3
The Magnitude of the Problem
It is difficult to accurately measure the burden of falsified and substandard drugs. As Chapter 2 mentions, some of the understanding of the problem comes from alerts in gray literature, including investigative journalism and industry and association reporting. Although these sources provide some insight, they do not provide an accurate estimate of the true magnitude of the problem. National regulatory authorities and drug companies keep records on fraudulent medicines; a broader understanding of the problem comes from peer-reviewed literature. There are few epidemiologically rigorous, peer-reviewed studies on the prevalence of falsified and substandard drugs.
This chapter presents the results of a cross section of government and industry data and peer-reviewed and gray literature about the global burden of falsified and substandard drugs. It does not summarize every study, but rather gives an overview of important trends.
Both industry and governments have data on medicines quality, but little of this information is public. There was a time when this was a conscious secrecy, an effort to avoid discrediting the public health system and drug companies, expressed in the Royal Pharmaceutical Society’s 1989 statement “no great publicity [about fake drugs] should be sought because it could damage public confidence in medicines” (Cockburn et al., 2005; More UK debate on counterfeits, 1989). The society has since changed its policy, but the essence of the problem remains. Governments may wish to control rumors that can be seen as damaging to their institutions. Similarly, drug companies, both innovator and generic, may withhold information about falsified and substandard medicines on the grounds that such stories discourage patient confidence in their products (Cockburn et al., 2005).
Key Findings and Conclusions
• Public data on the magnitude of the problem of substandard and falsified medicines are limited.
• The illegal trade and manufacture of medicines are a global problem, disproportionately affecting low- and middle-income countries.
• Countries with weak regulatory oversight and law enforcement attract illegitimate manufacturers, while countries with strict law enforcement repel them.
• Government and intergovernmental agencies, such as the U.S. Food and Drug Administration and Interpol, have taken action against substandard and falsified medicines.
There is a difference between secrecy and appropriate discretion with evidence for pending criminal prosecutions. The committee recognizes that undercover intelligence informs law enforcement agencies’ actions against criminals. Prematurely releasing confidential information about pharmaceutical crime can compromise an investigation. Too often, however, governments and industry withhold information years after incidents pass (Cockburn et al., 2005). Regulators should be able to access this data so that they can communicate it to the public as appropriate, as it informs consumer safety and can trigger epidemiological research on drug quality.
There is also value in sharing information on falsified and substandard medicines internationally. The modern pharmaceutical supply chain is complex. Drug manufacturers source chemicals from around the world, and different factories process ingredients into a final formulation that is packaged, repacked, and sold in many different countries. The chances that a drug quality problem in one country affects that country alone decrease when products travel along global supply chains. The interconnectedness of the drug supply chain makes it imperative that countries share information on falsified and substandard drugs.
The Pharmaceutical Security Institute Incident Reporting System
The Pharmaceutical Security Institute (PSI) is a nonprofit organization composed of the security departments of 25 major pharmaceutical companies
(PSI-Inc., 2012b). These companies share information on illegal pharmaceutical manufacture and trade. Because criminals who make and traffic illegal drugs target a wide range of companies’ products, cooperation and data sharing among companies adds depth to their collective understanding of the problem.
The institute maintains a secure database to which members report cases of fraudulent manufacture and mislabeling of drugs, as well as cases of fraudulent packaging. The database is organized into incidents, “discrete event[s] triggered by the discovery of counterfeit, illegally diverted, or stolen pharmaceuticals” (PSI-Inc., 2012a). A unique tracking number links every incident to a distinct date, time, place, and product. Incidents can vary in size; sometimes, small amounts of a single product are affected, other times large quantities of many products (PSI-Inc., 2012a). Some incidents last for years while others are resolved in 1 year. Incidents that lasted several years are dated with the year in which the incident started (PSI-Inc., 2012a).
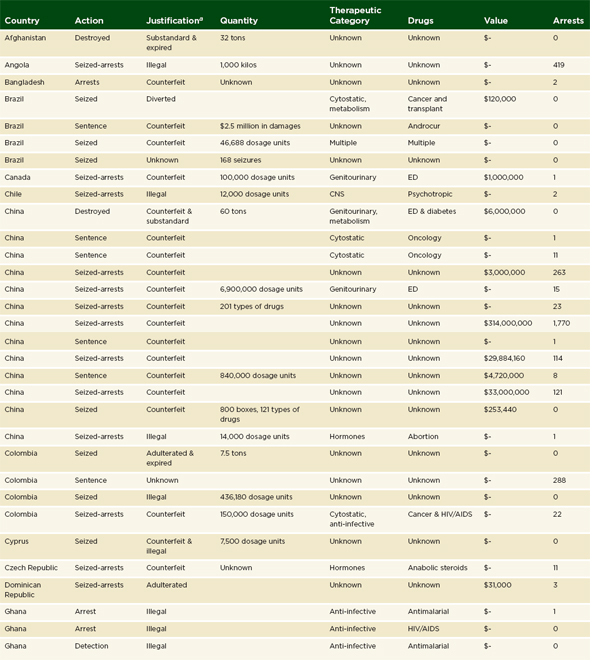
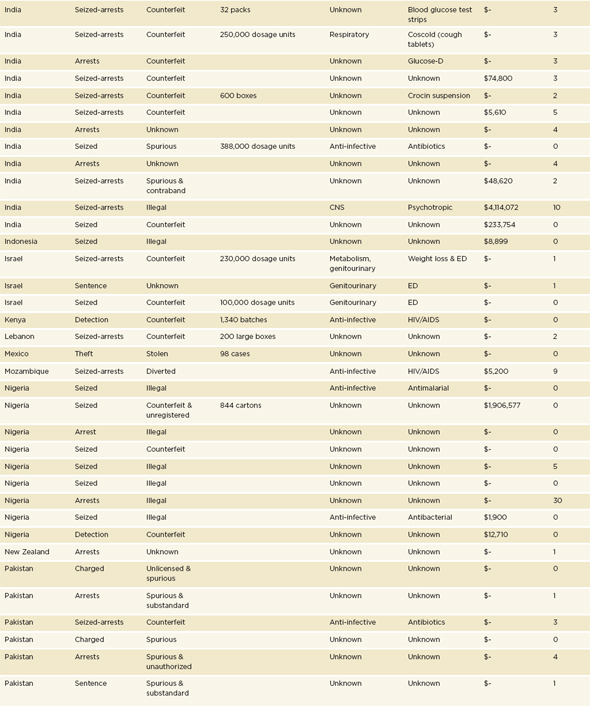
An analysis of the institute’s data gives an understanding of where law enforcement and regulators are active against the illegal trade and manufacture of drugs. Some countries with serious problems never appear in incident reports because there is little political will for action. For the same reason, some countries with transparent and accountable governments consistently appear in the ranking of numbers of seizures by countries (PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012). Table 3-1 presents the 2011 rankings of the top 12 countries where PSI members detected1 or where police, customs, or drug regulators seized falsified products.
The illegal manufacture and trade of medicines is transnational. Table 3-1 shows only those countries where government or industry staff found a bad product; many more countries stand to be affected by those products. If a shipment of falsified pills comes from China to the United States via India, then the incident report names all three countries. Table 3-2 ranks the 10 countries most often cited in PSI incident reports. The countries listed in Table 3-2 account for 56 percent of illegal manufacture, trade, or sale and 47 percent of diversion cases in the PSI 2011 database (personal communication, Thomas Kubic, PSI-Inc., December 26, 2012).
In addition to naming affected countries, every incident report in the PSI database mentions the drugs targeted. Every therapeutic class of drugs is represented in these reports, though genitourinary, anti-infective, and cardiovascular drugs are the most often implicated (PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012). Criminal interest in cardiovascular disease drugs is a new trend; only in 2011 did that class
______________________
1 Detection means confirming though chemical or package analysis that the product is not what it purports to be.
TABLE 3-1 Ranking of Seizures or Detections by Country, 2011
|
|
Country |
Incidents |
|
1 |
China |
279 |
|
2 |
United States |
141 |
|
3 |
Japan |
79 |
|
4 |
Germany |
62 |
|
5 |
Pakistan |
61 |
|
5 |
Peru |
61 |
|
7 |
Colombia |
59 |
|
8 |
United Kingdom |
56 |
|
9 |
South Korea |
47 |
|
10 |
Brazil |
45 |
|
10 |
Russia |
45 |
|
12 |
Taiwan |
44 |
SOURCE: PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012.
of medicines move into the top three most commonly targeted (PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012). This is consistent with other industry reports that drugs sold and restocked frequently are most often targeted (Mukherjee, 2012).
PSI member companies identified 1,623 counterfeiting incidents in 2011. In about half of these incidents (n = 810) companies were able to do product and packaging analysis. Investigators found that most samples were fraudulent in both product and packaging (see Figure 3-1). A false product in legitimate packaging was the second most common result; Chapter 5 discusses this problem in more detail.
Analysis of PSI data supports two main conclusions. First, falsified and substandard drugs are a global problem that affected at least 124 countries in 2011 (PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012). Twelve more countries were affected in 2011 than in 2010; African countries accounted for most of this increase (PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012). The data do not suggest anything about the relative burden of the problem in different countries, however. Indeed, countries with lax enforcement attract illegal manufacturers, and countries with vigorous law enforcement repel them.
TABLE 3-2 Ten Countries Most Named in PSI Incident Reports, 2011
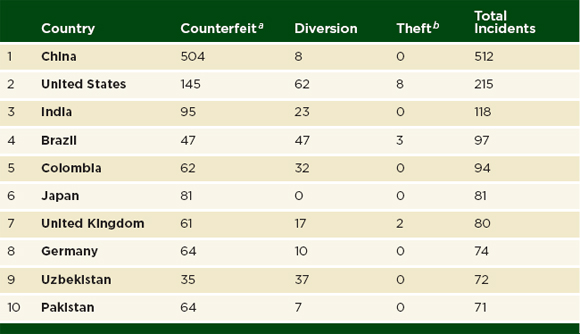
NOTE: PSI = Pharmaceutical Security Institute; WHO = World Health Organization.
aPSI uses the term counterfeit broadly, in accordance with the WHO definition: “one which is deliberately and fraudulently mislabeled with respect to identity and/or source. Counterfeiting can apply to both branded and generic products and counterfeit products may include products with the correct ingredients, wrong ingredients, without active ingredients, with insufficient quantity of active ingredient or with fake packaging” (WHO, 1992, 2009). See page 23.
bIncident reports include thefts worth roughly $100,000 or more.
SOURCE: PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012.
Second, PSI data suggest that the problem is borne disproportionately by low- and middle-income countries. Figure 3-2 shows Business Monitor International’s (BMI’s) global distribution of pharmaceutical sales; Figure 3-3 shows geographic distribution of PSI’s 2011 incidents. Admittedly, the Asia category in both figures includes rich countries such as Australia and Japan (personal communication, Mariam Kahn, Business Monitor International, October 23, 2012). The higher cost of living, higher incomes, and greater access to medicines in North America and Europe also account for these regions’ large share of pharmaceutical sales. Nevertheless, North America and Europe make up almost two-thirds of the world’s combined pharmaceutical sales but account for only a quarter of global trade in illegal medicines. PSI data come from the investigations of multinational, innovator pharmaceutical companies. One would expect a bias in these data to developed country markets, where PSI member companies earn most of their profits. However, even PSI data suggest a serious problem with falsified medicines in low- and middle-income countries.
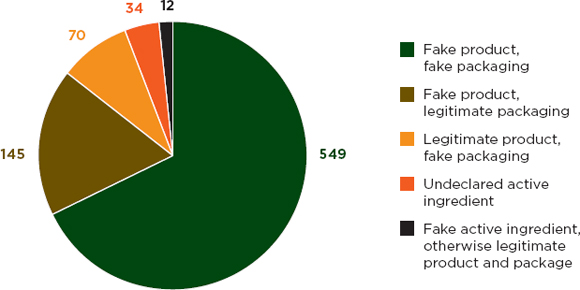
FIGURE 3-1 Results of packaging and product analysis, 2011.
SOURCE: PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012.
Government and Intergovernmental Investigations
National regulatory agencies have the main responsibility for monitoring drug safety (WHO, 2012a). This includes routine postmarket surveillance and enforcement of regulations. Much of their information about falsified and substandard drugs is confidential, but their publications give a sense of the types of violations regulators find.
The FDA Office of Criminal Investigations
The FDA Office of Criminal Investigations takes action against criminal violations of the Food, Drug, and Cosmetic Act, such as illegal drug manufacture, manufacture and sale of unapproved drugs, illegal importation, drug adulteration, and promotion of off-label uses for approved products (FDA, 2009a,b). In May 2012, the office briefed the committee on their work from 2003 to 2008. This presentation and a 2011 review of the agency’s drug criminal cases give some understanding of common problems with the drug supply in the United States (Devine and Jung, 2012; FDA, 2011). These data illustrate certain problems with the drug supply chain but are not a “scientific representation of current … trends or a comprehensive review of problems” (FDA, 2011, p. 4).
The FDA has investigated increasingly more drug quality cases since
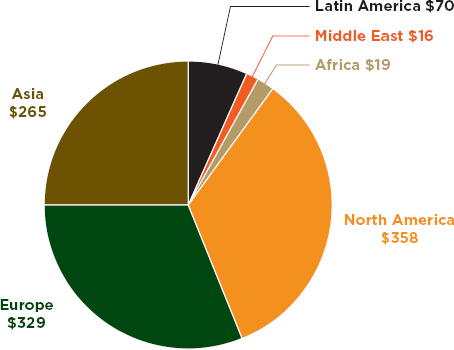
FIGURE 3-2 Geographic distribution of pharmaceutical sales in $U.S. billions, 2011 data.
SOURCE: BMI, 2012.
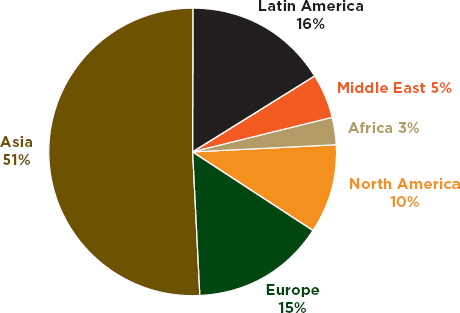
FIGURE 3-3 Geographic distribution of Pharmaceutical Security Institute incident reports, affected countries, 2011 data.
SOURCE: Personal communication, Thomas Kubic, PSI-Inc., October 18, 2012.
19972 (Devine and Jung, 2012). Solid oral dosage forms (pills and tablets) are the most commonly investigated product types (FDA, 2011). Zyprexa, Viagra, Lipitor, Zoloft, and Risperdal are the top 5 brand-name products implicated in the 10 highest-volume cases (FDA, 2011). Problems with individual criminals are more common than problems with negligent businesses; the FDA’s 2003-2008 review found that 86 percent of criminal investigations were of individual suspects, and 14 percent were of companies (FDA, 2011).
International Police Investigations
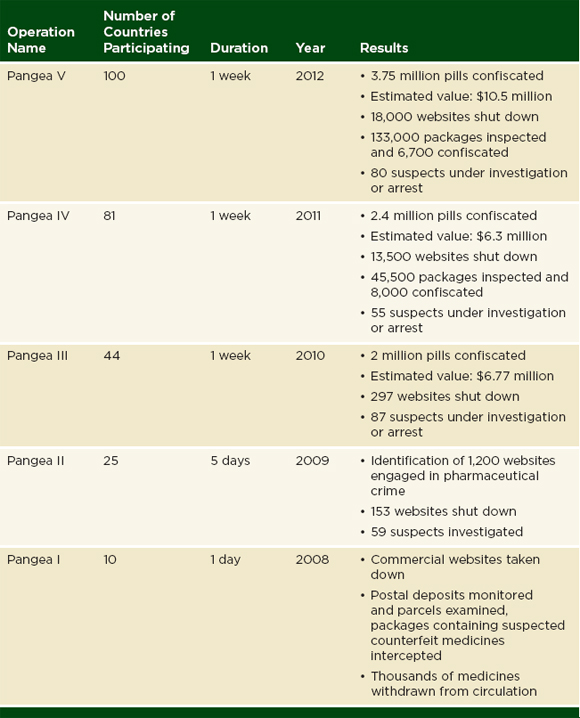
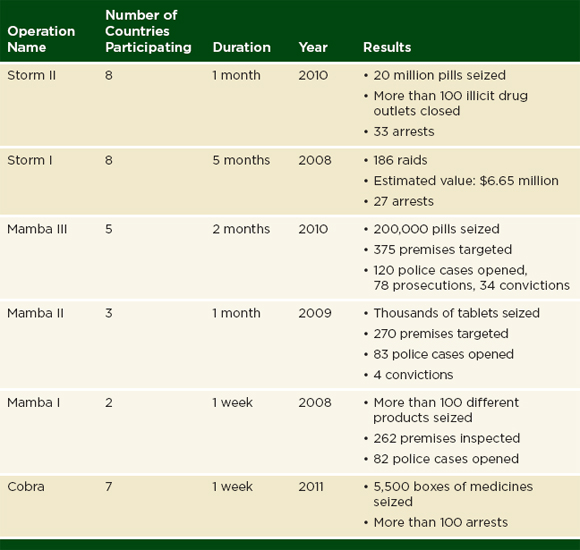
Interpol is an international organization that facilitates police cooperation around the world (Interpol, 2012c). The organization gives training and investigative support to police in 190 member countries (Interpol, 2012c). Pharmaceutical crime, which the organization defines as “counterfeiting and falsification of medical products, their packaging and associated documentation, as well as the theft, fraud, illicit diversion, smuggling, trafficking, [and] the illegal trade of medical products and the money laundering associated with it” has been an Interpol work area since 2005 (Interpol, 2012d; Plançon, 2012). Interpol organizes their work into four operations: Operation Pangea (against illegal online pharmacies), Operation Mamba (in East Africa), Operation Storm (in Southeast Asia), and Operation Cobra (in West Africa) (Interpol, 2012a). Tables 3-3 and 3-4 give an overview of these operations.
As Table 3-3 and 3-4 indicate, police have seized millions of pounds of suspect drugs in Interpol operations. Some police forces sample and report the quality of the products found in these seizures. The police are not obliged to undertake such investigations or report their results to Interpol; in many countries, testing even a small sample of confiscated product would overwhelm the national drug quality laboratory. Interpol does not publish information on the testing and sampling of seized products.
Interpol has raised international awareness about falsified and substandard drugs through their media campaigns and work with police (Interpol, 2011a; Mullard, 2010). Nevertheless, the information presented in Tables 3-3 and 3-4 does not indicate which kinds of drugs are targeted or if the problem is changing over time.
______________________
2 The FDA refers to these as counterfeit drug cases. The agency uses a definition of counterfeit that includes both falsified and substandard as this report defines them. See Table 1-2.
TABLE 3-3 Operation Pangea Against Online Pharmacies, 2008-2012
SOURCE: Adapted from Interpol, 2012b.
TABLE 3-4 Operations Storm, Mamba, and Cobra, 2008-2011
SOURCE: Adapted from Interpol, 2010a,b, 2011b.
CASE REPORTS AND CONVENIENCE SAMPLES
Scientific literature contains valuable reports of drug contamination; often clinicians uncover fake drugs in the course of their practice. Newspapers, court documents, and other gray literature sources also contain valuable information about drug quality lapses. Convenience surveys and case reports can be useful for identifying a problem in particular product lines or building momentum for further research.
The following brief analysis of convenience surveys and case reports indicates that drug quality lapses happen around the world. In countries with strong regulatory systems, the problems are often confined to gray market purchases of the so-called lifestyle drugs, medicines for erectile dysfunction and cosmetic conditions. In poor countries a wide range of products are compromised, including most essential medicines.
Key Findings and Conclusions
• Case studies and postmortem investigations generate interest in substandard and falsified drugs. These reports can drive epidemiological research.
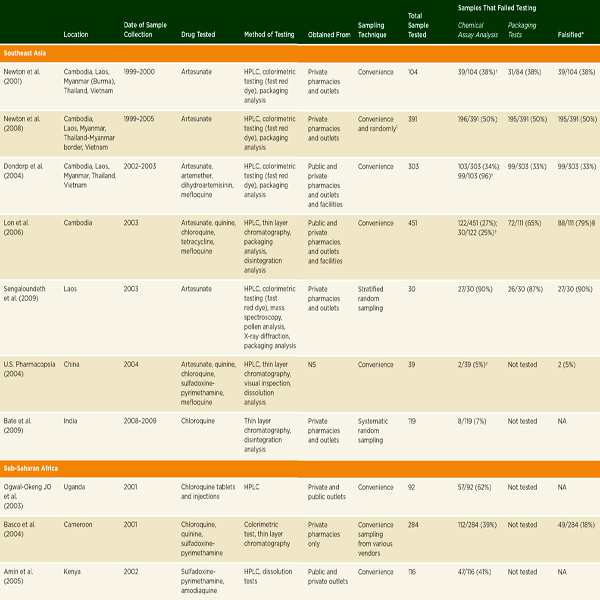
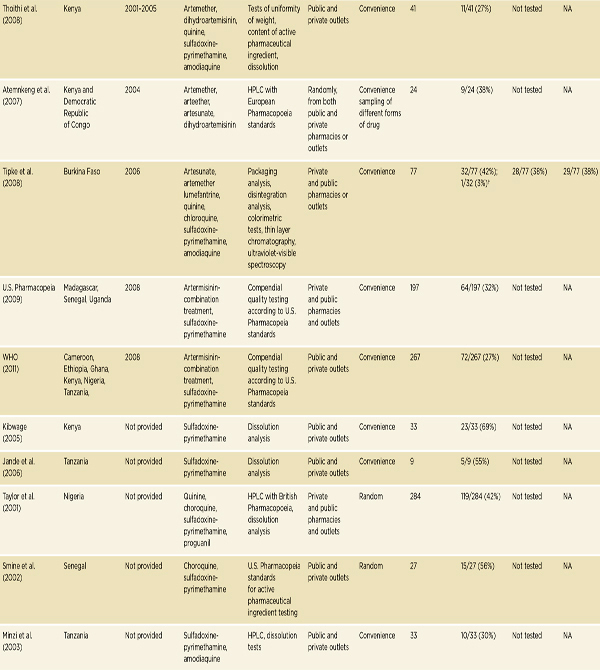
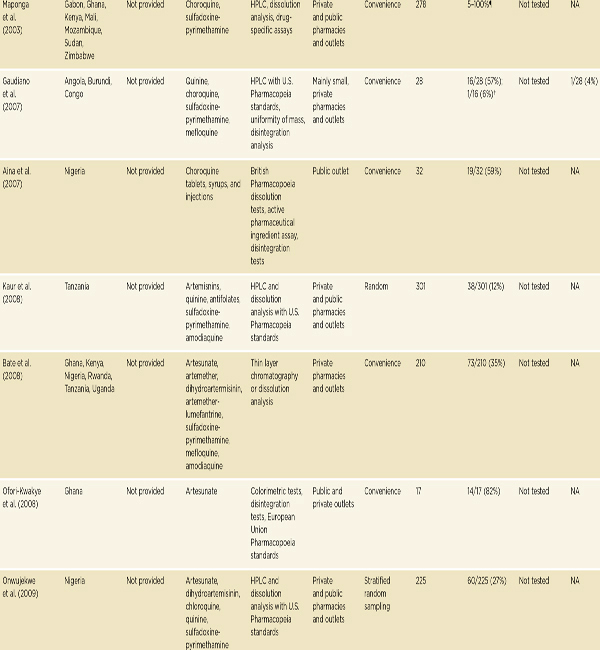
• Convenience surveys suggest serious problems with antimicrobial drug quality in low- and middle-income countries, and especially with antimalarial quality in sub-Saharan Africa and Southeast Asia.
A great deal of literature on falsified and substandard drugs describes problems that emerge only after patients have been harmed (Ravinetto et al., 2012). These reports do not set out to comment on the regional burden of poor drug quality or trends in compromised products, but they are useful for other reasons. Many of these stories receive significant media attention, encouraging public interest in the problem. Case studies also give understanding of the social and environmental conditions that foster problems with falsified and substandard drugs (Pew, 2012).
Patient case studies are a common type of incidental investigation. For example, the rapid deterioration and death of a Burmese patient with uncomplicated malaria triggered a drug analysis that found the medicines used to treat him were both falsified and substandard (Newton et al., 2006). A postmortem investigation in a previously healthy, 58-year-old Canadian woman found that her death was from acute metal poisoning from a variety of falsified and substandard drugs, many of them antianxiety and antidepressive medications she bought from the internet (Solomon, 2007).
Individual deaths can trigger drug quality investigations; mass causalities are clearly more likely to rouse suspicion. Chapter 2 describes one such incident, when a Panamanian physician reported on a spike in cases of acute renal failure, accompanied by neurological dysfunction, abdominal symptoms, urinary irregularities, anorexia, and fatigue (Rentz et al., 2008). A case-control investigation found diethyelene glycol poisoning to be the cause of the outbreak (Rentz et al., 2008). Later investigations, including a Pulitzer Prize–winning New York Times series, implicated falsified ingredients from China in an international poisoning crisis (Bogdanich, 2007; Rentz et al., 2008).
Newspaper reports and other gray literature sources also contain a wealth of information about drug quality problems. Monitoring this literature is a valuable way to follow what drugs are compromised and where. The U.S. Pharmacopeia’s Media Reports on Medicine Quality Focusing on
USAID-Assisted Countries is a useful gray literature compendium (PQM, 2012). Reports are organized by country and medicine affected. The compendium contains links to government and academic surveys as well. The reports presented in the compendium suggest that a range of drugs are compromised in low- and middle-income countries. Antimicrobial drugs are often mentioned, but oral contraceptives, the anti-influenza drug oseltamivir, antihypertensives, antidepressants, blood thinners, and drugs for erectile dysfunction are also frequently named (PQM, 2012). The last section lists international and global incidents, many tied to the internet (PQM, 2012).
The Pharmaceutical Security Institute also monitors gray literature; their open-source database contains publicly available records of the types of medicines compromised, arrests for pharmaceutical crime, and other details staff can glean from public reports (PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012). Table 3-5 summarizes the organization’s 2011 open-source review.
An overview of case studies and gray literature is helpful to understanding falsified and substandard drugs. Gray literature compendiums and peer-reviewed case studies indicate where and in what product lines drug quality problems occur. Such reports raise awareness of the problem and can trigger scientific investigation and convenience sampling. Gray literature reports do not often give details of quality testing of compromised samples, but they generally describe products so grossly and obviously compromised that confirmatory lab testing would be unnecessary.
A convenience sample is a no-probability sample chosen for its accessibility to researchers, not from an a priori sampling frame. Research on drug quality often uses convenience samples of pharmacies or dispensaries. Convenience studies are logistically simpler than probability-based studies and can be less expensive (Newton et al., 2009). Although useful for identifying problems, results of these studies cannot accurately estimate the population prevalence of poor-quality drugs. They do, however, suggest signals for further research (Newton et al., 2009). This section presents the results of some key convenience studies and review papers.
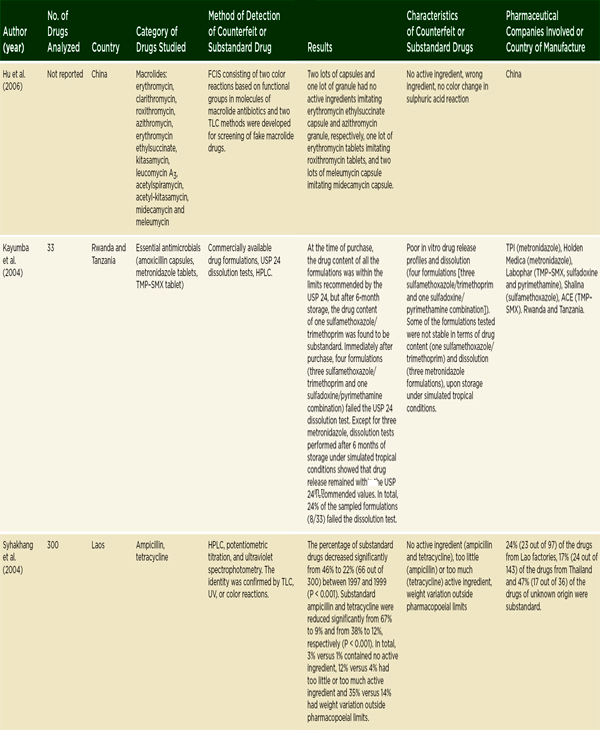
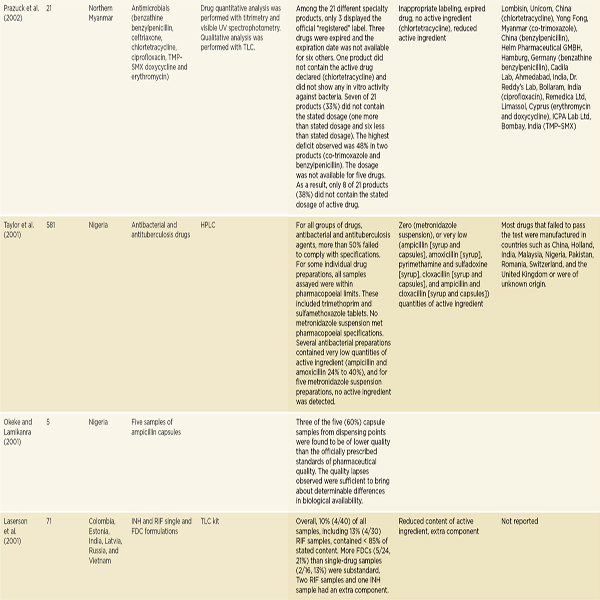
Antimicrobial drugs treat bacterial, viral, fungal, and parasitic diseases. There are considerable data to suggest that antimicrobial drug quality, particularly the quality of antibiotics and antimalarials, is a problem in low-and middle-income countries. In 2007 Kelesidis and colleagues conducted a comprehensive literature review on antimicrobial drug quality, reviewing
gray literature sources as well as English-language papers published between 1966 and 2006 (Kelesidis et al., 2007). They found that a lack of methodological detail prevented pooling or interpreting aggregate results (Kelesidis et al., 2007). As Table 3-6 indicates, they found reason for concern with antibiotic quality in low- and middle-income countries, though reports of poor-quality antibiotics surface all over the world, including the United States and Europe (Kelesidis et al., 2007). A year later, a study of 111 amoxicillin samples collected in four Arab countries found that 56 percent failed U.S. Pharmacopeia testing (Kyriacos et al., 2008). It is difficult, however, to draw firm conclusions about substandard drug production from these studies. Antibiotics degrade quickly in warm climates; it is hard to distinguish substandard manufacture from poor storage and handling.
Weaknesses in drug registration may complicate convenience surveys. When researchers test only authorized products, they bias their sample against the unregistered products used by the poorest (Seear et al., 2011). Some convenience samples have compared the quality of approved and unapproved products. Between 2008 and 2012, Bate and colleagues collected samples of 2,652 anti-infective drugs from around the world: 11 African cities, 3 Indian cities, Bangkok, Beijing, Istanbul, Moscow, and São Paulo. They found that less than one-third of products had stringent-regulatory agency approval or WHO prequalification (Bate et al., 2012). While only 1.01 percent of stringent-regulatory-agency–approved products failed quality testing, 6.80 percent of WHO-approved drugs failed, and 13.01 percent of products approved by neither the WHO nor a stringent regulatory agency failed (Bate et al., 2012). The report mentions that the failure rates were higher among samples from Africa than among samples from middle-income nations (Bate et al., 2012). Similarly, a WHO study in found that only 43 percent of essential anti-infective medicines3 sampled in Vietnam were registered, 20 percent of unregistered products failed pharmacopeial testing, but only 3 percent of registered ones failed (Wondemagegnehu, 1999).
Antimalarials in Southeast Asia and sub-Saharan Africa There is consistent survey evidence that antimalarial drugs, medicines bought around 200 million times per year, mostly for children under the age of 5, are often of poor quality (WHO, 2011b). The demand for these drugs is highest in sub-Saharan Africa and Southeast Asia, which together account for 94 percent of malaria cases (WHO, 2011b). Acute malaria episodes come on quickly and often; antimalarials are bought on short notice from the most
______________________
3 Including, but not limited to, amoxicillin, ampicillin, chloroquine, rifampicin, and co-trimoxazole.
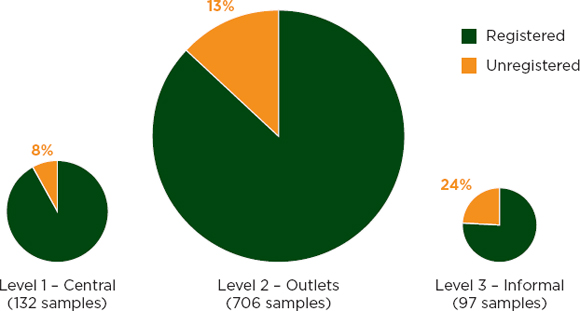
FIGURE 3-4 Registration status of antimalarial samples by distribution level.
SOURCE: WHO, 2011a.
convenient vendor. For these reasons, they are often the target of criminals and unscrupulous manufacturers.
A WHO study of antimalarial drug quality in six African countries4 used a stratified convenience sample (WHO, 2011a). In the spring of 2008, regulatory agency staff in the six countries collected samples from central stores, licensed outlets, and unlicensed markets (WHO, 2011a). Investigators screened all samples in the field and sent a subset for full quality-control testing (WHO, 2011a). Investigators found unregistered medicines least often at the central distribution level (see Figure 3-4). Quality analysis on a subset of products found no evidence that the unregistered drugs were of lower quality than the registered ones (see Figure 3-5) (WHO, 2011a).
Field screening of 893 samples detected ingredient failures in 12 percent (WHO, 2011a). Of the 267 samples selected for full quality-control testing, 28 percent failed (see Figure 3-6) (WHO, 2011a).
The WHO study findings are cause for concern, especially as they sampled heavily from national central medicine stores, the most controlled setting. A recent review paper includes some higher estimates of poor-quality antimalarial drugs (Nayyar et al., 2012). The review included 28 published and unpublished studies, mostly (n = 22) from convenience samples, but also 7 that included some type of randomized design (Nayyar et al., 2012).
______________________
4 Cameroon, Ethiopia, Ghana, Kenya, Nigeria, and Tanzania.
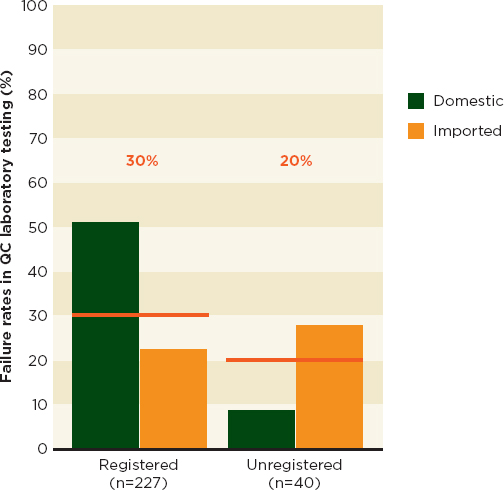
FIGURE 3-5 Quality failures of registered and unregistered antimalarials in 267 samples.
NOTE: QC = quality control.
SOURCE: WHO, 2011a.
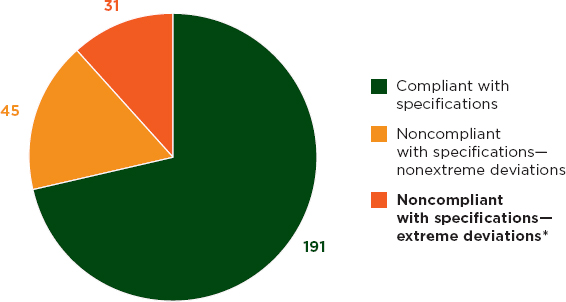
FIGURE 3-6 Results of full quality-control analysis in 267 samples.
*In this survey, extreme deviations were defined as a deviation by at least 20% from the declared content of one or more active ingredients, and/or dissolved percentage of one or more active ingredients less than the pharmacopoeial limit (Q) minus 25%.
SOURCE: WHO, 2011a.
Of 1,437 samples from Southeast Asia, 35 percent (n = 497) failed chemical testing. Of the 497 samples that failed chemical testing, 34 percent had no active ingredient; 4 percent had low active ingredient. In a subset of 919 samples with intact packaging and a verified, genuine packaging sample for comparison, 46 percent failed packaging analysis (Nayyar et al., 2012). Investigators classified all drugs failing packaging analysis as falsified, as well as those substandard drugs that contained no active ingredient or an ingredient not listed on the label (Nayyar et al., 2012). They classified 36 percent of 1,260 eligible samples as falsified.
Nayyar and colleagues used the same criteria to categorize samples from sub-Saharan African countries (Nayyar et al., 2012). Thirty-five percent (n = 796) of 2,297 samples failed chemical analysis. Forty-five percent of the studies reported active ingredient test results, finding that 121 (15 percent) had low active ingredient and 3 percent had excessive active ingredient (Nayyar et al., 2012). Only one study reported packaging analysis, and it found 36 percent failure (Nayyar et al., 2012). Nayyar and colleagues had fewer African samples (n = 389) from which to calculate the percentage of falsified drugs; they found 20 percent falsified (see Table 3-7) (Nayyar et al., 2012).
A consistent problem with all convenience surveys of drug quality is that they tend to sample heavily from the formal market: licensed pharmacies and dispensaries. Results of these studies will likely underestimate the burden of falsified and substandard drugs in places where much of the population buys essential medicines in unregulated bazaars. Sampling from these vendors is difficult, but a convenience sample of informal and private medicine sellers in Guyana and Surinam found 58 percent of the antimalarial samples from Guyana and all the samples from Surinam to be falsified or substandard (Evans et al., 2012). In a Burkina Faso study, Tipke and colleagues compared antimalarial drug quality in licit and illicit vendors. They found that 90 percent of samples from street vendors and open markets were substandard, and only 10 percent of samples from legal vendors were substandard (Tipke et al., 2008).
SYSTEMATIC RANDOM SAMPLES OF DRUG QUALITY
Drug quality is a global problem. Research estimating the precise extent of this problem is hard to find. There are few epidemiologically rigorous surveys of drug quality. This section presents the results of a few population-based random surveys of drug quality.
Kaur and colleagues analyzed antimalarial quality in drugs drawn from a systematic, random sample of a range of Tanzanian retail outlets, including drug stores, general stores, street hawkers, and medicine kiosks
Key Findings and Conclusions
• Few published studies use systematic random samples to estimate the burden of falsified and substandard drugs.
• Those that do indicate serious problems with antimalarial, antibiotic, and antituberculosis drugs in sub-Saharan Africa and Southeast Asia.
• Adherence to the Medicine Quality Assessment Reporting Guidelines would improve understanding of the problem.
(Kaur et al., 2008). Investigators stratified districts according to their participation in a national bed net program, chose districts at random from among the strata, and then surveyed 30 percent of wards in each study district (Kaur et al., 2008). They divided wards into major and nonmajor trading centers and drew half the samples from each type of market (Kaur et al., 2008). Between May and September 2005, investigators collected 1,080 samples from 2,474 vendors, one from each store that had them in stock on the day of the study visit (Kaur et al., 2008). After excluding 166 expired samples and 32 with no labeled expiry date, investigators had 882 samples, from which they systematically chose 301 for chemical analysis (Kaur et al., 2008); 12.2 percent failed quality testing (Kaur et al., 2008).
An older study in West Africa found more widespread quality problems. Taylor and colleagues collected 581 drugs from 35 randomly selected registered pharmacies in urban Nigeria (Taylor et al., 2001). They found 42 percent of antimalarials, 41 percent of antibacterials, and 54 percent of antituberculosis drugs outside of British Pharmacopoeia limits (Taylor et al., 2001). A stratified random sample of medicine shops and licensed pharmacies in Laos found 90 percent of artesunate samples failed quality testing (Sengaloundeth et al., 2009).
Researchers in southeast Nigeria attempted to include unlicensed private medicine dealers in their sample of antimalarial drug quality (Onwujekwe et al., 2009). They collected samples of a range of antimalarials from patent medicine dealers, pharmacies, public and private hospitals, and primary health care centers (Onwujekwe et al., 2009). Thirty-seven percent of drugs tested failed to meet U.S. Pharmacopeia specifications, by either not containing the active ingredient listed or containing it in low doses (Onwujekwe et al., 2009). Among the failed samples, 60 percent came from low-level shops, mostly the patent-medicine shops (Onwujekwe et al., 2009).

Malaria medicine dispensed at a clinic in Sittwe, Burma.
SOURCE: Paula Bronstein/Getty Images.
Though most epidemiologically rigorous research on drug quality has tested antimicrobial drugs, there is some information about other essential medicines. In a 2012 study, Stanton and colleagues prepared an exhaustive sampling frame of formal and informal drug sellers in three districts in Ghana (Stanton et al., 2012). They chose 75 vendors at random from the sampling frame, from which patient actors collected 101 samples of ergometrine and oxytocin, the thermally unstable, uterotonic drugs used to treat postpartum hemorrhage (Stanton et al., 2012). A total of 89 percent of samples failed pharmacopeial testing; none of the ergometrine samples and only 26 percent of oxytocin samples met pharmacopeial specifications (Stanton et al., 2012). All oxytocin samples (n = 46) were from unregistered manufacturers, though 18 were from manufacturers with registration pending; 69 percent of ergometrine samples (n = 38) came from unregistered manufacturers, though 11 were from manufacturers with registration pending (Stanton et al., 2012). All unregistered samples failed quality testing (Stanton et al., 2012).
The best estimates of the scope of the drug supply affected come from systematic, random sampling and testing of medicines drawn from a
representative cross section of the market. Such studies are uncommon (Seear, 2012). The expense of required assays, discussed further in Chapter 6, is one barrier, but a large part of the problem is logistical. The first step in drawing a systematic random sample of drugs is identifying the sampling frame, the list of every drug vendor in a given area. In developed countries, registered pharmacies and dispensaries are the only place most of the population gets medicine. In low- and middle-income countries, however, there is often an extensive pharmaceutical gray market. Identifying all the vendors is difficult and can be further complicated by the blurry lines between licit and illicit commerce (Seear et al., 2011). Health workers may supplement their incomes by selling medicine informally (Peters and Bloom, 2012); peddlers may trade medicines occasionally, along with any number of dry goods, at bazaars and flea markets. Without formative research to catalogue the sampling frame, research on medicines quality is vulnerable to bias.
There is also opportunity for bias in sample collection. Samples should be bought by patient actors, local study staff posing as shoppers who conceal from the vendor that they are working on an epidemiological investigation.
Without taking steps to protect study validity, the researchers risk wasting time and money on a study that does not produce reliable estimates. For example, in 2009 the Indian government conducted a massive survey of drug quality across the country, estimating that only 0.04 percent of drugs are substandard (CDSCO, 2009). Questions about the methodological rigor of the survey, particularly the choice of sampling frame and methods for sample collection, have called these results into question both within India and internationally (Bate, 2009, 2010; Pandeya, 2009).
The committee supports the guidelines on field surveys of medicine quality that Newton and colleagues proposed in March 2009 (Newton et al., 2009). They provide a standard protocol for collecting medicines samples and concrete advice on sampling techniques (Newton et al., 2009). More research adhering to the checklist in Table 3-8 would allow for a better understanding of the burden of falsified and substandard drugs, and it would facilitate valid comparisons of the problem among countries and over time.
The committee believes that more research in adherence with the guidelines put forth in Table 3-8 would give a better understanding of where and to what extent falsified and substandard medicines circulate. There is no substitute, however, for pharmacovigilance and postmarket surveillance. It is not a coincidence that falsified and substandard medicines circulate
Key Findings and Conclusions
• Falsified and substandard medicines circulate in countries where there are not sufficient systems to monitor drug safety and adverse events.
• There are currently no accurate estimates of the global burden of falsified and substandard drugs. This lack of clarity impedes coordinated international action.
• The WHO rapid alert system is a promising program to track falsified and substandard drugs in low- and middle-income countries.
in countries where there are not sufficient systems to monitor drug safety and adverse events. A 2012 IOM report called for greater international investment in low- and middle-income-country surveillance systems (IOM, 2012). The committee agrees with this report, especially the call for technical support for surveillance tools and protocols (IOM, 2012). National surveillance systems should work to detect signals of substandard and falsified drugs. Incorporating pharmacovigilance into the broader public health surveillance system will help ensure the system’s survival.
Recommendation 3-1: Governments should establish or strengthen systems to detect substandard, falsified, and unregistered medicines. This surveillance should be integrated with established public health surveillance systems. Analysis and reporting should precisely describe the product’s quality, packing, and registration.
As Chapter 4 explains, governments can be slow to act against falsified and substandard medicines. In emerging economies, officials may see enforcing drug quality standards as at odds with building the nascent manufacturing sector (IOM, 2012). It is also difficult to promote international effort against a threat as amorphous as fake medicines. Concrete data spur politicians and policy makers to action—information such as the number of doctor’s appointments repeated because of falsified and substandard drugs, the number of hospital beds taken by victims of pharmaceutical crimes, premature deaths from untreated disease, and productive years lost to society from medicine poisoning. Pharmacovigilance is the first step to generating the needed data. When pharmacovigilance systems indicate lack of medicines’ efficacy, these signals should be followed. In-depth investigations can eventually produce data on the specific consequences of falsified and substandard medicines.
TABLE 3-8 Medicine Quality Assessment Reporting Guidelines
|
Section and Topic |
Item |
Description |
|
Title/abstract/keywords |
1 |
• Identify the article as a study of medicine quality • Provide an abstract of what was done and what was found, describing the main survey methods and chemical analysis techniques used |
|
Introduction |
2 |
• Summarize previous relevant drug quality information and describe the drug regulatory environment • State specific objectives |
|
Methods |
||
|
Survey details |
3 |
The timing and location of the survey; when samples collected and when samples analyzed |
|
Definition |
4 |
The definitions of counterfeit, falsified, substandard, and degraded medicines used |
|
Outlets |
5 |
The type, including indices of size (e.g., turnover), of drug outlets sampled |
|
Sampling design |
6 |
• Sampling design and sample size calculation • Type and number of dosage units purchased/outlet • Definition of sampling frame • Question of interest, assumptions, sampling method(s) (including method of randomization if random sampling used) |
|
Samplers |
7 |
Who carried out the sampling and in what guise? What did the collector say in buying the medicines? |
|
Statistical methods |
8 |
Describe the data analysis techniques used |
|
Ethical issues |
9 |
Whether ethical approval was sought and whether the study encountered any ethical issues |
|
Packaging |
10 |
Packaging examination and reference standards |
|
Chemical analysis |
11 |
Chemical analysis and dissolution testing procedures and location(s) of laboratory. Description of validation and reference standards used |
|
Method validation |
12 |
Details of laboratory method validation results, including but not limited to: certificate of analysis for reference standard, within and between run repeatability (RSD% for n = 5–8), detection and quantization limits, accuracy observed for reference samples, linear range for all analytes, sample preparation recovery studies, selectivity. Possibly, validation against a reference method or inter-laboratory study |
|
Section and Topic |
Item |
Description |
|
Blinding |
13 |
Whether chemistry was performed blinded to packaging and vice versa |
|
Results |
||
|
Outlets |
14 |
The details of the outlets actually sampled, class of pharmacy (e.g., public, private for profit, private not for profit, informal, itinerant) |
|
Missing samples |
15 |
The reasons why any outlets chosen for sampling did not furnish a sample. Do these outlets differ systematically from those in which samples were obtained? |
|
Packaging and chemical results |
16 |
• Packaging and chemistry results and their relationship • Details of products sampled—how many, in what drug classes, countries of origin, batch numbers, manufacture and expiry dates • Results for each analysis—packaging, % active ingredient, dissolution • Additional information could be included in supplementary material |
|
Category of poor-quality medicine |
17 |
A clear statement for each medicine sample detected, whether the investigators class it as genuine, counterfeit, substandard, or degraded, with an explanation as to why and whether the medicine was registered with the government in the location(s) sampled |
|
State company and address as given on packaging |
18 |
If the names of companies and addresses are not given, give a reason as to why this information is not provided. |
|
Sharing data with the regulatory authority |
19 |
Whether the data shared with the appropriate regulatory agency |
|
Dissemination |
20 |
Description of any noncovert packaging features that would allow others to detect counterfeit medicines. If publication is not possible, consider disseminating via web-based supplementary material. |
|
Discussion |
||
|
Key Results |
21 |
Summarize key results with reference to study objectives |
|
Limitations |
22 |
Discussion of limitations of study, especially how robust the estimates of prevalence are and how applicable they may be to wider geographical areas. Discuss the direction and extent of any potential bias. |
|
Section and Topic |
Item |
Description |
|
Interpretation |
23 |
An interpretation of the results, in conjunction with prior studies, in relation to public health |
|
Intervention |
24 |
Whether interventions are thought appropriate and, if so, what type |
|
Other information |
||
|
Conflict of interest |
25 |
State any potential conflicts of interest. |
|
Funding |
26 |
Give the source of funding and role of funders in the study. |
NOTE: RSD = relative standard deviation.
SOURCE: Newton et al., 2009.
There is some reason to suspect problems with unregistered medicines in developing countries, but these problems resist detection (Amin et al., 2005). Postmarket surveillance systems, by definition, follow only those products registered and granted market authorization in a given country. The committee believes that unregistered medicines are as important a surveillance target as falsified and substandard ones. Research on the quality of unregistered medicines indicates that they are often of poor quality (Bate et al., 2010; Lon et al., 2006; Stanton et al., 2012; Wondemagegnehu, 1999). Furthermore, drugs for sale in a country where they are not registered have often been trafficked. Chapter 5 will explain why any product that has left the licit chain of custody is suspect.
A complete picture of the magnitude of the problem of poor-quality medicines depends on thorough and novel surveillance. This surveillance should advance systematic investigation of drug quality failures to build evidence for changing policy. The WHO is developing such a system and training surveillance staff in 10 counties on its use. The committee sees great promise in this system for other developing countries.
The WHO Global Capacity-Building Project
There are currently no accurate estimates of the global burden of falsified and substandard drugs. This lack of clarity hinders coordinated international action. Evidence suggests, however, that the problem is most common in low- and middle-income countries. Unscrupulous manufacturers and criminal cartels take advantage of the comparatively weak drug regulatory systems in these countries, knowing that the regulators are

A pharmacy in Macau advertises safe drugs.
SOURCE: Mark Obusan/Getty Images.
poorly equipped for surveillance or enforcement. A recent WHO project attempts to correct this problem by building a “coordinated, continuous, and ongoing global surveillance and monitoring system” for falsified and substandard drugs (WHO, 2012b, p. 11). The committee believes this project is promising for the 10 countries5 participating in the pilot program and, eventually, for the world.
The system makes use of rapid alert forms—Excel spreadsheets with mandatory fields and detailed guidance in dropdown menus (personal communication, Michael Deats, WHO, October 12, 2012). The investigator who finds an illegitimate drug completes the form and sends it to WHO headquarters, copying regional and country offices. At the WHO, the spreadsheet data populate a master database. Receipt of the form triggers a follow-up contact from a WHO investigator, who queries the reporter on the drug’s packaging, registration, and physical and chemical attributes and if the incident might be connected to other criminal activity (personal communication, Michael Deats, WHO, October 12, 2012). Table 3-9 shows more detail about the data collected in the rapid alert form.
______________________
5 Cambodia, Croatia, Georgia, Indonesia, Kyrgyzstan, Malaysia, the Philippines, Russia, Ukraine, and Vietnam.
TABLE 3-9 Information Collected in the WHO Rapid Alert System
|
Rapid Alert |
Details |
|
|
Reporting person |
Contact details |
|
|
Suspect product details |
Full details of all suspect products (up to 30 per report) |
|
|
How suspect product was discovered |
Regulated or unregulated supply chain |
|
|
Product analysis |
Laboratory results on medicine and packaging |
|
|
Photographs |
Photographs of product, packaging, method of concealment |
|
|
Impact on public health |
Record of adverse reactions in patients |
|
|
Action Taken |
Details |
|
|
Communication |
Details of recall or public announcements |
|
|
Dissemination |
Record of those other organizations, member states, or stakeholders informed |
|
|
Investigation |
Details of agencies involved in investigating the case |
|
|
Comments |
Specifically what aroused suspicion concerning the product |
|
NOTE: WHO = World Health Organization.
SOURCE: Personal communication, Michael Deats, WHO, October 12, 2012.
Regulators from the 10 pilot countries testing the rapid alert form and incident investigation system had training on the system in September 2012. The pilot testing ran until December 2012 (personal communication, Michael Deats, WHO, October 12, 2012). Already the system has allowed investigators to link incidents in multiple countries. Eventually, regulators may link falsified and substandard reporting to national pharmacovigilance systems, which would give more depth to information about lack of efficacy (personal communication, Michael Deats, WHO, October 12, 2012).
The lack of consensus on how to define falsified and substandard medicines has held back all public action on the topic, even surveillance.
The WHO project gets around this problem by recording problems with medicines and by not attempting to make the observed problems fit the confusing and contradictory national definitions of substandard, falsely labeled, spurious, counterfeit, etc.
Patient reporting triggers most investigations in the pilot countries. This depends on motivated and knowledgeable patients, and a longer-term improvement to the project might aim to increase reporting from health workers. However, Michael Deats, the WHO technical officer in charge of the program, explained that while he was working as a regulator in Britain, some of his best leads came from patients, especially ones who take medication for chronic disease. One informant was a patient in the habit of rubbing his pill in his hand before taking it and was immediately suspicious when the color rubbed off (personal communication, Michael Deats, WHO, November 11, 2012). A similar signal came from a patient accustomed to cutting his pills in half, who noticed irregular friability when he cut them (personal communication, Michael Deats, WHO, November 11, 2012).
The committee recognizes that building surveillance systems will be challenging in many countries. Nevertheless, taking steps to establish a system or to strengthen the existing system is a reasonable first step in most of the world. Developing countries may benefit from momentum for building surveillance among donor governments and international organizations (IOM, 2012). The WHO is also encouraging action in the same direction. At a meeting in November 2012, representatives of 200 member states agreed to develop instruments to more accurately measure the burden of substandard and falsified drugs (WHO, 2012c). To this end, the WHO capacity-building project is testing and developing surveillance tools specifically for use in low- and middle-income countries. As the pilot project goes on, the regulators and WHO staff may identify revisions that make the protocol more accessible in poor countries.
The WHO monitoring format advances understanding of the scope of the problem without depending on common variable names. The committee sees the WHO rapid alert system as an uncommonly thorough and precise tool for data collection. These data will inform tailored drug quality programs. For example, if the data indicate that substandard medicines are the main drug quality problem in one part of the world, then better regulation of manufacturers can do much to improve the problem. Similarly, if it becomes clear that a country has a problem with diverted medicines in commerce, then some of the distribution chain improvements presented in Chapter 5 would enhance the national drug safety program. Consistent use of this rapid alert form and eventually linking it to national pharmacovigilance systems would advance international discourse and give a more nuanced understanding of the extent and type of falsified, substandard, and unregistered medicines that circulate around the world.
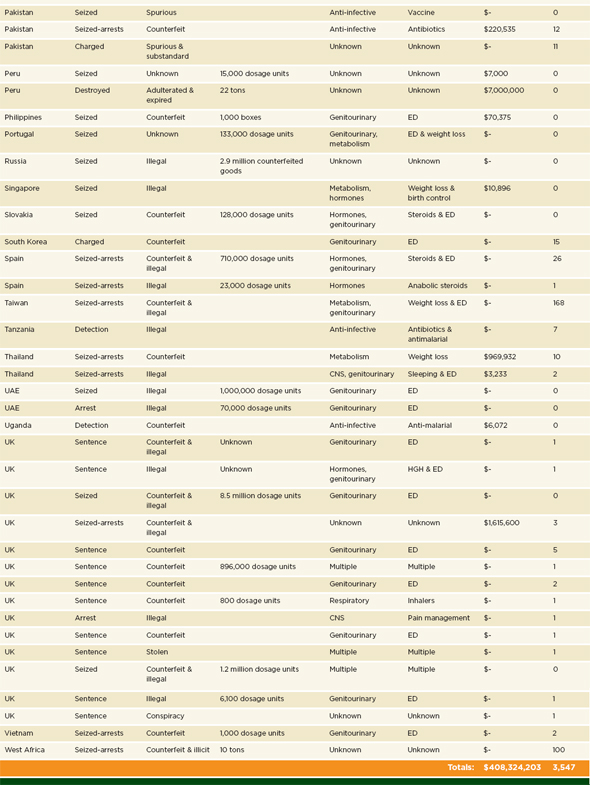
NOTES: $- = value unknown; CNS= central nervous system; ED = erectile dysfunction; HGH= human growth hormone; UAE = United Arab Emirates; UK= United Kingdom.
a As noted in footnote a under Table 3-2, PSI uses the term counterfeit broadly, in accordance with a WHO definition. See page 89. The organization’s open-source review presents each case as it is in the open source.
SOURCE: PSI data shared with the committee, Thomas Kubic, PSI-Inc., July 11, 2012.
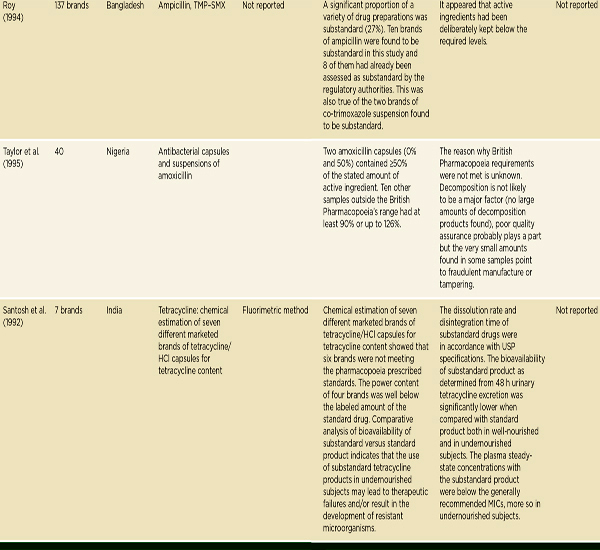
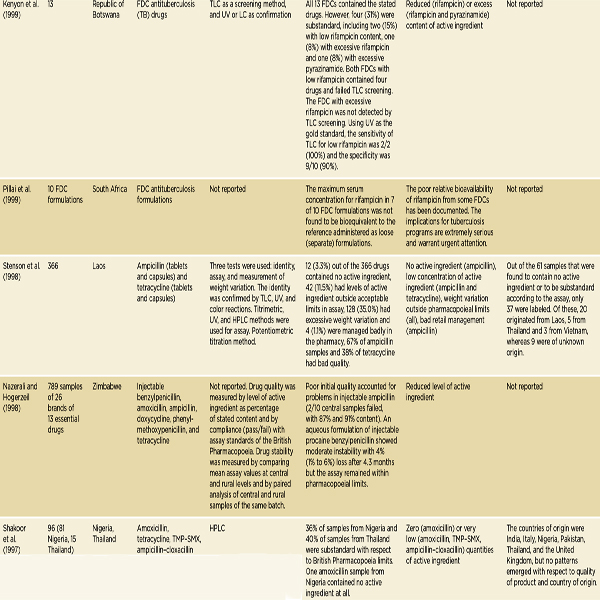
NOTE: The complete references for the studies cited in this table can be found in Kelesidis et al., 2007. Kelesidis and colleagues use the term counterfeit broadly, the way this report uses the term falsified. See page 23. FCIS = fast chemical identification test; FDC = fixed-dose combination; HCI = ondansetron hydrochloride; HPLC = high-performance liquid chromatography; INH = isoniazid; LC = liquid chromatography; MIC = minimum inhibitory concentration; RIF = rifampicin; TLC = thin layer chromatography; TMP-SMX = trimethoprim-sulfamethoxazole; USP = U.S. Pharmacopeia; UV = ultraviolet spectrophotometry.
SOURCE: Kelesidis et al., 2007. Reprinted with permission from Oxford University Press.
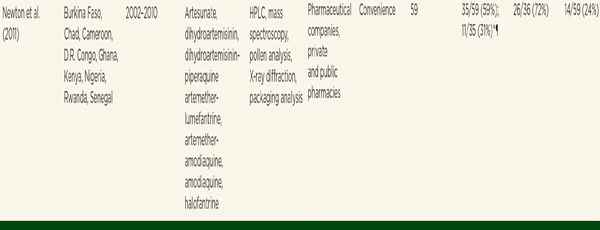
NOTE: DFID = Data are n/N (%), unless otherwise indicated. Samples failing chemical assay analysis might have failed packaging analysis; HPLC = high-perfomance liquid chromatography; NA = not applicable; NS = not specified.
* Falsified is used as a synonym for counterfeit.
† Samples with no active pharmaceutical ingredient.
‡ 115 samples from Laos were randomly selected.
§ Only tetracycline, quinine, and artesunate were tested.
¶ Varies substantially by drug and country; not included in analysis.
SOURCE: Nayyar et al., 2012. Reprinted from the Lancet with permission from Elsevier.
Amin, A. A., R. W. Snow, and G. O. Kokwaro. 2005. The quality of sulphadoxine-pyrimethamine and amodiaquine products in the Kenyan retail sector. Journal of Clinical Pharmacy and Therapeutics 30(6):559-565.
Bate, R. 2009. India’s counterfeit claims on counterfeit drugs. http://www.american.com/archive/2009/september/indias-counterfeit-claims-on-counterfeit-drugs (accessed September 24, 2012).
———. 2010. Delhi’s fake drug whitewash. Wall Street Journal, September 2.
Bate, R., L. M. K. Hess, and J. M. A. Attaran. 2012. Anti-infective medicine quality: Analysis of basic product quality by approval status and country of manufacture. Research and Reports in Tropical Medicine 3:57-61.
Bate, R., L. Mooney, and K. Hess. 2010. Medicine registration and medicine quality: A preliminary analysis of key cities in emerging markets. Research and Reports in Tropical Medicine 1:89-93.
BMI (British Monitor International). 2012. Global pharmaceutical sales. Data available from http://www.citibank.com/transactionservices/home/about_us/online_academy/docs/JDavis.pdf (accessed April 10, 2013).
Bogdanich, W. 2007. F.D.A. tracked poisoned drugs, but trail went cold in China. New York Times, June 17.
CDSCO (Central Drugs Standard Control Organization). 2009. Report on countrywide survey for spurious drugs. New Delhi: CDSCO.
Cockburn, R., P. N. Newton, E. K. Agyarko, D. Akunyili, and N. J. White. 2005. The global threat of counterfeit drugs: Why industry and governments must communicate the dangers. PLoS Medicine 2(4):e100.
Devine, J., and C. Jung. 2012. FDA efforts to build supply chain integrity. Paper presented at Committee on Understanding the Global Public Health Implications of Substandard, Falsified, and Counterfeit Medical Products: Meeting 2, Washington, DC, May 10.
Evans, L., V. Coignez, A. Barojas, D. Bempong, S. Bradby, Y. Dijiba, M. James, G. Bretas, M. Adhin, N. Ceron, A. Hinds-Semple, K. Chibwe, P. Lukulay, and V. Pribluda. 2012. Quality of anti-malarials collected in the private and informal sectors in Guyana and Suriname. Malaria Journal 11(1):203.
FDA (U.S. Food and Drug Administration). 2009a. Inspections, compliance, enforcement, and criminal investigations: Mission. http://www.fda.gov/ICECI/CriminalInvestigations/ucm123027.htm (accessed November 12, 2012).
———. 2009b. Inspections, compliance, enforcement, and criminal investigations: What OCI investigates. http://www.fda.gov/ICECI/CriminalInvestigations/ucm123062.htm (accessed November 12, 2012).
———. 2011. FDA conducts preliminary review of agency’s diversion and counterfeit criminal case information. Washington, DC: FDA Office of Criminal Investigations.
Interpol. 2010a. Operation Mamba. http://www.interpol.int/Crime-areas/Pharmaceutical-crime/Operations/Operation-Mamba (accessed November 12, 2012).
———. 2010b. Operation Storm. http://www.interpol.int/Crime-areas/Pharmaceutical-crime/Operations/Operation-Storm (accessed November 6, 2012).
———. 2011a. Interpol launches global campaign against fake medicines with powerful African voices. http://www.interpol.int/News-and-media/News-media-releases/2011/PR090 (accessed August 20, 2012).
———. 2011b. Operation Cobra. http://www.interpol.int/Crime-areas/Pharmaceutical-crime/Operations/Operation-Cobra (accessed November 12, 2012).
———. 2012a. Operations. http://www.interpol.int/Crime-areas/Pharmaceutical-crime/Operations (accessed November 6, 2012).
———. 2012b. Operations: Operation Pangea. http://www.interpol.int/Crime-areas/Pharmaceutical-crime/Operations/Operation-Pangea (accessed October 22, 2012).
———. 2012c. Overview. http://www.interpol.int/About-INTERPOL/Overview (accessed November 6, 2012).
———. 2012d. Pharmaceutical crime: A major threat to public health. http://www.interpol.int/Crime-areas/Pharmaceutical-crime/Pharmaceutical-crime (accessed November 6, 2012).
IOM (Institute of Medicine). 2012. Ensuring safe foods and medical products through stronger regulatory systems abroad. Washington, DC: The National Academies Press.
Kaur, H., C. Goodman, E. Thompson, K.-A. Thompson, I. Masanja, S. P. Kachur, and S. Abdulla. 2008. A nationwide survey of the quality of antimalarials in retail outlets in Tanzania. PLoS ONE 3(10):e3403.
Kelesidis, T., I. Kelesidis, P. I. Rafailidis, and M. E. Falagas. 2007. Counterfeit or substandard antimicrobial drugs: A review of the scientific evidence. Journal of Antimicrobial Chemotherapy 60(2):214-236.
Kyriacos, S., M. Mroueh, R. P. Chahine, and O. Khouzam. 2008. Quality of amoxicillin formulations in some Arab countries. Journal of Clinical Pharmacy and Therapeutics 33(4):375-379.
Lon, C. T., R. Tsuyuoka, S. Phanouvong, N. Nivanna, D. Socheat, C. Sokhan, N. Blum, E. M. Christophel, and A. Smine. 2006. Counterfeit and substandard antimalarial drugs in Cambodia. Transactions of the Royal Society of Tropical Medicine and Hygiene 100(11):1019-1024.
More UK debate on counterfeits. 1989. Scrip 1468(3).
Mukherjee, R. 2012. Now send SMS to find if pill is genuine. Times of India, September 23.
Mullard, A. 2010. Straight talk with … Aline Plancon. Nature Medicine 16(4):365-365.
Nayyar, G. M. L., J. G. Breman, P. N. Newton, and J. Herrington. 2012. Poor-quality antimalarial drugs in Southeast Asia and sub-Saharan Africa. Lancet Infectious Diseases 12(6):488-496.
Newton, P. N., S. J. Lee, C. Goodman, F. M. Fernández, S. Yeung, S. Phanouvong, H. Kaur, A. A. Amin, C. J. M. Whitty, G. O. Kokwaro, N. Lindegårdh, P. Lukulay, L. J. White, N. P. J. Day, M. D. Green, and N. J. White. 2009. Guidelines for field surveys of the quality of medicines: A proposal. PLoS Medicine 6(3):e1000052.
Newton, P. N., R. McGready, F. Fernandez, M. D. Green, M. Sunjio, C. Bruneton, S. Phanouvong, P. Millet, C. J. M. Whitty, A. O. Talisuna, S. Proux, E. M. Christophel, G. Malenga, P. Singhasivanon, K. Bojang, H. Kaur, K. Palmer, N. P. J. Day, B. M. Greenwood, F. Nosten, and N. J. White. 2006. Manslaughter by fake artesunate in Asia: Will Africa be next? PLoS Medicine 3(6):e197.
Onwujekwe, O., H. Kaur, N. Dike, E. Shu, B. Uzochukwu, K. Hanson, V. Okoye, and P. Okonkwo. 2009. Quality of anti-malarial drugs provided by public and private healthcare providers in south-east Nigeria. Malaria Journal 8(22). DOI: 10.1186/1475-2875-8-22.
Pandeya, R. 2009. Supply of fake drugs grossly overstated. http://www.livemint.com/Home-Page/rW90cqXqBIBdhqsoEG6ISK/Supply-of-fake-drugs-grossly-overstated.html (accessed October 2, 2012).
Peters, D. H., and G. Bloom. 2012. Developing world: Bring order to unregulated health markets. Nature 487(7406):163-165.
Pew. 2012. Case studies: How unsafe drugs can reach patients. Pew Health Group. http://www.pewhealth.org/reports-analysis/issue-briefs/case-studies-how-unsafe-drugs-can-reach-patients-85899388662 (accessed March 5, 2013).
Plançon, A. 2012. Presentation to the committee on understanding the global public health implications of substandard, falsified, and counterfeit medical products: Meeting 1, March 13.
PQM (Promoting the Quality of Medicines in Developing Countries). 2012. Media reports on medicine quality: Focusing on USAID-assisted countries. Rockville, MD: U.S. Pharmacopeia.
PSI-Inc. (Pharmaceutical Security Institute). 2012a. Counterfeit situation. http://www.psi-inc.org/counterfeitSituation.cfm (accessed April 25, 2012).
———. 2012b. Pharmaceutical Security Institute: Home. http://www.psi-inc.org/index.cfm (accessed October 16, 2012).
Ravinetto, R. M., M. Boelaert, J. Jacobs, C. Pouget, and C. Luyckx. 2012. Poor-quality medical products: Time to address substandards, not only counterfeits. Tropical Medicine &International Health 17(11):1412-1416.
Rentz, E. D., L. Lewis, O. J. Mujica, D. B. Barr, J. G. Schier, G. Weeraskera, P. Kuklenyik, M. McGeehin, J. Osterloh, J. Wamsley, W. Lum, C. Alleyne, N. Sosa, J. Motta, and C. Rubin. 2008. Outbreak of acute renal failure in Panama in 2006: A case-control study. Bulletin of the World Health Organization 86(10):737-816.
Seear, M. 2012. Pharmaceutical quality: An urgent and unresolved issue. Lancet Infectious Diseases 12(6):428-429.
Seear, M., D. Gandhi, R. Carr, A. Dayal, D. Raghavan, and N. Sharma. 2011. The need for better data about counterfeit drugs in developing countries: A proposed standard research methodology tested in Chennai, India. Journal of Clinical Pharmacy and Therapeutics 36:488-495.
Sengaloundeth, S., M. D. Green, F. M. Fernández, O. Manolin, K. Phommavong, V. Insixiengmay, C. Y. Hampton, L. Nyadong, D. C. Mildenhall, D. Hostetler, L. Khounsaknalath, L. Vongsack, S. Phompida, V. Vanisaveth, L. Syhakhang, and P. N. Newton. 2009. A stratified random survey of the proportion of poor quality oral artesunate sold at medicine outlets in the Lao PDR: Implications for therapeutic failure and drug resistance. Malaria Journal 8(172). DOI: 10.1186/1475-2875-8-172.
Solomon, S. 2007. BC woman killed by fake drugs bought online. National Review of Medicine, July 30.
Stanton, C., A. Koski, P. Cofie, E. Mirzabagi, B. L. Grady, and S. Brooke. 2012. Uterotonic drug quality: An assessment of the potency of injectable uterotonic drugs purchased by simulated clients in three districts in Ghana. BMJ Open 2(3):1-7.
Taylor, R. B., O. Shakoor, R. H. Behrens, M. Everard, A. S. Low, J. Wangboonskut, R. G. Reid, and J. A. Kolawole. 2001. Pharmacopoeial quality of drugs supplied by Nigerian pharmacies. Lancet 357:1933-1936.
Tipke, M., S. Diallo, B. Coulibaly, D. Störzinger, T. Hoppe-Tichy, A. Sie, and O. Müller. 2008. Substandard anti-malarial drugs in Burkina Faso. Malaria Journal 7(95). DOI: 10.1186/1475-2875-7-95.
WHO (World Health Organization). 1992. Counterfeit drugs: Report of a WHO/IFPMA workshop 1-3 April 1992. Geneva: WHO.
———. 2009. Medicines: Frequently asked questions. Geneva: WHO.
———. 2011a. Survey of the quality of selected antimalarial medicines circulating in six countries of sub-Saharan Africa. Geneva: WHO.
———. 2011b. World malaria report 2011: Summary and key points. Geneva: WHO.
———. 2012a. Immunization standards: National regulatory authorities. http://www.who.int/immunization_standards/national_regulatory_authorities/role/en/index.html (accessed November 6, 2012).
———. 2012b. Information leaflet for National Medicines Regulatory Authorities (NMRAs): Building global capactiy for surveillance and monitoring of “substandard/spurious/ falsely-labelled/falsified/counterfeit” medicines. Geneva: WHO.
———. 2012c. New global mechanism to combat substandard/spurious/falsely-labelled/ falsified/counterfeit medical products. Geneva: WHO.
Wondemagegnehu, E. 1999. Counterfeit and substandard drugs in Myanmar and Viet Nam: Report of a study carried out in cooperation with the governments of Myanmar and Viet Nam. Geneva: WHO.