8
Cells of Origin for Lung Cancer
OVERVIEW
The overall goals of this chapter are to estimate the alpha-energy dose to cells in the respiratory tract that give rise to lung cancers and to compare this dose at a particular level of exposure in uranium miners to that in members of the general population. The key question that the committee seeks to address and resolve in this chapter is which cells are specifically at risk for malignant transformation and the ultimate development of a lung cancer. The committee's dosimetry model incorporates the location of target cell nuclei in the epithelium. If this location could be determined precisely or even narrowed to a particular population of lung cells, then more accurate and less uncertain estimates of comparative doses to critical cell populations in different environments could be made. Thus, the committee reviewed evidence relevant to the question of whether all cells of the respiratory tract can be transformed to produce lung cancer or whether the risk is limited to a particular subpopulation such as the basal cells alone or basal cells and secretory cells. Also relevant is the location in terms of airway branching where the lung cancers originate, whether they arise exclusively from the cells in the central respiratory tract, whether they also can arise from cells of the lung periphery, or whether they can develop anywhere within the respiratory tract.
This chapter reviews current concepts and hypotheses of the cellular origins of cancer in general and lung cancer in particular. It considers the current debate about the proliferative capacity of secretory and basal cells and the possible roles of these cells in the origin of human lung cancers. Also considered are
data concerning the sites in the respiratory tract where lung tumors originate, particularly the question of whether they originate exclusively in the central respiratory tract. The evidence on histological types of lung cancers found in uranium miners and in the general population is reviewed for an indication of differences suggesting that different targets in the lung are affected in miners and nonminers. Based on these considerations, the committee sought to identify the cells that are most likely to be the progenitors of lung cancers so that dosimetry studies and modeling can be focused on this population.
To accomplish this goal, this chapter has been organized to review several lines of evidence. The structures of lung and airway epithelia are described, and the organization and function of the lung are considered briefly. The pathologies of major types of lung cancer are reviewed to provide a basis for understanding lung tumor types and their locations within the lung. Ultimately, the goal is to understand the cell type of origin for the tumors and the location of these vulnerable cells within the respiratory tract. Consideration of the cellular origin of lung tumors proceeds along three lines. First, the chapter explores the experimental evidence based on the cells of the lung that have the capacity for proliferation. These data suggest that the basal cells and secretory cells, the proliferative populations of the bronchi, are the probable cells of origin for the tumors. Second, this conclusion is supported by observations that bronchial cells can be propagated in cell culture and can be malignantly transformed. These cell cultures, which are presumed to arise from the proliferative cells within the bronchi, can be transformed by gene transfection and can give rise to a spectrum of cancers that resemble the major varieties of lung cancers. Third, observations of lung cancer in uranium miners show that they do not have a single type or a proportionate distribution of major types of lung cancer discrepant from the general population's. These three types of evidence indicate lung cancer development in the miners is not clearly distinct from that in the general population. Thus, although lung cancer occurs with an unusually high incidence in uranium miners, the process appears to be the same as that which occurs in the general population. For this reason it is presumed that the basic mechanisms of development of lung cancer, for example, the cells of origin and the location of these cells within the respiratory tract, should be the same in the two populations, miners and nonminers. These conclusions can be applied to guide dosimetric models for exposure of the cells at risk to radiation from exposure to radon daughters.
STRUCTURE OF THE LUNG
The lung has the principal function of exchanging gases between the body and the outside air. In its simplest description, the lung contains a conducting tube system composed of bronchi, bronchioles, and alveolar ducts for the flow of gases to and from the gaseous exchange surface in the alveoli.
In the alveoli, gases in the blood can interchange with gases in the alveolar spaces, facilitating the entrance of oxygen into the body and the exit of carbon dioxide. A complex system of blood vessels carries less oxygenated blood coming from the fight side of the heart through progressively smaller divisions until the blood reaches the small capillaries of the alveolar wall, where gas exchange occurs. After leaving these alveolar capillaries, the oxygenated blood passes through progressively larger vessels until it is returned to the left side of the heart for circulation throughout the body. Many systems within the lung support this primary function. The mucociliary apparatus includes mucous glands to produce secretions that protect the lung from microbial agents and mobilize inspired particles. Macrophages provide another defense mechanism. The cartilage and smooth muscle surrounding the bronchi provide mechanical structures that prevent the bronchi from collapsing. Other connective tissue cells and products produce a cohesive structure, yet one which preserves the high degree of elasticity needed for expansion and contraction of the lung to move gases within airways and air spaces.
The structure of the airways and, in particular, their epithelial morphology are very relevant for respiratory carcinogenesis. The epithelium of the bronchi is of pseudostratified columnar structure with ciliated cells, mucous goblet cells, and simpler secretory mucous cells (Morgenroth et al., 1982). The bronchial epithelium also contains basal cells and argentaffin neuroendocrine (Kulchitsky) cells. The Kulchitsky cells, despite producing neuroendocrine secretory products, may originate from the same endodermal precursors as the other cells of the bronchi and bronchioles. Submucosal mucous glands are found only around the bronchi. The bronchiolar epithelium is composed of ciliated cells, goblet cells, and Clara cells. Goblet cells progressively decrease and Clara cells increase in proportion from the proximal to the distal bronchioles. Epithelial thickness decreases from the main bronchi to the terminal bronchioles, although the decrease may not be uniform with progressive branchings of airways (Gastineau et al., 1972). Figure 8-1 illustrates the morphology of bronchial and bronchiolar epithelia. The alveolar epithelium is composed primarily of flat type 1 pneumocytes that line most of the alveolar surface and lesser numbers of larger, rounder type 2 pneumocytes.
The branching of the airways has been discussed previously in Chapter 5. Bronchi are identified by having smooth muscle, cartilage, and mucous glands. The mainstem bronchi begin at the branching of the trachea and divide to form the lobar bronchi. After two additional branchings these give rise to the segmental bronchi. After an average of about eight branchings of bronchi, the transition between bronchi and bronchioles is reached. There are numerous (about eight) additional branchings of the bronchioles before the transitions between bronchioles, alveolar ducts, and alveoli.
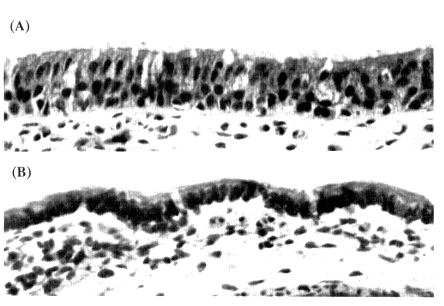
Figure 8-1 (A) Histology of the bronchial epithelium of a normal human lung. (B) Histology of the bronchiolar epithelium of a normal human lung. The bronchial epithelium (A) is organized as a pseudostratified columnar epithelium with ciliated and secretory mucous goblet cells lining the surface. Basal cells are seen occasionally along the basement membrane. Kulchitsky cells with argentaffin granules cannot be identified in this hematoxylin and eosin-stained slide. The bronchiolar epithelium is thinner and varies in organization from pseudostratified columnar to cuboidal epithelium, depending on how peripheral it is in the branching of bronchioles. The surface is lined by ciliated cells and goblet cells and/or Clara cells, depending on the level of branching of the bronchioles. Magnification = × 625. Hematoxylin and eosin.
PATHOLOGY OF LUNG CANCER
Cancer of the lung is the leading cause of cancer death in the United States (U.S. Department of Health and Human Services [DHHS], 1989). For males it has held this ranking for over two decades. It has recently emerged as the leading cause of cancer deaths among women in the United States. The majority of lung cancers have been attributed to the habit of cigarette smoking. The differing times that lung cancer became the preeminent lethal cancer in males and females in the United States relates to the differences in the demographics of cigarette smoking in cohorts of males and females during the past century. Similar observations have been made in other countries. Currently, the number of cigarette smokers is growing in a number of countries, and rising lung cancer rates have been observed or can be expected in the future in these locations.
Malignant tumors might be expected to develop from any of the cell types present in the lung that preserve the capacity to proliferate. In fact, the vast majority of lung cancers are thought to develop from epithelial cells that line the
airways (Spencer, 1977; Dunnill, 1982; Askin and Kaufman, 1985). Although benign and malignant neoplasms may develop from the connective tissues of the lung and tumors of mixed components (mixed tumors) have been reported, these are distinctly uncommon and not typically considered part of the lung cancers referred to here. For the purpose of this review, these are rare biological oddities that will be ignored. The epithelial cells that line the outer surface of the lung and the pleural cavities of the thorax are the source of tumors known as malignant mesotheliomas. Although these tumors are not exceptionally rare, and they do arise from cells that line the lung surface, they also are not typically considered part of the lung cancers referred to here. They are not included in the statistics on bronchogenic lung cancer and are not known to be associated with exposure to radon. These tumors are also excluded in this review.
As noted above, most lung cancers develop from the epithelial cells that line the airways. Lung cancers are also known as bronchogenic carcinomas because of their presumed origin from these airway epithelial lining cells. Lung cancers are a class of tumors that have several typical morphological patterns based on light microscopic examination. The most common morphological types of lung cancers are squamous cell carcinoma, small cell undifferentiated carcinoma, adenocarcinoma, and large cell undifferentiated carcinoma. Bronchioloalveolar cell carcinomas are a less common form of adenocarcinomas which are believed to arise from the cells that line the small peripheral airways (bronchioles) and/or alveolar spaces (Dunnill, 1982). These tumors represent a larger proportion of lung cancers in nonsmokers, in whom adenocarcinomas of all kinds are the most common type of lung tumor (Berg, 1970; DHHS, 1989). Again, as noted above, most types of lung cancers other than bronchogenic cancer are quite rare. For the purpose of this discussion, the four most common types of lung cancers are those listed above (see also Table 8-1). The four leading types of lung cancers have notable differences in their prognoses, response to therapy, and symptoms and signs that lead to their diagnosis. These four major lung cancer types are diagnosed and classified on the basis of histologic features, as determined by conventional light microscopy. The principal characteristics that distinguish the four tumor types are illustrated in Figure 8-2.
Squamous cell or "epidermoid" carcinoma (Figure 8-2A) is the most common form of lung cancer found in American men during the past 50 years. The primary lesion in these cancers is typically located in the central or middle zones of the lung, within the first four branchings of the bronchus, and they generally arise in an area of the bronchus with dysplastic squamous epithelium (Auerbach et al., 1957). Histologically, tumors of this type express characteristics of squamous epithelium. The tumor cells typically contain keratin and grow as if forming squamous surfaces, as in the skin. When these tumors are well differentiated, they form aggregates of keratin known as keratin pearls. Squamous cell tumors may be poorly differentiated with a solid sheet of polygonal cells in a mosaic pattern and may show less evidence of keratinization
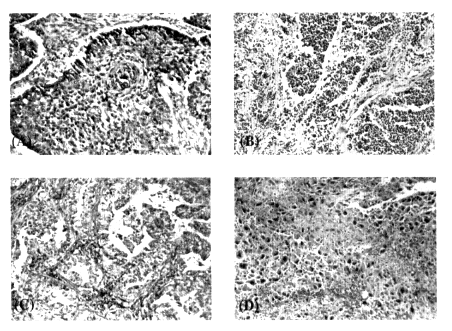
Figure 8-2 Photomicrographs of illustrative examples of each of the four major types of bronchogenic carcinoma. (A) Squamous cell carcinoma. (B) Small cell undifferentiated carcinoma. (C) Adenocarcinoma. (D) Large cell undifferentiated. Magnification = ×150. Hematoxylin and eosin stain.
or of intercellular bridges. These poorly differentiated regions may have central areas of necrosis. Cytologically, all of these tumors show various degrees of abnormal and heterogeneous nuclear structure.
Small cell undifferentiated carcinoma (Figure 8-2B), often referred to as oat cell carcinoma, most commonly presents as a central tumor mass. Unlike other lung cancers, the bulk of the tumor is often found in the lymph nodes and connective tissue at the hilum of the lung. Because the cells of these tumors often are comparatively small and quite homogeneous, and because they often fill the hilar lymph nodes, in the early years of this century they were thought to represent lymphomas. More recently, these tumors have been recognized as primary lung cancers. The tumor masses within the lung, which often are less noticeable than the metastatic secondary lesions, have been recognized as primary lesions. These tumors also arise in the epithelium of the bronchi in their first few branchings. Small cell undifferentiated carcinomas may have a variety of morphological patterns, including the classical comma-shaped oat cell variety, as well as small homogeneous round cells, fusiform cells. and larger intermediate cells. The cells of the tumors are usually very homogeneous, show little or no signs of any pattern of differentiation, and often have extensive
areas of necrosis within the tumors. Mitotic activity is often very extensive, and rapid growth that outstrips blood supply is thought to explain the frequent finding of necrosis within these tumors. All of the morphological forms of small cell undifferentiated carcinoma have poor prognoses and similar responses to therapy.
There are a variety of types of adenocarcinomas of the lung. Two of the less common types of adenocarcinomas include one rather rare tumor that resembles salivary gland neoplasms and is thought to arise from the cells of bronchial glands. A second type, the bronchioloalveolar carcinoma, is found peripherally in the lung and is thought to arise from the cells that line the terminal airways and/or alveoli. These tumors have a unique histological pattern characterized by growth along the alveolar septa, but without destruction of the underlying lung architecture. These less common tumors are not considered further in this review. The majority of adenocarcinomas (Figure 8-2C) are found centrally in the lung and are thought to arise from the bronchial epithelium. The term adenocarcinoma of the lung is usually applied to this type of neoplasm. Histologically, these adenocarcinomas show a variety of secretory or glandular patterns. They have more orderly glandular patterns in better differentiated tumors and are more solid in the less differentiated tumors. They generally contain mucins that are demonstrable with special stains. Because many cancers, particularly adenocarcinomas, metastasize to the lung, it is important to exclude the possibility that adenocarcinomas in the lung represent metastases from another organ. Adenocarcinomas are also thought to arise in scarred lungs, but the pathogenesis of so-called scar carcinomas is controversial. It is unclear in these cases whether the scar contributes to the formation of the tumor or whether the conditions that produce the tumor also produce a scar. Either or both situations may apply in specific cases (Madri and Carter, 1984).
Large cell undifferentiated carcinoma (Figure 8-2D) is the other major type of human epithelial lung cancer. These tumors are also typically localized in the central region of the lung. The major characteristic that has been used to distinguish this type of tumor is the large size of the cells and the almost complete lack of differentiated histological features when viewed by light microscopy. The tumor cells are typically very pleomorphic and have very prominent nucleoli. By light microscopy these tumors lack the cell bridging or nesting characteristic of squamous cell carcinomas and they lack keratin. Some of these tumors with pleomorphic cells and no evidence of glandular differentiation still showed the presence of mucin upon staining with special stains. Giant cell carcinomas of the lung are also thought to be a related lesion.
There have been a number of classification systems developed to distinguish different types of lung cancers, but the system developed by the World Health Organization is the most widely used (World Health Organization, 1982). Despite the availability of criteria, histopathological diagnosis is a subjective process that is affected by the quantity and quality of the specimen provided
TABLE 8-1 Most Common Types of Lung Cancer
|
Common Lung Cancer Type |
Percentage of Total Lung Cancers |
|
Squamous cell carcinoma |
40 |
|
Small cell carcinoma |
20 |
|
Adenocarcinoma |
25 |
|
Large cell carcinoma |
15 |
|
SOURCE: Askin and Kaufman (1985). |
|
for study. Differences in classification of lung cancer histopathology between observers and institutions have been well documented (Stanley and Matthews, 1981; Ives et al., 1983). Reliance on clinical reports unsubstantiated by histological diagnosis may introduce substantial misclassification. Because of observer variability, optimal classification of lung cancer cell types should incorporate a standardized review of original histological material by multiple pathologists using defined criteria. However, even such efforts to reduce errors of diagnosis do not ensure comparability between studies. Therefore, it should be noted that evidence on the distribution of tumor types varies among investigators and studies. This reflects, in part, the criteria used to specify tumor type and the techniques used to characterize the tumor. The most common types of pulmonary carcinomas and their approximate proportionate distributions, as observed in large studies of the general population using widely recognized and applied criteria (Carter and Eggleston, 1981; World Health Organization, 1982), are listed in Table 8-1. Bronchioloalveolar cell carcinomas and mesotheliomas, which occur with lower frequencies, and other rare types of lung cancers have been omitted.
That the above estimates of the frequency of occurrence of the most common lung cancers are reasonable can be seen if one considers them in comparison with another recent study (Percy et al., 1983). The overall proportions of squamous cell carcinoma, small cell undifferentiated carcinoma, adenocarcinoma, and large cell undifferentiated carcinoma are, respectively, about 35, 17, 25, and 9% of all lung cancers in the male population of the United States. If one omits the other 14% of tumors in this study, the proportions become 41, 20, 29, and 10%, respectively, which are similar to the distributions of lung cancer types listed above. In yet another study (Stanley and Matthews, 1981), the four major tumor cell types constituted 97% of the tumors, and the proportions were 38, 28, 18, and 13%, respectively. One of the biggest differences between these studies is the relative proportions of adenocarcinomas and large cell carcinomas, which, as noted above, may reflect the thoroughness of evaluation of the cases in the different studies. Table 8-2 compares the percentage distributions of lung cancers between smokers and nonsmokers. For nonsmokers, the most common type of lung cancer is adenocarcinoma, whereas small cell undifferentiated
TABLE 8-2 Distributions of Lung Cancers Between Smokers and Nonsmokers
|
|
Percentage of Lung Cancers |
|
|
Tumor Type |
Smokers |
Nonsmokers |
|
Squamous cell carcinoma |
47 |
14 |
|
Small cell carcinoma |
17 |
4 |
|
Adenocarcinoma |
10 |
57 |
|
Large cell carcinoma |
17 |
8 |
|
SOURCE: Data are for U.S. males and are from Berg (1970). |
||
carcinoma is very uncommon (Berg, 1970). Therefore, cigarette smoking is an example of an environmental exposure that appears to shift the distribution of histological types of lung cancers.
The information presented above largely concerns the characteristic of lung cancers as determined by light microscopy. The morphology of lung cancers becomes more complicated as these features are explored in greater depth and with more powerful investigative techniques (see next section). Among the examples of such problems are the unusual types of differentiation seen in lung tumors and the presence of characteristics of more than one pathway of differentiation within the same tumor and even within the same cells (Gatter et al., 1985).
The epithelial lining of the lower respiratory tract is not normally composed of squamous cells. Squamous epithelium arises in this location as a consequence of irritation of the normal epithelium. Cigarette smoke, infections, and environmentally and occupationally related inhalants are likely sources of irritation (Frank, 1982). Acute injuries to the respiratory epithelium can cause the loss of ciliated cells and produce a reparative reaction in which the injured area becomes covered by a thin and undifferentiated epithelial cell lining (see below). If the injury is severe, there can be loss of other cellular constituents with marked thinning of the epithelium to one-half or less of its original thickness. The appearance of the epithelial lining under these conditions can be that of a very irregular, cuboidal regenerative cell population with little differentiation. As irritation becomes repetitive or chronic, the resulting change in the airway lining is normally differentiated metaplastic squamous epithelium. This change can give rise to an epithelium with a thickness as much as twice that of the normal respiratory epithelium. The process leading to abnormal, atypical squamous epithelium is less well understood and occurs over a prolonged period of time (Saccomanno et al., 1965). This proposed sequence has been confirmed by the occasional observation of very early (occult) carcinomas of the lung. In such a patient, malignant cells may be found in the sputum on cytologic examination, and bronchoscopy is likely to show a ragged mucosal
surface in one of the subsegmental bronchi with dysplastic squamous epithelium and invasive squamous cell carcinoma contiguously (Auerbach et al., 1979). Squamous carcinoma of the lung, therefore, is presumed not to arise directly from the original respiratory epithelium but rather in areas of injury, repair, and squamous metaplasia and dysplasia that are induced in the bronchial epithelium.
Another issue relevant to dosimetry is the origin of small cell undifferentiated carcinomas and the relationship of these tumors to neuroendocrine cells of the respiratory tract. It is clear that the bronchial epithelium, like many other tissues and organs, contains a small proportion of cells with neuroendocrine secretory granules. These cells, which are most common in fetal life and progressively decrease with advancing age, are readily demonstrated with a Grimelius stain for neurosecretory granules. By electron microscopy, these cells have characteristic membrane-bound cytoplasmic granules that have the appearance of neurosecretory granules. It is believed that these cells can give rise to carcinoid tumors in the lungs and in other tissues. In the lungs, these tumors are usually not malignant. Because these cells have neurosecretory products and some of the same types of peptide hormones are found in small cell undifferentiated carcinomas, it has been suggested that these two types of tumors are related and are even in the same spectrum of tumors (Gould et al., 1978, 1983a,b). In experimental studies in hamsters, it has been reported that exposure to cigarette smoke caused an increase in the number of neuroendocrine cells of the respiratory tract (Tabassian et al., 1989) and that exposure to nitrosamine carcinogens combined with hyperoxia causes the development of neuroendocrine lung tumors (Schuller et al., 1988, 1990). Conversely, others have proposed that the small cell undifferentiated carcinomas arise from transformation of one of the major cell types of the respiratory epithelium (Pfeifer et al., 1989). This issue remains unresolved and requires further study, but one must entertain the possibility that small cell undifferentiated carcinomas may not originate from neuroendocrine cells of the respiratory tract. For example, other lung tumor cell types, particularly squamous cell carcinoma, are seen admixed with small cell carcinoma cells. It is possible that small cell undifferentiated carcinoma represents a more primitive form of these tumors or that these are tumors of a more primitive epithelial cell precursor (e.g., a stem cell). In these tumors of mixed tumor cell types, the clinical course generally reflects the poor prognosis of the small cell undifferentiated cancers.
For large cell undifferentiated carcinomas, the number of these cancers in a clinical series depends on the thoroughness with which cases are investigated and the techniques and criteria chosen to make this distinction (electron microscopy and histochemical staining versus light microscopy alone). When studied extensively, a substantial fraction of these tumors can be reclassified, most as adenocarcinomas and some as squamous cell carcinomas.
This problem of diagnostic specificity is not unique for large cell tumors. When lung cancers of most types are investigated extensively by electron
microscopic, histochemical, immunohistochemical, and molecular techniques, the system of classification described above is found to be too simple (Gatter et al., 1985). Many tumors have features of more than one lung cancer cell type. For example, some squamous tumors contain cells that have mucous granules and even neurosecretory granules in addition to keratins (McDowell et al., 1981). Thus, the separation of different types of tumors by light microscopy may be useful for guiding clinical therapy, but does not necessarily fully segregate the cellular products and structures in various lung cancer types. Different types of lung cancers appear to have a far greater degree of overlap than previously considered. This overlap suggests that various types of lung cancers may arise from cells of origin that are in the same family, perhaps a common bronchial precursor or stem cell (Gatter et al., 1985).
It is also important to consider some past observations and some recent trends in lung cancer epidemiology, since these may influence the types of lung cancers that will emerge in the future and thus may be relevant to radon exposure (DHHS, 1989). Although cigarette smoking is recognized as the leading cause of lung cancer, it is clear that several other environmental factors can influence lung cancer incidence by additive or synergistic interactions. Examples are occupational exposures to asbestos or bis-chloromethyl ether, which, when combined with cigarette smoking, causes a much higher than expected incidence of lung cancer. With increased use of cigarettes by females, lung cancer rates in females have risen over the past decades. The overall decrease in smoking, particularly among males during the past few years, has begun to be reflected in a decreasing lung cancer rate in young cohorts of males in the United States (Devesa et al., 1989). If this trend continues, there may be a general downturn in the overall incidence of lung cancers in the near future. This effect also could reflect the changed characteristics of cigarettes that are smoked now as compared with the characteristics of those consumed 30 to 50 yrs ago. Consistent with this point is the observation that the proportion of squamous cell carcinomas in a population of smokers is related to the number of cigarettes that they smoke each day (Weiss et al., 1977). Another recent trend is an increasing proportion of adenocarcinomas among lung cancer patients (Wu et al., 1986). It remains to be determined whether this change reflects different diagnostic techniques and criteria implemented in recent years, or whether this is a real change that has resulted from different patterns of cigarette smoking.
Observations of the pathology of human lung cancer lead to many unanswered questions concerning the basic mechanisms of the pathogenesis of lung cancers and the relationships between various types of lung cancers. The questions in turn lead to the pursuit of studies that apply the power of scientific investigation and apply modern experimental techniques toward their solution. It is these points that are addressed in the following section.
BASIC CONCEPTS OF LUNG CANCER DEVELOPMENT
Multistep Process of Lung Cancer Development
A body of clinical and experimental evidence supports the concept that the development of cancer is a multistep process (Armitage, 1985). Data from cancer registries show that the incidence of most cancers rises with age (Doll, 1971). The rising incidence of lung cancer with increasing age is consistent with a multistep process (Cross, 1987). The accumulation of multiple genetic alterations is thought to occur as the result of exposures to exogenous carcinogens such as tobacco smoke, together with a background of genetic changes that are believed to occur spontaneously. Clinical evidence also indicates that most cancers pass through premalignant stages prior to the development of overt clinical disease (Doll, 1971). The most thoroughly studied example of this concept is the evolution of squamous cell carcinoma of the uterine cervix through multiple premalignant and malignant steps. That this is a valid interpretation of the pathogenesis is attested to by the reduced incidence of invasive tumors when clinical intervention removes the early lesions. Similarly, a sequence of premalignant lesions of the bronchial epithelium in cigarette smokers has been shown to precede invasive lung cancer (Auerbach et al, 1961).
Compelling evidence in support of the concept that lung cancer development is a multistep process was derived from prospective cytologic studies in which the development of lung cancer in uranium miners was observed to occur over the course of several years (Saccomanno et al., 1964, 1965, 1974). By studying the cytologies of repetitive sputum samples from uranium miners, cytologic changes were found to proceed through a sequence of progressive steps from squamous metaplasia through various degrees of dysplasia, in situ carcinoma, and invasive carcinoma. Subsequently, comparable sequences of epithelial lesions have been found to precede overt cancers in a number of other tissues (Farber, 1984).
A framework for a more complete explanation of cancer as a multistep process has focused on recent developments in understanding the molecular genetics of human colon cancers (Fearon and Vogelstein, 1990). This analysis combines the results of chromosomal alterations detected in colon cancer cells from spontaneous cases and genetic predisposition syndromes and observations of changes in oncogenes and other genes by molecular genetics techniques. In the case of colon cancers, consistent deletions were found in chromosomes 5q, 17p, and 18q, and genetic loci at these deletion sites were incorporated into a model in which the alteration or loss of these normal genes was related to progressive transitions in the natural history of human colon cancers. One of these chromosomal loci (17p) contains the gene identified as p53, which has been identified as a tumor suppressor gene. When both alleles of this gene are
A,B = loss of two alleles of a tumor supressor gene via deletion or mutation
C = point mutation in Ha-ras or Ki-ras or related gene
D = genetic changes in the regulation of growth factor gene or receptor
E = altered regulation of c-myc or N-myc
Figure 8-3 Hypothesis of lung cancer development. Modified from Fearon and Vogelstein (1990).
functioning normally, they impede or prohibit the full neoplastic development of colon cells (and perhaps other cell types as well). When the functioning of the two alleles of this gene are lost by significant mutations of its DNA sequence and/or by partial or complete deletion of the gene, the suppressing effect on tumor development is lost. The identity and functions of the genes at the 5q and 18q loci are less well defined at present, but speculative interpretations have been offered based on available data. In addition to these changes, many colon cancers have been found to have mutated ras genes and hypomethylated DNA. These observations have been combined in a hypothetical genetic model of human lung cancer (Figure 8-3). Although this model concerns human colon cancer, it is reasonable to assume that the path to other human cancers, including lung cancer, is quite similar.
Genetic and Chromosomal Alterations in Lung Cancer
Chromosomal alterations are found in a large proportion of lung cancers. In small cell undifferentiated lung cancers, a high proportion of the cells have amplification of one of the myc genes, usually either N-myc or L-myc, and many have a deletion in the p21-p24 region of chromosome 3 (Waters et al., 1988). The human c-erbA-beta gene has been localized to the 3p21-3p25 region, and at least one copy of it was deleted in all six of the small cell lung cancer cell lines tested (Dobrovic et al., 1988). This gene is believed to specify a DNA-binding protein that has the features of a hormone receptor. As such, it may function as a tumor suppressor gene for small cell carcinomas and perhaps other lung cancers. In other studies of small cell carcinomas, loss of heterozygosity was found at 3p, 13q, and 17p (Harbour et al., 1988). In view of the fact that the retinoblastoma gene is located at 13q14, it was thought that alterations in the structure of this gene might be detected in small cell carcinomas. When a number of primary tumors and cell lines from small cell carcinomas were
tested, a number of structural abnormalities were detected in the retinoblastoma gene in these cells. Thus, the analysis of cytogenetic changes in lung cancers can be used to identify likely sites of recessive tumor suppressor genes that are involved in the malignant transformation of cells.
Weston et al. (1989) examined non-small-cell lung cancers for evidence of consistent DNA sequence deletions. Deletions from chromosomes 3, 11, 13, and 17 were found frequently, but the deletion frequency varied with the locus and tumor type. Deletions were detected by observations of loss of heterozygosity of the tested genetic loci in tumors of patients in comparison with the heterozygous state in uninvolved tissue from the patient. Sequence deletions in chromosome 17p were found in 8 of 9 squamous cell carcinomas but only 2 of 11 adenocarcinomas. Furthermore, in many of the cases of squamous cell carcinoma with 17p deletions, there was an associated chromosome 11 sequence deletion. For other loci, there were similar frequencies of alterations in squamous cell carcinomas and adenocarcinomas. Data for deletions in large cell carcinomas generally resembled those found in adenocarcinomas. These sites of chromosomal alteration are in proximity to the chromosomal location for previously identified tumor suppressor genes (e.g., p53 on 17p). Even though an earlier report had claimed that deletions in chromosome 3 were a consistent change in non-small-cell lung cancer, just as in small cell cancers (Kok et al., 1987), Weston et al. (1989) observed that only about half of the lung tumors showed DNA deletions in chromosome 3. A similar lower rate of allelic loss in the short arm of chromosome 3 was also observed by Rabbits et al. (1989). Therefore, this body of evidence shows that cytogenetic changes are valuable for identifying essential genetic alterations and guiding mechanistic studies of non-small-cell lung cancer.
A long list of alterations in the expression and functioning of oncogenes and growth factors and their receptors has been observed in lung cancers. Overexpression and amplification of genes of the myc family are very common in all forms of lung cancer but particularly notable in small cell lung cancers, which typically have overexpression and amplification of the N-myc and L-myc genes (Waters et al., 1988). In this case the extent of overexpression of these genes is related to the clinical progression of the cancers and is inversely related to prognosis and survival. Nigro et al. (1989) found that mutations in the p53 gene are common in various types of human cancers, including lung cancers. Most of these mutations occur in the portions of the gene that are highly conserved between species and thus likely to code for important functional sites of the p53 protein (gene product).
The c-raf-l and c-myc oncogenes are commonly overexpressed in small cell carcinomas. To evaluate the role of the abnormal expression of these genes in the induction of lung cancer, these two genes were transfected into Simian virus 40 large T antigen-immortalized cultures of human bronchial epithelial cells (Pfeifer et al., 1989). The transfected cells gave rise to malignantly
transformed lung cancer cells with a tumor morphology resembling that of large cell tumors but with evidence of neuroendocrine markers that are suggestive of small cell carcinomas. In fact, Harris and collaborators (Reddel et al., 1988; Pfeifer et al., 1989) have shown that they could malignantly transform human bronchial epithelial cells by transfection with a variety of oncogenes. When the transformed cells were xenotransplanted into nude mice, they gave rise to tumors that reproduced the entire spectrum of human bronchogenic carcinomas. Furthermore, the type of tumor that was generated appeared to relate to the oncogene that had been transfected. From these results it appears that each of the major types of bronchogenic cancers may have a common cell of origin, and also that the morphological type of tumor that results may be determined by the specific kind of genetic change that has occurred during the evolution of the tumor cell from this common precursor cell of origin. However, it is not known whether this common cell of origin is the basal cell, secretory cell, or some other unidentified bronchial cell.
CELLS FROM WHICH LUNG CANCERS DEVELOP
There are two important questions that must be addressed in determining where lung cancers originate. First, which kinds of cells in the human respiratory tract may be the progenitor cells from which lung cancers develop? Second, where in the branching of the respiratory airways do the cancers originate? If these target cells for cancer development, particularly those cancers caused by radon, can be defined, and if their distributions within the lung can be determined, this information could be used in formulating more valid dosimetry models of radon exposure. A refined dosimetry model of this type could specifically focus on the estimated location of the nuclei of these target cells in the airway epithelium (Crawford-Brown, 1987).
The first question is which cells in the human respiratory tract may be the progenitor cells from which lung cancers develop? Which cells are at risk for malignant transformation? Cancer typically occurs in tissues with cells that have a high rate of proliferation or in tissues in which cell proliferation occurs in response to injury. In adult tissues or for cell types in which cell proliferation does not occur, cancer is extremely rare. Thus, chronic irritation or injury was thought to be the etiologic factor for the development of cancer. Subsequently, the role of a variety of specific carcinogenic etiologic agents has come to be recognized, but cell proliferation clearly plays a significant role in the evolution of cancers (Kaufman and Cordeiro-Stone, 1990). The influence of cell proliferation as a contributing factor in the development of cancer presumably results from effects on the mitotic process and on DNA synthesis. Replicating DNA is vulnerable for a variety of reasons. First, replicating DNA is affected to a greater extent by chemical carcinogens than is nonreplicating DNA (Cordeiro-Stone et al., 1982). Second, replication of DNA that contains
carcinogen adducts may cause incorporation of incorrect nucleotides at sites of altered or excised bases. Third, some carcinogens may modify nucleotide precursors, and altered precursors may be incorporated into DNA. Fourth, DNA replication itself occurs with a low, but non-zero error rate. Situations that increase cell replication are likely to cause mutations strictly as the result of these errors. There appears to be a critical interrelationship between the repair and replication of DNA as factors in the etiology of cancer (Kakunaga, 1975; Kaufman and Cordeiro-Stone, 1990). If DNA replication proceeds through a damaged region prior to repair, there is a substantial risk of making errors during replication, which may cause a mutation to occur as the result of an alteration of the base sequence of the complementary DNA strand. A mutation does not occur if the repair of the damage precedes replication. Consequently, the relationship in time between the repair and replication of DNA may be a major determinant of the potential for the occurrence of mutations and also, presumably, of carcinogenesis.
It is presumed that the cells of the lung that are sensitive to radon-induced cancer are those that are not terminally differentiated but still have the capability for division and differentiation (Crawford-Brown, 1987). In the normal central respiratory tract, cell proliferation occurs at a very low rate. The turnover time for epithelial cells of the trachea and bronchi of normal mice and rats has been estimated to average about 3 to 7 weeks or even as long as 14 weeks (Shorter, 1970). In response to injury, cell proliferation rates are greatly increased, with a doubling time of less than 48 h. It was thought that cell renewal resulted from increased proliferation of basal cells of the tracheobronchial tract. Basal cells were thought to be the stem cells of the epithelium, and the progeny of the basal cells were thought to differentiate into the secretory, ciliated, and goblet cells that line the respiratory epithelium.
This belief was challenged by studies that examined the response of tracheal epithelium to mechanical injury (McDowell et al., 1979; Kauffman, 1980; Keenan et al., 1982). It was found that injury caused loss of the epithelial cells, and cells from the margins then migrated into the injured area and reepithelialized the surface. Flattened mucous cells and basal cells migrated over the denuded basement membrane and both types of cells proliferated to generate new cells in situ. McDowell et al. (1979) scraped the epithelium from hamster trachea and observed the regenerative process over the next several days. All cells had sloughed from the scraped area by 2 h, leaving a bare basement membrane. Simple flat cells migrated into the denuded area from adjacent epithelium by 6 to 12 h. By 24 h the defect was covered by a one-to-two-cell-thick layer of squamoid cells. At 48 to 72 h the layer of cells was thicker, with squamoid cells and undifferentiated cells. By 96 h the epithelium was restored and looked normal. This process was shown to involve migration and proliferation of mucous-secretory and basal cells and, initially, diminished differentiation of these cells. It was observed that mucous-secretory cells have
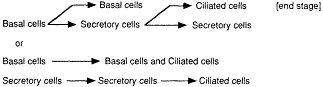
Figure 8-4 Pathways of bronchial cell proliferation and differentiation.
a greater proliferative potential than do basal cells during this regenerative process (Keenan et al., 1982). Furthermore, mucous-secretory cells give rise to both squamous and mucous cells. Ciliated cells form from mucous cell precursors via a longer maturation process (Keenan et al., 1982). In these areas following cell proliferation and migration, they often found squamous metaplasia. Therefore, they concluded that squamous metaplasia can arise from mucous cells as well as basal cells. Presumably, both cell types can undergo differentiation to either mucous or squamous cells. This range of differentiation for these cells was thought to explain the occurrence of human lung tumors with combined squamous and glandular differentiation (McDowell et al., 1978).
The idea that basal cells were not the only precursor of the airway epithelium was further strengthened by the results of studies in which isolated rat airway epithelial cells were used to repopulate denuded tracheal grafts. Nettesheim et al. (1990) showed that basal cells from the bronchi and Clara cells from the bronchioles are distinctly different stem cells. On repopulation of denuded tracheas, isolated Clara cells give rise to a cuboidal epithelium with only Clara cells and ciliated cells, such as are found in bronchioles. Isolated tracheal basal cells give rise to higher pseudostratified epithelium with the typical tracheobronchial cell constituents. Secretory cells were able to give rise to the same mixed population as basal cells (Nettesheim et al., 1990). In another study isolated rat basal cells and secretory cells were used separately to repopulate denuded tracheal grafts (Johnson and Hubbs, 1990). In contrast to the results of Nettesheim et al. (1990), basal cells gave rise to an epithelium of only basal and ciliated cells. Secretory cells gave rise to an epithelium of secretory, basal, and ciliated cells. This observation suggests that the secretory cell is the major progenitor for the bronchial epithelium and that basal cells may have a limited differentiation capacity (Johnson and Hubbs, 1990).
Results of these studies demonstrated that the basal and secretory cells throughout the respiratory epithelium have the capacity for cell proliferation. In fact, the vast majority of cell proliferation may occur in the secretory cells. This suggests two pathways for respiratory tract cell proliferation and cell renewal (Figure 8-4).
Thus, not only basal cells but also secretory cells of the tracheobronchial epithelium can proliferate. Therefore, all cells in the respiratory epithelium
except ciliated cells appear to have a proliferative capacity. Presumably, the same proliferative capacity is held by both basal and secretory cells in the human respiratory tract. Stimulation of cell proliferation is presumed to have an important role in lung cancer development in humans. It is plausible that injuries from infectious, toxic, or carcinogenic agents may stimulate cell proliferation, and that this proliferation may be critical for malignant transformation. Important genetic damage may occur during tissue repair. Therefore, mucosal areas with sustained increases in cell proliferation, such as focal metaplastic lesions, may be at particular risk. This kind of damage and stimulated cell division in bronchial epithelium is produced by cigarette smoke and may also be produced by exposure to radon daughters.
In humans, various etiological agents cause necrosis and shedding of the tracheobronchial epithelium and lead to acute and chronic forms of bronchitis (Spencer, 1977). As in experimental animals, repair of the denuded basement membrane involves an initial covering of the surface by a simple, squamoid epithelial layer, which is presumably composed of basal cells and undifferentiated secretory cells. This surface lining is often one-half (or less) than the thickness of the normal epithelium. If the injury to the bronchial epithelium is chronic or recurrent, squamous metaplasia often results. Such metaplastic lesions can be twice the thickness of the normal bronchial epithelium. Because cell proliferation may be particularly high, these mucosal lesions may be extremely vulnerable sites for the development of lung cancer.
The cell of origin for small cell undifferentiated carcinoma (oat cell lung cancers) is more obscure and remains a topic of debate. It is uncertain whether these tumors arise from typical airway epithelial cells or whether they are derived from neuroendocrine cells. It has been suggested that small cell and squamous cell cancers have the same cell of origin and that the differences in phenotypes between the two types of tumors result from different genetic changes (see the preceding section). Another possibility is that small cell tumors develop from the common precursor cell when the precursor cells are exposed to carcinogenic agents at exceptionally high rates.
Kreyberg and others thought that small cell carcinomas were undifferentiated squamous cell carcinomas (Dunnill, 1982). Because of cytological similarities between bronchial carcinoids and small cell undifferentiated lung cancers, opinion has evolved to regard them as the benign and malignant ends of a spectrum of neuroendocrine lung tumors (Gould et al., 1978, 1983a,b). Bronchial carcinoid tumors presumably originate from neuroendocrine Kulchitsky cells of the airway epithelium; therefore, it was thought that the small cell tumors also arose from these cells. The presence of neouroendocrine secretory granules in some small cell carcinomas was one part of the evidence suggesting that small cell undifferentiated carcinomas were neuroendocrine tumors. Godwin and Brown (1977) doubted that carcinoid and small cell undifferentiated
carcinoma shared a common precursor cell of origin. They noted that carcinoids and small cell carcinomas have very different epidemiologies. Small cell tumors have a marked predominance in males, and they are strongly linked to cigarette smoking and to occupational exposures; the epidemiology of bronchial carcinoids is not consistent. In fact, it has been suggested (Masse and Cross, 1989) that bronchogenic carcinomas of the four major cell types may all arise from the same endodermal progenitor cells. Secretory cells are the most likely progenitor cells for all of the bronchogenic tumor types, although basal cells may also have a role. From this perspective, neurosecretory cells may not be the origin of small cell undifferentiated carcinomas.
Lung cancers often have mixtures of histological patterns (McDowell et al., 1978; Cotran et al., 1989). Most commonly, small cell undifferentiated and squamous cell patterns or squamous cell and adenocarcinoma patterns are found together. Based on these observations, it has been suggested that all histological variants of bronchogenic carcinomas (small cell undifferentiated carcinoma, squamous cell carcinoma, adenocarcinoma, and large cell undifferentiated carcinoma) have a common origin in the endodermal cells that make up the respiratory epithelium. From this perspective, it is possible that all of the histological types of bronchogenic carcinoma represent a phenotypic (and genetic) spectrum of malignant disease derived from a common cell of origin.
The second question is where in the branching of the respiratory airways do the cancers develop? This is another topic that has been the subject of much debate. There are about eight branchings of large airways (bronchi) and about five branchings of bronchioles (James, 1988). Some believe that most bronchogenic carcinomas arise near the hilus of the lung (Ellett and Nelson, 1985; Cotran et al., 1989; Bair, 1990). In fact, it has been suggested that 75% originate from first-, second-, or third-order bronchi, and most of the remainder arise in more distal bronchi (Cotran et al., 1989). If one accepts this position, few lung cancers arise peripherally, and those that do are largely adenocarcinomas. There are several forms of adenocarcinoma, and this diversity adds to the uncertainty about the site of origin for adenocarcinomas. Acinar adenocarcinomas are thought to arise in segmental or subsegmental bronchi (Gibbs and Seal, 1984) and to develop from bronchial surface epithelium or bronchial mucous glands (Dunnill, 1982; Kodama et al., 1982). In contrast, there seems to be a general consensus that the bronchioloalveolar cell form of adenocarcinoma arises from peripheral lung cells, but it is still unclear to what extent alveolar type II cells and bronchiolar Clara cells are the cells of origin.
Spencer's (1977) views concerning the site of origin of lung cancers within the lung are widely cited. He noted that estimates of the proportion of lung tumors that are peripheral in origin vary because of the methods used to determine location (X ray or tissue specimen, or other approaches) and the patient population studied (specimens from surgical patients versus autopsy specimens). His estimate was that 50 to 60% of lung cancers were
peripheral, where peripheral implied small distal bronchi and bronchioles. By this accounting, 40% of the lung cancers were more proximal than the segmental bronchi. In contrast, in large studies of autopsy cases, Bryson and Spencer (1951) found that only 2.8% were peripheral tumors and Strang and Simpson (1953) found that 8% were peripheral. Walter and Pryce (1955) noted that 50% of resected lung cancers arose from central bronchi and 50% arose from smaller bronchi or bronchioles. Since these cancers were thought to be resectable clinically, they probably represent a selected subpopulation of all lung cancers.
Bronchial basal cell hyperplasia and squamous metaplasia are typically antecedents to dysplastic proliferative lesions and occur in areas near bronchogenic cancers of various histologic types (Dunnill, 1982). Squamous metaplasia, dysplasia, and carcinoma in situ of the bronchus are usually multifocal and commonly occur in the central bronchi prior to the appearance of lung cancers (Spencer, 1977). Metaplasia, dysplasia, and carcinoma in situ are most common at sites of bronchial bifurcations. Auerbach et al. (1957) found widespread carcinoma in situ and early invasive carcinoma in the central respiratory tract of about 90% of patients with invasive carcinomas. Similarly, Valentine (1957) found extensive squamous metaplasia in the bronchi of patients with lung cancer. The occurrence of hyperplastic, metaplastic, and dysplastic lesions as well as localized cancers in major bronchi strongly argues for the importance of the central region of the respiratory tract as the site of origin for most lung cancer development.
To evaluate the risk for development of lung cancer, it is necessary to consider more than the type of cell at risk and its location within the respiratory tract. Factors that influence the risk for lung cancer development in uranium miners exposed to radon at work must be considered, and this must be compared with the effects of radon in the average population at home. In both populations the risk may depend greatly on the rate of cell proliferation in the respiratory tract cells and the effects of exposure to radon at different dose levels and to cigarette smoke. In nonsmokers the rate of turnover of tracheobronchial cells may be very low, but it is increased periodically during tissue repair following respiratory tract infections or occasional exposures to inhaled pollutants. The rates of cell turnover in the respiratory tract of individuals chronically exposed to agents that injure and kill cells are higher. The increase in proliferation rates will depend on the types and amounts of injurious agents to which they are exposed. Tobacco smoke is certainly the leading injurious agent; radon and other inhaled agents can also produce respiratory injuries. Furthermore, various exposures can potentiate each other. In the case of chronic pulmonary injuries, the reparative process may produce alterations in pulmonary structure (e.g., brochiectasis and emphysema) that adversely effect pulmonary function, and this in turn may exacerbate the toxicity of the inciting agent by impeding clearance mechanisms or slowing repair. The extent of exposure to radon,
tobacco smoke, and other injurious agents, the chronicity of this exposure, and the presence of chronic changes of pulmonary structure and function can affect cell proliferation rates in respiratory epithelia and perhaps have a major effect on the risk of lung cancer.
In conclusion, bronchogenic cancers arise predominantly in the central respiratory tract from epithelial cells with the capacity for proliferation. Secretory cells have the capacity for proliferation and squamous and mucous cell differentiation. Therefore, these cells, not just basal cells, throughout the central respiratory tract must be included in dosimetry calculations (James, 1988). Dosimetry cannot be focused on basal cells alone, but the dose must be averaged over the whole central bronchial epithelium.
TYPES AND INCIDENCE OF LUNG CANCER IN URANIUM MINERS
Next consider the incidence and types of lung cancer in uranium miners and compare these observations with lung cancer in the general population, in people who are not miners but who are exposed to radon in their homes. As reviewed below, there are no clear differences between the lung cancers in uranium miners and lung cancers that develop in the general population. Therefore, the cell of origin and the location of lung cancers in the general population and in uranium miners are presumed to be the same.
The issue of the percentage distribution of major types of lung cancer in uranium and other underground miners exposed to radon daughters has been reviewed thoroughly in a previous report (NRC, 1988). Samet (1989) reviewed nearly 20 studies of the relationship between exposure to radon and its decay products and the risk of lung cancer. Several studies showed an excess of small cell cancers compared with lung cancers in the general population. Cancers observed in Schneeberg miners were identified as lymphosarcomas, but probably were small cell undifferentiated lung cancers. Tumors found later in autopsies of miners from the adjacent Joachimshal region had a preponderance of small cell undifferentiated carcinomas. An excess of small cell undifferentiated carcinomas of the lung have also been reported among more recent uranium miners from Canada and New Mexico and iron ore miners from Sweden and Great Britain who were exposed to radon daughters (NRC, 1988). Nearly all of these miners were also cigarette smokers.
The most extensive studies of radon daughter exposure and lung cancer were those by Saccomanno and associates on the uranium miners of the Colorado Plateau (Saccomanno et al., 1971, 1974, 1986, 1988, 1989). The studies considered a population of 16,000 uranium miners from 1957 to 1987 and provided extensive data about the types of lung cancer that resulted from radon daughter exposure in comparison to the estimated total radon exposure. There were 383 cases of lung cancers in this population: 356 occurred in smokers, and 25 were nonsmokers (2 cases were unknown). Estimates of radon exposure
levels were available for 82.5% of cases. The majority of lung cancers found in this population were small cell undifferentiated carcinomas. Whereas small cell tumors constituted as much as 76% of lung cancers in this population in 1964, it had declined to 22% by the late 1970s. During this time the proportion of squamous cell carcinomas increased to make up the difference. The small cell tumor excess found earlier (Saccomanno et al., 1989) occurred in smoking miners who had been exposed to very high levels of radon (> 1,200 working level months [WLM]). Overall, there is very little difference now in the histology of lung cancers in uranium miners compared with that in the general population (Saccomanno et al., 1989). Although three-fourths of the miners were smokers, more than 90% of the lung cancers were in smokers. They interpreted this as evidence of a synergistic interaction between the effects of cigarette smoking and radon exposure.
Although data for nonsmoking uranium miners are sparse, there is no exceptional association between uranium mining and small cell cancers in non-smoking miners. In fact, the cell type distribution in nonsmoking uranium miners was comparable to the distribution observed in the general population (NRC, 1988). Small cell carcinomas did not predominate in the 21 cases of lung cancer occurring in Navajo uranium miners. Seven of these cancers were small cell carcinomas, eight were squamous cell carcinomas, four were adenocarcinomas, and two were large cell carcinomas. While small cell carcinomas was not the predominant cell type in this series, the proportion of subjects with this cell type (33%) was greater than that expected from the distribution of lung cancer histopathology in non smokers. Saccomanno et al. (1989) reviewed the 383 cases of lung cancer to characterize the histological type of the lung cancers in smoking and nonsmoking miners (Table 8-3). The number of tumors in nonsmokers was small, but they interpreted the distribution of tumor types as not dramatically different between smokers and nonsmokers. Slightly less than half of the cases of lung cancer in nonsmoking uranium miners from the Colorado Plateau region were small cell carcinomas, while the remainder were of other cell types. All the cases of lung cancer in the nonsmoking miners were in miners exposed to high aggregate radon doses (>301 WLM) (Saccomanno et al., 1988).
These considerations should guide interpretation of the cancer risks of exposure to radon daughters in uranium miners, who generally also smoke cigarettes, as the risk in the overall population who do not mine uranium but who may be exposed to radon daughters at much lower levels is contemplated. In this regard, it is clear that the bulk of U.S. uranium miners who developed lung cancers in previous years were exposed at vastly higher rates than are typical for the general environment or even homes with the highest ambient radon levels. The rate of radon daughter exposure in miners has decreased progressively over a number of years. Most of the U.S. uranium miners were also smokers.
TABLE 8-3 Percentage Distribution of Major Lung Cancer Types Among Miners
|
|
Uranium Miners |
||
|
Tumor Type |
U.S. Males |
Smokers |
Nonsmokers |
|
Squamous cell carcinoma |
35 |
36 |
40 |
|
Small cell carcinoma |
17 |
32 |
24 |
|
Adenocarcinoma |
25 |
13 |
12 |
|
Large cell carcinoma |
9 |
12 |
20 |
|
SOURCE: Cothem (1990). |
|||
In summary, the proportions of lung cancers of different cell types that are induced by cigarette smoking or by uranium mining or the combination are essentially the same when viewed over a lifetime (Cross, 1987; Cothem, 1990). An excess of small cell undifferentiated carcinomas was seen in U.S. uranium miners, particularly those that smoked cigarettes, in early years of epidemiological observation. Presumably, small cell carcinoma occurs earlier as a result of smoking and exposure to radon daughters. While small cell carcinomas were the first to appear, all forms of lung cancer were increased by exposure to radon daughters (Cross, 1987). The tumors that appeared after a longer latency were predominantly squamous cell tumors. As time has gone by, the proportions of the various types of lung cancers among uranium miners has come to resemble the distribution of lung cancers observed in the general population that largely reflect the consequences of cigarette smoking. Since there are no extreme differences between the lung cancers that develop in uranium miners compared with the lung cancers that develop in the general population, it is presumed that the cells of origin and the locations of origin of cancers within the lungs of the general population and uranium miners are the same. Adenocarcinomas in nonsmokers may be a possible exception as it appears to be less frequent for nonsmoking miners than for nonsmokers in the general population (c.f. Tables 8-2 and 8-3). Future data from nonsmoking miners who die at older ages may provide further information.
DISCUSSION
Lung cancer has been described here as a disease that develops as the result of a process that involves multiple steps and that usually proceeds over many years. This interpretation is derived from observations made over many years in high-risk populations, particularly uranium miners, most of whom smoked cigarettes. This view of lung cancer as a multistep process has led to efforts
to define the multiple individual steps. This has met with much success in the past few years as the result of studies of tumor cell cytogenetics and genetic changes in oncogenes and other genetic loci. As the consistencies increase in the catalog of changes found in lung cancers, the pattern of critical genetic changes in these tumors begins to emerge. Concurrent with these advances, there has been an advance in understanding of the growth and differentiation potential of cells of the respiratory tract. It is recognized that basal cells are not the only proliferative cells in the central respiratory tract and that the cells with mucous differentiation have the capacity to proliferate and, as such, may be cells at risk for malignant transformation.
With regard to the identification of the critical target cell population in the lungs that may be the progenitor for human lung cancers, there are several points that can be made. The vast majority of the lung cancers that are seen in smokers and uranium miners develop in the central respiratory tract. They have been estimated to arise largely in the first three to five branchings of the bronchi; to add a margin of safety to exposure estimates, it might be useful to consider exposures that occur in the bronchi through eight branchings. This appears to exclude the lung periphery, which is thought to be the site of origin of bronchioloalveolar cell tumors; these tumors are relatively uncommon in the U.S. population. Whereas peripheral lung adenocarcinomas and even small cell carcinomas may arise in the peripheral lung of rodents, these appear to be very uncommon in humans, presumably based on patterns of exposure and the cell populations in the human respiratory tract at that level. Since adenocarcinomas are, if anything, underrepresented among the lung cancers in uranium miners, it is likely that effects of radon on bronchial gland cells are minimal. The observation that the secretory cells of the central airways can proliferate changes the way that the central respiratory tract must be viewed with regard to the cells at risk for transformation. It appears that the majority of the cells in the central bronchial epithelium, and not just the basal cells, could undergo transformation. Although the number of lung cancers in nonsmoking uranium miners is small, there are no strong differences between the proportions of lung cancers of different histologic types in smoking miners, nonsmoking miners, and the general population. The uranium miners have had a small relative increase in small cell lung tumors and a relative paucity of adenocarcinomas. Consequently, it is unlikely from the available data that the tumors induced in the lungs by low-level ambient exposures to radon differ greatly in type or location from those found in miners. This indicates that exposure modeling should focus on the basal and secretory cells of the bronchial epithelium in the first five (or eight) branchings of the bronchi as the likely target cells for lung cancer development.
REFERENCES
Armitage, P. 1985. Multistage models of carcinogenesis. Environ. Health Perspect. 63:195-201.
Askin, F. B., and D. G. Kaufman. 1985. Histomorphology of human lung cancer. Pp. 17-21 in Carcinogenesis, A Comprehensive Survey. Vol. 8, Cancer of the Respiratory Tract: Predisposing Factors, M. J. Mass, D. G. Kaufman, J. M. Siegfried, V. E. Steele, and S. Nesnow, eds. New York: Raven Press.
Auerbach, O, J. B. Gere, J. M. Pawlowski, G. E. Muehsem, H. J. Smolin, and A. P. Stout. 1957. Carcinoma-in-situ and early invasive carcinoma occurring in the tracheobronchial trees in cases of bronchial carcinoma. J. Thoracic Surg. 34:298-309.
Auerbach, O., A. P. Stout, E. C. Hammond, and L. Garfinkel. 1961. Changes in bronchial epithelium in relation to cigarette smoking and in relation to lung cancer. N. Engl. J. Med. 265:253-267.
Auerbach, O., E. C. Hammond, and L. Garfinkel. 1979. Changes in bronchial epithelium in relationship to cigarette smoking, 1955-1960 vs. 1970-1977. N. Engl. J. Med. 300:381-385.
Bair, W. J. 1990. Overview of ICRP Respiratory Tract Model. Radiat. Prot. Dosim., in press.
Berg, J. 1970. Epidemiology of the different histologic types of lung cancer. Pp. 93-104 in Morphology of Experimental Respiratory Carcinogenesis, P. Nettesheim, M. G. Hanna, and J. W. Deatherage, eds. Oak Ridge, Tenn.: U.S. Atomic Energy Commission.
Bryson, C. C., and H. Spencer. 1951. Carcinoma of the bronchus: A clinical and pathological survey of 866 cases. Qu. J. Med. 20:173-188.
Carter, D, and J. C. Eggleston. 1981. Tumors of the Lower Respiratory Tract. Atlas of Tumor Pathology. Second Series. Fascicle 17. Washington, D.C.: Armed Forces Institute of Pathology.
Cordeiro-Stone, M., M. D. Topal, and D. G. Kaufman. 1982. DNA in proximity to the site of replication is more alkylated than other nuclear DNA in s Phase 10T1/2 cells treated with N-methyl-N-nitrosourea. Carcinogenesis 3:1119-1127.
Cothern, C. R. 1990. Indoor air radon. Rev. Environ. Contam. Toxicol. 111:1-60.
Cotran, R. S., V. Kumar, and S. L Robbins. 1989. Pp. 797-810 in Pathologic Basis of Disease. Philadelphia: W. B. Saunders Co.
Crawford-Brown, D. J. 1987. Dosimetry. Pp. 173-213 in Environmental Radon, C. R. Cothern and J. E. Smith, eds. New York: Plenum Press.
Cross, F. T. 1987. Health effects. Pp. 215-248 in Environmental Radon, C. R. Cothern and J. E. Smith, eds. New York: Plenum Press.
Devesa, S. S., W. J. Blot, and J. F. Fraumeni, Jr. 1989. Declining lung cancer rates among young men and women in the United States: A cohort analysis. J. Natl. Cancer Inst. 81:1568-1571.
Dobrovic, A., B. Houle, A. Belouchi, and W. E. C. Bradley. 1988. erb-A-related sequence coding for DNA-binding hormone receptor localized to chromosome 3p21-3p25 and deleted in small cell lung carcinoma. Cancer Res. 48:682-685.
Doll, R. 1971. The age distribution of cancer: Implications for models of carcinogenesis. J. R. Soc. Med. 134:133-166.
Dunnill, M. S. 1982. Pp. 293-334 in Pulmonary Pathology. New York: Churchill Livingstone.
Ellett, W. H., and N. S. Nelson. 1985. Epidemiology and risk assessment: Testing models for radon-induced lung cancer. Pp. 79-107 in Indoor Air and Human Health, R. B. Gammage and S. V. Kaye, eds. Chelsea, Mich. : Lewis Publishers, Inc.
Farber, E. 1984. Chemical carcinogenesis: A current biological perspective. Carcinogenesis 5:1-5.
Fearon, E. R., and B. Vogelstein. 1990. A genetic model for colorectal tumorigenesis. Cell 61:759-767.
Frank, A. L. 1982. The epidemiology and etiology of lung cancer. Clin. Chest Med. 3:219-228.
Gastineau, R. M., P. J. Walsh, and N. Underwood. 1972. Thickness of the bronchial epithelium with relation to exposure to radon. Health Phys. 23:857-860.
Gatter, K. C., M. S. Dunnill, K. A. F. Pulford, A. Heryet, and D. Y. Mason. 1985. Human lung tumours: A correlation of antigenic profile with histological type. Histopathology 9:805-823.
Gibbs, A. R., and R. M. E. Seal. 1984. The histological varieties of bronchial carcinoma. Pp. 129-145 in Bronchial Carcinoma, M. Bates, ed. Berlin: Springer-Verlag.
Godwin J. D., and C. C. Brown. 1977. Comparative epidemiology of carcinoid and oat-cell tumours of the lung. Cancer 40:1671-1673.
Gould, V. E., A. D. Yannopoulos, S. C. Sommers, and J. A. Terzakis. 1978. Neuroendocrine cells in dysplastic bronchi. Am. J. Pathol. 90:49-56.
Gould, V. E., R. I. Linnoila, V. A. Memoli, and W. H. Warren. 1983a. Neuroendocrine cells and neuroendocrine neoplasms of the lung. Pathol. Annu. 18:287-330.
Gould, V. E., R. I. Linnoila, V. A. Memoli, and W. H. Warren. 1983b. Neuroendocrine components of the bronchopulmonary tract: Hyperplasia, dysplasia and neoplasms. Lab. Invest. 49:519-537.
Harbour, J. W., et al. 1988. Abnormalities in structure and expression of the human retinoblastoma gene in SCLC. Science 241:353-357.
Ives, J. C., P. A. Buffler, and S. D. Greenberg. 1983. Environmental associations and histopathologic patterns of carcinoma of the lung: The challenge and dilemma in epidemiologic studies. Am. Rev. Respir. Dis. 128:195-209.
James, A. C. 1988. Lung dosimetry. Pp. 259-309 in Radon and Its Decay Products in Indoor Air, W. W. Nazaroff and A. V. Nero, eds. New York: John Wiley & Sons.
Johnson, N. F., and A. F. Hubbs. 1990. Epithelial progenitor cells in the rat trachea. Am. J. Resp. Cell Molec. Biol., in press.
Kakunaga, T. 1975. The role of cell division in the malignant transformation of mouse cells treated with 3-methylcholanthrene. Cancer Res. 35:1637-1642.
Kauffman, S. L. 1980. Cell proliferation in the mammalian lung. Int. Rev. Exp. Pathol. 22:131-191.
Kaufman, D. G., and M. Cordeiro-Stone. 1990. The roles of cell proliferation and gene replication in neoplastic transformation. Pp. 143-151 in Growth Regulation and Carcinogenesis, W. R. Paukovits, ed. Boca Raton, Fla.: CRC Press.
Keenan, K. P., J. W. Combs, and E. M. McDowell. 1982. Regeneration of hamster tracheal epithelium after mechanical injury. Cell Pathol. 41:231-252.
Kodama, T., Y. Shimosato, and T. Kameya. 1982. Histology and ultrastructure of bronchogenic and bronchial gland adenocarcinoma (including adenoid cystic and mucoepidermoid carcinomas) in relation to histogenesis. Pp. 147-166 in Morphogenesis of Lung Cancer, Y. Shimasoto, M. R. Melamed, and P. Nettesheim, eds. Boca Raton, Fla.: CRC Press.
Kok, K., et al. 1987. Deletions of a DNA sequence at the chromosomal region 3p21 in all major types of lung cancer. Nature 330:578-581.
Madri, J. C., and D. Carter. 1984. Scar cancers of the lung: Origin and significance. Hum. Pathol. 15:625-631.
Masse, R., and F. T. Cross. 1989. Risk considerations related to lung modeling. Health Phys. 57(Suppl. 1):283-289.
McDowell, E. M., P. J. Becci, L. A. Barrett, and B. F. Trump. 1978. Morphogenesis and classification of lung cancer. Pp. 445-519 in Pathogenesis and Therapy of Lung Cancer, C. C. Harris, ed. New York: Marcel Dekker.
McDowell, E. M., P. J. Becci, W. Schurch, and B. F. Trump. 1979. The respiratory epithelium. VII. Epidermoid metaplasia of hamster tracheal epithelium during regeneration following mechanical injury. J. Natl. Cancer Inst. 62:995-1008.
McDowell, E. M., T. S. Wilson, and B. F. Trump. 1981. Atypical endocrine tumors of the lung. Arch. Pathol. Lab. Med. 105:20-28.
Morgenroth, K., M. T. Newhouse, and D. Nolte. 1982. Pp. 9-96 in Atlas of Pulmonary Pathology. London: Butterworth Scientific.
National Research Council (NRC). 1988. Pp. 445-488 in Health Risks of Radon and Other Internally Deposited Alpha-Emitters. Washington, D.C.: National Academy Press.
Nettesheim, P., et al. 1990. The role of clara cells and basal cells as epithelial stem cells of the conducting airways. Pp. 99-111 in Biology, Toxicology and Carcinogenesis of Respiratory Epithelium, D. G. Thomassen and P. Nettesheim, eds. Washington, D.C.: Hemisphere Publishing Corp.
Nigro, J. M., et al. 1989. Mutations in the p53 gene occur in diverse human tumour types. Nature 342:705-708.
Percy, C., J. W. Horm, and T. E. Goffman. 1983. Trends in histologic types of lung cancer, SEER, 1973-1981. Pp. 153-159 in Lung Cancer: Causes and Prevention, M. Mizell and P. Correa, eds. Deerfield Beach, Fla.: Verlag Chemie International.
Pfeifer, A. M. A., G. E. Mark, L. Malan-Shibley, S. Graziano, P. Amstad, and C. C. Harris. 1989. Cooperation of c-raf-1 and c-myc protooncogenes in the neoplastic transformation of simian virus 40 large tumor antigen-immortalized human bronchial epithelial cells. Proc. Natl. Acad. Sci. USA 86:10075-10079.
Rabbits, P., et al. 1989. Frequency and extent of allelic loss in the short arm of chromosome 3 in nonsmall-cell lung cancer. Genes, Chromosomes, and Cancer 1:95-105.
Reddel, R., et al. 1988. Human bronchial epithelial cells neoplastically transformed by v-Ki-ras: Altered response to inducers of terminal squamous differentiation. Oncogene Res. 3:401-408.
Saccomanno, G., V. E. Archer, R. P. Saunders, L. A. James, and P. A. Bechler. 1964. Lung cancer of uranium miners on the Colorado Plateau. Health Phys. 10:1195-1201.
Saccomanno, G., R. P. Saunders, V. E. Archer, O. Auerbach, M. Kuschner, and P. A. Bechler. 1965. Cancer of the lung: The cytology of sputum prior to the development of carcinoma. Acta Cytol. 9:413-423.
Saccomanno, G., V. Archer, O. Auerbach, M. Kuschner, R. P. Saunders, and M. G. Klein 1971. Histologic types of lung cancer among uranium miners. Cancer 27:515-523.
Saccomanno, G., V. E. Archer, O. Auerbach, R. P. Saunders, and L. M. Brennan. 1974. Development of carcinoma of the lung as reflected in exfoliated cells. Cancer 33:256-270.
Saccomanno, G., C. Yale, W. Dixon, O. Auerbach, and G. C. Huth. 1986. An epidemiological analysis of the relationship between exposure to Rn progeny,
smoking and bronchogenic carcinoma in the U-mining population of the Colorado Plateau
—1960-1980. Health Phys. 50:605-618.Saccomanno, G., G. C. Huth, O. Auerbach, and M. Kuschner. 1988. Relationship of radioactive radon daughters and cigarette smoking in the genesis of lung cancer in uranium miners. Cancer 62:1402-1408.
Saccomanno, G., G. C. Huth, O. Auerbach, and M. Kuschner. 1989. The histology of neoplasia in uranium miners with smoking and radon exposure evaluation. Pp. 53-62 in Proceedings of the 24th Annual Meeting, N. H. Harley, ed. Bethesda, Md.: National Council on Radiation Protection and Measurements.
Samet, J. M. 1989. Radon and lung cancer. J. Natl. Cancer Inst. 81:745-757.
Schuller, H. M., K. L. Becker, and H. P. Witschi. 1988. An animal model for neuroendocrine lung cancer. Carcinogenesis 9:293-296.
Schuller, H. M., H. P. Witschi, et al. 1990. Pathobiology of lung tumors induced in hamsters by 4-(methylnitros-amino)-l-(3-pyridyl)-l-butanone and the modulating effect of hyperoxia. Cancer Res. 50:1960-1965.
Shorter, R. G. 1970. Cell kinetics of respiratory tissues, both normal and stimulated. Pp. 45-61 in Morphology of Experimental Respiratory Carcinogenesis, P. Nettesheim, M. G. Hanna, and J. W. Deatherage, eds. Oak Ridge, Tenn.: U.S. Atomic Energy Commission.
Spencer, H. 1977. Pp. 115-149, 773-860 in Pathology of the Lung. Oxford: Pergamon Press.
Stanley, K. E., and M. J. Matthews. 1981. Analysis of a pathology review of patients with lung tumors. J. Natl. Cancer Inst. 66:989-992.
Strang, C. and J. A. Simpson. 1953. Carcinomatous abscess of lung. Thorax 8:11-28.
Tabassian, A. R., E. S. Nylen, R. I. Linnoila, R. H. Snider, M. M. Cassidy, and K. L. Becker. 1989. Stimulation of hamster pulmonary neuroendocrine cells and associated peptides by repeated exposure to cigarette smoke. Am. Rev. Resp. Dis. 140:436-440.
U.S. Department of Health and Human Services (DHHS). 1989. A Report of the Surgeon General. Reducing the Health Consequences of Smoking, 25 Years of Progress. Rockville, Md.: U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control, Office of Smoking and Health.
Valentine, E. H. 1957. Squamous metaplasia of the bronchus. Cancer 10:272-279.
Walter, J. B., and D. M. Pryce. 1955. The site of origin of lung cancer and its relation to histologic type. Thorax 10:117-126.
Waters, J. J., J. M. Ibson, P. R. Twentyman, N. M. Bleehan, and P. H. Rabbits. 1988. Cytologic abnormalities in human small cell lung carcinoma: Cell lines characterized for myc gene amplification. Cancer Genet. Cytogenet. 30:213-223.
Weiss, W., S. Altan, M. Rosenzweig, and M. A. Weiss. 1977. Lung cancer type in relation to cigarette dosage. Cancer 39:2568-2572.
Weston, A., J. C. Willey, R. Modali, H. Sugimura, E. M. McDowell, J. Resau, B. Light, A. Haugen, D. L. Mann, B. F. Trump, and C. C. Harris. 1989. Differential DNA sequence deletions from chromosomes 3, 11, 13, and 17 in squamous-cell carcinoma, large-cell carcinoma, and adenocarcinoma of the human lung. Proc. Natl. Acad. Sci. USA 86:5099-5103.
World Health Organization. 1982. International Histological Classification of Tumors, No. 1: Histological Typing of Lung Tumors, 2nd ed. Am. J. Clin. Pathol. 77:123-136.
Wu, A. H., B. E. Henderson, D. C. Thomas, and T. M. Mack. 1986. Secular trends in histologic types of lung cancer. J. Natl. Cancer Inst. 77:53-56.




























