1
Introduction
Radon (radon-222), an inert gas under usual environmental conditions, is a naturally occurring decay product of radium-226, the fifth daughter of uranium-238 (Figure 1-1). Both uranium-238 and radium-226 are present in most soils and rocks, although the concentrations vary widely (National Council on Radiation Protection and Measurements, 1984a). As radon forms, some atoms leave the soil or rock and enter the surrounding soil or water. Consequently, radon is ubiquitous in indoor and outdoor air. Radon decays with a half-life of 3.82 days into a series of solid, short-lived radioisotopes that are collectively referred to as radon daughters, radon progeny, or radon decay products (Figure 1-1). Two of these progeny, polonium-218 and polonium-214, emit alpha particles. When these radon progeny are inhaled and release alpha particles within the lungs, the cells lining the airways may be damaged and lung cancer may ultimately result.
As information on air quality in indoor environments accumulated, it became apparent that radon and its progeny are invariably present in indoor environments and that concentrations vary widely, even reaching in some dwellings levels found in underground mines. The well-documented excess cases of lung cancer among underground miners exposed to radon raised concern that exposure to the gas might also be a cause of lung cancer in the general population. Although the problem of indoor radon was well known in the scientific community by the late 1970s, it did not receive great public attention in the United States until a widely publicized incident in 1984. During routine monitoring, a worker in a Pennsylvania nuclear power plant was found to be contaminated with radioactivity. This contamination was subsequently
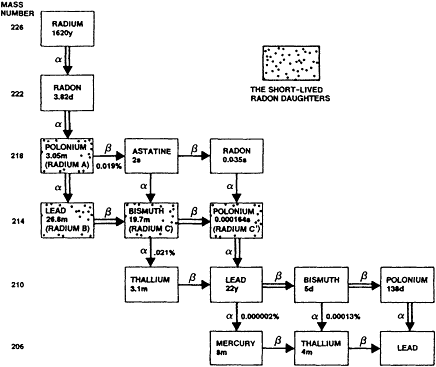
Figure 1-1 The radon decay chain.
traced to radon in his home, which was located on a geologic formation known as the Reading Prong. High levels of radon have now been measured in other homes on the Reading Prong and throughout the United States (Logue and Fox, 1985; Cohen, 1986; Nero et al., 1986; U.S. Environmental Protection Agency, 1988).
Recognition that the problem of indoor radon is widespread has prompted action by the EPA and some states. The pamphlet ''A Citizen's Guide to Radon,'' first published by EPA in 1986, sets out the risks of radon and provides guidance on action to be taken at various concentrations (EPA, 1986). Although EPA does not have a statutory basis for regulating concentrations of radon indoors, its action guidelines are the most widely considered national recommendations on acceptable concentrations indoors. In 1988, EPA and the Centers for Disease Control jointly recommended that nearly all homes in the United States be tested for radon and in 1989 the EPA called for testing of schools. In many regions, radon testing is routinely performed at the time of sale of homes. Thus, during the 1980s, the health effects of radon assumed importance for virtually
the entire population, and property owners were faced with making a decision about testing and potentially paying for mitigation.
The evidence from epidemiological investigations of miners justifies concern about the hazard posed by indoor radon. Radon has been linked to excess cases of lung cancer in underground miners since the early decades of the twentieth century. As early as the 1500s, Agricola described unusually high mortality from respiratory diseases among underground metal miners in the Erz mountains of eastern Europe. The disease, termed Bergkrankheit, probably represented lung cancer and silicosis and tuberculosis, which are common diseases of underground miners. In 1879, Harting and Hesse (1879) reported autopsy findings in miners of Schneeberg in Germany that documented an occupational hazard of lung cancer, although they did not identify the disease as primary cancer of the lung. Early in the twentieth century, further pathological studies showed that the miners developed primary carcinoma of the lung (Arnstein, 1913; Rostoski et al., 1926).
Measurement of radon in the mines in eastern Europe early in this century documented the presence of radon at concentrations that would be considered high by present standards. By the 1930s, excess cases of lung cancer were demonstrated among miners in Joachimsthal, which is on the Czechoslovakian side of the Erz Mountain range, and radon was also found in the air of these mines. Radon was considered to be a likely cause of lung cancer in Joachimsthal miners (Pirchan and Sikl, 1932), but the causal role of radon was not uniformly accepted until the biological basis for carcinogenesis by radon was better understood and further epidemiological evidence documented excess cases of lung cancer in other groups of exposed miners (Seltser, 1965; Hueper, 1966; Lundin et al., 1971). Bale's 1951 memorandum showing that the progeny of radon, rather than radon itself, deliver alpha energy to the respiratory tract was an important advance (Bale, 1980).
Epidemiological evidence on radon and lung cancer, as well as other diseases, is now available from about 20 different groups of underground miners (Samet, 1989). Excess occurrences of lung cancer have been found in uranium miners in the United States, Czechoslovakia, France, and Canada and in other underground miners exposed to radon decay products, including Newfoundland fluorspar miners, Swedish and U.S. metal miners, British and French iron miners, and Chinese and British tin miners (National Research Council [NRC], 1988). Many of these studies include information on the exposure of the miners to radon progeny and provide estimates of the quantitative relationship between exposure and lung cancer risk (Lubin, 1988; Samet, 1989). In view of the contrasting methodologies used in these investigations, the coefficients describing the change in excess relative risk per unit exposure span a remarkably narrow range (Table 1-1).
The risk of indoor radon has been primarily assessed by using risk assessment approaches that extend the findings of the studies of miners to the
TABLE 1-1 Relative Risk Coefficients for Lung Cancer from Epidemiological Studies of Underground Minersa
general population. Epidemiological investigations in the general population can potentially provide direct estimates of the risks of indoor radon. The results of studies of indoor radon have been published (Samet, 1989), and numerous investigations are now in progress throughout the world (U.S. Department of Energy/Commission of the European Communities [DOE, 1989]). However, difficult methodological problems limit the accuracy of these studies (Lubin et al., 1990), and the studies of miners will probably remain the principal basis for estimating the risks of indoor radon for the immediate future.
Extrapolation of the lung cancer risks observed in underground miners to the risks for the general population who are exposed to radon indoors is subject to uncertainties related to the differences between the physical environments of homes and mines, the circumstances and temporal patterns of exposure in the two environments, and the potential biological differences between miners and the general population (Table 1-2 and Figure 1-2).
With regard to the physical differences between homes and mines, the activity-weighted particle size distributions tend to be different, with there being more activity in the ultrafine mode (unattached fraction) in homes. The air of many mines was contaminated by other carcinogens, such as diesel exhaust, and by toxic and irritant dusts and gases, such as silica, uranium, and oxides of nitrogen. In general, mining-related jobs underground would demand breathing rates higher than those demanded by more sedentary indoor activities. A greater minute ventilation of miners would tend to increase the dose to target cells at a particular concentration.
The miners included in the epidemiological studies were generally occupationally exposed to radon progeny for a relatively small proportion of their lives, whereas exposure indoors takes place across the entire life span, albeit at varying rates. The miners were largely adult males with a high proportion of smokers, whereas the general population includes males and females, smokers and nonsmokers, of all ages (Table 1-2). Thus, an assumption must also be
TABLE 1-2 Potentially Important Differences Between Exposure to Radon in the Mining and Home Environments
|
PHYSICAL FACTORS |
|
Aerosol characteristics: Greater concentrations in mines; differing size distributions |
|
Attached/unattached fractions: Greater unattached fractions in homes |
|
Equilibrium of radon/decay products: Highly variable in homes and mines |
|
ACTIVITY FACTORS |
|
Amount of ventilation: Probably greater for working miners than for persons indoors |
|
Pattern of ventilation: Patterns of oral/nasal breathing not characterized |
|
BIOLOGICAL FACTORS |
|
Age: Miners have been exposed during adulthood; entire spectrum of ages exposed indoors |
|
Gender: Miners studied have been exclusively male; both sexes exposed indoors |
|
Exposure pattern: Miners exposed for variable intervals during adulthood; exposure is lifelong for the population |
|
Cigarette smoking: The majority of the miners studied have been smokers; only a minority of U.S. adults are currently smokers |
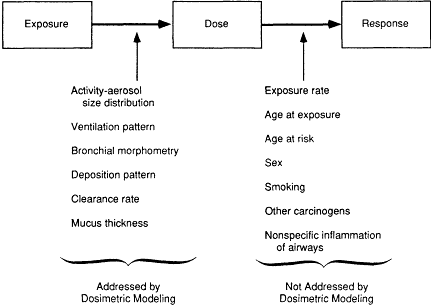
Figure 1-2 Factors influencing the relationship between radon exposure and the risk of lung cancer.
made concerning the interaction between radon progeny and cigarette smoking, the principal cause of lung cancer (U.S. Department of Health and Human Services, 1989). Inflammation of the airways of miners by irritant dusts and gases might also have influenced the response to radon progeny.
The need to make assumptions concerning these physical and biological factors that may lead to different risks of radon exposure in mines and in homes and the associated uncertainties have reduced confidence in the projections of the public's risk of radon-related lung cancer; in fact, some scientists and policymakers discard the risk projections based on the studies of miners as too uncertain and call for empiric evidence that radon causes lung cancer in the general population.
Methods are available, however, for characterizing the uncertainties associated with the factors determining the relationship between exposure to radon and the dose of alpha energy delivered to target cells in the respiratory tract. Using models of the respiratory tract, the dose to target cells in the respiratory epithelium, which lines the lung's airways, can be estimated for the circumstances of exposure in the mining and indoor environments. In such comparative analyses reported to date, the relationship between exposure and dose, and hence the potency of radon as a carcinogen, has generally been found to be comparable in these two settings (NCRP, 1984b; James, 1988; NRC, 1988). For some parameters, the input data needed for these analyses are limited, and controversy remains concerning the application of the findings for miners to the entire general population of infants, children, adult females, and adult males. For example, the International Commission on Radiological Protection (ICRP) concluded (ICRP, 1987) that risks were threefold greater for those in the group from 0 to 20 years of age as compared with the risks in older persons; in contrast, the time-dependent risk model developed by the Committee on the Biological Effects of Ionizing Radiation (BEIR IV) (NRC, 1988) suggests that there are reduced effects for exposures received during childhood.
Thus, further consideration of the relation between exposure to radon and the radiation dose and its biological effectiveness as delivered to the respiratory tract is warranted by the continuing scientific controversy concerning the risks of exposure to indoor radon and by the potential policy implications of risk projections of radon-related lung cancer in the general population. Such risk projections serve as the basis for establishing action guidelines for judging the safety of the nation's homes, schools, and offices and for guiding potentially costly mitigation of unacceptable concentrations (EPA, 1986). In response to this immediate and compelling rationale, the present committee was charged with considering the dosimetry of radon and its decay products in the mining and indoor environments. This charge does not fully cover all uncertainties related to extrapolating risks derived from studies of miners to risks for the general population (Figure 1-2 and Table 1-2). To the extent possible, the
committee has also considered biological factors relevant to this extrapolation, including age at exposure, duration of exposure, sex, cigarette smoking, and the effects of other contaminants in the air of mines. This chapter provides a general background for the committee's detailed review and conclusions.
CONCENTRATION, EXPOSURE, AND DOSE
For historical reasons, the concentration of radon progeny in mines has generally been expressed as working levels (WL), where 1 WL is any combination of progeny in 1 liter of air that ultimately releases 1.3 × 105 MeV of alpha energy during decay (Holaday et al., 1957). Concentrations of radon in indoor air have most often been expressed as picocuries (pCi) per liter, a unit for the rate of decay; a radon concentration of 1 pCi/liter translates to about 0.005 WL under the usually assumed conditions of equilibrium between radon and its progeny in a home. In international (SI) units, the activity of radon per unit mass of air is expressed as becquerels (Bq) per cubic meter; at radioactive equilibrium between radon and its progeny, 1 WL corresponds to 3.7 × 103 Bq/m3.
The working level month (WLM) was developed to describe exposure to radon progeny in underground mines (Holaday et al., 1957). Exposure to 1 WL for 170 h equals 1 WLM of exposure. Because most persons spend much more time than 170 h at home each month, a concentration of 1 WL in a residence results in an exposure much greater than 1 WLM on a monthly basis. For example, during 1 month, if the concentration of radon in a home were 1.0 WL, a child spending 75% of the time at home would receive an exposure of 3.2 WLM, whereas an adult spending 60% of the time at home would receive 2.5 WLM. Exposure in SI units is expressed in Joule-hours (Jh) per m3, and 1 WLM is 3.5 × 10-3 Jh/m3.
The relationship between exposure to radon progeny, measured as WLM or Jh/m3, and dose to cells in the respiratory tract, considered as targets for carcinogenesis, is extremely complex and is dependent on both biological and nonbiological factors, including the physical characteristics of the inhaled air, the amount of air inhaled, breathing patterns, and the biological characteristics of the lung (see Chapter 9). Because the dose of alpha energy delivered to target cells in the lungs cannot be measured directly, modeling approaches are used to simulate the sequence of events from inhalation of radon progeny to cellular injury by alpha particles. These complex models generally incorporate biological factors, including airway geometry, mucociliary clearance, particle deposition, ventilation pattern, and location of the target cells, and physical factors, including the aerosol size distribution and the proportion of progeny not attached to particles. In the past, the terms attached and unattached fractions have been used to refer to radon progeny presumed to be attached and unattached to atmospheric particles. However, these terms are more appropriately replaced
by the term activity-weighted particle size distribution. Recent evidence suggests that the unattached progeny are in ultrafine clusters in the 0.5- to 3-nm size range and that unattached and attached fractions cannot be distinctly separated (see Chapter 6). Factors for converting exposure to an absorbed radiation dose can be calculated by using dosimetric models of the respiratory tract, but the range of published dose conversion factors is wide (James, 1988). To convert absorbed dose to tissue dose equivalent in rem, or sieverts in the SI system, the absorbed dose in rads or grays is multiplied by a quality factor for alpha radiation.
RISK ASSESSMENT FOR INDOOR RADON
To estimate the lung cancer hazard associated with indoor radon, information on exposure levels in dwellings is used in a risk projection equation, or model, that describes the increment in the occurrence of lung cancer per unit exposure. In the principal models in use at present, the risk coefficients describing the relationship between exposure and lung cancer occurrence are derived from studies of miners (NCRP, 1984b; ICRP, 1987; NRC, 1988). The models project the excess occurrences of lung cancer following exposure across the lifetimes of the exposed persons. Although each of the models incorporates risk coefficients from the studies of miners, the biological assumptions underlying the models differ (Table 1-3), and risk projections from the models may vary substantially (Land, 1989).
The incidence of (and mortality from) lung cancer rises sharply from about
TABLE 1-3 Features of Selected Risk Projection Models for Radon and Lung Cancer
|
Feature |
NCRP |
ICRP |
BEIR IV |
|
Form of model |
Attributable risk |
Relative risk |
Relative risk |
|
Time-dependent |
Yes; risk declines exponentially after exposure |
No |
Yes; risk declines as time since exposure lengthens |
|
Lag interval |
5 years |
10 years |
5 years |
|
Age at exposure |
No effect of age at exposure |
Threefold increased risk for exposures before age 20 years |
No effect of age at exposure |
|
Age at risk |
Risk commences at age 40 years |
Constant relative risk with age |
Lower risks for ages 55 years and older |
|
Dosimetry adjustment |
Increased risk for indoor exposure |
Decreased risk for indoor exposure |
No adjustment |
|
Risk coefficient |
10 × 10-6/year/WLM |
Excess relative risks: 1.9%/WLM at ages 0-20 years and 0.64%/WLM for ages 21 years and above |
Excess relative risk of 2.5%/WLM but modified by time since exposure |
ages 50 through 80 yr. Risk projection models inherently assume a relationship between the added risk from radon progeny and the background risk of lung cancer. The model of the NCRP (1984b) assumes additivity of the risks and a time-dependent decline in risk following exposure. In contrast, the model of the ICRP (1987) assumes that the background rate is multiplied by the additional risk associated with radon progeny. The model developed by the BEIR IV committee (NRC, 1988) is also multiplicative, but it incorporates a time-dependent decline in risk.
Regardless of the form of a model, its application to the indoor environment requires consideration of the relationship between exposure and dose in the mining environment and in the indoor environment. In this regard, the three models described in Table 1-3 make different assumptions. The BEIR IV model makes no adjustment, whereas the model of the ICRP reduces the risks by 20% for adults in the general population, and the NCRP model increases the risks by 40% for the general population, assuming 0.5 rad/WLM for the mining environment and 0.7 rad/WLM for the indoor environment.
With regard to other factors that potentially influence the risks of radon in homes and in mines, the three models also incorporate different assumptions for some factors. As noted, the ICRP model increases risk for exposures before age 20 yr and the NCRP model assumes that risk commences at age 40 yr. In the BEIR IV model, risk varies with attained age. With regard to smoking, the NCRP model is additive, whereas the other two models are multiplicative.
SUMMARY
The finding that radon and its decay products are invariably present in indoor environments has prompted concern that lung cancer caused by radon is a public health problem. At present, the risks of indoor radon can be estimated best by using risk coefficients derived from epidemiological studies of underground miners in risk models. However, application of the evidence from the mining environment to the indoor environment requires assumptions and introduces substantial uncertainty. Some uncertainties associated with this extrapolation can be estimated by comprehensive consideration of the dosimetry of radon decay products in the indoor and mining environments. However, potentially important biological factors that also introduce uncertainty are not addressed by dosimetric modeling.
REFERENCES
Arnsrein, A. 1913. Sozialhygienische Untersuchungen Über die Bergleute in den Schneeberger Kobaltgruben. Wein. Arbeit Geb. Soz. Med. 5:64-83.
Bale, W. F. 1980. Memorandum to the files, March 14, 1951: Hazards associated with radon and thoron. Health Phys. 38:1062-1066.
Cohen, B. L. 1986. A national survey of 222Rn in U.S. homes and correlating factors. Health Phys. 51:175-183.
Harting, F. H., and W. Hesse. 1879. Der lungenkrebs, die Bergkrankheit in den Schneeberger gruben. Vjschr. Gerichtl. Med. Offentl. Gesundheitswesen 31:102-132, 313-337.
Holaday, D. A., D. E. Rushing, R. D. Coleman, et al. 1957. Control of Radon and Daughters in Uranium Mines and Calculations on Biologic Effects. DHEW Publ. No. (PHS) 57-494. Washington, D.C.: U.S. Government Printing Office.
Hueper, W. C. 1966. Occupational and Environmental Cancers of the Respiratory Tract. New York: Springer-Verlag.
International Commission on Radiological Protection (ICRP). 1987. Lung Cancer Risk from Indoor Exposures to Radon Daughters. ICRP Publ. No. 50. Oxford: Pergamon Press.
James, A. C. 1988. Lung dosimetry. Pp. 259-309 in Radon and Its Decay Products in Indoor Air, W. W. Nazaroff and A. V. Nero, Jr., eds. New York: John Wiley & Sons.
Land, C. E. 1989. The ICRP 50 Model. Pp. 115-126 in Proceedings of the Annual Meeting of the National Council on Radiation Protection and Measurements. Bethesda, Md.: National Council on Radiation Protection and Measurements.
Logue, J., and J. Fox. 1985. Health hazards associated with elevated levels of indoor radon—Pennsylvania. Morbid. Mortal. Weekly Rep. 34:657-658.
Lubin, J. H. 1988. Models for the analysis of radon-exposed populations. Yale J. Biol. Med. 61:195-214.
Lubin, J. H., J. M. Samet, and C. Weinberg. 1990. Design issues in epidemiologic studies of indoor exposure to Rn and risk of lung cancer. Health Phys. 59:807-817.
Lundin, F. E., Jr., J. K. Wagoner, and V. E. Archer. 1971. Radon Daughter Exposure and Respiratory Cancer: Quantitative and Temporal Aspects. NIOSH-NIEHS Monogr. No. 1. Washington, D.C.: National Institute of Occupational Safety and Health-National Institute of Environmental Sciences.
National Council on Radiation Protection and Measurements (NCRP). 1984a. Exposure from the Uranium Series with Emphasis on Radon and Its Daughters. NCRP Report No. 77. Bethesda, Md.: National Council on Radiation Protection and Measurements.
National Council on Radiation Protection and Measurements (NCRP). 1984b. Evaluation of Occupational and Environmental Exposure to Radon and Radon Daughters in the United States. NCRP Report No. 78. Bethesda, Md.: National Council on Radiation Protection and Measurements.
National Research Council (NRC). 1988. Health Risks of Radon and Other Internally Deposited Alpha-Emitters. BEIR IV. Committee on the Biological Effects of Ionizing Radiation. Washington, D.C.: National Academy Press.
Nero, A. V., M. B. Schwehr, et al. 1986. Distribution of airborne 222Rn concentrations in U.S. homes. Science 234:992-997.
Pirchan, A., and H. Sikl. 1932. Cancer of the lung in the miners of Jachymov (Joachimsthal). Am. J. Cancer 4:681-722.
Rostoski, O., E. Saup, and G. Schmorl. 1926. Die Bergkrankheit der Erzbergleute in Schneeberg in Sacksen ("Schneeberger Lungenkrebs"). Z. Krebforsch. 23:360-384.
Samet, J. M. 1989. Radon and lung cancer. J. Natl. Cancer Inst. 81:745-757.
Seltser, R. 1965. Lung cancer and uranium mining: A critique. Arch. Environ. Health 10:923-935.
U.S. Department of Energy, Office of Energy Research, Office of Health and Environmental Research. 1989. International Workshop on Residential Radon Epidemiology, Washington, D.C. CONF-8907178. Springfield, Va.: National Technical Information Service.
U.S. Department of Health and Human Services. 1989. Reducing the Health Consequences of Smoking: 25 Years of Progress. A Report of the Surgeon General. Office on Smoking and Health, Center for Chronic Disease Prevention and Health Promotion, Centers for Disease Control, Public Health Service, U.S. Department of Health and Human Services. DHHS Publ. No. (CDC) 89-8411. Washington, D.C.: U.S. Department of Health and Human Services.
U.S. Environmental Protection Agency (EPA). 1986. A Citizen's Guide to Radon. What It Is and What To Do About It. EPA Publ. No. 86-004. Washington, D.C.: U.S. Government Printing Office.
U.S. Environmental Protection Agency (EPA). 1988. Final Report on the 1987 State Survey Results. Washington, D.C.: Office of Radiation Programs, U.S. Environmental Protection Agency.











