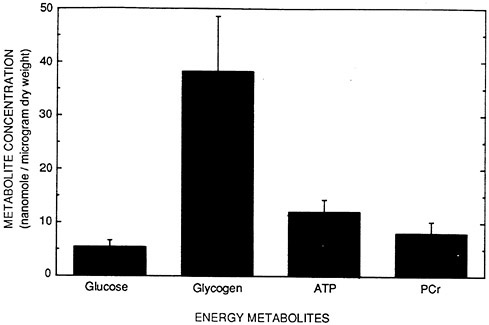
FIGURE 1 The normal rabbit corneal epithelium possesses a large amount of glycogen, a small amount of free glucose, and moderate amounts of ATP and PCr. The error bars represent one standard deviation, highlighting the relative precision of each assay.
DISCUSSION
Earlier research efforts have not been able to document a coordinated picture of the corneal energy metabolite distribution pattern because of a limitation of bulk extraction methods: essentially only one assay can be performed per cornea. The microfluorometric technique permits numerous assay repetitions per cornea. Regional assays can be performed as well, allowing separate determinations of epithelial, stromal, and endothelial metabolite levels. Epithelial data are presented in bar-chart format in Figure 1 to illustrate the interrelationship between metabolites in the normal rabbit cornea.
The corneal epithelium in the rabbit possesses a large amount of storage glucose or glycogen. Although free glucose is suggested to be present in adequate amounts to sustain purely anaerobic activity (Riley, 1969), studies have shown glycogen mobilization to be quite responsive in times of stress (Uniacke and Hill, 1972; Thoft and Friend,1975). With the microfluorometric
technique it will be possible to document the relationship between free glucose and glycogen throughout the cornea as contact-lens-wearing time is varied.
Prior researchers have inferred, because of a decreased oxygen availability during contact lens wear, that glycogen depletion represents an increase in epithelial glucose consumption as a result of glycolytic compensation. However, if stromal pH changes reflect actual epithelial intracellular conditions, then the glycogen depletion may represent merely decreased activity in the enzyme system responsible for glycogen synthesis or, on the other hand, increased activity in the enzyme system responsible for glycogen degradation. Such a condition could field increased levels of glucose within the involved tissue, perhaps beneficial for ready availability to fuel an elevated rate of glycolysis or perhaps detrimental as a source of increased hydrostatic pressure.
Note that PCr, a high-energy phosphate bond reservoir, is found at approximately two-thirds the levels of ATP. The relatively high energy reserve has been used to explain the ATP sparing, at the expense of PCr, that occurs following corneal exposure to ultraviolet radiation (Lattimore, 1988). A similar relationship between ATP and PCr likely exists in association with contact lens wear; it then will be possible to depict the interaction between energy stores and fuel stores, thereby allowing a more complete determination of the influence that contact lens wear exerts on corneal function.
REFERENCES
Bachman, W.G. 1988 Extended-wear Soft and Rigid Contact Lens: Operational Evaluation Among Army Aviators. United States Army Aeromedical Research Laboratory Technical Report No. 88-17. Fort Rucker, Ala. 36362–5292.
Barr, R.E. and J.P. Schoessler 1980 Corneal endothelial response to rigid contact lenses American Journal of Optometry and Physiological Optics 57:267–274.
Benjamin, W.J. 1988 Corneal oxygen, how much is enough? International Contact Lens Clinic 13:58.
Bergmanson J.P.G., C.M. Ruben, and L.W.-F. Chu 1985 Epithelial morphological response to soft hydrogel contact lenses British Journal of Ophthalmology 69:373–379.
Bonano, J.A., and K.A. Polse 1986 Effect of contact lens wear on stromal pH. American Journal of Optometry and Physiological Optics 63:58–62.
Efron, J., and S. Brennan 1987 In search of the critical oxygen requirement of the cornea. Contax July:6.
Fatt, I., and D. Linn 1976 Oxygen tension under a soft or hard gas-permeable contact lens in the
presence of tear pumping. American Journal of Optometry and Physiological Optics 58:104–107.
Fatt, I., and R. St. Helen 1971 Oxygen tension under an oxygen permeable contact lens. American Journal of Optometry and Physiological Optics 48:545–548.
Fatt, I., M.T. Bieber, and S.D. Poe 1969 Steady state distribution of oxygen and carbon dioxide in the in vivo cornea of an eye covered by a gas-permeable contact lens. American Journal of Optometry and Physiological Optics 64:3–14.
Francois, J.F. 1983 The rabbit corneal epithelium after wearing hard and soft contact lenses. Contact Lens Association of Ophthalmologists Journal 9:267–274.
Hamano, H., and M. Hori 1983 Effect of contact lens wear on the mitoses of corneal epithelial cells. Contact Lens Association of Ophthalmologists Journal 9:133–137.
Holden, B.A., Sweeney, D.F., and G. Sanderson 1984 The minimum precorneal oxygen tension to avoid corneal edema. Investigative Ophthalmology and Visual Science 24:218–222.
Holden, B.A., D.F. Sweeney, and A. Vannas 1985 Effects of long term extended contact lens wear on the human cornea Investigative Ophthalmology and Vision Science 26:1489–1501.
Holden, B.A., R. Ross, and J. Jenkins 1987 Hydrogel contact lenses impede carbon dioxide efflux from the human cornea. Current Eye Research 6:1283–1290.
Kamiya, C. 1982 A study of corneal endothelial response to contact lenses. Contact Lens and Intraocular Lens Medicine Journal :92–95.
Lattimore, M.R. 1989 The effect of ultraviolet radiation on the energy metabolism of the corneal epithelium of the rabbit. Photochemistry and Photobiology 49:175–180.
Lemp, M.A., and J.B. Gold 1986 The effects of extended wear hydrophilia contact lenses on the human corneal epithelium. American Journal of Ophthalmology 101:Z74–Z77.
Lowry, O.H., and J.V. Passonneau 1972 A Flexible System of Enzymatic Analysis . New York: Academic Press
Mandell, R.B., and R. Farrell 1980 Corneal swelling at low atmospheric oxygen pressures. Investigative Ophthalmology and Visual Science 19(6):697–702.
McCandless, D.W. 1985 Octanoic acid induced coma and reticular formation energy metabolism Brain Research 335:131–134.
McCandless, D.W., and M.S. Abel 1985 Hypoglycemia and cerebral energy metabolism. Pp. 27–41in D.W. McCandless ed., Cerebral Energy Metabolism and Metabolic Encephalogothy. New York: Plenum Press.
McCandless, D.W., and F.C. Schwartzenburg 1982 Audiosenic seizure induced changes in energy metabolites in cerebral portional and cerebellar lasers. Epilepsia 23:481–484.
National Research Council 1965 Guide for Laboratory Animals Facilities and Care. Institute of Laboratory Animal Resources, Committee on Revision of the Guide for Laboratory Animals. Washington, D.C.: National Academy of Sciences.
O'Neal, M.R., and K.A. Polse 1986 In vivo assessment of mechanisms controlling corneal hydration. Investigative Ophthalmology and Visual Science 26:849–853.
Polse, K.A. 1979 Tear flow under hydrogel contact lenses. Investigative Ophthalmology and Visual Science 18:409–412.
Polse, K.A., and R.B. Mandell 1970 Critical oxygen tension at the corneal surface. Archives of Ophthalmology 84:505–509.
Rao, G.N., E.L. Shaw, E.J. Arthur, and J.V. Aquavella 1979 Endothelial cell morphology and corneal detursescence. Annals of Ophthalmology 11:885–890.
Rao, G.N., J.V. Aquavella, S.H. Goldberg, and S.L. Berk 1984 Pseudo-phakic bullous keratopathy. Ophthalmology 91:1135–1140.
Riley, M.V. 1969 Glucose and oxygen utilization of the rabbit cornea. Experimental Eye Research 8:193–200
Schoessler, J.P. 1983 Corneal endothelial polymelithism associated with extended wear. International Contact Lens Clinic 10:148–151.
Snyder, A. 1982 Corneal thickness and endothelial cell changes associated with daily and extended contact lens wear . Master's thesis, Ohio State University.
Stocker, E.G., and J.P. Schoessler 1985 Corneal endothelial polymelathism induced by PMMA contact lens wear Investigative Ophthalmology and Visual Science 26:867–888.
Sweeney, D.F., B.A. Holden, A. Vannas, N. Efron, H. Swarbrick, M. Kotow, and T. Chan-Ling 1985 The clinical significance of corneal endothelial polymelathism. Investigative Ophthalmology and Visual Science (ARVO Suppl) 26 :53.
Thoft, R.A, and J. Friend 1972 Corneal epithelial glucose utilization. Archives of Ophthalmology 81:58– 62 1975 Biochemical aspects of contact lens American Journal of Ophthalmology 80:139–145.
Uniacke, C.A., and R.M. Hill 1972 The depletion course of epithelial glycogen with corneal anoxia. Archives of Ophthalmology 87:56–59.
Weissman, B.A., P.A. Blaze, S. Ingles, and N. Wheeler 1988 Open-eyed corneal swelling secondary to hydrogel contact lens wear American Journal of Optometry and Physiological Optics 65:272–276.
Zantos, S.G., and B.A. Holden 1977 Transient endothelial cell changes soon after wearing soft contact lenses. American Journal of Optometry and Physiological Optics 54:856–861.










