3
Indexing Value in Medicare: The Role of Geographic Area Performance
An important part of the committee’s statement of task focuses on “whether Medicare payment systems should be modified to provide incentives for high-value, high-quality, evidence-based, patient-centered care through adoption of a value index (based on measures of quality and cost) that would adjust payments on a geographic area basis.” As described in Chapter 1, the committee defined health care value as the equivalent of net benefit: the amount by which overall health benefit and/ or well-being produced by care exceeds (or falls short of) the costs of producing it. Here “health benefit and/or well-being” and “costs” are assessed as health outcomes and Medicare and other payer spending for goods and services, respectively. To decide whether to recommend Medicare adjustment of provider and hospital payments based on this definition of value, the committee had to define the scope and evaluate the conceptual and empirical dimensions of a geographically based value index.
DEFINING A GEOGRAPHICALLY BASED VALUE INDEX
Value indexes can take specific forms and serve many purposes. In general, a value index is a relative measure of value—for example, a measure of improvement in patient-centered, clinical health outcomes per unit of resources used in one area relative to the national average. In health care, indexes can be used to adjust hospital or provider reimbursement rates based on measures of relative performance. For example, the Centers for Medicare & Medicaid Services’ (CMS’s) hospital value-based purchasing program and physician payment modifier (authorized under Sections
3001 and 3007 of the Patient Protection and Affordable Care Act [ACA], respectively) adjust hospital and provider payments according to observed hospital and individual provider performance compared with national averages (CMS, 2013a,b).
This chapter focuses primarily on a geographically based value index. Section 1159 of the Affordable Health Care for America Act (H.R. 3962), on which the committee’s charge is based, asked the Institute of Medicine (IOM) to consider different value indexes, including a value index that would adjust provider payments based on regional composite measures of quality and cost. Such an index would be designed to encourage high-value care by tying provider reimbursements to the indexed performance of an area.
In earlier legislation, lawmakers in the U.S. House of Representatives and U.S. Senate introduced separate bills (H.R. 2844 and S. 1249, respectively), both entitled the Medicare Payment Improvement Act of 2009. It proposed that a geographically based value index replace the physician work component of the geographic practice cost indexes, which help standardize differences in resource costs (physician work, practice expenses, and malpractice insurance) across geographic areas (CMS, 2010).1 The geographically based value index, as a relative value ratio, would adjust a portion of provider reimbursements under the Medicare Physician Fee Schedule according to a region’s average Medicare Part A and B spending and health care quality for the Medicare population. The bills’ sponsors hoped that, by “linking rewards to the outcomes for an entire payment area,” providers within a given area would coordinate care, thereby improving health care quality and reducing inefficiencies.2
With this historical context in mind, the committee limited its evaluation of a geographically based value index to a relative ratio that would use area-level composite measures of clinical health outcomes and cost to adjust individual hospital and provider payments under Medicare Parts A, B, and D (a geographic value index).
CONCEPTUAL ASSESSMENT OF A GEOGRAPHIC VALUE INDEX
The U.S. health care “system” is fragmented at the national, state, community, and practice levels. Rather than networks of interrelated components it comprises a wide range of smaller health care systems and solo
_________________
1As described in Chapter 1, the IOM produced the report Geographic Adjustment in Medicare Payment—Phase 1: Improving Accuracy, which provides recommendations for improving the accuracy of Medicare’s current geographic adjustment factors. Detailed descriptions of the geographic practice cost indexes and the hospital wage index are included in that report.
2Health Care Reform, CR S6491, 111th Cong., Congressional Record 155, no. 87, daily ed. (June 11, 2009).
practitioners operating independently. These systems span diverse populations, providers, and geographic areas, increasing the complexity of the overarching health care system.
Within the U.S. health care system, health care decision making generally occurs at the level of the individual practitioner or organization, such as a hospital or physician group (IOM, 2001, 2010), not at the geographic region level.3 Payments that target these actors are more likely than those targeting geographic regions to trigger behavioral change because providers will be accountable for the value of health care services delivered (McKethan et al., 2009). Whether a geographic value index is an appropriate policy depends on whether payment modifications pursuant to the payment model effectively shift provider behavior toward greater efficiency (i.e., using fewer resources) without substantially diminishing health care outcomes.
A geographic value index does not target an appropriate level of clinical decision making to trigger behavioral change at the patient-provider level. In fact, a geographic value index is not designed to target any level of actual decision making. Rather, it treats all providers in a geographic area alike, assuming that area-level payment modifications will incentivize the various decision makers within an area to coordinate care and improve efficiencies across the area. However, two practical considerations suggest otherwise.
First, collaboration among competing providers, absent clinical and financial integration, may raise antitrust issues (Kass and Linehan, 2012). Second, payment modifications targeting large areas do not always link individual physician behaviors to spending increases or decreases. Consequently, a physician (or physician group practice) that reduces volume sees not a proportional increase in payment but reduced income (MedPAC, 2007). For example, the sustainable growth rate (SGR) system, which is designed to decrease Medicare Part B spending each year automatically if Medicare expenditures exceed Medicare spending targets in the previous year (and vice versa), has not incentivized providers to constrain spending growth. Rather, overall spending has increased annually since 2003 (Hahn and Mulvey, 2011).4 Moreover, Medicare’s current fee-for-service reimbursement structure allows providers to maintain reimbursement levels despite cuts to individual-service payments by increasing the volume of services provided.
Although setting provider payments by region, as under a geographic
_________________
3Public health measures, such as educational programs, may be directed at the geographic region level. However, such interventions typically are not covered under Medicare and are the domain of public health agencies, such as the Centers for Disease Control and Prevention and state and county health departments (Salinsky, 2010).
4The SGR system is an imperfect example because Congress has never allowed payment decreases to take effect when Medicare’s actual total expenditures have exceeded targets.
value index, would be more targeted than the current SGR system, it raises similar concerns about altering provider behavior. Regions large enough to have year-to-year stability in spending (e.g., hospital referral regions [HRRs], metropolitan core-based statistical areas [CBSAs]) are in most cases still too large for any individual provider to have enough influence over total expenditures to alter provider behavior patterns (MedPAC, 2007). An exception is when a single delivery system dominates care in an area. In that limited case, payment policies targeting geographically based government units are functionally equivalent to targeting the relevant decision-making unit. However, this scenario does not pertain to the vast majority of the country. Thus, the likelihood that geographically based payments would incentivize all providers in a geographic region to work together to improve value in the region without centralized decision making is low.
In recent years, multiple stakeholders (e.g., payers, providers, employers local governments) have formed region- or community-based collaboratives focused on improving the value of health care for their populations.5 In some cases, health care payments may appropriately target these collaboratives. Like accountable care organizations and other integrated organizations, these collaboratives vary in size and organizational structure, and the area they serve may or may not comport with traditional geographic units. Moreover, collaboratives emerge episodically as a result of local initiatives and leadership and are not necessarily tied to central budgets within their communities. Thus, it is unreasonable for long-term payment policies promoting high-value care to use geographic areas as a shortcut for distributing financial resources to health care decision makers within these collaboratives.
Conclusion 3.1. A geographically based value index is unlikely to promote more efficient behaviors among individual providers and thus is unlikely to improve the overall value of health care.
EMPIRICAL ASSESSMENT OF A GEOGRAPHIC VALUE INDEX
A geographic value index for Medicare would have to generate hospital and provider payments perceived as fair. Proponents of a geographic value index argue that paying more (per unit of service or in total) to providers in areas that are better stewards of health care resources and less to providers in regions that are poor stewards is fair policy. Given the fragmented
_________________
5These collaboratives may implement a range of initiatives, including but not limited to improvement in data collection and dissemination, efficient promotion of health service delivery, and provision of financial incentives for high-value care (Alliance for Health Reform, 2013).
structure of health care delivery within the United States, however, area-level payments are fair only under certain conditions. First, all hospitals and providers within an area must be equally deserving of reward (or penalty), implying they behave similarly. Second, assuming that all providers behave similarly, performance levels in high-value areas must be achievable in low-value areas through elimination of inefficiencies. In other words, differences in measured value between low- and high-spending areas cannot include differences stemming from underlying health status and other acceptable sources of variation. As described fully in Chapter 2, the committee commissioned original analyses to test these premises empirically. Relevant results are discussed below.
Variation in Health Care Spending by Unit of Analysis
To determine whether provider organizations within an identified area behave similarly, the committee examined patterns of health care spending across subregions, service types, and clinical condition categories, as well as condition-specific quality measures across HRRs. As noted above, if providers do not behave similarly, a fairness problem arises whereby low-value providers are rewarded simply by virtue of practicing in areas that are on average high-value (the reverse is also true). Starting with HRRs, the committee examined the amount of variation within progressively smaller units of analysis (hospital services area [HSA], hospital, practice, and individual provider levels).
Spending Variation at the Hospital Service Area Level Within Hospital Referral Regions
The committee investigated the extent and range of variation in spending in subregions within HRRs to test whether the HRR is an appropriate geographic unit upon which to base provider payment. HSAs are nested within HRRs, with an average of 11 per HRR, although there is considerable variability (a range of 1 to 76 HSAs per HRR).6 As one measure of variability within an HRR, the committee examined the ratio of the highest-spending to the lowest-spending HSA within each HRR for HRRs that contain at least three HSAs. At the median (50th percentile) of these ratios, the highest-spending HSA spends 24 percent more than the lowest-spending HSA in the same HRR (Acumen, LLC, 2013, p. 41). In the 76 HRRs with the highest ratios (those above the 75th percentile), the highest-spending
_________________
6Personal communication, Jonathan S. Skinner, Ph.D., Dartmouth Institute for Health Policy and Clinical Practice, Geisel School of Medicine, February 12, 2013.
HSA within each HRR spends at least 36 percent more than the lowest-spending HSA within that HRR.
An analogous assessment decomposed the variance across all HSAs as the sum of variation among HSAs within an HRR and among HRRs. In analyses for the committee, University of Pittsburgh investigators found that about 59 percent of the variation in adjusted HSA Medicare drug spending is within HRRs, compared with 41 percent among HRRs (University of Pittsburgh, 2013, p. 13). Looking at adjusted drug spending, for example, Manhattan (New York) is one of the highest-spending HRRs, while Albuquerque (New Mexico) is one of the lowest-spending, yet the lowest-spending HSA in Manhattan spends less than 25 percent of what is spent by the HSAs within Albuquerque. In addition to heterogeneity in spending within HRRs, there is substantial heterogeneity in utilization patterns.
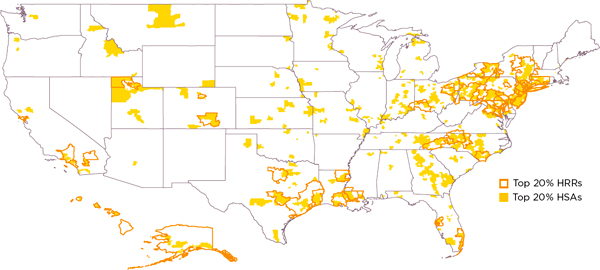
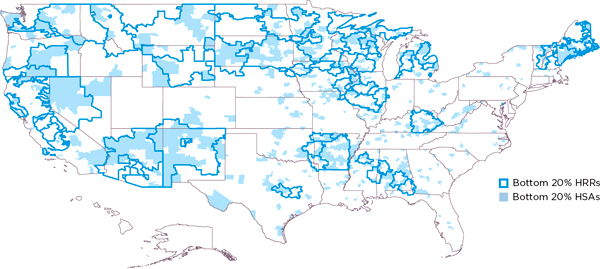
To illustrate local variation in drug spending and utilization, Pittsburgh investigators calculated that about half of the HSAs located within the borders of HRRs in the highest-drug-spending HRR quintile are not in the highest-drug-spending quintile of HSAs, and approximately half of the HSAs in the lowest-drug-spending quintile of HRRs are not in the lowest-drug-spending quintile of HSAs. Figures 3-1 and 3-2 illustrate this quintile analysis for adjusted drug spending for HRRs and HSAs. The light pink and light blue shaded areas lying outside the heavy lines are, respectively, high- and low-drug-spending HSAs that are not in high- or low-drug-spending HRRs. Conversely, the nonshaded areas within the heavy lines are, respectively, not high- and not low-drug-spending HSAs that lie within high- and low-drug-spending HRRs. In sum, these maps show remarkable misalignment of high- and low-drug-spending HSAs and HRRs.
To quantify further the magnitude of variation occurring at geographic levels smaller than HRRs, Precision Health Economics, LLC (PHE) conducted regression analyses to identify the proportion of HRR-level variation in mean spending and utilization that is attributable to HSAs. As Table 3-1 demonstrates, 45 percent of all HRR-level variation in total Medicare spending occurs among HSAs within HRRs. Overall, PHE found that approximately 40 to 70 percent of variation in spending and utilization remained after controlling for HRR characteristics. Collectively, these findings demonstrate considerable variation in spending and utilization that can be explained not by HRR-level factors but by factors at the smaller, HSA geographic level or even below that level—within HSAs.
Spending Variation at the Hospital Level Within Hospital Referral Regions
Hospitals within the same HRR vary substantially in their resource use, as can be seen from the committee’s analysis of Dartmouth data on
TABLE 3-1
Hospital Referral Region Variation in Selecteda Components of Medicare Spending Attributable to Hospital Services Areas (HSAs)
| Spending Component | Proportion of Variation Due to HSAs | ||
| Total Spending | 0.45 | ||
| Input-Price-Adjusted Spending | 0.41 | ||
| Inpatient Admissions | 0.62 | ||
| Outpatient Visits | 0.52 | ||
| Prescription Fills | 0.56 | ||
| Emergency Department Visit Days | 0.70 | ||
| Imaging Encounters | 0.53 | ||
NOTE: Input-price-adjusted spending controls for age, age-sex interaction, partial-year enrollment, year, and health status. Findings are based on a random effects model; results are similar with a fixed-effects model.
a The committee was limited in the number of utilization measures it could investigate across Medicare and commercial databases due to time and budget constraints. Hence, post acute care was not included in the committee’s main analysis. PHE’s analyses reflected those limitations.
SOURCE: Developed by the committee based on data from Precision Health Economics Medicare analysis.
variation in hospital spending for cohorts of patients treated for three major conditions—stroke, hip fracture, and heart attack.7 Variation among hospitals exists in both lower- and higher-spending HRRs, meaning there are high-spending hospitals in low-spending regions and low-spending hospitals in high-spending regions. Figures 3-3, 3-4, and 3-5, respectively, display observed variation in Medicare spending at the hospital level within each HRR for the above three clinical conditions, after adjustment for input price and health status. The higher the point in the figure, the greater is the variation among hospitals in the HRR. For example, referencing the right-most point in Figure 3-3, in the HRR that spends approximately $45,000 per stroke patient, the difference between spending for hospitals in the 75th and 25th percentiles is around 17 percent of the median value. Differences between hospitals at more extreme points, such as the 90th and 10th percentiles, would, of course, be even larger. In short, Figures 3-3 through 3-5 demonstrate that hospitals within HRRs do not tend to be uniformly high- or low-cost.
_________________
7This analysis was limited to HRRs with four or more hospitals with data on spending for a given condition.
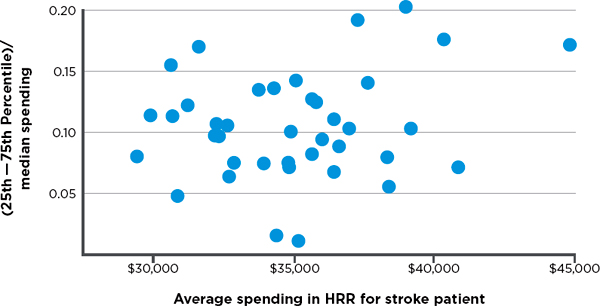
FIGURE 3-3 Variation in price- and risk-adjusted Medicare spending for stroke in a hospital referral region.
SOURCE: Committee analysis of unpublished Dartmouth data (personal communication, Jonathan S. Skinner, Ph.D., Dartmouth Institute for Health Policy and Clinical Practice, Geisel School of Medicine, February 6, 2013).
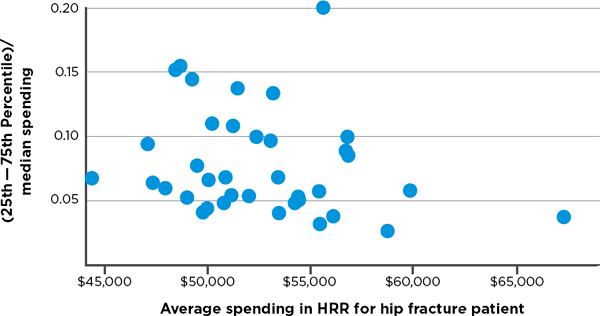
FIGURE 3-4 Variation in price- and risk-adjusted Medicare spending for hip fracture in a hospital referral region.
SOURCE: Committee analysis of unpublished Dartmouth data (personal communication, Jonathan S. Skinner, Ph.D., Dartmouth Institute for Health Policy and Clinical Practice, Geisel School of Medicine, February 6, 2013).
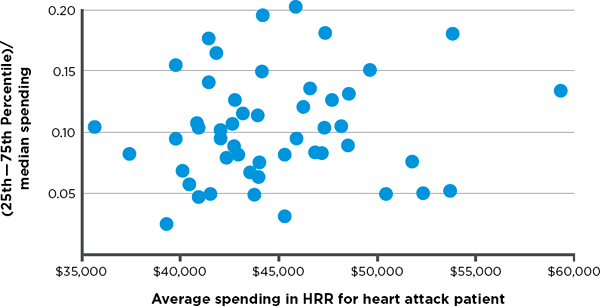
FIGURE 3-5 Variation in price- and risk-adjusted Medicare spending for heart attack in a hospital referral region.
SOURCE: Committee analysis of unpublished Dartmouth data (personal communication, Jonathan S. Skinner, Ph.D., Dartmouth Institute for Health Policy and Clinical Practice, Geisel School of Medicine, February 6, 2013).
Spending Variation Within Provider Practices
Variation also is found among physicians within the same specialty (e.g., cardiology) (Cherkin et al., 1994; Lucas et al., 2010; MedPAC, 2009). The committee could not examine variation below the hospital level (i.e., at the provider level) in its analyses of Medicare data because of restrictions included in data use agreements. Fortunately, commercial insurance data were available. For more than two dozen clinical conditions, Blue Cross Blue Shield of Massachusetts (BCBSMA) regularly examines variation in practice patterns among Massachusetts physicians within particular specialties, comparing physicians with peers in the same practice as well as all comparable specialists across the state. BCBSMA applies episode treatment groups to establish patient populations with defined clinical conditions and then identifies one or two salient differences among physicians in their treatment of those patients. For example, one measure is the tendency of primary care physicians to refer patients with a new episode of knee pain to an orthopedic surgeon. Another is the rate at which cardiologists prescribe angiotensin-converting enzyme (ACE) inhibitors versus more expensive, branded angiotensin receptor blockers for patients with simple hypertension. For each condition, these data indicate that variation among specialists who work in the same group practice is as great as that among
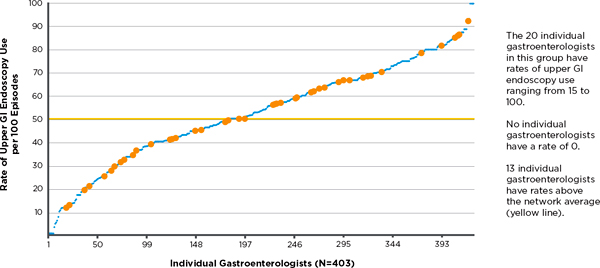
FIGURE 3-6 Use of upper gastrointestinal (GI) endoscopy among gastroenterologists treating gastroesophageal reflux disease.
NOTE: Inflammation of the esophagus, without surgery, Group X, 2009. Rate = episodes with upper GI endoscopy/total episode treatment groups.
SOURCE: Personal communication, Dana Gelb Safran, Blue Cross Blue Shield of Massachusetts, July 17, 2011.
specialists across the entire state.8 For example, Figure 3-6 shows almost as much variation in the use of upper gastrointestinal (GI) endoscopy for patients with gastroesophageal reflux disorder seen by a gastroenterologist (a major driver of spending in that specialty) among 20 physicians within a single practice (denoted by the yellow triangles) as exists for all gastroenterologists in the state (denoted by the blue dots).9
Spending Variation at the Individual Provider Level Across Clinical Conditions
Even individual physician performance varies across different measures of performance. A study by Partners HealthCare of six primary care physicians (PCPs) within the same practice group found that individual levels of utilization and quality varied across nine distinct measures associated with diabetes, cholesterol, and hypertension control; ordering of radiology tests and generic prescriptions; and rates of admissions and emergency department visits (Partners HealthCare, 2012). No single physician was high or low across all measures; instead, each was above average for some and below average for others. Similar analyses have been generated for more than 1,100 PCPs and many specialty groups within the Partners Health System. These data demonstrate the difficulty of classifying individual physicians as high- or low-value providers using composite measures. The committee was not provided with standard errors for these analyses, and some of the variation observed is random. Nonetheless, this variation at the physician level suggests that variation among providers within HSAs may be substantial, so the foregoing estimates of variation within HRRs attributable to variation among HSAs are a lower bound on variation among all providers within an HRR. In short, it is highly unlikely that all physicians within an HSA practice similarly. As a result, area-level performance calculations would likely mischaracterize the actual value of services delivered by many providers and hospitals, resulting in unfair payments.
Conclusion 3.2. Substantial variation in spending and utilization remains as units of analysis get progressively smaller.
_________________
8Personal communication, Dana Gelb Safran, Senior Vice President, Blue Cross Blue Shield of Massachusetts, July 17, 2011.
9It should be noted that the patients are likely somewhat heterogeneous, so that some of this variation is attributable to differences in the nature of patients seen by each physician rather than the physician’s style of practice. Nonetheless, the variation is, in all likelihood, substantially larger than what patient-level factors could explain when averaged across all patients seen by a physician.
Consistency of Health Care Quality Within an Area
The delivery of health care has become increasingly specialized. Although claims-based quality measures are sparse in some specialized clinical areas, they are plentiful and robust in others (CMS, 2011). Because a geographic value index calculates a composite quality score for a region, many providers in an area will be evaluated on measures not applicable to their practice. Therefore, for a single uniform geographic value index to generate fair reimbursement rates, data would at a minimum have to indicate that performance across a wide range of quality measures was relatively consistent within an area.
Testing this notion, the committee drew on a small sample of the large universe of quality measures, estimating pairwise correlations between 18 condition-specific Medicare quality indicators (Acumen, LLC, 2013, p. 129). Correlations ranged from -0.38 (between diabetes retinal screening and cholecystectomy measures) to 0.67 (between chronic obstructive pulmonary disease [COPD] and congestive heart failure [CHF] admissions). Bivariate correlations showed that approximately 38 percent of quality measures are negatively correlated with each other, 40 percent have correlation coefficients between 0 and 0.19, and only one-fifth have correlation coefficients above 0.20. In short, areas with high scores on some quality measures do not necessarily have high scores on others, particularly if the measures relate to conditions treated by different types of specialists.
To explore the nature of relationships among quality indicators, Harvard examined 31 individual measures: 12 process measures,10 readmissions within 30 days of discharge (based on Healthcare Effectiveness Data and Information Set [HEDIS] specifications), 12 prevention quality indicators (PQIs) measuring potentially avoidable hospitalizations,11 and seven patient safety indicators (PSIs) (Harvard University, 2012, pp. 48, 52).12 HRR-level pairwise correlations between the 10 process measures varied from -0.18 (radiation therapy following breast-conserving therapy and appropriate treatment for low back pain) to 0.52 (antidepressant treatment and treatment with beta blockers following an acute myocardial infarction), but the correlations for most were less than 0.20. The 12 preventable admission rates were highly correlated with each other. Patient safety indicators were not highly correlated (all correlations were less than 0.20).
_________________
10Chosen from the National Committee for Quality Assurance’s (NCQA’s) 2011 Healthcare Effectiveness Data and Information Set (HEDIS) or recommended by the relevant specialty society.
11These measures were developed by the Agency for Healthcare Research and Quality (AHRQ) and were designed to measure the quality of ambulatory care.
12These measures were also developed by AHRQ and were designed to measure adverse events associated with medical errors.
And correlations across measure types (for example, process measures with preventable hospitalizations) were generally low, with a few exceptions (Harvard University, 2013).
Collectively, these findings suggest that an area in which providers deliver high-value treatment for one condition may well contain providers who deliver low-value treatment for other conditions. This finding again demonstrates that provider performance within an area is not homogeneous.
Consistency of Health Care Spending and Utilization Within an Area
Spending and utilization measures across conditions are more highly correlated within an HRR than are the quality measures just described (see Tables 3-2 and 3-3). Nonetheless, an HRR that uses many services to treat a given condition (e.g., prostate cancer) does not necessarily use many services to treat another (e.g., low back pain). In that example, the correlation across HRRs for Medicare beneficiaries is 0.485 and for the commercial population is 0.43.
Conclusion 3.3. Quality across conditions and treatments varies widely within HRRs; spending and utilization across conditions are moderately correlated within HRRs.
“Acceptable” and “Unacceptable” Sources of Geographic Variation
Some proponents of a geographic value index contend that area-level payments may be appropriate even if provider behavior varies within an area, reasoning that after controlling for acceptable sources of variation, any remaining variation represents inefficiencies correctable through area-level payment incentives. To evaluate this assertion, the committee commissioned analyses (see Chapter 2) to identify potential sources of variation and quantify their possible influences.13 It is important to underscore again here that characterizing this unexplained variation as “acceptable” or “unacceptable” is impossible, as associated sources cannot be observed within the available data. As established by the detailed results and related discussion in Chapter 2, however, the robustness of this body of findings points to important residual variation in health care spending and utilization, indicative of inefficiencies not necessarily remediable at the geographic level.
_________________
13Note that unacceptable variation can arise from both overuse and underuse.
TABLE 3-2
Pearson Correlation Coefficients for Medicare Beneficiary Utilization (Risk-Adjusted Per-Member-Per-Month Cost) Across Cohorts
| Lower Back Pain | Cataracts | Congestive Heart Failure | Breast Cancer | Prostate Cancer | Cholecystectomy | |
| Lower Back Pain | 1.00 | |||||
| Cataracts | 0.477 | 1.00 | ||||
| Congestive Heart Failure | 0.907 | 0.483 | 1.00 | |||
| Breast Cancer | 0.574 | 0.311 | 0.583 | 1.00 | ||
| Prostate Cancer | 0.485 | 0.230 | 0.502 | 0.524 | 1.00 | |
| Cholecystectomy | 0.593 | 0.353 | 0.624 | 0.406 | 0.406 | 1.00 |
SOURCE: Acumen, LLC, 2013.
TABLE 3-3
Pearson Correlation Coefficients for Input-Price-Adjusted Spending Across Cohorts in the Harvard MarketScan Commercial Population
| Lower Back Pain | Cataracts | Congestive Heart Failure | Breast Cancer | Prostate Cancer | Cholecystectomy | |
| Lower Back Pain | 1.00 | |||||
| Cataracts | 0.35 | 1.00 | ||||
| Congestive Heart Failure | 0.52 | 0.36 | 1.00 | |||
| Breast Cancer | 0.49 | 0.52 | 0.37 | 1.00 | ||
| Prostate Cancer | 0.43 | 0.33 | 0.39 | 0.49 | 1.00 | |
| Cholecystectomy | 0.40 | 0.36 | 0.39 | 0.42 | 0.36 | 1.00 |
SOURCE: Harvard University, 2012.
Relationships Between Spending or Utilization and Quality
Use of a geographic value index would require that area-level performance be observable in reliable relationships between health care resource use and health care quality. Absent such reliable relationships, geographically based value adjustment would have, on balance, no predictable effect on overall quality. Thus, assuming that composite measures of health care spending and health outcomes are used to measure value, the case for an area-wide payment adjuster is stronger if a payment change has consistent effects on the quality measures making up the composites.
The committee’s commissioned analyses did not reveal a consistent relationship between condition-specific utilization and area-wide condition-specific quality measures in the Medicare population. The measures are scaled such that a positive correlation indicates that higher condition-specific utilization is associated with higher-quality care. Positive correlations between quality and utilization varied from 0.005 for radiation therapy for breast cancer to 0.085 for disease-modifying antirheumatic drugs dispensed for arthritis (0.085). Negative correlations between quality and utilization varied from -0.012 for antiplatelet prescription over 12 months for coronary heart disease (CHD) to -0.483 for bronchodilator prescription 30 days following an acute COPD event (Acumen, LLC, 2013, pp. 104-111, 131).
In the commercial population, Harvard found that positive correlations between quality measures and spending varied from 0.02 for the preventable acute admissions composite to 0.30 for antibiotics following pneumonia diagnosis (Harvard University, 2012, pp. 19-21, 58). Negative correlations between quality and spending ranged from -0.07 for readmissions and the preventable admissions composite to -0.13 for the preventable chronic admissions composite. Spending, however, can be disaggregated into price and quantity components. Holding input price constant and focusing on variation in the quantity of services, positive correlations between utilization and quality ranged from 0.06 for breast cancer screening mammography to 0.09 for readmissions. Negative correlations between utilization and quality varied from -0.06 for antibiotics following pneumonia diagnosis to -0.57 for imaging following complaint of low back pain (Harvard University, 2012, pp. 18-23, 58).
Although the committee believes several of these correlations are subject to measurement error because of small samples, there is no affirmative evidence of a consistent relationship between spending and quality. Few of the correlations differ substantially from zero.14
_________________
14This is so even though mechanical relationships between quality measures and utilization cause some correlations to be artificially strong. For example, the outcome for the COPD admissions quality measure is an inpatient admission. As the rate of COPD admissions increases in a region, indicating a lower quality of COPD care, utilization, by definition, necessarily increases.
In sum, acknowledging the limitations and challenges to interpretation of the quality analyses, overall the committee found no evidence of reliable associations between disease- or condition-specific measures of utilization and disease- or condition-specific measures of quality. As a result, a geographic value index employing these measures could affect some health outcomes negatively and others positively.
Findings from the committee’s commissioned empirical analyses are consistent with those of a recent systematic review demonstrating an inconsistent relationship between health care quality and cost (Hussey et al., 2013). Of 61 studies selected for review, 21 (34 percent) found a positive or mostly positive association, 18 (30 percent) found a negative or mostly negative association, and 22 (36 percent) found an inconsistent or no association between health care quality and cost. Further, the authors of the review concluded that the magnitude of the cost-quality association was generally low or moderate from the perspective of clinical significance.
Conclusion 3.4. HRR-level quality is not consistently related to spending or utilization in Medicare or the commercial sector.
It is important to note that this particular conclusion is limited to area-level, composite measures of value derived from administrative claims data and should not be interpreted as condemning initiatives to improve health care value. Nor does it imply that particular providers in low- or high-cost areas currently are being compensated appropriately.
RECOMMENDATION 2: Congress should not adopt a geographically based value index for Medicare. Because geographic units are not where most health care decisions are made, a geographic value index would be a poorly targeted mechanism for encouraging value improvement. Adjusting payments geographically, based on any aggregate or composite measure of spending or quality, would unfairly reward low-value providers in high-value regions and punish high-value providers in low-value regions.
Acumen, LLC. 2013. Geographic variation in spending, utilization and quality: Medicare and Medicaid beneficiaries. Washington, DC: Institue of Medicine.
Alliance for Health Reform. 2013. Select community quality initiatives map. http://allhealth.org/community-initiatives.asp (accessed February 19, 2013).
Cherkin, D. C., R. A. Deyo, K. Wheeler, and M. A. Ciol. 1994. Physician variation in diagnostic testing for low back pain. Who you see is what you get. Arthritis & Rheumatism 37(1):15-22.
CMS (Centers for Medicare & Medicaid Services). 2010. Medicare program: Payment policies under the physician fee schedule and other revisions for Part B for CY 2011. Federal Register 75(288):73170-73860.
———. 2011. Claims-based quality measures. http://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeedbackProgram/downloads/claims_based_measures_with_descriptions_num_denom_excl.pdf (accessed July 18, 2013)
———. 2013a. Hospital value-based purchasing. http://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/hospital-value-based-purchasing/index.html?redirect=/hospital-value-based-purchasing (accessed April 4, 2013).
———. 2013b. Value-based payment modifier. http://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeedbackProgram/ValueBasedPaymentModifier.html (accessed April 4, 2013).
Hahn, J., and J. Mulvey. 2011. Medical physician payment updates and the sustainable growth rate (SGR) system. Washington, DC: Congressional Research Service.
Harvard University. 2012. Geographic variation in health care spending, utilization, and quality among the privately insured. Washington, DC: Institute of Medicine.
———. 2013. Harvard HRR-level quality measures correlations. Washington, DC: Institute of Medicine.
Hussey, P. S., S. Wertheimer, and A. Mehrotra. 2013. The association between health care quality and cost: A systematic review. Annals of Internal Medicine 158(1):27-34.
IOM (Institute of Medicine). 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
———. 2010. Leadership commitments to improve value in health care: Finding common ground: Workshop summary. Washington, DC: The National Academies Press.
Kass, J. E., and J. S. Linehan. 2012. Fostering healthcare reform through a bifurcated model of fraud and abuse regulation. Journal of Health & Life Sciences Law 5(75):79-93.
Lucas, F. L., B. E. Sirovich, P. M. Gallagher, A. E. Siewers, and D. E. Wennberg. 2010. Variation in cardiologists’ propensity to test and treat: Is it associated with regional variation in utilization? Circulation: Cardiovascular Quality and Outcomes 3(3):253-260.
McKethan, A., M. Shepard, S. L. Kocot, N. Brennan, M. Morrison, N. Nguyen, R. D. Williams, and N. Cafarella. 2009. Improving quality and value in the U.S. health care system. Washington, DC: Engelberg Center for Health Reform, Brookings Institution, and Avalere Health, LLC.
MedPAC (Medicare Payment Advisory Commission). 2007. Assessing alternatives to the sustainable growth rate system. Washington, DC: MedPAC.
———. 2009. Measuring regional variation in service use. In Report to the Congress: December 2009. Washington, DC: MedPAC.
Partners HealthCare. 2012. Variation within a single group of primary care physicians at Partners Healthcare, Inc. Boston, MA: Partners HealthCare, Inc.
Salinsky, E. 2010. Governmental public health: An overview of state and local public health agencies. Washington, DC: National Health Policy Forum.
University of Pittsburgh. 2013. Geographic variation in health care spending and promotion of high-value health care Medicare Part D. Washington, DC: Institute of Medicine.