MORTALITY IN THE CHILDREN OF ATOMIC BOMB SURVIVORS AND CONTROLS1
JAMES V.NEEL
Department of Human Genetics, University of Michigan Medical School, Ann Arbor, Michigan 48104
HIROO KATO
Department of Statistics. Atomic Bomb Casualty Commission, Hiroshima, and Hiroshima Branch, Japanese National Institute of Health, Hiroshima, Japan
WILLIAM J.SCHULL
Center for Demographic and Population Genetics, Graduate School of Biomedical Sciences at Houston, The University of Texas, Houston, Texas 77025
Manuscript received July 15, 1973
Revised copy received October 24, 1973
ABSTRACT
A continuing study of mortality rates among children born to survivors of the atomic bombings and a suitable group of controls has been updated; the average interval between birth and verification of death or survival is 17 years. The mortality experience is now based on 18,946 children liveborn to parents one or both of whom were proximally exposed, receiving jointly an estimated dose of 117 rem; 16,516 children born to distally exposed parents receiving essentially no radiation; and 17,263 children born to parents not in Hiroshima or Nagasaki at the time of the bombings. No clearly significant effect of parental exposure on child's survival can be demonstrated either by a contingency X2 type of analysis or regression analysis. On the basis of the regression data, the minimal gametic doubling dose of radiation of this type for mutations resulting in death during (on the average) the first 17 years of life among liveborn infants conceived 0–13 years after parental exposure is estimated at 46 rem for fathers and 125 rem for mothers. On the basis of experimental data, the gametic doubling dose for chronic, low-level radiation would be expected to be three to four times this value for males and as much as 1000 rem for females.
ONE of the possible manifestations of exposure to ionizing radiation is a decrease in the life expectancy of children born to radiated parents, because of the induction of deleterious mutations in the gonadal tissues of these parents. We have previously described in detail a study of the mortality through a mean follow-up period of nine years among a group of children liveborn between 1946 and 1958 to survivors of the atomic bombings of Hiroshima and Nagasaki, and a suitable group of controls, reviewing also the pertinent literature (KATO, SCHULL
|
Reproduced, with permission, from Genetics, vol. 76, © 1974 by the Genetics Society of America. |
|
1 |
This study was undertaken as one aspect of the program of the Atomic Bomb Casualty Commission, a cooperative research agency of the U.S. National Academy of Sciences—National Research Council and the Japanese National Institute of Health of the Ministry of Health and Welfare, with funds provided by the U.S. Atomic Energy Commission, the Japanese National Institute of Health, and the U.S. Public Health Service. |
and NEEL 1966). No difference in mortality rates was demonstrated. The present report will extend the study for another eight years (through December 31, 1969), thus providing a near-complete accounting of what might be termed prereproductive mortality. Although the actual number of deaths since the prior review of the mortality experience is rather small, publication at this time is prompted by the additional fact that a significant revision of dose estimates has recently become available, thus facilitating a more meaningful estimate of the minimum dose of radiation necessary to increase by 100% the mutation rate resulting in this type of effect (i.e., the so-called doubling dose).
As originally defined, the study population consisted of three cohorts of children, drawn from both Hiroshima and Nagasaki, as follows:
THE STUDY POPULATION
|
Group I— |
either or both parents <2000 meters from hypocenter at time of bombing (ATB), |
|
Group II— |
neither parent <2000 meters but either or both >2500 meters from hypocenter ATB, and |
|
Group III— |
both parents not in city ATB. |
A detailed description of how these groups were assembled will be found in the earlier paper (KATO, SCHULL and NEEL 1966). The number of liveborn infants in each of the panels when they were established was reported as 17,721,17,870, and 17,828, respectively. Although we are interested in total zygote loss, stillborn infants have been excluded from the present study because a definitive analysis of stillbirth frequency between 1948 and 1953, the period during which the majority of the study births occurred, has been presented elsewhere; it revealed no relation of the phenomenon to parental radiation (NEEL and SCHULL 1956). The results of that analysis will not be altered by the present findings. Parents of group III children of course received no radiation exposure ATB. Furthermore, it has been clear for some time that the exposure received by the parents of group II children was negligible, usually less than 1 rad. Thus both groups II and III are “control” groups. However, for a variety of reasons it has proven difficult to estimate the dosage received by the parents of group I children.
Early attempts to estimate for genetic purposes the amount of radiation received by survivors relied heavily on such medical parameters as epilation and petechiae, in addition to distance from hypocenter and shielding (NEEL and SCHULL 1956). The only unclassified distance-dose curve then available was that published in “The Effects of Atomic Weapons” (Los Alamos Scientific Laboratory, 1950), for a “nominal” atomic bomb. The studies of RITCHIE and HURST (1959) and ARAKAWA (1960), together with the declassification of distance-dose curves specifically estimated for Hiroshima and Nagasaki, then resulted in the adoption by the Atomic Bomb Casualty Commission (ABCC), the joint U.S.-Japan official follow-up agency, of the so-called T57 Dose Scale, which assigned individual doses solely on the criteria of distance from hypocenter and shielding. This was the basis for the estimates of radiation exposure in our first publication
on the mortality experience of these three groups of children (KATO, SCHULL and NEEL 1966); we simply adopted for group I and group II parents the estimate of the mean dose to individuals falling in these distance categories. However, AUXIERet al. (1966) then published a revision of the two distance-dose curves, even as much more detailed shielding data were assembled. The result was the adoption by the ABCC of the T65 Dose Scale, based again solely on distance and shielding (MILTON and SHOHOJI 1968), and each surviving individual within 2500 meters ATB has now been assigned a dose. In developing this dose scale, independent estimates were made of gamma and neutron doses, and total dose expressed in rad's. The present analysis is based on these individual dose estimates.
However, not unexpectedly, some problems regarding the assignment of individual doses persist. Thus, a few individuals give apparently valid histories of radiation sickness when their T65 doses are well below those thought necessary to radiation sickness. Conversely, a few individuals who report proximity to the hypocenter with very little shielding ATB must on the T65 schedule be assigned doses clearly incompatible with survival. In both instances, this probably results from confusion about position or shielding or both at the time of the explosions. The distribution of estimated doses among the parents of group I children is such that the preponderance of estimated values fall in the range of 50 to 150 rad, although some few individuals have estimated values greater than 600 rad.
Among the parents of the subjects of this study there were, when the T65 dose schedule became available, instances where one or both parents were deceased or no longer resided in Hiroshima or Nagasaki, i.e., were not available for dosimetry studies. This was relatively unimportant in the case of parents of group II and III children, since it was clear that they were, for the purposes of this study, to be considered essentially unirradiated, but seriously impeded dose estimates for the parents of group I children. There were also a number of parents of group I children who were unable to supply adequate shielding or location data ATB. Unfortunately, for these various reasons, there were 2885 parents of group I or II children from whom no proper radiation history could be obtained. It is clear that the proximally exposed among these parents received considerable doses of radiation. The exclusion of their children from the study would result in a very real loss of data. Accordingly, such parents have been assigned a radiation dose on the basis of their own very approximate histories or of third-person histories. Where shielding data were available, they were incorporated into the dose estimate; where they were not, dose was assigned solely on the basis of distance from the hypocenter.
We turn now to the problem in dosimetry raised by the substantial neutron component in the Hiroshima spectrum but its lesser representation in Nagasaki (cf. JABLONet al. 1970). Three of the most reliable symptoms of radiation sickness are epilation, petechiae, and severe gingivitis. The Joint Commission (OUGHTERSONet al. 1951) reported that of 3506 survivors in Hiroshima living 20 days following the bombings, whose histories placed them within 2000 meters of the hypocenter (their exposure groups A, B, and C), 1223 (35%) reported
epilation and/or petechiae. Of 3471 similarly positioned survivors from Nagasaki, the corresponding figure was 1072 (31%). The Commission's data are not presented in such a manner that one can consider the frequency of individuals with one or more of the three cardinal symptoms just enumerated. Granted some confusion in the evaluation of radiation sickness, it seems the great majority of persons actually did develop these findings. The minimum dose thought to elicit these symptoms in man is the equivalent of 200 roentgens of gamma rays (Los Alamos Scientific Laboratory, 1950). These data suggest that the biologically effective dose (for these end points) was as great in Hiroshima as in Nagasaki.
Additional data on this point are available because the majority of the parents of group I children were drawn from the study of N EEL and SCHULL (1956). The mothers of these children registered their pregnancies well in advance of delivery, at which time a brief radiation history was obtained. Among 4245 mothers registering in Hiroshima who were within 2000 meters of the hypocenter at the time of the bombings, 660 persons (16%) reported epilation and/or petechiae and/or oral lesions, including necrotic gingivitis. The similar figure for Nagasaki for 1636 mothers was 329 persons (20%). We have discussed the inadequacy of these data in our study in some detail (NEEL and SCHULL 1956). Nevertheless, they provide a crude guide to biological dosimetry. Like the foregoing data collected just following the bombings, they suggest relatively little difference between the two cities.
If we calculate the rad dose in the two cities assuming an RBE for neutrons of 1, then the mean rad dose for individuals exposed within 2000 meters is estimated as 55.9 for Hiroshima and 136.7 for Nagasaki. This lower mean for Hiroshima is at variance with the facts, just presented, that within 2000 meters, the symptoms of radiation sickness were quite similar in the two cities. JABLONet al. (1970) have recently pointed out that there are striking discrepancies between the two cities in the relationship of symptomatology to T65 dose when the RBE for neutrons is taken as 1, but that these differences disappear when the RBE is placed at 4–5.
If we intend to recognize the neutron component in calculating the genetically effective dose, the RBE figure of greatest pertinence is specifically for genetic effects. The data for mammals are not so extensive as one might desire; we will accept RUSSELL'S appraisal that “in general, for a given absorbed dose, neutrons prove to be far more mutagenic than X and gamma rays, namely, of the order of 5 or 6 times both for oocytes and for the rising part of the dose curve for spermatogonia. This is for acute radiation” (RUSSELL 1967; see also SEARLE 1967; SEARLE, EVANS and WEST 1969; and SEARLE and PHILLIPS 1971). The RBE undoubtedly varies with both the absolute levels and the proportions of neutrons and gamma rays in the total dose. The RBE therefore varies from survivor to survivor. In this complex situation we will for now set the average RBE at 5 for the types of genetic damage (point mutations, small deletions, unbalanced translocation, nondisjunction) which we may assume are being measured in this study. Incorporation of an RBE of 5 for neutrons into the distance-shielding data necessitates that we now measure dosage in rem units. All estimates were rounded down to the nearest integral value.
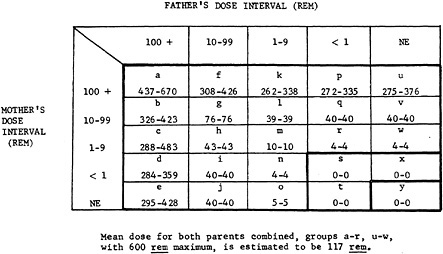
FIGURE 1.—The total mean dose in rem received by both parents combined for the 25 subclasses of children defined in this study. With those exceptions described in the text, children falling into subdivisions a–r and u–w correspond to group I children of the previous report, those in subdivisions s, t, and x, to group II, and those in y to group III. The average parental dose for all parents of group I children is estimated to be 117 rem; of group II, 0 rem; and of group III, 0 rem. The lower figure in each box is mean dose after reduction of all dose estimates greater than 600 rem to 600 rem; the higher figure is the estimate before that adjustment.
In order to project some better idea of the distribution of doses, and to facilitate the statistical analysis (see below), the radiated parents of group I and II children have been subdivided into four classes, according to the rem equivalents of the T65 dose, namely, <1 rem, 1–9 rem, 10–99 rem, and 100+rem. There is also the category of unexposed parents. With this subdivision 25 subclassifications of the total material result, shown in Figure 1. In general, the parents of group I children fall into subclassifications a-r and u-w, the parents of group II children into subclassifications s, t, and x, and the parents of group III children into subclassification y. The fact that parents in subgroups s, t, and x are shown as receiving no radiation when in fact they often received a fraction of a rem is due to the practice of rounding down to the nearest integer. The first entry in each box in Figure 1 represents the estimated mean dose in rems received by the father and mother together. In computing this, whenever the estimated T65 rem dose for an individual exceeded 600, it has been reduced to that figure, on the grounds that this is the approximate maximum dose of whole or near-whole body radiation compatible with survival, and doses estimated as greater than that result from confusion over position ATB or shielding or both. However, it has not been possible to introduce a corresponding counter-correction, for parents who in their histories may inadvertently have exaggerated the amount of shielding or distance from hypocenter ATB. The magnitude of this correction is indicated by the second figure in each box, which is the estimate of the mean before estimated doses greater than 600 rem were reduced to that level.
Ascertainment of the survival status of the children in each panel utilized the system of compulsory family registration (koseki) which has existed in Japan since the last quarter of the nineteenth century. All deaths in a family are routinely entered in this record. The koseki record is kept in that civil administrative unit which the family regards as its ancestral home (honseki). At the time the study was undertaken, the honseki had been established for each father. Thus, the death of any child in the study groups could be ascertained by consulting personally or by mail the koseki. In case of death, cause of death was updated for mortality through 1969. Because of the manner in which the groups were assembled, the age of the living children at the end of 1969 ranged from 11 to 23. In only 51 instances where the child was of Japanese parentage was it impossible to obtain information on child's survival; these children have been omitted from the analysis.
Mortality, especially infant mortality, is influenced by many variables, such as parents' age, birthweight, and socioeconomic conditions. If the three study groups differ significantly in regard to these conditions, failure to take this into consideration might lead to spurious inferences concerning radiation effects. Through city birth records and the records of the earlier genetic study from which the present mortality study evolved (NEEL and SCHULL 1956) it was possible in 1963 to obtain data on virtually all births in the panel with respect to maternal age, paternal age, parity, birthweight, duration of pregnancy, and, of course, year of birth. In an effort to extend this information, in 1964 a mail survey was undertaken of a random 10% of the parents in the total sample, with respect to present parental occupation, parental schooling, survival status of the parents, the size of the home (as measured by the standard module of Japanese mats) and the number of persons normally residing therein, food expenditures per person per month, and child's nutrition, as measured by consumption of such dietary items as fish, meat, eggs, and milk. A detailed analysis of these findings has already been presented (KATO, SCHULL and NEEL 1966). It can be summarized with the simple statement that in general the unexposed control parents (who came to Hiroshima and Nagasaki following the bombings) were slightly younger and had a little more education and somewhat higher occupation ratings than the exposed. The differences as tested by contingency x2 were of borderline statistical significance. In general, these differences in the distribution of extraneous socioeconomic variables, together with the fact that parents' ages are slightly older in the exposed groups than in the control groups, are such as might lead to an inflation of mortality rates in the children of the ‘heavily' (proximally) exposed parents as contrasted to the other comparison groups. However, the data do not permit a rigorous treatment of this possible bias. We return to this subject later.
STATISTICAL CONSIDERATIONS
Two analyses of the data have been undertaken. The first is a simple contingency x2 analysis of a 5x5 table defined by the radiation classes of father and mother as shown in Figure 1, the cell entries being the proportion of deceased among the children of parents with that radiation history. Expectations were corrected for age of child (i.e., year of birth), sex, and city.
The second has been a stepwise regression analysis (see, for example, DRAPER and SMITH 1966), based on 52,725 individuals, of the form
y=a+bx1+cx2+dx3+ex4
where y=probability of death,
x1=year of birth
x2=sex of child
x3=father's dose in rem, and
x4=mother's dose in rem
The variables have been entered into the analysis in the order of the amount of variance each accounts for, with a recomputation of the contribution of each preceding variable as each successive variable is added to the regression. The analysis was performed on an IBM 1440 computer.
THE DATA
Table 1 displays the results of the contingency x2 analysis. During the immediate post-war years, infant and childhood mortality declined significantly in Japan. In the composition of these study groups, the limiting factor on size of group was the number of children born to relatively heavily irradiated parents. There was an excess of potential controls among children falling into categories II and III. Accordingly in constituting the three groups, groups II and III were balanced with group I with respect to year of birth, and this factor does not confound this analysis. The expected number of deaths for each cell is calculated on the basis of the same probability of death for each age (and sex) balanced cell, based on the rate observed in the total sample. Traumatic (accidental) deaths (322 in number) have been included in the tabulations and analyses of Table 1, on the general thesis that such accidents often fall on prepared soil. However, an analysis which excludes them yields substantially the same result. The table also presents the total number of years at risk. The mean age of the subjects contributing to each cell can of course be derived by dividing the latter figure by the number of subjects. The contingency x2 is well below the level of significance.
When the numbers in the data cells were assembled to correspond as closely as possible to the definition of the three study groups followed in our first treatment of this material (by distance rather than dosage), there were certain discrepancies from the numbers given earlier (KATO, SCHULL and NEEL 1966). In the course of preparing this analysis, it was discovered that in the previous analysis, the stillbirths occurring in one segment of the data (GE-3) had, contrary to tabulation instructions, not been excluded from the base (denominator) in the calculation of rates. Since there were 694 stillbirths, this resulted, with cumulative mortalities at the time of the study of 6–7%, in an underestimate of the true rate of approximately 0.1%. This error, which in no way alters the conclusions of the earlier paper, has been corrected. Beyond this, in the change from a classification based on location ATB as obtained at the time of pregnancy registration to one based on radiation as estimated from a special study conducted some ten years later, there have been transfers of parents from one group to another. Some parents who on the original questionnaire administered as long ago as 1948 indicated absence from either city ATB, have now on subsequent contacts altered their history to presence. In part this may stem from confusion regarding the
TABLE 1 The mortality experience of the 25 groups of children defined in this study (Data are for both Hiroshima and Nagasaki, sexes combined. Doses given are in rem units)
|
Father |
|||||||
|
100+ |
10–99 |
1–9 |
<1 |
NE |
Total |
||
|
Mother |
|||||||
|
100+ |
20 |
17 |
10 |
28 |
186 |
261 |
Observed |
|
22.60 |
11.08 |
7.06 |
32.85 |
180.40 |
253.99 |
Expected* |
|
|
0.88 |
1.53 |
1.42 |
0.85 |
1.03 |
1.03 |
0/E |
|
|
323 |
169 |
110 |
511 |
2920 |
4033 |
No. Subjects |
|
|
6409 |
3165 |
2053 |
9718 |
53866 |
75211 |
P.Y.† |
|
|
10–99 |
18 |
61 |
19 |
55 |
239 |
392 |
Observed |
|
14.30 |
63.32 |
17.88 |
58.79 |
262.12 |
416.41 |
Expected* |
|
|
1.26 |
0.96 |
1.06 |
0.94 |
0.91 |
0.94 |
0/E |
|
|
220 |
855 |
266 |
880 |
4129 |
6350 |
No. Subjects |
|
|
4139 |
17569 |
5154 |
17007 |
77583 |
121452 |
P.Y.† |
|
|
1–9 |
16 |
16 |
31 |
31 |
123 |
217 |
Observed |
|
11.77 |
17.42 |
29.59 |
36.50 |
127.94 |
223.22 |
Expected* |
|
|
1.36 |
0.92 |
1.05 |
0.85 |
0.96 |
0.97 |
0/E |
|
|
180 |
259 |
413 |
567 |
2024 |
3443 |
No. Subjects |
|
|
3406 |
5028 |
8276 |
10730 |
37837 |
65277 |
P.Y.† |
|
|
<1 |
46 |
65 |
35 |
309 |
590 |
1045 |
Observed |
|
48.81 |
64.32 |
28.03 |
295.97 |
582.05 |
1019.18 |
Expected* |
|
|
0.94 |
1.01 |
1.25 |
1.04 |
1.01 |
1.03 |
0/E |
|
|
768 |
993 |
429 |
4268 |
9207 |
15665 |
No. Subjects |
|
|
14413 |
18806 |
8102 |
84069 |
172168 |
297558 |
P.Y.† |
|
|
NE |
73 |
67 |
37 |
195 |
1071 |
1443 |
Observed |
|
59.67 |
74.96 |
37.06 |
175.13 |
1098.37 |
1445.19 |
Expected* |
|
|
1.22 |
0.89 |
1.00 |
1.11 |
0.98 |
1.00 |
0/E |
|
|
1030 |
1281 |
619 |
3041 |
17263 |
23234 |
No. Subjects |
|
|
18240 |
23013 |
11234 |
53889 |
324390 |
430766 |
P.Y.† |
|
|
Total |
173 |
226 |
132 |
618 |
2209 |
3358 |
Observed |
|
157.15 |
231.10 |
119.62 |
599.24 |
2250.88 |
3357.99 |
Expected* |
|
|
1.10 |
0.98 |
1.10 |
1.03 |
0.98 |
1.00 |
0/E |
|
|
2521 |
3557 |
1837 |
9267 |
35543 |
52725 |
No. Subjects |
|
|
46607 |
67581 |
34819 |
175413 |
665844 |
990264 |
P.Y.† |
|
|
x2=22.46 d.f.=24 (expectation set by grand total). * Basis of survival of total sample. † P.Y.=person years. |
|||||||
precise city limits (often somewhat vaguely defined in Japan) and, since the radiation dose at the periphery of the city was minimal, this is a shift from one type of control to another. However, there were also 78 changes from a report of
TABLE 2 The regression of death among liveborn infants on year of birth, sex, father's radiation dose, and mother's radiation dose, as determined by a stepwise regression procedure (DRAPER and SMITH 1966)
|
Factor |
Partial correlation coefficient |
Proportion of variation explained (R2) |
Coefficient of regression |
|
Year of birth |
-0.08274 |
0.00684 |
-0.00588± 0.000307** |
|
Sex |
0.02031 |
0.00725 |
0.00988± 0.002119** |
|
Father's dose |
0.008516 |
0.00733 |
0.000029± 0.000015 |
|
Mother's dose |
0.000075 |
0.00733 |
0.000000± 0.000012 |
|
** p<0.01. |
|||
“not in city” to a report of a position in city ATB at which more than 1 rem unit was received, including 14 in which the conjoint dose exceeded 100 rem. To some extent this may be due to the fact that our original histories of parental radiation were usually obtained from the mothers alone, or her surrogate, whereas the later histories have been obtained directly from the persons concerned. Anyone familiar with the problem of obtaining accurate histories concerning highly emotional subjects will not be too surprised at these minor changes.
The results of the stepwise regression analysis are shown in Table 2. A year-of-birth effect on mortality emerges as quite significant, as does the well-known sex effect (male death rates greater than female in the early years of life). The regression of proportion of death on mother's exposure is non-significant, as is the regression on father's exposure, but the latter term is just below the 5% level. One might in this situation elect to apply a one-tailed test of significance. In this case, the regression becomes significant at the 5% level.
As noted earlier, a ten percent sample survey in 1964, some ten years after the atomic bombings, revealed small socioeconomic differences between families where one or both parents were exposed and families for which neither parent was exposed. Socioeconomic status changed rapidly in postwar Japan and the sample was relatively small, but the possibility had to be considered that these differences were influencing the variable of interest. The most appropriate way to examine this possibility is through a regression analysis which excludes the cell (y) in Figure 1 comprised of children both of whose parents were unexposed. The results of such an analysis are given in Table 3. Sample size is now reduced from 52,725 to 35,462. The differences between the results of the two analyses are negligible.
TABLE 3 The regression of death among liveborn infants on year of birth, sex, father's radiation dose, and mother's radiation dose after exclusion from the study material of those control children neither of whose parents were exposed ATB
|
Factor |
Partial correlation coefficient |
Proportion of variation explained (R2) |
Coefficient of regression |
|
Year of birth |
-0.08323 |
0.00693 |
-0.00598± 0.000378** |
|
Sex |
0.02008 |
0.00733 |
0.00983± 0.002600** |
|
Father's dose |
0.008578 |
0.00740 |
0.000031± 0.000019 |
|
Mother's dose |
0.001916 |
0.00740 |
0.000006± 0.000016 |
|
** p<0.01. |
|||
DISCUSSION
There is, then, no clear evidence thus far for an increased death rate in the children born to the survivors of the atomic bombings of Hiroshima and Nagasaki. These data are in keeping with most of the results on experimental mammals such as mice, rats, and pigs, where substantially larger amounts of radiation have been delivered, in some experiments over several generations, without the clear demonstration of net dominant deleterious effects (KOHN 1960; SUGAHARA 1964; FRÖLEN 1965; CHAPMANet al. 1964; COX and WILLHAM 1962; D.F.COX 1964; RODERICK 1964; but see RUSSELL 1955, and RUSSELL and RUSSELL 1959). These data also continue to be in keeping with other, more limited data on man (MACHT and LAWRENCE 1955; CROW 1955; KAPLAN 1958; D.W.COX 1964; TANAKA and OHKURA 1958), although in most of these series the numbers are so small and the dosage so low that no real test of hypothesis is involved. As is our custom, we enter the usual caveat, that this failure to demonstrate an unequivocal effect of this type does not constitute evidence there were no effects.
In a previous paper (KATO, SCHULL and NEEL 1966) employing preliminary dose estimates, these “negative” findings were used to generate a minimum estimate of the doubling dose for mutations resulting in death in liveborn carriers in the first generation after their origin. Using an arcsin transformation, the angular difference (as expressed in degrees) which could be demonstrated between two samples of size 17,721 (the size of group I) with respect to death rate was derived. It was shown that if the Type II error (ß) was set at 0.05, then on the hypothesis of an increased death rate among the children of heavily radiated parents (i.e., one-tailed significance test), the sample was large enough to detect an angular difference of 1.0. As the cumulative death rate in the two control groups among liveborn infants was close to 6.0% at the time of this evaluation, we could detect
a difference between the children in group I and either group II or III of approximately 0.8–1.0%.
In order to make a calculation of the doubling dose, we needed an estimate of the percent of liveborn children who each generation die before age 9 (the average age of that sample) because of mutation (point and chromosomal) in the preceding generation. This is an extremely difficult figure to generate in the present state of knowledge. In this study, pregnancies were generally registered during the sixth month of gestation, and if at the time of termination there was any sign of life whatsoever, the child was classified as liveborn. The data thus include terminations occurring during the last trimester. We have previously, on the basis of both theoretical and enumerative considerations, suggested that 0.005 of a cohort of liveborn children so defined will die before age 9 as a result of mutation in the preceding generation, and we will retain this estimate for this paper, although with improving standards of medical practice it may be on the high side. Half of this was assumed to be contributed by gross chromosomal mutation and half by so-called point mutation (which might include small deletions). Quite similar figures are found in the recent report of the Subcommittee on Genetic Effects of the Advisory Committee on the Biological Effects of Ionizing Radiations (1972). The calculation then proceeded as follows:
where a=joint parental dose,
b=mortality due to spontaneous mutation in the preceding generation,
c=increase in mortality excluded by present data, (conservatively taken to be 0.010), and
d=minimal doubling dose.
In view of the possibility that per unit exposure, the recovery of mutations from the offspring of irradiated males and females differ, it should be pointed out that this calculation tacitly assumes equal exposures of fathers and mothers, which is only approximately correct (Table 1). In the previous situation, it was calculated by this means and with these assumptions and facts that the doubling dose of radiation of this type to either or both parents for dominant mutations resulting in the death of liveborn infants prior to reproduction was not less than 50 rad. Since the data on the neutron component of the radiation spectrum of the atomic bombs were not yet available, this calculation was in terms of rad rather than rem units, and so did not take into consideration the greater RBE of neutrons. In the present situation, with the new data on dosage, we can modify the previous estimate as follows:
The most vulnerable aspect of this calculation is the estimate that 0.005 of liveborn infants will die prior to maturity because of chromosomal and “point” mutation in the preceding generation. Given our present knowledge of dominant mutation and chromosomal abnormalities, this figure can scarcely be below 0.003. On the other hand, the radiation dose is probably conservative, because of
the correction downward to an estimate of 600 of all individual T65 rem dose estimates above 600, without a corresponding correction for spuriously low estimates. This correction avoids the embarrassment of assigning to individuals doses of acute whole-body radiation obviously incompatible with survival. However, without this correction, the estimate in rem for those cells where one or both parents received 100+rem units would be 63 to 233 rem higher (Table 1). We have also employed the conservative figure of 5 for the genetic RBE of fast neutrons. Let us assume the average total dose was only 20 rem greater. Then, taken with the above mentioned 0.003, the minimal estimate of doubling dose becomes
Thus the two most probable sources of error in the estimate tend to be partially compensating.
It should perhaps be emphasized that this estimate is for zygotic doubling dose, i.e., the amount of radiation necessary to produce the number of mutations which as a result of spontaneous mutation are “normally” present in those zygotes which result in liveborn infants. We are indebted to DR. JAMES CROW for reminding us that the usual estimates of doubling dose, for such organisms as Drosophila and the mouse, are gametic doubling dose. The conversion is achieved by dividing the foregoing estimates by a factor of 2.
The fact that the present analysis is of the regression type permits an alternative and more powerful approach to the question of the doubling dose than the simple contrast of two groups. If the hypothesis is that parental radiation can only increase the death rate in children (one-tailed significance test), then from Table 2 we see that for fathers the data exclude at the 5% level a regression term greater than 0.000029+1.645×0.000015=0.000054, and on the assumption that the father contributes half of the mutations resulting in prereproductive death among liveborn infants, the minimum* gametic doubling dose is ![]() For mothers the corresponding regression value excluded at the 5% level is 0.000020, and the minimum gametic doubling dose is estimated to be 125 rem. In a situation where the parents are radiated equally, the minimum average conjoint zygotic doubling dose is 67 rem. Now the age span covered by this estimate is, on average, the first 17 years of life.
For mothers the corresponding regression value excluded at the 5% level is 0.000020, and the minimum gametic doubling dose is estimated to be 125 rem. In a situation where the parents are radiated equally, the minimum average conjoint zygotic doubling dose is 67 rem. Now the age span covered by this estimate is, on average, the first 17 years of life.
As described earlier, an analysis has also been performed excluding the children of unexposed parents. The estimates of minimum gametic doubling dose based on this analysis is essentially the same for the fathers (44 rem) but somewhat lower for mothers (77 rem). The differences in the magnitude of the coefficients of regression in the two analyses are well within random fluctuation, and because of the greater precision of estimation afforded by the larger numbers, we prefer the analysis of Table 2. However, a slightly more conservative estimate of minimal doubling dose (but with less precision) emerges from the analysis of Table 3.
|
* |
The term “minimum” as here employed is synonymous with “97.5% lower confidence limit”. |
As noted above, the effects of paternal radiation approach significance, and, in fact, with a one-tailed significance test, are significant at the 5% level. In a question of this importance, we believe a finding should be unequivocally significant before being trumpeted as such. It is worth noting, however, that if with the further accumulation of data this regression became significant, while not departing greatly from its present value, then the estimate of the gametic doubling dose for male radiation of this type, on the assumption that 0.0025 of liveborn infants die prior to the age of reproduction because of spontaneous mutation in their fathers in the preceding generation, becomes approximately ![]() For the present, it is more prudent to be guided by the doubling dose which the data exclude.
For the present, it is more prudent to be guided by the doubling dose which the data exclude.
The approximate nature of these calculations is obvious. They apply to only one type of genetic effect, albeit the one which probably has maximum psychological effect on human populations, since early deaths of the type under study here are often accompanied by physical defect and chronic disease. We recognize that in the experimental investigations of the mouse, the doubling dose for genetic effects of the type here under consideration has been higher than for a variety of other types of genetic effects (summary in LÜNING and SEARLE 1971) and the same may yet be shown to be true for man. For the present we note only that these estimates of minimal doubling dose for males and females fall within the range of recent consensuses from the study of other mammals (UNITED NATIONS 1972; ADVISORY COMMITTEE 1972). The need for more readily quantifiable and more objective measures of genetic damage in man continues to be great, and may be met by recent technical developments which permit large-scale screening of exposed populations for mutations affecting protein structure (cf. NEEL 1971; NEEL, TIFFANY and ANDERSON 1973).
The subjects of this study were conceived within one month to thirteen years after parental exposure. RUSSELL (1965) has shown that although per unit exposure the recovery of mutations is as great or greater in the offspring of radiated females as of radiated males for offspring conceived during the first six weeks following exposure, there was no evidence of an increased mutation rate among later litters of these same females. It is unknown whether this same phenomenon applies to the human female or, if so, what the appropriate time scale is. The data of the present study are consistent with the male-female difference observed in mice, although far from the level of significance. It may be anticipated that in the case of man, there will on the average be a considerable interval between exposure and reproduction (assuming cumulative exposures during the prereproductive and reproductive periods). The present data therefore seem to be the type appropriate to the calculation of a doubling dose for deleterious genetic effects from high intensity, short burst radiation. However, it must also be borne in mind that in the mouse the mutational yield from spaced or low dose radiation appears to be less than that from single-dose, quickly administered radiation by a factor of 3 or 4 in the male and 20 in the female, the latter figure still based on relatively scanty data (RUSSELL, RUSSELL and KELLEY 1958;
RUSSELL, RUSSELL and CUPP 1959; RUSSELL 1963; SEARLE and PHILLIPS 1968; BACHELOR, PHILLIPS and SEARLE 1969; LYON, PHILLIPS and BAILEY 1972). Thus, under the usual conditions of human exposure (small, widely spaced doses of radiation), if human spermatogonia and oocytes respond to radiation as do those of the mouse, with the present argument the gametic doubling dose of radiation for mutations resulting in pre-reproductive mortality in liveborn infants (sexes combined) is unlikely to be less than 138 rem for males and over 1000 rem for females.
LITERATURE CITED
Advisory Committee on the Biological Effects of Ionizing Radiations , National Academy of Sciences—National Research Council, 1972. The effects on populations of exposure to low levels of ionizing radiation. National Research Council, Washington.
ARAKAWA, E.T., 1960Radiation dosimetry in Hiroshima and Nagasaki atomic bomb survivors . New Engl. J.Med.263:488–493.
AUXIER, J.A., J.S.CHEKA, F.F.HAYWOOD, T.O.JONES and J.H.THORNGATE, 1966Free-field radiation-dose distributions from the Hiroshima and Nagasaki bombings. Health Physics12:425–429.
BACHELOR, A.L., R.J.S.PHILLIPS and A.G.SEARLE, 1969The ineffectiveness of chronic irradiation with neutrons and gamma rays in inducing mutations in female mice. Brit. J. Radiol.42:448–451.
CHAPMAN, A.B., J.L.HANSEN, G.B.HAVENSTEIN and N.E.MORTON, 1964Genetic effects of cumulative irradiation on prenatal and early postnatal survival in the rat. Genetics50: 1029–1042.
COX, D.F., 1964Effects of radiation on litter size in swine. Genetics50:1025–1028.
COX, D.F. and R.L.WILLHAM, 1962Genetic effects of irradiation on early mortality in swine. Genetics47:785–788.
COX, D.W., 1964An investigation of possible genetic damage in the offspring of women receiving multiple diagnostic pelvic X-rays. Amer. J.Hum. Genet.16:214–230.
CROW, J.F., 1955A comparison of fetal and infant death rates in the progeny of radiologists and pathologists. Amer. J.Roentgenol.73:467–471.
DRAPER, N.R. and H.SMITH, 1966Applied Regression Analysis.Wiley, New York.
FRÖLEN, H., 1965The effect on the length of life in the offspring of X-irradiated male mice. Mutation Res.2/3:287–292.
JABLON, S., S.FUJITA, K.FUKUSHIMA, T.ISHIMARU and J.A.AUXIER, 1970RBE of neutrons in atomic bomb survivors: Hiroshima-Nagasaki. Atomic Bomb Casualty Commission Technical Report 12–70. p. 36. Also: Symposium on Neutrons in Radiobiology, Nov. 11–14, 1969. Oak Ridge, Tenn. USAEC Conf.—691106. pp. 547–577.
KAPLAN, I.I., 1958The treatment of female sterility with X-ray therapy directed to the pituitary and ovaries. Amer. J.Obstet. Gynecol.76:447–453.
KATO, H., W.J.SCHULL and J.V.NEEL, 1966A cohort-type study of survival in the children of parents exposed to atomic bombings. Amer. J.Hum. Genet.18:339–373.
KOHN, H.I., 1960The effect of paternal X-ray exposure on the secondary sex ratio in mice (F1 generation). Genetics45:771–778.
Los Alamos Scientific Laboratory, 1950The Effects of Atomic Weapons.G.P.O., Washington.
LÜNING, K.G. and A.G.SEARLE, 1971Estimates of the genetic risks from ionizing radiation. Mutation Res.12:291–304.
LYON, M.F., R.J.S.PHILLIPS and H.J.BAILEY, 1972Mutagenic effects of repeated small radiation doses to mouse spermatogonia . Mutation Res.15:185–190.
MACHT, S.H. and P.S.LAWRENCE, 1955National survey of congenital malformations resulting from exposure to roentgen radiation. Amer. J.Roentgenol.73:442–466.
MILTON, R.C. and T.SHOHOJI, 1968Tentative 1965 radiation dose estimation for atomic bomb survivors . ABCC Technical Report 1–68.
NEEL, J.V., 1971The detection of increased mutation rates in human populations. Perspectives Biol. Med.14:522–537.
NEEL, J.V. and W.J.SCHULL, 1956The Effect of Exposure to the Atomic Bombs on PregnancyTermination in Hiroshima and Nagasaki.National Academy of Sciences—National Research Council, Publ. 461 : Washington, D.C.
NEEL, J.V., T.O.TIFFANY and N.G.ANDERSON, 1973Approaches to monitoring human populations for mutation rates and genetic disease, pp. 105–150. In: Chemical Mutagens: Principles and Methods for their Detection, Vol. 3. Edited by A.HOLLAENDER. Plenum Press, New York .
OUGHTERSON, A.W., G.V.LEROY, A.A.LIEBEN, E.C.HAMMOND, H.C.BARNETT, J.D.ROSENBAUM and B.A.SCHNEIDER, 1951Medical Effects of Atomic Bombs.The Report of the Joint Commission for the investigation of the Effect of the Atomic Bomb in Japan. Army Institute of Pathology. 5 vols.
RITCHIE, R.H. and G.S.HURST, 1959Penetration of weapons radiation, application to the Hiroshima-Nagasaki studies. Health Physics1:390–404.
RODERICK, T.H., 1964Summary of the general discussion of the symposium. Genetics50: 1213–1217.
RUSSELL, W.L., 1955Genetic effects of radiation in mice and their bearing on the estimation of human hazards, pp. 382–383. In: Proc. Int. Conf. on the Peaceful Uses of Atomic Energy, Vol. II. United Nations, New York.—, 1963The effect of radiation dose rate and fractionation on mutation in mice. pp. 205–217. In: Repair from Genetic Radiation. Edited by F.SOBELS. Pergamon Press, Oxford.—, 1965Effect of the interval between irradiation and conception on mutation frequency in female mice. Proc. Natl. Acad. Sci. U.S.54:1552–1557.—, 1967Factors that affect the radiation induction of mutations in the mouse . Ann. Acad. Brasil, Cienc 39 (Suppl.): 66–75.
RUSSELL, W.L. and L.B.RUSSELL, 1959Radiation-induced genetic damage in mice. pp. 179– 188. In: Progress in Nuclear Energy, Vol. II, Series VI, Biological Sciences. Pergamon Press, London.
RUSSELL, W.L., L.B.RUSSELL and M.B.CUPP, 1959Dependence of mutation frequency on radiation dose rate in female mice. Proc. Natl. Acad. Sci. U.S.45:18–23.
RUSSELL, W.L., L.B.RUSSELL and E.M.KELLEY, 1958Radiation dose rate and mutation frequency. Science128:1546–1550.
SEARLE, A.G., 1967Progress in mammalian radiation genetics, pp. 469–481. In: Intern. Congr.of Radiation Research 1966. Edited by G.SILINI. North Holland Publishing Co., Amsterdam.
SEARLE, A.G. and R.J.PHILLIPS, 1968Genetic insensitivity of the mouse dictyate oöcyte to chronic irradiation , pp. 17–25. In: Effects of Radiation on Meiotic Systems.International Atomic Energy Agency, Vienna.—, 1971The mutagenic effectiveness of fast neutrons in male and female mice . Mutation Res.11:97–105.
SEARLE, A.G., E.P.EVANS and B.J.WEST, 1969Studies on the induction of translocations in mouse spermatogonia . Mutation Res.7:235–240.
Subcommittee on Genetic Effects, Advisory Committee on the Biological Effects of Ionizing Radiations , 1972.
SUGAHARA, T., 1964Genetic effects of chronic irradiation given to mice through three successive generations. Genetics50:1143–1158.
TANAKA, K. and K.OHKURA, 1958Evidence for genetic effects of radiation in offspring of radiological technicians. Jap. J.Hum. Genet.3:135–145.
United Nations Scientific Committee on the Effects of Atomic Radiation , 1972Ionizing radiation: Levels and Effects, Vol. 2. United Nations, New York. Corresponding Editor: G.LEFEVRE

















