Experiences with MDR TB in Other Countries
Key Messagesa
• An intensive program of treatment for MDR TB in prisons in Tomsk Oblast cured more than 80 percent of cases, and mortality rates in Tomsk are now half those in the rest of Russia.
• A community-based treatment program in KwaZulu-Natal province cured more than half of MDR TB patients despite major weaknesses in the health care system.
• A community-based model with a hospital/health center component and a home-based component cured more than 70 percent of MDR TB cases in Cambodia and was so successful that it has been replicated in Ethiopia.
__________________
a Identified by individual speakers.
Three speakers at the workshop provided descriptions of experiences in addressing MDR TB in countries other than China. (Specific issues are discussed in detail in the following chapters in the context of pediatric populations, infection control, rapid diagnosis, treatment across the spectrum of drug resistance, and SLDs.) In all the countries discussed—Cambodia, Ethiopia, Russia, and South Africa—intensive treatment programs based on rapid diagnosis and tailored regimens produced impressive cure rates while overcoming serious obstacles to the delivery of care.
MDR TB IN THE RUSSIAN FEDERATION1
Russia has a very complex health care system. Following the collapse of the Soviet Union, the poverty rate in the Russian Federation rose dramatically. Depending on the measure used, somewhere between 35 and 60 percent of the population was living in poverty.
As unemployment rose throughout the 1990s, rates of TB rose as well, from a low of 34 per 100,000 in 1991 to a high of 90 per 100,000 in 2000. Russia also shares with the United States the distinction of having some of the highest rates of incarceration in the world, at close to 700 per 100,000 citizens, and the rate of incarceration in a country tracks with its TB incidence (Stuckler et al., 2008).
The cure rates from WHO’s DOTS approach in Russia were very low, observed Salmaan Keshavjee, Harvard Medical School. Instead of the 90+ percent that one would expect in a population of people where drugs were effective, cure rates were in the 60s, and as time went on, the cure rates for the DOTS regimen continued to decline. At the same time, the rate of MDR TB increased—from 6.7 percent in 1999 to 14.4 percent in 2010 among new cases of TB and from 10 percent to 30 percent among all cases of TB.2
The spread of MDR TB in Russia had several causes. The prevalence of drug resistance was already high; MDR TB was spreading in the air; HIV infections were increasing; incarceration rates were high; and patients were being treated in hospitals, leading to nosocomial transmission, especially during the intensive phase of treatment. In addition, the pharmaceutical supply systems had broken down, so drug supplies were limited, and drug regimens were inadequate; outpatient systems for observing therapy and managing side effects were lacking as well. More generally, this was happening in an environment characterized by considerable substance abuse, weakened family structures, and people feeling isolated from their communities and surroundings.
In 1998, Partners In Health began working in Tomsk Oblast at the invitation of the Russian Ministry of Justice and the Open Society Institute, which had a program to combat TB in the oblast’s prisons. Tomsk is in western Siberia and has a population of about 1 million spread over a
__________________
1 This section is based on the presentation by Salmaan Keshavjee, Director, Program in Infectious Disease and Social Change, Department of Global Health and Social Medicine, Harvard Medical School.
2 At the time that DOTS became a global program, international advisors were advising the Russians not to treat MDR TB. In 1996, WHO’s Groups at Risk wrote, “MDR TB is too expensive to treat in poor countries; it detracts attention and resources from treating drugsusceptible disease.” Later, it also was claimed that “best-practice short-course chemotherapy might even reduce the incidence of MDR TB where it had already become endemic” (Dye et al., 2002).
large area. The incidence of TB in the Tomsk prison sector rose substantially in the 1990s, to a high of more than 7,000 per 100,000. The overall prevalence in prisons was more than 20,000 per 100,000. The percentage of MDR TB among new cases was 28 percent and among retreatment cases was 54 percent.
In the nonincarcerated sector, TB rates also had risen, to more than 100 per 100,000. And for all of Tomsk Oblast—including the prison population, the nonincarcerated population, new cases, and retreatment cases—about 41 percent of TB patients had MDR TB.
Many of these patients were resistant not just to ethambutol, isoniazid, pyrazinamide, rifampicin, and streptomycin but also to SLDs used for TB and other diseases. According to the Tomsk Oblast Tuberculosis Services, 49 percent were resistant to kanamycin, 10 percent to capreomycin, and 54 percent to ethionamide, potentially as a result of cross-resistance with isoniazid. Levels of resistance to cycloserine and fluoroquinolones were low. As a result of these resistance patterns, standardized regimens were not sufficient; individual resistance results were necessary to treat patients properly.
Partners In Health worked with the local TB laboratory services and the Massachusetts State Laboratory Institute to ensure that every patient underwent high-quality DST so that the strain with which they were infected could be characterized and their therapy tailored. While the drug susceptibility tests were pending, patients were placed on an empirical seven-drug regimen. When the test results came back, they were used to tailor an appropriate five-drug regimen. Patients took these drugs twice per day for 18 to 24 months. Almost all experienced side effects, which were managed within the program. Patients also had severe comorbidities, such as diabetes and alcoholism, that could worsen the tolerance of medication. Managing side effects is particularly important, Keshavjee emphasized, because it can greatly reduce the rates of default from a treatment program.
Among the first cohort of 244 patients, 45 percent had spent time in prison. Forty-two percent had body mass indexes (BMIs) lower than 18.5. About 50 percent had problems of substance abuse. And two-thirds had cavitary and bilateral disease, so their disease was advanced.
The Partners In Health and Tomsk physicians, assisted early on by the British humanitarian agency MERLIN and later by the Russian Red Cross, worked closely together, focused on ensuring that every patient finished treatment and had the best possible outcome by seeking programmatic solutions to overcome barriers to care. Solutions included improvements to facilities to strengthen infection control, transportation assistance for patients and health workers, a greater choice of treatment sites, and the provision of food assistance. For example, patients could receive both treatment and food at TB hospitals, outpatient hospitals, TB dispensaries, rural
TB offices, and doctors’ clinics, as well as during home visits. Patients also received social assistance, such as psychological support, help with government services, clothing in the winter, and help with job searches. These incentives for patients were part of a comprehensive package of medical and social assistance.
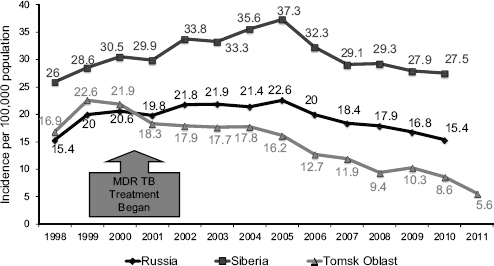
Among the prisoners in the first cohort of patients, the cure rate for MDR TB cases was approximately 81 percent, with 12 percent defaulting, 4 percent failing treatment, and 3 percent dying. TB mortality in the Tomsk prison system declined from 384 per 100,000 in 1999 to essentially zero, and it has remained very low (Figure 5-1). For the prison and nonincarcerated population combined, the cure rate was 77 percent, with approximately 5 percent dying. Even among the 29 XDR TB cases in the first cohort, the cure rate was about 48 percent.
In the second cohort, however, the cure rates were not as high, and the default rates were higher. Analysis revealed that rapid expansion of the program had not been matched by building the infrastructure necessary to deal with patients who needed extra levels of care. The solution was to develop a system of patient accompaniment focused on providing care for those at higher risk of default—a “sputnik” (or “fellow traveler”) in Russian. A nurse was trained to look after five to seven patients, with responsibility for ascertaining how to get these patients to adhere to a
SOURCE: Keshavjee, 2013. Presentation at the IOM workshop on the Global Crisis of Drug-Resistant Tuberculosis and the Leadership of the BRICS Countries: Challenges and Opportunities.
treatment regimen. Patients who defaulted on their treatment were recommended to the sputnik program. This program combined a personal connection to patients, in that nurses would know where and how to reach them, with rigorous management of adverse events, so that patients were comfortable taking their medicines. More important, it shifted the burden of nonadherence from the patient to the program, calling for sound programmatic interventions.
Among the 51 patients who entered the sputnik program between December 2006 and November 2008, adherence was 80 percent for 5 patients who restarted a new treatment course and rose from 52 to 81 percent for the 46 patients who were continuing previous treatment. The cure rate was 64 percent, with 17 percent defaulting. An additional 7.5 percent who were transferred out because of arrests were eventually cured outside of the program. Keshavjee characterized these results as a “reasonably good” cure rate. The cost of the program was somewhere between $500 and $600 a year—“a good value for a program that prevents further transmission of drug-resistant strains of TB and death.”
Among the lessons learned from experiences in Tomsk is that transmission can be interrupted, but ways must be found to deliver care to all patients. Tomsk received support from the Global Fund in 2004, which made it possible to treat all of the patients with MDR TB. That same year, MDR TB cases as a proportion of all TB in Tomsk started declining. Building up delivery mechanisms in the ambulatory sector—both facility- and home-based treatment—allowed large numbers of patients to be treated.
TB prevalence in Tomsk has fallen below the overall rates for the Russian Federation, and mortality rates are now one-third of what they are in the rest of the country. Keshavjee emphasized, in conclusion, that programs to treat DR TB can benefit TB programs in general and should not be seen as competing with those programs.
COMMUNITY-BASED CARE IN SOUTH AFRICA3
Among the 11 districts in KwaZulu-Natal province, the rate of XDR TB in the district that includes Tugela Ferry was 10 times the rate in any other district, with more than 50 percent of MDR TB cases being XDR, said Kristina Wallengren, K-RITH, University of KwaZulu-Natal, and THINK. Furthermore, the prevalence of MDR was very high, at 30 MDR TB cases per 100,000 population, with the actual number likely to be at least twice as large because of sampling deficiencies.
__________________
3 This section is based on the presentation by Kristina Wallengren, Clinical Advisor, K-RITH, Nelson R. Mandela School of Medicine, University of KwaZulu-Natal; and Chief Executive Officer, THINK.
In 2007, the only MDR TB treatment available in the province was through King George V Hospital, which had 160 beds. Treatment guidelines at the time called for patients to be hospitalized during the 6 months of intensive treatment. With at least 3,000 MDR TB cases in the province every year, capacity was obviously far too low. The hospital increased the turnover of patients, causing more patients to be discharged from the hospital while they were still infectious.
Once discharged, patients were treated in their own communities, in clinics where health care providers had no training in MDR TB treatment or its side effects. In addition, patients were expected to return to the central hospital once a month to collect their medication for the 2 years of total treatment, at times from distances of several hundred kilometers.
Conditions in both central and local hospitals were suboptimal for the burden. There were no isolation facilities, so MDR and XDR TB patients in the hospitals were kept in the same wards, and N95 respirators were not worn.
Under the circumstances, community-based treatment was the only option. The South African Department of Health was wary of having MDR and XDR TB patients treated in their homes because of the possibility of household transmission. However, without a feasible alternative and with the acknowledgment of WHO, the South African Department of Health decided to pilot community-based treatment in four pilot sites in KwaZulu-Natal. The program, carried out with no external funding, was based on the Partners In Health experience in Peru. Each of the four sites had a local hospital with a few beds dedicated to MDR TB cases where patients stayed only if and as long as necessary to stabilize treatment. Other patients started their MDR TB treatment without spending a single day in the hospital.
The best combination, Wallengren said, was to have patients spend 1 or 2 weeks in the hospital. During this time, social workers could visit their homes to see if they had a stable food source and an environment conducive to supporting MDR TB treatment. This period also provided an opportunity to educate patients and their families about the importance of adhering to treatment and about how to respond to side effects.
During the 6 months of intensive treatment, an injection team consisting of a driver and a nurse visited the homes of patients 5 days per week to give the injectable medication kanamycin. Patients who lived close to the clinic went there daily to receive their injections. Patients saw their doctors monthly to review the continued treatment.
In an evaluation of the community-based versus the hospital-based treatment, data from 1,549 MDR TB patients who were followed for 2 years showed that about 51 percent of the patients in the four community-based sites had been cured, compared with 34 percent in the centralized, hospital-based setting. Patients in the community also culture converted much earlier
than patients in the hospital-based setting. More patients in the centralized hospital had previous TB or MDR TB as compared with the decentralized cohort, and the latter patients were more likely to be smear positive at diagnosis. Fifty-four percent of patients in the hospital-based setting had a successful outcome compared with 60 percent in the decentralized sites.4 However, there was great variation across the four community-based sites, among which successful treatment outcomes ranged from 52 to 72 percent. Other predictors of success were being female; being older than 30; and, for the more than 70 percent of patients in the study who were HIV positive, being on antiretroviral medications.
A second evaluation looked at the performance of the health care system and how it affected the outcomes of MDR TB treatment (Loveday et al., 2013). The evaluation examined the context, the intervention, the mechanism, and the output for health system delivery. According to the evaluation, the four pilot decentralized sites showed substantial differences. The cure rates at the four sites ranged from 46 to 62 percent, and death and default rates also differed significantly. Health outcomes closely tracked the performance scores of the four sites. The factors affecting these differences included ownership of the MDR TB problem at both the district and facility levels, the stability of service (including how long doctors and nurses remained at a site), the integration of services such as TB care and HIV treatment, the availability of drugs, and the quality of care.
Wallengren grounded these statistics in the experiences of a representative patient. During some months the patient was able to receive complete treatment, but in other months treatment was compromised by such factors as bad weather, stock-outs of drugs, strikes of health care workers, and lost medical records (Figure 5-2). In such cases, “it’s not the patients who are unwilling to take the treatment or defaulting,” said Wallengren. “It’s the health system that is failing them.”
Wallengren ended on a positive note by observing that in the best-performing pilot site for community-based treatment, treatment success was achieved in more than 70 percent of cases, with 62 percent being cured, in a population where the HIV positive rate was high. She termed these results “phenomenal.” Furthermore, the program has been so successful that it has been expanded to six sites in KwaZulu-Natal, and Wallengren and her colleagues have been developing training materials so the program can be rolled out much more widely.
__________________
4 Not all patients with a successful treatment outcome could be deemed a “treatment success.” Treatment success is defined as having completed treatment and being identified as cured based on five consecutive culture-negative samples collected at least 30 days apart in the final 12 months of treatment (WHO, 2008).
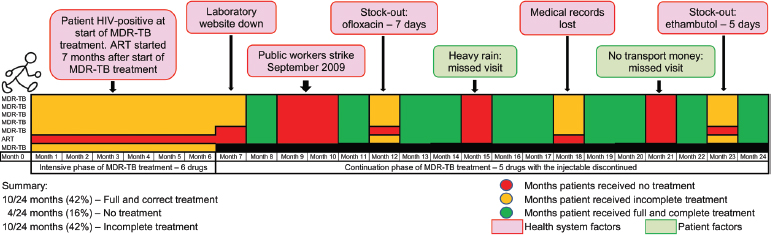
FIGURE 5-2 Many factors have compromised MDR TB treatment for a typical patient in KwaZulu-Natal province.
NOTE: ART, antiretroviral treatment.
SOURCE: Loveday et al., 2013. Reprinted with permission of the International Union Against Tuberculosis and Lung Disease. Copyright © The Union.
DIRECT COLLABORATION IN CAMBODIA AND ETHIOPIA5
After the Cambodian genocide of the 1970s, more than a million refugees fled to refugee camps on the Thai-Cambodian border. Because of the social disruption within the country, there had been no TB treatment since before 1975, and there was no TB program in place among the emergency interventions implemented in the refugee camps on the Thai-Cambodian border. In the 1980s, a TB program was developed by the American Refugee Committee, which used commonsense interventions such as providing food to patients and making use of patient supporters, often family members, who could accompany patients through therapy. This program served as the first demonstration of treatment of TB patients in a war zone and was one of the earliest demonstrations of successful DOTS.6 By the time the border camps closed down in 1993, about 10,000 people had been cured.
In 1994, drawing on the success of the earlier program, the Cambodian Health Committee developed a novel community-based approach to TB and later to AIDS treatment, said Anne E. Goldfeld, Professor of Medicine, Harvard Medical School; and Co-Founder, Global Health Committee (GHC) (a nongovernmental organization [NGO] that has worked in Cambodia as the Cambodian Health Committee since 1994 and is based in the United States at Harvard Medical School). Working in one of the poorest parts of Cambodia, where TB prevalence was an estimated 700 cases per 100,000, the program had cured more than 32,000 people through December 2012. Its community DOTS approach has been scaled up to the entire country of 15 million people and is a major reason why new cases have plummeted in Cambodia—recently recognized by WHO as a global achievement.
As part of this program, the Cambodian Health Committee developed an MDR TB treatment program in 2006 based on the same community-based strategies, with a strong emphasis on case finding. The program uses a community-based model with hospital/health center and home-based components. Training, national guidelines, monitoring of the quality of services, active case finding, and strengthening of the national system are other components of the program. About one-quarter of the 282 patients began treatment at home, while the others were hospitalized for at least 1 month.
__________________
5 This section is based on the presentation by Anne E. Goldfeld, Professor of Medicine, Harvard Medical School; and Co-Founder, Global Health Committee (GHC).
6 DOTS is a WHO TB control strategy used widely throughout the world. The five elements of DOTS are (1) political commitment with increased and sustained financing; (2) case detection through quality-assured bacteriology; (3) standardized treatment, with supervision and patient support; (4) an effective drug supply and management system; and (5) monitoring and evaluation system and impact measurement. For more information, see http://www.who.int/tb/dots/en (accessed August 16, 2013).
For those initiating at home, treatment supporters and health center nurses came to patients’ homes to give injections on a daily basis, and combined teams from the Cambodian Health Committee and Ministry of Health made monthly home visits for follow-up, management of side effects, and monitoring. Of the MDR TB patients treated from 2006 through 2010, 70 percent were cured or completed therapy, and less than 8 and 20 percent defaulted or died, respectively.
In 2008, the Cambodian Health Committee began a program in Ethiopia, which, like Cambodia, is mostly rural and very poor, has been affected by conflict, and has a high TB burden. A 2008 survey estimated that about 6,000 new MDR TB cases appeared each year in Ethiopia, yet no program was in place to treat these patients. Training for the Ethiopian MDR TB program took place both in Ethiopia and in Cambodia in 2008, but the program initially was limited because of a lack of availability of SLDs, the absence of an isolation ward at the centralized pulmonary hospital, human resource limitations, only partial laboratory testing, and no outpatient system. The first cohort of patients was admitted to St. Peter’s Hospital in February 2009, in an isolation ward that was being reserved for bird flu and was completely empty. Supplies of SLDs made available through private donations from the Jolie-Pitt Foundation and through a drug donation from Eli Lilly and Company (“Eli Lilly”) were used until previously promised SLDs from the Global Drug Facility (GDF) gradually arrived over the course of that year. By the end of 2010, a pilot for outpatient treatment was in place, the program had been expanded to northern Ethiopia, and almost 160 patients were being treated for MDR TB.
As of the time of the workshop, in January 2013, 631 patients had been initiated on MDR TB therapy, 107 of whom had been cured or completed treatment, with another 446 on active treatment. Fifty-eight patients had died, and just 10 had had their treatment interrupted or defaulted.
The key to success in Ethiopia, as in Cambodia, has been providing multisectoral support to help patients, said Goldfeld. For outpatients and patients being treated in the community, intensive monitoring, monthly home visits, nutritional support, and social support, including a transportation allowance for returning to the hospital or clinic, are all part of the comprehensive package.
The expansion of the program from Cambodia to Ethiopia represents a south-to-south transfer of an integrated hospital-based and community-based treatment program. It demonstrates that rapid scale-up of MDR TB treatment is possible and effective, said Goldfeld. However, the program continues to face challenges, including a lack of laboratory capacity; the need for funding from international donors; and XDR TB, which remains very expensive to treat in Ethiopia.










