Engineering Tissue-to-Tissue Interfaces and
the Formation of Complex Tissues
HELEN H. LU
Columbia University
Two significant challenges in the field of tissue engineering are the simultaneous formation of multiple types of tissues and the functional assembly of these tissues into complex organ systems (e.g., the skeletal, muscular, or circulatory systems). These challenges are particularly important for orthopedic regenerative medicine, as musculoskeletal motion requires synchronized interactions among many types of tissue and the seamless integration of bone with soft tissues such as tendons, ligaments, or cartilage. These tissue-to-tissue interfaces are ubiquitous in the body and exhibit a gradient of structural and mechanical properties that serve a number of functions, from mediating load transfer between two distinct types of tissue to sustaining the heterotypic cellular communications required for interface function and homeostasis (Benjamin et al. 1986; Lu and Jiang 2006; Woo et al. 1988). But these critical junctions are prone to injury (from trauma or even exercise and daily activity) and unfortunately do not regenerate after standard surgical repair, thus compromising graft stability and long-term clinical outcome (Friedman et al. 1985; Lu and Jiang 2006; Robertson et al. 1986). Consequently, there is a need for grafting systems that support biological fixation or integrative repair of soft tissues.
BACKGROUND
Through a combination of cells, growth factors, and/or biomaterials, the principles of tissue engineering (Langer and Vacanti 1993; Skalak 1988) have been readily applied to the formation of a variety of connective tissues such as bone, cartilage, ligament, and tendon both in vitro and in vivo. More recently, emphasis has shifted from tissue formation to tissue function (Butler et al. 2000), with a
focus on imparting biomimetic functionality to orthopedic grafts and enabling their translation to the clinic.
But clinical translation remains elusive as researchers seek to understand how to achieve biological fixation or functional integration of tissue-engineered orthopedic grafts—of bone, ligaments, or cartilage—with each other and/or with the host environment. The challenge is rooted in the complexity of the musculo-skeletal system and the structural intricacy of both hard and soft tissues. These tissues, each with a distinct cellular population, must operate in unison to facilitate physiologic function and maintain tissue homeostasis. It is thus not surprising that the transition between various tissue types is characterized by a high level of heterogeneous structural organization that is crucial for joint function.
As shown in Figure 1, ligaments and tendons with direct insertions into bone exhibit a multitissue transition consisting of three distinct but continuous
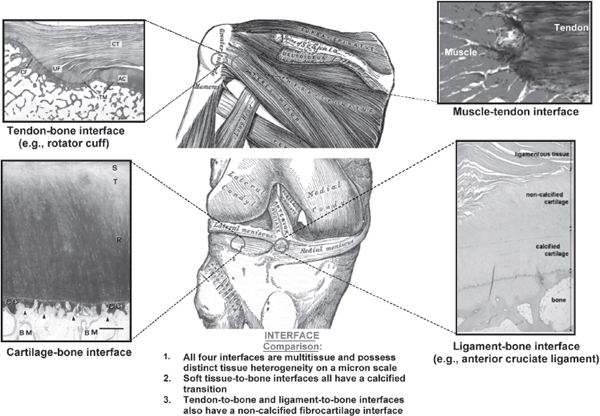
FIGURE 1 Common orthopedic tissue-to-tissue interfaces. Significant structural and compositional homology exists in the orthopedic tissue-to-tissue interfaces of the tendon-bone (Benjamin and Ralphs 1998), muscle-tendon (Larkin et al. 2006), cartilage-bone (Hunziker et al. 2002), and ligament-bone junctions (Iwahashi et al. 2010). Regeneration of these complex junctions is essential for integrative soft tissue repair and treatment of massive, multitissue injuries. Tendon-to-bone interface: AC = articular cartilage, B = bone, CF = calcified fibrocartilage, CT = connective tissue, TM = tidemark, UF = uncalcified fibrocartilage. Cartilage-to-bone interface: BM = bone marrow space, CC = calcified cartilage, R = radial zone, S = superficial zone, T = transitional zone.
regions of ligament, fibrocartilage, and bone (Benjamin et al. 1986; Cooper and Misol 1970; Wang et al. 2006). The fibrocartilage interface is further divided into noncalcified and calcified regions. In light of this complexity, effective tissue engineering must incorporate strategic biomimicry or the prioritization of design parameters in order to regenerate the intricate tissue-to-tissue interface and ultimately enable seamless graft integration and functional repair.
MECHANISMS OF INTERFACE REGENERATION
The mechanisms underlying the formation, repair, and maintenance of tissue-to-tissue boundaries are not well understood. In particular, it is not known how distinct boundaries between different types of connective tissues are reestablished after injury. It is likely that mechanical loading (Killian et al. 2012) as well as chemical and biological factors play a role in this complex process.
It has long been observed that when tendon is resutured to its original attachment site, cellular organization resembling that of the native insertion occurs in vivo (Fujioka et al. 1998). Investigators have also reported that, although healing after ligament reconstruction does not lead to the reestablishment of the native insertion, a layer of interface-like tissue forms in the bone tunnel (Blickenstaff et al. 1997; Grana et al. 1994; Rodeo et al. 1993). These observations suggest that when trauma or surgical intervention results in nonphysiologic exposure of normally segregated tissue types (e.g., bone or ligament), interactions between the resident cell populations (e.g., osteoblasts in bone, fibroblasts in tendon, stem cells/progenitor cells in both tissues) are critical for initiating and directing the repair response that leads to reestablishment of a fibrocartilage interface between soft tissue and bone.
Specifically, it has been hypothesized that osteoblast-fibroblast interactions mediate interface regeneration through heterotypic cellular interactions that can lead to phenotypic changes or transdifferentiation of osteoblasts and/or fibroblasts (Lu and Jiang 2006). Moreover, these interactions may induce the differentiation of stem cells or resident progenitor cells into fibrochondrocytes and thereby promote the regeneration of the fibrocartilage interface. This hypothesis has been validated using coculture and triculture models of interface-relevant cell populations (Jiang et al. 2005; Wang et al. 2007), models that offer simple and elegant methods to systematically investigate cell-cell interactions (Bhatia et al. 1999; Hammoudi et al. 2010).
When ligament fibroblasts and osteoblasts were cocultured using a model permitting both physical contact and cellular interactions, it was observed that these controlled interactions altered cell growth and upregulated the expression of interface-related matrix markers. These cellular interactions have a downstream effect, either inducing cell transdifferentiation or causing the recruitment and differentiation of progenitor or stem cells for fibrocartilage formation. When this hypothesis was tested in triculture, it was noted that under the influence of
osteoblast-fibroblast interactions, stem cells from the bone marrow began to differentiate toward a chondrocyte-like phenotype, producing a matrix similar in composition to that of the interface.
These intriguing findings suggest that heterotypic cellular communications play a regulatory role in the induction of interface-specific markers in progenitor or stem cells, and demonstrate the effects of these interactions in regulating the maintenance of soft tissue-to-bone junctions. The nature of the regulatory cytokines secreted and the mechanisms underlying these interactions are not known, but cell communication is likely to be significant for interface regeneration as well as homeostasis. Therefore the optimal interface scaffold must promote interactions between the relevant cell populations residing in each interface region.
INTERFACE STRUCTURE-FUNCTION
RELATIONSHIP AND DESIGN INSPIRATION
From a structure-function perspective, the complex multitissue organization of the soft tissue-to-bone junction is optimized to sustain both tensile and compressive stresses experienced at the ligament-to-bone junction. Numerous characterization studies (Benjamin et al. 1986; Bullough and Jagannath 1983; Matyas et al. 1995; Moffat et al. 2008; Oegema and Thompson 1992; Ralphs et al. 1998; Spalazzi et al. 2004; Thomopoulos et al. 2003; Woo et al. 1988) have revealed remarkable organizational similarities among many tissue-to-tissue interfaces (Figure 1). They often consist of a multitissue, multicell transition and exhibit a controlled distribution of mineral content that, along with other structural parameters such as collagen fiber organization, results in a gradient of mechanical properties progressing from soft tissue to bone.
Direct measurement of interface mechanical properties has been difficult due to the complexity and relatively small scale of the interface, generally ranging from 100 μm to 1 mm in length. Instead, knowledge of insertion material properties has been largely derived from theoretical models.
Moffat and colleagues (2008) recently performed the first experimental determination of the compressive mechanical properties of the anterior cruciate ligament (ACL)-bone interface in a neonatal bovine model. They evaluated the incremental displacement field of the fibrocartilage tissue under the applied uni-axial strain by coupling microcompression with optimized digital image correlation analysis of pre-and postloading images. Deformation decreased gradually from the fibrocartilage interface to bone, and these changes were accompanied by a gradual increase in compressive modulus. The interface also exhibited a region-dependent decrease in strain, and a significantly higher elastic modulus was found for the mineralized fibrocartilage compared to the nonmineralized region. These region-specific mechanical properties enable a gradual transition rather than a sudden increase in tissue strain across the insertion, thereby minimizing the formation of stress concentrations and enabling load transfer from soft to hard tissues.
Given the structure-function dependence inherent in the biological system, these regional changes in mechanical properties are likely correlated to matrix organization and composition across the interface. Partition of the fibrocartilage interface into nonmineralized and mineralized regions likely has a functional significance, as increases in matrix mineral content have been associated with higher mechanical properties in connective tissues.
Evaluation of the insertion site using Fourier transform infrared imaging (Spalazzi et al. 2007) and X-ray analysis revealed an increase in calcium and phosphorous content progressing from ligament to interface and then to bone. A narrow exponential transition in mineral content, instead of a linear gradient of mineral distribution, was detected progressing from the nonmineralized to the mineralized interface regions. Moreover, the increase in elastic modulus progressing from the mineralized to the nonmineralized fibrocartilage interface region was shown to be positively correlated (Moffat et al. 2008) with the presence of calcium phosphate.
These observations have yielded invaluable clues for the design of biomimetic scaffolds for engineering tissue-to-tissue interface. Specifically, a stratified or multiphased scaffold will be essential for recapturing the multitissue organization observed at the soft tissue-to-bone interface. To minimize the formation of stress concentrations, the scaffold should exhibit phase-specific structural and mechanical properties, with a gradual increase in the latter across the scaffold phases. Spatial control of mineral distribution on a stratified scaffold can impart controlled mechanical heterogeneity similar to that of the native interface. Compared to a homogeneous structure, a scaffold with predesigned, tissue-specific matrix inhomogeneity can better sustain and transmit the distribution of complex loads inherent at the multitissue interface.
It is important to bear in mind that the phases of a stratified scaffold must be interconnected and preintegrated with each other, to ensure the formation of compositionally distinct yet structurally contiguous multitissue regions. Furthermore, interactions between interface-relevant cells serve important functions in the formation, maintenance, and repair of interfacial tissue. Therefore, precise control over the spatial distribution of these cell populations is also critical for multitissue formation and interface regeneration. Consideration of these biomimetic parameters should guide and optimize the design of stratified scaffolds for promoting the formation and maintenance of controlled matrix heterogeneity and interface regeneration.
BIOINSPIRED SCAFFOLD DESIGN FOR
INTERFACE TISSUE ENGINEERING
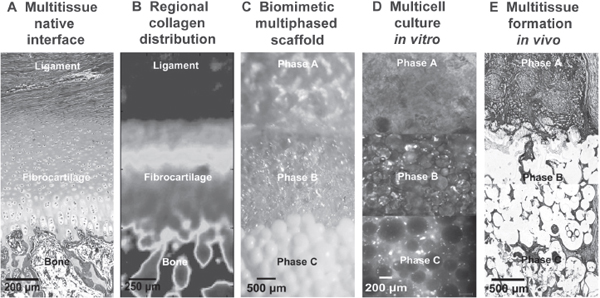
Inspired by the native ACL-to-bone interface, Spalazzi and colleagues (2006, 2008) pioneered the design of a triphasic scaffold (Figure 2C) for the regeneration of this challenging interface. The scaffold’s three continuous phases are each engineered for a specific tissue region of the interface: Phase A is a polymer fiber
mesh for fibroblast culture and soft tissue formation, Phase B consists of polymer microspheres and is designed for fibrochondrocyte culture, and Phase C is composed of sintered polymer-ceramic composite microspheres for bone formation (Lu et al. 2003). The innovative design is in essence a single scaffold system with three compositionally distinct yet structurally continuous phases, all designed to support the formation of multitissue regions across the ligament-bone junction.
To form the ligament, interface, and bone regions, fibroblasts, chondrocytes, and osteoblasts were seeded onto Phases A, B, and C, respectively. Interactions between these cell types on the stratified scaffold were evaluated both in vitro (Spalazzi et al. 2008) and in vivo (Spalazzi et al. 2006). Extensive tissue infiltration and abundant matrix deposition were observed, with tissue continuity maintained across scaffold phases. Interestingly, matrix production compensated for the decrease in mechanical properties that accompanied scaffold degradation, and three continuous regions of ligament, interface, and bone-like matrix were formed in vivo (Figure 2E).
In addition to stratified scaffolds, there is tremendous interest in designing scaffolds with a gradient of properties—that is, with a relatively gradual and continuous transition in either composition or structural organization, resulting in a linear gradient in mechanical properties (Harris et al. 2006; Seidi et al. 2011; Singh et al. 2008). These novel scaffolds with either a compositional (Erisken et al. 2008; Li et al. 2009) or chemical factor (Phillips et al. 2008; Singh et al. 2010) gradient offer direct regional control and allow for scaffold heterogeneity that mimics the complex native interface. They may thus address the need to recapitulate the complex transition of mechanical and chemical properties that are characteristic of tissue-to-tissue junctions.
Design challenges in engineering biomimetic gradients revolve around scale—how best to recapitulate the micro- to nanoscale gradients that have been reported at the tissue-to-tissue interface. The stratified scaffold approach may represent a simpler strategy, whereby a gradation of key compositional and functional properties is preestablished by focusing on forming specific tissue regions of interest and preintegrating them through stratified design. In any case, it is necessary to adopt strategic biomimicry in functional interface scaffold design and to prioritize design parameters for interface regeneration based on the type of interface to be regenerated, the type and severity of injury, and the patient’s age and overall health.
In addition to scaffold design, it is expected that cellular contributions will play a pivotal role in mediating the regeneration and homeostasis of the gradation of compositional and mechanical properties at the interface. For example, Ma and colleagues (2009) used cell self-assembly to form bone-ligament-bone constructs by culturing engineered bone segments to ligament monolayers. Paxton and colleagues (2009) also reported promising results when evaluating the use of a polymer ceramic composite and RGD peptide to engineer functional ligament-to-bone attachments.
SUMMARY AND FUTURE DIRECTIONS
The biomimetic interface tissue engineering approach described in this paper is rooted in an in-depth understanding of the inherent structure-function relationship at the tissue-to-tissue interface. The studies discussed indicate that controlling cellular response via coculture, triculture, or growth factor distribution on multiphased scaffolds is a critical emerging strategy to enable the development of local gradients on a physiologically relevant scale.
Many soft tissues connect to bone through a multitissue interface populated by multiple cell types that minimize the formation of stress concentrations while enabling load transfer between soft and hard tissues. In the event of injury or other disruption, reestablishment of tissue-to-tissue interfaces is critical for the formation of multitissue systems and the promotion of integrative tissue repair.
Investigations into the mechanism of interface regeneration have revealed the role of mechanical loading as well as heterotypic cellular interactions in directing the formation, repair, and maintenance of the tissue-to-tissue interface. Moreover, functional and integrative repair may be achieved by coupling both cell- and scaffold-based approaches. The vast potential of stratified scaffold systems is evident because (1) they are designed to support multitissue regeneration by mediating heterotypic cellular interactions and (2) they can be further refined by incorporating well-controlled compositional and growth factor gradients as well as the use of biochemical and biomechanical stimulation to encourage tissue growth and maturation.
Interface tissue engineering will be instrumental for the ex vivo development and in vivo regeneration of integrated musculoskeletal tissue systems with biomimetic functionality. Yet there remain a number of challenges in this exciting area. These include the need for a better understanding of the structure-function relationship at the native tissue-to-tissue interface and of the mechanisms that govern interface development and regeneration. Furthermore, the in vivo host environment and the precise effects of biological, chemical, and physical stimulation on interface regeneration must be thoroughly evaluated to enable the formation and homeostasis of the new interface. Physiologically relevant in vivo models are needed to determine the clinical potential of designed scaffolds.
The successful regeneration of tissue-to-tissue interfaces through a bioinspired approach may promote integrative and functional tissue repair and enable the clinical translation of tissue engineering technologies from bench to bedside. Moreover, by bridging distinct types of tissue, interface tissue engineering will be instrumental for the development of integrated musculoskeletal organ systems with biomimetic complexity and functionality.
REFERENCES
Benjamin M, Ralphs JR. 1998. Fibrocartilage in tendons and ligaments: An adaptation to compressive load. Journal of Anatomy 193(Pt 4):481–494.
Benjamin M, Evans EJ, Copp L. 1986. The histology of tendon attachments to bone in man. Journal of Anatomy 149:89–100.
Bhatia SN, Balis UJ, Yarmush ML, Toner M. 1999. Effect of cell-cell interactions in preservation of cellular phenotype: Cocultivation of hepatocytes and nonparenchymal cells. Journal of the Federation of American Societies for Experimental Biology 13:1883–1900.
Blickenstaff KR, Grana WA, Egle D. 1997. Analysis of a semitendinosus autograft in a rabbit model. American Journal of Sports Medicine 25:554–559.
Bullough PG, Jagannath A. 1983. The morphology of the calcification front in articular cartilage: Its significance in joint function. Journal of Bone and Joint Surgery 65:72–78.
Butler DL, Goldstein SA, Guilak F. 2000. Functional tissue engineering: The role of biomechanics. Journal of Biomechanical Engineering 122:570–575.
Cooper RR, Misol S. 1970. Tendon and ligament insertion: A light and electron microscopic study. Journal of Bone and Joint Surgery (American volume) 52:1–20.
Erisken C, Kalyon DM, Wang H. 2008. Functionally graded electrospun polycaprolactone and beta-tricalcium phosphate nanocomposites for tissue engineering applications. Biomaterials 29:4065–4073.
Friedman MJ, Sherman OH, Fox JM, Del Pizzo W, Snyder SJ, Ferkel RJ. 1985. Autogeneic anterior cruciate ligament (ACL) anterior reconstruction of the knee: A review. Clinical Orthopaedics and Related Research 196:9–14.
Fujioka H, Thakur R, Wang GJ, Mizuno K, Balian G, Hurwitz SR. 1998. Comparison of surgically attached and non-attached repair of the rat Achilles tendon-bone interface: Cellular organization and type X collagen expression. Connective Tissue Research 37:205–218.
Grana WA, Egle DM, Mahnken R, Goodhart CW. 1994. An analysis of autograft fixation after anterior cruciate ligament reconstruction in a rabbit model. American Journal of Sports Medicine 22:344–351.
Hammoudi TM, Lu H, Temenoff JS. 2010. Long-term spatially defined coculture within three-dimensional photopatterned hydrogels. Tissue Engineering Part C Methods 16(6):1621–1628.
Harris BP, Kutty JK, Fritz EW, Webb CK, Burg KJ, Metters AT. 2006. Photopatterned polymer brushes promoting cell adhesion gradients. Langmuir 22(10):4467–4471.
Hunziker EB, Quinn TM, Häuselmann HJ. 2002. Quantitative structural organization of normal adult human articular cartilage. Osteoarthritis Cartilage 10:564–572.
Iwahashi T, Shino K, Nakata K, Otsubo H, Suzuki T, Amano H, Nakamura N. 2010. Direct anterior cruciate ligament insertion to the femur assessed by histology and 3-dimensional volume-rendered computed tomography. Arthroscopy 26: S13–S20.
Jiang J, Nicoll SB, Lu HH. 2005. Co-culture of osteoblasts and chondrocytes modulates cellular differentiation in vitro. Biochemical and Biophysical Research Communications 338:762–770.
Killian ML, Cavinatto L, Galatz LM, Thomopoulos S. 2012. The role of mechanobiology in tendon healing. Journal of Shoulder and Elbow Surgery 21(2):228–237.
Langer R, Vacanti JP. 1993. Tissue engineering. Science 260:920–926.
Larkin LM, Calve S, Kostrominova TY, Arruda EM. 2006. Structure and functional evaluation of tendon—skeletal muscle constructs engineered in vitro. Tissue Engineering 12:3149–3158.
Li XR, Xie JW, Lipner J, Yuan XY, Thomopoulos S, Xia YN. 2009. Nanofiber scaffolds with gradations in mineral content for mimicking the tendon-to-bone insertion site. Nano Letters 9:2763–2768.
Lu HH, Jiang J. 2006. Interface tissue engineering and the formulation of multiple-tissue systems. Advances in Biochemical Engineering/Biotechnology 102:91–111.
Lu HH, El Amin SF, Scott KD, Laurencin CT. 2003. Three-dimensional, bioactive, biodegradable, polymer-bioactive glass composite scaffolds with improved mechanical properties support collagen synthesis and mineralization of human osteoblast-like cells in vitro. Journal of Biomedical Materials Research 64A:465–474.
Ma J, Goble K, Smietana M, Kostrominova T, Larkin L, Arruda EM. 2009. Morphological and functional characteristics of three-dimensional engineered bone-ligament-bone constructs following implantation. Journal of Biomechanical Engineering 131:101017.
Matyas JR, Anton MG, Shrive NG, Frank CB. 1995. Stress governs tissue phenotype at the femoral insertion of the rabbit MCL. Journal of Biomechanics 28:147–157.
Moffat KL, Sun WH, Pena PE, Chahine NO, Doty SB, Ateshian GA, Hung CT, Lu HH. 2008. Characterization of the structure-function relationship at the ligament-to-bone interface. Proceedings of the National Academy of Sciences of the United States of America 105:7947–7952.
Oegema TR Jr, Thompson RC Jr. 1992. The zone of calcified cartilage: Its role in osteoarthritis. In Kuettner KE, Schleyerbach R, Peyron JG, Hascall VA, eds. Articular Cartilage and Osteoarthritis. New York: Raven Press. Pp. 319–331.
Paxton JZ, Donnelly K, Keatch RP, Baar K. 2009. Engineering the bone-ligament interface using polyethylene glycol diacrylate incorporated with hydroxyapatite. Tissue Engineering Part A 15:1201–1209.
Phillips JE, Burns KL, Le Doux JM, Guldberg RE, Garcia AJ. 2008. Engineering graded tissue interfaces. Proceedings of the National Academy of Sciences of the United States of America 105:12170–12175.
Ralphs JR, Benjamin M, Waggett AD, Russell DC, Messner K, Gao J. 1998. Regional differences in cell shape and gap junction expression in rat Achilles tendon: Relation to fibrocartilage differentiation. Journal of Anatomy 193(Pt 2):215–222.
Robertson DB, Daniel DM, Biden E. 1986. Soft tissue fixation to bone. American Journal of Sports Medicine 14:398–403.
Rodeo SA, Arnoczky SP, Torzilli PA, Hidaka C, Warren RF. 1993. Tendon-healing in a bone tunnel: A biomechanical and histological study in the dog. Journal of Bone and Joint Surgery (American volume) 75:1795–1803.
Seidi A, Ramalingam M, Elloumi-Hannachi I, Ostrovidov S, Khademhosseini A. 2011. Gradient biomaterials for soft-to-hard interface tissue engineering. Acta Biomaterialia 7(4):1441–1451.
Singh M, Berkland C, Detamore MS. 2008. Strategies and applications for incorporating physical and chemical signal gradients in tissue engineering. Tissue Engineering Part B: Reviews 14(4):341–366.
Singh M, Sandhu B, Scurto A, Berkland C, Detamore MS. 2010. Microsphere-based scaffolds for cartilage tissue engineering: Using subcritical CO(2) as a sintering agent. Acta Biomaterialia 6:137–143.
Skalak R. 1988. Tissue engineering: Proceedings of a workshop, held at Granlibakken, Lake Tahoe, California, February 26-29. New York: Liss.
Spalazzi JP, Costa KD, Doty SB, Lu HH. 2004. Characterization of the mechanical properties, structure, and composition of the anterior cruciate ligament-bone insertion site. Transactions of the Orthopedic Research Society 29, Poster #1271.
Spalazzi JP, Doty SB, Moffat KL, Levine WN, Lu HH. 2006. Development of controlled matrix heterogeneity on a triphasic scaffold for orthopedic interface tissue engineering. Tissue Engineering 12:3497–3508.
Spalazzi JP, Boskey AL, Lu HH. 2007. Region-dependent variations in matrix collagen and mineral distribution across the femoral and tibial anterior cruciate ligament-to-bone insertion sites. Transactions of the Orthopaedic Research Society (abstract 0891), San Diego.
Spalazzi JP, Dagher E, Doty SB, Guo XE, Rodeo SA, Lu HH. 2008. In vivo evaluation of a multiphased scaffold designed for orthopaedic interface tissue engineering and soft tissue-to-bone integration. Journal of Biomedical Materials Research A 86(1):1–12.
Thomopoulos S, Williams GR, Gimbel JA, Favata M, Soslowsky LJ. 2003. Variations of biomechanical, structural, and compositional properties along the tendon to bone insertion site. Journal of Orthopaedic Research 21:413–419.
Wang IE, Mitroo S, Chen FH, Lu HH, Doty SB. 2006. Age-dependent changes in matrix composition and organization at the ligament-to-bone insertion. Journal of Orthopaedic Research 24:1745–1755.
Wang IE, Shan J, Choi R, Oh S, Kepler CK, Chen FH, Lu HH. 2007. Role of osteoblast-fibroblast interactions in the formation of the ligament-to-bone interface. Journal of Orthopaedic Research 25:1609–1620.
Woo SL, Maynard J, Butler DL, Lyon RM, Torzilli PA, Akeson WH, Cooper RR, Oakes B. 1988. Ligament, tendon, and joint capsule insertions to bone. In Woo SL, Buckwalter JA, eds. Injury and Repair of the Musculoskeletal Soft Tissues. Savannah GA: American Academy of Orthopedic Surgeons. Pp. 133–166.