2
Total Artificial Hearts: Technological Potential and Research and Development Costs
THIS CHAPTER FIRST ADDRESSES the likelihood of total artificial hearts (TAHs) providing useful treatment of end-stage heart disease. The evaluation examines the current technological potential and limitations of the devices and compares these estimates with data on existing treatments for end-stage heart disease. Then, because actually fulfilling the potential of a technology is directly related to the further research and development provided, current and future funding from the National Heart, Lung, and Blood Institute (NHLBI) are examined. Finally, general considerations for industry R&D decisions, rather than costs, are briefly discussed; determining the costs of future industry R&D for TAHs is not possible.
Since the early 1980s, the major use of TAHs and ventricular assist devices (VADs) (together referred to as mechanical circulatory support systems, or MCSSs) has been for temporary support while the heart muscle recovers function or the patient awaits a donor heart. Appreciable data now exist for temporaryuse of MCSSs. Although such devices have in a few instances functioned for many months, and although a very few TAHs have been implanted with the intent of long-term use, relatively little information exists that is useful for determining the efficacy and risks of long-termuse. This evaluation of the technological opportunities and barriers in the development of TAHs is therefore based primarily on information from temporary use of TAHs and VADs, animal trials, and in vitro testing.
From these data, it clearly is possible to make a reasonably accurate assessment of the technical state of MCSSs that takes into account their biocompatibility and the mechanical reliability of the systems or components. On other questions, such as long-term technological efficacy, clini-
cal complications and effectiveness,1 and impact on quality of life, it is more difficult to ascertain the future potential of MCSSs. Clinical trials of a totally implantable long-term left ventricular assist device are scheduled to begin in 1992; the data from these trials will have broad implications for continuing development of long-term TAHs and VADs.
TEMPORARY CIRCULATORY SUPPORT
The use of MCSSs for temporary support has evolved, in the absence of totally implantable systems, to meet patient needs and to evaluate the feasibility of mechanically substituting for the pumping action of the heart. These systems have all been connected or “tethered” to cumbersome external power sources by skin-penetrating tubes or wires, limiting acceptance as permanent devices and increasing the risk of infection. They have, however, provided rather substantial and impressive information on pumping efficacy, mechanical reliability, and morbidity and mortality risks that is helpful in predicting the long-term outcomes of using totally implanted devices.
Temporary MCSS use is intended to assist or replace the heart for a limited period of time. Temporary devices support patients until transplantation with a donor heart or while they are recovering from acute depression of cardiac function that is thought to be reversible (e.g., after cardiopulmonary bypass). Although temporary clinical use of MCSSs has been successful in some instances for over a year, the purpose is not to provide permanent circulatory support.
Several MCSSs are currently available for temporary use under a Food and Drug Administration (FDA) investigational device exemption (IDE) 2 (see Table 2.1). Most devices in temporary use are VADs, used to support either the left ventricle, right ventricle, or both. Centrifugal external VADs are not considered here, because their maximum period of use is measured in days.
As of early 1991, the only TAH approved by FDA for temporary investigational use was the Pennsylvania State University heart, also known as the Hershey heart. The Symbion/Jarvik TAH was used for temporary support until 1990, when its IDE was withdrawn by FDA because of questions concerning production and quality control practices on the part of the manufacturer; it is still used for temporary support outside the United States.
From an engineering perspective, certain specific MCSS components and
|
1 |
Efficacy is the measure of a medical technology's effect on a disease state under ideal clinical conditions. Effectiveness, in contrast, is the measure of a technology's effect during general clinical use. |
|
2 |
FDA grants approval of an IDE for trial use of a device in humans. |
TABLE 2.1 MCSSs That Are Currently Available
|
Devices available under investigational device exemptions (IDEs) Short-term external devices ABIOMED VADa Pierce-Donachy VADb Short-term internal devices Novacor LVADc Thermo Cardiosystems LVADc Pennsylvania State University TAHc Devices under development Long-term internal devices ABIOMED/Texas Heart Institute TAHd Nimbus/Cleveland Clinic TAHd Pennsylvania State University/Sarns-3M TAHd University of Utah TAHd Novacor LVADd Thermo Cardiosystems LVADd |
|
aPump on stand adjacent to bed. bPump strapped externally to the abdomen. cPump is implantable but connected to an external power source. dPump is fully implantable, power transmitted through the skin. eNo longer available under IDE in the United States. LVAD, left ventricular assist device; MCSSs, mechanical circulatory support systems; TAH, total artificial heart; VAD, ventricular assist device. SOURCES: Graham and Chalmers, 1989; Macoviak et al., 1990; Rosenberg,1990. |
designs, such as the electronic control system, are effective whereas other elements of the devices still need improvement. Nevertheless, there is general consensus that MCSSs can temporarily reverse or stabilize a patient's physiologic abnormalities. As with early heart valves and other cardiovascular devices, continued R&D investment is needed to eliminate or minimize complications associated with the devices, to take full advantage of the various strengths of the different device designs, and to expand device capabilities from temporary use to long-term circulatory support.
LONG-TERM DEVICES UNDER DEVELOPMENT
Long-term use of devices is intended to meet a permanent need to assist or replace the heart. The four U.S. development groups currently involved
in developing TAHs under contract to NHLBI are ABIOMED/Texas Heart Institute, Nimbus/Cleveland Clinic, Pennsylvania State University/Sarns-3M, and the University of Utah (see Table 2.1). As of 1991, none of these groups had reached the stage of development appropriate for the initiation of clinical trials.
In the category of VADs, two developers of long-term devices are Novacor and Thermo Cardiosystems, Inc. (TCI). The Novacor long-term implantable VAD is a refinement of that manufacturer's externally powered, temporary VAD. Use of this short-term Novacor VAD in humans has extended in a few instances up to one year. The fully implantable long-term Novacor VAD is beginning its animal trials in 1991 and is expected to enter human trials in 1992. The TCI VAD is a fully implantable device for long-term use that is under development through an NHLBI contract. In vivo trials of the device have been conducted in animals, and a pneumatically driven tethered version has been used for support before transplantation in 33 patients. This VAD uses textured blood-contacting surfaces, enabling the growth of a living lining of endothelial cells within the device that could reduce the likelihood of thromboembolic events.3 FDA has also approved a TCI electrically powered, tethered VAD for investigational temporary use in five patients (Altman, 1991).
ENGINEERING ASSESSMENT OF MECHANICAL CIRCULATORY SUPPORT DEVICES4
Testing of mechanical circulatory devices occurs in three phases: bench testing, also known as in vitro device readiness testing; in vivo trials in animals; and clinical trials in humans. Bench testing of a circulatory support device provides an evaluation of engineering reliability, in particular of its design and manufacture. Animal trials provide biological and physiological information including safety and complications, and are particularly necessary for evaluating whether or not to perform trials in humans. Clinical-trial or investigational use of devices in humans is conducted under an IDE to determine efficacy. All three, used together, help determine the potential benefit of mechanical circulatory support devices and are also specific steps toward FDA approval of the general use and marketing of a device (see Figure 2.1).
|
3 |
Thromboembolism is the blocking of a blood vessel (embolization) by a particle of a blood clot (thrombus) that has broken away from the clot where it formed. |
|
4 |
This section draws heavily on a background paper (Rosenberg, 1990) commissioned by the Institute of Medicine committee from Gerson Rosenberg, Ph.D., a bioengineer at Pennsylvania State University. |
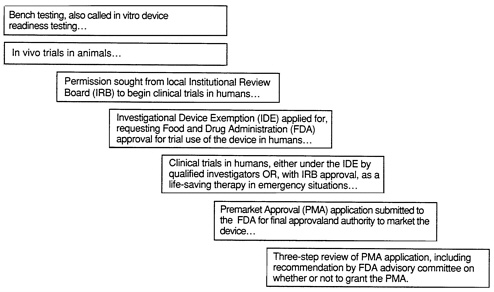
FIGURE 2.1 Testing and approval process for a life-sustaining or implantable medical device.
Mechanical Failure
There are two general kinds of mechanical failure with circulatory support devices. A “soft” device failure occurs when the malfunction is correctable and is not so critical that the patient's life is in immediate danger; a “hard” failure is a major mechanical failure that results in serious risk of death. Based on experience with MCSSs employed for temporary use, the risk of either type of mechanical failure is low.
In one institution, for example, 40 Jarvik TAHs were used over the course of three years. During this time no mechanical failures occurred in the drive system and mechanical dysfunction was limited to one artificial valve that needed to be replaced (Muneretto et al., 1990). Similar positive results are evident in another analysis of reported use, which indicates that of 186 clinical applications of short-term pneumatic TAHs (Symbion/ Jarvik devices for the most part), approximately 1 percent of the patients experienced any mechanical failure, as did approximately 1.5 percent of all those who received one or two VADs (Rosenberg, 1990).
Recent bench testing of the pumping system of the long-term Novacor VAD involved 12 devices operating unattended for 2 years in a simulated in vivo situation. This rigorous reliability testing was, however, a trial of a small number of devices in a simulated environment, and therefore the positive results of the trial are not directly transferable to evaluation of the device's clinical potential. Most notably, saline solution and mechanical
valves, rather than blood and tissue valves, had to be used for the in vitro experiment and the implantable power source was not tested. Nevertheless, the in vitro testing results correlate with the low reported rates of device failure in short-term clinical use. Because failures in such a bench test are most likely to occur during the early stages of the test, the results of the two-year Novacor VAD bench trial support the clinical evidence that mechanical failure of MCSSs is not likely to be a significant limitation on their use.
Device Components as Limiting Factors
Two technical areas of MCSSs that have been inadequately examined are the valves and the batteries. Mechanical wear of valves over time is a particular limitation. The valves currently in use were not designed for use with circulatory support devices but were developed as stand-alone implants, a use that subjects them to less material stress. Valves currently appear to have acceptable but not optimal durability for two or more years of use. Accelerated testing and analysis of stress on the valve elements would enable better estimates of the component's longevity (Rosenberg, 1990).
Longevity of power sources is another concern, despite continuing work on designing next-generation power sources. A primary difficulty is the patient's need to rely on the implanted back-up battery for short periods of time. The longevity of this internal battery must not be significantly impaired by such frequent use, and it must recharge quickly and efficiently enough to be available in an emergency.
One possible solution to the power-source limitation, currently being pursued by a corporation, is the development of a lithium battery (Post and Takeuchi, 1990). Another solution would be thermal power sources; NHLBI has for several years supported two thermal power developers, who reported promising results at the annual contractors ' conference in December 1990.5 A specific advantage of thermal power sources is that the patient would be independent of the external battery pack, with recharging limited to perhaps one hour for every eight hours of continuous operation (Butler et al., 1990; White et al., 1990). Further exploration of alternative power sources might lead to significant improvements in patient quality of life and could obviate the longevity problem of current implantable batteries.
|
5 |
In these implanted thermal power systems, heat from a storage reservoir containing a mixture of lithium fluoride-lithium chloride salts generates energy to drive a piston and power the pump. An advantage of this power system is the elimination of electronic components and several moving parts, with consequently minimized mechanical wear. |
The development of safe biomaterials, specifically for surfaces that come into contact with blood, also has been a limitation on MCSS technology; the polyurethane currently in use was developed in the 1960s. The incidence of thromboembolism, a serious clinical complication, is highly responsive to the type and use of biomaterials and the configuration of the pumping chamber. Recent interest in improved biomaterials, including new biomedical elastomers for blood-contacting surfaces, may reduce this barrier to MCSS and other medical device development; two corporations are beginning development of improved biomaterials. Among the goals are materials with improved biocompatibility, fatigue resistance, and ease of manufacture.
The success of further R&D in addressing the problems just enumerated will have a direct impact on levels of clinical complications and quality of life. These levels, in turn, will greatly affect the probability of developing long-term devices that are effective for routine clinical use.
Device Longevity
When calculating the costs of developing a long-term MCSS, a pivotal question is whether the effectiveness of the current devices can eventually be extended beyond two years. Current assumptions about MCSSs are that, with further R&D and the option of easily replacing implanted batteries and other components, device longevity can eventually be extended to 5 and perhaps 10 years. It is also anticipated that component failure or wear will become predictable and that some components can be easily repaired. Although such assumptions of improved device longevity and maintenance have not formed a basis for the committee 's evaluation and recommendations, a device with an average 2-year longevity will clearly be used in many fewer patients than one expected to last 5 to 10 years.
CLINICAL COMPLICATIONS WITH MECHANICAL CIRCULATORY SUPPORT DEVICES
There has been extensive temporary use of MCSSs, particularly ventricular assist devices, with over 1,300 documented applications. Of these, 400 patients have been temporarily supported before transplantation and 68 percent of these lived to receive a donor heart (Miller et al., 1990). There do not appear to be significant differences in the outcomes of immediate transplantation and transplantations preceded by MCSS use. The major risk factors for mortality with transplantation after MCSS support are irreversible noncardiac organ dysfunction and infection at the time of transplantation; the same major risks apply when a donor heart is implanted without prior use of an MCSS. When organ dysfunction is moderate and reversible,
mechanical circulatory support can actually normalize liver, kidney, and respiratory function while the patient is awaiting transplantation (McGee et al., 1989; Pifarre et al., 1990).
For patients successfully supported to transplantation, the causes of death vary. With patients who used an MCSS for fewer than 30 days —generally those with the greatest severity of illness—kidney failure, infection, and bleeding had the highest correlation with the patient's death following transplantation. For patients supported for more than 30 days, infection and rejection of the donor heart were the most frequent causes of death, results comparable to those for direct transplantation. These data suggest that, despite an initial risk in implanting the device, there is a relative lack of additional ill effects, once an MCSS is implanted, from duration of use (Miller et al., 1990).
As to clinical complications and biocompatibility, data from the Clinical Registry of Mechanical Ventricular Assistance (Miller et al., 1990) show that the major complicating factors for all MCSS use are bleeding and the subsequent need for reoperation, which occur shortly after initial implantation and are generally related to the severity of illness and consequent alteration in coagulation factors. Kidney failure and infection, respiratory failure, biventricular heart failure during VAD use, and thromboembolism are the other major complications (see Table 2.2). Although particular de-
TABLE 2.2 Clinical Complications with Short-Term Use of MCSSs
|
Percent of Patients Experiencing the Complication |
||
|
Complication |
With a VAD (n = 1,221) |
With a TAH (n = 186) |
|
Bleeding and reoperation |
30 |
28 |
|
Kidney failure |
25 |
19 |
|
Infection |
17 |
21 |
|
Respiratory failure |
16 |
13 |
|
Biventricular failure (VAD only) |
16 |
— |
|
Thromboembolic complications |
12 |
13 |
|
Emboli |
7 |
9 |
|
Thrombi |
5 |
4 |
|
LVAD, left ventricular assist device; MCSSs, mechanical circulatory support systems; TAH, total artificial heart; VAD, ventricular assist device. SOURCE: Combined Registry for the Clinical Use of Mechanical VentricularAssist Pumps and the Total Artificial Heart, verbal communicationto G. Rosenberg, 1990. |
||
vices can excel in specific situations, the overall data are very similar for the short-term use of TAHs and the short-term use of uni- or biventricular assist devices.
The major clinical risks with use of circulatory support devices are, therefore, failure of major organs, bleeding, infection, and thromboembolism. Of these, it is reasonable to predict that the incidence of infection will be lower because the skin-penetrating tubes used with temporary devices will not be necessary with a totally implantable MCSS. Predictions about the implications of long-term MCSS use, however, such as the number of strokes resulting from thromboembolism, can be made less confidently because they depend on current estimates of the future state of the technology.
Data on thromboembolism in particular are very dependent on device design and biomaterials. For all clinical MCSS use for all indications, there is experience of approximately 12 percent either of thrombosis in the device and tubing or of embolism (Rosenberg, 1990) but rates of thromboembolic events vary considerably. These rates depend heavily on factors such as the patient's condition, surgeon's experience, and anticoagulant drug therapies used after implantation, and also on the design of the specific device used. For instance, one group of 12 patients using a VAD with low thromboembolic rates had no such events, whereas 17 percent of 54 patients using another VAD did; both groups received antithrombotic drug therapies (McGee et al., 1989; Termuhlen et al., 1989). To create the best possible device, the optimal combination of MCSS design, components, and materials must be determined (Didisheim et al., 1989; Rosenberg, 1990).
ALTERNATIVE TECHNOLOGIES FOR PREVENTING AND TREATING END-STAGE HEART DISEASE
The conventional treatment modality for end-stage heart disease is medical treatment with pharmaceutical products. When such treatment does not suffice, or when a patient experiences acute difficulties, alternative treatments are considered. Like the use of medication, these treatments can prevent or ameliorate some of the clinical consequences of end-stage heart disease. They are not, however, able to prevent end-stage heart disease itself. Most notable among these current and emerging alternative technologies are: short-term intra-arterial assist devices, surgical techniques for augmenting the function of the heart muscle with a skeletal muscle, and heart transplantation.
Conventional Medical Treatment
The conventional medical treatment of congestive heart failure has evolved over the past century and continues to change with the introduction of new
pharmaceuticals and the acquisition of new understanding about pathophysiologic mechanisms. Medical treatment is first directed at identifying and treating underlying causes and precipitating factors. The work of the heart is reduced by restricting physical activity and by pharmacologically adjusting blood pressure and cardiac function. Digitalis glycosides are used as positive inotropic agents that increase the force of contraction of the failing heart. Salt restrictions and diuretic agents contract intravascular fluid volume and adjust intracardiac pressures to ameliorate the signs and symptoms of vascular congestion. Varying potency and specificity for different portions of the kidney tubule enable these diuretics to be concentrated according to the severity of the disease. Vasodilating drugs reduce the work of the failing heart and thus relieve the symptoms of congestive heart failure. These are the only drugs that have been shown in prospective randomized studies to prolong the survival of patients with congestive heart failure, but to date this has been a short-lived benefit. More potent positive inotropic and vasodilating drugs are available for intravenous use in the intensive care unit for treatment of acute decompensation, but these are not currently practical for long-term management.
In summary, in spite of many advances in the conventional medical therapy of congestive heart failure, the primary goals of therapy remain those of symptomatic relief and palliation of a chronic and inexorably progressive disorder. Ongoing basic and clinical research may offer more hopeful medical therapeutic approaches in the future.
Other Cardiac Assistance Technologies
The intra-aortic balloon pump provides mechanical assistance to left ventricular function on a temporary basis. It is usually inserted into the thoracic aorta through the femoral artery and employs a helium-inflated balloon that deflates in cardiac systole and inflates in diastole. This action reduces the pressure against which the left ventricle contracts and augments diastolic pressure and thus coronary flow. The pump increases cardiac output to a maximum of about 20 percent.
The Nimbus Hemopump6 is a small device, usually inserted via the femoral artery into the left ventricle, which assists blood flow by propelling blood from the left ventricle to the ascending aorta. It provides greater augmentation of cardiac blood flow than does the intra-aortic balloon pump, but its use is currently limited to several days to a week and it is not yet approved for general use.
Both the intra-aortic balloon and Nimbus blood pumps are generally used
|
6 |
Nimbus Hemopump is a registered trademark. |
to maintain patients who are waiting for their hearts to recover normal function after a massive insult to the myocardium, such as might occur after open heart surgery or acute myocardial infarction, or during percutaneous transluminal coronary angioplasty. There are clear applications for their short-term use, but because they can be used for only limited periods of time and because the patient is essentially bed-bound, they are not long-term solutions to end-stage heart disease (Frazier et al., 1990; Macoviak et al., 1990).
Cardiomyoplasty is the process of relocating a skeletal muscle from the back to a position around or near the heart and training it, through electrical stimulation, to contract regularly and thereby aid circulation. Because it takes several weeks to adapt skeletal muscle fibers for this function, cardiomyoplasty is not suitable for emergency circumstances. Having been used investigationally in only a hundred or so patients worldwide, it is not yet clear how much the procedure can improve standard clinical indicators of health.
In summary, pharmaceutical treatment, intra-aortic devices, and cardiomyoplasty can improve or ameliorate the condition of only a portion of patients with end-stage heart disease, typically those in comparatively better health. In particular, these treatment alternatives usually cannot improve the health status of moribund patients (those in New York Heart Association functional Class IV),7 or postpone their deaths for more than a few weeks. For these patients heart transplantation provides the only available alternative, but is of limited potential given the small supply of donor hearts.
Heart Transplantation
When comparing TAHs with heart transplantation, the first consideration is that the supply of donor hearts, currently about 2,000 annually in the United States, is inadequate and will continue to be insufficient to meet demand. The population currently eligible for donor hearts is a subset of the larger population that would be eligible for TAHs. Given a device that functions well, many of those patients who do not currently receive a donor organ would benefit from a device. Even among those patients who are clearly eligible for transplantation, many will prefer receiving a long-term device to waiting for an available donor heart.
The second consideration is that heart transplantation and circulatory
|
7 |
New York Heart Association functional classification for Class IV: Patients with cardiac disease resulting in inability to carry on any physical activity without discomfort. Symptoms of cardiac insufficiency or of the anginal syndrome may be present even at rest. If any physical activity is undertaken, discomfort is increased (New York Heart Association, 1964). |
support devices are not always suitable for the same individual, and therefore will not necessarily be competing for patients. The criteria for heart transplantation are rather restrictive; there are both absolute and relative contraindications. Absolute contraindications include predictable immune system rejection of the donor organ and comorbidities such as diabetes, cerebrovascular or liver disease, carcinoma, or chronic obstructive pulmonary disease (Graham and Chalmers, 1989). Relative contraindications include overall health too poor to allow waiting for a donor heart; poor tolerance of immunosuppressant side effects; and advanced age, which currently militates against the use of comparatively rare donor hearts. TAHs will have fewer contraindications than transplantation and therefore would be potentially useful for patients who are not eligible for transplantation as well as those who cannot survive the wait for a donor heart.
In the first two years following heart transplantation the most serious risks are infection and immune system rejection of the donor heart. A further risk for heart transplantation recipients, still relatively unexamined for lack of long-term data, is accelerated atherosclerosis of the donor heart. This condition afflicts at least 20 percent of all transplant recipients and may become the most important cause of late mortality or retransplantation (Davies and Al-Tikriti, 1989; Baughman, 1990).
The goals in using circulatory support devices are to prolong life while maintaining an acceptable quality of life. The average survival rate for cardiac transplantation patients after five years is currently about 70 percent. In predicting future use when TAHs and heart transplantation are both available, it will be important to determine whether early TAH performance can be maintained over a 5- and then a 10-year span of clinical use.
SUMMARY OF THE CURRENT AND FUTURE STATE OF TOTAL ARTIFICIAL HEART TECHNOLOGY
Major limitations on long-term, widespread use of mechanical circulatory support are the devices' current power sources, biomaterials, valves, and design. Design challenges include extending device longevity, reducing the size of MCSSs to make them available to smaller adults and children, and improved, smaller configurations of both components and systems. The level of success in overcoming these limitations will directly affect the ultimate usefulness and effectiveness of MCSSs.
In short-term MCSS use, the results of in vitro and in vivo trials indicate that perioperative bleeding, major organ failure, infection, and thromboembolism are the main clinical complications. The data for short-term VADs and TAHs are similar when considered, overall, by category (see Table 2.2) but within the VAD category, performance levels vary in terms of the clinical
complications and overall performance of the specific devices. Long-term use of totally implantable circulatory support devices has not yet occurred; the results of early long-term VAD trials will be crucial in refining current estimates about the long-term routine use of TAHs.
Finally, comparison with other technologies for treating end-stage heart disease indicates that well-performing long-term TAHs could provide care to end-stage heart disease patients that is currently unavailable through heart transplantation, other technologies, or established drug treatment.
RESEARCH AND DEVELOPMENT COSTS OF TOTAL ARTIFICIAL HEARTS
Stages of the Innovative Process
A brief examination of the various stages of the innovation process in medical technology will help to put the research and development process for MCSSs in perspective. Most analysts would propose stages in the innovative process that include basic or fundamental research; applied research and development; product development and testing; introduction to use; and diffusion of use. Two observations about these stages are needed, however.
First, many scientists, implicitly or explicitly, see the stages as a linear progression, in which development and use depend on advances in basic science. But recent observers have begun to demonstrate that the stages often have an iterative relationship to each other: basic research may stimulate further basic research as often as it drives applied research; applied research may feed findings and questions to basic research as it receives vital information from basic research; and development and use of a technology may generate both basic and applied research questions, as well as benefit from research results. Although the dependence of technology on basic science is well established, the frequent contribution of technological advance to scientific progress is less clear but often equally important (Gelijns, 1990).
Second, many scientists and engineers involved in the R&D process view “technology-push” as the driving force in innovation, whereas economists and managers tend to place more weight on “demand-pull” as the source of innovation. As Gruber and Marquis (1969) noted long ago, neither of these opposing views is adequate. Successful innovation stems both from the integration of new scientific and technical information and from a recognition of potential market demand. These observations hold for the MCSS R&D process.
Support for the Artificial Heart Program
One purpose of the IOM committee's deliberations is to advise NHLBI in its decision on R&D resource allocations for circulatory support devices. For 1989, estimated federal expenditures on health-related R&D totaled $10.5 billion (IOM, 1990). The federal funds spent specifically on MCSS research and development are disbursed through NHLBI grants and contracts. From the inception of the artificial heart program in 1964 through fiscal year (FY) 1989, a total of $264.4 million has been spent on a wide variety of targeted programs and grants for MCSS R&D (see Figure 1.5).
In FY 1990 estimated costs for MCSS research were $8.2 million in nontargeted grants and $7.3 million in contracts, for a combined total of $15.5 million. This sum represents 2.7 percent of the $581 million allocated by NHLBI to R&D on heart and vascular diseases in FY 1990, and is 1.4 percent of the total NHLBI 1990 budget of $1,072 billion (see Figure 2.2) (NHLBI, 1990a; NHLBI, 1990b).
It is beyond the committee's abilities to precisely estimate the amounts of NHLBI funds needed to carry MCSS development forward. Device developers presented their estimates of further needed funding, but the
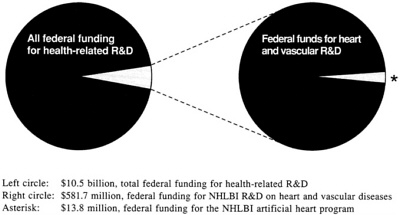
FIGURE 2.2 Representation of federal funding for the artificial heart program, FY 1989 and FY 1990. NHLBI, National Heart, Lung, and Blood Institute; R&D, research and development. SOURCES: Division of National Cost Estimates, 1987; IOM, 1990; Office of National Cost Estimates, 1990; NHLBI, 1990a, 1990b, 1991; Washington Post, 1990.
committee does not feel comfortable with these figures because they substantially exceed previous levels of NHLBI support. Instead, the committee's best estimates are that for VAD development as recommended (see Chapter 10) about $2 million per year will be needed, over and above the amounts already committed for the Novacor clinical trial, during the early-1990s interim period. Support for further VAD clinical trials may be needed thereafter.
The recommended interim funding of TAH development will likely total $3 to $6 million per year for a two- to three-year period, depending on the number of contracts extended after peer-review consideration of the developers' progress. If development continues thereafter, the support required from NHLBI will likely total, over 10 years, between $30 and $90 million for the two final R&D phases (see Chapter 6).
For all MCSS development, the committee notes the advantages of funding multiple research teams. Existing circulatory support systems differ in terms of design, efficiency, and perhaps susceptibility to clinical complications. Continued R&D on several fronts could make it possible to develop MCSSs with optimal capabilities in all of these areas.
Considering the Costs of Research and Development to Industry
In general, the actual costs of medical technology R&D are not well documented or are unknown, and individual corporations are reluctant to release financial data on their R&D programs (Gelijns, 1990). Nevertheless, a few broad observations are possible. The most data are available for pharmaceutical R&D; it is known, for example, that in 1991 development of the “average” pharmaceutical cost a total of $231 million (DiMasi et al., in press; Grabowski, 1991). Even for pharmaceutical R&D, however, “[a]ctual development costs vary greatly from drug to drug, year to year, and company to company, depending on whether the definitions used are uniform or change, in addition to actual [cost] changes” (Spilker, 1989). For medical devices, the uncertainty and difficulty of comparing cost estimates increase, but it is clear that many factors, including the nature of the device, its stage in the development process, the costs of mass production, and quality assurance and testing requirements, are potential considerations in calculating total costs. Chapter 3 and Chapter 9 address other aspects of industry R&D.
CONCLUSIONS
The committee believes that, with the further development likely to occur, the MCSSs currently being developed can reverse the physiological effects of heart failure and improve quality of life for two years or more.
Five-year models appear possible in the near term; however, it is not altogether clear what engineering and materials science advances will be required to achieve highly reliable 5- to 10-year devices. As discussed in Chapter 4, the extent to which MCSSs prolong life and improve its quality will be critical factors in determining their clinical value and the extent of their use.
The committee is uncertain about the costs to NHLBI and industry of the necessary technical improvements in MCSSs and the time required to achieve the necessary level of device longevity. Nevertheless, it does not consider the levels of R&D funding needed from NHLBI to be large enough to constitute, in and of themselves, a reason for discontinuing or cutting back the artificial heart program. The committee further notes the importance of continued funding of multiple research teams so as to profit from the strengths of each.
REFERENCES
Altman, L. K. 1991. F.D.A. approves use in tests of pump to aid failing heart. The New York Times (January 4), pp. A1, A15.
Baughman, K. 1990. Presentation at the public meeting of the Institute of Medicine Committee to Evaluate the Artificial Heart Program of the National Heart, Lung, and Blood Institute. July 13, 1990 .
Butler, K. S., T. Brown, T. Rintoul, R. Kiraly, C. Davies, H. Harasaki, et al. 1990. Development of a thermal energy powered LVAS. Cardiovascular Science and Technology: Basic & Applied, II. Presentation at the annual conference on cardiovascular science and technology, Louisville, Ky.
Davies, H., and S. Al-Tikriti. 1989. Coronary arterial pathology in the transplanted human heart. International Journal of Cardiology 25:99-118.
Didisheim, P., D. B. Olsen, D. J. Farrar, P. M. Portner, B. P. Griffith, D. G. Pennington, et al. 1989. Infections and thromboembolism with implantable cardiovascular devices . Transactions of the American Society of Artificial Internal Organs 35:54-70.
DiMasi, J. A., R. Hansen, H. Grabowski, and L. Lasagna. In press. The cost of innovation in the pharmaceutical industry. Journal of Health Economics, Vol. 10.
Division of National Cost Estimates, Office of the Actuary, Health Care Financing Administration. 1987. National health expenditures, 1986-2000. Health Care Financing Review 8(4):1-36.
Frazier, O. H., R. K. Wampler, J. M. Duncan, W. E. Dear, M. Macris, S. M. Parnis, et al. 1990. First human use of the Hemopump, a catheter-mounted ventricular assist device. Annals of Thoracic Surgery 49:299-304.
Gelijns, A. C. 1990. Comparing the development of drugs, devices, and clinical procedures . In: Institute of Medicine, Medical Innovation at the Crossroads: Vol. I. Modern Methods of Clinical Investigation. A. C. Gelijns, ed. Washington, D.C.: National Academy Press, pp. 147-201.
Grabowski, H. 1991. The changing economics of pharmaceutical research and development . In: Institute of Medicine, Medical Innovation at the Crossroads:
Vol. II. Public Policy and the Economics of Medical Innovation. A. C. Gelijns and E. A. Halm, eds. Washington, D.C.: National Academy Press, pp. 35-52.
Graham, T. R., and J. A. C. Chalmers. 1989. Temporary mechanical ventricular support: Part 1. British Journal of Hospital Medicine 41:420-425.
Gruber, W. H., and D. G. Marquis. 1969. Introduction. In: Factors in the Transfer of Technology. W. H. Gruber and D. G. Marquis, eds. Cambridge, Mass.: The Massachusetts Institute of Technology Press, pp. 3-8.
IOM (Institute of Medicine). 1990. Funding Health Sciences Research: A Strategy to Restore Balance. F. E. Bloom and M. A. Randolph, eds. Washington, D.C.: National Academy Press.
Macoviak, J. A., K. A. Dasse, and V. L. Poirier. 1990. Mechanical cardiac assistance and replacement. Cardiology Clinics 8(l):39-53.
McGee, M. G., S. M. Parnis, T. Nakatani, T. Myers, K. Dasse, W. D. Hare, et al. 1989. Extended clinical support with an implantable left ventricular assist device. Transactions of the American Society of Artificial Internal Organs 35:614-616.
Miller, C. A., W. E. Pae, Jr., and W. S. Pierce. 1990. Combined registry for the clinical use of mechanical ventricular assist pumps and the total artificial heart in conjunction with heart transplantation: Fourth official report—1989 . Journal of Heart Transplantation 9:453-458.
Muneretto, C., G. Rabago, Jr., A. Pavie, Ph. Leger, I. Gandjbakhch, Y. Sasako, et al. 1990. Mechanical circulatory support as a bridge to transplantation: Current status of total artificial heart in 1989 and determinants of survival . Journal of Cardiovascular Surgery 31:486-491.
NHLBI (National Heart, Lung, and Blood Institute). 1990a. Profile of the National Heart, Lung, and Blood Institute artificial heart program. Photocopy.
NHLBI. 1990b. Background information provided at the request of the Institute of Medicine Committee to Evaluate the Artificial Heart Program of the National Heart, Lung, and Blood Institute. Photocopy .
NHLBI. 1991. National Heart, Lung, and Blood Institute: Fact Book, Fiscal Year 1990. Rockville, Md.: National Heart, Lung, and Blood Institute.
New York Heart Association. 1964. Diseases of the Heart and Blood Vessels: Nomenclature and Criteria for Diagnosis(6th ed.). Boston: Little Brown and Company.
Office of National Cost Estimates, Office of the Actuary, Health Care Financing Administration. 1990. National health expenditures, 1988. Health Care Financing Review 11(4):1-41.
Pifarre, R., H. J. Sullivan, A. Montoya, M. Bakhos, J. Grieco, B. K. Foy, et al. 1990. The use of the Jarvik-7 total artificial heart and the Symbion ventricular assist device as a bridge to transplantation. Surgery 108:681-685.
Post, C. J., and E. S. Takeuchi. 1990. Recent advances in the development of a rechargeable lithium battery for left ventricular assist devices. Cardiovascular Science and Technology: Basic & Applied, II. Presentation at the annual conference on cardiovascular science and technology, Louisville, Ky.
Rosenberg, G. 1990. Technological Opportunities and Barriers in the Development of Mechanical Circulatory Support Systems. Background paper commissioned
by the Institute of Medicine Committee to Evaluate the Artificial Heart Program of the National Heart, Lung, and Blood Institute.
Spilker, B. 1989. Multinational Drug Companies: Issues in Drug Discovery and Development. New York: Raven Press, p. 488.
Termuhlen, D. F., M. T. Swartz, D. G. Pennington, L. R. McBride, E. A. Szukalski, J. E. Reedy, et al. 1989. Thromboembolic complications with the Pierce-Donachy ventricular assist device. Transactions of the American Society of Artificial Internal Organs 35:616-618.
Washington Post. 1990. 1989 Health-care outlay up 11%, HHS reports. (December 21) p. A3.
White, M. A., R. Kiraly, and R. L. Whalen. 1990. Investigation of thermal ventricular assist system, TVAS. Cardiovascular Science and Technology: Basic & Applied, II. Presentation at the annual conference on cardiovascular science and technology, Louisville, Ky.


















