3
Extrapolation of Doses and Risk per Unit Exposure from Mines to Homes
INTRODUCTION
It is believed that initiating or promotion of the genetic changes in target cells that lead to radiation-induced cancer is caused by specific interactions of ionizing radiation events with DNA in the cell nucleus (NRC, 1990). At the low doses of concern in the assessment of exposure to radon progeny in the home, it is presumed that the likelihood of these carcinogenic events occurring is directly proportional to the radiation dose received by the cell nucleus. More fundamental discussion of the dosimetry and radiobiology of alpha particles can be found in the BEIR IV report (NRC, 1988).
The purpose of this chapter is to translate exposures to radon progeny under the different environmental conditions encountered in mines and homes into the biologically effective dose delivered in each environment. The committee's approach to this task was to apply a dosimetric model based on an up-to-date interpretation of the relevant physiological and biological factors (see Chapters 7 and 8) with current knowledge of the variability of radon progeny aerosol conditions in mines and homes (reviewed in Chapter 2). Details of the dosimetric model together with a summary of the experimental data and underlying assumptions on which the model is based are given in Chapter 9.
This chapter is concerned with evaluating dose conversion coefficient that links exposure to dose. More specifically, the aim is to compare values of the exposure-dose conversion coefficient that apply to subjects exposed in the home with those applicable to underground miners, for the purpose of extrapolating to
the home environment the exposure-risk relationships found in epidemiological studies of underground miners. Using the terminology of the BEIR IV report (NRC, 1988), if exposure is expressed in the commonly used unit working level month (WLM), the risk per unit exposure in the home, (Risk)h/(WLM)h, can be related to that in mines, (Risk)m/(WLM)m, by a dimensionless factor, K.
On the premise that risk is related primarily to the doses received by the appropriate cellular targets, the value of the risk extrapolation factor K is given by the ratio of the doses that result from unit exposure in each environment.
The dose conversion coefficients (Dose/WLM) for miners and for adults and children exposed in the home and the corresponding values of the factor K are evaluated below. The factor K is referred to as the dosimetric risk extrapolation factor or, for convenience, as the K factor. The variability of dose per WLM exposure and the K factor over a range of representative exposure scenarios in mines and homes are examined in this chapter. The sensitivities of these predicted values to parameters of dosimetric modeling that remain uncertain are also determined.
REPRESENTATIVE EXPOSURE CONDITIONS
Radon Progeny Aerosols
Underground mining involves a variety of work activities, from which three representative and characteristically different exposure scenarios are identified in Chapter 6. These scenarios are rock-breaking, ore-winning, and other dusty activities (referred to below as mining); transport and maintenance work in haulageways; and time spent in less active and dusty areas (referred to as lunchrooms). The respective unattached fractions of potential alpha energy (fp) and the activity median thermodynamic diameter (AMTD) of the attached radon progeny aerosols that are assumed to characterize each of these categories of exposure are given in Table 3-1.
Chapter 6 also identifies several characteristically different exposure scenarios in the home. These are distinguished as time spent in living rooms and in bedrooms. The normal living room is considered to be free of strong sources of aerosol particles. The radon progeny aerosol is characteristically different in a room in which cigarettes are smoked (referred to as a smoker), one in which an electric motor is being used (e.g., during vacuuming), or a
TABLE 3-1 Summary of Radon Progeny Aerosol Characteristics Assumed to Represent Exposure Conditions in Mines and Homes
|
Exposure Scenario |
fp |
AMD of Room Aerosol (µm) |
AMD of Aerosol in Respiratory Tract (µm) |
|
Mine |
|||
|
Mining |
0.005 |
0.25 |
0.5 |
|
Haulage driftsa |
0.03 |
0.25 |
0.5 |
|
Lunch room |
0.08 |
0.25 |
0.5 |
|
Living Room |
|||
|
Normal |
0.08 |
0.15 |
0.3 |
|
Smoker—average |
0.03 |
0.25 |
0.5 |
|
Smoker—during smoking |
0.01 |
0.25 |
0.5 |
|
Cooking/vacuuming |
0.05 |
0.02/0.15b (15%/80%) |
0.02/0.3 (15%/80%) |
|
Bedroom |
|||
|
Normal |
0.08 |
0.15 |
0.3 |
|
High |
0.16 |
0.15 |
0.3 |
|
a As noted in Chapter 2, a reconsideration of early aerosol size measurements has indicated that the aerosol AMTD in haulage drifts and other areas away from active mining is shifted to 0.15 µm. The effect on calculated K factors of this revision of the committee's assumed value of 0.25 µm is noted at the end of the chapter. b The radon progeny aerosol produced by cooking/vacuuming has three size modes; 5% of potential alpha energy is unattached, 15% has an AMD of 0.02 µm and 80% has an AMD of 0.15 µm. The 0.02-µm AMD mode is hydrophobic and does not increase in size within the respiratory tract. |
|||
kitchen and connecting rooms during cooking. The radon progeny aerosol in a normal bedroom is considered to be the same as that in the normal living room. However, in well-insulated homes with low exchange between indoor and outdoor air, the particle loading of bedroom air may be substantially less, giving rise to a high value of the unattached fraction. The corresponding values of the unattached fraction and radon progeny aerosol size that are assumed to represent these various exposure scenarios in the home are given in Table 3-1.
In both mine and home atmospheres, unattached radon progeny are considered to have a characteristic diffusion coefficient of 0.035 cm2/s (see Chapter 6). This corresponds to a particle of diameter 0.0011 µm. The size of the unattached progeny is assumed to remain constant in the respiratory tract. However, the condensation nuclei to which radon progeny attach are unstable in saturated air. They are assumed to grow rapidly on inhalation, such that the AMTD of the radon progeny aerosol within the respiratory tract is double that in ambient air (Sinclair et al., 1974). To evaluate the contributions to respiratory tract deposition made by impaction and sedimentation (see Chapter 9), the density of these hygroscopically enlarged particles is taken to be unity.
TABLE 3-2 Levels of Physical Exertion and Average Minute Volumes Assumed for Underground Miners and for Adults in the Home
|
|
Average VE (liters/min) |
||
|
Exposure Scenario |
Level of Exertion |
Man |
Woman |
|
Underground mine |
|||
|
Mining |
25% heavy work/75% light work |
31 |
— |
|
Haulageway |
100% light work |
25 |
— |
|
Lunchroom |
50% light work/50% rest |
17 |
— |
|
Home—Living room |
|||
|
Normal and smoker |
50% light work/50% rest |
17 |
14 |
|
Cooking/vacuuming |
75% light work/25% rest |
21 |
17 |
|
Home—Bedroom |
|||
|
Normal and high |
100% sleep |
7.5 |
5.3 |
BREATHING RATES AND CALCULATION OF DOSE PER UNIT EXPOSURE
The breathing rates of miners at work underground and adults and children under various circumstances in the home are discussed in Chapter 7. To evaluate dose per unit exposure in these situations, the committee assumed the levels of exertion and corresponding minute volumes (VE) given in Table 3-2 for adults and in Table 3-3 for children and infants. The dose per unit exposure for each subject and for each exposure scenario (defined in Table 3-1) was obtained by combining individual values of the primary exposure-dose conversion coefficient that were calculated using the dosimetric model described in Chapter 9. Examples of these primary exposure-dose conversion coefficients are tabulated in Chapter 9. An individual conversion coefficient relates to a specific radon progeny aerosol size and level of exertion (i.e., sleep, rest, light exercise, or heavy exercise). The dependence of primary exposure-dose conversion coefficients on radon progeny aerosol size and level of exertion for male and female adults, children, and infants is illustrated and discussed in Chapter 9.
In order to derive exposure-dose conversion coefficients for a population of underground miners, the committee calculated the individual time-weighted doses for mining and haulageway work. The corresponding average dose-weighted minute volume corresponds to a minute volume of 28 liters/min. To represent overall exposure in the home, the committee used a time-weighted average of individual values of the exposure-dose conversion coefficient calculated for the normal living room and normal bedroom. The corresponding time-averaged dose-weighted minute volumes are 12.3 liters/min for an adult male, 9.7 liters/min for an adult female, 8.8 liters/min for a 10-yr-old child,
TABLE 3-3 Levels of Physical Exertion and Average Minute Volumes Assumed for Children and Infants in the Home
|
Exposure Location and Subject |
Level of Exertion |
Average VE(liters/min) |
|
|
Living room |
|||
|
Child, age 10 yr |
50% light work/50% rest |
12.4 |
|
|
Child, age 5 yr |
50% light work/50% rest |
7.4 |
|
|
Infant, age 1 yr |
50% light work/50% rest |
4.8 |
|
|
Infant, age 1 mo |
30% light work/70% sleep |
1.5 |
|
|
Bedroom |
|||
|
Child, age 10 yr |
100% sleep |
5.2 |
|
|
Child, age 5 yr |
100% sleep |
4.0 |
|
|
Infant, age 1 yr |
100% sleep |
2.6 |
|
|
Infant, age 1 mo |
100% sleep |
1.3 |
|
5.7 liters/min for a 5-yr-old child, 3.7 liters/rain for a 1-yr-old infant, and 1.4 liters/min for a 1-mo-old infant.
COMPARATIVE DOSES FROM RADON PROGENY IN MINES AND HOMES
Target Cells in Bronchial Epithelium
In uranium miners, and also in the general population, the majority of lung cancers arise from the epithelium of bronchial airways (Chapter 8). In this tissue both secretory cells and basal cells are considered to be targets for lung cancer development. The bronchial airways include all airways in which the epithelium is supported by a thick wall of connective tissue and cartilage. These airways are generally larger than 2 mm in caliber in an adult male (see Chapter 9 for the corresponding dimensions in other people). The bronchi are represented in the dosimetric model by airway generations one through eight, in which the trachea is termed generation zero. The committee used the model to calculate doses received by target cells in each generation and then averaged the results to express an exposure-dose conversion coefficient for bronchial epithelium as a whole.
The exposure-dose conversion coefficients calculated for a miner and for an adult male exposed to radon progeny at home are shown in Figure 3-1. Values are shown separately for secretory and basal cells, which are treated as discrete targets. Figure 3-1 also shows the effect of plausible, but different, assumptions about the clearance behavior of radon progeny after deposition on an airway surface (see Chapter 9 for a discussion of the clearance model). The alternative assumptions considered here are (1) radon progeny are effectively insoluble, i.e., they are retained in mucus and cleared progressively toward the throat, and (2) that the progeny are partially soluble, i.e., 30% of the activity deposited in each airway generation is assumed to be taken up rapidly by epithelial tissue.
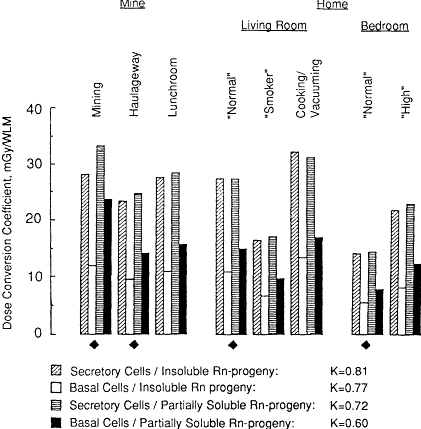
Figure 3-1 Comparison of dose conversion coefficients for a miner and for a man exposed to radon progeny at home. The histograms represent various conditions of exposure in each environment, which are described in the text. Four values of the dose conversion coefficient are shown for each situation. From left to right, these represent the doses calculated for secretory and basal cell nuclei in bronchial epithelium, if radon progeny are assumed to be insoluble, and for the same target cell nuclei if pan of the radon progeny activity is assumed to be taken up by the epithelium. The diamond-shaped symbol indicates conditions that are considered to represent a ''typical'' exposure in each environment: for the mine, an equal mixture of active "mining" and work in a "haulageway" and for the home, equal exposure in the "normal" living room and bedroom. Additional conditions of exposure are also shown, to indicate the degree of variability of the dose conversion coefficient in each environment. The numerical factor K represents the ratio of the "typical" dose conversion coefficient for the man exposed at home to that for the miner.
It is seen from the data in Figure 3-1 that the calculated exposure-dose conversion varies substantially with exposure conditions and with the choice of target cells (secretory or basal cells), but varies to a lesser extent with the assumed clearance behavior. In absolute terms, the conversion coefficient varies from a maximum of 33 milligrays (mGy)/WLM (to secretory cells) during active mining, with similar values for exposure in the home to the radon progeny aerosol produced by cooking/vacuuming, to a minimum of 5.6 mGy/WLM (to basal cells) for an adult male sleeping in a normal bedroom. However, for the purpose of deriving a dosimetric risk extrapolation factor (the K factor), the choice of target cell population and the assumptions about clearance behavior are less influential. Figure 3-1 shows that the corresponding values of the K factor, which represent the ratios of dose conversion coefficients from exposure in the home to that in a mine, vary from 0.81 to 0.60 for secretory cell and basal cell targets, respectively.
As noted earlier, the K factor is derived by averaging the exposure-dose conversion coefficients calculated for the normal living room and bedroom and comparing the result with the average value calculated for mining and haulageway work. Exposure-dose conversion coefficients calculated for additional exposure scenarios (defined in Table 3-1) are also shown in Figure 3-1 (see also Figures 3-2 to 3-8). These are included to illustrate the variability of exposure-dose conversion coefficients, and, potentially, the K factor as well, with particular conditions in the home or mine.
LOCALIZED VERSUS REGIONAL DOSES
The extent of sensitive tissue within the lung and the factors that influence this are not well understood (see also Chapter 8). It is therefore prudent to examine the effect on risk extrapolation of choosing alternative tissues as the reference target. Figure 3-2 shows values of the exposure-dose conversion coefficient and K factor that are obtained when the reference tissue is taken to be the epithelium in just the lobar and segmental bronchi (generations 2-5 in the model), the epithelium in the bronchioles (generations 9-15 in the model), or epithelial cells in the alveoli. These values are compared in Figure 3-2 with the dose conversion coefficients derived for the bronchial airways as a whole by averaging doses received by secretory and basal cell targets. It is seen from the data in Figure 3-2 that exposure-dose conversion coefficients calculated for the lobar and segmental bronchi are uniformly higher than those calculated for the bronchi as a whole (by about 30%). Values calculated for the bronchioles are lower than those for the bronchi, on average by about 50%, and the coefficients calculated for alveolar epithelial cells are only about 2% of those for the bronchi. However, this marked variation of the exposure-dose conversion coefficient between different target tissues has a relatively small impact on the extrapolation of risk from the mine to the home. The K factor is
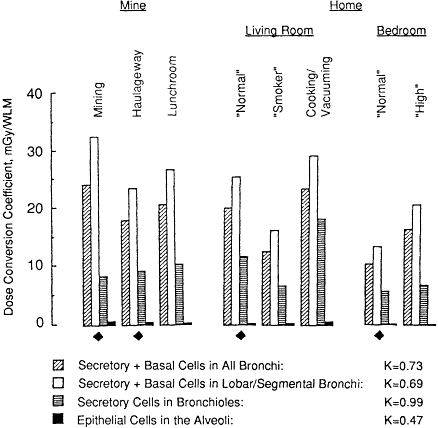
Figure 3-2 Comparison of dose conversion coefficients calculated for sensitive cells in various tissues of the lung. Values are shown by histograms for four cell populations. They represent the average doses received by: secretory and basal cell nuclei throughout the bronchial epithelium, the subpopulation of these cell nuclei in the epithelium of just the lobar and segmental bronchi, secretory cell nuclei in the bronchioles, and the alveolar epithelium. The various exposure conditions in the mine and the home are the same as those described for Figure 3-1.
found to be 0.73 for the bronchi as a whole, 0.69 for just lobar and segmental bronchi, 0.99 for the bronchioles, and 0.47 for alveolar epithelial cells (Figure 3-2). In the dosimetric comparisons that follow, the reference target tissue is assumed to be the bronchial epithelium as a whole.
INFLUENCE OF MODELING UNCERTAINTIES
As discussed in Chapter 9 neither the efficiencies of the nose and mouth in filtering unattached radon progeny from inhaled air nor the theoretical calculation
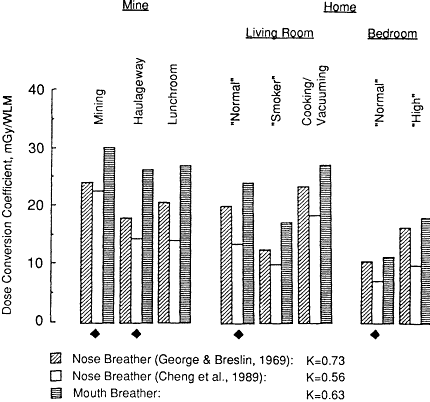
Figure 3-3 Influence of the assumed filtration efficiencies of the nasal and oral airways on the calculated bronchial dose conversion coefficient.
of radon progeny deposition in the bronchi (where airflow is complex) is well established. The impact of these uncertainties on calculated exposure-dose coefficients and the K factor are discussed here. A further source of uncertainty is the degree to which the attached radon progeny aerosols in various exposure situations are hygroscopic and increase in size within the respiratory tract.
The influence of radon progeny filtration by the nasal and oral passages is shown in Figure 3-3. Values of the exposure-dose coefficient are shown in Figure 3-3 for secretory cell targets in the bronchi. The values calculated by using a model of nasal deposition consistent with the human experimental data obtained by George and Breslin (1969) are compared in Figure 3-3 with values based on the recent data from nasal casts that were reported by Cheng et al. (1989). The respective data and the empirical nasal deposition models that are derived from them are discussed in Chapter 9. The lower exposure-dose conversion coefficients implied by the data of Cheng et al.'s result from corresponding
reductions in the dose contributed by unattached progeny. According to Cheng et al., only about 15% of the inhaled unattached progeny are able to pass through the nasal passages without depositing there, whereas George and Breslin's data, and also other data from nasal casts (Strong and Swift, 1990), indicate that the nasal penetration efficiency is between 30 and 40%. Values of the exposure-dose conversion coefficient shown in Figure 3-3 for a mouth breather were calculated on the assumptions that the oral filtration efficiency is half that estimated for the nose from George and Breslin's (1969) data and that a typical mouth breather inhales partly through the nose (see Chapter 9 for a discussion of these assumptions). The resulting K factors are 0.73 according to the committee's preferred estimate of nasal deposition efficiency (George and Breslin, 1969), 0.56 according to the data of Cheng et al. (1989) from nasal casts, and 0.63 for a mouth breather.*
The finding that the K factor for mouth breathers is lower than the value of 0.73 estimated by the committee for nose breathers is at first surprising. However, breathing partly through the mouth has a more complex effect for underground miners than it does for a subject exposed to radon in the home. The mine aerosol is assumed to grow rapidly to reach an AMD of 0.5 µm in nasal or oral passages. At the higher rates of airflow that occur when all inspired air passes through the nose, deposition of the attached radon progeny within the nose is significant. Larger particles, which would otherwise tend to deposit in the bronchi, are lost from the aerosol. Filtration of larger aerosol particles is lower on two counts if the inhaled air is split between the nose and mouth: the airflow rate through the nose is reduced (decreasing the inertial deposition efficiency of the nose), and the inertial deposition efficiency of the oral passageway is comparatively low. However, if the incidence of mouth breathing among underground miners is substantially greater than that among subjects at home, K factors derived purely for nose breathers may be biased toward artificially high values.
The effect on calculated exposure-dose conversion coefficients by using bronchial deposition efficiencies based on empirical observations made from experiments with bronchial casts (Cohen et al., 1990), rather than a purely theoretical analysis, is shown in Figure 3-4 (see Chapter 9 for a discussion of the data and theoretical deposition models). It is seen from the data in Figure 3-4 that the assumption of enhanced bronchial deposition efficiencies reported by Cohen et al. gives uniformly higher conversion coefficients. However, the
|
* |
As noted in Chapter 9, new experimental studies of the penetration of unattached radon progeny through nasal casts, carded out after the committee completed its work, are found to support the data of Cheng et al. (1989) and no longer to support the lower values of nasal deposition that are obtained from George and Breslin's (1969) study. After further experimental verification, it may become preferable to evaluate K factors based on the higher nasal deposition efficiency reported by Cheng et al. The effect of higher nasal deposition is evaluated in a note to Table 3-4. |
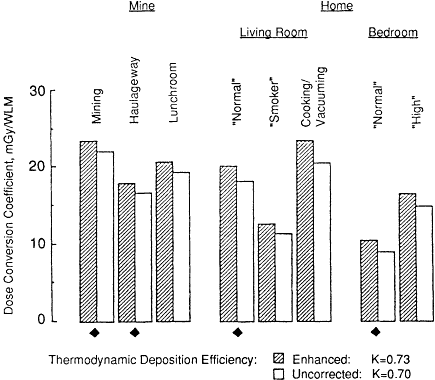
Figure 3-4 Influence of the enhanced bronchial deposition efficiency observed experimentally for submicron aerosol particles by Cohen et al. (1990) on the calculated dose conversion coefficient.
effect on the K factor is marginal. The empirically enhanced value of the K factor is 0.73, and the uncorrected theoretical value is similar at 0.70.
The sensitivities of calculated exposure-dose conversion coefficients and the K factor to the assumption made by the committee that the attached radon progeny aerosol grows in the respiratory tract to double its size in ambient air are examined in Figure 3-5. In this case, the values shown in Figure 3-5 are averages of dose conversion coefficients calculated for secretory and basal cell targets. It is seen that this assumed aerosol growth tends to increase conversion coefficients for exposures in mines but to decrease those calculated for exposures in the home. These effects arise because an aerosol of 0.5 µm in AMD (the size that a mine aerosol is assumed to attain by growth in the respiratory tract) is deposited more efficiently in the bronchi than is an aerosol of 0.25-µm AMD (the ambient size of the mine aerosol), whereas the reverse holds for growth of the smaller ambient aerosol generally found in the home.
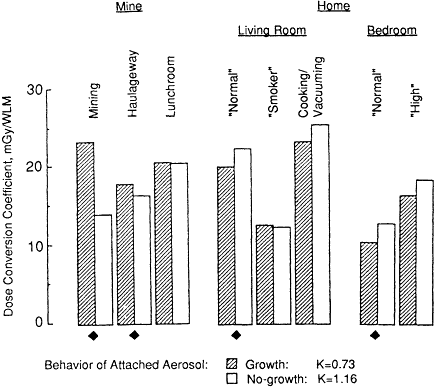
Figure 3-5 Influence of the assumed growth of radon progeny aerosols in the humid air of the respiratory tract on the calculated bronchial dose conversion coefficient.
If the attached radon progeny aerosols in both mines and homes were, in fact, stable in the respiratory tract, the K factor would increase to 1.16, which is significantly higher than the committee's estimate of 0.73.
DOSES TO ADULT FEMALES
Bronchial exposure-dose conversion coefficients calculated for a female exposed to radon progeny in the home (a female homemaker) are compared in Figure 3-6 with the values presented in Figure 3-1 for an underground miner. Variations of the dose conversion coefficient with exposure conditions and with the type of target cell considered (secretory or basal cell) are similar to those calculated for a male (compare Figure 3-6 with Figure 3-1). However, the K factors are somewhat lower for a female. The K factor is 0.72 for secretory
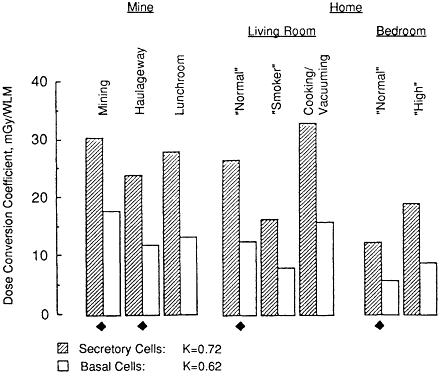
Figure 3-6 Comparison of the bronchial dose conversion coefficients calculated for exposure of a female homemaker to radon progeny with those for a miner.
cell targets and 0.62 for basal cells in a female compared with 0.76 and 0.69, respectively, for a male.
DOSES TO CHILDREN AND INFANTS
Exposure-dose conversion coefficients calculated for children and infants exposed to radon progeny in the home are compared in Figure 3-7 with those for adult males and females. The values shown in Figure 3-7 apply to secretory cell targets in the bronchial epithelium of each subject. In this example, the conversion coefficients calculated for subjects breathing entirely through the mouth are compared with those calculated for those breathing entirely through the nose. Although it is unlikely that any subject breathes entirely through the mouth, this behavior is examined here to indicate an upper bound for the dose conversion coefficient in each case. The ratio of dose conversion coefficients calculated for 100% oral breathing relative to 100% nasal breathing is 1.44 ± 0.02 (standard deviation [SD] between subjects) for exposure in the living room
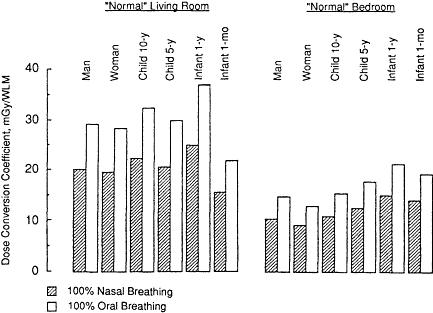
Figure 3-7 Influence of subject age and gender on the bronchial dose conversion coefficient calculated for exposure to radon progeny under "normal" conditions at home in the living room and bedroom. Values are compared for subjects breathing entirely through the nose or mouth.
and 1.40 ± 0.01 for exposure in the bedroom. During activities not demanding high work levels, the majority of people breathe through the nose unless they are talking (or crying in the case of an infant) or suffering from nasal congestion (see Chapter 7).
It is seen from Figure 3-7 that the exposure-dose conversion coefficient is generally higher for children than for adults, but only slightly so. The corresponding values of the K factor (relative to exposures of underground miners) for each subject are summarized in the concluding section of this chapter. The K factors are given in Tables 3-4 and 3-5, for normal subjects and for nose and mouth breathers, respectively.
VARIABILITY OF THE DOSE CONVERSION COEFFICIENT IN HOMES
The dose conversion coefficients shown in Figure 3-7 apply to exposure conditions in living rooms and bedrooms that are assumed to represent normal (or typical) situations (these were defined in Table 3-1). The effect of more extreme aerosol conditions is examined in Figure 3-8. In Figure 3-8 the dose
TABLE 3-4 Summary of K Factors Calculated for Normal Healthy Subjectsa
|
Subject |
Target Region |
Target Cell |
Radon Progeny Solubility |
K Factor ± SDb |
|
Man |
Bronchi |
Secretory |
Insoluble Part-soluble |
0.81 ± 0.25 0.72 ± 0.22 |
|
|
|
Basal |
Insoluble Part-soluble |
0.77 ± 0.25 0.60 ± 0.21 |
|
Man |
Lobar/segmental |
Secretory |
Insoluble Part-soluble |
0.78 ± 0.24 0.66 ± 0.20 |
|
|
Bronchi |
Basal |
Insoluble Part-soluble |
0.75 ± 0.24 0.59 ± 0.19 |
|
Man |
Bronchioles |
Secretory |
Mean |
0.99 ± 0.48 |
|
Man |
Bronchi |
Secretory + basal |
Mean |
0.73 ± 0.23 |
|
Man |
Bronchi |
Secretory |
Mean |
0.76 ± 0.23 |
|
|
Bronchi |
Basal |
Mean |
0.69 ± 0.22 |
|
Woman |
Bronchi |
Secretory |
Mean |
0.72 ± 0.26 |
|
|
Bronchi |
Basal |
Mean |
0.62 ± 0.24 |
|
Child, 10 yr |
Bronchi |
Secretory |
Mean |
0.83 ± 0.28 |
|
|
Bronchi |
Basal |
Mean |
0.72 ± 0.26 |
|
Child, 5 yr |
Bronchi |
Secretory |
Mean |
0.83 ± 0.23 |
|
|
Bronchi |
Basal |
Mean |
0.72 ± 0.22 |
|
Infant, 1 yr |
Bronchi |
Secretory |
Mean |
1.00 ± 0.29 |
|
|
Bronchi |
Basal |
Mean |
0.87 ± 0.28 |
|
Infant, 1 mo |
Bronchi |
Secretory |
Mean |
0.74 ± 0.21 |
|
|
Bronchi |
Basal |
Mean |
0.64 ± 0.20 |
|
a Use of the nasal deposition efficiency reported by Cheng et al. (1989), and also on a revised estimate of 0.15 µm for the characteristic aerosol size in the underground haulage drifts (see Table 3-1), would yield smaller K factors. The mean values of the K factor calculated for bronchial target cells become: Adult male, 0.58 vis à vis 0.73; adult female, 0.55 vis à vis 0.67; child age 10 yr and age 5 yr, 0.64 vis à vis 0.78; infant age 1 yr, 0.78 vis à vis 0.94; infant age 1 mo, 0.56 vis à vis 0.69. b ±SD refers to the standard deviation of the calculated K factor for the five home environments and three mine environments shown in Figure 3.1. |
||||
conversion coefficients calculated for each subject exposed to radon progeny in a living room in the presence of an active cigarette smoker (where the unattached fraction of potential alpha energy, fp, is assumed to be only 1%, and the AMD of the attached aerosol is 0.25 µm) are compared with the values shown in Figure 3-7 for nose breathers exposed to the normal living room atmosphere (where fp is 8% and the aerosol AMD is 0.15 µm). In this case, when cigarettes are being smoked in a room, the exposure-dose conversion coefficient is, on average only 47% of the normal value (±1% standard deviation between subjects). Figure 3-8 also shows the effect of the higher unattached fraction (16%) expected in a bedroom when the rate of exchange with outdoor air is low. The exposure-dose conversion coefficient is then calculated to be 51% higher (±5% standard deviation) than the values applicable to the normal bedroom atmosphere.
TABLE 3-5 Summary of the Effect on Calculated K Factors of Nasal or Oral Breathing Habit
|
|
|
Breathing Habit of Reference Miner |
|
|
Subject |
Target Cell |
Nasal |
Oral |
|
K Factor for Nasal Breathers in the Home |
|||
|
Man |
Secretory |
0.76 |
0.56 |
|
|
Basal |
0.66 |
0.51 |
|
Woman |
Secretory |
0.72 |
0.53 |
|
|
Basal |
0.62 |
0.48 |
|
Child, 10 yr |
Secretory |
0.83 |
0.61 |
|
|
Basal |
0.72 |
0.55 |
|
Child, 5 yr |
Secretory |
0.83 |
0.61 |
|
|
Basal |
0.72 |
0.55 |
|
Infant, 1 yr |
Secretory |
1.00 |
0.73 |
|
|
Basal |
0.87 |
0.66 |
|
Infant, 1 mo |
Secretory |
0.74 |
0.54 |
|
|
Basal |
0.64 |
0.49 |
|
K Factor for Oral Breathers in the Home |
|||
|
Man |
Secretory |
0.88 |
0.64 |
|
|
Basal |
0.81 |
0.59 |
|
Woman |
Secretory |
0.84 |
0.61 |
|
|
Basal |
0.76 |
0.55 |
|
Child, 10 yr |
Secretory |
0.97 |
0.71 |
|
|
Basal |
0.88 |
0.64 |
|
Child, 5 yr |
Secretory |
0.95 |
0.70 |
|
|
Basal |
0.87 |
0.63 |
|
Infant, 1 yr |
Secretory |
1.16 |
0.85 |
|
|
Basal |
1.06 |
0.77 |
|
Infant, 1 mo |
Secretory |
0.82 |
0.60 |
|
|
Basal |
0.75 |
0.55 |
EFFECTS OF AIRWAY DISEASE ON DOSE
The results of modeling the effects of airway diseases on the exposure-dose conversion coefficients calculated for underground miners are shown in Figure 3-9. Three distinct disease conditions are considered:
Bronchitis, in which the epithelium is assumed to be normal, but the overlying mucus is assumed to have an abnormal thickness of 30 µm and the mucous clearance rates in each airway generation are assumed to be one-half the normal values. In this case, secretory cells are assumed to be the principal targets.
Hyperplasia of bronchial epithelium, in which the bronchial epithelium is assumed to be twice the normal thickness (at 100 µm) and is devoid of secretory cells. Basal cell nuclei, which are assumed to be the sensitive targets, are located in an abnormally thick layer (30-µm thick) at a depth of 70 µm. It is assumed that the epithelial surface is devoid of cilia and is lined only by a
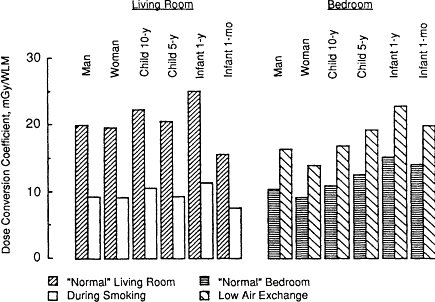
Figure 3-8 Influence of aerosol conditions in the living room and bedroom on the dose conversion coefficients calculated for various subjects exposed to radon progeny.
thin layer of fluid (3-µm thick). In this case, the committee assumed that there is no movement of deposited radon progeny toward the throat.
Local regeneration of bronchial epithelium in response to epithelial injury, in which case the epithelium is assumed to be one-half the normal thickness, such that secretory cell nuclei occur in a 15-µm-thick layer at a 5-µm depth and basal cell nuclei occur in an 8-µm-thick layer at a 17-µm depth. The epithelium is again assumed to be devoid of cilia and to be covered only by a 3-µm layer of fluid for which clearance is ineffective. The principal targets are assumed to be the nuclei of secretory cells.
Comparison of the conversion coefficients calculated for miners under these assumed conditions of disease with values calculated for healthy subjects yields the somewhat complex effects shown in Figure 3-9. The dose to secretory cell nuclei in the bronchial epithelium of a bronchitic miner is estimated to be between 40 and 50% of that for a healthy subject. The ratio of doses depends to this rather limited extent on the conditions of exposure and the assumed solubility of radon progeny. In the case of epithelial hyperplasia, however, the deep-lying basal cell targets receive no dose if the deposited radon progeny remain in the thin layer of fluid that is assumed to cover the epithelium. Alternatively, if the progeny are assumed to be partially soluble and 30% of
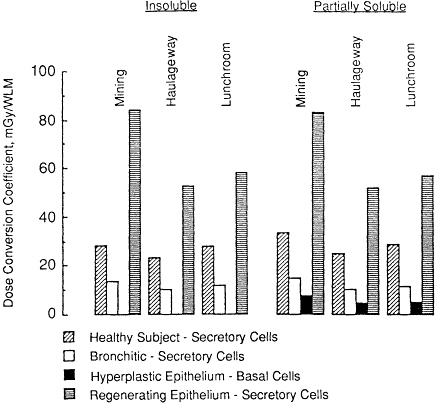
Figure 3-9 Modeled effects of bronchial airway disease in miners on the calculated dose conversion coefficient for exposure to radon progeny. Values calculated for subjects with bronchitis (secretory cells are the assumed targets), or for those with hyperplasia of bronchial epithelium (where the targets are assumed to be basal cells) or regenerating epithelium (secretory cell targets), are compared with values calculated for miners with healthy bronchial epithelium. Dose conversion coefficients calculated on the assumption that radon progeny are insoluble (i.e., that they remain in mucus) are shown separately from the corresponding values calculated if part of the progeny activity is assumed to be taken up by the bronchial epithelium.
the deposited activity is taken up by the epithelial tissue, the dose received by the basal cell nuclei at the site of hyperplasia is calculated to be approximately 20% of the average dose received by secretory cell nuclei in a healthy subject. In the case of local areas of regenerating epithelium, it is estimated that the target secretory cell nuclei receive between two- and threefold higher doses than the average dose for a healthy subject. The actual ratio of doses within this range again depends on the exposure conditions and the assumed solubility characteristics of the deposited radon progeny.
Similar dosimetric effects are calculated to arise for subjects with these
TABLE 3-6 Summary of K Factors Calculated for Bronchial Target Cells in Adult Males with Airway Disease Compared to Healthy and Diseased Miners
|
Subject/Disease |
Target Cell |
Radon-Progeny Solubility |
K Factor ± SDa |
|
Diseased subject in the home (cf. healthy miner) |
|||
|
Bronchitis |
Secretory |
Insoluble Part-soluble |
0.34 ± 0.10 0.28 ± 0.08 |
|
Hyperplasia |
Basal |
Insoluble Part-soluble |
0 0.18 ± 0.06 |
|
Epithelial injury (regeneration) |
Secretory |
Insoluble Part-soluble |
1.58 ± 0.38 1.38 ± 0.35 |
|
Healthy subject in the home (cf. diseased miner) |
|||
|
Bronchitis |
Secretory |
Insoluble Part-soluble |
1.75 ± 0.56 1.70 ± 0.53 |
|
Hyperplasia |
Basal |
Insoluble Part-soluble |
1.84 ± 0.63 |
|
Epithelial injury (regeneration) |
Secretory |
Insoluble Part-soluble |
0.30 ± 0.11 0.31 ± 0.11 |
|
a ±SD refers to the standard deviation of the calculated K Factor for the five home environments and three mine environments shown in Figure 3-1. b No dose to diseased miner. |
|||
airway diseases when they are exposed in the home. The overall effects of airway disease on the estimated K factor, for dosimetric extrapolation of risk, are summarized in Table 3-6.
COMPARATIVE DOSES FROM RADON AND THORON PROGENY
The dose conversion coefficients calculated for exposure to thoron progeny in a mine or a home are compared in Figure 3-10 with the values applicable for an adult male exposed to radon progeny. For thoron progeny, the unattached fraction of potential alpha energy, fp, is assumed to be 0.1% during active mining, 1% in an underground haulageway, and normally, 2% indoors in the home. The AMD of the attached thoron progeny aerosol is assumed to be 0.25 µm in all cases in ambient air and 0.5 µm within the respiratory tract.
It is found that the dose from exposure to thoron progeny is determined principally by the behavior of lead-212, which has a relatively long radioactive half-life (10.6 h). The assumed solubility characteristics and clearance behavior are then found to have a substantially greater effect on calculated doses than they do on calculated doses for radon progeny. Dose conversion coefficients calculated separately on the alternative assumptions that thoron progeny are insoluble (i.e., remain in mucus) or are partially soluble and partially taken up
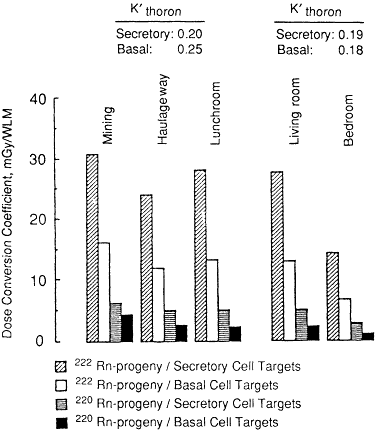
Figure 3-10 Comparison of bronchial dose conversion coefficients calculated for exposure of a man to radon progeny in a mine or at home with those calculated for exposure to thoron (220Rn) progeny.
by epithelial tissue are given in Chapter 9. The exposure-dose conversion coefficients shown in Figure 3-10 were obtained by averaging the values calculated for both types of assumed clearance behavior.
It is seen from the data in Figure 3-10 that, for unit exposure to thoron progeny in a mine or in the home, secretory cell nuclei are expected to receive only about 20% of the dose that they receive from the same exposure to radon progeny. Basal cell nuclei are estimated to receive a somewhat higher dose from unit exposure to thoron progeny in a mine than they do in the home (25% of the dose from radon progeny in a mine compared with 18% in the home). The dosimetric risk extrapolation factors K'thoron for exposure to thoron
progeny in the home relative to radon-222 in mines, Figure 3-10 were obtained by substituting these relative doses in the following equation:
SUMMARY OF DOSIMETRIC RISK EXTRAPOLATION FACTORS
Dosimetric risk factors (K factors) for extrapolating the observed risk of radon progeny exposure in mines to domestic settings that were derived in this chapter for various reference target tissues and different subjects are summarized in Table 3-4. K factors derived with respect to miners and subjects in the home who breath habitually through both the mouth and nose are summarized in Table 3-5. Finally, the K factors derived with respect to subjects with airway disease are summarized in Table 3-6.
REFERENCES
Cheng, Y. S., D. L. Swift, Y. F. Su, and H. C. Yeh. 1989. Deposition of radon progeny in human head airways. Pp. 29-30 in Inhalation Toxicology Research Institute Annual Report 1988-89. LMF-126, Albuquerque, N.M.: Lovelace Biomedical and Environmental Research Institute.
Cohen, B. S., R. G. Sussman, and M. Lippmann. 1990. Ultrafine particle deposition in a human tracheobronchial cast. Aerosol Sci. Technol. 12:1082-1091.
George, A. C., and A. J. Breslin. 1969. Deposition of radon daughters in humans exposed to uranium mine atmospheres. Health Phys. 17:115-124.
National Research Council (NRC). 1988. Health Risks of Radon and Other Internally Deposited Alpha-Emitters. BEIR IV. Committee on the Biological Effects of Ionizing Radiation. Washington, D.C.: National Academy Press.
National Research Council (NRC). 1990. Committee on the Biological Effects of Ionizing Radiations. Health Effects of Exposure to Low Levels of Ionizing Radiation. BEIR V. Washington, D.C.: National Academy of Press.
Sinclair, D. R., R. J. Countess, and G. S. Hoopes. 1974. The effect of relative humidity on the size of atmospheric aerosol particles. Atmos. Environ. 8:1111-1117.
Strong, J. C., and D. L. Swift. 1990. Deposition of 'unattached' radon daughters in models of the human nasal and oral airways. Paper presented at the 29th Hanford Life Sciences Symposium, Indoor Radon and Lung Cancer: Reality or Myth? October 16-19, 1990, Richland, Wash.





















