APPENDIX D
Intensity Influence
The photocatalytic process at a TiO2 particle can be schematically represented in a simplified reaction scheme as:

The actual reaction mechanism is more complicated and can involve a number of intermediates, such as hydroxyl radicals species adsorbed on the TiO2 surface, radicals, and radical ions.
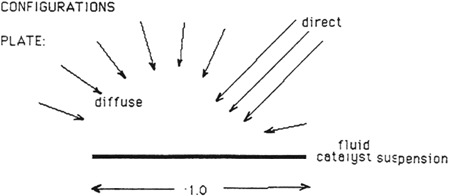
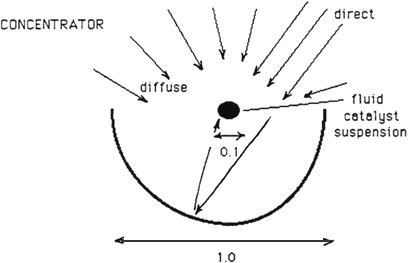
A summary of literature and Solar Energy Research Institute data [1] for the effect of intensity [irradiance (photons/area-time)] on the photocatalyzed rate of oxidation indicates that at or above I sun near-ultraviolet equivalence, the reaction rate varies as the square root of intensity. This square root dependence can be rationalized by the increased importance of processes like e-h+ recombination [Eq. (4)] and the reaction of photogenerated e- and h+ with intermediates. A collector also brings a reflectance loss. The impact of these two phenomena on the achievable rate is now estimated (see Figure D-1).
A. Photon Rate Arriving at Catalyst
i. Plate: (direct + diffuse)(area)
= (Id + 0.25Id)(1.0)
= 1.25 Id (photons/time)
ii. Concentrator:
(direct)(collector x-section) + (diffuse)(tube x-section)
(0.8 Id)(1.0) + (0.25Id)(0.1)
(reflectance = 0.8)
= 0.825Id