9
Strengthening Health Systems for an Effective HIV/AIDS Response
BACKGROUND AND CONTEXT FOR SYSTEMS DEVELOPMENT AND FUNCTIONING FOR HEALTH
A health system includes “all the organizations, institutions, and resources that are devoted to producing health actions. A health action is defined as any effort, whether in personal health care, public health services or through intersectoral initiatives, whose primary purpose is to improve health” (WHO, 2000, p. xi). The primary objectives of a health system are to improve health by achieving the best attainable average level of population health and minimizing the differences between individuals and groups. National governments are ultimately responsible for the performance of health systems and for ensuring the well-being of their populations (WHO, 2000). To meet the ambitious goal of equitable access to health, member states of the World Health Organization (WHO) have committed to providing universal health coverage, defined as “access to key promotive, preventive, curative and rehabilitative health interventions,” at an affordable cost for all members of a population (WHO Secretariat, 2005, p. 1; World Health Assembly, 2005).
Over the past decade (2001–2010), international donors (particularly high-income countries and multilateral institutions) provided more than $185 billion in development assistance for health to low- and middle-income countries (IHME, 2011). Much of this funding has been directed to programs and interventions for specific diseases (e.g., HIV/AIDS, tuberculosis) and for other health focus areas (e.g., maternal and child
health). Large global health initiatives such as PEPFAR, the Global Fund, and the Global Alliance for Vaccines and Immunization have facilitated the tremendous increase in development assistance for health, but there is concern about the effects, intended and unintended, of these initiatives on partner country health systems (Bärnighausen et al., 2012; Biesma et al., 2009; Grépin, 2012a; Levine and Oomman, 2009; Samb et al., 2009). There is widespread consensus within the global health community on the need to strengthen health systems in order to improve health outcomes and meet global targets such as universal health coverage and the health-related Millennium Development Goals1 (Shakarishvili, 2009; Task Force on Global Action for Health System Strengthening, 2008; WHO, 2009). Many of the largest donors and multilateral organizations involved in global health have faced challenges in scaling up services because of health systems weaknesses and have responded by supporting interventions specifically designed to strengthen components of the health system (Palen et al., 2012; Shakarishvili, 2009).
In 2007, WHO developed a framework for health systems strengthening (HSS) that identifies six building blocks corresponding with the essential functions of health systems:
1. Leadership and governance,
2. Financing,
3. Information,
4. Medical products, vaccines, and technologies (shortened to “medical products and technologies” by the committee),
5. Health workforce, and
6. Service delivery (WHO, 2007a).
These building blocks are interdependent, and the relationships between the building blocks deserve as much attention as the individual components (WHO, 2007a, 2009). The building block framework, illustrated in Figure 9-1, has been adopted by the Office of the U.S. Global AIDS Coordinator (OGAC) and others stakeholders that are emphasizing the prioritization, organization, and execution of activities in the essential area of strengthening health systems (Friedman et al., 2011; OGAC, 2009f).
Large donor-funded global health initiatives interact with each building block within partner country health systems. Despite sharing the same goal as partner country health systems—to improve health outcomes—initiatives such as PEPFAR can have negative as well as positive effects on these sys-
__________________
1 In 2000, world leaders committed to the United Nations Millennium Declaration and adopted eight Millennium Development Goals to reduce the most important determinants and consequences of poverty (United National General Assembly, 2000).
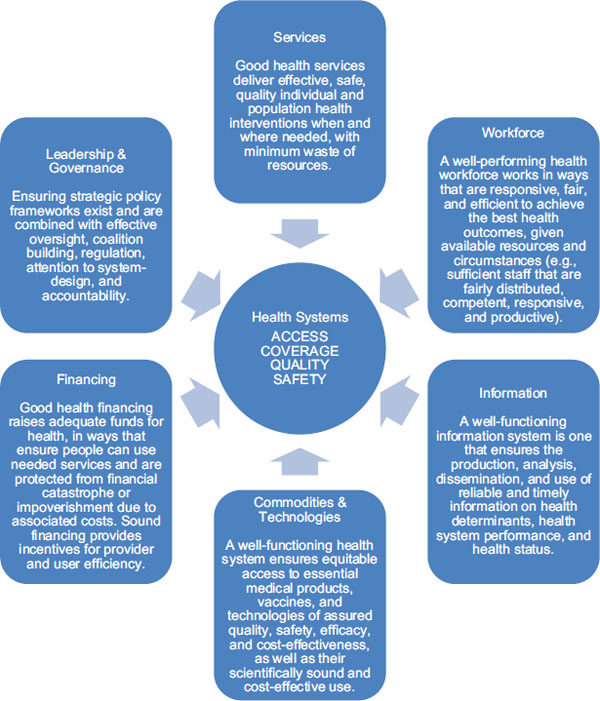
FIGURE 9-1 Representation of WHO’s six building blocks for effective health systems.
SOURCE: Adapted from IOM and NRC, 2010 and WHO, 2007a.
tems. Several studies have examined the effects of global HIV/AIDS initiatives and broader global health initiatives on health systems. Positive effects have included strengthened infrastructure and laboratories, scale-up of HIV/AIDS service delivery, improved primary health care services, a slowing of HIV/AIDS-related deaths among the health workforce through the provision of antiretroviral treatment, greater participation of stakeholder
groups, and increased funding to nongovernmental organizations (NGOs) and faith-based bodies (Biesma et al., 2009; Samb et al., 2009; Yu et al., 2008). Negative effects on health systems include the reallocation of or reduction in funding for other health or non-health priorities; attrition in the public health or primary care workforce as a result of increased incentives to work for donor-funded programs; and the “distortion of recipient countries’ national policies, notably through distracting governments from coordinated efforts to strengthen health systems and re-verticalization of planning, management and monitoring and evaluation systems” (Biesma et al., 2009, p. 239; Samb et al., 2009; Yu et al., 2008). In general, the evidence is mixed and limited for determining whether effects are positive or negative (Biesma et al., 2009; Samb et al., 2009; Yu et al., 2008). In recent years, there has been more research devoted to the interaction between global health initiatives and health systems, and this research has produced recommendations for ensuring that health systems are strengthened, not weakened by global health initiatives.
The ability of societies generally, as well as public health and clinical care entities in particular, to address the HIV epidemic is contingent upon functioning health systems. The term “health system” is intentionally broad, referring to all of the societal resources mobilized to achieve and preserve health, and thus a health systems approach to constraints offers a different lens from that of a disease-specific response (see Table 9-1) (Mills, 2007). Many scholars have argued that investments in response to scaling up disease-specific services could be more appropriately targeted to interventions that broadly strengthen health care systems (Travis et al., 2004; Yu et al., 2008). In 2009 the WHO Maximizing Positive Synergies Collaborative Group issued five recommendations (paraphrased here) for improving the joint effectiveness of large global health programs and partner country health systems: (1) prioritize health system strengthening, (2) agree on and track health system strengthening indicators, (3) align planning and resource allocation between global health initiatives and country health systems, (4) generate more reliable data for the costs and benefits of strengthening health systems, and (5) commit to increased national and global health financing that is more predictable in order to support sustainable and equitable growth of health systems (Samb et al., 2009). The challenge for global health donors is that health system interventions require long-term investments and the longer time lags between interventions and outcomes make such interventions more difficult to measure and evaluate (Bärnighausen et al., 2012).
TABLE 9-1 Health System Constraints with Potential Disease-Specific and Health System Responses
| Constraint | Disease-Specific Response | Health-System Response | ||
| Financing | ||||
| Financial inaccessibility: inability to pay, informal fees | Permit exemptions or reduce prices for focal diseases | Develop risk pooling strategies | ||
| Service Delivery | ||||
| Physical inaccessibility: distance to facility | Provide outreach for focal diseases | Reconsider plans for long-term capital investment and planning for facilities | ||
| Poor quality of care among providers in the private sector | Provide trainings for private-sector providers | Develop systems for accreditation and regulation | ||
| Workforce | ||||
| Inappropriately skilled staff | Implement continuous education and training workshops aimed at developing skills in focal diseases | Review basic medical and nursing training curricula to ensure basic training includes necessary and appropriate skills | ||
| Poorly motivated staff | Offer financial incentives to reward delivery of priority services | Institute appropriate performance review systems, create greater clarity around performance roles and expectations, review salary structures and promotion procedures | ||
| Leadership and Governance | ||||
| Weak planning and management | Provide continuous education and training workshops aimed at developing planning and management skills | Restructure ministries of health, recruit and develop a cadre of dedicated managers | ||
| Lack of intersectoral action and partnership | Create special disease-focused cross-sectoral committees and task forces at the national level | Build systems of local government that incorporate representatives from health, education, and agriculture and that promote accountability of local governance structures to the people | ||
SOURCE: Adapted from Travis et al., 2004.
OVERVIEW OF PEPFAR’S HEALTH SYSTEMS STRENGTHENING ACTIVITIES
As part of the current Institute of Medicine (IOM) evaluation of PEPFAR, Congress mandated an assessment of PEPFAR’s effects on health systems, “including on the financing and management of health systems
and the quality of service delivery and staffing.”2 This section provides a brief history of PEPFAR’s approach to HSS; this is followed by a more in-depth discussion of PEPFAR activities related to each building block of the health system.
History of PEPFAR’s Approach to and Increasing Focus on HSS
In PEPFAR’s first Five-Year Strategy, OGAC articulated the importance of supporting national strategies, laboratory systems, workforce training, and information systems because these components of health systems were essential for scaling up quality services (OGAC, 2005b). Recognizing that partner country health systems were not prepared to support needed services, OGAC committed to providing “targeted technical assistance, training, and funding to improve and expand the infrastructure necessary to ensure optimal delivery of HIV/AIDS treatment services” (OGAC, 2004, p. 39). Although “evidence demonstrates that scale-up of HIV services has produced stronger health systems and, conversely, that stronger health systems were critical to the success of the HIV scale-up” (Palen et al., 2012, p. S113), some have argued that the disease-specific nature of the PEPFAR program may have undermined a coordinated approach to health planning and delivery (Bärnighausen et al., 2011; Hanefeld, 2010; OGAC, 2009f).
OGAC has recognized the largely ad hoc nature of HSS interventions during the first phase of the PEPFAR program (2004–2009) and also the lack of a strategic focus on strengthening each building block of the health system (OGAC, 2009f). PEPFAR-supported HSS interventions were largely disease-specific or somewhere on the continuum between disease-specific and a broader health system response (see Table 9-1). The reauthorization legislation provided the opportunity for PEPFAR to formally identify and support strategies to “strengthen overall health systems in high-prevalence countries, including support for workforce training, retention, and effective deployment, capacity building, laboratory development, equipment maintenance and repair, and public health and related public financial management systems and operations”3 as well as for PEPFAR and partner country governments to commit to a “deeper integration” of HIV services into existing national programs and systems.4 The reauthorization legislation laid out goals for PEPFAR to strengthen health policies and systems for not only HIV/AIDS, but also tuberculosis and malaria, in support of increasing
__________________
2 Tom Lantos and Henry J. Hyde United States Global Leadership Against HIV/AIDS, Tuberculosis, and Malaria Reauthorization Act of 2008, P.L. 110-293, 110th Cong., 2nd sess. (July 30, 2008), ![]() 101(c), 22 U.S.C. 7611(c)(2)(B)(ii).
101(c), 22 U.S.C. 7611(c)(2)(B)(ii).
3Ibid., ![]() 301(c)(5)(D), 22 U.S.C. 2151b-2(d)(6)(G)(ii).
301(c)(5)(D), 22 U.S.C. 2151b-2(d)(6)(G)(ii).
4Ibid., ![]() 301(c)(6), 22 U.S.C. 2151b-2(d)(8).
301(c)(6), 22 U.S.C. 2151b-2(d)(8).
partner country ability to deliver efficient, effective, and evidence-based services.5 This further enabled PEPFAR’s engagement and promotion of other stakeholders, such as civil society, to participate in a country’s HIV/AIDS response.
In its second phase (2009-2013), PEPFAR “emphasizes the incorporation of health systems strengthening goals into its prevention, care and treatment portfolios,” including the training and retaining “health care workers, managers, administrators, health economists, and other civil service employees critical to all functions of a health system” (OGAC, 2009d, p. 8). In response to the reauthorizing legislation’s goals and objectives for health systems, PEPFAR’s second Five-Year Strategy not only articulated its commitment to health systems in terms of activities and resources, but also specified that it would be cognizant and more considerate of health systems activities’ effects when planning prevention, care, and treatment services within partner countries (OGAC, 2009f). The second Five-Year Strategy also specified that PEPFAR could be a platform for improving other health conditions, especially because of its work in HSS to ensure quality and expanded care and treatment services, including antiretroviral therapy (ART) (OGAC, 2009d). In 2009 PEPFAR developed a strategic framework to help PEPFAR mission teams plan HSS activities by identifying the focused investments needed to achieve service delivery objectives, spillover effects, and targeted leveraging of other programs and donors (OGAC, 2009f). Specific OGAC guidance and PEPFAR activities related to each building block are described in the sections that follow.
PEPFAR Funding for HSS
Broadly speaking, funding for PEPFAR HSS activities is captured in three budget codes: Health Systems Strengthening, Strategic Information, and Laboratory Infrastructure (see Box 9-1) (OGAC, 2011c). Although funding for Strategic Information and Laboratory Strengthening can be traced to HSS efforts in the Health Information and Medical Products and Technologies building blocks, funding cannot be disaggregated for efforts in the other building blocks. Over the years, PEPFAR’s budget code definitions were revised, but HSS activities have generally included broad policy reform efforts, system-wide approaches (e.g., supply chain, procurement, and information), and capacity building for financial and program management (OGAC, 2008a, 2010a). Other activities that contribute to HSS, such as those associated with service delivery, especially human resources for health training (HRH), may not be reported in the HSS budget codes (Palen
__________________
5Ibid., ![]() 204(a), 22 U.S.C. 7623(a)(1)(A).
204(a), 22 U.S.C. 7623(a)(1)(A).
BOX 9-1
PEPFAR Budget Code Definitions for HSS
Health Systems Strengthening: “include[s] activities that contribute to national, regional or district level systems by supporting finance, leadership and governance (including broad policy reform efforts including stigma, gender etc.), institutional capacity building, supply chain or procurement systems, [strengthening of local coordinating mechanisms for implementation of] Global Fund programs [or other external grants,] and donor coordination” (OGAC, 2011c, p. 184).
Laboratory Infrastructure: includes “development and strengthening of laboratory systems and facilities to support HIV/AIDS-related activities including: strengthening of laboratory leadership and management; purchase of equipment and commodities; strengthening of laboratory supply and equipment management systems; promotion of quality management systems, laboratory monitoring and evaluation, and laboratory information systems; and provision of staff training and other technical assistance” (OGAC, 2011c, p. 156).
Strategic Information: “[a]ims to build capacity in country for HIV/AIDS behavioral and biological surveillance, facility surveys, monitoring program results, reporting results, supporting health information systems, supporting countries to establish and/or strengthen such systems, supporting training and retention of local cadres of personnel needed to direct all SI activities, and related analyses and data dissemination activities” (OGAC, 2011c, p. 165).
SOURCE: OGAC, 2011c.
et al., 2012), so the amounts presented in Figure 9-2 may underrepresent PEPFAR’s investments in HSS.
Over time, as shown in Figure 9-2, funding for the three budget codes most directly related to HSS, as a proportion of all PEPFAR funding has increased from nearly 12 percent in fiscal year (FY) 2006 to nearly 18 percent in FY 2011 (data for two of the three budget codes were not reported in FY 2005) (OGAC, 2005a, 2006b, 2007c, 2008b, 2010b, 2011d,e). Funding for these three budget codes increased from $175 million in FY 2006 to $769 million in FY 2011. Initially, Strategic Information and Laboratory Strengthening activities received a greater share of funding, but over time more funding has been directed to the Health Systems Strengthening budget code. From FY 2006 to FY 2011, 38 percent of PEPFAR funding for HSS was directed to the Health Systems Strengthening budget code, 33 percent to Laboratory Strengthening, and 29 percent to Strategic Information.
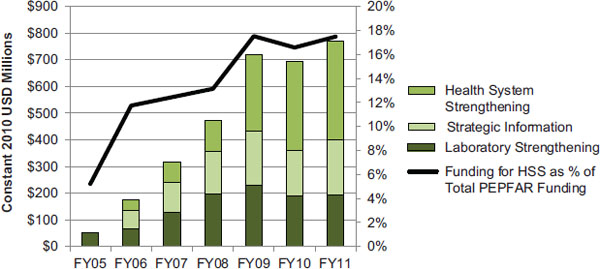
FIGURE 9-2 PEPFAR funding for HSS (country activities) (constant 2010 USD millions).
NOTES: These data represent funding for country activities planned through the Health Systems Strengthening, Strategic Information, and Laboratory Strengthening budget codes. For FY 2005, data were not reported for the Health Systems Strengthening and Strategic Information budget codes. To compare data most accurately over time, data are presented in constant 2010 USD. These totals do not include funding for the Medical Education Partnership Initiative (MEPI) and the Nursing/Midwifery Education Partnership Initiative (NEPI), which are supported by PEPFAR.
SOURCE: OGAC, 2005a, 2006b, 2007c, 2008b, 2010b, 2011d,e.
Committee’s Approach to the Assessment of PEPFAR by Health Systems Building Block
The committee systematically collected and analyzed data about PEPFAR activities and effects for each health systems building block; these data included semi-structured interviews with key stakeholders, programmatic and financial data, and other published information including peer-reviewed and grey literature, PEPFAR/OGAC guidance documents, and a targeted review of 2008 and 2010 PEPFAR Country Operational Plans (COPs) for a subset of countries. Based on these analyses, the sections below describe PEPFAR’s inputs and activities within each of the WHO health systems building blocks. PEPFAR inputs include funding for HSS activities and the strategic documents (such as the authorizing and reauthorizing legislation, 5-year strategies, and programmatic guidance) that provide direction for HSS activities. Although funding information is not disaggregated by each building block’s activities, it is presented where available. Reflecting the interdependent nature of the building blocks, many of PEPFAR’s activities involve two or more building blocks. In the following sections, PEPFAR activities are discussed within the context of the most relevant building block for each activity, based on the intent of the activity as it was described in semi-structured interviews and guidance documents. Finally, the committee drew upon these data to outline PEPFAR’s historic and current achievements and made recommendations for future directions for HSS efforts.
Introduction
The Leadership and Governance building block represents the most critical function of the health system: stewardship (WHO, 2000, 2007a). Broadly speaking, stewardship has been defined as the “careful and responsible management of the well-being of the population” (WHO, 2000, p. viii). The stewardship function in health systems is quite complex and has been difficult to operationalize since its proposal by WHO in 2000. In 2001, WHO organized a technical consultation through which experts recommended several considerations for a refined definition of stewardship and for conceptualizing “more tangible elements for better assessment of stewardship in a particular country” (WHO, 2001, p. 2). In this redefined definition, stewardship incorporates much of what is described as governance with an emphasis on the role of government; it differs from governance in its style or approach to particular tasks more than in its scope. Stewardship should be ethical, inclusive, and proactive. By reflecting the cultural, political, and societal norms in each country’s context, stewardship can facilitate addressing interactions between the health system and other aspects of society as well as influence other stakeholders in the private and other sectors. Stewardship includes the mobilization of multisectoral stakeholders to produce positive changes that address today’s challenges while maintaining a long-term perspective in order “to develop lasting solutions, to build the capacity to solve the problems of the future, and to foster continuous improvement” (WHO, 2001, p. 4).
For health systems, stewardship includes priority setting, strategy and policy development, multisectoral collaboration and coalition building, oversight and guidance for the whole health system (public and private), and regulation of all actors involved in the health system (WHO, 2007a). Although national governments are ultimately responsible for the performance of their health systems, other entities and institutions from the private sector and civil society may be involved in or carry out some of the functions of stewardship (IOM and NRC, 2009; WHO, 2000, 2007a).
Leadership and Management
In 2007, WHO organized another international consultation on improving leadership and management for health. Reports from that consultation stated that leadership and management are complex concepts relevant to many different parts of the health system and acknowledged the different, yet complementary, roles of leaders and managers. Leaders were identified as essential for setting a strategic vision and planning and
mobilizing efforts toward the realization of that vision, while skilled and motivated managers work throughout a health system to “ensure effective organization and utilization of resources to achieve results” and meet the objectives set forth in the strategic vision (WHO, 2007b, p. 1). Good leadership and management were described as key to effectively using resources devoted to health to achieve measurable results, particularly by “providing direction to, and gaining commitment from, partners and staff, facilitating change, and achieving better health services through efficient, creative and responsible deployment of people and other resources” (WHO, 2007b, p. 1). Additionally, the report stated that countries needed to develop and implement overall plans for leadership and management and that external donor assistance should coherently support these plans.
Governance
In the broadest sense, “[g]overnance refers to the structures, rules, and processes that societies use to organize and exercise political power to identify and achieve objectives [. . .] Governance includes, but is not synonymous with, government [. . .] National governance refers to the way in which a country organizes political power within its territory and controls interactions among local, sub-national, and central governmental authorities” (IOM and NRC, 2009, p. 206). Governance for the health system has been defined by WHO as “the wide range of functions carried out by governments as they seek to achieve national health policy objectives” and includes identifying the health needs of a population, setting priorities, strategic planning, policy development and implementation, and regulation of different types of actors within the health system (WHO, 2012c, p. 1). To achieve good governance, governments must have the capacity to “plan, manage, and regulate policy, financial resources, and service delivery” with efficiency, effectiveness, openness, transparency, and accountability (Brinkerhoff and Bossert, 2008; Fox et al., 2010, p. 12).
Effective governance of the HIV/AIDS response requires a multisectoral approach that is responsive to and inclusive of other government sectors as well as the private sector and civil society (Brinkerhoff and Bossert, 2008). HIV/AIDS impacts all the social and economic sectors within a country, so to be truly effective, national responses to HIV/AIDS must be multisectoral (Piot and Coll Seck, 2001). The health sector may be the focus of a country’s HIV/AIDS response (and donor support), but comprehensive responses involve other sectors, such as finance, education, labor, transportation, military, policy, women, and young people (UNAIDS, 2009). Frameworks and principles for good governance seem to be rooted in historical development from postwar conflicts and other activities that have threatened the principles of democracy, security, and the rights of people around the
globe. While there are emerging theories and frameworks for global health governance, the current frameworks for good governance are described as building on the “fundamental values of human rights, the rule of law and democracy,” and the principles of equity, participation, accountability and solidarity, in addition to promoting stability, preventing conflict, and facilitating social and economic progress (COE, 2005; Committee of Ministers, 2010, p. 391).
Entities in many PEPFAR countries are receiving a considerable amount of donor funding for health and development issues, and governance includes the management of these resources “in ways that promote national leadership, contribute to the achievement of agreed policy goals, and strengthen national management systems” (WHO, 2012c, p. 1). Although national governments are charged with the responsible and transparent management of resources as one of the functions of stewardship, a significant amount of donor funding for health and HIV/AIDS is provided to nongovernmental organizations as opposed to governmental institutions. This complicates efforts by national governments to effectively manage and oversee resources that they do not directly receive. The challenges to governance associated with the mechanisms and approaches of donor funding are discussed further in the section on the financing building block and in Chapter 10.
There are many examples of strong government engagement in and management of HIV/AIDS responses in low- and middle-income countries. In the 1980s and 1990s, the Government of Uganda, led by President Museveni, brought together government institutions and civil society organizations (CSOs) to form a nationally integrated response that eventually became known as the “multisectoral approach” (Grebe, 2009). In Rwanda, the government is “fully engaged and in command” of the HIV/AIDS programs within the country (PEPFAR/Rwanda, 2010, p. 1); it “insists on ownership of all development plans and has asked all partners to adhere to them” (Logie et al., 2008, p. 259). The Ministry of Health coordinates donor assistance for health through the Health Sector Cluster Group, and “health sector partners, including the USG, are signatories to the Sector-Wide Approach” (PEPFAR/Rwanda, 2010, p. 1).
The Role of Nongovernmental Leadership
Although partner country governments bear the primary responsibility for HIV/AIDS responses, the complexity of the disease requires “broad societal partnerships” (Grebe, 2009, p. 3). In many countries, CSOs provide a critical stewardship function by enabling “access and facilitating participation of societal groups” (Brinkerhoff and Bossert, 2008, p. 3). Global advocacy for HIV/AIDS and donor-funded global health initiatives have catalyzed stronger involvement of civil society in decision-making processes
(Yu et al., 2008). The Global Fund requires that funding proposals be developed with representatives from all sectors, including government, civil society, the business sector, and people living with HIV (PLHIV) through broad partnerships called Country Coordinating Mechanisms (CCMs) (Global Fund, 2012). PEPFAR’s Partnership Framework process also requires the participation of key partners from civil society, community- and faith-based organizations, the private sector, other bilateral and multilateral partners, and international organizations (OGAC, 2009a). In addition to direct involvement in planning and decision-making processes, civil society can also play an important role in advocating for government action or resources as well as in monitoring for good governance and responsiveness to the needs of marginalized or vulnerable populations (Grebe, 2009; Samb et al., 2009).
The private and civil sectors also play an important role in the delivery of health services, particularly those supported through donor funding (OGAC, 2009a; Yu et al., 2008). PEPFAR and the Global Fund channel considerable amounts of funding to nongovernmental and community-based organizations, which has expanded the delivery of services beyond the public sector. The increased involvement of civil society has also contributed to the decentralization of health management and highlighted the need for capacity building to ensure legitimacy, accountability, and transparency by nongovernmental service providers (Samb et al., 2009).
Perspectives on Partner Country Context
Most partner country governments have embraced HIV/AIDS as a health priority, and multiple stakeholders who were interviewed asserted that governments are engaged in leading the response (272-1-USG; 272-12-USNGO; 240-5-PCGOV; 240-33-USG; 636-2-USG; 461-8-PCGOV; 461-25-ML; 396-7-PCGOV; 396-18-USG; 166-25-USG; 934-7-PCGOV), particularly the ministries of health (240-1-USG; 240-3-USG; 272-20-PCNGO; 461-16-USG; 934-28-PCNGO; 331-6-CCM; 636-3-USG).6 Interviewees attributed the success of national HIV/AIDS responses to robust leadership and, in particular, stated that such robust leadership is important for mobilizing national responses to HIV/AIDS and raising support from external donors (461-8-PCGOV; 240-7-PCGOV):
__________________
6 Country Visit Exit Synthesis Key: Country # + ES Country Visit Interview Citation Key: Country # + Interview # + Organization Type Non-Country Visit Interview Citation Key: “NCV” + Interview # + Organization Type Organization Types: United States: USG = U.S. Government; USNGO = U.S. Nongovernmental Organization; USPS = U.S. Private Sector; USACA = U.S. Academia; Partner Country: PCGOV = Partner Country Government; PCNGO = Partner Country NGO; PCPS = Partner Country Private Sector; PCACA = Partner Country Academia; Other: CCM = Country Coordinating Mechanism; ML = Multilateral Organization; OBL = Other (non-U.S. and non-Partner Country) Bilateral; OGOV = Other Government; ONGO = Other Country NGO.
‘The President asked all leaders in the nation and communities to “not sit down” about HIV.’ (461-8-PCGOV)7
‘MOH [Ministry of Health] definitely drives the agenda and programming, even when partners have disagreements.’ (636-3-USG)
Where strong leadership was absent, HIV/AIDS programs were described as having been built outside the government (272-1-USG).
In addition to what it heard about the presence of high-level leadership in many partner countries, the committee also heard about gaps in national leadership and management skills (240-19-USACA; 587-22-USG; 196-11-USNGO; 636-9-USG; 116-7-USG; 116-11-PCGOV; 166-13-PCGOV; 935-2-USG; 935-12-USPS; 935-24-USNGO), a lack of a government commitment to the HIV/AIDS response (196-19-PCNGO; 196-20-PCNGO; 196-6-USNGO; 461-13-USACA; 636-9-USG; 636-21-USNGO), persistent challenges with leadership at the sub-national level (272-1-USG; 240-3-USG; 587-22-USG; 636-9-USG; 166-13-PCGOV; 116-23-USPS; 935-12-USPS), authoritarian leadership (587-3-USG), a lack of capacity for implementation (331-43-USG; 116-2-USG; 461-4-USG; 461-8-USG; 240-22-PCNGO; 272-20-PCNGO; 935-14-USG), poor coordination and collaboration among government entities or programs (196-11-USNGO; 461-19-USG; 240-33-USG; 636-21-USNGO), and ‘diminishing’ leadership (196-7-PCNGO; 542-11-PCNGO; 461-7-PCNGO; 461-25-ML).
Conclusion: Many stakeholders reported that there is strong leadership in partner countries for the HIV/AIDS response, both within government and in nongovernmental sectors. However, in some countries there are still challenges related to governance and management capacity for the maintenance and sustainability of the HIV/AIDS response.
PEPFAR Inputs
Guidance
PEPFAR’s authorizing legislation recognized the importance of “determined national leadership” for addressing HIV/AIDS epidemics.8 This legislation supported building leadership capacity, particularly at the community level, and specified training and “the development and implementation of national and community-based multisectoral strategies and programs” as
__________________
7 Single quotations denote an interviewee’s perspective with wording extracted from transcribed notes written during the interview. Double quotations denote an exact quote from an interviewee either confirmed by listening to the audio-recording of the interview or extracted from a full transcript of the audio-recording.
8 United States Leadership Against HIV/AIDS, Tuberculosis, and Malaria Act of 2003, P.L. 108-25, 108th Cong., 1st sess. (May 27, 2003), ![]() 2(15).
2(15).
mechanisms to achieve improved capacity.9 In its first Five-Year Strategy, PEPFAR pledged to coordinate programs with partner country policies and strategies, and it identified four strategies to build leadership at all levels of the HIV/AIDS response (OGAC, 2004, p. 20):
• “Engaging heads of state and other government officials through bilateral diplomatic interventions and multilateral forums
• Reaching out to a broad range of community and religious leaders and private institutions to generate multisectoral leadership and responses to HIV/AIDS
• Using the tools of public diplomacy and communications to inform and engage new partners, including media, in the fight against HIV/AIDS
• Using diplomatic interventions in bilateral and multilateral forums with donor nations, and communications tools with the public and private institutions, to raise additional resources for global AIDS.”
Partnership Frameworks
As specified in the reauthorization legislation, PEPFAR II focuses on ensuring the sustainability of programs and activities rather than mounting an emergency response.10 The reauthorization legislation permitted a joint, intergovernmental framework for cooperation between the U.S. government (USG), partner country governments, and other partners as a mechanism to support the transition from an emergency response to a “public health and development approach to HIV/AIDS.”11 Originally called “compacts” in this legislation, these mechanisms are now known as Partnership Frameworks (PFs). “The purpose of a Partnership Framework is to provide a 5-year joint strategic framework for cooperation between the USG, the partner government, and other partners to combat HIV/AIDS in the country through technical assistance and support for service delivery, policy reform, and coordinated financial commitments. At the end of the 5-year time-frame, the expectation is that, in addition to results in the prevention, care and treatment of HIV/AIDS, country governments will be better positioned to assume primary responsibility for the national responses to HIV/AIDS in terms of management, strategic direction, performance monitoring, decision-making, coordination, and, where possible, financial support and service delivery” (OGAC, 2009a, p. 3). PFs were framed as an
__________________
9Ibid., ![]() 301(a)(2), 22 U.S.C. 2151b(d)(6)(B).
301(a)(2), 22 U.S.C. 2151b(d)(6)(B).
10Supra, note 2 at ![]() 4, 22 U.S.C. 7603(1)(C).
4, 22 U.S.C. 7603(1)(C).
11Supra, note 2 at ![]() 301(d)(2), 22 U.S.C. 2151b-2(e)(1)(B).
301(d)(2), 22 U.S.C. 2151b-2(e)(1)(B).
opportunity to accelerate PEPFAR’s “transition of PEPFAR support from direct service provision to increased provision of technical assistance to governments” (OGAC, 2009a, p. 4). PFs, although not legally binding in either country of the partnership, are guided by the following principles:
• Country ownership
• Sustainability
• Support for coordination of country resources
• USG interagency collaboration
• Engagement and participation
• Strategic framework
• Flexibility
• Progress toward policy reform and increased financial accountability
• Integration of HIV/AIDS into strengthened health systems and a broader health and development agenda
• Monitoring and evaluation
• Collaborative but not contractual
• Transparency
• “Do no harm” (highlighting PEPFAR’s continued support of existing implementing partner service delivery systems while the transition to country ownership occurs over time) (OGAC, 2009a)
Within the context of these principles, PFs provide a broad overview of the goals, contributions, and targets for PEPFAR, as well as other partner country actors, to address HIV/AIDS in accordance with the national HIV/AIDS strategy. An additional document, the Partnership Framework Implementation Plan (PFIP), provides more detail on: the scope of activities to be carried out; the indicators that will be used to set targets and monitor progress; 5-year targets; specific commitments of the USG, partner country, and other partners; and a plan for monitoring progress. Together, the PF and the PFIP, once signed, serve as the basis for annual PEPFAR country operational planning (OGAC, 2009a). As of July 5, 2012, 19 countries and 2 regions have signed PFs. Fourteen of these countries have completed the next step of the process and have drafted PFIPs, but only four PFIPs had been signed as of July 2012 (OGAC, 2012a). Once signed by the partner country government and the USG, PFs were intended to be executed as the primary planning vehicle for the respective roles and contributions of governments and other stakeholders for the country’s HIV/AIDS response (OGAC, 2009a). The committee learned from OGAC and implementing partners that when extenuating circumstances hindered the development of PFs (e.g., political turmoil in a country), strategic plans could be submitted in lieu of PFs (NCV-9-USG; 542-13-USG). The committee regarded the PFs as the primary tool for PEPFAR to contribute to strengthening and ensuring lead-
ership and governance of the HIV/AIDS response within partner countries. These strategic planning mechanisms are further discussed in Chapter 10 on progress toward transitioning to a sustainable response.
PEPFAR Activities
Since its beginning, PEPFAR has supported partner country leadership and governance primarily through technical assistance (TA), which is defined by OGAC as “the identification of need for and delivery of practical program and technical support,” and training of human resources (OGAC, 2007b, p. 97). In response to request for programmatic data, OGAC provided the data from PEPFAR I for three indicators that measure the number of local organizations that received TA and three indicators that measure the number of individuals trained in activities related to leadership and governance.
Technical Assistance
PEPFAR implementing partners have provided TA to governmental and nongovernmental entities involved in the leadership and governance of national HIV/AIDS responses in order to build capacity for designing, implementing, and evaluating HIV/AIDS programs (OGAC, 2007b). PEPFAR collected data on three categories of TA, many of which overlap with other health systems building blocks (see definitions in Box 9-2). Because this TA was intended to strengthen some key stewardship functions, the indicator data are presented here, and the concepts are discussed in later sections that incorporate examples from the interview data. From FY 2006 to FY 2009, PEPFAR-supported partners more than tripled the total number of local organizations receiving technical assistance, from more than 11,000 to almost 36,000 (see Table 9-2).
Interviewees described PEPFAR support for ministries of health (461-8-PCGOV; 240-1-USG; 240-19-USACA; 935-24-USNGO; 542-5-USPS), HIV/AIDS coordinating bodies (461-8-PCGOV; 240-1-USG; 935-22-PCGOV), Global Fund CCMs (331-6-CCM; 587-6-CCM; 636-3-USG), and other ministries involved in the HIV/AIDS response (461-8-PCGOV; 272-17-USG; 636-3-USG; 935-10-USG; 935-14-USG; 935-19-USG). In one country visit an interviewee pointed out that PEPFAR is the only external donor that provides support for governance capacity building, specifically by working with the country’s Global Fund CCM (587-6-CCM). Interviewees also noted that PEPFAR-supported technical assistance has included support for building the capacity of partner country governments to oversee or regulate both the public and the private sectors (240-12-USG; 331-28-PCGOV; 196-11-USNGO). In some countries PEPFAR “seconds”—temporarily transfers—staff or provides salary support for key technical positions in the ministries of health (240-19-USACA;
BOX 9-2
OGAC Definitions of Technical Assistance (TA)
Related to Leadership and Governance
“TA should include regular technical communications and information dissemination sustained over a period of time. TA can be provided through a combination of strategic approaches and dissemination strategies including individualized and on-site peer and expert consultation, site visits, ongoing consultative relationships, national and/or regional meetings, consultative meetings and conferences, conference calls and web-casts, development and implementation of training curricula” (OGAC, 2007b, p. 97).
TA for HIV-related policy development includes activities “that aim to broaden and strengthen political and popular support for HIV/AIDS policies and programs; improve the operational environment for these programs, including better planning and financing; ensure that accurate, up-to-date information informs policy decisions; and build in-country and regional capacity to participate in policy development” (OGAC, 2007b, p. 101).
TA for HIV-related institutional capacity building may include strategic planning; registration; financial management; human resource management; networks development; commodities, equipment and logistics management; and infrastructure development (OGAC, 2007b, p. 102).
TA for strategic information refers to “activities that aim to strengthen HIV/AIDS surveillance, HMIS [health management information systems] and M&E [monitoring and evaluation]. Examples include providing local organizations with technical assistance in the following areas: developing or improving M&E models, methods and tools for collecting, analyzing, disseminating and using data; establishing or improving information systems; developing or improving program monitoring, planning and or conducting targeted program evaluations including operations research; monitoring and disseminating best practices to improve program efficiency and effectiveness; and/or improving data quality” (OGAC, 2007b, p. 97).
SOURCE: OGAC, 2007b.
116-7-USG; 116-11-PCGOV; 166-6-USG; 166-15-USACA; 166-30-ONGO). Several stakeholders identified investment in management personnel, which PEPFAR was supporting, as the greatest priority for country ownership (396-55-USG; 272-2-USG; 166-13-PCGOV; 116-23-USPS; 935-4-PCGOV).
PEPFAR has also supported capacity building for NGOs and CSOs, including faith-based organizations (FBOs) and community-based orga-
TABLE 9-2 PEPFAR Indicators Related to Leadership and Governance (Organizations)
| FY 04 | FY 05 | FY 06 | FY 07 | FY 08 | FY 09 | |
| Number of local organizations in thousands provided with technical assistance for: | ||||||
| For HIV-related policy development (14.1) | 3.5a | 2.9a | 2.2 | 2.5 | 7.0 | 7.3 |
| For HIV-related institutional capacity building (14.2) | 5.5 | 6.8 | 10.9 | 13.8 | ||
| For strategic information activities (13.1) | — | — | 3.7 | 8.5 | 11.2 | 14.5 |
NOTES: For FY 2004 and FY 2005, data were reported by the 15 focus countries. For FY 2006—FY 2009, the number of countries that reported data varied by year from 26 to all 31 of the countries that are the focus of this evaluation (see Chapter 2). The numbers in parentheses are the PEPFAR indicator numbers as published in OGAC's Indicators Reference Guide for FY 2007 reporting/FY 2008 planning (OGAC, 2007b). Local organizations refer to governmental or nongovernmental organizations with headquarters in a country or region served by PEPFAR. Data are presented in thousands. Indicator 13.1 was not reported prior to FY 2006.
a In FY 2004 and FY 2005, indicators 14.1 and 14.2 had not yet been separated by OGAC and were reported as a single result.
SOURCE: Program monitoring indicators provided by OGAC.
nizations (CBOs) (196-9-USNGO; 636-3-USG; 935-10-USG; 935-14-USG; 542-5-USPS; 331-34-USNGO). PEPFAR support has enabled a network of AIDS service organizations to coordinate activities across districts through a secretariat and community councils (636-11-PCNGO) and has enabled CSOs to plan and fiscally manage programs, including reporting on activities (587-21-PCNGO). In several countries, PEPFAR partners sub-contract with local governments and other entities and are devoting time and resources to building management and administrative capacity so that these implementing partners will be able to sustain the HIV/AIDS response (NCV-5-USACA; NCV-6-USNGO; NCV-16-USNGO).
Training
In conjunction with TA, PEPFAR also supports training for individuals in HIV-related policy development, institutional capacity building, and stigma and discrimination reduction (see Table 9-3). Training for HIV-related policy development and institutional capacity building serves the same purposes as TA (see definitions in Box 9-2). Again, some of these trainings may overlap with other building blocks.
Interpretation of these data is difficult; one individual may have received training (and been counted) in more than one area, and the same individuals may have been trained or retrained in multiple years. Although output indicators are the only data available from OGAC, during country
TABLE 9-3 PEPFAR Indicators Related to Leadership and Governance (Individuals)
| FY 04 | FY 05 | FY 06 | FY 07 | FY 08 | FY 09 | |
| Number of individuals in thousands trained: | ||||||
| In HIV-related policy development (14.3) | 24.1a | 45.9a | 28.3 | 26.9 | 21.1 | 23.5 |
| In HIV-related institutional capacity building (14.4) | 45.4 | 69.6 | 83.5 | 102.6 | ||
| In HIV-related stigma and discrimination reduction (14.5) | — | — | 63.6 | 186.1 | 168.0 | 103.6 |
NOTES: For FY 2004 and FY 2005, data were reported by the 15 focus countries. For FY 2006—FY 2009, the number of countries that reported data varied by year from 29 to all 31 of the countries that are the focus of this evaluation (see Chapter 2). The numbers in parentheses are the PEPFAR indicator numbers as published in OGAC's Indicators Reference Guide for FY 2007 reporting/FY 2008 planning (OGAC, 2007b). “A training must have specific learning objectives, a course outline or curriculum, and expected knowledge, skills and/or competencies to be gained by participants;” to be counted, individuals must attend the full training (OGAC, 2007b, p. 104). Double counting of individuals is to be avoided. Data are presented in thousands. Indicator 14.5 was not reported prior to FY 2006.
aIn FY 2004 and FY 2005, indicators 14.3 and 14.4 had not yet been separated by OGAC and were reported as a single result.
SOURCE: Program monitoring indicators provided by OGAC.
visits, the committee heard several examples of PEPFAR-supported activities and initiatives to increase health workforce capacity for leadership and management, including educational exchange programs (166-3-USG; 166-11-USG), integrating leadership and management skills and training into public health curricula (240-8-USG; 934-38-PCGOV), mentorship (166-11-USG; 116-7-USG), support for sub-national (e.g., district) management teams (935-4-PCGOV; 935-24-USNGO; 116-7-USG), and training programs and workshops (331-43-USG; 934-38-PCACA; 116-7-USG; 116-4-USG; 196-1-USG; 586-13-USG; 240-12-USG).
Policy Development
PEPFAR is also building capacity for policy development to support the implementation of HIV/AIDS services and activities. Contributions to policy development and strengthening, as a part of leadership and governance to plan and oversee a national response, can occur through direct negotiations with national counterparts or through less direct efforts to influence dialogue around topics or issues with policy implications. Throughout the course of its interview data gathering, the committee heard several examples of efforts by PEPFAR mission teams and implementing partners to shape or influence policy in the countries in which they work. Examples included successfully supporting the recognition of populations at elevated risk in national HIV strategy documents (331-ES; 166-ES); supporting the development of national guidelines related to the prevention of mother-to-child transmission (PMTCT) (240-24-USG; 636-9-USG), palliative care (116-13-PCNGO), and
counseling and testing (196-11-USNGO; 396-23-USG; 272-13-USG; 542-8-USNGO; 331-18-USNGO); and bolstering efforts to improve the rights of women, including protection against gender-based violence and support and vocational training for sex workers, particularly adolescent girls (272-12-USNGO; 166-17-USG; 166-27-PCNGO). As one interviewee said,
‘The studies that partners do and the influence [that] PEPFAR has been able to bring to policies, guidelines, and standards is tremendous and has been a major impact.’ (272-22-USG)
However, the committee found that even where new policies and laws have been adopted or enacted, implementation was reported to be very limited or difficult (240; 331; 196; 636; 166; 272; 461). Some examples of laws and policies that are particularly challenging to implement or enforce were those laws and policies protecting or addressing women’s land rights (240-22-PCNGO), laws protecting PLHIV (331-11-PCNGO), policies addressing HIV prevention for sex workers (196- 10-PCGOV), and guidelines for procurement (166-22-USPS). One interviewee (461-18-USG) specifically highlighted policy implementation as an area that PEPFAR could help address. There was no single, consistent reason offered by the interviewees for why policy implementation had not occurred in their various countries. One reason offered was that the government lacked commitment to policies ensuring women’s access to services (e.g., educating women about their rights or expanding service delivery), so there was no budget for implementation activities (240-22-PCNGO). In another country, the lack of local and national ownership of laws and policies resulted in limited enforcement of these laws and policies by officials (196-7-PCNGO; 196-10-PCGOV; 196-23-PCNGO).
Across the partner countries visited by the evaluation teams, data use emerged as an important theme for program planners and implementers at the national level. Some interviewees noted the lack of useful data available for policy formation, although the reasons for this limitation varied by partner country. Specifically, interviewees cited the need to conduct more surveillance or surveys because of a dearth of comprehensive data on specific populations (166-5-USG; 240-9-USG), the lack of formal processes for using the results of assessments (587-9-USG), weak linkages between national academic and research institutions and the Ministry of Health (116-23-USPS), and the failure to use data if they didn’t support the government’s priorities (587-2-USG). In contrast, in other countries, there were conscious attempts by the national governments to use data to inform program and policy decisions (196-1-USG; 116-8-USG; 934-24-PCGOV; 331-24-PCGOV; 272-6-ML; 396-9-PCGOV). The introduction of data into discussions about health policy was also mentioned by interviewees as a way of bringing awareness to sustainability challenges (116-23-USPS).
At the national level within partner countries, PEPFAR has played varying roles in creating more supportive policy environments. However, this has not been achieved in every partner country, and PEPFAR’s engagement has varied with respect to different topics. There is evidence of successful engagement for policies related to clinical services and labs, but PEPFAR’s efforts on broader structural issues are more mixed (461-18-USG; 240-21-PCGOV; 240-24-USG; 636-9-USG; 116-13-PCNGO; 116-19-PCACA; 196-10-PCGOV; 272-13-USG; 331-3-USG; 396-22-USG).
Strategic Planning
National strategic plans for the HIV/AIDS response can be used to capture or bring together multiple partners and stakeholders (331-27-PCGOV). Interviewees from PEPFAR mission teams and implementing partners described PEPFAR support for a wide range of national frameworks and strategic plans related to the HIV/AIDS response (272-12-USNGO; 272-1-USG), including those for laboratories (396-22-USG; 396-55-USG; 240-21-PCGOV; 331-17-USG) and human resources for health (240-12-USG; 934-38-PCACA).
Across countries, interviewees mentioned PEPFAR-supported TA for assisting the national government with planning, budgeting, rollout, and coordination (272-17-USG; 935-2-USG; 935-10-USG; 935-19-USG; 116-7-USG; 166-3-USG; 166-16-PCGOV). For example, in one country PEPFAR was part of a task force for developing a new national strategic plan for the HIV/AIDS response (240-7-PCGOV). PEPFAR partners are also supporting sub-national levels of the government (e.g., regions and districts) with planning tools, such as costing and resource mapping, to help in the prioritizing, planning, and budgeting of implementation activities (240-12-USG; 272-17-USG; 272-20-PCNGO; 196-1-USG; 196-6-USG; 116-7-USG).
Interviewees identified a number of challenges to national strategic planning, including turnover within ministries and sub-national departments of health, which resulted in the loss of skills and required retraining and rebuilding relationships (116-7-USG; 116-11-PCGOV; 542-8-USNGO; 542-21-USNGO; 196-6-USG; 272-16-PCNGO); turnover within PEPFAR mission teams (116-6-USG; 116-26-USG; 116-27-USG); and differing timeframes or cycles for PEPFAR and partner country budget years and multi-year strategic plans (166-13-PCGOV; 166-ES; 396-6-PCGOV; 396-7-PCGOV; 196-6-USG; 461-16-USG).
The 2007 IOM evaluation of PEPFAR I recommended increased support for country leadership through improved coordination with partner country governments, with a focus on transparency and participation during the annual planning process (IOM, 2007). In PEPFAR II, OGAC has instructed PEPFAR mission teams to align and harmonize PEPFAR planning documents (e.g., PFs and the COPs) with national strategies through consultations with partner country governments; ultimately, partner country governments must approve the strategic direction of the PEPFAR program (OGAC, 2011a).
However, PEPFAR mission teams struggled to collaborate with partner country governments that had competing health priorities or, specifically, those that did not view the HIV/AIDS response as a priority (542-2-USG; 396-18-USG). Some interviewees noted that PEPFAR was providing considerable funding for the HIV response while the government really needed or wanted funding for a broader approach to health (240-1-USG; 934-5-USG). For example, in one country where PEPFAR and Global Fund funding made up the overwhelming majority of the government’s total budget for health, the partner country government felt like PEPFAR was driving the priorities of the HIV response (240-21-PCGOV). Interviewees identified some challenges to joint strategic planning, including unrealistic targets or expectations for funding (240-33-USG) and situations in which the priorities of the partner country government did not align with OGAC/USG guidance or PEPFAR focus areas (935-17-USG; 196-11-USNGO; 636-3-USG). Although there were examples of misalignment of priorities between the USG and partner country priorities (240-1-USG; 240-7-PCGOV; 331-6-CCM), there were also many instances in which PEPFAR was supporting the government’s vision (240-2-USG; 636-3-USG; 331-15-USG; 116-16-PCGOV). Many stakeholders believed that strategic alignment had improved during PEPFAR II (240-20-ML; 935-8-PCGOV; 116-16-PCGOV; 166-13-PCGOV), and some PEPFAR partners mentioned forming agreements or signing memorandums of understanding as tools to ensure alignment with government priorities (272-15-PCNGO; 166-30-ONGO).
This evaluation committee learned from interview data that the way cooperative planning is defined has evolved over time and has ranged from joint planning before resources are allocated, to the review and adoption of select activities from the strategic plans that donors decide they wish to support, and to partner country governments taking the lead in instructing donors about the priorities needed for their HIV/AIDS responses (see Chapter 4 on funding).
PEPFAR Achievements
PEPFAR supports strengthening partner country leadership and governance primarily through training and technical assistance, and it collaborates with partner countries to improve strategic planning and to develop policies to guide national HIV/AIDS responses. Several interviewees felt that PEPFAR had a positive effect on leadership and management capacity (331-1-USG; 587-2-USG; 116-16-USG; 116-23-USPS; 240-12-USG; 935-13-PCGOV; 935-14-USG), but the committee had no data with which to understand the impact of these activities. The committee reached several conclusions regarding leadership and governance:
Conclusion: Intergovernmental planning among partner country governments, other national stakeholders, and external donors is a critical activity that is needed for the current and future responses
to HIV/AIDS. For the USG support for PEPFAR countries, this type of planning is the primary tool for ensuring leadership and governance as well as a vehicle for joint planning efforts that support the principles of ownership, mutual transparency, and mutual responsibility and accountability.
Conclusion: Over time PEPFAR has increasingly provided stronger support for partner country planning and the development of national frameworks, policies, and strategic plans. There is variable alignment or harmonization with partner country planning processes, which are primarily driven by national government priorities. It is reasonable that the USG, like all donors, has its own considerations and requirements for funding decisions. Nonetheless, PEPFAR has made progress in making its considerations a part of a joint planning process rather than a displacement of country priorities.
Conclusion: PEPFAR has supported training for management and leadership to build capacity for improved functioning of health systems with a variety of activities, including curriculum development, mentorship, and shorter-term trainings and workshops. However, the focus and outputs of these training efforts are varied, and it was difficult for the committee to determine the impact of these efforts from the data currently available.
Conclusion: PEPFAR’s capacity building approach has been holistic and includes developing human resources; strengthening financial management; and building organizational capacity at national, provincial, and district levels and across government, private, and civil society sectors. Despite these efforts, leadership and financial management capacity were frequently mentioned as challenges to effective HIV/AIDS responses.
Background and Context
Health financing includes the “mobilization, accumulation, and allocation of money to cover the health needs of the people, individually and collectively, in the health system” and has two goals: “(i) to raise sufficient funds and (ii) to provide financial risk protection to the population” (WHO, 2010c, p. 72). In most developing countries, resources for health come from both domestic and external sources. Domestic sources
include private spending (typically out-of-pocket expenditures) and public spending (national and local government expenditures). External sources include official development assistance for health (DAH), from both bilateral and multilateral sources, as well as funding from private donors and philanthropic organizations. Ideally, financial risk protection involves combining elements of prepayment schemes (and less reliance on out-of-pocket expenditures), risk pooling (such as health insurance), and incentives for health care providers to offer priority interventions efficiently (WHO, 2000). In 2010, WHO’s World Health Report addressed health financing for achieving universal access to health services.12 The report identified three challenges for achieving universal access: the availability of resources, an overreliance on direct payments at the time people need care, and the inefficient and inequitable use of resources (WHO, 2010e).
The Financing building block overlaps significantly with Leadership and Governance. This section of the report presents some information on the availability of resources for health in PEPFAR countries, followed by a discussion of PEPFAR efforts to build capacity for health financing in partner countries. Accountability and transparency are two governance functions of particular importance for health financing. Transparency, or the sharing of and access to information regarding the allocation of resources, is a necessary mechanism for accountability (Brinkerhoff and Bossert, 2008). Partner country governments, as well as donors, need to be able to account for the use of financial resources for health as well as for the outcomes and results achieved with such resources (WHO, 2010c). For example, in 2012, the Kaiser Family Foundation’s Global Health Policy Report and Voice of America News reported that hundreds of Zimbabweans marched in the capital to highlight concerns about how the funds raised from the country’s AIDS levy were being used (KFF, 2012; Mhofu, 2012).
Context of Domestic Financing
Since 2000, domestic spending on health in developing regions has increased dramatically; in 2009, government spending on health was $411 billion in developing regions, nearly 16 times as much as total DAH (IHME, 2011). Domestic spending is critical for achieving health goals, and it funds the core components of health systems, such as infrastructure and salaries, whereas donor funding is often earmarked or directed to specific diseases (e.g., the Global Fund and PEPFAR) (IHME, 2011) (240-1-USG; 240-2-USG; 934-5-USG). Many governments have recognized the importance of increasing domestic
__________________
12 Universal access is defined as ensuring that “all people have access to services and do not suffer financial hardship paying for them” (WHO, 2010e, p. ix).
funding for health in order to achieve universal access to essential health services and to achieve the health-related Millennium Development Goals.
In 2001, African heads of state gathered at a conference in Abuja, Nigeria, and pledged to increase spending on health to at least 15 percent of government spending in what became known as the Abuja Declaration (OAU, 2001). The Abuja Declaration recognized that AIDS created a “state of emergency in the continent,” and African leaders committed to mobilize the resources necessary to combat HIV/AIDS, tuberculosis, and other infectious diseases (OAU, 2001, p. 5; WHO, 2011). According to a recent report on progress toward the commitments made at Abuja, only one country—Tanzania—had met the Abuja goal by 2009, reflecting a lack of “appreciable progress in terms of the commitments the AU [African Union] governments make to health” (WHO, 2011, p. 5). The report acknowledged that it is important to consider a country’s overall level of spending for health in addition to considering the proportion of government spending on health. Between 2000 and 2010, per capita expenditure on health increased in all 31 PEPFAR countries that were considered during the committee’s evaluation (see Table 9-4). In 16 of the 29 countries for which data are available, external resources for health as a percentage of total health expenditure also increased between 2000 and 2010 (WHO, 2012b). Figure 9-3 presents external resources for health as a percentage of total health expenditure for 2010.
The committee sought data on partner country government expenditures for HIV/AIDS responses from National Health Accounts and National AIDS Spending Assessments for the 31 countries that are the focus of this evaluation. However, data were unavailable for many countries and years, so the committee was unable to examine trends in partner country HIV/AIDS funding. In a descriptive analysis of HIV/AIDS and health expenditures from 65 countries, Amico et al. (2010) found wide regional variability in HIV/AIDS expenditures. In 2007, sub-Saharan Africa countries directed more than 19 percent of total health expenditure ($4.08 per capita) to HIV/AIDS-related activities. Central and South American countries spent $2.63 per capita on HIV/AIDS activities, and this represented just slightly more than 1 percent of total health expenditure. Amico et al. also found that a one unit increase in prevalence predicted a nearly $4 million increase in HIV/AIDS funding after controlling for total health spending, foreign direct investment, gross domestic product, and population size (Amico et al., 2010).
Context of External Funding
External assistance has become an important source of funding for health for many countries. Annual DAH from bilateral and multilateral donors in-
TABLE 9-4 Total Expenditure on Health per Capita at Exchange Rate
| 2000 | 2010 | % Increase | |
| Angola | $15.8 | $123.2 | 680.1 |
| Sudan | $12.2 | $83.9 | 588.7 |
| Ukraine | $35.7 | $234.4 | 555.7 |
| Rwanda | $9.0 | $55.5 | 513.9 |
| Russian Federation | $96.0 | $525.3 | 447.5 |
| Ghana | $12.3 | $ 67.0 | 444.0 |
| China | $43.4 | $220.9 | 408.9 |
| Indonesia | $15.1 | $76.9 | 408.1 |
| Lesotho | $26.6 | $108.9 | 308.7 |
| Zambia | $18.0 | $72.9 | 305.0 |
| Botswana | $151.8 | $614.6 | 304.9 |
| Viet Nam | $20.9 | $82.9 | 296.7 |
| Nigeria | $17.1 | $62.8 | 267.0 |
| Tanzania | $10.1 | $30.9 | 206.0 |
| Uganda | $15.6 | $46.7 | 199.2 |
| Ethiopia | $5.3 | $15.7 | 195.5 |
| Guyana | $54.3 | $158.8 | 192.5 |
| Namibia | $126.0 | $361.3 | 186.6 |
| India | $19.6 | $54.2 | 177.2 |
| Malawi | $9.4 | $25.6 | 172.3 |
| Thailand | $66.0 | $179.1 | 171.5 |
| Swaziland | $75.3 | $203.1 | 169.7 |
| South Africa | $251.3 | $648.7 | 158.1 |
| Cambodia | $18.6 | $45.2 | 142.6 |
| Kenya | $19.0 | $36.8 | 93.6 |
| Côte d'Ivoire | $31.8 | $59.7 | 88.0 |
| Dominican Republic | $174.8 | $323.3 | 85.0 |
| Haiti | $25.7 | $46.4 | 80.7 |
| Mozambique | $14.1 | $21.3 | 51.3 |
| Democratic Republic of the Congo | $13.3 | $15.8 | 18.4 |
| Zimbabwe | $60.0 | — | — |
NOTES: Data represent total health expenditure per capita expressed at average exchange rate for that year in USD. Total health expenditure includes government and private expenditure on health and may include donor funding. Countries are sorted by the percentage increase from 2000 to 2010. Data have been rounded. No data were available for Zimbabwe after 2001.
SOURCE: WHO, 2012b (retrieved November 2012).
creased from approximately $11 billion in 2000 to nearly $28 billion in 2011 (IHME, 2011). DAH doubled between 2000 and 2008, but it has slowed in recent years because of the global economic recession (IHME, 2011). In the United Kingdom and the United States, economic uncertainty has prompted discussions about reducing development assistance (IHME, 2011).
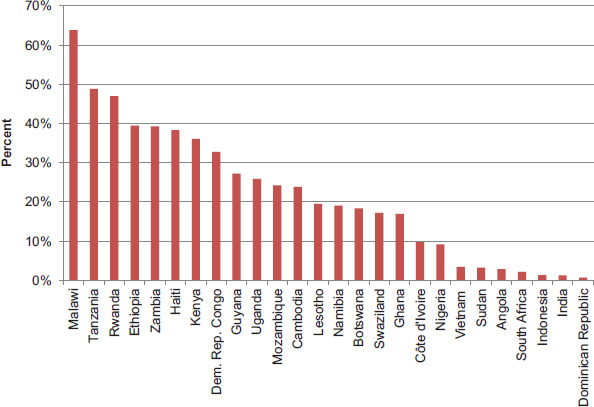
FIGURE 9-3 External resources for health as percent of total health expenditure, 2010. NOTES: Russia and Zimbabwe have been excluded from the figure because there were no data available. China, Thailand, and Ukraine have been excluded from the figure because less than 0.5 percentage of their total health expenditures came from external resources in 2010.
SOURCE: WHO, 2012b (retrieved November 2012).
Debt relief is another form of external assistance that can be used for health financing. Many heavily indebted poor countries (HIPCs) qualify for debt relief through the HIPC Initiative and the Multilateral Debt Relief Initiative. In eligible countries, bilateral and multilateral creditors agree to forgive some of the national debt burden, which reduces spending on debt service (e.g., interest) and increases the availability of domestic resources for social spending on such issues as health and education (IMF, 2012). In a recent report on progress toward the Abuja Declaration commitments, WHO suggested that countries can increase their investment in health by “making larger claims on their funds from debt relief” (WHO, 2011, p. 4). Of the 31 countries included in the committee’s evaluation, 12 qualify for assistance under the HIPC Initiative (IMF, 2012).
Although external assistance is a primary source of resources for health and HIV/AIDS in developing countries, there are serious concerns about the sustainability of donor funding. The question of whether funds from PEPFAR and other donors have led to a displacement of government funding for HIV continues to be an open question (Garg et al., 2012; Lu et al., 2010). In some of the countries visited, the committee heard from a
variety of interviewees that partner country governments had diverted domestic resources away from health in response to the contributions of donors (166-13-PCGOV; 461-4-USG; 461-15-USG; 461-17-PCNGO; 935-2-USG). Donor dependency and other challenges associated with the sustainability of national HIV/AIDS responses are discussed in Chapter 10.
Coordinating Financial Resources for the HIV/AIDS Response
Many developing countries receive external assistance for HIV/AIDS from multiple donors and funding mechanisms (see Chapter 4 for more information). Bilateral and multilateral donors may provide funding directly to partner country governments, while others fund projects implemented by civil society or faith-based organizations. Coordinating these resources can be a challenge for partner country governments, a theme the committee heard often during country visits (935-14-USG; 461-12-PCGOV; 542-6-ML), but several structures have been proposed to improve coordination (Spicer et al., 2010). In the late 1980s, WHO’s Global Programme on AIDS introduced the concept of National AIDS Commissions, and, as described in Chapter 1, the “Three Ones” principles call for National AIDS Coordinating Authorities to take responsibility for coordinating resources for and implementation of national HIV/AIDS responses (HLSP Institute, 2006; Spicer et al., 2010; UNAIDS, 2004). In 2002, the Global Fund introduced the concept of CCMs—broad partnerships with representatives from all sectors, including government, civil society, the business sector, and PLHIV—to coordinate the development of Global Fund proposals and the implementation of grants (Global Fund, 2012; Spicer et al., 2010). In some countries, one or more of these entities have been integrated, but in others the existence of parallel coordination structures is a challenge for the effective governance of the HIV/AIDS response (331-9-PCNGO) (Spicer et al., 2010).
The way in which donor funding is provided can affect a government’s ability to coordinate and manage donor resources. Some donor funding is provided as budget support—this funding goes directly to government treasuries and may or may not be earmarked for specific purposes. Many countries have a basket funding mechanism through which donors contribute resources to a common pool and the government is able to allocate resources as needed to support national priorities. Finally, some donors provide project-based funding, which may bypass government accounting and budgeting systems and go directly to implementing partners (USAID, 2007). Donors may provide aid through any or all of these approaches. In some countries, partner country governments struggled to successfully coordinate donor funding, implement projects, and ensure accountability with multiple streams of funding (935-14-USG; 461-12-PCGOV; 396-16-PCGOV; 166-ES). Partner country governments also reported to other interviewees that it
was difficult to coordinate the response without control over the money (542-6-ML; 396-16-PCGOV). Across countries, interviewees agreed that funding provided through budget support or basket funding mechanisms was preferable to project-based funding. Many interviewees identified direct budget support as a way to increase country ownership of the response (NCV-9-USG; 240-21-PCGOV; 240-33-USG; 166-34-PCGOV; 542-6-ML), to build capacity (240-5-PCGOV; 240-21-PCGOV; 240-23-PCGOV; 331-4-PCGOV; 166-22-USPS; 166-34-PCGOV; 461-25-ML), and to ensure sustainability (331-4-PCGOV).
Several countries use basket funding to increase the coordination of donor resources, and some coordinate resources and activities through a Sector-Wide Approach mechanism, which is a type of basket funding approach (166-13-PCGOV; 166-16-PCGOV; 116-18-PCNGO; 461-19-USG; 935-2-USG; 331-ES; 934-29-USNGO; 934-ES; 196-ES). Interviewees described basket funding as a way to avoid fragmenting the national strategy by funding streams (461-5-PCGOV), to reduce transaction costs associated with funding from and reporting to multiple donors (116-5-PCGOV), and to allow donors to support the priorities set by the government (461-21-ONGO). In one country, basket funds were used to support the planning for and delivery of services by district health councils (935-24-USNGO); in another, this mechanism of funding was used to support CSOs that provided HIV/AIDS services (461-19-USG; 461-12-PCGOV). In a few countries, the governments wanted to have partners contribute to a basket fund or pooled funding mechanisms, but donors refused or were wary because of concerns about government mismanagement or the inefficient use of the money; in the most severe cases, such concerns prompted donors to withdraw from basket or pooled funding mechanisms (331-29-PCGOV; 636-4-PCGOV; 461-12-PCGOV; 166-22-USPS; 116-ES).
PEPFAR Inputs
PEPFAR funding provides external assistance to partner country HIV/AIDS responses and is delivered in different ways in different countries. PEPFAR funds are not generally contributed toward basket funding mechanisms because the U.S. Congress has placed legislative requirements and limitations on how PEPFAR resources may be used (NCV 9-USG). Because it is difficult to account for how basket funding is used (and to report on indicators related to such funding), most PEPFAR mission teams do not provide direct PEPFAR funding to such funding mechanisms (NCV-9-USG; 636-4-PCGOV; 934-2-USG; 934-25-USPS; 116-2-USG; 935-9-USG). There are some exceptions, where PEPFAR contributes to a funding pool that supports a partner country AIDS coordinating authority or a sector-wide approach to health (116-18-PCNGO; 196-12-PCGOV). As described in Chapter 4, some PEPFAR funding is provided to partner country governments and other local entities directly as prime partners or indirectly as subpartners. The committee’s assessment of prime partner
funding in a subset of 13 countries from FY 2004 to FY 2010 revealed that during this time period, 9 percent of PEPFAR funding was provided to partner governments as prime partners and 24 percent was provided to nongovernmental entities based in partner countries (including nonprofit organizations, for-profit firms, and academic institutions) (see Chapter 4 for more information about PEPFAR prime partners). In general, PEPFAR has been criticized for “a lack of willingness to coordinate with other donors” (Spicer et al., 2010, p. 3). Representatives from multilateral and other bilateral donors supported that criticism, describing coordination with PEPFAR as a challenge (196-13-OBL; 240-20-ML), but many noted that coordination of donors is a challenge in general and not necessarily unique to PEPFAR (331-5-ML; 461-25-ML; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS).
Guidance
OGAC has highlighted capacity building for governmental and nongovernmental organizations as one of its strategies to ensure sustainability and country ownership, and the key activities for capacity building include improving financial management and accounting systems as well as strategic information activities that inform financial management (OGAC, 2004, 2008a, 2009f). In particular, PEPFAR’s first Five-Year Strategy called for the strengthening of financial, administrative, and management systems for implementing partners, particularly NGOs and CBOs, to build capacity for the delivery of HIV/AIDS services (OGAC, 2004). Capacity building activities will be discussed in this chapter, while the implications for sustainability and country ownership will be discussed in Chapter 10.
The reauthorization legislation authorized the U.S. Treasury “to provide assistance for advisors and partner country finance, health, and other relevant ministries to improve the effectiveness of public finance management systems in partner countries.”13 In its second Five-Year Strategy, PEPFAR identified the long-term (3- to 5-year) objective of strengthening both partner country governance and the financing of the partner countries’ health systems in order to contribute to country ownership and sustainability (OGAC, 2009f). “PEPFAR’s long-term goal is to see more management and operation of bilateral programs conducted by the countries themselves, with financial support through the Global Fund. In order to promote this goal, PEPFAR is working to improve grant performance, quality, and consistency of services, and transparent and accountable financial management” for both PEPFAR and Global Fund grants (OGAC, 2009e, pp. 14–15).
__________________
13Supra, note 2 at ![]() 204(a), 22 U.S.C. 7621(b)(1).
204(a), 22 U.S.C. 7621(b)(1).
PEPFAR Activities
Technical Assistance/Capacity Building
Across countries, PEPFAR funding has supported TA to partner country governments for planning, budgeting, and coordination (272-17-USG; 116-11-PCGOV). Often this TA is directed to both national and sub-national levels of the government (240-12-USG; 272-17-USG; 935-14-USG; 116-23-USPS; 166-22-USPS).
PEPFAR partners have also been working to strengthen the financial management capacity of partner country NGOs (272-11-PCNGO; 240-12-USG; 240-19-USACA; 935-14-USG; 196-9-USNGO; 542-5-USPS), FBOs (240-26-PCNGO; 196-20-PCNGO), and service providers (331-34-USNGO; 636-15-PCNGO; 934-8-USNGO; 272-3235-PCNGO). Often, partner country governments and other entities will be subpartners of PEPFAR prime partners that actively work to build capacity for grants and financial management (240-12-USG; 196-9-USNGO; 116-4-USG; 272-16-PCNGO). One prime partner described regular visits to recipient organizations that were used to transfer skills about proper procedures for reporting and to keep track of funding (NCV-5-USACA). Another prime partner subcontracts directly with district-level governments in partner countries and works through government systems to build financial and management capacity (NCV-6-USNGO). Other strategies include incorporating health financing topics into the economics curriculum at local universities (116-23-USPS), mentoring district health personnel and management teams (116-7-USG; 935-12-USPS), and seconding staff for temporary assignments in national and regional governments (240-12-USG).
Generating and Mobilizing Domestic Resources
Many partner country governments have committed to (240-3-USG; 331-5-ML; 331-15-USG; 587-1-USG; 272-5-PCGOV) or have already begun increasing domestic resources for their HIV/AIDS responses (396-2-PCGOV; 331-28-PCGOV). In some situations, the government has taken financial responsibility for specific components of the response, such as the procurement of antiretrovirals (ARVs) (272-22-USG; 934-25-USPS; 196-10-PCGOV; 542-9-PCGOV) or salaries for health care workers (240-2-USG; 331-15-USG; 636-7-PCNGO; 196-7-PCNGO). However, several interviewees noted that partner country governments are unable to take over total financing for the response (587-14-PCGOV; 587-22-USG), particularly at the current level supported by donors (240-3-USG; 272-32/35-PCNGO; 272-36-USG; 461-8-PCGOV; 935-10-USG), and that in some cases, governments are unwilling to contribute more resources because of competing health and development priorities (331-6-CCM; 461-15-USG; 396-18-USG).
Interviewees from partner country governments recognized the need to mobilize existing resources and to generate new domestic resources to ensure sustainability of the HIV/AIDS response (461-6-PCGOV; 331-28-PCGOV; 331-29-PCGOV; 240-7-PCGOV; 196-10-PCGOV; 116-5-PCGOV; 116-13-PCGOV). In a few countries, additional resources have been mobilized through requirements that govern-
ment ministries or agencies dedicate a specific portion of funding to the HIV/AIDS response (331-27-PCGOV; 636-4-PCGOV; 636-11-PCNGO; 166-16-PCGOV). Interviewees identified the need for greater advocacy aimed at convincing partner country governments to increase resources (542-8-USNGO; 935-12-USPS; 461-19-USG). Through an international workshop, PEPFAR has supported partner country organizations (governmental and nongovernmental) to advocate for more government investment in the HIV/AIDS response (461-19-USG). In some countries, PEPFAR is training CSOs to advocate for more funding to local government and also supporting them to mobilize resources from non-USG sources (196-9-USG).
‘PEPFAR needs to help countries look at ways to make this a sustainable domestic response, for example, how to mobilize domestic resources. The direction is that countries will be required to invest.’ (461-5-PCGOV)
In order to generate new domestic resources for the HIV/AIDS response, several countries are exploring—or are interested in exploring—innovative financing mechanisms such as AIDS trust funds or levies (934-7-PCGOV; 934-10-PCGOV; 934-46-PCGOV; 934-42-PCACA; 331-6-CCM; 331-27-PCGOV; 166-34-PCGOV; 461-5-PCGOV), but the committee found limited evidence that PEPFAR is supporting such mechanisms (see Box 9-3). A few stakeholders mentioned expanding health insurance as another mechanism for increasing resources for health (116-13-PCGOV; 272-5-PCGOV; 116-23-USPS). PEPFAR support for insurance schemes is described below in the section that discusses removing financial barriers to access.
BOX 9-3
Select Innovative Financing Mechanisms from
Committee-Collected Interview Data
Interviewees expressed interest in the following strategies to increase domestic resources available for the health sector or HIV/AIDS activities:
• Tax credit/rebate or increased taxes from private/corporate sector (331-6-CCM)
• Value-added tax (VAT) to support national health insurance (331-6-CCM)
• National fund (331-6-CCM); HIV/AIDS fund is necessary so government has a dedicated pot of money for HIV (331-27-PCGOV)
• AIDS trust fund (935-8-PCGOV; 331-40-PCPS); National HIV/AIDS Trust Fund funded through tax on consumer commodities (461-5-PCGOV; 116-16-PCGOV)
Global Fund–Related Support
As reported in Chapter 4, PEPFAR and the Global Fund are the largest donors for HIV/AIDS worldwide, and many countries receive both PEPFAR and Global Fund support. Chapter 4 also describes some of the ways in which PEPFAR and Global Fund activities are aligned in partner countries. In most countries, partner country governments are the Principal Recipients of Global Fund grants, so this money moves through the government systems (116-11-PCGOV; 240-7-PCGOV). Several countries have experienced delays in Global Fund disbursements or had Global Fund applications rejected due to
• lack of absorptive capacity (331-47-USG)
• pipeline issues (396-15-USNGO)
• supply chain management or procurement problems (lack of capacity) (542-21-USNGO; 636-3-USG; 166-3-USG; 166-31-USG)
• failure to meet deadlines (116-11-PCGOV)
• lack of capacity to complete Global Fund monitoring and evaluation (M&E) or reporting requirements (166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS)
• financial management or mismanagement (166-3-USG; 166-31-USG; 934-12-CCM)
• failure to expend funding in a timely manner (636-16-USG)
‘One of the challenges in terms of the Global Fund is the issue of grants management by the MOH [Ministry of Health]; moving money is an issue as the MOH sits on the funding.’ (636-16-USG)
Turnover within high-level government leadership has also affected governments’ ability to access funding, particularly when staff members with grant management experience leave (116-11-PCGOV; 166-22-USPS). To address these issues, PEPFAR mission teams in 19 countries include Global Fund Liaisons or Advisors who support partner country management and implementation of Global Fund resources (240-8-USG; 166-31-USG; 196-6-USG; NCV-11-USG). A portion of the USG’s contribution to the Global Fund is withheld each year to support technical assistance with CCMs, National AIDS Commissions, ministries of health, and Global Fund Principal Recipients to increase productivity and improve the management of and structures related to Global Fund resources (NCV-11-USG; 331-6-CCM; 196-6-USG; 542-5-USPS; 166-3-USG) (Bilimoria, 2012). The Grant Management Solutions project, led by Management Science for Health, provides support in multiple areas, such as “governance and leadership challenges; financial and grants management; procurement and supply management; monitoring, evaluation, and reporting; and improving the skills and participation of civil society organizations and local consultants” (MSH, 2009). In 2011, OGAC announced the Country Collaboration Initiative, through which additional funding was available to PEPFAR
mission teams to increase coordination and collaboration with Global Fund–supported activities within countries and “to optimize Global Fund grant performance” (Goosby, 2011; OGAC, 2012b, p. 59).
The committee heard about specific examples of Global Fund–related support during interviews with partner country stakeholders. In one country, PEPFAR supports the salary of a Global Fund coordinator who works with various departments in the Ministry of Health (MOH) that implement Global Fund–supported projects. This coordinator ensures that everyone stays on schedule and that reporting deadlines are met (116-11-PCGOV). In another country, a PEPFAR partner helped the MOH set up a Global Fund Management Unit to support the financial management, monitoring and evaluation, and supply chain management of Global Fund grants (166-22-USPS). PEPFAR funding also supports secondments, or temporary assignments, of technical staff to work directly with the MOH staff in the Global Fund Management Unit (166-22-USPS; 166-31-USG). In some countries, staff members from PEPFAR mission teams sit in on or are members of the Global Fund CCM (331-6-CCM; 636-3-USG; 934-12-CCM; 196-26-USG; 166-31-USG; 396-12-USG).
PEPFAR has supported the development of Global Fund proposals and applications (240-20-ML; 331-6-CCM; 331-47-USG; 542-21-USNGO; 116-8-USG; 196-ES). PEPFAR has also supported the strengthening of systems and structures necessary to implement programs with Global Fund resources (331-47-USG), including CCMs (587-6-CCM; 240-12-USG; 934-12-CCM). In some countries, PEPFAR supports capacity building of the NGOs that are Principal Recipients of Global Fund money (240-12-USG; 240-29-USNGO; 542-2-USG).
In addition to supporting partner countries, the USG, through OGAC/PEPFAR, has been instrumental in the formation of the Global Fund and the structures through which it operates (NCV-16-USG). The USG holds a permanent seat on the board of the Global Fund, which is currently held by Ambassador Eric Goosby. PEPFAR provides “critical guidance to and oversight of” the organization (Bilimoria, 2012, p. 1416). More recently, OGAC has established a PEPFAR-funded position based in Geneva for a liaison between PEPFAR and the Global Fund to assist with day-to-day coordination (NCV-21-ML). PEPFAR has also been working with the Global Fund to provide country information to facilitate decision making, because PEPFAR has a greater presence on the ground in countries through PEPFAR mission teams (NCV-11-USG).
Tracking Resources
The ability to measure and track government expenditure on health allows governments to be transparent about resource allocation and to “identify opportunities to improve resource flows” (OGAC, 2009f, p. 21). Access to this information makes it possible for civil society to hold govern-
ments accountable, but the data on government health spending are often incomplete, of poor quality, or unavailable (IHME, 2011). As of August 2012, at least 15 PEPFAR countries (out of 31) had costed national HIV/AIDS plans (WHO, 2012a). However, interviewees in several countries identified the lack of expenditure data, costing data, or both, as a challenge to planning and sustainability (331-10-PCGOV; 587-6-CCM; 935-4-PCGOV; 166-5-USG).
In recent years, PEPFAR has increased its support for partner country governments to quantify expenditures and project funding needs through National Health Accounts (NHAs), National AIDS Spending Assessments (NASAs), costing efforts, and GAP analysis (Goosby, 2012b; Holmes et al., 2012; OGAC, 2009f). PEPFAR partners have supported costing and modeling efforts that have helped governments project costs and resource needs (240-12-USG; 331-10-PCGOV; 587-7-PCGOV; 587-10-USG; 272-22-USG; 272-36-USG; 396-16-PCGOV; 935-2-USG; 461-15-USG; 934-25-USPS). Some of these efforts have contributed to increased partner country capacity to forecast procurement needs for ARVs and other commodities (331-10-PCGOV; 272-1-USG; 272-20-PCNGO). PEPFAR supported-partners have also provided training and technical assistance to build capacity for partner country governments for tracking health and HIV/AIDS expenditures through NHAs or NASAs (587-10-USG; 116-23-USPS; 461-15-USG). However, in one country, staff trained in data collection for NHAs frequently left the MOH, necessitating frequent retraining and challenging efforts to institutionalize the NHA process and for routine use of the data (116-23-USPS).
Interviewees frequently mentioned the lack of transparency about PEPFAR funding as a challenge for understanding HIV/AIDS expenditures in partner countries. Partner country governments have been frustrated by not knowing where PEPFAR money was going (240-5-PCGOV; 240-7-PCGOV; 240-20-ML; 331-4-PCGOV; 331-10-PCGOV; 542-6-ML; 935-8-PCGOV; 116-2-USG; 116-16-PCGOV). Some interviewees wondered how partner country governments could be expected to hold implementers accountable if the governments did not know where the money was going (935-8-PCGOV; 461-11-PCGOV). Interviewees from partner country governments described challenges in tracking the amount of money that PEPFAR was providing to support particular services and activities (e.g., home-based care, community outreach, condom provision) (240-5-PCGOV; 240-7-PCGOV; 240-20-ML; 331-4-PCGOV; 331-10-PCGOV; 116-16-PCGOV; 935-8-PCGOV) as well as challenges in tracking the amount of money that PEPFAR was providing to specific implementing partners (e.g., district governments, local NGOs, U.S.-based universities) (331-4-PCGOV; 116-16-PCGOV). Partner country governments wanted to know more than what’s been planned in the COPs—they wanted to know where (geographically) the money is going and what services are being supported so that they can identify unmet needs. This has opened the door for increased dialogue with partner country governments (NCV-9-USG).
In at least one country, this situation was reversed, and the PEPFAR mission team described difficulties in planning activities due to a lack of
transparency from the partner country government about the amount of money it would invest in the HIV/AIDS response (636-3-USG). Representatives from other donors and multilateral organizations reported that donors other than the USG often are also not transparent with partner country governments about the amount of funding that donors will provide (331-5-ML; 587-2-USG).
Removing Financial Barriers to Access
Direct payments, or payments required for a service at the time it is delivered, are one of the largest barriers to access to health services (WHO, 2010e). To reduce reliance on direct payments, governments should encourage risk-pooling, prepayment approaches in which “payments [are] made in advance of an illness, pooled in some way and used to fund health services for everyone who is covered—treatment and rehabilitation for the sick and disabled, and prevention and promotion for everyone” (WHO, 2010e, p. xiv; Xu et al., 2007). In some countries visited by the committee, access to HIV/AIDS services is free (240-15-USG; 196-17-PCGOV; 935-12-USPS; 935-13-PCGOV; 636-4-PCGOV), but in others a fee is assessed in order to acquire the drugs needed to survive, which creates a financial barrier to access (240-15-USG; 935-19-PCGOV; 396-25-PCGOV; 934-15/16-PCGOV). Transportation costs were also described as a financial barrier for accessing services (935-19-PCGOV; 396-25-PCGOV); experience in at least one country showed that there was a large uptake of services when the fees were eliminated (240-1-USG).
Some PEPFAR countries have or are moving toward national health insurance programs (331-5-ML; 240-12-USG; 272-5-PCGOV; 272-20-PCNGO; 166-13-PCGOV), but these are not always sufficient to reduce financial barriers to access (WHO, 2010e). In Rwanda, the government has implemented a community-based insurance scheme called Mutuelles, which has improved the utilization of maternal and child care services and reduced catastrophic household spending on medical expenses (Lu et al., 2012). Other PEPFAR partner countries rely on the private sector to provide access to insurance (116-23-USPS) or have a mixed system in which private insurers cover some of the population and the government covers the poor (396-4-PCGOV). However, HIV/AIDS treatment and ARVs are not always covered through these insurance schemes (331-6-CCM; 396-16-PCGOV), and PLHIV have struggled to pay monthly premiums, which has been a barrier to accessing care (331-6-CCM; 331-32-PCNGO; 331-38-USPS).
PEPFAR is supporting access to insurance in several partner countries by supporting the implementation of national insurance schemes (240-12-USG), piloting social insurance strategies (240-12-USG), piloting a membership-managed insurance fund (935-14-USG), piloting a group insurance scheme (935-14-USG), costing health services for incorporation in insurance benefits (396-16-PCGOV), and exploring potential insurance providers (636-4-PCGOV).
PEPFAR Achievements
In a few countries, stakeholders have been able to use information from PEPFAR-supported NHAs and NASAs for policy decisions and evidence-based advocacy. In one country, for example, NHA data contributed to an understanding of the sources of health resources and affected policy decisions regarding resource allocations (116-23-USPS). In this example, data from NHAs revealed low rates of insurance contribution, and stakeholders used this information to advocate for employers to contribute more to the health costs of their employees (116-23-USPS). PEPFAR-supported costing studies in one country improved the government’s ability to plan and budget (935-2-USG), and PEPFAR has supported capacity building for financial management and resource mobilization at multiple levels and across sectors (240-12-USG; 272-17-USG; 272-3235-PCNGO; 935-14-USG; 116-11-PCGOV; 116-23-USPS; 166-22-USPS; 331-34-USNGO; 636-15-PCNGO; 934-8-USNGO). As a result of increased capacity, in a few countries, local implementing partners have transitioned from subpartners to prime partners and have received direct PEPFAR funding (NCV-5-USACA; NCV-6-USNGO). However, support to improve financial management capacity and accountability is still needed in many countries. Interviewees identified the need for increased financial management capacity at national (240-3-USG; 331-30-USPS; 166-13-PCGOV; 461-12-PCGOV; 636-16-USG) and sub-national levels (240-19-USACA; 116-23-USPS; 196-13-OGOV; 196-28-USG).
Conclusion: Although there are nascent efforts in PEPFAR for the costing of services and the projecting of needs to help countries develop a costed HIV/AIDS response, PEPFAR has not yet systematically implemented assistance for partner countries to develop resource mobilization plans, conduct costing activities and resource projections, or identify funding needs.
Background and Context
A health information system (HIS) is a set of components and procedures organized to generate sound and reliable health information “to enable decision-makers at all levels of the health system to identify problems and needs, make evidence-based decisions on health policy and allocate scarce resources optimally” (Health Metrics Network 2008; WHO, 2010c, p. 44). In addition to data generation, the key functions of an HIS are data compilation, analysis and synthesis, and communication and use of data (WHO, 2010c). Although HISs are sometimes equated with program monitoring and evaluation—and, indeed, having an HIS in place is essen-
tial for being able to successfully carry out monitoring and evaluation of health programs—an HIS also serves broader objectives, such as supporting patient and health facility management, enabling planning, and providing information for situational and trends analyses (WHO, 2010c).
The Health Metrics Network, which helps countries and other partners strengthen HISs for evidence-based decision making, has developed an HIS framework made up of six components—HIS resources, indicators, data sources, data management, information products, and dissemination and use of information—organized into three categories: inputs, processes, and outputs (see Figure 9-4). HIS inputs include the legislative environment and resources that are put into the HIS (Health Metrics Network, n.d.). Indicators are used to measure the effectiveness of health systems and should reflect change over time. Data sources for the HIS can vary from facility-level information to periodic nationwide surveys; gathering the appropriate data can be challenging because it requires coordination across sectors (Health Metrics Network, 2008) (see Figure 9-5). The use of information generated by an HIS can vary from day-to-day operational decisions to longer-term strategic decision making; therefore, having multiple data sources is necessary (see Figure 9-5).
Data management, information products, and dissemination and use rely heavily on having a skilled workforce to analyze and interpret health information. Sharing and analyzing data for use in decision making can be a challenge when there are limited available resources and capacity for these processes, as is the case in many low-income countries. Because each component of an HIS requires infrastructure and skilled human resources, it is not surprising that in many countries HISs continue to be inadequate for meeting stakeholder needs (AbouZahr and Boerma, 2005).
PEPFAR Inputs
Guidance
In 2004, PEPFAR’s first Five-Year Strategy recognized that existing HISs in many partner countries lacked the capacity to provide the information necessary to monitor and manage interventions. The strategy called for the design of “country-appropriate HIV management information systems,” to be built from the ground up, and for the integration of these systems into regional or national HISs (OGAC, 2004, p. 73). The strategy planned to develop and improve partner country capacity to collect client-, facility, and district-level and surveillance information (OGAC, 2004). During PEPFAR I, support for information systems, surveillance, and monitoring and evaluation was referred to as “upstream support” because it was often
FIGURE 9-5 Data needs and sources at different levels of the health care system.
NOTE: KAP = knowledge, attitudes, and practices.
SOURCE: AbouZahr and Boerma, 2005.
provided at the national level and benefitted multiple sites and programs (OGAC, 2005b) (see Figure 11-1).
From FY 2006 to FY 2011, 4 to 5 percent of total funding for PEPFAR country activities was budgeted for strategic information activities (OGAC, 2006b, 2007c, 2008b, 2010b, 2011d,e). Until FY 2011, strategic information activities included monitoring and reporting PEPFAR partner results as well as surveillance, surveys, and efforts to strengthen partner country information systems (OGAC, 2008a, 2009b, 2010a). The FY 2012 COP guidance advised mission teams that activities planned under the strategic information budget code should aim “to build individual, institutional, and organizational capacity in country” for strategic information activities (OGAC, 2011b, p. 68).
PEPFAR Activities
Through technical assistance and support and capacity building, PEPFAR mission teams and partners have supported the development of national plans and frameworks for monitoring the HIV/AIDS response
(240-23-PCGOV; 166-1-USG) and have supported and participated in national-level M&E working groups (636-1-USG; 587-9-USG; 396-19/20-USG) (PEPFAR/Ethiopia, 2007; PEPFAR/Thailand, 2009). PEPFAR has also supported building capacity for national health management information systems (HMISs), national M&E and reporting systems for HIV/AIDS (240-19-USACA; 636-ES), the integration of various national data systems (196-8-ML; 196-11-USNGO) (PEPFAR/Botswana, 2009), and surveys and surveillance (934-21-USG; 331-1-USG; 240-8-USG). See Chapter 11 for additional information on PEPFAR’s support of surveys and surveillance.
Given the limited capacity for collecting and using strategic information at the onset of and during PEPFAR I, mission teams invested in strategic information capacity building efforts focused on strengthening HIV/AIDS-related M&E and surveillance (OGAC, 2005b, 2006a). Mission teams reported on two PEPFAR indicators to monitor capacity building for strategic information activities (including M&E, surveillance, and HMIS) (OGAC, 2005c, 2007b). From FY 2004 to FY 2009, the number of individuals trained and the number of local organizations provided with TA for strategic information activities with PEPFAR support greatly increased (see Table 9-5). Interpretation of these data is difficult because individuals may have been trained or retrained in multiple years and local organizations may have been provided with TA in multiple years.
Most of PEPFAR’s investments in strengthening HISs have been related to training and analytics, supply chain issues, human resource information systems, laboratory information management systems, patient record management systems, and electronic health records (see Box 9-4 for examples of PEPFAR-supported information systems). The committee heard few examples of integration of these systems, a fact that reflects disciplinary and
TABLE 9-5 PEPFAR Indicators Related to Strategic Information and Information Systems
| FY 04 | FY 05 | FY 06 | FY 07 | FY 08 | FY 09 | |
| Number of individuals in thousands trained in strategic information (13.2) | 9.3 | 18.0 | 42.6 | 38.7 | 53.0 | 64.5 |
| Number of local organizations with technical assistance in thousands for strategic information activities (13.1) | — | — | 3.7 | 8.5 | 11.2 | 14.5 |
NOTES: For FY 2004 and FY 2005, data were reported by the 15 focus countries. For FY 2006–FY 2009, the number of countries that reported data varied by year from 26 to all 31 of the countries that are the focus of this evaluation (see Chapter 2). The numbers in parentheses are the PEPFAR indicator numbers as published in OGAC’s Indicators Reference Guide for FY 2007 reporting/FY 2008 planning (OGAC, 2007b). Data are presented in thousands.
SOURCE: Program monitoring indicators provided by OGAC.
BOX 9-4
Select Examples of PEPFAR-Supported Information Systems
• Human Resource Information System (HRIS) (240-12-USG; 166-12-USG; 116-7-USG)
• Patient information management system (PEPFAR/Botswana, 2009)
• Electronic medical records (166-15-USACA; 166-ES; 116-ES; 116-9-PCNGO; 636-1-USG; 461-13-USACA)
• Commodities/logistics/supply chain management information systems (240-2-USG; 240-5-PCGOV; 331-12-USG; 331-43-USG; 331-38-USPS; 166-12-USG). In one partner country, a PEPFAR-supported local partner has developed an electronic medical record system and shared relevant data for drug procurement in order to quantify drug needs (116-9-PCNGO)
• Pharmaceutical information management (542-21-USNGO; 240-5-PCGOV)
• Laboratory/Laboratory Information Management Systems (LIMSs) (166-11-USG; 166-12-USG; 331-15-USG; 331-17-USG; 331-38-USPS; 396-22-USG; 636-14-USNGO)
SOURCES: PEPFAR/Botswana (2009) and country visit interview data.
structural barriers within partner countries as well as software interoperability issues.
When PEPFAR began, partner country information systems were often fragmented, and it was difficult for PEPFAR mission teams to integrate or strengthen existing parallel systems. In many countries, PEPFAR originally set up or supported parallel information systems rather than strengthening existing national HISs (331-24-PCGOV; 587-2-USG; 537-9-USG; 636-1-USG; 636-9-USACA; 166-4-USG; 166-12-USG; 166-34-PCGOV; 272-27-USG; 461-11-PCGOV; 396-ES), often because of the challenges associated with national systems. Interviewees noted that partner country HISs often did not capture the information that PEPFAR mission teams needed to report to OGAC (636-9-USG; 166-1-USG; 166-12-USG; 166-10-USNGO; 272-27-USG; 461-15-USG; 461-20-PCPS; 934-21-USG; 116-12-PCNGO).
Routine monitoring of HIV/AIDS service delivery at the facility or patient level requires more indicators than are collected by many HISs, a factor that contributed to the development of M&E systems for HIV/AIDS that operate outside broader HISs (166-10-USNGO; 461-11-PCGOV; 934-46-PCGOV). Among the challenges with these parallel systems were that some stakeholders bypassed the national system (461-11-PCGOV; 934-21-USG) and reported only to funders (461-11-PCGOV) and that some partner countries had no way to aggregate the data collected through parallel systems (331-5-ML; 331-10-PCGOV; 331-24-PCGOV).
HIV/AIDS M&E Systems
Although M&E systems represent a subset of an HIS, efforts to strengthen M&E systems can contribute to strengthening the overall HIS. In many partner countries, PEPFAR has built capacity at the national level by supporting national M&E plans and frameworks (636-18-ONGO; 331-1-USG; 461-16-USG; 934-46-PCGOV) and providing technical support (636-18-ONGO; 331-1-USG; 166-12-USG; 934-21-USG). PEPFAR has also supported sub-national levels (636-18-ONGO; 240-12-USG; 461-15-USG) and organization and facility levels (636-18-ONGO; 196-11-USNGO; 116-12-PCNGO; 166-12-USG) to strengthen national M&E systems. PEPFAR has supported the development of M&E guidelines and processes (240-23-PCGOV; 934-21-PCGOV; 196-11-USNGO), data collection and management tools (934-21-USG; 934-46-PCGOV; 196-11-USNGO; 331-23-USNGO; 331-34-USNGO; 935-ES; 587-14-PCGOV) (PEPFAR/Botswana, 2007), data quality management (196-11-USNGO; 636-18-ONGO), and national reporting systems (196-11-USNGO). Finally, PEPFAR partners have also supported M&E training for health care workers (e.g., doctors, nurses, etc.) as well as data clerks (636-18-ONGO) and government employees (331-1-USG; 331-15-USG). See Chapter 11 on knowledge management for additional information on PEPFAR’s activities related to HIV/AIDS M&E systems.
Health Management Information System
An HMIS provides information to assist in the management and planning of health programs rather than the delivery of services (WHO, 2004). Although parallel information systems for the collecting and reporting of PEPFAR indicators were created in many countries, PEPFAR has also supported the development and implementation of HMISs (396-19/20-USG; 196-ES). In the developing world, most HMISs collect only cross-sectional patient data with few systems collecting longitudinal, patient-based data, an imbalance that can affect the strategic planning and management of health programs. The lack of more comprehensive data reported in a timely manner across sectors, among levels, and from many providers to the central depository in the countries will also affect program planning and management, including efforts for quality assurance or improvement. Many interviewees felt that strengthening national HMISs would improve the availability, quality, and use of data for decision making (934-38-PCACA; 934-46-PCGOV; 240-8-USG; 272-22-USG).
Formats of HMISs in PEPFAR partner countries vary; some are paper-based (116-12-PCNGO; 934-21-USG), some are electronic, and some are a mix (331-15-USG). Transitioning to an electronic HMIS requires significant financial resources (934-21-USG). In several countries, PEPFAR has supported partner country governments to plan for (PEPFAR/Botswana, 2009) and to roll out or implement national HMISs (636-1-USG; 116-16-PCGOV; 934-21-PCGOV; 196-11-USNGO; 587-9-USG; 331-3-USG; 240-8-USACA; 240-19-USACA; 240-20-ML) (PEPFAR/Botswana, 2007, 2009).
PEPFAR has also supported capacity building for increased or improved workforce capacity for HMISs, such as through ‘pre-service training at regional health colleges for health information technicians’ (240-12-USG) or by support for in-service training when new systems were initiated (636-1-USG).
As part of strengthening partner country systems, PEPFAR has supported the development of Laboratory Information Management Systems (LIMSs) and in many cases has introduced electronic LIMSs. PEPFAR has generally supported an incremental approach within partner countries—introducing LIMSs in a few larger laboratories and then gradually expanding to additional laboratories (396-22-USG; 240-16-USG; 636-14-USNGO). For example, in one partner country, PEPFAR first supported an integrated LIMS at four labs and then expanded to an additional five laboratories the following year (636-14-USNGO). In another partner country, PEPFAR focused on developing a LIMS for hospital laboratories and HIV testing laboratories, working in 10 sites (396-22-USG). In a country with the greatest level of integration described, the LIMS was automated and interfaced to standard software, and all databases were networked and fed into a central repository of data, which served not only as backup but also as a means for data to be available at the national level so that the laboratory services could make informed decisions about procurement and larger policy decisions (636-14-USNGO). To build capacity and manage the LIMSs, PEPFAR has provided training for cadres of workers as well as salary support for key LIMS staff (636-14-USNGO; 166-11-USG). For additional information about PEPFAR’s efforts related to laboratory strengthening, see the Laboratory subsection later in this chapter in the Medical Products and Technologies section.
Integration of Information Systems
Partner countries often have multiple, separate health information sources. Some interviewees expressed a desire to integrate information, monitoring and evaluation, and surveillance systems for HIV and other diseases (636-18-ONGO; 934-46-PCGOV; 331-24-PCGOV; 240-ES; 116-ES; 116-9-PCNGO; 116-16-PCGOV; 935-ES; 196-8-ML), and PEPFAR has provided considerable support to streamline and integrate multiple information and reporting systems (196-ES; 196-11-USNGO; 240-2-USG; 934-10-PCGOV). In one partner country PEPFAR supported combining parallel district HISs into one national HMIS in order to make data available centrally (331-24-PCGOV). One country’s MOH choose to use the HIV information system as a model for bringing together disparate components of health information (196-11-USNGO). However, interviewees did not always see the integration of information systems as the best approach (116-16-PCGOV; 196-8-ML). As described by one interviewee, ‘integration would be difficult—there is very different data collected for communicable diseases, like HIV’ (196-8-ML).
Individual Capacity Building
In some countries, strategic information (SI) skills were new competencies introduced in the country’s workforce (587-10-USG; 461-16-USG) or were nascent efforts that, even with very little training, made the workers marketable and afforded them different job opportunities (272-32/35-PCNGO; 166-12-USG; 587-7-PCGOV). PEPFAR II has supported training for HISs, M&E, and surveillance at all levels: the national government (636-1-USG), sub-national levels of the government (396-9-PCGOV; 116-7-USG; 166-7-PCGOV; 331-15-USG) (PEPFAR/Botswana, 2009), and facilities and partners (240-17-PCGOV; 240-8-USG; 396-29-PCGOV; 196-21-PCGOV; 636-15-PCNGO). However, interviewees from at least one country mentioned that, because of the attrition of staff trained in M&E (M&E skills make them more marketable), they were continuously training (934-21-PCGOV; 934-46-PCGOV). PEPFAR has also provided salary support for key health information positions within national (166-30-ONGO) (PEPFAR/Botswana, 2009) and sub-national institutions (166-15-USACA).
In addition, PEPFAR has contributed to strengthening health workforce capacity for M&E, HMIS, surveillance, and the use of health information by supporting partner country education programs and institutions to integrate SI skills into pre-service curricula at local universities and colleges (331-1-USG; 331-15-USG; 331-34-USNGO; 240-12-USG) (PEPFAR/Botswana, 2007). PEPFAR has also funded efforts to produce health workers with SI skills through Masters of Public Health programs (934-38-PCACA; 396-55-USG), programs for health information technicians (240-12-USG), certificate programs for HMIS (PEPFAR/Ethiopia, 2007), and CDC’s Field Epidemiology and Laboratory Training Programs (see the Workforce building block in this chapter for more information on these programs).
Challenges
Despite PEPFAR’s efforts to build national and individual capacity to plan for, collect, manage, and use HIV/AIDS and other health information, interviewees across countries reported challenges that their organizations faced in their efforts to strengthen HISs. These included inadequate financial resources for the HISs (166-5-USG); a lack of national-level capacity (196-8-ML; 166-1-USG; 461-11-PCGOV; 461-16-USG; 934-46-PCGOV; 396-19-USG; 396-20-USG), which was exacerbated by the attrition of skilled and trained employees (934-21-PCGOV; 331-5-ML; 196-1-USG; 396-ES; 272-ES); and the lack of national-level commitment (587-25-ML; 636-ES).
Issues related to human resources were also described as challenges to improving data collection and management, including inadequate numbers of trained professionals (396-9-PCGOV; 934-46-PCGOV; 587-ES; 461-ES), a lack of capacity (in skills and knowledge) (461-11-PCGOV; 587-9-USG; 331-34-USNGO), a lack of incentives (331-6-CCM; 396-9-PCGOV), and low salaries (331-23-US NGO; 396-9-PCGOV). Interviewees also
described various infrastructure-related challenges to collecting and managing information, including issues with roads and transportation (636-1-USG), electricity (587-9-USG), Internet access (934-21-PCGOV; 116-12-PCNGO), and technology (both hardware and software) (934-5-USG; 934-21-PCGOV; 587-9-USG). In some countries, challenges with the timely reporting of data from sub-national to national levels impaired the use of data for programs and policies (934-46-PCGOV; 166-12-USG).
Another challenge identified by interviewees, which was more specific to PEPFAR’s approach, was that strengthening HIV/AIDS information systems did not always result in strengthening the overall HIS system (166-12-USG; 461-11-PCGOV; 396-2-USG; 934-25-USPS).
“I think also PEPFAR remains constrained by the fact that, and this is changing again, to some extent, but it’s still HIV. So still, when you talk about health system strengthening, it’s not health system strengthening. And I think that’s more obvious here because HIV was pulled out of the health system, in a way. But it’s also true in other countries where people have been maneuvered into, I think, more basically HIV systems that overlap with broader health systems. [. . .] [Y]ou can strengthen those, but they’re still at the cost of the other systems unless you really look at staffing across the system, look at skill-based [. . .] allocation of staffing.” (396-45-USNGO)
However, an interviewee in another country offered a contrasting view, saying that, despite the fact that PEPFAR has an HIV mandate, most of the activities of that partner country’s mission team SI technical working group were broad, health sector strengthening projects (166-12-USG).
PEPFAR Achievements
Despite contributing to parallel systems for collecting health information, PEPFAR has at the same time supported capacity building for national information systems across countries and improved the availability of quality information regarding HIV/AIDS (Samb et al., 2009). In several instances, interviewees credited PEPFAR support for improved HMISs (240-2-USG; 240-3-PCGOV; 240-7-PCGOV; 240-20-ML; 461-4-USG; 587-9-USG). A partner country government official and an USG interviewee in the same country agreed on this issue:
‘Before PEPFAR, HR capacity was very low, HMIS was very poor, and the laboratory and health system in general was very poor, very weak. PEPFAR has contributed to strengthening the health system.’ (240-7-PCGOV)
“Before PEPFAR, HMIS was a challenge—used to be very weak. PEPFAR has helped to strengthen, pilot, and implement a system, which has now been rolled out to almost all facilities. PEPFAR has streamlined all information systems. The government is leading this effort, but PEPFAR has supported the government in leadership at every level.” (240-2-USG)
Over time there has been a shift in PEPFAR’s dialogue concerning the alignment of PEPFAR’s M&E system with national HISs. In some countries, interviewees described increased efforts to align PEPFAR monitoring indicators with national HISs (240-20-ML; 331-18-USNGO; 636-9-USACA; 636-18-ONGO; 166-12-USG; 461-18-USG). Interviewees said that the capacity building for and the strengthening of national information systems increased during PEPFAR II (166-22-USPS). As one interviewee said, ‘PEPFAR has supported a lot of information systems and better management of data but, “there is still a long way to go”’ (587-9-USG).
Conclusion: Despite initial PEPFAR-specific systems for program monitoring data, PEPFAR has worked with partner country governments to integrate and strengthen health information systems, including work that has strengthened partner country LIMS. However, ongoing support to strengthen partner country health information systems—and better alignment and integration with those systems—is needed to enhance timely data availability and quality for use in strategic program planning, resource allocation, and commodities procurement.
MEDICAL PRODUCTS AND TECHNOLOGIES
Commodities and Supply Chain Management
Background and Context
Consistent access to diagnostic reagents, medicines, vaccines, and technologies requires a system of links that run from unprocessed raw materials to the delivery of the finished product; this system is commonly referred to as a supply chain (CSCMP, 2010). Supply chain management includes operational components such as quantification, procurement, inventory management, distribution, and data collection and reporting as well as “the coordination and collaboration of staff, levels, and functions” (USAID DELIVER Project, 2011, p. 1). Effective supply chains require “an understanding of patient needs, captured in forecasts and supply plans, which
then guide procurement and supply to satisfy those needs” (SCMS, 2012, p. 4).
However, in many settings in low-income countries considerable challenges remain in achieving and maintaining well-functioning supply chain management, from the macro level (infrastructure issues, including laws related to the importation of commodities, regulatory policies and approaches for pharmaceuticals, and local drug manufacturing capacity, that could play a contributory role to sustainability) to the micro level (poor tracking systems within clinical care systems). It is critically important to manage the supply chain effectively and efficiently, particularly with a lifelong infection like HIV, to ensure that there are no stock-outs of essential medications, such as antiretrovirals and other drugs to prevent or treat opportunistic infections. In order to avoid such stock-outs and emergency shipments, accurate planning and forecasting must be established; this requires coordination, transparent financing, and procurement plans for the necessary goods (Lalvani et al., 2010). Practical and efficient warehouses are also vital; they must be constructed in a methodical way to hold the stocks of quality goods (e.g., HIV test kits, ARVs, etc.) at the appropriate temperature and settings (USAID DELIVER Project, 2011). Adequate in-country technical capacity for freight and logistics is necessary to help maintain an organized schedule of deliveries, and coordination between supply-chain managers and program-service managers can ensure that the necessary commodities are available on a regular and consistent basis (OGAC, 2011c). Accountability is also necessary to protect against the misuse of products and ensure product reliability. Smooth transitions from one link in the supply chain management system to the next help to ensure proper product delivery and use, creating more successful HIV/AIDS programs in the areas that need it the most.
PEPFAR Inputs
Guidance The first PEPFAR Five-Year Strategy articulated the necessity of effective supply chain management in order to provide diagnostic reagents, drugs, materials, and equipment for HIV/AIDS programs. To support effective supply chain management, the strategy committed to training supply chain management personnel and strengthening health logistics systems. The strategy also pledged to coordinate supply chain management systems to “reduce and eliminate diversion, counterfeiting, and the sale of HIV/AIDS products and supplies on the black market” (OGAC, 2004, pp. 12–13).
The FY 2009 COP guidance identified improving commodity distribution and control as a key area for building capacity in both the governmental and nongovernmental sectors (OGAC, 2008a). The FY 2012 COP guidance identified the following activities to address multiple components
of the supply chain in order to ensure the continued availability of key health commodities (OGAC, 2011b, p. 33):
• “Support/encourage the development and implementation of a national strategic plan for supply chain.
• Contribute to an adequately trained and well-performing supply chain workforce, including capacity building activities and transitioning roles and responsibilities to partner government counterparts.
• Coordinate with other donors, and leverage other donor inputs, for supply chain system strengthening activities.
• Improve the availability and use of information within the supply chain system for decision making.”
PEPFAR Activities
In its early years PEPFAR procurement of ARVs made possible the rapid scale-up of HIV treatment. To address challenges with partner country supply chain infrastructure and systems, in 2005, OGAC established the Supply Chain Management System (SCMS) (SCMS, 2012). SCMS is managed by the Partnership for Supply Chain Management, a nonprofit organization that has two managing partners (JSI Research and Training Institute, Inc., and Management Sciences for Health) and 13 member organizations from the nonprofit, commercial private, and academic sectors (PFSCM, 2010) (see Box 9-5). SCMS works in three areas (PFSCM, 2010):
1. Procurement and distribution of essential medicines and supplies
2. Technical assistance to strengthen existing supply chains
3. Collaboration with in-country and global partners to coordinate efforts in these areas
Capacity building for supply chain management To build partner country capacity for supply chain management, SCMS provides technical assistance for the strengthening of local supply chains (Jamieson, 2011). SCMS is working with partner country educational institutions in seven countries to “incorporate supply chain management modules into health worker training curricula” (SCMS, 2012, p. 6).
Interviewees described various PEPFAR-supported capacity building efforts at many levels of the supply chain. At the national level, PEPFAR partners have provided support for the development and implementation of policies related to pharmaceuticals (such as national medicines policies and essential medicines lists) (636-20-PCGOV; 542-5-USPS; 196-11-USNGO) and for strengthening regulatory authority in order to ensure drug quality (542-21-USNGO; 240-
BOX 9-5
SCMS Member Organizations
• 3i Infotech
• Booz Allen Hamilton
• Crown Agents
• i+solutions
• JSI Research & Training Institute, Inc.
• Management Sciences for Health
• The Manoff Group
• MAP International
• North-West University, South Africa
• Northrop Grumman
• PHD
• UPS Supply Chain Solutions
• Voxiva
SOURCE: PFSCM, 2010.
12-USG). PEPFAR is supporting human resource capacity at all levels of the supply chain system, from providing direct salary support (116-2-USG; 934-5-USG; 934-25-USPS) and technical assistance (636-20-PCGOV; 331-38-USPS; 116-2-USG; 116-4-USG; 166-3-USG; 461-18-USG; 587-6-CCM; 587-10-USG; 935-9-USG; 542-21-USNGO) for key national-level positions (e.g., in the MOH) to training, supportive supervision, and mentorship for health care workers (636-20-PCGOV; 240-12-USNGO; 461-13-USACA; 331-30-USPS), including study tours abroad to learn skills for proper supply chain management (587-11-PCGOV). PEPFAR partners have contributed to strengthened supply chains through the development of standard operating procedures and the dissemination of best practices (240-29-USNGO; 542-21-USNGO; 636-20-PCGOV; 166-32-USPS; 331-38-USPS) (Botswana Ministry of Health, 2012). In several countries, PEPFAR is supporting national or central medical stores to improve warehousing and storage capacity (461-13-USACA; 461-15-USG; 166-6-USG; 166-32-USPS; 935-9-USG). PEPFAR has also supported innovative distribution systems (934-25-USPS) and purchased vehicles to expand distribution capacity (240-5-PCGOV; 934-25-USPS).
PEPFAR partners have supported data collection and data management activities to monitor the drug supply and to provide information for quantification (166-6-USG), including electronic data systems for capturing and reporting data (636-20-PCGOV; 542-21-USNGO; 331-38-USPS; 166-32-USPS). PEPFAR support for the information needed for forecasting and procurement is also discussed in the information building block in this chapter.
Procurement In most countries, PEPFAR funding supports the procurement of ARVs in coordination with other donors and domestic resources, but the type of arrangement varies. In a few countries visited by the committee, the Global Fund procures first-line ARVs, and PEPFAR procures second-line ARVs or pediatric formulas, or both (240-5-PCGOV; 587-13-USG; 587-22-USG; 461-13-USACA). In most countries, the majority of ARVs are procured with Global Fund (116-11-PCGOV; 331-16-USG; 935-4-PCGOV) or PEPFAR (461-4-USG; 166-13-PCGOV; 396-4-PCGOV; 396-7-PCGOV) funding, but in some countries, the government has taken partial to complete responsibility for funding ARVs (636-4-PCGOV; 542-2-USG; 542-9-PCGOV; 272-1-USG; 196-10-PCGOV; 934-25-USPS).
In 2011, 71 percent of ARVs funded by PEPFAR were delivered by SCMS (SCMS, 2012). Interviewees from many countries mentioned that SCMS was responsible for the procurement of ARVs (240-5-PCGOV; 587-6-CCM; 166-6-USG; 166-8-USG; 461-13-USACA; 461-17-PCNGO; 396-4-PCGOV; 396-41-PCGOV; 934-12-CCM; 934-25-USPS). In several countries, PEPFAR was also supporting procurement of other commodities, such as diagnostic reagents, condoms (587-8-PCGOV; 196-6-USG; 934-10-PCGOV; 934-25-USPS; 116-19-PCACA; 166-11-USG), and drugs to treat tuberculosis (TB) and other opportunistic infections (166-6-USG; 396-7-PCGOV; 396-41-PCGOV; 934-12-CCM). In some countries, partner country governments have been unable to demonstrate adequate forecasting or quantification for the procurement of drugs (and other supply chain issues) (331-ES; 587-ES; 116-ES; 934-ES; 935-ES), so the Global Fund has refused to release funding (636-3-USG; NCV-11-USG). Interviewees from some countries described PEPFAR as a “safety net” for the government and gave examples of cases in which PEPFAR has provided buffer stocks or emergency procurement to address shortages or stock-outs (331-43-USG; 587-1-USG; 587-22-USG; 166-31-USG; 272-1-USG; 272-5-PCGOV; 272-22-USG; 934-5-USG; 116-2-USG; 935-14-USG). Shortages or stock-outs may be caused by partner country government financial crises (934-25-USPS); a failure to project needs, plan, or procure adequately (331-43-USG; 587-22-USG); temporary situations in which demand exceeds supply (272-1-USG); delays in disbursements from the Global Fund or other donors (196-6-USG; 116-2-USG; 934-5-USG; 587-22-USG); or transitions in support for procurement from one donor to another (934-5-USG). In some countries, PEPFAR-supported programs such as SCMS have set up procurement and supply chain systems outside of the partner country systems (166-22-USPS). In one country, interviewees noted that the supply chain for antiretroviral therapy must be separate and parallel to the national system in order for ARVs to reach treatment sites (116-18-PCNGO).
Partner country challenges Across partner countries, interviewees described common challenges with procurement and supply chain management (196-7-PCNGO; 196-26-USG; 331-43-USG; 636-9-USACA; 116-2-USG; 116-16-PCGOV; 166-6-USG; 166-31-USG; 461-17-PCNGO; 461-25-ML; 542-21-USNGO). In several countries, insufficient quantification and forecasting, often due to the unavailability of necessary data, had threatened the regular availability of drugs and commodities and resulted
in stock-outs (116-16-PCGOV; 331-12-USG; 331-28-PCGOV; 331-38-USPS; 272-20-PCNGO; 461-14-USG; 542-21-USNGO). Stakeholders in nearly all countries struggled with stock-outs, which constrained the delivery of treatment for HIV (934-17-PCGOV; 461-7-PCNGO; 461-8-PCGOV; 116-12-PCNGO; 587-1-USG; 587-22-USG; 587-18-PCGOV; 331-43-USG), TB (542-21-USNGO), and opportunistic infections (240-25-PCGOV; 587-18-PCGOV; 196-17-PCGOV; 461-17-PCNGO; 116-12-PCNGO; 166-34-PCGOV), and which reduced access to HIV testing (116-12-PCNGO; 934-17-PCGOV; 461-15-USG; 166-5-USG; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS; 166-32-USPS; 240-33-USG). Challenges with procurement at the national level, including corruption (542-2-USG; 542-3-USG; 542-11-PCNGO), also threatened effective supply chain management (542-21-USNGO; 331-30-USPS). In some countries, this was attributed to government regulations or processes that caused delays in timely procurement (331-17-USG; 331-28-PCGOV; 542-21-USNGO; 240-3-USG; 240-8-USG; 240-33-USG; 240-21-PCGOV). A weak capacity for financial management was mentioned as a barrier for every step of the supply chain (331-2-USG; 166-6-USG; 166-31-USG; 116-2-USG). Commodity storage (240-24-USG; 331-38-USPS; 116-2-USG), distribution (331-6-CCM; 331-38-USPS; 166-6-USG; 166-31-USG; 542-21-USNGO), and logistics (e.g., tracking inventory) (331-17-USG; 166-11-USG; 166-22-USPS) were also mentioned as challenges. Although national supply chains were often inadequate or overwhelmed, parallel supply chains serving different donors or disease programs were also described as inefficient and as leading to coordination challenges among partners and donors (396-18-USG; 396-21-USG; 396-57-USG; 636-9-USACA; 331-12-USG; 331-38-USPS; 542-21-USNGO).
PEPFAR Achievements
Across countries, PEPFAR has improved supply chain management capacity for forecasting, procurement, and distribution (240-2-USG; 240-3-USG; 240-5-PCGOV; 240-12-USG; 331-38-USPS; 587-10-USG; 935-9-USG; 935-14-USG; 272-20-PCNGO; 934-5-USG; 934-25-USPS). PEPFAR’s efforts to strengthen partner country supply chains have also had a positive impact on Global Fund programs (240-12-USG; 331-38-USPS; 587-6-CCM). In addition to increasing access to HIV-related commodities (e.g., ARVs and condoms) (272-25-USG; 116-9-PCNGO; 240-2-USG), PEPFAR has also contributed to the increased availability of medicines and commodities for other health issues (166-4-USG). When partner countries have faced stock-outs due to supply chain constraints or delays in disbursements from the Global Fund and other donors, PEPFAR has often provided buffer stocks of medicines and commodities on an emergency basis to prevent the interruption of treatment and to ensure the continuity of care (935-8-PCGOV; 935-17-USG; 587-1-USG; 461-10-PCNGO; 116-9-PCNGO; 240-7-PCGOV; 331-43-USG). PEPFAR partners have been actively involved in convincing ministries of health to use the most effective first-line ARV regimens (196-11-USPS; 272-20-PCNGO).
Conclusion: PEPFAR has improved the capacity of partner country governments to quantify, forecast, procure, store and warehouse,
distribute, and track commodities, but challenges to assuring consistent and reliable supply chain functioning remain in many countries. These challenges are a common issue across countries and are not PEPFAR-specific. Reliable supply chains will be critical for sustainable and cost-efficient HIV/AIDS responses and for avoiding disruptions to the clinical care and treatment of people living with HIV/AIDS.
Laboratory Infrastructure
Functioning laboratories, with the capacity to run screening, diagnostic, and clinical laboratory tests, are fundamental to the monitoring and management of patients with HIV/AIDS and other diseases (Gershy-Damet et al., 2010). In PEPFAR’s early years, access to and the quality of laboratory services was a major challenge in partner countries (Cohen, 2007; Sturchio and Cohen, 2012). A lack of prioritization and leadership (e.g., no national policies or strategic plans), inadequate workforce capacities, and dilapidated infrastructures affected laboratory systems, which tended to have limited available resources. In many countries, limited laboratory capacity was “a major barrier” or “rate-limiting step” for the scale-up of HIV/AIDS and other health services that was necessary to meet the Millennium Development Goals (Birx et al., 2009, p. 849; WHO, 2008, p. 1).
Within the past 5 years, global stakeholders have declared their commitment to strengthening laboratory systems, particularly in Africa. In January 2008, 33 countries and 3 multilateral organizations signed the Maputo Declaration on Strengthening of Laboratory Systems (WHO, 2008). The Maputo Declaration called on national governments to prioritize support to laboratory systems through the development of national laboratory policies, national laboratory strategic plans, and departments of laboratory systems within the various countries’ ministries of health in order to address the challenges that limit the scale-up of services for tuberculosis, malaria, and HIV diagnosis and care. The Maputo Declaration also called on donors and partners to support these national efforts and to coordinate and collaborate with each other to support the strengthening of laboratory systems, including efforts to build public–private partnerships (WHO, 2008).
PEPFAR Inputs
Guidance During PEPFAR I, support for laboratory services focused on the provision of those basic services that were needed for HIV diagnosis and care (Justman et al., 2009). PEPFAR’s first Five-Year Strategy committed to improving laboratory capacity for HIV testing and treatment monitoring as well as to training laboratory technicians in order to quickly
expand HIV/AIDS services (OGAC, 2004). COP guidance through 2009 emphasized a programmatic focus on increasing the availability and quality of laboratory services at various levels of the health system in partner countries through the purchase of equipment and commodities, the provision of quality assurance, staff training, and technical assistance (OGAC, 2008a). During PEPFAR II, the focus shifted to the strengthening of broader laboratory systems and included such activities as “quality management systems, equipment maintenance, training, and infrastructure” (Justman et al., 2009, p. S30). PEPFAR country programs were directed to focus on increasing quality assurance of laboratory services, on efforts to achieve accreditation, on training for laboratory technicians and management, and on the transition of laboratory programs to in-country partners (OGAC, 2011b). The second Five-Year Strategy recognized the potential to leverage increased laboratory capacity for other diseases, such as malaria and TB, and highlighted support for laboratories as one of PEPFAR’s contributions to the U.S. Global Health Initiative (OGAC, 2009f).
PEPFAR indicators for laboratory infrastructure During PEPFAR I, laboratory-related indicators were limited to the following output measures:
• the number of laboratories with capacity to perform HIV tests and CD4 tests and/or lymphocyte tests,
• the number of individuals trained in laboratory-related activities, and
• the number of tests performed at USG-supported laboratories in the areas of HIV testing, TB diagnostics, syphilis testing, and HIV disease monitoring (OGAC, 2007b).
In 2009, OGAC released the Next Generation Indicator Guidance, which included two slightly modified indicators for laboratory infrastructure to reflect this new focus:
• the number of testing facilities (laboratories) with capacity to perform clinical laboratory tests and
• the percent of testing facilities (laboratories) that are accredited according to national or international standards (OGAC, 2009c).
PEPFAR’s reauthorization legislation included a new target for the training of 140,000 health care workers, including laboratory experts (Birx et al., 2009).
Funding OGAC has defined a budget code for laboratory strengthening activities (see Box 9-1). From FY 2005 to FY 2011, PEPFAR provided more than $1 billion for laboratory strengthening (OGAC, 2005a, 2006b, 2007c, 2008b, 2010b, 2011d,e).
PEPFAR Activities
PEPFAR has supported a wide range of laboratory-strengthening activities that have been described in the published literature and that were reported by country visit interviewees. In several countries PEPFAR supported the development of national laboratory strategic plans (240-21-PCGOV; 331-3-USG; 116-19-PCACA; 396-22-USG) and also guidelines and standard operating procedures (196-10-PCGOV; 116-19-PCACA; 272-13-USG). PEPFAR support for laboratory strengthening has often been aligned with partner country strategic plans, and in many countries, PEPFAR partners have assisted countries with the development of laboratory strategic plans (396-22-USG; 396-55-USG; 240-21-PCGOV; 331-17-USG).
Interviewees confirmed PEPFAR support for the construction and refurbishment of partner country laboratories at multiple levels, including national reference labs (587-7-PCGOV; 587-13-USG; 240-2-USG), provincial or regional labs (240-12-USG; 240-21-PCGOV; 166-11-USG; 934-5-USG), and local labs (461-3-USG). For example, in Ethiopia, PEPFAR has supported the National Laboratory Strategic Plan of the Ethiopian Health and Nutrition Research Institute by renovating and equipping the national reference laboratory, four regional hospitals, and six regional laboratories (Justman et al., 2009). In many countries, PEPFAR has provided lab equipment (240-2-USG; 934-24-PCGOV; 934-28-PCNGO; 396-22-USG; 396-41-PCGOV; 116-19-PCACA; 331-16-USG) and supplies such as reagents, test kits, and consumables (166-11-USG; 240-12-USG; 461-8-PCGOV; 934-2-USG; 934-21-USG; 934-25-USPS; 934-28-PCNGO).
PEPFAR funds several partners to provide TA for laboratory systems across partner countries (ASCP, 2012; BD, 2012; Diallo, 2011). Several interviewees offered descriptions of PEPFAR support for laboratory information systems (331-15-USG; 166-11-USG; 396-22-USG), including numerous reports of PEPFAR efforts to establish quality-management, quality-assurance, and quality-improvement programs (240-19-USACA; 240-21-PCGOV; 166-4-USG; 272-12-USNGO; 461-19-USG; 396-2-USG; 396-22-USG; 934-5-USG). Interviewees in several countries reported participating in PEPFAR’s Strengthening Laboratory Management Towards Accreditation Initiative as well as receiving other PEPFAR support for lab accreditation (116-19-PCACA; 166-11-USG; 934-28-PCNGO; 331-15-USG; 461-18-USG; 396-2-USG; 396-22-USG) (see Box 9-6).
PEPFAR has supported human capacity development for laboratory skills through south-to-south workshops in Nigeria, and the U.S. Centers for Disease Control and Prevention opened the African Center for Integrated Laboratory Training in South Africa (Justman et al., 2009). Interviewees from nearly all of the countries that the committee visited de-
BOX 9-6
PEPFAR’S Laboratory Systems
Strengthening Initiatives Over Time
In 2007, PEPFAR launched a laboratory systems strengthening initiative to be implemented by a public–private partnership with Becton, Dickinson and Company, the Centers for Disease Control and Prevention (CDC), national ministries of health, and national reference laboratories (OGAC, 2007a; Sturchio and Cohen, 2012). This approach to strengthening laboratories includes technical assistance (training and mentorship), process improvement related to quality management and specimen referrals, curriculum and leadership development, and strategic facilitation of planning meetings and project management (Thompson, 2011). As of July 2012, the initiative had been launched in Uganda, Ethiopia, Mozambique, and South Africa, with plans to expand to more countries (BD, 2012).
In 2009, the CDC launched the Strengthening Laboratory Management Towards Accreditation program, a series of workshops designed to improve laboratory management through a task-based framework and mentoring kit that provides information on the day-to-day tasks necessary for ensuring quality services and achieving accreditation (Yao et al.,2010).
In 2009, PEPFAR partnered with the World Health Organization Regional Office for Africa (WHO/AFRO) to launch a five-step framework to help African laboratories achieve accreditation through a star ranking system (Gershy-Damet et al., 2010; Wenner, 2009). The accreditation process involves assessing laboratories and verifying the implementation of laboratory standards to ensure that services are “accurate, traceable, and reproducible” (Gershy-Damet et al., 2010, p. 394; Palen et al., 2012; WHO, 2007a).
scribed how PEPFAR has supported the strengthening of human resources for laboratory systems, including support for pre-service training (116-7-USG; 116-19-PCACA; 166-6-USG; 166-11-USG; 272-13-USG; 461-18-USG) and in-service training (240-2-USG; 240-15-USG; 240-19-USACA; 331-15-USG; 331-16-USG; 331-28-PCGOV; 116-19-PCACA; 166-11-USG; 272-13-USG; 461-13-USACA; 396-18-USG; 934-28-PCNGO).
Although data were available for only some years of the program, PEPFAR indicators reported to OGAC clearly reflect an increased emphasis on laboratory strengthening. During PEPFAR I, the number of individuals trained in the provision of lab-related activities increased from 3,131 in FY 2004 to 60,037 in FY 2009. During the same time period, the number of testing facilities in partner countries with the capacity to perform clinical lab tests increased from 282 to 7,211 (see Figure 9-6).
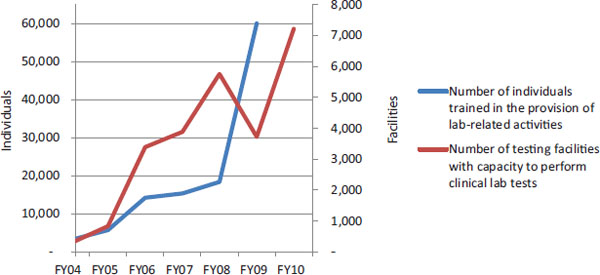
FIGURE 9-6 Select indicators related to PEPFAR’s laboratory activities.
NOTES: For FY 2004 and FY 2005, data were reported by the 15 focus countries. For FY 2006–FY 2010, the number of countries that reported data varied by year from 26 to all 31 of the countries that are the focus of this evaluation (see Chapter 2). For “Number of testing facilities [. . .],” FY 2004 to FY 2009 numbers correspond to PEPFAR I indicator 12.1, which has a slightly different definition: Number of laboratories with capacity to perform (1) HIV tests and (2) CD4 tests and/or lymphocyte tests (OGAC, 2007b). This indicator only captures USG-supported testing facilities and does not represent national capacity.
SOURCE: Program monitoring indicators provided by OGAC.
Despite PEPFAR’s achievements with laboratory strengthening in partner countries, challenges remain. In many countries capacity is still limited to large labs or urban areas, and the process of transporting samples or specimens to labs creates delays between testing and getting results (636-17-PCGOV; 240-24-USG; 272-13-USG; 461-10-PCNGO). Some interviewees described challenges in monitoring patient CD4 counts or identifying ARV resistance caused by limited capacity (166-11-USG; 166-15-USACA; 461-10-PCNGO; 934-15-PCGOV; 636-17-PCGOV). Across countries, stakeholders expressed the need for more (and newer) laboratory equipment (461-10-PCNGO; 116-12-PCNGO; 116-19-PCACA; 166-15-USACA; 934-34-USNGO) yet also cited equipment maintenance as a challenge (240-8-USG; 240-21-PCGOV; 331-17-USG; 934-17-PCGOV; 934-28-PCNGO; 396-22-USG). Laboratory networks are also affected by procurement and supply chain challenges (described in the previous section), which can result in stock-outs of reagents, test kits, and other laboratory commodities (587-18-PCGOV; 196-10-PCGOV; 116-12-PCNGO; 116-19-PCACA; 166-15-USACA; 934-28-PCNGO). In many countries, shortages of appropriately trained laboratory personnel (e.g., lab technicians) are the greatest barriers to expanding access to laboratory services (272-13-USG; 461-13-USACA; 461-18-USG; 934-2-USG; 166-11-USG). Ongoing challenges include adequate pre-service training, lack of management capacity, and site supervision, which are critical for quality services (396-22-USG; 166-12-USG) (Justman et al., 2009).
PEPFAR Achievements
Although challenges remain, the improvement of laboratories under PEPFAR support and guidance has been a signature achievement of the program. There are well-documented examples of how PEPFAR has helped transform labs, such as in the Caribbean region, where accreditation was attained and quality assurance systems were built, and in Nigeria, where lab deficiencies (e.g., failure to monitor ambient temperatures or failure to post HIV rapid-testing algorithms) were reduced from 13 percent to 2 percent (Abimiku et al., 2010; Alemnji et al., 2012). During the committee’s country visits, interviewees emphasized this theme with near universality.
PEPFAR support for labs has increased partner country capacity to provide laboratory services such as HIV testing (331-17-USG; 587-2-USG; 240-2-USG), viral load testing (934-5-USG), and HIV resistance testing (240-2-USG). Interviewees from several countries observed that PEPFAR support for laboratories has had spillover effects for their entire health systems (240-2-USG; 240-19-USACA; 331-17-USG; 331-28-PCGOV; 116-28-USACA; 166-11-USG; 461-18-USG; 396-1-USG; 396-60-USG). They reported increasing capacity for testing of the blood supply (166-11-USG) and for the diagnosis and treatment of diseases other than HIV (331-17-USG; 166-4-USG; 166-11-USG), such as tuberculosis (240-2-USG; 331-28-PCGOV), malaria (240-2-USG), and influenza (240-2-USG). In many countries, PEPFAR was the only donor supporting laboratory systems (240-21-PCGOV; 116-4-USG; 166-11-USG).
PEPFAR-supported technical assistance for laboratory information systems has improved the management and sharing of information (166-11-USG). Interviewees reported that PEPFAR efforts have resulted in improved specimen transport and decreased turnaround times for getting lab results (240-24-USG; 331-38-USPS; 272-13-USG; 934-2-USG; 116-19-PCACA). PEPFAR-supported laboratories in some partner countries have achieved accreditation (240-33-USG; 396-22-USG).
Conclusion: PEPFAR’s laboratory efforts have had a fundamental and substantial impact on laboratory capacity in countries. This laboratory infrastructure and capacity has been, and can continue to be, leveraged to improve the functioning of countries’ entire health systems.
Background and Context
The association between an available and competent health workforce and key indicators of morbidity and mortality has long been noted both domestically and internationally (Chen et al., 2004; Speybroeck et al., 2006). WHO has set a critical threshold of 2.3 doctors, nurses, and midwives per
1,000 population as essential for achieving 80 percent coverage of maternal and child health services within a country. If all countries were able to achieve WHO’s minimal target goal of 2.3 health professionals per 1,000 population, not only would it allow progress toward HIV/AIDS and other global health targets, but also it would greatly advance the health status in countries with the highest burdens of disease (DeLuca et al., forthcoming). In 2006, 57 countries failed to meet this target and were considered to have a severe workforce crisis (GHWA, 2010). Of these 57 countries, 36 are in sub-Saharan Africa, home to two-thirds of all people living with HIV globally (and where an estimated 75 percent of adults do not know their status despite scale-up of testing programs and campaigns) (Dayrit et al., 2011; Gilliam et al., 2012). There is a critical need to expand the production and retention of doctors and nurses as well as other frontline clinical providers such as pharmacists and managers (King and Fomundam, 2010; Kober and Van Damme, 2006; Scheffler et al., 2008). Given current levels of pre-service training, it has been estimated that, even after ignoring most forms of attrition, “it would take 36 years for physicians and 29 years for nurses and midwives to reach WHO’s recent target of 2.28 professionals per 1,000 population” in 12 sub-Saharan African workforce crisis countries, with some countries never reaching that target (Kinfu et al., 2009, p. 225).
PEPFAR Inputs
Guidance
PEPFAR’s authorizing legislation provided increased resources to support the training of health care workers, which were needed “particularly at the community and provincial levels,” as well as other community workers and leaders in order to address the HIV epidemic.14 The legislation also recognized the negative impact that emigration was causing on national health workforce capacities in sub-Saharan Africa and called for an analysis of related challenges and strategies for retention of medical and public health personnel.15 The first Five-Year Strategy identified technical assistance for and training of health care professionals, community-based groups, and faith-based organizations as strategies for building local capacity (OGAC, 2004).
In 2009, OGAC elevated the importance of health workforce activities by requiring that PEPFAR mission teams describe these activities in a new section of the COPs: the Human Capacity Development narrative. Previously, this information was captured in the Health Systems Strengthening
__________________
14Supra, note 8 at ![]() 2(21)(A).
2(21)(A).
15Supra, note 8 at ![]() 101(b)(3)(T).
101(b)(3)(T).
narrative. In addition, a secondary cross-cutting budget attribution was added for Human Capacity Development (OGAC, 2008a).16 Workforce activities to develop human capacity include
• human resources for health strategy development and workforce planning,
• Human Resource Information Systems (HRIS),
• training (pre-service, longer-term, and for task shifting),
• performance assessment,
• retention strategies,
• twinning and volunteers,
• management and leadership development, and
• support for salaries (OGAC, 2008a).
With PEPFAR II’s transition to capacity building and sustainability efforts came an emphasis on the goal of training and retaining “health care workers, managers, administrators, health economists, and other civil service employees critical to all functions of a health system” (OGAC, 2009d, p. 8). PEPFAR’s reauthorization legislation expanded the scope of support for health workforce activities and created a new target to train and retain at least 140,000 new health care professionals and paraprofessionals. The target emphasized training for “critically needed doctors and nurses” in order to strengthen partner country capacity to deliver primary health care and help partner countries achieve the WHO-identified critical threshold of 2.3 doctors, nurses, and midwives per 1,000 population.17 The reauthorization legislation called for building the capacity of partner country institutions in order to promote pre-service training of and postsecondary education for health professionals. OGAC’s FY 2010 COP guidance prioritized the development and retention of health care workers in public and nongovernmental settings (OGAC, 2009b). During PEPFAR II workforce efforts focused on pre-service training (or the training of new health care workers) as well as “task-shifting, innovative retention strategies, reemployment, and additional training of health care personnel across the WHO six building blocks of health” (OGAC, 2009f, p. 24).
PEPFAR Funding for Health Care Worker Salaries
Although COP guidance considered salary support to be a component of human capacity development, there are limitations on how PEPFAR
__________________
16 Secondary cross-cutting budget attributions “are designed to capture all funding associated with a cross-cutting program, regardless of program area” (OGAC, 2008a, p. 68).
17Supra, note 2 at ![]() 101(a), 22 U.S.C. 7611(a)(4)(J).
101(a), 22 U.S.C. 7611(a)(4)(J).
funding may be used to support health care worker salaries.18 Prime partners and subpartners may use PEPFAR funding “to pay for time-limited contractors to carry out activities essential to HIV/AIDS program goals” and NGOs may hire personnel to work for their organizations, government institutions, or government health facilities (USAID, 2009, p. 32). PEPFAR funding may also be used to provide bonuses or incentives to public- or private-sector health care workers who achieve certain targets within a performance-based financing scheme. However, PEPFAR funding cannot be used to pay for “salaries of permanent civil service employees on public health institution payrolls” or salary supplements, known as “top-ups,” to employees of partner country governments (USAID, 2009, p. 32). Although PEPFAR cannot support recurrent salaries for government workers, in many countries PEPFAR funding is used to second, or lend, health workforce personnel to government entities (Stash et al., 2012; U.S. Ethiopia GHI Team, 2010). In some countries, PEPFAR support includes top-ups and allowances for health workers at nongovernmental facilities (Hanefeld, 2008; The Maximizing Positive Synergies Academic Consortium, 2009).
PEPFAR Activities
Across countries, PEPFAR has provided support for government leadership related to the health workforce (e.g., planning and monitoring) (240-ES; 934-ES; 587-ES; 116-ES; 166-ES), training and education (240-ES; 636-ES; 934-ES; 587-ES; 461-ES; 331-ES; 196-ES; 116-ES; 166-ES; 272-ES; 396-ES), health worker retention (240-ES; 636-ES; 116-ES; 461-ES; 935-ES), and capacity building of health professional training institutions and universities (240-ES; 587-ES; 116-ES; 166-ES; 461-ES; 934-ES; 396-ES; 331-ES).
Workforce Planning
WHO’s Global Health Workforce Alliance and others have emphasized that countries should have a national plan for workforce development and that investments from bilateral and multilateral donors should be long-term, dependable, and contribute toward the progress of country-led plans (GHWA, 2011; IOM, 2009). Many PEPFAR countries have been making progress in developing HRH plans (GHWA, 2011; WHO, 2012a). Interviewees described various aspects of PEPFAR support for planning and management of national health workforces, such as support for national
__________________
18 These limitations refer to funding from the Global Health and Child Survival appropriations account, which is the largest source of PEPFAR funding (in FY 2010, 86 percent of PEPFAR funding was appropriated through the Global Health and Child Survival account) (OGAC, 2011d). For more information on the PEPFAR funding process, see Chapter 4.
HRH strategies or plans (240-12-USG; 934-38-PCACA); development of a human resources unit within the MOH (587-10-USG); human resource information systems (240-12-USG; 116-7-USG; 166-12-USG); and support for workforce licensing, registration, or regulation (240-12-USG; 116-13-PCNGO).
In-Service Training
In the early phase of PEPFAR, most health worker training involved in-service trainings (short-term training for current health workers) related to HIV testing, ARV management, and other focused, protocol-specific approaches needed to allow health systems to rapidly scale up HIV services. PEPFAR II transitioned to a focus on pre-service training (or the training of new health care workers), task-shifting (or task-sharing), developing and employing retention strategies, and “reemployment and additional training of health care personnel [including for primary care] across the WHO six building blocks of health” (OGAC, 2009f, p. 24).
Until FY 2010, OGAC indicators related to training captured both new training and retraining of individuals (OGAC, 2005c). These indicators track the number of persons trained in each topic without accounting for duplication across topics or years, so while the committee cannot determine how many unique individuals were trained each year, the data show that PEPFAR supported more than 6 million training experiences in various program areas from FY 2005 to FY 2009 (see Table 9-6). In FY 2010, PEPFAR supported nearly 450,000 in-service trainings (but data were not provided to the committee disaggregated by as many program areas as the data from FY 2005 to FY 2009) (see Table 9-7). The committee’s interpretation of these output data was limited as there are no data available on whether individuals that attended trainings were retained in the country, in the health sector, or in positions providing HIV/AIDS-related services.
Despite the renewed focus on pre-service training during PEPFAR II, in-service training is appropriate in some situations. Interviewees from PEPFAR partners described in-service training being used to disseminate new service delivery guidelines (587-5-PCGOV; 636-17-PCGOV) as well as to offer refresher courses for experienced staff (587-5-PCGOV; 396-56-USNGO; 935-23-PCNGO; 934-22-USNGO; 272-26-PCNGO). PEPFAR supports in-service training for multiple cadres of health workers in partner country health systems, including
• doctors (396-29-PCGOV; 542-8-USNGO; 542-11-PCNGO);
• nurses (240-5-PCGOV; 331-12-USG; 636-9-USACA; 542-11-PCNGO);
• volunteers (240-5-PCGOV; 240-7-PCGOV; 272-25-PCNGO);
• pharmacists (396-29-PCGOV);
TABLE 9-6 PEPFAR Indicators Related to Workforce Training(FY 2004–FY 2009)
| FY 2004 | FY 2005 | FY 2006 | FY 2007 | FY 2008 | FY 2009 | |
| Number of individuals in thousands trained: | ||||||
| In blood safety (3.2) | 2.2 | 7.9 | 6.6 | 7.7 | 9.7 | 9.6 |
| In injection safety (4.1) | 4.4 | 14.0 | 55.5 | 78.1 | 67.9 | 75.0 |
| To provide HIV/AIDS prevention programs promoting abstinence and/or being faithful (2.2) | 116.6 | 174.5 | 346.3 | 333.7 | 456.8 | 567.2 |
| To promote HIV/AIDS prevention through other behavior change beyond abstinence and/or being faithful (5.3) | 51.0 | 92.9 | 190.6 | 238.1 | 207.7 | 242.5 |
| To provide HIV palliative care (including TB/HIV) (6.3) | 27.0 | 71.2 | 145.1 | 129.7 | 159.6 | 162.7 |
| To provide treatment for TB to HIV-infected individuals (diagnosed or presumed) (7.3) | 10.5 | 14.9 | 19.9 | 28.5 | 43.3 | 50.2 |
| In counseling and testing according to national and international standards (9.3) | 14.2 | 22.1 | 35.5 | 52.8 | 54.5 | 68.9 |
| In HIV-related community mobilization for prevention care and/or treatment (14.6) | — | — | 87.8 | 100.7 | 95.1 | 94.6 |
| In the provision of lab-related activities (12.2) | 3.1 | 5.7 | 14.2 | 15.4 | 18.4 | 60.0 |
| In strategic information (13.2) | 9.3 | 18.0 | 42.6 | 38.7 | 53.0 | 64.5 |
| In HIV-related policy development (14.3) | 24.1a | 45.9a | 28.3 | 26.9 | 21.1 | 23.5 |
| In HIV-related institutional capacity building (14.4) | 45.4 | 69.6 | 83.5 | 102.6 | ||
| In HIV-related stigma and discrimination reduction (14.5) | — | — | 63.6 | 186.1 | 168.0 | 103.6 |
| Number of health workers in thousands trained: | ||||||
| Or retrained in the provision of PMTCT services (1.4) | 24.4 | 28.5 | 36.2 | 31.7 | 50.8 | 61.6 |
| To deliver ART services (11.5) | 12.3 | 36.6 | 59.0 | 62.6 | 69.3 | 84.1 |
| Number of providers/caretakers in thousands trained: | ||||||
| In caring for OVC (8.2) | 22.5 | 74.8 | 164.4 | 228.0 | 255.0 | 273.1 |
NOTES: For FY 2004 and FY 2005, data were reported by the 15 focus countries. For FY 2006–FY 2010, the number of countries that reported data varied by year from 29 to all 31 of the countries that are the focus of this evaluation (see Chapter 2). The numbers in parentheses are the PEPFAR indicator numbers as published in OGAC’s Indicators Reference Guide for FY 2007 reporting/FY 2008 planning(OGAC, 2007b). Data are presented in thousands. ART = antiretroviral therapy; OVC = orphans and vulnerable children; PMTCT= prevention of mother-to-child transmission.
aIn FY 2004 and FY 2005, indicators 14.3 and 14.4 had not yet been separated and were reported as a single result.
SOURCE: Program monitoring indicators provided by OGAC.
TABLE 9-7 PEPFAR Indicators Related to Workforce Training (FY 2010)
| Result | Target | (%) | |
| Number of new health care workers in thousands who graduated from a pre-service training institution within the reporting period (H2.1.D) | 10.8 | 12.4 | (87) |
| Nurses | 4.4 | 0 | |
| Midwives | 0.4 | 0 | |
| Doctors | 1.4 | 0 | |
| Number of community health and para-social workers in thousands who successfully completed a pre-service training program (H2.2.D) | 136.8 | 102.6 | (133) |
| Number of health care workers in thousands who successfully completed an in-service training program within the reporting period (H2.3.D) | 448.1 | 384.0 | (117) |
| Pediatric treatment | 13.4 | 20.7 | (64) |
| Male circumcision | 2.2 | 4.9 | (46) |
NOTES: The numbers in parentheses are the PEPFAR indicator numbers as published in OGAC’s Next Generation Indicators Reference Guide (OGAC, 2009c). Data are presented in thousands. Indicator H2.1.D includes training for clinical professionals (including doctors, nurses, midwives, laboratory scientists, pharmacists, social workers, medical technologists, and psychologists), other clinical health workers (including, but not limited to, clinical officers, medical and nursing assistants, lab and pharmacy technicians, auxiliary nurses, auxiliary midwives, testing and counseling counselors), and nonclinical health workers (including workers in a health ministry, hospital and facility administrators, managers, monitoring and evaluation advisors, epidemiologists, and other professional staff critical to health service delivery and program support) (OGAC, 2009c).
SOURCE: Program monitoring indicators provided by OGAC.
• medics (587-10-USG) and emergency surgical officers (240-12-USG);
• social workers and counselors (and those working with youth and families) (272-14 -PCNGO; 272-15-PCNGO; 272-20-PCNGO; 272-21-PCNGO; 166-12-USG; 396-12-USG; 396-29-PCGOV);
• laboratory technicians and other personnel (240-2-USG; 240-19-USACA; 116-19-PCACA; 166-11-USG; 272-13-USG; 461-13-USACA; 461-18-USG; 396-22-USG);
• leaders, program managers, and program supervisors (240-7-PCGOV; 587-10-USG; 587-13-USG; 116-7-USG; 116-11-USG; 272-15-PCNGO; 396-55-USG; 934-38-PCACA);
• supply chain personnel (240-12-USG; 934-25-USNGO; 461-13-USACA); and
• data clerks and managers, M&E personnel (196-21-PCGOV; 636-1-USG; 636-18-ONGO; 166-11-USG; 396-29-PCGOV).
In some countries, interviewees expressed the need for continuous in-service training because of frequent turnover of staff and the resulting loss of institutional memory (542-11-PCNGO; 587-7-PCGOV; 587-8-PCGOV; 272-6-ML; 272-32/35-PCNGO; 331-12-USG; 331-44-USNGO; 240-19-USACA; 934-21-USG; 934-45-USNGO; 396-6-PCGOV; 396-41-PCGOV; 396-56-USNGO; 935-2-USG; 166-6-USG; 166-11-USG).
Pre-Service Training
Only 1 year of data for Next Generation Indicator H2.1.D is available to measure progress toward the PEPFAR II goal of training 140,000 new health workers. (See Boxes 9-7 and 9-8 for more information about PEPFAR-supported initiatives to strengthen pre-service training.) This indicator reflects the number of clinical (e.g., doctors, nurses, midwives, laboratory scientists, pharmacists, social workers, medical technologists, and psychologists) and nonclinical (e.g., administrators, managers, monitoring and evaluation advisors, epidemiologists, and other professional staff) health workers who graduated from a university-based or university-affiliated program of at least 6 months in duration (OGAC, 2009c).
The Medical Education Partnership Initiative (MEPI) provides $130 million in direct funding over 5 years to 13 African medical schools for institutional support and management. Each medical school was chosen through a competitive grant process and is partnered with a U.S.-based university. These partnerships have three overarching themes:
1. Increasing capacity through “enhancements in the quantity and quality of medical education in funded schools and in their respective countries. This effort includes increased admissions, curricular innovations, graduate medical education enhancement, and faculty training and support” (Mullan et al., 2012, p. 1564).
2. Retention of “both faculty and graduates to further build the capacity of each school and graduates of the schools in their respective countries” as well as improving geographic distribution of the graduates (Mullan et al., 2012, p. 1564).
3. Regionally relevant research, which is important for the generation of new knowledge and as an instrument of faculty development and retention.
MEPI is coordinated by the George Washington University School of Public Health and Health Services, in partnership with the African Centre for Global Health and Social Transformation in Kampala, Uganda. The coordinating center is responsible for evaluating each grantee’s program and providing technical support. MEPI is funded through OGAC and the National Institutes of Health and receives administrative support from the Health Resources and Services Administration.
SOURCE: Mullan et al., 2012.
Nurses and midwives play an important role in the delivery of health services, particularly in African countries, and PEPFAR has supported capacity building for nurses through various initiatives. In 2006, PEPFAR funded the Global HIV/AIDS Nursing Capacity Building program, implemented by Georgetown University, to build nursing leadership and develop regional networks of nurses to support mentoring (Georgetown University Medical Center, 2006). In 2009, Columbia University’s International Center for AIDS Care and Treatment Programs (ICAP) was awarded a 5-year grant to continue building capacity for nursing through the ICAP Nurse Capacity Initiative (INCI). INCI is active in seven African countries, where it supports national-level nursing strategies and leadership as well as pre-service and in-service training of nurses to increase knowledge and skills (Dohrn, n.d.).
In 2010, OGAC launched the Nursing Education Partnership Initiative (NEPI) to build the capacity of nursing and midwifery schools in partner countries. Through NEPI, PEPFAR provides direct funding to national government working groups that choose the education models and interventions and the schools where they will be implemented (Dohrn, n.d.; Palen et al., 2012).
Indicator H2.2.D captures the number of community health or para-social workers (CHSWs) who completed a pre-service training program with PEPFAR support. Although CHSWs do not count toward the legislative target, these jobs are often the first step in entering the health workforce and they contribute to the pipeline for health workers (OGAC, 2009c). In FY 2010, nearly 11,000 doctors, nurses, and midwives graduated from pre-service training institutions, and nearly 138,000 CHSWs completed a pre-service program with PEPFAR support (see Table 9-7). CHSWs—who are the preponderance of trainees—can contribute significantly to the HIV/AIDS response, but only if sufficient training, remuneration, and oversight support systems are provided (Celletti et al., 2010; Jerome and Ivers, 2010). CHSWs may be trained to deliver health services at various levels of complexity, and they can “improve access to care and services despite the shortage of higher level health care professionals” (Jerome and Ivers, 2010, p. S69). In some partner countries, PEPFAR has supported training for CHSWs, some of whom are deployed in rural areas in order to increase access to services (240-12-USG; 331-12-USG; 461-13-USACA; 166-18-USNGO). PEPFAR partners may also provide financial support, mentoring, and supervision to community health workers who deliver home-based care (934-29-USNGO; 272-7-USG).
Educational/Training Institutions
PEPFAR and other global health donors have been criticized for supporting in-service training and salary support for existing health workers over pre-service training to expand the health workforce (Grépin, 2012b; Vujicic et al., 2012), but since its inception PEPFAR has supported the production of health workers in partner countries through capacity building of educational institutions. During its first phase, PEPFAR supported twinning arrangements between U.S.-based and partner country education institutions to build capacity in diverse areas, such as strengthen nursing HIV/AIDS education in Tanzania (OGAC, 2006b). Partner country stakeholders and PEPFAR mission teams reported PEPFAR support for academic institutions that offer multiple levels of pre-service training: bachelor’s (240-12-USG; 116-7-USG), master’s (240-2-USG; 240-12-USG; 240-38-PCACA), and doctoral programs (240-12-USG; 240-19-PCACA; 331-12-USG). Interviewees in nearly all countries mentioned PEPFAR support for curriculum development (240-2-USG; 240-19-USACA; 587-10-USG; 116-13-PCNGO; 166-19-PCGOV; 166-22-USPS; 461-13-USACA; 396-2-USG; 934-21-USG) and faculty educators (240-2-USG; 331-12-USG; 166-11-USG; 166-22-USPS; 934-44-PCACA; 116-7-USG; 396-39-USG) at local institutions.
PEPFAR has also supported the production of health professionals by providing start-up costs for Field Epidemiology and Laboratory Training Programs (FELTPs) in six countries (Ethiopia, Mozambique, Nigeria, Rwanda, South Africa, and Tanzania) and continuing financial support to a Field Epidemiology Training Program (FETP) in Zimbabwe (Nsubuga et al., 2011). The first FETPs were established in Zimbabwe and Uganda in the early 1990s as partnerships between ministries of health, universities, and sub-national level of government in partner countries (Mukanga et al., 2010). The first FELTP was established in Kenya in 2004; it added a laboratory component to the FETP curriculum. FELTPs combine didactic classes and fieldwork to build competencies in epidemiology and public health laboratory management. After completing the 2-year program, graduates are usually awarded master’s degrees in fields such as public health, applied or field epidemiology, or applied epidemiology and laboratory management (Nsubuga et al., 2011). In 2004, a review of alumni data from the Zimbabwe and Uganda FETPs showed that 85 percent of graduates were still working in their home country 3 years after completing the program, as compared with 40 percent of graduates from medical and other health schools in Africa (Mukanga et al., 2010).
In 2010, recognizing the need to “increase the quantity, quality, and relevance of health care workers” in partner countries, OGAC established the Medical Education Partnership Initiative (MEPI) and the Nursing/Midwifery Education Partnership Initiative (NEPI) to support medical and nursing education institutions across Africa (Palen et al., 2012, p. S115) (see Boxes 9-7 and 9-8). The MEPI and NEPI initiatives are longer-term
investments in African universities intended to improve the quality of physician and nursing education and to increase the quantity of health workers (Mullan et al., 2012; OGAC, 2011f). In the short term, however, these initiatives with a small amount of funding (about $163 million total over 5 years) will not produce large cohorts of front-line clinicians or other health workers to directly staff government or other sector health facilities (Mullan et al., 2012; OGAC, 2011g). Recently, PEPFAR, the Peace Corps, and the Global Health Service Corps launched a public–private partnership to send U.S. medical and nursing professionals to serve as short-term adjunct faculty in overseas medical or nursing schools (Peace Corps, 2012). In 2013, the Global Health Service Partnership will pilot programs in Malawi, Uganda, and Tanzania, supporting 36 volunteers to serve as medical and nursing educators in each country (Global Health Service Corps, 2012). PEPFAR is supporting a similar effort in Rwanda, where the government is partnering directly with 13 U.S.-based universities to strengthen medical and nursing education as part of the country’s Human Resources for Health Program (Nash, 2012; Rwanda Ministry of Health, 2012). The U.S.-based universities will pay the salaries of full-time medical, nursing, dentistry, and health management faculty serving for 1 year as educators in Rwandan academic institutions; the Government of Rwanda will provide a housing allowance (Duke University, 2012). The government estimates that after 8 years it will be “positioned to sustain the improved health workforce on its own without foreign aid” (Rwanda Ministry of Health, 2012).
Retention of Health Workforce
Despite tremendous efforts to train health care workers in partner countries, nearly every country has faced workforce shortages that have challenged the proper functioning of the health system (396-7-PCGOV; 396-9-PCGOV; 240-21-PCGOV; 331-6-CCM; 461-8-PCGOV; 587-10-USG; 587-25-ML; 196-16-PCGOV; 636-2-USG; 166-13; 116-23; 272-13-USG; 934-15-PCGOV). In 2010, the member states of the World Health Assembly adopted the Global Code of Practice on the International Recruitment of Health Personnel, which was developed to provide an ethical framework for international recruitment that attempts to minimize the migration of health workers from countries with severe workforce shortages (WHO, 2010d). However, the principles and practices described in the code are voluntary, and in many countries external migration is a challenge (636-2-USG; 636-7-PCNGO; 240-15-USG; 116-7-USG; 272-2-USG; 935-2-USG). Many elements contribute to migration (either internal, to a different provider or organization, or external, to another country) and attrition (the decision to leave the workforce), including low salaries (331-4-PCGOV; 240-24-USG; 542-11-PCNGO; 935-2-USG), a lack of career and professional development opportunities (587-5-PCGOV; 934-17-PCGOV), poor management or supervision (587-5-PCGOV), and poor workplace conditions (636-
2-USG; 240-19-USACA) (Willis-Shattuck et al., 2008). Low salaries were a frequently cited cause of attrition, not only for clinicians and laboratory personnel, but also for support staff engaged in monitoring and evaluation (396-9-PCGOV; 461-4-USG; 587-9-USG; 934-22-USNGO), supply chain management (331-17-USG), financial management (240-19-USACA; 331-34-USNGO), and other roles. The workplace conditions that were identified as challenges to health workforce retention include lack of access to basic services, such as electricity and running water (636-2-USG; 166-11-USG), and lack of Internet access (240-19-USACA). In many cases health care workers are overwhelmed because of understaffing, and attrition and migration increase the burden on those left behind (331-16-USG; 331-44-USNGO; 587-13-USG; 196-16-PCGOV; 636-17-PCGOV; 116-16-PCGOV; 272-13-USG; 272-25-USG; 935-4-PCGOV). A systematic review of 20 retention studies in low-income countries found that “adequate resources and appropriate infrastructure can improve morale significantly” (Willis-Shattuck et al., 2008). Interviewees agreed that it is necessary to provide an enabling environment with access to appropriate equipment and resource materials in order to retain employees, particularly in rural areas (636-7-PCNGO; 240-19-USACA). Some interviewees felt that opportunities for staff development—‘creating conducive environments for learning and teaching by providing laboratories, reference materials, textbooks, video-conferencing, Internet’ (240-19-USACA)—could contribute to greater staff retention, and there were examples of situations in which this strategy was successful: ‘health center does not experience a lot of staff turnover because there is electricity, water, and accommodations are provided for the staff’ (636-22-PCNGO).
Partner country interviewees described PEPFAR support for financial and nonfinancial incentives for retaining health workers, such as the provision of health care to health care workers and their families (240-12-USG), opening private wings in public hospitals so that physicians can work in both sectors (240-12-USG; 240-33-USG), ensuring access to the necessary medical equipment (116-7-USG; 240-19-USACA), and creating an enabling environment with access to informational resources (240-19-USACA). As described above, PEPFAR funding may not be used to supplement salaries for public-sector health workers unless the bonus payments are tied to performance indicators. In at least one country, per diems offered to workers who attended in-service trainings were considered to be an informal method of increasing salaries (240-12-USG). PEPFAR partners in some countries are providing performance-based top-ups, or bonus payments, when employees work overtime (935-13-PCGOV). Interviewees provided examples of successful efforts to retain health workers by increasing salaries, but these salary increases were not supported by PEPFAR (934-25-USPS; 116-5-PCGOV; 116-7-USG); some interviewees expressed a desire to provide salary top-ups in order to motivate staff and attract well-qualified employees (166-1-USG; 166-6-USG). Sometimes employees leave the public sector,
and then PEPFAR seconds them back to government institutions and provides higher salaries (240-33-USG).
There has been concern that PEPFAR funding may have indirectly contributed to a workforce migration in some countries, whereby higher pay scales for the HIV programs could induce a siphoning-off of workers from primary care and other general health sectors. In Mozambique, for example, more doctors are lost from clinical practice to internal migration, such as to the private sector, than to external migration (Sherr et al., 2012). Interviewees from partner country governments confirmed that private-sector (both for-profit and nonprofit) wages often exceed public-sector wages, creating a strong “pull factor” away to non-public-sector jobs (331-4-PCGOV; 240-5-PCGOV; 240-19-PCGOV; 587-7-PCGOV; 587-18-PCGOV; 934-17-PCGOV; 116-7-USG; 166-5-USG; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS). In one partner country, increased donor funding for HIV/AIDS had both negative and positive effects; health care workers were recruited away from rural areas to work in the capital or away from primary care to HIV services, but increased funding had also increased job opportunities, which gave health care workers who had previously emigrated an incentive to return back to the country (935-2-USG). In particular, interviewees attributed some migration of health workers away from the public sector to PEPFAR funding (935-2-USG; 396-55-USG; 587-18-PCGOV; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS; 240-33-USG).
Task-Shifting
Given the growing evidence about the trajectory of need for the cascade of services from testing to the provision of ART and patient retention, as well as evidence concerning the benefits of earlier provision of antiretroviral therapy,19 it is clear that there is a crucial need for adequate numbers of providers who can initiate and manage ongoing antiretroviral drug use. This includes doctors (who are extremely scarce in low-income country settings) as well as nurses and other associated clinicians with additional training who are capable of managing HIV including the dispensing of medication. Task shifting involves the reassignment of clinical tasks to different cadres of workers (Callaghan et al., 2010). A study of one such model at the PEPFAR-funded AMPATH system in western Kenya found that clinician nurse–led monitoring significantly reduced mortality (Braitstein et al., 2012). Similarly, in South Africa nurse-monitored ART has been found to
__________________
19 The latest guidance from the U.S. Department of Health and Human Services recommends ART for all HIV-infected individuals (Panel on Antiretroviral Guidelines for Adults and Adolescents, 2012). The latest WHO guidance recommends treatment for “all patients with CD4 counts of .350 cells/mm3 irrespective of the WHO clinical stage” (WHO, 2010a, p. 24).
improve treatment outcomes at an efficient cost (Brennan et al., 2011; Long et al., 2011).
A growing body of literature shows that providers other than physicians (e.g., nurse-practitioners, physician assistants, clinical officers, and other associate clinicians) may provide care that is at least equivalent in quality to that provided by physicians for such tasks as determining eligibility and deciding to initiate ART, prescribing medication, and managing treatment (Callaghan et al., 2010; Laurant et al., 2005; Mullan and Frehywot, 2007; Sherr et al., 2010; Shumbusho et al., 2009). In some countries, non-physician providers have been crucial for expanding access to ART, and successful task-shifting models may be able to be extended to other health priorities beyond HIV, including non-communicable diseases, which are increasingly prevalent in low- and middle-income countries (Dohrn et al., 2009; Lekoubou et al., 2010; Sanne et al., 2010). El-Sadr and colleagues, in a summary of PEPFAR achievements, concluded that “PEPFAR’s adoption and support of task-shifting principles has enabled the expansion of HIV treatment in countries with some of the most severe health workforce constraints” (El-Sadr et al., 2012, p. S97).
Task-shifting may support efficient models of service delivery, which will be increasingly required as a growing number of people living with HIV are identified and supported for lifetime care (Fulton et al., 2011). The benefits of task-shifting—which requires training and support rather than simply adding more work to different cadres—include increased access to life-saving treatment; improved workforce skills mix, retention, and health-system efficiency; and likely cost advantages (Zachariah et al., 2009). The need to maximize available resources within health systems (including the implementation of effective quality-improvement systems) will need to be supported as PEPFAR moves into its next phase of work (Leatherman et al., 2010). It has been observed that these efficiencies are necessary “to achieving the goal of universal access to treatment as well as the sustainability of these programmes” (Grépin, 2012a, p. 1).
Task-shifting or task-sharing may occur at all levels of the workforce. For example, the initiation of antiretroviral therapy may be reassigned from doctors to nurses or health officers (240-2-USG; 272-20-NGO; 934-10-PCGOV; 636-4-PCGOV; 116-7-USG; 166-6-USG; 461-14-USG); HIV testing may be reassigned from registered nurses to lay counselors or community health workers (272-13-USG; 934-10-PCGOV); and nonclinical health care workers or volunteers may take responsibility for helping newly diagnosed patients navigate the health system or adhere to ARV regimens or for providing psychosocial support, all of which reduces the burden of clinical health care workers (240-15-USG; 331-19-USNGO; 166-29-PCGOV). Some countries have introduced new cadres of health workers, including associate clinicians, who require less training than doctors but more than nurses (240-2-USG; 166-6-USG; 166-23-USG; 116-19-PCACA), and workers with training in a
particular set of tasks (e.g., case management or home-based care) (240-15-USG). Where there are few doctors, these associate clinicians are trained to do tasks that are usually done by doctors (166-6-USG), such as the diagnosis and treatment of tuberculosis (331-12-USG). PEPFAR has assisted partner country governments in restructuring the health workforce and developing strategies for task-shifting (272-7-USG; 240-15-USG). In several countries, interviewees described other ways in which PEPFAR partners are supporting task-shifting, including technical assistance (272-20-PCNGO), assessments of need for new cadres of workers (116-23-USPS), developing protocols to insure new cadres deliver appropriate services (116-9-PCNGO), mentoring of these new cadres (240-24-USG), evaluating task-shifting efforts (166-1-USG; 461-1-USG), and training for nurse-initiated ART (272-20-PCNGO). PEPFAR has also supported training for new cadres of associate clinician health workers, such as “health officers” or “clinical officers,” to complement task-shifting efforts (240-2-USG; 240-3-USG; 240-5-PCGOV; 240-7-PCGOV; 166-6-USG; 166-23-USG; 116-19-PCACA). Some interviewees felt that these new cadres of workers were more likely to stay in the country than doctors or nurses (116-7-USG). In one country where task-shifting had not been formalized, PEPFAR was supporting research to provide the government with evidence to contribute to policy development (935-14-USG).
PEPFAR Achievements
PEPFAR has supported millions of training encounters, but without data on the number of health care workers working for PEPFAR programs or supported by PEPFAR funding, it is difficult to determine PEPFAR’s impact on the quantity, distribution, productivity, and motivation of health care workers in partner countries (Oomman et al., 2010). In many countries, PEPFAR partners are working on innovative solutions for workforce shortages, such as providing financial and nonfinancial incentives to retain health workers and exploring task-shifting strategies.
Conclusion: PEPFAR’s contribution to health workforces in partner countries has over time been appropriately directed to more pre-service production. Nonetheless, partner countries continue to have considerable need for health workforce development and retention. PEPFAR can contribute to fulfilling that need by leveraging and maximizing its investments in collaborative efforts to build the capacity of health professional training schools, which would improve the ability of countries to address not only HIV but also the dual burden of infectious and non-communicable diseases that many high-burden countries increasingly face. Adherence by partner countries to the Global Code of Practice on the International Recruitment of Health Personnel and follow-through on commit-
ments to the Abuja Declaration could support both sustainability of their own health workforces and country ownership.
Background and Context
The service delivery building block of health systems includes managing, integrating, and scaling up health services as well as innovative strategies to deliver and improve services (WHO, 2012d). Service delivery, for prevention as well as for the treatment and care of disease, is in many ways the purpose of a health system and not simply one of its components. Service delivery stands on a pillar composed of the other blocks: leadership and governance, financing, health information, access to essential medicines and commodities, and health workforce (see Figure 9-7). Thus, challenges within the other building blocks also affect the delivery of health services. Inadequate levels of human resources (workforce), high costs (financing), and lack of the capacity to monitor patients (information) are constraints for expanding and delivering quality services.
Although the most visible aspect of health systems may be government-funded health services, nongovernmental actors from the private sector, civil society, and communities often contribute to the delivery of health services as well. The private for-profit and nongovernmental sectors play an important role in the delivery of health services (OGAC, 2009a), especially in poor countries where out-of-pocket expenditure for health care needs can be high and can become a large portion of expenditures by the poor (Xu et al., 2007). Although not directly reflected in the six building blocks, the engagement of communities in the health care system is a key factor in the impact that service delivery has on population health. During several country visits, interviewees noted the importance that contributions from private and civil society sectors, particularly faith communities, have for service delivery in urban areas (240-ES; 331-ES; 636-ES; 116-ES; 166-ES; 272-ES; 935-ES; 461-ES; 934-ES). Each building block of the health system includes private-sector actors (USAID, 2010), which are further discussed in Chapter 10.
Effective service delivery also depends critically on standards, guidance, and accountability mechanisms to ensure access to quality services (WHO, 2010b). The utilization of services, retention in care, and adherence to health worker advice are just as important to successful service delivery as are other inputs. And to make an already complex model more complicated, these system components are interrelated. Clients respond not only to the cost of services, but also to their quality, including, for example, the delivery of services in ways that respect their dignity (Gilson, 2003). Quality services are characterized by the essential dimensions of safety, effec-
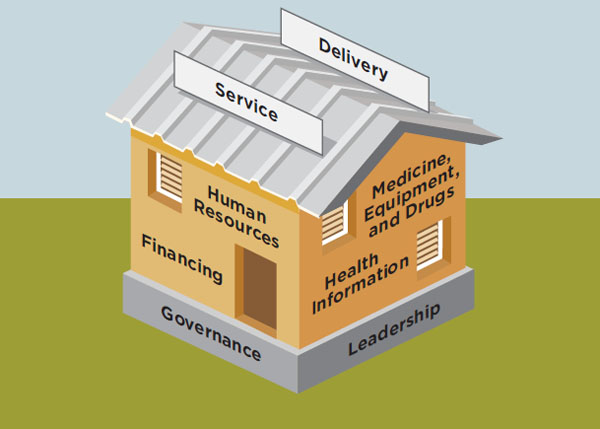
FIGURE 9-7 Health system building blocks represented as a house.
SOURCE: Adapted from Lancet, 2009.
tiveness, integration, continuity, and people-centeredness (WHO, 2010b). While developing improved drug regimens or new laboratory tests is clearly important, true innovation in health systems approaches will come from developing new service delivery models.
Since its inception, PEPFAR has supported an enormous expansion of service delivery. In many countries, PEPFAR has supported the development of health systems infrastructure through the construction (240-5-PCGOV; 240-7-PCGOV; 240-12-USG; 935-9-PCGOV) and renovation of existing facilities (240-7-PCGOV; 240-9-USG; 240-12-USG; 240-24-USG; 116-12-PCNGO; 116-19-PCACA; 166-11-USG; 166-15-USACA; 272-22-USG; 935-12-USPS; 935-24-USNGO; 461-13-USACA; 396-32/33/34-PCGOV) such as health centers, clinics, and hospitals (240-7-PCGOV; 240-12-USG; 240-25-PCGOV; 240-33-USG; 935-12-USPS; 935-24-USNGO); laboratories (240-12-USG; 240-19-USACA; 240-33-USG; 331-17-USG; 587-7-PCGOV; 636-3-USG; 116-19-PCACA; 166-11-USG; 116-15-USACA; 935-9-PCGOV); training centers (240-12-USG) and universities (240-19-USACA); and warehouses for storing commodities (240-12-USG; 587-7-PCGOV; 935-9-USG). Interviewees noted that construction and renovation have sustainable, “horizontal” impacts beyond dealing with HIV/AIDS that strengthen the health sector in general and that contribute to increasing access to services (240-3-USG; 240-7-PCGOV; 240-19-USACA; 240-25-PCGOV; 240-33-USG; 166-15-USACA). PEPFAR activities and achievements in prevention, care, and treatment services for HIV/AIDS are discussed in Chapter 5 on prevention and Chapter 6 on care
and treatment. The committee chose to assess PEPFAR’s cross-cutting and capacity building efforts related to service delivery from a systems-level perspective. In particular, the following discussion reflects the committee’s efforts to understand PEPFAR’s broader contribution to increasing the quality and integration of health services.
Particularly given the constrained global economic climate and the growing responsibility to provide lifelong care to those living with HIV, it is important for PEPFAR and other stakeholders involved in the global HIV/AIDS response to understand how to best identify and propagate efficient models for the prevention, care, and treatment of HIV/AIDS. The challenge is to lower the unit cost of providing HIV/AIDS-related health services while simultaneously ensuring that there is an adequate quality of services to achieve viral suppression for those already living with HIV/AIDS, which improves health and reduces the risk of transmission from this population (Goosby, 2012a). These have been, and continue to be, nearly impossible tasks if one does not address the underlying health care and public health systems.
Quality of Service Delivery
PEPFAR Inputs
Guidance Since its inception, PEPFAR has emphasized its goal of providing quality services for HIV/AIDS and has recognized the importance of interventions across the health systems building blocks in order to achieve this goal (OGAC, 2006a). PEPFAR’s first Five-Year Strategy committed to developing the infrastructure, staff, and capacity necessary to provide high-quality services through technical assistance for the development of appropriate protocols for service delivery and the training of health care providers at all levels of the health system (OGAC, 2004). The FY 2009 COP guidance specified strengthening quality assurance programs as a key activity for building government and nongovernment capacity (OGAC, 2008a). OGAC guidance on the development of Partnership Frameworks identified the need for building government capacity to regulate services provided in the nongovernmental sector and for capacity building across sectors for the delivery of quality services (OGAC, 2009a). PEPFAR’s second Five-Year Strategy noted that “access to quality services for all health conditions remain[ed] problematic in some areas” (OGAC, 2009d, p. 12). One USG official noted that in the beginning, ‘it was almost more important that you had clinics for treatment and programs for prevention without looking at quality’ (NCV-3-USG). Although PEPFAR met and exceeded its HIV treatment service delivery targets during the first phase (see Part II of the report), OGAC recognized that “in some countries, this focus did not
fully translate to a broader service delivery impact across the health sector” (OGAC, 2009d, p. 12).
PEPFAR Activities
PEPFAR has supported a range of efforts to ensure and improve the quality of HIV/AIDS and other health services. Although interviewees said that initially there was insufficient attention paid to quality during the scale-up of services to meet PEPFAR’s ambitious enrollment targets (272-15-PCNGO; 166-15-USACA; 461-17-PCNGO; 935-2-USG), in recent years there has been an increased focus on ensuring the quality of services (240-2-USG; 166-15-USACA), and in some countries, interviewees identified new approaches to improving the quality of services as a primary objective of the PEPFAR program (331-30-USPS; 331-43-USG; 116-1-USG; 396-21-USG).
In nearly all countries, interviewees described interventions with the health workforce as crucial for ensuring the delivery of quality services. PEPFAR supports pre-service (934-5-USG; 331-16-USG; 331-38-USPS; 116-7-USG) and in-service training (934-5-USG; 331-16-USG; 166-15-USACA; 935-13-PCGOV) to build the capacity of health care providers to deliver quality services. PEPFAR partners also use strategies such as supportive supervision (934-22-USNGO; 331-30-USPS; 116-12-PCNGO; 935-22-PCGOV) and mentorship (934-5-USG; 240-2-USG; 240-15-USG; 272-24-USG; 935-23-PCNGO) to improve the ability of health care workers to deliver high-quality services. In countries that are piloting or implementing task-sharing models of care and treatment, PEPFAR is supporting in-service training and mentorship programs to ensure the quality of service delivery (240-2-USG; 240-15-USG). PEPFAR partners are also working to build leadership and management capacity for the monitoring of services (331-12-USG; 331-38-USPS; 331-43-USG; 240-12-USG; 240-19-USACA; 636-6-USG; 587-10-USG).
PEPFAR supports quality assurance (QA) activities (396-12-USG; 934-5-USG; 272-24-USG; 331-12-USG; 587-9-USG; 935-17-USG) and quality improvement (QI) activities (636-6-USG; 461-8-PCGOV; 331-43-USG; 934-44-PCACA; 587-13-USG; 196-1-USG; 272-9-USG; 935-23-PCNGO). Traditionally, QA has referred to retrospective inspections that measure compliance with standards, while QI refers to ongoing approaches to improve the processes and systems necessary for delivering high-quality services (Agins, 2007; HRSA, 2013). Often interviewees used these terms interchangeably, but across countries the committee heard about processes and tools used by PEPFAR partners to monitor and improve quality, including the client-oriented provider-efficient methodology, which was also described as innovative; the continuous quality improvement approach; and the HIVQUAL and HEALTHQUAL projects (331-30-USPS; 331-44-USNGO; 587-9-USG; 587-13-USG; 587-18-PCGOV; 396-55-USG; 461-18-USG; 935-23-PCNGO; 166-15-USACA). Although slightly different, these approaches all involve examining various steps in the service delivery process, identifying weaknesses or barriers to de-
livering high-quality services, and developing solutions to improve the quality of service delivery. In particular, PEPFAR supports HIVQUAL and HEALTHQUAL in 14 countries, and recent evaluations in Thailand, Uganda, and Mozambique showed that the use of this model led to increased clinical performance in HIV care (HEALTHQUAL International, 2011, 2012; Thanprasertsuk et al., 2012).
In many countries, interviewees described the need to develop agreed-upon standards of care before service quality could be assessed against the standard processes described above (240-2-USG; 461-18-USG; 166-15-USACA; 331-12-USG; 331-30-USPS; 116-20-USNGO; 542-21-USNGO). Some PEPFAR partners provide technical assistance to support partner countries in the development and implementation of standards of care for services (240-2-USG; 196-11-USNGO; 331-12-USG; 331-16-USG; 461-18-USG; 166-15-USACA; 116-20-USNGO; 272-9-USG).
There was widespread agreement across countries about the importance of collecting and using data to monitor the quality of services (240-19-USACA; 272-18-PCNGO; 272-32-PCNGO; 396-55-USG; 461-18-USG; 166-15-USACA; 587-9-USG; 116-9-PCNGO; 934-22-USNGO), including the use of both paper (such as maternal care registries and child health passports) and electronic registries and health records (116-2-USG; 272-20-PCNGO; 587-18-PCGOV; 935-23-PCNGO). To increase the availability of data for monitoring the quality of services, PEPFAR partners have supported the development of electronic patient tracking systems (166-15-USACA; 935-23-PCNGO) and tools for collecting and tracking information (934-45-USNGO; 461-19-USG; 935-23-PCNGO; 331-30-USPS). In Uganda, the PEPFAR program has incorporated quality-specific indicators into the M&E systems of treatment partners, including “the retention of patients on ART and adherence to preventive care guidelines (such as the use of cotrimoxazole)” (OGAC, 2006a, p. 46). More information about PEPFAR’s support for various activities related to strengthening the generation and use of health data is presented in the Information building block.
PEPFAR Achievements
Interviewees in many countries reported that PEPFAR partners deliver high-quality HIV/AIDS services (272-15-PCNGO; 196-1-USG; 461-13-USACA; 461-18-USG) and that efforts to improve the quality of HIV/AIDS services have had some success (587-18-PCGOV; 934-15-PCGOV; 461-13-USACA; 461-15-USG; 331-12-USG; 331-30-USPS; 331-44-USNGO; 272-15-PCNGO; 935-23-PCNGO). In some countries, people chose not to seek health care services because of perceptions that the services were of poor quality; specific mention was made of such issues as dilapidated facilities, a lack of medical supplies and commodities, and unqualified or unmotivated staff (240-22-PCNGO; 240-24-USG). In addition to focused efforts to improve the quality of services, interviewees mentioned the contributions that PEPFAR activities across the health systems building blocks have made to improving quality,
such as increasing the availability of medicines and medical supplies (240-24-USG; 934-5-USG; 542-21-USNGO), increased laboratory capacity (934-5-USG; 935-2-USG), and improving the infrastructure (196-21-PCGOV; 116-19-PCACA; 396-32/33/34-PCGOV).
Integration of Services
PEPFAR Inputs
Guidance From the beginning, a fundamental principle of PEPFAR has been to integrate prevention, treatment, and care services for HIV/AIDS (OGAC, 2004), and the reauthorization legislation called specifically for the integration of services to ensure a continuum of care for those affected by HIV/AIDS.20 OGAC’s guidance for the development of Partnership Frameworks called for the integration of “existing parallel service delivery systems with [. . .] government-coordinated and managed health system[s]” in order to facilitate government leadership of HIV/AIDS programs (OGAC, 2009a, p. 14). There has also been increasing global recognition of the importance of access to family planning services for HIV-positive women who want to space or limit births, and OGAC has emphasized the importance of linking HIV/AIDS and family planning programs (OGAC, 2011a).
In 2009, President Obama proposed the Global Health Initiative (GHI), a 6-year $63 billion effort to develop a comprehensive and highly integrated strategy for all U.S. global health programming (OGAC, 2009f). One of the principles of the GHI is to “increase impact through strategic integration and coordination” through support for “holistic planning and programming among health and development programs” as well as through support for integration “where effective and efficient” (OGAC, 2009f, p. 7). As the largest component of the GHI, PEPFAR support for the delivery of coordinated and integrated services is described in the program’s second Five-Year Strategy. Some of the goals during PEPFAR II are to integrate PEPFAR quality interventions with other health and development programs; to ensure improved coordination with the partner country governments and between U.S. agencies; and to identify and implement efficiencies to maximize the ability of proven programs not only to provide HIV care, treatment, and prevention, but also to respond to the overall health needs faced by PLHIV, their families, and their communities. The strategy described several areas where PEPFAR could support greater integration, such as co-location of HIV/TB services, co-location of HIV and reproductive health services, expanded workforce training to provide health care workers with a strong background in primary care, linking PEPFAR food and nutrition programs with other development initiatives, incorporating HIV prevention messages
__________________
20Supra, note 2 at ![]() 101(a), 22 U.S.C. 7611(a)(4)(D).
101(a), 22 U.S.C. 7611(a)(4)(D).
into educational programs, and expanding economic strengthening and microfinance opportunities for PLHIV (OGAC, 2009f).
PEPFAR Activities
Despite OGAC’s early articulation of the principles of integration for HIV/AIDS services, the emergency approach during PEPFAR I, coupled with the condition of many country health systems, resulted in parallel or vertical systems being established for the delivery of HIV/AIDS services in some countries, particularly in the case of HIV treatment services, which had previously been unavailable in the public sector in nearly all countries. In some countries visited, interviewees described HIV care and treatment facilities that had been built, staffed, and supplied separately from other government health facilities (451-4-USG; 935-ES; 240-ES; 272-ES; 396-ES; 542-ES). However, interviewees noted a renewed focus on integration during PEPFAR II, including a transition from parallel systems to integration and from projects to programs (587-7-PCGOV; 461-18-USG; 396-18-USG). In particular, the GHI offered a fresh opportunity for USG agencies to think about integration (116-2-USG; 331-14-USG; 396-60-USG).
During the committee’s country visits, partner country government, multilaterals, and USG participants described integration as an essential part of the HIV/AIDS response (934-12-CCM; 587-10-USG; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS) and pointed to efforts to integrate as many services as possible into the overall health system (587-10-USG). In several partner countries, there was strong government support and commitment for integration (166-10-USNGO; 331-2-USG; 240-24-USG; 461-11-PCGOV). Both country partners and the USG had similar assumptions about the benefits that service integration has for the health system in general, such as maximizing the value of human resources (587-5-PCGOV; 587-6-CCM; 396-39-USG; 934-10-PCGOV), preventing stigma (396-23-USG; 331-2-USG), increasing access to services for HIV/AIDS (240-15-USG) and other health needs (934-12-CCM; 934-44-PCACA; 396-60-USG), increasing acceptability of services to patients (331-2-USG), integrating the assistance at the donor partner level in order to strengthen the health sector overall (166-34-PCGOV), and reducing costs (934-10-PCGOV; 272-20-PCNGO) in order to create efficiencies for both the health system and its clients. However, service integration should not be assumed to be cost-neutral in the initial efforts and for some time after integration (IOM, 2010). Although evidence suggests that some types of HIV/AIDS service integration can be cost-effective, more research is necessary to determine the most efficient processes of integration (Sweeney et al., 2012).
Integration of different HIV services PEPFAR has supported many different models for the integration of different HIV/AIDS services, particularly
through systems of linkages and referrals at the facility and community levels.
Facility level Many interviewees defined effective integration of HIV services as the delivery of multiple services (prevention, care, treatment) within a single facility (396-21-USG; 396-39-USG; 240-19-USACA; 934-15-PCGOV; 196-17-PCGOV; 461-18-USG; 542-11-PCNGO; 116-12-PCNGO). PEPFAR partners are supporting integration of HIV services at multiple levels, including technical assistance for national guidelines for service integration (240-19-USACA), technical assistance to build capacity of facilities to deliver integrated services (396-21-USG; 461-18-USG; 240-29-USNGO), and training to build capacity of health care workers to provide integrated services (396-21-USG). The co-location of services provides more convenient access to services and reduces the time and transportation costs for patients that would otherwise have to visit multiple locations or return for multiple visits (934-15-PCGOV; 396-44-PCGOV; 272-20-PCNGO). Some interviewees believed that integration of HIV services would reduce the cost of service delivery, particularly in rural areas (240-2-USG; 934-10-PCGOV; 272-20-PCNGO).
Generally, this “one-stop shop” model is used for delivery of outpatient services—such as counseling and testing, prevention education, treatment for opportunistic infections, and palliative care—and patients in need of inpatient services are referred to hospitals (196-17-PCGOV). In many countries, patients can access prevention and care services at local health centers or clinics, but must be referred to upper-level clinics or hospitals for initiation or delivery of ART (196-17-PCGOV; 934-15-PCGOV; 461-18-USG). To reduce the loss to follow-up that occurs when patients do not access the services that they are referred to, one country is piloting efforts to send teams of health workers from district hospitals to community rural health centers to initiate patients on treatment within communities (934-10-PCGOV). PEPFAR support for strengthening laboratory services (described previously in this chapter) has expanded access to CD4 testing and treatment monitoring at lower-level facilities (166-11-USG; 461-18-USG; 934-45-USNGO). In some countries, patients accessing clinical services at health facilities were referred to NGOs or CSOs to access additional HIV services such as community support groups and home-based care (396-21-USG; 331-44-USNGO; 240-19-USACA).
Community level In many countries, PEPFAR supports NGOs and CSOs that provide community- or home-based services and are important linkages to health facilities (396-8-PCNGO; 272-15-PCNGO; 240-19-USACA; 636-6-USG; 166-23-USG; 272-7-USG; 331-34-USNGO). In one country, local partners supporting community-based nutrition interventions and psychosocial support services help to identify patients in need of PMTCT services and refer them to the appropriate facilities (636-6-USG). In several countries, HIV prevention education activities were integrated into HIV counseling and testing and care services
in order to reach different settings and populations, such as communities in general, women, parents, in-school youth, people living with HIV/AIDS, people who inject drugs, and members of civil society organizations (331-18-USNGO; 272-12-USNGO; 396-21-USG; 196-20-PCNGO; 331-24-PCGOV; 331-44-USNGO; 396-40-PCGOV; 542-11-PCNGO; 196-21-PCGOV). Interviewees identified civil society organizations as critical for informing people about the availability of services, as well as linking vulnerable populations to health services (196-24-PCNGO; 396-21-USG; 396-32-PCGOV; 396-44-PCGOV; 542-11-PCNGO; 331-18-USNGO; 331-44-USNGO). In several countries, referrals were tracked by providing examination cards or referral forms to patients (272-15-PCNGO; 396-31-PCGOV; 196-21-PCGOV; 934-22-USNGO). The efforts in these countries could be reproduced in other countries to address the challenge of tracking patients or verifying that services were actually provided (272-15-PCNGO; 587-13-USG; 587-5-PCGOV).
Integrating HIV with other health services Interviewees across countries emphasized PEPFAR’s efforts to integrate HIV services with other health services, including maternal and child health (MCH) services, reproductive health and family planning services, and services for tuberculosis. Effective referral systems are sometimes necessary when services for different diseases, such as TB and HIV, are co-located but delivered separately within a single facility (240-19-USACA). (TB/HIV integration is discussed in Chapter 6 and is therefore not discussed here.) Interviewees reported that PEPFAR is supporting innovations to support and integrate medical and psychosocial services for addressing the needs of PLHIV; women who are survivors of intimate partner violence; sex workers seeking to change their job skills; orphans and other vulnerable children, including street children; people who inject drugs; and others with psychosocial needs by using mobile outreach teams, by training social workers (including curriculum development), and by encouraging multisectoral collaboration for including social workers in a variety of settings as a needed cadre of workers for health services (396-40-PCGOV; 396-21-USG; 542-11-PCNGO; 542-12-PCGOV; 542-14-PCGOV; 166-27-PCNGO; NCV-10-USG; 587-8-PCGOV; 934-31-PCNGO).
In many countries, PEPFAR-supported partners have integrated reproductive health services—such as family planning and cervical cancer screening—with HIV services (166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS; 116-18-PCNGO; 166-10-USNGO; 935-21-PCGOV; 240-24-USG; 461-21-PCNGO; 542-11-PCNGO; 934-8-USNGO; 934-17-PCGOV; 272-21-PCNGO; 636-17-PCGOV; 331-14-USG). PEPFAR-supported partners are also supporting HIV testing and treatment for PMTCT as part of an integrated package of MCH services (166-10-USNGO; 272-24-USG; 935-19-PCGOV). In some countries, interviewees said that government support had facilitated the integration of these services (166-10-USNGO; 116-10-USNGO; 934-10-PCGOV), but interviewees in other countries described vertically organized government systems for health ser-
vices (542-6-PCGOV; 396-22-USG), including family planning, which made integration of these services a challenge (331-16-USG; 396-45-USNGO).
PEPFAR support has also contributed to the integrated delivery of food and nutrition interventions and HIV services, particularly for children and moderately to severely malnourished patients on ART (240-2-USG; 116-20-USNGO; 331-16-USG; 396-42-PCGOV). Where food and nutrition interventions are not integrated with HIV services, PEPFAR-supported partners are providing linkages to other nutritional support (636-6-USG; 240-15-USG); referring patients for food subsidies (especially children living with elders and individuals living in child-headed households) (166-8-USG); and developing nutritional guidelines and tools (935-10-USG).
Other examples Interviewees in a number of different countries described integration of a variety other services with HIV services, including increasing access to potable water (331-14-USG; 934-14-PCGOV; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS), providing insecticide-treated bed nets and malaria prophylaxis (331-14-USG; 636-17-PCGOV), and incorporating chronic disease management into home-based care services (587-18-PCGOV). In one country, PEPFAR supported the integration of separate reference laboratories for tuberculosis, immunology, and malaria (166-11-USG). In several countries, PEPFAR has supported the efforts to provide HIV prevention messages and testing services in workplace settings with the intention to link those who test positive for HIV to appropriate care and treatment services (272-25-USG; 396-50-PCGOV; 587-14-PCGOV; 636-4-PCGOV; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS). Additional models and approaches to integrating HIV/AIDS services are presented in Box 9-9.
Challenges for Service Integration
Many interviewees described challenges to the goals of integrating prevention, treatment, and care services for HIV/AIDS as well as integrating HIV/AIDS services with other health services. Integration initiatives faced challenges despite the integration policy being well accepted (331-16-USG) and the development, in some countries, of guidelines for integration (240-24-USG). Interviewees reported a lack of integration at several levels of the delivery of services, a lack that was attributed to still treating HIV as a separate entity (240-24-USG; 935-ES; 240-ES; 272-ES; 396-ES; 542-ES). In one country, interviewees said that every region has an HIV clinic but that these clinics are not integrated into the country’s health system (331-12-USG). Interviewees reported loss to follow-up when patients are referred from counseling and testing to care and treatment programs (587-12-USG; 587-13-USG; 240-23-PCGOV; 196-11-USNGO; 272-9-USG; 461-14-USG), poor linkages from PMTCT and ART programs to family planning services (240-15-USG; 240-24-USG; 935-10-USG); and ART working as a standalone program in some cases (240-19-USACA).
BOX 9-9
Select Examples of PEPFAR-Supported Models
and Approaches to Service Integration
Integration into Primary Care: Efforts by both the government and PEPFAR to integrate HIV/AIDS into primary care while simultaneously strengthening health systems are in progress in many countries (587-13-USG; 396-39-USG; 331-44-USNGO; 934-14-PCGOV; 272-17-USG; 196-7-PCNGO). The committee heard several examples of co-locating various services at primary-level clinics, including counseling and testing, treatment of opportunistic infections, ART, PMTCT, nutrition, and health promotion (272-20-PCNGO; 461-18-USG; 331-16-USG; 396-42-PCGOV; 166-10-USNGO; 935-19-PCGOV). There are also efforts to expand strategies such as the Integrated Management of Childhood Illness and integrate early infant diagnosis into primary health care (272-24-USG). In some instances, national-level leadership felt that HIV care and treatment services were too complex to be delivered at the primary care level (587-13-USG; 934-14-PCGOV). Hospices were connected to primary facilities through referrals (272-7-USG). One interviewee called for integrating prevention and HIV counseling and testing, along with linkages to NGO services, with primary care (196-11-USNGO).
Integration of HIV/AIDS Within the National Health Care System or Existing Platforms: Interviewees described integration with existing platforms for service delivery as an opportunity to address other needs besides HIV (934-12-CCM; 934-44-PCACA; 396-60-USG) as well as to reduce costs (934-10-PCGOV; 272-20-PCNGO). Several interviewees considered reducing costs to be critically important because of challenges with funding (934-12-CCM; 196-23-PCNGO; 116-12-PCNGO; 272-20-PCNGO; 587-5-PCGOV; 331-44-USNGO). Interviewees noted that scaling up successful integration models could promote sus-
Challenges specific to partner country governments Interviewees reported vertically structured government health systems as a challenge to integration because of the lack of connections between separate government programs for managing diseases (396-22-USG; 396-45-USNGO; 542-6-ML; 587-3-USG; 272-24-USG). An interviewee in one country said that the vertical funding of national programs made it difficult to carry out integration at the national level (331-16-USG). Some interviewees expressed concern about integrating HIV services into health systems with weak primary care services or health systems where health care workers are already overburdened or in short supply (587-2-USG; 587-13-USG; 272-20-PCNGO; 116-6-USG; 935-10-USG). The decentralization taking place in the health care system of some countries disrupted the flow that was already established among the different service delivery levels (240-15-USG). Finally, political support for integration varied among countries; some partner country
tainability (196-22-PCGOV; 542-11-PCNGO). In many countries, PEPFAR support has strengthened health information systems, workforce capacity, and supply chain networks, and through the integration of services, these investments can be leveraged to improve non-HIV-related health outcomes (116-2-USG; 396-55-USG; 166-6-USG).
Family-Centered Approach: PEPFAR-supported partners in some countries are implementing or considering family-centered or family-based models of service delivery through which services are provided to adults and children at the same time; services may be delivered in clinics or homes (396-21-USG; 636-6-USG; 116-7-USG; 116-12-PCNGO; 240-24-USG; 240-2-USG). These approaches create efficiencies because they require fewer visits for the patients and less staff to address the whole family in the same visit (587-13-USG). PEPFAR has supported national-level policies for the rollout of family-centered approaches and the development of training materials for health workers delivering family-centered services (240-24-USG; 396-21-USG).
Health Information: Improving data systems also contributed to having a stronger referral system. Some countries implemented electronic referral systems that “down-referred” patients from higher-level to lower-level or smaller clinics for drug management and then “up-referred” them when they needed clinicians (587-12-USG; 461-11-PCGOV; 272-20-PCNGO). Interviewees reported several initiatives to improve patient monitoring using electronic data systems and linking maternal and child care databases (116-2-USG), by using child health passports to track babies born to HIVpositive mothers (116-2-USG), and by integrating MCH registers with PMTCT (587-5-PCGOV). These efforts were implemented to address the challenge of monitoring HIV patients and to decrease the loss to follow-up in such patients (587-5-PCGOV; 587-13-USG).
governments were hesitant or resistant to supporting integration of HIV services into primary care (396-39-USG; 587-13-USG). Interviewees were concerned about the limited funding for integration efforts coming from the government (116-12-PCNGO; 272-20-PCNGO), such as the lack of funding for follow-up and care after testing in the transgender community (196-23-PCNGO).
Challenges specific to PEPFAR Several interviewees said that PEPFAR’s initially vertical approach to scaling up HIV services was responsible for some of the current challenges for integration (396-39-USG; 272-7-USG; 461-4-USG). In some countries, interviewees noted that USG agencies or partners in-country worked separately and at different service delivery levels, which contributed to broken referrals and linkages between services (240-15-USG; 461-14-USG; 935-24-USNGO). These challenges have been overcome in some
countries where stronger coordination has been developed across USG agencies (272; 542; 935) and with the change of the approach described in the PEPFAR’s second Five-Year Strategy. Other interviewees were unsure about PEPFAR’s position on or support for different models of integration and wanted more clarity about how to move forward on integration (116-2-USG; 396-7-PCGOV).
PEPFAR supported many integration initiatives, some of them as projects or pilots, which meant that the funding stopped when the project ended. For example, a PEPFAR-supported project on PMTCT services integration that trained hospital staff to support pediatric care and link maternal care with pediatric care was found to be successful but had to stop (331-44-USNGO). Some interviewees characterized funding coming from PEPFAR as inflexible and narrowly focused and mentioned that they were not allowed to fund non-HIV activities even when, in their opinion, these would impact HIV (587-3-USG; 396-12-USG). Interviewees said partners usually had a positive view of integration until they were competing for funding (331-16-USG).
Access and capacity challenges One of the challenges to integration concerned access at the community level to testing, care and treatment, and other services when the facilities that provided all the needed services were not close to the patient or there was a lack of appropriate facilities to which to refer patients (461-14-USG; 240-25-PCGOV; 272-15-PCNGO). This problem was compounded when there was a lack of adequate infrastructure (587-3-USG). There were, however, several activities to improve or build infrastructure to facilitate integration efforts, including, for example, improvements in laboratory services (240-19-USACA; 166-10-USNGO; 166-11-USG). The availability of enough providers and adequate training for providers were also described as critical challenges for efforts to integrate services, such as family planning for PLHIV and care for children and families (935-13-PCGOV; 116-7-USG). This was one area where many activities were put in place to address the lack of skilled personnel, especially in PMTCT, MCH, and family planning services (934-17-PCGOV; 587-5-PCGOV; 166-10-USNGO; 396-21-USG; 935-13-PCGOV).
Data challenges Limitations in information systems were often described as one of the challenges to facilitating effective linkages and referral systems. One country, for example, offers anonymous HIV testing but does not link test results to a name or code so it is difficult to track patients from testing to care (587-13-USG). In other countries, lack of data sharing or data compatibility between different facilities (e.g., mobile clinics, health centers, NGOs) made it difficult to determine whether patients were truly lost to follow up or simply accessing services at different facilities (935-10-USG; 587-5-PCGOV).
Stigma Stigma presented a challenge for linking patients who test HIV positive to care and treatment services when patients are in denial and refuse to accept their HIV-positive status or when patients go out of their way to seek services outside of their community (636-6-USG; 542-16-PCGOV; 331-7-PCNGO). Furthermore, there was also a concern that integrating services would make it easier to identify patients as being HIV-positive (396-44-PCGOV), but many interviewees felt that integrating services would reduce stigma (116-7-USG; 331-7-PCNGO; 934-21-PCGOV). PEPFAR funding was used to support implementing partners in the provision of quality services that would ensure the confidentiality of patients (396-44-PCGOV).
Future challenges Interviewees offered some insight on ways to move forward on integration. They said it was important to do more strategic planning and health systems strengthening (396-55-USG). Within countries, some interviewees felt that PEPFAR partners delivering different services could work together to strengthen linkages and referrals (396-21-USG; 166-10-USNGO). According to interviewees, linkages to ART, including linkages between HIV diagnosis and ART, could be strengthened by supporting community groups to track clients and help reduce loss to follow-up (461-18-USG). One interviewee spoke of the need for a network to respond to the HIV epidemic that is composed of the health, education, and social welfare systems—and even the church—sharing the same strategy and providing all the available resources (396-8-PCNGO).
PEPFAR Achievements
Most of the discourse in the literature has focused on the extent to which PEPFAR’s efforts have resulted in parallel systems for HIV/AIDS services and the consequences for the rest of the health care system, both theoretical (Travis et al., 2004) and empirical (Biesma et al., 2009; Brugha et al., 2010a,b; Dutta et al., 2012; Grépin, 2012a,b; Hanefeld, 2010; Kruk et al., 2012; Shiffman, 2008; Shiffman et al., 2009). During the committee’s country visits, however, the renewed emphasis on integration of services in PEPFAR II was considered a successful part of the HIV/AIDS response (934-12-CCM; 587-10-USG; 166-9-ML/OBL/USACA/USNGO/PCNGO/PCPS). Interviewees reported that progress had been achieved in integration with continuity of care for HIV patients from point of testing to treatment to follow-up in the community (116-12-PCNGO), in coordination for disbursement of drugs among PEPFAR-supported government outpatient clinics (396-29-PCGOV), and in using other USG funding (non-PEPFAR) to build service linkages (934-2-USG). In one country, PEPFAR programs were very well integrated with the government health system and government facilities as well as with other donor activities (636-16-USG).
Conclusion: PEPFAR’s impressive achievements in service delivery represent the success of a largely disease-specific approach, which had both positive and negative effects on partner country national health systems. In some countries, an early emphasis in PEPFAR implementation on increasing the volume of services to meet targets for service delivery resulted in disease-specific programming, which did not always facilitate service integration. PEPFAR has articulated the goal of increased integration of services and has had some success. Many stakeholders in partner countries have identified an interest or need for greater integration of HIV services into the general health system. The best practices for integrating services—such as those for HIV and TB, reproductive health, and primary care—need to be identified, evaluated, and scaled up.
Other Service Delivery Issues
In the published literature, the evidence on PEPFAR’s spillover effects to other health services appears to be mixed. In PEPFAR-supported facilities in sub-Saharan Africa, Kruk et al. found that the number of patients receiving ART, the availability of support groups for PLHIV, and the availability of advanced HIV infrastructure (e.g., onsite laboratories and electronic databases) were statistically associated with an increased volume of facility births by women who were HIV-negative (Kruk et al., 2012). In Kenya there is some evidence that HIV treatment scale-up may have supported health delivery efficiency as measured by costs, rather than becoming a trade-off (Dutta et al., 2012). In an analysis of overall donor funding for HIV (not exclusively PEPFAR funding), Grépin found that HIV funding may have had a positive effect on maternal health services but a negative effect on child immunization services (Grépin, 2012b). Data from Zambia showed both positive and negative synergies between the scale-up of non-HIV services and the scale-up of HIV services supported by PEPFAR and the Global Fund (Brugha et al., 2010b).
In contrast to the attention given to possible parallel systems and the goal of service integration, there is relatively little PEPFAR-wide experience in health system innovations, such as performance-based financing, other incentive programs, or risk pooling. One exception appears to be Rwanda, where PEPFAR helped support several innovative strategies, including performance-based financing; the Mutuelles, Rwanda’s national community insurance system; and an innovative approach to human resources for health (Logie et al., 2008; Lu et al., 2012). Rwanda has pioneered performance-based financing in primary health care (Basinga et al., 2011). The U.S. Agency for International Development, with financial
support from PEPFAR, has examined the impact of financial incentives for HIV-related treatment performance targets.
Performance-Based Financing
Performance-based financing (PBF) is an innovative funding strategy that rewards “the delivery of specific services to encourage higher coverage, better quality or improved health outcomes” (WHO, 2010e, p. 75). PBF links funding to outputs or outcomes, and there is some evidence that PBF can help strengthen health systems and increase the quality of service delivery (Holmes et al., 2012; Palen et al., 2012; Samb et al., 2009). OGAC has identified PBF as a innovation for improving efficiencies, health outcomes, and sustainability (OGAC, 2011a,c,d). In some countries, PEPFAR partners are supporting PBF strategies in which the disbursement of funding is dependent on a partner or sub-partner reaching a specific target or result (166-5-USG) (EGPAF, 2012). One PEPFAR-supported partner is piloting PBF with its subpartners. The organization monitors indicators of performance from district health clinics, and roughly 20 to 25 percent of each grant is dependent on achieving specific targets (166-10-USNGO). One interviewee wanted PEPFAR to engage in more PBF (461-15-USG). Another asserted that PBF by an external partner such as PEPFAR undermines national authority and that poor quality data for monitoring performance makes implementation of PBF a challenge (331-5-ML). In Rwanda and Côte d’Ivoire, PEPFAR is supporting partner country governments to implement PBF (Holmes et al., 2012; Logie et al., 2008).
There are a number of ways in which PEPFAR, in both phases of the program, has contributed to the strengthening of health systems in high-HIV burden countries in the areas of workforce development, training, and retention; health information tools and systems development; health financing, with an emphasis on financial management capacity building; and capacity building and opportunities for the practical application for leadership and governance with the Partnership Frameworks and Implementation Plans. PEPFAR has had exemplary achievements in the strengthening of laboratory infrastructure and procurement systems for health commodities and technologies, and it has demonstrated positive spillover effects in the areas of blood safety and medical injection safety for overall individual and population health.
Initially, as an emergency response, PEPFAR sometimes developed and supported parallel components of health systems, including the delivery of services outside of partner country health systems; separate processes for
procurement and supply chain management; and monitoring, evaluation, and reporting outside of national information systems. In some countries, an early emphasis on increasing service volume to meet service delivery targets did not always facilitate service integration. As PEPFAR is transitioning from an emergency to a sustainable response, it has placed an increased emphasis on strengthening and integrating with system components in partner countries. In order to avoid undermining areas of non-HIV-related health care delivery, and to ensure that stretched health systems can deliver care for all populations, PEPFAR, in conjunction with partner country governments and other external donors, may be able to synergize even further efforts for overall health system strengthening.
Recommendation 9-1: To support the delivery of HIV-related services, make progress toward sustainable management of the HIV response, and contribute to other health needs, PEPFAR should continue to implement and leverage efforts that have had positive effects within partner country health systems. PEPFAR should maintain efforts in all six building blocks but have a concerted focus on areas that will be most critical for sustaining the HIV response, especially workforce, supply chain, and financing.
Further considerations for implementation of this recommendation:
• An important focus for PEPFAR’s future activities and policies should be support for partner country capacity to locally produce and retain clinical, nonclinical, and management professionals whose training and scope of practice are appropriate and optimized for the tasks needed. MEPI and NEPI have provided a starting point for the training of physicians and nurses; however, the training of associate clinician providers and other cadres will also be critical to the sustainable management of the response. In addition, PEPFAR needs to augment its efforts to build partner country capacity to track the placement of trained workers, to promote retention, and to develop long-term human resources plans. (See also the discussion and recommendation for capacity building in Chapter 10 on progress toward a sustainable response.)
• Building on the progress made through the public–private partnership with the Supply Chain Management System, PEPFAR should enhance and expand efforts with a greater focus on capacity building for accountable supply chain management in partner countries. The aim of this improved capacity should be
to gradually shift to local or regional leadership, coordination, and management to ensure a reliable supply chain for essential medicines and commodities.
• Financing and leadership and governance are particularly critical for the sustainable management of the HIV response; this area is addressed in Recommendation 10-1 (see Chapter 10).
• To contribute to the knowledge base for health systems strengthening, PEPFAR should include this area in its research and evaluation agenda and its knowledge dissemination efforts. (See also recommendations for PEPFAR’s knowledge management in Chapter 11.)
Abimiku, A. G., T. Croxton, E. Akintunde, B. Okelade, J. Jugu, S. Peters, P. Dakum, W. Blattner, and N. Constantine. 2010. Experiences in establishing a PEPFAR-supported laboratory quality system in Nigeria. American Journal of Clinical Pathology 134(4):541-549.
AbouZahr, C., and T. Boerma. 2005. Health information systems: The foundations of public health. Bulletin of the World Health Organization 83(8):578-583.
Agins, B. D. 2007. HIVQUAL-International. The HRSA-HAB Global Quality Improvement Center: The way forward. Presented at PEPFAR Track 1 Partners Meeting, Washington, DC.
Alemnji, G. A., S. Branch, A. Best, M. Kalou, B. Parekh, W. Waruiru, E. Milstrey, W. Conn, J. N. Nkengasong, and S. Lecher. 2012. Strengthening national laboratory health systems in the Caribbean region. Global Public Health 7(6):648-660.
Amico, P., C. Aran, and C. Avila. 2010. HIV spending as a share of total health expenditure: An analysis of regional variation in a multi-country study. PLOS ONE 5(9):e12997.
ASCP (American Society for Clinical Pathology). 2012. ASCP shows its far-reaching impact on PEPFAR countries at International AIDS Conference. http://www.ascp.org/Newsroom/ASCP-Shows-Its-Far-Reaching-Impact-on-PEPFAR-Countries-at-International-AIDS-Conference.html (accessed February 14, 2013).
Bärnighausen, T., D. E. Bloom, and S. Humair. 2011. Going horizontal—shifts in funding for GH interventions. New England Journal of Medicine 364(23):2181-2183.
Bärnighausen, T., D. E. Bloom, and S. Humair. 2012. Health systems and HIV treatment in sub-Saharan Africa: matching intervention and programme evaluation strategies. Sexually Transmitted Infections 88(2):e2.
Basinga, P., P. J. Gertler, A. Binagwaho, A. L. Soucat, J. Sturdy, and C. M. Vermeersch. 2011. Effect on maternal and child health services in Rwanda of payment to primary health-care providers for performance: an impact evaluation. Lancet 377(9775):1421-1428.
BD (Becton, Dickinson and Company). 2012. BD and PEPFAR launch Labs for Life to strengthen laboratories in regions heavily burdened by disease. http://www.bd.com/contentmanager/b_article.asp?Item_ID=26813&ContentType_ID=1&BusinessCode=20001&d=BD+Worldwide&s=&dTitle=&dc=&dcTitle= (accessed February 14, 2013).
Biesma, R. G., R. Brugha, A. Harmer, A. Walsh, N. Spicer, and G. Walt. 2009. The effects of global health initiatives on country health systems: a review of the evidence from HIV/AIDS control. Health Policy and Planning 24(4):239-252.
Bilimoria, N. F. 2012. Lessons learned from a decade of partnership between PEPFAR and the Global Fund: A case study from Tanzania. Health Affairs 31(7):1415-1421.
Birx, D., M. de Souza, and J. N. Nkengasong. 2009. Laboratory challenges in the scaling up of HIV, TB, and malaria programs: The interaction of health and laboratory systems, clinical research, and service delivery. American Journal of Clinical Pathology 131(6):849-851.
Botswana Ministry of Health. 2012. Health commodities storage best practices. Ministry of Health, Republic of Botswana and the Supply Chain Management System (SCMS).
Braitstein, P., A. Siika, J. Hogan, R. Kosgei, E. Sang, J. Sidle, K. Wools-Kaloustian, A. Keter, J. Mamlin, and S. Kimaiyo. 2012. A clinician-nurse model to reduce early mortality and increase clinic retention among high-risk HIV-infected patients initiating combination antiretroviral treatment. Journal of the International AIDS Society 15(1):7.
Brennan, A. T., L. Long, M. Maskew, I. Sanne, I. Jaffray, P. MacPhail, and M. P. Fox. 2011. Outcomes of stable HIV-positive patients down-referred from a doctor-managed antiretroviral therapy clinic to a nurse-managed primary health clinic for monitoring and treatment. AIDS 25(16):2027-2036.
Brinkerhoff, D., and T. Bossert. 2008. Health governance: Concepts, experience, and programming options. Washington, DC: USAID, Health Systems 20/20, Policy Brief.
Brugha, R., J. Kadzandira, J. Simbaya, P. Dicker, V. Mwapasa, and A. Walsh. 2010a. Health workforce responses to global health initiatives funding: a comparison of Malawi and Zambia. Human Resources for Health 8:19.
Brugha, R., J. Simbaya, A. Walsh, P. Dicker, and P. Ndubani. 2010b. How HIV/AIDS scale-up has impacted on non-HIV priority services in Zambia. BMC Public Health 10:540.
Callaghan, M., N. Ford, and H. Schneider. 2010. A systematic review of task-shifting for HIV treatment and care in Africa. Human Resources for Health 8:8.
Celletti, F., A. Wright, J. Palen, S. Frehywot, A. Markus, A. Greenberg, R. A. Teixeira de Aguiar, F. Campos, E. Buch, and B. Samb. 2010. Can the deployment of community health workers for delivery HIV services response to health workforce shortages? Results of multicountry study. AIDS 24:S45-S57.
Chen, L., T. Evans, S. Anand, J. I. Boufford, H. Brown, M. Chowdhury, M. Cueto, L. Dare, G. Dussault, G. Elzinga, E. Fee, D. Habte, P. Hanvoravongchai, M. Jacobs, C. Kurowski, S. Michael, A. Pablos-Mendez, N. Sewankambo, G. Solimano, B. Stilwell, A. de Waal, and S. Wibulpolprasert. 2004. Human resources for health: Overcoming the crisis. Lancet 364(9449):1984-1990.
COE (Council of Europe). 2005. Warsaw Declaration and Action Plan. Council of Europe Warsaw Summit, May 16-17.
Cohen, G. M. 2007. Access to diagnostics in support of HIV/AIDS and tuberculosis treatment in developing countries. AIDS 21:S81-S87.
Committee of Ministers. 2010. Recommendation CM/Rec(2010)6 of the Committee of Ministers to member states on good governance in health systems. European Journal of Health Law 17(4):389-401.
CSCMP (Council of Supply Chain Management Professionals). 2010. Supply chain management terms and glossary. http://cscmp.org/sites/default/files/user_uploads/resources/downloads/glossary.pdf (accessed December 12, 2012).
Dayrit, M. M., C. Dolea, and N. Dreesch. 2011. Addressing the human resources for health crisis in countries: How far have we gone? What can we expect to achieve by 2015? Revista Peruana de Medicina Experimental y Salud Pública 28(2):327-336.
DeLuca, M. A., A. E. Kurth, and A. Hagopian. 2013 (forthcoming). Investing strategically in the global health workforce. In Transforming the Global Health Workforce, edited by M. A. DeLuca and A. Soucat. New York: New York University Press.
Diallo, A. 2011. PEPFAR II: The transition from an emergency program to building sustainability. Presented at 2011 APHL Annual Meeting, Omaha, Nebraska.
Dohrn, J. n.d. Nursing Education Partnership Initiative (NEPI). Presented at 2011 MEPI Symposium, Johannesburg, South Africa.
Dohrn, J., B. Nzama, and M. Murrman. 2009. The impact of HIV scale-up on the role of nurses in South Africa: Time for a new approach. Journal of Acquired Immune Deficiency Syndromes 52(Suppl 1):S27-S29.
Duke University. 2012. Rwanda Human Resources for Health Program: Partnership with the Government of Rwanda, Ministry of Health. Terms of reference for internal medicine and pediatrics applicants. https://globalhealth.duke.edu/sites/default/files/files/rwanda_hrh_internal_medicine_and_pediatrics_job_description_03_14_12.pdf (accessed December 4, 2012).
Dutta, A., N. Wallace, P. Savosnick, J. Adungosi, U. M. Kioko, S. Stewart, M. Hijazi, and B. Gichanga. 2012. Investing in HIV services while building Kenya’s health system: PEPFAR’s support to prevent mother-to-child HIV transmission. Health Affairs 31(7):1498-1507.
EGPAF (The Elizabeth Glaser Pediatric AIDS Foundation). 2012. Transitioning large-scale HIV care and treatment programs to sustainable national ownership: The Project Heart experience. Washington, DC: EGPAF.
El-Sadr, W., C. Holmes, P. Mugyenyi, H. Thirumurthy, T. Ellerbrock, R. Ferris, I. Sanne, A. Asiimwe, G. Hirnschall, R. Nkambule, L. Stabinski, M. Affrunti, C. Teasdale, I. Zulu, and A. Whiteside. 2012. Scale-up of HIV treatment through PEPFAR: A historic public health achievement. Journal of Acquired Immune Deficiency Syndromes 60(Suppl 3): S96-S104.
Fox, L. M., N. Ravishankar, J. Squires, T. Williamson, and D. Brinkerhoff. 2010. Rwanda health governance assessment. Bethesda, MD: Health Systems 20/20.
Friedman, E., I. Katz, E. Kiley, E. Williams, and A. Lion. 2011. Global Fund’s support for health systems strengthening interventions: A reference guide. Bethesda, MD: Physicians for Human Rights, Health Systems 20/20 project, Abt Associates Inc.
Fulton, B. D., R. M. Scheffler, S. P. Sparkes, E. Y. Auh, M. Vujicic, and A. Soucat. 2011. Health workforce skill mix and task shifting in low income countries: a review of recent evidence. Human Resources for Health 9(1):1.
Garg, C. C., D. B. Evans, T. Dmytraczenko, J. A. Izazola-Licea, V. Tangcharoensathien, and T. T. Ejeder. 2012. Study raises questions about measurement of “additionality,” or maintaining domestic health spending amid foreign donations. Health Affairs 31(2):417-425.
Georgetown University Medical Center. 2006. Georgetown Program to Build Nursing Capacity for HIV/AIDS Care in Africa. http://explore.georgetown.edu/news/?ID=15686 (accessed October 16, 2012).
Gershy-Damet, G.-M., P. Rotz, D. Cross, E. H. Belabbes, F. Cham, J.-B. Ndihokubwayo, G. Fine, C. Zeh, P. A. Njukeng, S. Mboup, D. E. Sesse, T. Messele, D. L. Birx, and J. N. Nkengasong. 2010. The World Health Organization African Region laboratory accreditation process. American Journal of Clinical Pathology 134(3):393-400.
GHWA (Global Health Workforce Alliance). 2010. Will we acheive universal access with health workforce we have? Geneva: GHWA, WHO.
GHWA. 2011. Reviewing Progress, Renewing Commitment. Progress report on the Kampala Declaration and Agenda for Global Action. Geneva: GHWA, WHO.
Gilliam, B. L., D. Patel, R. Talwani, and Z. Temesgen. 2012. HIV in Africa: Challenges and directions for the next decade. Current Infectious Disease Reports 14(1):91-101.
Gilson, L. 2003. Trust and the development of health care as a social institution. Social Science & Medicine 56(7):1453-1468.
Global Fund. 2012. Country Coordinating Mechanisms. http://www.theglobalfund.org/en/ccm (accessed October 12, 2012).
Global Health Service Corps. 2012. The joint program. http://globalhealthservicecorps.org/joint-program (accessed November 20, 2012).
Goosby, E. 2011. PEPFAR’s partnership with the Global Fund improves the response to HIV/AIDS. In DipNote: U.S. Department of State Official Blog. http://blogs.state.gov/2011/05/article/pepfars-partnership-global-fund-improves-response-hivaids (accessed May 14, 2013).
Goosby, E. 2012a. The President’s Emergency Plan for AIDS Relief: Marshalling all tools at our disposal toward an AIDS-free generation. Health Afffairs 31(7):1593-1598.
Goosby, E. M. D. 2012b. The way forward: maximizing our impact through shared responsibility and smart investments. Journal of Acquired Immune Deficiency Syndromes 60(Suppl 2):S44-S47.
Grebe, E. 2009. The emergence of effective “AIDS response coalitions”: A comparison of Uganda and South Africa. Presented at Mobilizing Social Capital in a World with AIDS Workshop, Salzburg, Austria.
Grépin, K. A. 2012a. Efficiency considerations of donor fatigue, universal access to ARTs and health systems. Sexually Transmitted Infections 88(2):75-78.
Grépin, K. A. 2012b. HIV donor funding has both boosted and curbed the delivery of different non-HIV health services in sub-Saharan Africa. Health Affairs 31(7):1406-1414.
Hanefeld, J. 2008. Impact beyond intent: the role of global health initiatives in ART roll-out. Evidence from Zambia and South Africa. Presented at XVII International AIDS Conference, Mexico City.
Hanefeld, J. 2010. The impact of Global Health Initiatives at national and sub-national level—a policy analysis of their role in implementation processes of antiretroviral treatment (ART) roll-out in Zambia and South Africa. AIDS Care 22(Suppl 1):93-102.
Health Metrics Network. 2008. Framework and standards for country health information systems. Second Edition. Geneva: Health Metrics Network, WHO.
Health Metrics Network. n.d. Components of a strong health information system: A guide to the HMN Framework. Geneva: Health Metrics Network, WHO.
HEALTHQUAL International. 2011. HEALTHQUAL International Brief 2(2).
HEALTHQUAL International. 2012. HEALTHQUAL International: A Public Health Approach to Quality Management. Who We Are. http://www.healthqual.org/who-we-are (accessed July 24, 2012).
HLSP Institute. 2006. Roles and responsibilities of National AIDS Commissions: Debates and issues. United Kingdom: HLSP Institute.
Holmes, C. B., J. M. Blandford, N. Sangrujee, S. R. Stewart, A. DuBois, T. R. Smith, J. C. Martin, A. Gavaghan, C. A. Ryan, and E. P. Goosby. 2012. PEPFAR’s past and future efforts to cut costs, improve efficiency, and increase the impact of global HIV programs. Health Affairs 31(7):1553-1560.
HRSA (U.S. Health Resources and Services Administration). 2013. What is the difference between Quality Improvement and Quality Assurance? http://www.hrsa.gov/healthit/toolbox/HealthITAdoptiontoolbox/QualityImprovement/whatarediffbtwqinqa.html (accessed January 31, 2013).
IHME (Institute for Health Metrics and Evaluation). 2011. Financing global health 2011: Continued growth as MDG deadline approaches. Seattle: IHME.
IMF (International Monetary Fund). 2012. Debt relief under the Heavily Indebted Poor Countries (HIPC) Initiative. http://www.imf.org/external/np/exr/facts/hipc.htm (accessed October 12, 2012).
IOM (Institute of Medicine). 2007. PEPFAR implementation: Progress and promise. Washington, DC: The National Academies Press.
IOM. 2009. The U.S. commitment to global health: Recommendations for the public and private sectors. Washington, DC: The National Academies Press.
IOM. 2010. Promoting cardiovascular health in the developing world: A critical challenge to achieve global health. Washington, DC: The National Academies Press.
IOM and NRC (National Research Council). 2009. Sustaining Global Surveillance and Response to Emerging Zoonotic Diseases. Washington, DC: The National Academies Press. IOM and NRC. 2010. Strategic approach to the evaluation of programs implemented under The Tom Lantos and Henry J. Hyde U.S. Global Leadership Against HIV/AIDS, Tuberculosis, and Malaria Reauthorization Act of 2008. Washington, DC: The National Academies Press.
Jamieson, D. 2011. Efficiency gains through smarter transportation and pooled procurement. Presented at PEPFAR’s Smart Investments to Save More Lives: Efficiencies, Innovation, Impact, Washington, DC.
Jerome, G., and L. Ivers. 2010. Community health workers in health systems strengthening: A qualitative evaluation from rural Haiti. AIDS 24:S67-S72.
Justman, J. E., S. Koblavi-Deme, A. Tanuri, A. Goldberg, L. F. Gonzalez, and C. R. Gwynn. 2009. Developing laboratory systems and infrastructure for HIV scale-up: A tool for health systems strengthening in resource-limited settings. Journal of Acquired Immune Deficiency Syndromes 52:S30-S33.
KFF (Kaiser Family Foundation). 2012. Zimbabwean AIDS activists march to National AIDS Council demanding accountability for AIDS Levy funds. http://kgh.preview.kff.org/Daily-Reports/2012/October/11/GH-101112-Zimbabwe-AIDS-Levy.aspx (accessed October 11, 2012).
Kinfu, Y., M. R. Dal Poz, H. Mercer, and D. B. Evans. 2009. The health worker shortage in Africa: Are enough physicians and nurses being trained? Bulletin of the World Health Organization 87(3):225-230.
King, R. C., and H. N. Fomundam. 2010. Remodeling pharmaceutical care in Sub-Saharan Africa (SSA) amidst human resources challenges and the HIV/AIDS pandemic. International Journal of Health Planning and Management 25(1):30-48.
Kober, K., and W. Van Damme. 2006. Public sector nurses in Swaziland: Can the downturn be reversed? Human Resources for Health 4:13.
Kruk, M. E., A. Jakubowski, M. Rabkin, B. Elul, M. Friedman, and W. El-Sadr. 2012. PEPFAR programs linked to more deliveries in health facilities by African women who are not infected with HIV. Health Affairs 31(7):1478-1488.
Lalvani, P., P. Yadav, K. Curtis, and M. Bernstein. 2010. Increasing patient access to antiretrovirals. Washington, DC: Center for Global Development.
Lancet. 2009. Health in South Africa: An executive summary for the Lancet series. South Africa Series Executive Summary Core Group.
Laurant, M., D. Reeves, R. Hermens, J. Braspenning, R. Grol, and B. Sibbald. 2005. Substitution of doctors by nurses in primary care. Cochrane Database of Systematic Reviews (2):CD001271.
Leatherman, S., T. G. Ferris, D. Berwick, F. Omaswa, and N. Crisp. 2010. The role of quality improvement in HSS. International Journal for Quality in Health Care 22(4):237-243.
Lekoubou, A., P. Awah, L. Fezeu, E. Sobngwi, and A. P. Kengne. 2010. Hypertension, diabetes mellitus and task shifting in their management in sub-Saharan Africa. International Journal of Environmental Research and Public Health 7(2):353-363.
Levine, R., and N. Oomman. 2009. Global HIV/AIDS Funding and Health Systems: Searching for the win-win. Journal of Acquired Immune Deficiency Syndromes 52:S3-S5.
Logie, D. E., M. Rowson, and F. Ndagije. 2008. Innovations in Rwanda’s health system: Looking to the future. Lancet 372(9634):256-261.
Long, L., A. Brennan, M. P. Fox, B. Ndibongo, I. Jaffray, I. Sanne, and S. Rosen. 2011. Treatment outcomes and cost-effectiveness of shifting management of stable ART patients to nurses in South Africa: An observational cohort. PLOS Medicine 8(7):e1001055.
Lu, C., M. T. Schneider, P. Gubbins, K. Leach-Kemon, D. Jamison, and C. J. L. Murray. 2010. Public financing of health in developing countries: A cross-national systematic analysis. Lancet 375:1375-1387.
Lu, C., B. Chin, J. L. Lewandowski, P. Basinga, L. R. Hirschhorn, K. Hill, M. Murray, and A. Binagwaho. 2012. Towards universal health coverage: An evaluation of Rwanda Mutuelles in its first eight years. PLOS ONE 7(6):e39282.
Mhofu, S. 2012. Zimbabwe HIV activists push for government accountability. http://www.voanews.com/content/zimbabwe_hiv_activists_push_for_government_accountability/1524053.html (accessed October 10, 2012).
Mills, A. 2007. Strengthening health systems. In The Commonwealth Health Ministers Book. London, UK: Commonwealth Secretariat.
MSH (Management Sciences for Health). 2009. Global presence: Grant Management Solutions (GMS). http://www.msh.org/global-presence/grant-management-solutions.cfm (accessed January 1, 2013).
Mukanga, D., O. Namusisi, S. N. Gitta, G. Pariyo, M. Tshimanga, A. Weaver, and M. Trostle. 2010. Field epidemiology training programmes in Africa—where are the graduates? Human Resources for Health 8:18.
Mullan, F., and S. Frehywot. 2007. Non-physician clinicians in 47 sub-Saharan African countries. Lancet 370(9605):2158-2163.
Mullan, F., S. Frehywot, F. Omaswa, N. Sewankambo, Z. Talib, C. Chen, J. Kiarie, and E. Kiguli-Malwadde. 2012. The Medical Education Partnership Initiative: PEPFAR’s effort to boost health worker education to strengthen health systems. Health Affairs 31(7):1561-1572.
Nash, K. 2012. UMSON participating in pioneering effort to improve health care in Rwanda. http://nursing.umaryland.edu/news/4462 (accessed December 4, 2012).
Nsubuga, P., K. Johnson, C. Tetteh, J. Oundo, A. Weathers, J. Vaughan, S. Elbon, M. Tshimanga, F. Ndugulile, C. Ohuabunwo, M. Evering-Watley, F. Mosha, O. Oleribe, P. Nguku, L. Davis, N. Preacely, R. Luce, S. Antara, H. Imara, Y. Ndjakani, T. Doyle, Y. Espinosa, D. Kazambu, D. Delissaint, J. Ngulefac, and K. Njenga. 2011. Field epidemiology and laboratory training programs in Sub-Saharan Africa from 2004 to 2010: Need, the process, and prospects. Pan African Medical Journal 10(24).
OAU (Organization of African Unity). 2001. Abuja Declaration on HIV/AIDS, tuberculosis and other related infectious diseases. Presented at African Summit on HIV/AIDS, Tuberculosis and Other Related Infectious Diseases, Abuja, Nigeria.
OGAC (Office of the U.S. Global AIDS Coordinator). 2004. The President’s Emergency Plan for AIDS Relief: U.S. five-year global HIV/AIDS srategy. Washington, DC: OGAC.
OGAC. 2005a. Emergency Plan for AIDS Relief fiscal year 2005 operational plan: June 2005 update. Washington, DC: OGAC.
OGAC. 2005b. Engendering bold leadership: The President’s Emergency Plan for AIDS Relief. First annual report to Congress. Washington, DC: OGAC.
OGAC. 2005c. The President’s Emergency Plan for AIDS Relief: Indicators, reporting requirements, and guidelines for focus countries. Washington, DC: OGAC.
OGAC. 2006a. Action today, a foundation for tomorrow: The President’s Emergency Plan for AIDS Relief. Second annual report to Congress. Washington, DC: OGAC.
OGAC. 2006b. The U.S. President’s Emergency Plan for AIDS Relief fiscal year 2006: Operational plan. 2006 August update. Washington, DC: OGAC.
OGAC. 2007a. The power of partnerships: The President’s Emergency Plan for AIDS Relief. Third annual report to Congress. Washington, DC: OGAC.
OGAC. 2007b. The President’s Emergency Plan for AIDS Relief: Indicators, reporting requirements, and guidelines. Indicators reference guide: FY2007 reporting/FY2008 planning. Washington, DC: OGAC.
OGAC. 2007c. The U.S. President’s Emergency Plan for AIDS Relief fiscal year 2007: Operational plan. 2007 June update. Washington, DC: OGAC.
OGAC. 2008a. The President’s Emergency Plan for AIDS Relief: FY2009 country operational plan Guidance. Washington, DC: OGAC.
OGAC. 2008b. The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) fiscal year 2008: PEPFAR operational plan. June 2008. Washington, DC: OGAC.
OGAC. 2009a. Guidance for PEPFAR partnership frameworks and partnership framework implementation plans. Version 2.0. Washington, DC: OGAC.
OGAC. 2009b. The President’s Emergency Plan for AIDS Relief: FY2010 country operational plan guidance: Programmatic considerations. Washington, DC: OGAC.
OGAC. 2009c. The President’s Emergency Plan for AIDS Relief: Next generation indicators reference guide. Version 1.1. Washington, DC: OGAC.
OGAC. 2009d. The U.S. President’s Emergency Plan for AIDS Relief: Five-year strategy. Washington, DC: OGAC.
OGAC. 2009e. The U.S. President’s Emergency Plan for AIDS Relief five-year strategy. Annex: PEPFAR and the global context of HIV (December 2009). Washington, DC: OGAC.
OGAC. 2009f. The U.S. President’s Emergency Plan for AIDS Relief five-year strategy. Annex: PEPFAR’s contribution to the Global Health Initiative. Washington, DC: OGAC.
OGAC. 2010a. The President’s Emergency Plan for AIDS Relief: FY2011 country operational plan guidance appendices. Washington, DC: OGAC.
OGAC. 2010b. The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) fiscal year 2009: PEPFAR operational plan. November 2010. Washington, DC: OGAC.
OGAC. 2011a. The President’s Emergency Plan for AIDS Relief: FY2012 country operational plan guidance. Washington, DC: OGAC.
OGAC. 2011b. The President’s Emergency Plan for AIDS Relief: FY2012 country operational plan guidance appendices. Washington, DC: OGAC.
OGAC. 2011c. The President’s Emergency Plan for AIDS Relief: FY2012 country operational plan guidance technical considerations. Washington, DC: OGAC.
OGAC. 2011d. The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) fiscal year 2010: PEPFAR operational plan. April 2011. Washington, DC: OGAC.
OGAC. 2011e. The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) fiscal year 2011: PEPFAR operational plan. December 2011. Washington, DC: OGAC.
OGAC. 2011f. The U.S. President’s Emergency Plan for AIDS Relief: Seventh annual report to Congress. Washington, DC: OGAC.
OGAC. 2011g. United States addresses human resources for health in Africa with daunch of the Nursing Education Partnership Initiative (NEPI). http://www.state.gov/r/pa/prs/ps/2011/11/177307.htm (accessed February 27, 2013).
OGAC. 2012a. Email communication between OGAC staff and IOM: “RE: Followup request: Partnership frameworks and implementation plans.” Washington, DC: OGAC.
OGAC. 2012b. The President’s Emergency Plan for AIDS Relief: FY2013 country operational plan guidance. Washington, DC: OGAC.
Oomman, N., D. Wendt, and C. Droggitis. 2010. Zeroing in: AIDS donors and Africa’s health workforce. Washington, DC: Center for Global Development.
Palen, J., W. El-Sadr, A. Phoya, R. Imtiaz, R. Einterz, E. Quain, J. Blandford, P. Bouey, and A. Lion. 2012. PEPFAR, health system strengthening, and promoting sustainability and country ownership. Journal of Acquired Immune Deficiency Syndromes 60(Suppl 3): S113-S119.
Panel on Antiretroviral Guidelines for Adults and Adolescents. 2012. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. http://www.aidsinfo.nih.gov/contentfiles/lvguidelines/adultandadolescentgl.pdf (accessed October 3, 2012).
Peace Corps. 2012. Peace Corps, PEPFAR and Global Health Service Corps launch public-private partnership to boost training for health professionals in developing countries. http://www.peacecorps.gov/resources/media/press/1986 (accessed November 20, 2012).
PEPFAR (U.S. President’s Emergency Plan for AIDS Relief )/Botswana. 2007. PEPFAR country operational plan FY2008. Washington, DC: OGAC.
PEPFAR/Botswana. 2009. PEPFAR country operational plan FY2010. Washington, DC: OGAC.
PEPFAR/Ethiopia. 2007. PEPFAR country operational plan FY2008. Washington, DC: OGAC.
PEPFAR/Rwanda. 2010. Country operational plan FY2011. Washington, DC: OGAC.
PEPFAR/Thailand. 2009. PEPFAR country operational plan FY2010. Washington, DC: OGAC.
PFSCM (Partnership for Supply Chain Management, Inc). 2010. About us. http://pfscm.org/pfscm/about (accessed August 13, 2012).
Piot, P., and A. M. Coll Seck. 2001. International response to the HIV/AIDS epidemic: Planning for success. Bulletin of the World Health Organization 79(12):1106-1112.
Rwanda Ministry of Health. 2012. Human resources for health program. Program overview. http://hrhconsortium.moh.gov.rw/about-hrh/program-overview (accessed May 14, 2013).
Samb, B., T. Evans, M. Dybul, R. Atun, J.-P. Moatti, S. Nishtar, A. Wright, F. Celletti, J. Hsu, J. Y. Kim, R. Brugha, A. Russell, and C. Etienne. 2009. An assessment of interactions between global health initiatives and country health systems. Lancet 373(19541040): 2137-2169.
Sanne, I., C. Orrell, M. P. Fox, F. Conradie, P. Ive, J. Zeinecker, M. Cornell, C. Heiberg, C. Ingram, R. Panchia, M. Rassool, R. Gonin, W. Stevens, H. Truter, M. Dehlinger, C. van der Horst, J. McIntyre, R. Wood, and C.-S. S. Team. 2010. Nurse versus doctor management of HIV-infected patients receiving antiretroviral therapy (CIPRA-SA): a randomised non-inferiority trial. Lancet 376(9734):33-40.
Scheffler, R. M., J. X. Liu, Y. Kinfu, and M. R. Dal Poz. 2008. Forecasting the global shortage of physicians: an economic- and needs-based approach. Bulletin of the World Health Organization 86(7):516B-523B.
SCMS (Supply Chain Management System). 2012. Six years of saving lives through stronger public health supply chains: A report on SCMS contributions to PEPFAR results. Arlington, VA: SCMS.
Shakarishvili, G. 2009. Building on health systems frameworks for developing a common approach to health systems strengthening. Prepared for the World Bank, the Global Fund, and the GAVI Alliance Technical Workshop on Health Systems Strengthening, Washington, DC.
Sherr, K., M. A. Micek, S. O. Gimbel, S. S. Gloyd, J. P. Hughes, G. C. John-Stewart, R. M. Manjate, J. Pfeiffer, and N. Weiss. 2010. Quality of HIV care provided by non-physician clinicians and physicians in Mozambique: a retrospective cohort study. AIDS 24:S59-S66.
Sherr, K., A. Mussa, B. Chilundo, S. Gimbel, J. Pfeiffer, A. Hagopian, and S. Gloyd. 2012. Brain drain and health workforce distortions in Mozambique. PLOS ONE 7(4):e35840.
Shiffman, J. 2008. Has donor prioritization of HIV/AIDS displaced aid for other health issues? Health Policy and Planning 23(2):95-100.
Shiffman, J., D. Berlan, and T. Hafner. 2009. Has aid for AIDS raised all health funding boats? Journal of Acquired Immune Deficiency Syndromes 52(Suppl 1):S45-S48.
Shumbusho, F., J. van Griensven, D. Lowrance, I. Turate, M. A. Weaver, J. Price, and A. Binagwaho. 2009. Task shifting for scale-up of HIV care: Evaluation of nurse-centered antiretroviral treatment at rural health centers in Rwanda. PLOS Medicine 6(10).
Speybroeck, N., Y. Kinfu, M. R. D. Poz, and D. B. Evans. 2006. Reassessing the relationship between human resources for health, intervention coverage and health outcomes. Geneva: WHO.
Spicer, N., J. Aleshkina, R. Biesma, R. Brugha, C. Caceres, B. Chilundo, K. Chkhatarashvili, A. Harmer, P. Miege, G. Murzalieva, P. Ndubani, N. Rukhadze, T. Semigina, A. Walsh, G. Walt, and X. Zhang. 2010. National and subnational HIV/AIDS coordination: Are global health initiatives closing the gap between intent and practice? Global Health 6:3.
Stash, S., J. Cooke, M. Fisher, and A. Kramer. 2012. Competing Pressures for U.S. PEPFAR in Botswana: rising ambitions, declining resources. Washington, DC: Center for Strategic and International Studies.
Sturchio, J. L., and G. M. Cohen. 2012. How PEPFAR’s public-private partnerships achieved ambitious goals, from improving labs to strengthening supply chains. Health Affairs 31(7):1450-1458.
Sweeney, S., C. D. Obure, C. B. Maier, R. Greener, K. Dehne, and A. Vassall. 2012. Costs and efficiency of integrating HIV/AIDS services with other health services: A systematic review of evidence and experience. Sexually Transmitted Infections 88(2):85-99.
Task Force on Global Action for Health System Strengthening. 2008. G8 Hokkaido Toyako Summit follow-up. Global action for health system strengthening: Policy recommendations to the G8. Presented at G8 Hokkaido Toyako Summit, Toyako, Japan.
Thanprasertsuk, S., S. Supawitkul, R. Lolekha, P. Ningsanond, B. D. Agins, M. S. McConnell, K. K. Fox, S. Srisongsom, S. Chunwimaleung, R. Gass, N. Simmons, A. Chaovavanich, S. Jirajariyavej, T. Leusaree, S. Akksilp, P. A. Mock, S. Chasombat, C. Lertpiriyasuwat, J. W. Tappero, and W. C. Levine. 2012. HIVQUAL-T: Monitoring and improving HIV clinical care in Thailand, 2002-08. International Journal for Quality in Health Care 24(4):338-347.
The Maximizing Positive Synergies Academic Consortium. 2009. Interactions between global health initiatives and health systems: Evidence from countries. World Health Organization’s Maximizing Positive Synergies Project. Geneva: WHO.
Thompson, K. 2011. PEPFAR/BD partnership to expand laboratory capacity. Presented at PEPFAR’s Smart Investments to Save More Lives: Efficiency, Innovation and Impact, Washington, DC.
Travis, P., S. Bennett, A. Haines, T. Pang, Z. Bhutta, A. A. Hyder, N. R. Pielemeier, A. Mills, and T. Evans. 2004. Overcoming health-systems constraints to achieve the Millennium Development Goals. Lancet 364(9437):900-906.
UNAIDS (Joint United Nations Programme on HIV/AIDS). 2004. “Three Ones” key principles. Conference Paper 1, International Conference on AIDS and STIs in Africa (ICASA), Nairobi, Kenya.
UNAIDS. 2009. Monitoring the Declaration of Commitment on HIV/AIDS: Guidelines on Construction of Core Indicators: 2010 Reporting. Geneva: UNAIDS.
United Nations General Assembly. 2000. United Nations Millennium Declaration, resolution adopted by the General Assembly, A/RES/55/2. New York: United Nations.
U.S. Ethiopia GHI (Global Health Initiative) Team. 2010. Ethiopia Global Health Initiative Strategy. Washington, DC/Addis Ababa, Ethiopia: U.S. Ethiopia GHI Team.
USAID (U.S. Agency for International Development). 2007. The health systems assessment approach: A how-to manual. Arlington, VA: USAID, Health Systems 20/20, Partners for Health Reformplus, Quality Assurance Project, and Rational Pharmaceutical Management Plus.
USAID. 2009. Guidance on the definition and use of the global health and child survival account. Washington, DC: USAID.
USAID. 2010. Strengthening health systems by engaging the private health sector: Promising HIV/AIDS partnerships. Washington, DC: USAID.
USAID DELIVER Project. 2011. The logistics handbook: A practical guide for the supply chain management of health commodities. Arlington, VA: USAID DELIVER project.
Veenstra, N., and A. Whiteside. 2005. Economic impact of HIV. Best Practice & Research Clinical Obstetrics & Gynaecology 19(2):197-210.
Vujicic, M., S. E. Weber, I. A. Nikolic, R. Atun, and R. Kumar. 2012. An analysis of GAVI, the Global Fund and World Bank support for human resources for health in developing countries. Health Policy and Planning 27(8):649-657.
Wenner, M. 2009. New plan seeks to accelerate African diagnostic capacity. Nature Medicine 15(9):978.
WHO (World Health Organization). 2000. The world health report 2000. Health systems: Improving performance. Geneva: WHO.
WHO. 2001. Report on WHO meeting on the stewardship function in health systems. Geneva: WHO.
WHO. 2004. Developing health management information systems: A practical guide for developing countries. Geneva: WHO.
WHO. 2007a. Everybody’s business: Strengthening health systems to improve health outcomes: WHO’s framework for action. Geneva: WHO.
WHO. 2007b. Towards better leadership and management in health: Report on an international consultation on strengthening and leadership and management in low-income countries. Geneva: WHO.
WHO. 2008. The Maputo Declaration on Strengthening of Laboratory Systems. Presented at Consensus Meeting on Clinical Laboratory Testing Harmonization and Standarization, Maputo, Mozambique.
WHO. 2009. Systems thinking for health systems strengthening. Geneva: WHO.
WHO. 2010a. Antiretroviral therapy for HIV infection in adults and adolescents: Recommendations for a public health approach—2010 rev. Geneva: WHO.
WHO. 2010b. Key components of a well functioning health system. Geneva: WHO.
WHO. 2010c. Monitoring the building blocks of health systems: A handbook of indicators and their measurement strategies. Geneva: WHO.
WHO. 2010d. The WHO global code of practice on the international recruitment of health personnel. Sixty-third World Health Assembly—WHA63.16. Geneva: WHO.
WHO. 2010e. The world health report: health systems financing: The path to universal coverage. Geneva: WHO.
WHO. 2011. The Abuja Declaration: Ten years on. Geneva: WHO.
WHO. 2012a. Country planning cycle database. http://www.nationalplanningcycles.org/home.shtml (accessed August 14, 2012).
WHO. 2012b. Global health expenditure database. http://apps.who.int/nha/database/DataExplorerRegime.aspx (accessed November 2, 2012).
WHO. 2012c. Health systems. Governance. http://www.who.int/healthsystems/topics/stewardship/en/index.html (accessed August 7, 2012).
WHO. 2012d. Health systems. Health service delivery. http://www.who.int/healthsystems/topics/delivery/en/index.html (accessed August 16, 2012).
WHO Secretariat. 2005. Social health insurance: Sustainable health financing, universal coverage, and social health insurance. Report by the Secretariat. Geneva: WHO.
Willis-Shattuck, M., P. Bidwell, S. Thomas, L. Wyness, D. Blaauw, and P. Ditlopo. 2008. Motivation and retention of health workers in developing countries: a systematic review. BMC Health Services Research 8:247.
World Health Assembly. 2005. Sustainable health financing, universal coverage, and social health insurance. World Health Assembly Resolution 58.33. Fifty-Seventh World Health Assembly.
Xu, K., D. B. Evans, G. Carrin, A. M. Aguilar-Rivera, P. Musgrove, and T. Evans. 2007. Protecting households from catastrophic health spending. Health Affairs 26(4):972-983.
Yao, K., B. McKinney, A. Murphy, P. Rotz, W. Wafula, H. Sendagire, S. Okui, and J. N. Nkengasong. 2010. Improving quality management systems of laboratories in developing countries: an innovative training approach to accelerate laboratory accreditation. American Journal of Clinical Pathology 134(3):401-409.
Yu, D., Y. Souteyrand, M. A. Banda, J. Kaufman, and J. H. Perriens. 2008. Investment in HIV/AIDS programs: does it help strengthen health systems in developing countries? Global Health 4:8.
Zachariah, R., N. Ford, M. Philips, S. Lynch, M. Massaquoi, V. Janssens, and A. D. Harries. 2009. Task shifting in HIV/AIDS: opportunities, challenges and proposed actions for sub-Saharan Africa. Transactions of the Royal Society of Tropical Medicine and Hygiene 103(6):549-558.







































































































