11
Economics of Malaria Control
WHERE WE WANT TO BE IN THE YEAR 2010
There will be a great change in the way economic reasoning and analysis are applied to malaria. Health priorities no longer will be set according to the absolute burden of disease, but will be based on what can actually be done about a given disease problem. Average cost-effectiveness, once an ubiquitous and poorly applied tool of analysis, will have been discarded. Improved collection and better use of data will allow for more rigorous analyses of the marginal effectiveness and costs of programs. Policymakers will recognize that malaria is a number of different diseases and that control interventions must be situation specific. Further, they will have a better understanding of how various policy instruments, applied at different levels of intensity, affect disease outcomes.
WHERE WE ARE TODAY
Applications of Economics to Malaria
Research on the economics of malaria has traditionally fallen into two categories: studies that document the economic burden (costs) of the dis-
ease, and those that examine the cost-effectiveness of interventions aimed at preventing or controlling the disease.
The Economic Costs of Malaria
The primary cost of malaria is its contribution to mortality and morbidity. Because precise data on morbidity and mortality are often lacking, it is not surprising that estimates of the disease's economic impact are vague and imprecise. A number of factors make calculating economic impact very difficult. For one, the disease affects each geographic region, and the individual communities within them, differently. The economic impact of malaria-related death also varies according to the age of those who die. In Africa and other highly endemic regions, where most deaths are among infants and young children, the impact on productivity is lower than in areas of low to moderate endemicity, where the burden of disease falls primarily on adults, who are the main breadwinners or primary caretakers (Over et al., 1990).
Most economic impact studies attempt to measure the effects of episodes of malarial illness on lowered worker productivity, a literature reviewed by Barlow and Grobar (1986). Researchers have estimated that each case of malaria causes between 5 and 20 days of disability (Van Dine, 1916; Russell and Menon, 1942; Malik, 1966; Conly, 1975). A common convention in the literature has been to use seven days of work lost per bout of malaria whenever this parameter is needed to assess a program and cannot be independently estimated (Sinton, 1935, 1936; Quo, 1959; San Pedro, 1967-1968; Niazi, 1969).
A number of studies have attempted to measure malaria's impact on worker output. One study found that malaria prevalence did have an effect on rice production (Audibert, 1986) and another that found farm families with malaria cleared 60 percent less land than did families free of the disease (Bhombore et al., 1952).
Other researchers have shown that in agricultural settings malaria can influence the choice of crops. Conly (1975) observed that farmers replaced higher-value crops, whose growing season coincided with the peak malaria season, with lower-value crops with different growing seasons. A similar analysis revealed that new settlers in agricultural areas may decide, a priori, to plant crops that are less sensitive to interruptions in cultivation (e.g., root crops) to avoid the potential impact of malaria (Rosenfield et al., 1984). De Castro (1985) showed that families with at least one sick member tended to shift the workload to healthy family members, thereby reducing (and also spreading) the net economic costs of the disease.
Despite the work of these and other researchers, the precise effect of malaria on productivity is still an open question. Using direct measure-
ments of physical ability, for example, at least one study demonstrated that malaria had no effect at all on productivity (Brohult et al., 1981).
Another approach focuses on the potential economic benefits of new agricultural or industrial development made possible by malaria control efforts (Wernsdorfer and Wernsdorfer, 1988). For example, successful malaria control efforts may allow workers to enter previously malarious mining areas, thereby increasing an individual's or company's earnings. If successful control efforts were not undertaken, the costs of malaria in this scenario would be the potential (but unearned) profits that would have accrued from the new mining activity (Griffith et al., 1971). In a similar vein, Sinton (1935, 1936) documents a number of cases in India where malaria prevented expansion into new areas, with substantial losses in forgone earnings.
In a recent study of the impact of malaria in four African countries (Burkina Faso, Chad, Rwanda, and the Congo) a distinction was made between “direct costs” (the out-of-pocket expense of treatment, including transportation and allied expenses) and “indirect costs” (earnings forgone by adults while sick or while caring for sick children, and discounted future earnings lost due to child mortality) (Shepard et al., 1990). In these countries, between three and seven days of production were lost per case of malaria. Extrapolating to the whole of Africa, an average of 2.1 days of output per person were lost, or $1.70 per capita per year.
All of these analytical approaches have shortcomings. For instance, the focus on days lost from work or output forgone is too narrow. A more appropriate measure of “cost” might be the “compensating variation,” or the amount of money necessary to return a sick individual to the same level of utility, happiness, satisfaction, and sense of well-being enjoyed prior to the onset of disease. Such a measure would capture the subjective and psychological impact of the illness and would be more inclusive than simply looking at the effect of malaria on worker productivity. Regardless of its appeal, there are no good methods for obtaining this number.
It is possible, however, to infer a lower bound for compensating variation by calculating the total costs of malaria treatment borne by families and individuals. These costs include not only payment for treatment but also the time and money spent on transportation, family care for patients, and preventive measures at the household and community levels. All of these calculations are affected by other variables, of which the most important may be access to appropriate care, and are subject to significant underestimation. (For example, there may be costs incurred by the individual before formal treatment is sought, and some individuals may decide that the costs of seeking treatment and forgoing income outweigh the uncertain costs of letting the disease run its course.)
To illustrate how this approach can be applied, a study in Thailand
(Kaewsonthi, 1989) estimated, among other things, patient costs associated with seeking local treatment for malaria prior to receiving care at a government-supported health center. These “external” expenses amounted to an average of $20 per positive case, nine times the average Thai minimum daily wage. For Thailand as a whole, external costs equaled about 45 percent of what the government itself spent to treat these patients. This is probably a considerable underestimation of actual patient costs, since productive time lost before and after seeking treatment—which may depend in part on the speed of the service provided and varies within and among countries—was not taken into account.
It is clear from this review of the literature that the analytical approaches used to study the costs of malaria are incomplete and difficult to compare with one another. The result, as noted by Andreano and Helminiak (1988), is that “we remain woefully ignorant of the social and economic effect of malaria in those countries of the world where it is prevalent.”
Costs and Effects of Malaria Control
While there has been progress in estimating the economic burden of malaria, estimates of the relative costs and impacts of interventions used to control the disease are less satisfactory. The predominant analytical method has used cost-effectiveness to help policymakers choose among policy alternatives. The primary measure, the cost-effectiveness ratio, represents the costs of a given intervention divided by a given outcome, with the outcome usually expressed in terms of the number of cases prevented (lives or life-years saved, the latter sometimes corrected for quality and with future years discounted).
Average cost-effectiveness ratios are widely used, but they can be quite misleading and should be viewed with great caution (Doubilet et al., 1986). Barlow and Grobar (1986) and Mills (1987) have summarized the results of a number of studies that calculated the costs per year of life saved (Table 11-1) and cost-benefit ratios (Table 11-2) for malaria control efforts in various countries over the past two decades. Table 11-1 also includes calculations of the cost per discounted quality adjusted life-years (QALY) saved by the program under analysis (Jamison and Mosley, 1990).1
These data are most striking because of their variability; it is hard to make any generalizations about them. The costs per case prevented ranged from $2.10 to $260 (in 1987 dollars), and cost-benefit ratios ranged from 2.4 to 150. The higher cost-benefit figures make malaria control seem of
|
1 |
QALYs attempt to adjust for sickness or premature death. Healthy years or days are given higher weights than unhealthy days or years. Often, as with basic accounting principles, the value of future years is discounted to the present. |
TABLE 11-1 Cost Effectiveness of Malaria Control
|
Cost (1987$) |
|||||
|
Author(s) |
Country |
Method |
per Case Prevented |
per Death Averted |
per Discounted QALY Saved |
|
Barlow (1968) |
Sri Lanka |
Insecticide |
78 |
2.80 |
|
|
Cohn (1973) |
India |
Insecticide |
2.10 |
7.00 |
|
|
Gandahusada et al. (1984) |
Indonesia |
Insecticide |
83-102 |
275-618 |
|
|
Hedman et al. (1979) |
Liberia |
Vector control and chemotherapy |
14 |
143 |
|
|
Kaewsonthi and Harding (1984) |
Thailand |
Vector control and chemotherapy |
27-74 |
90-760 |
|
|
Mills (1987) |
Nepal |
Vector control and chemotherapy |
1.30-172 |
2.80-255 |
|
|
Molineaux and Gramiccia (1980) |
Nigeria |
Vector control and chemotherapy |
259 |
1,500-2,650 |
|
|
Ortiz (1968) |
Paraguay |
Insecticides |
60 |
71 |
|
|
Walsh and Warren (1979) |
LDCs |
Vector control |
990 |
34 |
|
|
SOURCES: Barlow and Grobar (1986); Mills (1987); authors' calculations. |
|||||
TABLE 11-2 Cost-Benefit Ratios in Malaria Control
|
Author(s) |
Country |
Method |
Cost-Benefit Ratio |
|
Barlow (1968) |
Sri Lanka |
Insecticide |
146 |
|
Griffith et al. (1971) |
Thailand |
Chemoprophylaxis |
6.5 |
|
Khan (1966) |
Pakistan |
Eradication program |
4.9 |
|
Livadas and Athanassatos (1963) |
Greece |
Eradication program |
17.3 |
|
Niazi (1969) |
Iraq |
Eradication program |
6.0 |
|
Ortiz (1968) |
Paraguay |
Insecticides |
3.6 |
|
San Pedro (1967-1968) |
Philippines |
Eradication program |
2.4 |
|
Democratic Republic of Sudan (1975) |
Sudan |
Control program |
4.6 |
|
SOURCE: Barlow and Grobar (1986). |
|||
utmost economic importance, while the lower figures make malaria control appear no more urgent than a number of other government programs.
Unfortunately, there are no simple methodological explanations for this variability that could guide future cost-benefit calculations. Differences in data quality, the assumptions used in the analyses, the definition of relevant costs, the length of the study period, the discount rate applied, and the coverage and purpose of the original intervention all contribute to, but do not account for, the variation. For example, the Garki Project study (Molineaux and Gramiccia, 1980), which generated an estimate of $260 saved for every case of malaria prevented per year, included as part of overall project costs the expenditures on research and monitoring that accompanied the intervention. Some studies added in administrative costs to their computations, while others included only the cost of materials. While some calculations were based on small pilot projects (Gandahusada et al., 1984), others were based on large national efforts (Barlow, 1968). Finally, case fatality rates were assumed for those countries with data only on cases averted per year. Costs per QALY are sensitive to these assumptions. 2
Even if standardized research methods were used, the cost-benefit and cost-effectiveness ratios from these studies would vary greatly. That is because this variation has more to do with the inherent variability of malaria than with differences in research design. There are four underlying reasons for the wide variations seen in these cost-effectiveness ratios: levels of endemicity, temporal variability, organization structure, and returns to scale.
Levels of Endemicity Differences in the ecologic, epidemiologic, and social characteristics of each study region help determine the effectiveness of control programs per dollar spent (see Chapter 10). While the most crucial of these is the degree of endemicity, other factors, such as the behavior of the mosquito population, the presence of nonimmune individuals, and the degree of parasite resistance to chloroquine, also contribute.
Temporal Variability Depending on the degree of endemicity, there can be substantial variations in malaria prevalence within the same region at different times during the year or between years. Since costs per death or illness averted are likely to vary inversely with the prevalence rate, the usefulness of cost calculations based on a single year is questionable. Consequently, policy options should be evaluated on the basis of their “expected” value, taking into account the distribution of prevalences at different times. This is particularly important if costs include a fixed component, such as the administrative costs for control organizations, that does not depend on the disease prevalence in any one year.
|
2 |
Following Lancaster (1990), case fatality rates were assumed to be 1 percent for India, 0.25 percent for Liberia and Nigeria, 3 percent for Paraguay, and a range of 0.2 to 1 percent for the rest of Asia, based on the figure for India and data from Malaysia. |
Organizational Structure Control programs may be highly structured and single-purpose (vertical), or they may be integrated with other components of the health care system and somewhat unstructured (horizontal). Assessing control policies in integrated settings requires that the costs of providing general health care to be separated from costs specific to malaria control. As a rule, the higher the volume of cases, the more similar the costs of integrated and free-standing programs are likely to be (Mills, 1987). In areas with few cases, however, integrated programs can cost considerably less, since health personnel can more efficiently serve patients with different health needs, as demand requires.
Returns to Scale The intensity of the intervention has a substantial effect on both costs and effectiveness. The concept of returns to scale is useful for determining what level of intervention is appropriate, given its expected effect on some desirable outcome. Although certain costs are relatively fixed (facilities, staff salaries), others increase as the workload increases. In addition, many malaria control programs that rely on single interventions experience diminishing returns. The expense of vector control activities, for example, will increase as the program moves into peripheral areas where there are fewer people and, possibly, fewer mosquitoes per unit area covered. Similarly, the costs of case management will rise when there is a shift from passive to active case detection. Analyses that estimate costs only at a single level of intensity will be of limited value if extrapolations are made to higher levels of intensity. Barlow and Grobar (1986) suggest that because cost estimates for parasitic disease control programs are uncertain, a combination of policies (a “portfolio” of instruments) should be used. However, even if program personnel were armed with perfect knowledge of the costs and effects of various strategies, a diversified approach would still be desirable given the reality of diminishing returns to any single instrument (see Example 3 below).
Operational Research
Much of the best recent research on the economics of malaria control has been designed to answer specific questions about service delivery in specific geographic areas. Using this type of operational research it is relatively easy to gauge costs, since changes in scale are not an issue. Incremental benefits can also be assessed in relation to local epidemiology
and institutional and administrative conditions. Such exercises can greatly improve resource allocation and program decisions by local-level managers.
In two studies (Kaewsonthi, 1989; Mills, 1987), comparisons among techniques of vector control, and between vector control and therapy, were illuminated by carefully calculating the costs at the local level. The authors also were able to propose a set of practical recommendations based on their analyses. Mills, for example, was able to suggest reducing active malaria case detection and increasing investment in malaria clinics (or other treatment-based facilities) in Nepal. According to that study, either alternative was more cost-effective than spraying pesticide to kill mosquitoes.
Although few studies have done so, operational research can be used to assess “decreasing effectiveness”—the point at which the cost-effectiveness ratio of a given intervention drops enough to merit a reevaluation of its worth. Using such a method, Ettling et al. (1990) documented the increasing costs of expanding clinic coverage in a district in Thailand, making clear the trade-off between numbers of cases treated and the cost per case. Researchers at the Centers for Disease Control (Sudre et al., 1990) examined the relative costs of alternative drug therapies under differing degrees of chloroquine resistance. The salient variables in this decision-making process, including patient compliance with drug regimens, were also identified.
A review of the regulation and control of pharmaceuticals in Africa south of the Sahara (Foster, 1990) highlights the savings and improved health resulting from the effective use of drugs, and the costs of misdiagnosis, prescribing errors, and compliance failure. Another study took a more theoretical approach: the relative merits of over-the-counter and prescription drug sales were examined, and the possible loss of accurate drug use of the former was compared with its potential for increasing access and affordability (Hammer, in press). Other ongoing work has highlighted the problems of malaria-related infant and child mortality in endemic regions of Africa. One conclusion emerging from that work is that prenatal prophylaxis, undertaken early in pregnancy and especially for first pregnancies, can reduce low birth weight and infant mortality. From a policy perspective, this raises the issue of how women who have not felt sick from malaria for years and who do not normally seek prenatal care can be convinced to take preventive measures. All of these studies suggest that improved education of prescribers or patients about drug therapies can have beneficial effects.
Economics and Policymaking
In any analysis of any government program, such as malaria control, it is important to realize that different decision makers have fundamentally
different perspectives. The top levels of government, for example, must weigh the value of resources put into the health sector as opposed to other sectors, such as education or agriculture. Within the health sector, policymakers must choose between malaria and other diseases. Administrators of malaria control or primary health care programs, for their part, must allocate resources among a variety of interventions, such as spraying or case management. Local project managers may have to select among different vector control interventions or drug protocols.
Studies of the economic costs of malaria control generally do not provide the information that would be most useful to policymakers. In lieu of firm quantitative estimates of the costs of various malaria control interventions, qualitative judgments (with variable degrees of documentation) are often made. This approach may be adequate in many cases, but local decision makers would be greatly aided if more precise estimates were available. Further, to be truly effective, decisions affecting policy must take into account what is truly within the control of the policymaker. The relationships among the various instruments available to the policymaker, the objectives of the program, and program costs also must be determined.
Determining Policy Options
Determining what policy options (instruments) are available for disease control is more complicated than it seems. Epidemiologic research typically identifies risk factors associated with a specific disease; however, the degree to which these risk factors can be affected by policy is not always clear.
Instruments In the case of malaria, while it may be possible to identify the effect of vectorial capacity on the incidence of disease, neither vectorial capacity nor any of its components are directly controllable by policymakers (see Chapter 7), nor do policymakers control the number or proportion of houses sprayed with pesticide or, by extension, the death rate of mosquitoes. The relevant instrument actually is the hiring and training of sprayers; whether they actually spray houses, or do so successfully, is another matter. Similarly, for drug therapy, although the technical effectiveness of a specific protocol may be well documented, the degree to which that protocol affects disease prevalence depends on how many people seek treatment and how well they comply with the drug regimens. In both of these instances, the intervening variable is a function of some kind of managerial or monitoring capability, the presence or absence of which determines how effectively the policy instrument affects the program in question.
In fact, many aspects of malaria case management depend on the structure of the health care system. The government's role and ability to influ-
ence outcomes thus are quite context specific. Malaria policy in situations in which health care is provided within the family or by private-sector traditional healers, physicians, nurses, or pharmacists will be different than in cases in which the bulk of services are provided by public primary health facilities or malaria clinics. The instruments applied in the former might be taxes, subsidies, or information campaigns geared to encouraging the use of certain drugs; in the latter, the instrument might be drug protocols that more directly control the types and dosages of drugs used. For example, in the case of drug therapy, the relevant policy options are more likely to include information, education, and communication (IEC) services, or active case detection methods, rather than simply the choice of drug.
Outcomes When the results, or outcome, of an intervention can be valued in some unit of account such as money, cost-benefit analysis becomes possible and investments in health can be compared with those for other government-supported activities such as road building. Short of this, intermediate outcomes can be selected and compared across projects. In health, the number of lives saved is one such measure.
Once an outcome, such as lives saved, is chosen, the impact of each dollar spent or program pursued on the outcome needs to be determined. For a number of reasons, this type of information is hard to come by. While a policy option may be directed toward a specific objective, its effects may be felt in other areas: vector control aimed at halting malaria transmission may affect the transmission of other diseases or may degrade the environment; changes in water management practices can affect agriculture; more accurate diagnostic techniques may improve the treatment of other diseases. These external effects, positive or negative, cannot be ignored in calculations of cost-effectiveness, but they are difficult to track and measure.
A policy's effectiveness (and its marginal benefit) also varies according to the extent to which it is implemented; this leads, in turn, to either higher or lower costs (the marginal cost) per life saved. If a new clinic is built, the per-capita costs of the initial investment will decline if the number of users increases over time. In contrast, the effectiveness of an information campaign may decline and per-capita costs therefore increase as more and more is spent to reach those living in remote areas.
Epidemiology and Economics
The relationship between economics and malaria epidemiology in the policy arena has largely been expressed in the rather static calculation of “burden of disease.” The goal of an economics-based approach, how-
ever, is to describe how people make choices and prescribe how governments should make them. The fact that malaria affects hundreds of million of people does not, in and of itself, make the disease a priority for policymakers. What matters to the economist (and to policymakers and those directly in charge of control programs) is whether something can be done about the disease. Policy choices should be made on the basis of the effects that those policies will have on outcomes, not on the size of the problem.
An important exception to this concept is found in basic research, where the benefits of investing in a project are speculative. In cases like this, policymakers might opt to invest heavily, despite an uncertain return, in the hope that the payoff will be large. For any disease, the burden of the disease at least defines the upper bound of a control project's value.
The examples that follow illustrate four fundamental economic concepts: first, the absolute magnitude of the burden of a disease is not a proper criterion for setting priorities in health; second, changes in costs that occur at different levels of intensity of activity affect the optimal allocations of resources; third, this pattern of costs requires a mix of malaria control strategies; and fourth, average cost-effectiveness is not a proper criterion for setting policy priorities. A fifth example illustrates the use of the epidemiologic paradigms, discussed in Chapter 10, for economic decision making.
Example 1: The Burden of Disease and Priority Setting
There are two diseases. Disease A kills 1,000 people per year, and disease B kills 30 people per year. It so happens that it costs $100 per person to prevent people from dying of disease A and $50 per person to prevent death from disease B. The Ministry of Health has $10,000 for disease control. How should it spend the money?
If priorities are set by ranking diseases by “importance,” as measured by mortality rates, disease A wins easily since it kills more than 30 times as many people as disease B. If all resources were devoted to disease A, the Ministry could save 100 lives ($10,000/100). However, if resources were devoted to each of the diseases in proportion to their mortality rates, disease A would receive $9,700, disease B would get the rest, and a total of 103 lives would be saved: six disease-B patients (using the $300) and 97 disease-A patients. The ministry could do even better, though, were it to give the $1,500 needed to eradicate the less important disease, disease B, and use the remaining $8,500 to treat patients who have disease A. In this case, 115 patients would be saved: 30 with disease B and 85 with disease A.
In this example, priorities set by looking only at the burden of disease are exactly backwards. It is the disease with the lower total burden that
should have the first priority, not the one that is more prevalent. Interventions should be ranked in order of the marginal effect of each additional unit of input, whether in dollars, hours, or number of patients seen. Interventions are carried out in this order until the budget is exhausted (as in this case) or until the marginal effect is no longer greater than the marginal cost of other uses of these funds. For instance, if it were determined that each life saved was worth $60 (either because that is how much saving lives from all other causes would cost or, more contentiously, because someone decided that was what lives are worth), then only disease B would warrant intervention.
Example 2: Decreasing Returns
In this instance, the cost of controlling disease B remains at $50 per life saved, but costs associated with disease A are now different: 100 people can be saved at a cost of $25 apiece, but the remaining 900 would cost $100, as in example 1. This situation might arise if there were different techniques for dealing with the disease and the cheaper technique was subject to some capacity limits. Alternatively, there may be only one technique that becomes less effective as its use increases. For example, drug therapy may be inexpensive for patients treated in a clinic setting but quite expensive if health workers have to go into the field to detect and treat cases. With this cost structure and the same $10,000 budget, what should the Ministry of Health do?
If disease A is a priority because of the burden-of-disease argument, 175 lives are saved: 100 at a cost of $2,500 and 75 at a cost of $7,500. More lives could be saved, however, if the criterion used in example 1 were applied. First, $2,500 spent on disease A would save 100 lives. Next, attention would turn to disease B, where 30 lives could be saved for $1,500. The remaining $6,000 could then be spent on the more expensive technique for controlling disease A, saving 60 lives, for a total of 190 lives saved. If the marginal benefits are compared with the marginal cost of $60, as in example 1, the first 100 people suffering from disease A and the 30 people suffering from B would be saved. The rest of the money would be used elsewhere in the health sector (or in another part of the economy).
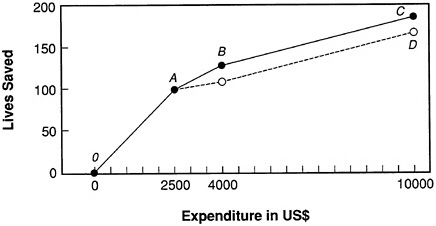
Figure 11-1 shows the relationship between money spent and lives saved in example 1 and example 2. Path OABC represents the strategy of using the $25 technique (from O to A) for disease A, followed by spending on disease B (A to B), and finishing with a return to the less effective technique for A (B to C). Had all resources gone to disease A, we would have followed path OAD, which completely misses the opportunity to reduce mortality due to disease B.
Example 3: More Decreasing Returns
There is only one disease, disease A, but there are two possible interventions. For vector control, the cost is $25 per person for the first 100 individuals who benefit and $100 thereafter. This difference in cost may reflect the relative success of focal spraying, which by definition is limited in its applicability, contrasted with control operations that are more widespread and have a lower success rate. The second intervention, passive case detection, can save 200 people for a cost of $10 apiece if they are treated at a clinic. However, if active detection is required, the cost rises to $33.33 per person.
Given the same budget constraint of $10,000, the optimal strategy would be to treat the 200 people who come to the clinic (costing $2,000), conduct focal spraying to save another 100 lives (costing $2,500), and use active case detection methods to exhaust the budget, saving an additional 165 lives. This strategy favors a mixture of techniques as costs increase and returns decrease for single interventions.
Example 4: Average Versus Marginal Costs and Benefits
As in example 3, there are two techniques: vector control and drug therapy. Focal spraying is very effective. It costs $10 per life and can save 200 lives; after that, other vector control techniques are needed which cost $100 to save one life. Drug therapy costs $40 per person. The country's malaria program splits the budget in half, giving $5,000 to vector control and the same amount to malaria clinics for drug procurement. Overall, the
policy saves 355 lives (200 from focal spraying, costing $10 per person, 30 from spraying at a cost of $100 per person, and 125 ($5,000/$40) from spending on drug therapy).
An accountant appraises the program and discovers that the average cost of a life saved by vector control is only $21.74 ($5,000/230), while for antimalarial drug therapy, each life saved costs $40. The accountant recommends the vector control operations be expanded at the expense of the clinics.
This is precisely the wrong approach. While the average cost of vector control operations is indeed $21.74, the marginal cost is $100, since the limits of focal spraying (which substantially increased the calculation of average effectiveness) have been reached and the less effective technique of widespread spraying is being used. The correct policy decision would be to reduce vector control spending to $2,000 and use the remaining funds to purchase antimalarial drugs. The number of lives saved would then total 400.
Example 5: Economics and Epidemiologic Paradigms
Depending on the degree of endemicity, control policies have had varying degrees of effectiveness. The most dramatic improvements have occurred in areas of low endemicity, where eradication is at least possible. Even in highly endemic areas, real (though possibly temporary) gains may be possible, and short-run benefits may be worth pursuing. The epidemiologic paradigms in Chapter 10 can be used to determine appropriate malaria control policies. This example illustrates how such polices vary with the particular type of malaria.
Assume that two policies are available: drug therapy and pesticide spraying operations, which can be done in one, two, or three cycles per season. Drug therapy will always save 60 lives per $1,000 spent. On the other hand, the effectiveness of vector operations, in terms of deaths averted per dollar, varies by region. In an urban area where the vectorial capacity is relatively low and spraying can reduce transmission, each round of spraying costs $1,000. A program with one round of spraying saves 100 lives, one with two rounds saves 150 lives, and one with three rounds saves 175 lives. In a forest region, the costs of the program are the same but the numbers of lives saved for the three levels of effort are 10, 15, and 18, respectively. How should these malaria control programs in these areas spend their $3,000 budgets?
In the urban area, the optimal allocation is a single round of spraying, saving 100 lives, with the remainder spent on drug therapy, saving an additional 120 lives, for a total of 220 lives saved. A second round of spraying would have saved only an additional 50 lives for the extra $1,000,
compared with 60 lives saved from drugs. The return on the third round of spraying is even worse. In the forest area, even the first round of spraying is not worth the cost. All resources should go to drugs. This will save 180 lives—fewer than in the urban setting, but the best that can be done.
The paradigm approach may also be used to infer the appropriateness of control policies intended to be used in different environments. The inference is not always directly related to overall effectiveness, however, as was true above. In highland fringe areas, which are prone to epidemic malaria, the cost structure may be as follows: a single cycle of spraying saves 75 lives; a second round, 140; and a third round, 170. At every level of spraying effort, vector control in this region yields fewer lives saved than in the periurban areas. However, the appropriate policy in this case is to use two cycles of spraying instead of one. This is because the marginal benefit of the second round of spraying in the epidemic areas is 65 lives per $1,000 (140 - 75), which is still better than the 60 lives saved by drug therapy.
These points are all illustrated in Figure 11-2. For each of the three regions, the number of lives saved by each round of spraying is plotted against costs. For comparison, the number of lives saved by using antimalarial drugs is also plotted as more is spent on therapy (at rate of 60 per $1,000). As a rule, vector operations should be expanded as long as the slope of the kinked line is steeper than the line representing drug therapy. The appropriate stopping points are marked A, B, and C, for urban, forest and highland fringe areas, respectively.
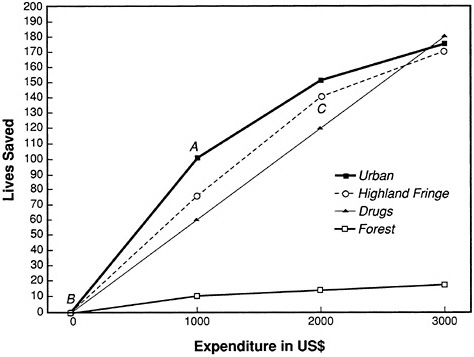
FIGURE 11-2 Example 5: visual representation.
RESEARCH AGENDA
The most appropriate malaria control policies will often require a mix of interventions. This mix will vary from one relatively small region to another, and the information needed to assess the effectiveness of each intervention often does not exist.
Decision Making Skills
Malaria is a complicated disease. Malaria control programs need to use a mix of strategies to achieve optimal results, since each instrument is likely to be subject to diminishing returns with expanded use. Further, packages of interventions will be different for each particular type of malaria. Given resource limitations, policymakers need to know the cost and effectiveness of the interventions at their disposal, and they must be aware of the changes in these costs and effects at different scales of implementation and levels of intensity. Unfortunately, the vast majority of policymakers are not aware of these relationships.
RESEARCH FOCUS: Definition of the range of policy choices relevant to malaria control at different levels of decision making, including health care financing, public investments, facility location, education campaigns, and preventive and treatment options.
RESEARCH FOCUS: Assessment of the costs and effectiveness of malaria interventions applied at different levels of intensity in specific areas.
Data Collection
There is a general need for routine collection of economic and financial data related to malaria control. Such information should be epidemiologic in nature, emphasizing the measurement of variables that can be affected by changes in policy, and should be at a level of detail that is useful to decision makers.
RESEARCH FOCUS: Assessment of the fixed and variable costs associated with malaria control programs; de-
termination of how the latter are affected by changes in the level of program effort.
RESEARCH FOCUS: Comparison of the level of effort devoted to malaria control with changes in disease incidence, deaths averted, or cases detected, with the goal of linking specific policy instruments (e.g., houses sprayed and hours worked) to specific epidemiologic types of malaria.
Information, Education, and Communication Programs
IEC programs, or health education generally, may be of great value in improving drug compliance, encouraging women to seek prenatal care, helping people recognize the symptoms of malaria, and encouraging the adoption of personal protection measures. Neither the technology (costs and effectiveness) of providing this information, the effect of this information on behavior, nor the effect of behavior on incidence is well known.
RESEARCH FOCUS: Assessment of the impact of IEC programs on human behavior in different regions.
Resources
The epidemiology of malaria is often shaped by activities in other sectors (e.g., agriculture, industry, and housing). Similarly, malaria often poses an economic cost to activities within these sectors, as discussed in this chapter. Given the limited funds spent on health by many countries, any contribution to malaria control efforts by non-health-related ministries, sectors, and authorities within a malaria endemic country could prove beneficial in the long term. (For example, agricultural extension workers could collect information about water management or insecticide use and its relationship to malaria transmission during their routine activities.)
RESEARCH FOCUS: Determination of how better to take advantage of the resources of both governmental and nongovernmental entities for malaria control within malaria endemic countries.
REFERENCES
Andreano, R., and T. Helminiak. 1988. Economics, health, and tropical disease: a review. Pp. 19-72 in Economics, Health and Tropical Diseases, Herrin, A. N., and P. L. Rosenfield, eds. Manila: University of the Philippines School of Economics.
Audibert, M. 1986. Agricultural non-wage production and health status: a case-study in a tropical environment. Journal of Development Economics 24:275-291.
Barlow, R. 1968. The Economic Effects of Malaria Eradication. Ann Arbor: Bureau of Public Health Economics, University of Michigan.
Barlow, R., and L. M. Grobar. 1986. Costs and benefits of controlling parasitic diseases. PHN Technical Note 85-17. Washington, D.C.: World Bank.
Bhombore, S. R., C. B. Worth, and K. S. Nanjundiah. 1952. A survey of the economic status of villagers in a malarious irrigated tract in Mysore State, India, before and after DDT residual insecticidal spraying. Indian Journal of Malariology 6:355-365.
Brohult, J., L. Jorfeldt, L. Rombo, A. Bjorkman, P.-O. Pehrson, V. Sirleaf, and E. Bengtsson. 1981. The working capacity of Liberian males: a comparison between urban and rural populations in relation to malaria. Annals of Tropical Medicine and Parasitology 75:487-494.
de Castro, B. 1985. Development of Research-Training Project in Socio-Economics of Malaria Eradication in Colombia. Geneva: World Health Organization.
Cohn, E. J. 1973. Assessing the costs and benefits of anti-malaria programs: the Indian experience. American Journal of Public Health 63:1086-1096.
Conly, G. N. 1975. The Impact of Malaria on Economic Development: A Case Study. Washington, D.C.: Pan American Health Organization.
Democratic Republic of Sudan. 1975. National Health Programme 1977/78-1983/84, Khartoum: Government Printing Office.
Doubilet, P., M. C. Weinstein, and B. J. McNeil. 1986. Use and mis-use of the term “cost-effective” in medicine. New England Journal of Medicine 314:253-256.
Ettling, M. B., S. Krachaiklin, K. Thimasarn, and D. Shepard. 1990. Evaluation of Malaria in Maesot, Thailand. Thailand: Malaria Division, Ministry of Public Health.
Foster, S. D. 1990. Improving the Supply and Use of Essential Drugs in Sub-Saharan Africa PRE Working Paper Series 457. Washington, D.C.: World Bank.
Gandahusada, S., G. A. Fleming, Sukamto, T. Damar, Suwarto, N. Sustriayu, Y. H. Bang, S. Arwati, and H. Arif. 1984. Malaria control with residual fenitrothion in Central Java, Indonesia: an operational-scale trial using both full and selective coverage treatments. Bulletin of the World Health Organization 62:783-794.
Griffith, D. H. S., D. V. Ramana, and H. Mashaal. 1971. Contribution of health to development. International Journal of Health Services 1:253-318.
Hammer, J. In press. To prescribe or not to prescribe: on the regulation of pharmaceuticals in less developed countries. Social Science and Medicine.
Hedman, P., J. Brohult, J. Forslund, V. Sirleaf, and E. Bengtsson. 1979. A pocket of controlled malaria in a holoendemic region of West Africa Annals of Tropical Medicine and Parasitology 73:317-325.
Jamison, D. T., and W. H. Mosley. 1990. Selecting disease control priorities in developing countries (3rd draft). Pp. 1-66 in Disease Control Priorities in Developing Countries, Jamison, D. T., and W. H. Mosley, eds. Washington, D.C.: Population, Health and Nutrition Division, World Bank.
Kaewsonthi, S. 1989. Costs and performance of malaria surveillance and monitoring in Thailand: a retrospective study based on apportionment of expenditure under budget headings.
Social and Economic Research Project Reports No. 5. Geneva: UNDP/World Bank/WHO Special Programme on Research and Training in Tropical Diseases.
Kaewsonthi, S., and A. G. Harding. 1984. Cost and performance of malaria surveillance in Thailand. Social Science and Medicine 19:1081-1097.
Khan, M. J. 1966. Estimate of economic loss due to malaria in West Pakistan. Pakistan Journal of Health 16:187-193.
Lancaster, H. O. 1990. Expectations of Life. New York: Springer-Verlag.
Livadas, G. A., and D. Athanassatos. 1963. The economic benefits of malaria eradication in Greece. Rivista di Malariologia 42:178-187.
Malik, I. H. 1966. Economic advantages of anti-malaria measures amongst the rural population Publication No. 137. Lahore: The Board of Economic Inquiry.
Mills, A. 1987. Economic study of malaria in Nepal: the cost effectiveness of malaria control strategies. London: Evaluation and Planning Center, London School of Hygiene and Tropical Medicine.
Molineaux, L., and G. Gramiccia. 1980. The Garki Project: Research on the Epidemiology and Control of Malaria in the Sudan Savanna of West Africa. Washington, D.C.: Population, Health and Nutrition Division, World Bank.
Niazi, A. D. 1969. Approximate estimates of the economic loss caused by malaria with some estimates of the benefits of M.E.P. in Iraq. Bulletin of Endemic Diseases 2:28-39.
Ortiz, J. R. 1968. Estimate of the cost of a malaria eradication program. Bulletin of the Pan American Health Organization 64:14-17.
Over, M., R. Ellis, J. Huber, and O. Solon. 1990. The consequences of adult ill-health. Pp. 4.1-4.25 in The Health of Adults in the Developing World, Feachem, R., T. Kjellstrom, C. J. L. Murray, M. Over, and M. Phillips, eds. Washington, D.C.: World Bank.
Quo, W. K. 1959. Malaria Information. Unpublished document. Geneva: World Health Organization.
Rosenfield, P. L., F. Golladay, and R. K. Davidson. 1984. The economics of parasitic diseases: research priorities. Social Science and Medicine 19:1117-1126.
Russell, P. F., and M. K. Menon. 1942. A malario-economic survey in rural South India. Indian Medical Gazette 77:167-180.
San Pedro, C. 1967-1968. Economic costs and benefits of malaria eradication. Philippines Journal of Public Health 12:5-24.
Shepard, D. S., U. Brinkmann, M. Ettling, and R. Sauerborn. 1990. Economic Impact of Malaria in Africa. Arlington: Vector Biology and Control Project, U.S. Agency for International Development.
Sinton, J. A. 1935. What malaria costs India, nationally, socially and economically. Records of the Malaria Survey of India 5:223-264, 413-489.
Sinton, J. A. 1936. What malaria costs India, nationally, socially and economically. Records of the Malaria Survey of India 6:96-169.
Sudre, P., J. G. Breman, D. McFarland, and J. P. Koplan. 1990. Treatment of Chloroquine-Resistant Malaria in African Children: A Cost Effective Analysis. Atlanta: Centers for Disease Control.
Van Dine, D. L. 1916. The relation of malaria to crop production. Scientific Monthly November:431-439.
Walsh, J. A., and K. S. Warren. 1979. Selective primary health care: an interim strategy for disease control in developing countries. New England Journal of Medicine 301:967-974.
Wernsdorfer, G., and W. H. Wernsdorfer. 1988. Social and economic aspects of malaria and its control. Pp. 1421-1471 in Malaria, Principles and Practice of Malariology, Wernsdorfer, W. H., and I. McGregor, eds. New York: Churchill Livingstone.