Removing Carbon Dioxide
from the Atmosphere:
Possibilities and Challenges of Air Capture
CHRISTOPHER W. JONES
Georgia Institute of Technology
More than 150 years of fossil fuel combustion has increased the global atmospheric carbon dioxide (CO2) concentration from approximately 280 ppm in preindustrial times to almost 400 ppm today. Given the link between the rising atmospheric CO2 concentration and global climate change, the world is now “carbon-constrained,” and scientists, engineers, segments of the public, and scientifically literate policymakers are pushing for rapid development of alternative energy sources. However, the coupling of population growth and an ever-higher global standard of living means that energy demand will continue to increase. Although a worldwide effort is focused on development and deployment of renewable energy technologies, development is outpaced by energy demand. For this reason, fossil energy will continue to supply the preponderance of global energy for generations—and global CO2 emissions will keep rising, hastening climate change.
CO2 emissions from fossil fuel combustion are associated with three broad categories: (1) electricity production from coal- or gas-fired power plants (33–50% of total), (2) land, air, or sea transportation (~33%), and (3) other industrial uses. Global climate and energy strategies addressing anthropogenic emissions have focused on capturing the CO2 emitted from the world’s largest point sources, coal-fired power plants. This can be done in a variety of ways, for example by modification of existing plants to capture the CO2 produced (i.e., postcombustion capture, or PCC) or by designing new plants that enable more efficient CO2 capture. However, these approaches, even if widely adopted, would address only the 33–50% of CO2 emissions associated with large point sources.
The most difficult CO2 emissions to address are those associated with transportation. Onboard CO2 capture from mobile sources such as automobiles and
airplanes is currently impractical. While the electrification of passenger vehicles is (very slowly) shifting some energy use for transportation to large electricity-generating point sources, some mobile CO2 sources, such as planes, will likely never be electrified. Thus, alternative technologies for addressing CO2 emissions from mobile sources are needed.
DIRECT CAPTURE OF CO2 FROM AIR (“AIR CAPTURE”)
In 1999, Klaus Lackner first proposed the widespread development and deployment of devices that extract CO2 directly from the atmosphere as a way to address global CO2 emissions and climate change (Lackner et al. 1999). Although initially considered an alternative to capture from large point sources, the direct capture of CO2 from the air, or “air capture,” is generally considered a complementary technology to point source capture. Implementation of the two technologies together could allow long-term use of fossil energy while slowing or mitigating the impacts of anthropogenic CO2 emissions on climate change. Furthermore, unlike other climate mitigation options—often described as geo-engineering, whereby humans tinker with the planet to influence climate—CO2 capture from air may be a safer option, a form of traditional pollution control.
Why, then, have PCC and air capture not been widely implemented? Because, in the absence of a price on emitted carbon, there is no incentive for the private sector to adopt such technologies. A recent study published by the US Department of Energy suggests that 90% of the coal-fired power plants in the United States could implement PCC at a cost of approximately $60 per ton of CO2 captured (Nichols 2011). However, as an emerging technology, there are far fewer detailed technoeconomic descriptions of air capture processes, and the limited reports offer a wide array of estimated costs. One study of air capture processes based on CO2 absorption using basic alkaline hydroxide solutions suggested costs of $500–1,000/ton CO2 (House et al. 2011), whereas an evaluation of a second-generation technology based on use of supported amine adsorbents estimated costs closer to $100/ton CO2 (Kulkarni and Sholl 2012).
TECHNICAL CHALLENGES OF AIR CAPTURE
Most gas separation processes considered for air capture are based on CO2-absorbing liquids or CO2-adsorbing solids, with the overall process passing through cyclical stages of adsorption and desorption, as shown in Figure 1. This approach is common and used in a variety of scalable gas separation technologies. However, compared to most large-scale gas separation processes, air capture has a unique challenge associated with the ultradilute nature of CO2 (~400 ppm) in the atmosphere. Yet it also has some key advantages over PCC; for example, it can be located anywhere in the world, because ambient air is largely uniform in composition; in addition, impurities in fossil fuel exhaust (e.g., nitrogen and sulfur
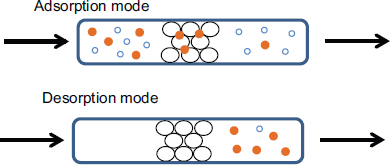
FIGURE 1 Gas separation processes based on adsorption onto solid are cyclical in nature. An adsorption cycle (top) selectively removes some gas species (small solid sphere) by adsorption on a solid (large shaded sphere), yielding a purified exhaust, followed by a desorption cycle (bottom) that liberates a concentrated product, most often induced by a pressure and/or temperature change.
oxides) are present in ambient air at a much lower level, precluding the need for gas pretreatment to remove these species. This allows for siting processes at locations appropriate for CO2 use or sequestration, negating the need for transport of concentrated CO2 in pipelines over long distances.
There are five important criteria for an economically scalable air capture process.
1. Because of the low concentration of CO2 in air, very large volumes of air must be moved through the process—about 125 times and 375 times more than for CO2 capture from a natural gas- or coal-fired power plant, respectively, assuming an equivalent capture fraction. Thus, to prevent excessive energy requirements for gas movement, the process must have very low pressure drops associated with the air flow.
2. Also associated with the low ambient CO2 concentration, the process must use materials and/or fluids with high CO2 capture capacities, such as those with a very high density of adsorption sites and/or very strong CO2-adsorbent interactions.
3. Favorable adsorption kinetics are important to enable short cycle times (long cycle times lead to impractical plant sizes associated with large inventories of adsorption media).
4. Because absorption and adsorption are exothermic processes, the removal of CO2 from the capture media for concentration is endothermic and can require significant energy input. This regeneration energy must be provided at low cost, ideally in the form of low-grade waste heat.
5. Finally, the process equipment and adsorption media must have a suitably long lifetime because the above factors will make air capture a capital-
intensive process with large plant sizes compared to many traditional gas separation processes.
AIR CAPTURE VIA ADSORPTION ON AMINES
A wide variety of CO2-adsorbing materials have been considered for use in PCC processes. In contrast, the scope of materials for air capture applications is dramatically decreased because processes must operate near ambient temperature and pressure and offer good adsorption capacities under ultradilute conditions. Supported amine materials, a class of solids functionalized with organic amine sites, are the only materials available that offer large CO2 capacities under air capture conditions and operate near ambient temperature. Several research groups have recently reported the suitability of such materials for CO2 capture from ultradilute gases (e.g., Goeppert et al. 2012).
One process that shows the potential to meet the five key criteria involves supported amine adsorbents coated on a high-surface-area structured contactor (an object that contacts flowing fluid), such as a ceramic monolith. Such contactors are already produced on a large scale for use in catalytic exhaust gas clean-up and offer a low-cost route to high surface areas with low pressure drops. Flow of air through the adsorbent-lined channels at high velocity allows for rapid CO2 adsorption kinetics. Desorption is achieved by flowing low-grade saturated steam (70–105°C) through the monoliths and over the adsorbent layer, providing both a thermal and concentration driving force for desorption. Steam in this temperature range can be obtained as low-grade waste heat from a variety of industrial processes or produced via solar-thermal heating at low cost. The concentrated CO2 product is produced from the steam/CO2 mixture via condensation or compression. Finally, the robustness of the monolith contactor offers promise for long-term stability of adsorbent materials.
The above description is only one process possibility; undoubtedly other promising approaches are being actively researched as well.
OUTLOOK FOR AIR CAPTURE
CO2 capture from ambient air, or “air capture,” is an emerging technology that, if deployed on a large scale alongside traditional postcombustion capture, could play a critical role in stabilizing or even reducing global atmospheric CO2 levels. But its development is almost entirely in the hands of private enterprise: although almost $7 billion federal dollars have been spent on research and development on methods to capture carbon from large point sources, the total federal investment for CO2 capture from air may be as little as $300,000. Thus essentially all investment in air capture technologies in the United States has been private, and, in the absence of a carbon tax, initial deployments of such technologies will be focused on profitable industrial use, such as in greenhouses or enclosed algal
bioreactors, or for enhanced oil recovery. Without policy changes or a reallocation of federal research dollars to this area, initial deployments will focus on profit-generating applications, and technological developments that might support implementation of air capture as a climate change mitigation strategy will be deferred to the future.
DISCLOSURE
The author collaborates with and has a financial interest in Global Thermostat, LLC, a company actively engaged in commercializing technology for CO2 capture from ultradilute gases.
REFERENCES
Goeppert A, Czaun M, Prakash GKS, Olah GA. 2012. Air as the renewable carbon source of the future: An overview of CO2 capture from the atmosphere. Energy and Environmental Science 5:7833–7853.
House KZ, Baclig AC, Ranjan M, van Nierop EA, Wilcox J, Herzog HJ. 2011. Economic and energetic analysis of capturing CO2 from ambient air. Proceedings of the National Academy of Sciences of the United States of America 51:20428–20433.
Kulkarni AR, Sholl DS. 2012. Analysis of equilibrium-based TSA processes for direct capture of CO2 from air. Industrial and Engineering Chemistry Research 51:8631–8645.
Lackner KS, Ziock H-J, Grimes P. 1999. Carbon dioxide extraction from air: Is it an option? Proceedings of the 24th International Conference on Coal Utilization and Fuel Systems, Clearwater, FL.
Nichols C. 2011. Coal-Fired Power Plants in the United States: Examination of the Costs of Retrofitting with CO2 Capture Technology, Revision 3. DOE/NETL Report 402/102309. Washington, DC: Department of Energy, National Energy Technology Laboratory.






