APPENDIX F
Knowledge Base and Status of Technology of Solar Fuels
Direct Decomposition of H2X (X = O, S)
Concentrated sunlight is a unique energy source capable of heating materials to temperatures in excess of 1000°C, without the combustion products associated with fossil fuels and without the inefficiency of electricity production associated with plasma arc furnaces [1]. In addition, concentrated sunlight is an intense source of high-energy photons that can be harnessed to drive photochemical systems. Utilization of a direct catalytic absorption receiver takes advantage of both, by passing the concentrated sunlight through a transparent window onto a catalyst bed where the sunlight is absorbed in depth. Thus, the heat and the photons are delivered directly to sites at which endothermic reactions occur.
1. Direct Decomposition of Water
The decomposition of liquid water at 25°C gives gaseous hydrogen and oxygen. This occurs at wavelengths below 200 nm, which are absorbed by water but which solar energy cannot supply. A suitable photocatalytic redox system is therefore required to promote water photolysis utilizing solar energy. No systems are presently available for practical applications.
(b) Thermolysis [4] needs >2000°C for splitting water thermally; only 4 percent of water is thermolyzed.
2. Decomposition of Hydrogen Sulfide
(a) Thermolysis [5]
Hydrogen sulfide is an industrial waste product, recovered in large quantities in the sweetening of natural gas and in the removal of organically bound sulfur from petroleum and coal. The quantity of organically bound sulfur recoverable from fossil fuels in the United States alone
amounts to about 40 billion metric tons. In 1979, 1.76 million metric tons of sulfur was recovered from natural gas in the United States, and another 2.31 million metric tons was recovered from petroleum. In Canada the recovered sulfur amounted to 6.1 million metric tons. The heating value of the associated hydrogen comes to 9.1 x 107 GJ per year or about 2900 MW. This represents, on an annual basis, the equivalent of 1.92 x 109 kg (17 million barrels) of gasoline. If the hydrogen could be recovered, it could be used to upgrade 6.8 x 109 kg of coal to produce 59 million barrels of high-grade hydrocarbon fuel.
Current practice uses the Claus process to recover the sulfur from hydrogen sulfide, with the hydrogen oxidized to water to produce low-grade process heat.
Although an economic evaluation of hydrogen sulfide processing has not been carried out, use of solar energy to produce hydrogen and sulfur from H2S promises some economic advantages, making it more advantageous in the short term than the water-splitting process: (1) sulfur is more valuable than oxygen; (2) the H2S process works at much lower temperatures (1000° to 2000°K); (3) solar collection efficiency is much higher (reradiation losses are minimized at the lower temperatures); (4) heliostat and concentrator areas are reduced to 55 percent from those needed for water splitting; (5) an H2S plant would cost 62 percent of that for the water process; and (6) for equal energy supplied, 1 kg of water processed produces 0. 111 kg of H2, versus 26.8 kg of H2S processed to yield 1.756 kg of H2 and 25.2 kg of S.
(b) Thermocatalytic [6]
Transition metal sulfides (MoS2 and WS2) have been examined as possible catalysts for the effective decomposition of hydrogen sulfide at a temperature of 500° to 800°C. Other catalysts such as alumina, nickel/molybdenum sulfide, and cobalt/molybdenum sulfide have also been utilized, using a 1.5 kW solar furnace as the heat source.
Hybrid Systems
1. Photochemical/Thermochemical/Electrochemical Cycles
(a) The Yokohama Mark 5 and 6 cycles
A combination of several reactions (photochemical, thermochemical, etc.) presents advantages with respect to solar energy utilization as it embraces both the heat generated in a solar reactor and the high-energy photons available.
Various multistage hybrid systems for water decomposition by solar energy have been developed by the Yokohama group in Japan [3,7,8]. The systems are based on an appropriate combination of photochemical and electrochemical reactions (Yokohama Mark 5) or on photochemical, electrochemical, and thermochemical processes (Yokohama Mark 6). Both systems use I2 as a sensitizer. Figure F-1 depicts a scheme of the experimental system. The aqueous solution of I2 and Fe2+ (as FeSO4) ions is passed as a thin film in front of the Fresnel lens. The HI and Fe3+ ions produced are subsequently reduced electrochemically in two separate cells, producing H2 and O2 and reforming I3- and Fe2+ ions. The fraction of nonabsorbed light is collected by the Fresnel lens and directed onto a thermoelectric device (a p/n junction) whereby electrons and holes are produced on the semiconductor electrodes and subsequently used to produce electric power. For the Mark 5 system, the energy demand cannot be self-supplied. The overall efficiency of the absorbed light is about 20 percent. In the Mark 6 system, the energy demand is supplied by the thermochemical and electrochemical processes. The efficiency of the photochemical conversion process
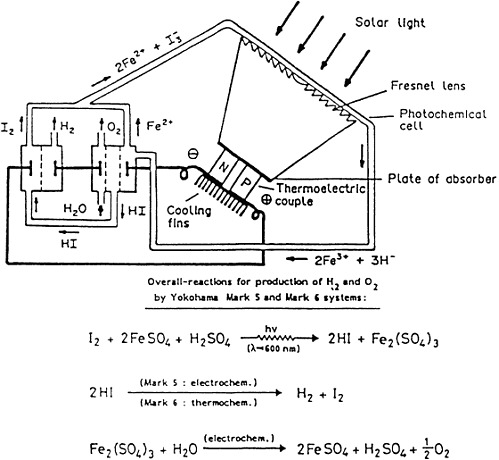
depends on the light intensity, concentration, and the nature of the reactants; it is approximately 15 to 20 percent. One disadvantage is the reversibility of the photochemical reaction.
(b) The Yokohama Mark VII cycle
In the improved Yokohama Mark VII hybrid system [8,9], the cycle has been modified to be:
2Fe3(PO4)2 + 3I2 + 2H3PO4 ——> 6FePO4 + 6HI (photochemical)
6HI ——> 2H2 + 2I2 (thermochemical)
6FePO4 + 3H2O ——> 2Fe3(PO4)2 + 2H3PO4 3/2 O2 (electrolytic)
The modified experimental setup is illustrated in Figure F-2 [9].
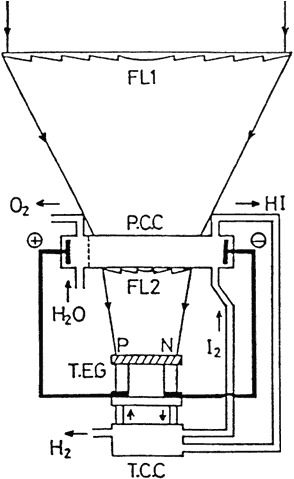
Figure F-2 A model scheme of the Yokohama Mark VII: FL1, FL2, Fresnel lenses; PCC, photoelectric cell; TEG, thermoelectric generator; TCC, thermochemical chamber.
Source: [9]
In this case, all three reactions are carried out by solar energy. The photochemical reaction and the electrolysis are carried out in a photon-assisted electrolyzer that evolves HI and O2. The light beam passing through the photoelectrochemical cell is collected and heats up the thermoelectric generator which supplies electricity to the electrolyzer, and the wasted heat is also used for the HI decomposition.
This system is advantageous for the hydrogen producing portion of the cycle; the efficiency is low at present.
The hybrid systems described above are promising devices for a technical solar energy utilization, although nothing is known about any eventual corrosion problems and stability, among other factors.
2. Thermochemical/Electrochemical System Using Metal/Metal Hydride (M = Na) Couples [10]
The metal/metal hydride processes can be used to produce hydrogen from water through the direct use of high-temperature heat. They are hybrid cyclic systems that consist of an electrolytic step and a thermochemical step. In the electrolytic step, a hydrogenous intermediate product (MH) and oxygen are first produced from water at 250° to 400°C. The presence of a hydrogen acceptor (M), which acts as a depolarizer, leads to smaller electrolytic voltage requirements for the process. The metal hydride, MH, is subsequently decomposed in the thermochemical step into M and H2 at high temperatures (700° to 900°C). The high-temperature heat leads to considerably higher efficiencies in comparison to purely electrolytic hydrogen generation processes. A small demonstration scale plant produced 20 cm3 of hydrogen per hour.
3. Thermolytic/Electrochemical Process [11]
The experimental setup used to examine this hybrid process is shown in Figure F-3. With this apparatus (temperature in reactor = 2,500 K at 105 Pa pressure), 90 percent of the H2 formed was recovered. For direct normal irradiance of 700 W/m-2, the product gas rate was 3.21 × 10-3 mol of H2 per hour; the decomposed molar fraction of water was 2.9 percent, and the overall thermal efficiency for the thermolysis was 1.1 percent.
A feasibility analysis for an industrial-scale plant module for thermolysis (only) to give 172 GJ per year equivalent of thermolytic hydrogen (200 mol of H2 per hour for an energy input of 1191.3 MJ per hour) was performed. If the waste heat were recovered within the process to generate additional steam to drive a unipolar electrolyzer, the production rate for H2 could be doubled to give a total yearly capacity of 344 GJ in equivalent hydrogen. Efficiencies and the relevant costs for hydrogen produced are given in Table F-1.
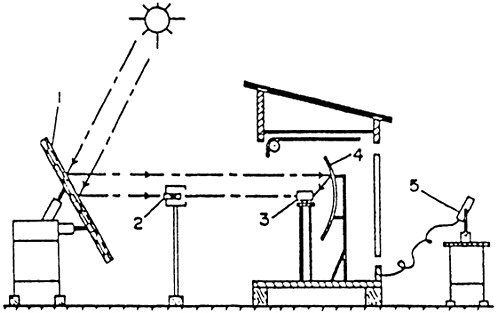
Figure F-3 Experimental setup for the thermolytic/electrolytic decomposition of water: (1) nonfocusing heliostat, (2) heliostat, (3) reactor, (4) reflector, (5) pyrheliometer.
Source: [11].
Table F-1 First Law (energy) and Second Law (exergy) Efficiencies for the Thermolysis/Electrolysis System and Related Costs
|
Parameter |
Thermolysis |
Hybrid System |
|
Energy efficiency |
4.11% |
5.39% |
|
Exergy efficiency |
3.39% |
4.44% |
|
Cost of hydrogen |
$68/GJ |
$46/GJ |
The exergy analysis shows the major losses (72 percent) in this system to be in the receiver/reactor (steam generator, steam superheater, and reactor).
If the concentrator costs were $20 m-2, the above costs for hydrogen come down to $58 per GJ for thermolytic hydrogen and $38 per GJ for hydrogen from the hybrid system. These costs can be compared with those for hydrogen production from other solar methods: $60 to $115 per GJ for PV-electrolytic hydrogen [12] and $15 to $70 per GJ for hybrid thermochemical hydrogen [13].
4. Thermochemical and Hybrid Systems Based on the Sulfur Family Cycles
(a) The Mark Processes [14]
The various Mark processes are based principally on the thermal decomposition of H2SO4 acid at high temperatures (1123 K):
H2SO4 ——> H2O + SO2 + 1/2 O2
Three closures are possible for the cycle:
(i) electrochemical
SO2 + 2H2O ——> H2SO4 + H2 (Westinghouse, Ispra Mark 11)
(ii) hybrid (thermal followed by electrochemical)
SO2 + 2H2O + Br2 ——> H2SO4 + 2 HBr (300 K)
2 HBr ——> H2 + Br2 (electrochemical)
(iii) thermal
SO2 + 2 H2O + I2 ——> H2SO4 + 2 HI (393 K)
2 HI ——> H2 + I2 (573 K)
(General Atomics; Ispra Mark 16)
While detailed cost evaluations have not been done, the Ispra people believe that costs from the Mark processes, with thermal efficiencies greater than 35 percent, would be comparable to the costs for advanced water electrolysis.
(b) (i) The Cristina Process (vector = hot air) [15]
Sunlight is intermittent (diurnal vs. nocturnal). To avoid shutdown operations during nighttime
hours in a hydrogen production solar plant, a process has been devised (the Cristina process) in which SO2 is produced during sunshine hours and operates in reverse as a sulfuric acid synthesis process at a required rate to produce high-temperature heat during nighttime operations by using a small part of the stored SO2. In this way the chemical process is operated continuously, thereby eliminating thermal inertia and start-up problems.
During sunlight:
H2SO4nH2O ——> H2SO4 + nH2O (600 K)
H2SO4 ——> H2O + SO3 (600–1000 K)
SO3 ——> SO2 + 1/2 O2 (1100–1200 K)
During nocturnal operations the last two steps are reversed using oxygen from the air.
The system would produce 106 mol of SO2, per hour when coupled to a receiver/solar system producing 106 GJ of heat per hour operating 2333 hours per year. When coupled to a hydrogen-producing step, as in the Mark 11 or Mark 13 cycle operating 7000 hours annually and using external electric energy, the system would produce 0.62 × 106 GJ of hydrogen per year.
The cost of sulfuric acid decomposition by the solar Cristina process was estimated to be $31 per GJ of hydrogen. With the cost of solar heat set at about $32 per GJ of hydrogen and that of the hydrogen-producing step set at approximately $5 per GJ of hydrogen, the total cost for producing solar hydrogen is about $68 per GJ.
(b) (ii) The Cristina Process (vector = hot oxygen) [16]
The Cristina process was improved by using hot oxygen as the vector. With these modifications, for a 106, GJ/year solar plant, the overall efficiency of solar hydrogen production is about 38 percent and the solar hydrogen cost is $15 to $70 per GJ of hydrogen, depending on cost parameters [16].
Aparametric analysis carried out on this version of the solar Cristina process showed the following: (1) the cost of H2 varies linearly with the process heat cost, capital charge rate, and electricity costs; (2) the solar H2 cost is most sensitive to the process heat cost (doubling the process heat cost increases H2 cost by 65 percent); (3) doubling the capital charge rate increases the solar H2 cost by 25 percent; (4) doubling the electricity costs increases the solar H2, cost by 8 percent; (5) the cost of the solar H2 varies from $15 to $70 per GJ of hydrogen — the typical cost of solar hydrogen for 15 percent capital charge rate, $10 per GJ of solar thermal energy and $0.02 kWh of electrical energy is about $33 per GJ of hydrogen [a recalculation of the Cristina-Mark process in (b)(i) above gives a cost for solar H2 of $53 per GJ under the same conditions]; and (6) the typical solar hydrogen cost of $33 per GJ is about three times the cost of nuclear hydrogen ($10 per GJ) — due to the intermittence of the solar source and the high cost of solar heat.
(c) The General Atomics (GA) Sulfur/Iodine Process
There are four principal steps in the GA process [17]:
(i) water splitting
SO2 + I2 + 2H2O ——> H2SO4 + 2HI
(ii) decomposition of sulfuric acid
H2SO4 ——> SO2 + H2O + 1/2 O2 (870 K)
(iii) separation of HI, I2, and H2O (a major source of inefficiency)
(iv) decomposition of HI
2HI ——> H2 + I2
This process was first devised for nuclear energy to be the primary energy source; overall process thermal efficiency is about 46 percent [14]. When the GA process is coupled to a solar thermal heat source, the hydrogen production costs range from $40 to $50 per GJ of hydrogen for 2333 hours of operation per year, a 27 percent capacity factor and a 20 percent return on investment [14]. The major cost (about 76 percent) is due to the solar/process interface and sulfuric acid decomposition; separation of HI is also expensive (and inefficient).
Another study that compared the GA thermochemical S/I2 cycles against water vapor electrolysis was carried out by Leach and Copeland, first proposed in 1986 [18] and subsequently in 1990 [17]; the latter was not updated. Leach and Copeland claim that water vapor electrolysis in a high-temperature solid oxide cell is more economical than thermochemical hydrogen production by the GA process. Equipment costs for the electrolysis process are lower, the process efficiency
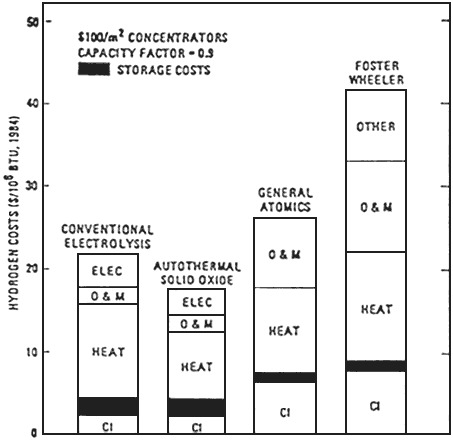
FIGURE F-4 Electrolysis with a 600°C receiver versus S/I2 with a 900°C receiver (1 BTU = 1.055 kJ).
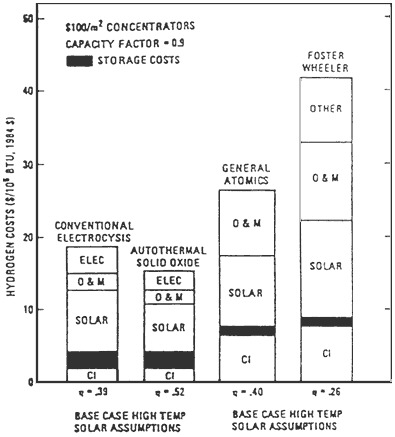
FIGURE F-5 Electrolysis versus S/I2 high-temperature reciever.
is higher, and there is less technical risk in adapting it to a solar heat source. Their conclusions appear to be at variance with other studies carried out on thermochemical hydrogen production.
The costs of hydrogen produced by the two solar water-splitting processes (the GA process vs. the water vapor electrolysis process) are compared in Figures F-4 and F-5 [17,18].
An analysis of energy from coal versus solar energy shows that the latter will be competitive with coal for electricity production (if the Department of Energy's solar thermal goals are realized). Leach and Copeland conclude that the opposite is true for hydrogen production. Solar thermal energy is competitive for electricity production because energy conversion equipment cost reductions are possible with a clean heat source; for hydrogen production via existing water splitting, equipment costs for solar plants are higher than for plants that consume fossil fuels. Also, known water-splitting processes, that convert solar energy to stored chemical energy in hydrogen are less efficient than commercial processes that convert stored energy in fossil fuels to energy in hydrogen [17]. Because of the historically highly unreliable costs of fossil fuels, it is not possible to conclude that solar hydrogen will be uneconomical in the year 2010.
(d) The UT-3 (MASCOT) Process [8,19–23]
This multistep thermochemical water decomposition cycle was proposed in 1978 by the Yokohama group. The reactions are heterogeneous gas/solid reactions and are carried out continuously in
fixed bed reactors by transferring gaseous reactants from one reactor to another without moving the reactants [14].
A bench-scale model of the UT-3 system was fabricated and operated for 200 hours. A cost evaluation (1985) for this process on an industrial scale was done by TOYO Engineering Corporation and sponsored by JAERI. The output was 20,000 nm3 per hour; the primary energy source was a high-temperature gas-cooled nuclear reactor (HTGR); the thermal efficiency depended on the power generation efficiency. If power is generated at 30 percent, the overall thermal efficiency is about 45 percent. The capital cost of the plant was 12 billion yens (1985), and the cost of heat from the HTGR was 3.8 yen per 1000 Kcal (or 908 yen per GJ).
The UT-3 process continues to be actively examined by the Japanese. A more recent analysis is given by Aochi et al. [20].
REFERENCES
1. Fish, J.D. ''Application of Solar Technology to Fuel Production Chemical Processing and Thermochemical Energy Transport: Status and Future." Advances in Solar Technology, Vol. 3, (W.H. Bloss and F. Pfisterer, eds.), Pergamon Press, Oxford (1987).
2. Getoff, N. "Basic Problems of Photochemical and Photoelectrochemical Hydrogen Production from Water." Int. J. Hydrogen Energy, 9, 997 (1984).
3. Getoff, N. "Photoelectrochemical and Photocatalytic Methods of Hydrogen Production: A Short Review." Int. J. Hydrogen Energy, 15, 407 (1990).
4. Dinga, G.P. "A4. "Hydrogen: The Ultimate Fuel and Energy Carrier." Int. J. Hydrogen Energy, 14, 777 (1989).
5. Noring, J.E., and Fletcher, E.A. "High Temperature Solar Thermochemical Processing — Hydrogen and Sulfur from Hydrogen Sulfide." Energy, 7. 651 (1982).
6. Bishara, A., Salman, O.A., Khraishi, N., and Marafi, A. "Thermochemical Decomposition of Hydrogen Sulfide by Solar Energy." Int. J. Hydrogen Energy, 12, 679 (1987).
7. Ohta, T. "Photochemical and Photoelectrochemical Hydrogen Production from Water." Int. J. Hydrogen Energy, 13, 333 (1988).
8. Ohta, T., and Abe, I. "Hydrogen Energy Research and Developments in Japan." Int. J. Hydrogen Energy, 10, 275 (1985).
9. Kamiya, N., Ohta, T., and Asano, N. "Hybridized Hydrogen Production System with Fe/I. Photochemical Reaction in Concentrated Phosphoric Acid." Int J. Hydrogen Energy, 9, 563 (1984).
10. Herzog, F., and Glaubitz, D. "Production of Hydrogen from the Na/NaH Process." Int. J. Hydrogen Energy, 15, 13 (1990).
11. Baykara, S.Z., and Bilgen, E. "An Overall Assessment of Hydrogen Production by Solar Water Electrolysis." Int. J. Hydrogen Energy, 14, 881 (1989).
12. Hammache, A., and Bilgen, E. Proc. ASES Ann. Meet., p. 108 (1987).
13. Bilgen, E. Sol. Energy 41, 1 (1988).
14. Funk, J.E., and Knoche, K.F. "Hydrogen by Thermochemical Water Splitting." P. 2894 in Advances in Solar Energy Technology, Vol. 3, (W.H. Bloss and F. Pfisterer, eds.), Pergamon Press, Oxford, (1987).
15. Bilgen, C., Broggi, A., and Bilgen, E. "The Solar Cristina. Process for Hydrogen Production." Sol. Energy 36, 267 (1986).
16. Bilgen, E. "Solar Hydrogen Production by Hybrid Thermochemical Processes." Sol. Energy, 41, 199 (1988).
17. Leach. J.W., and Copeland, R.J. "Solar Hydrogen Production: The Sulfur/Iodine Cycle Versus Water Vapor Electrolysis." Int. J. Energy Systems, 10, 55 (1990).
18. Leach, J.W., and Copeland, R.J. "Solar Hydrogen Production: The Sulfur/Iodine Cycle Versus Water Vapor Electrolysis." P. 702 in 21st Proceedings of the Intersociety Energy Conversion Engineering Conference, American Chemical Society, Washington D.C., p. 702 (1986).
19. Nakayama, T., Yoshioka, H., Furutani, H., Kameyama, H., and Yoshida, K. "MASCOT — A Bench-Scale Plant for Producing Hydrogen by the UT-3 Thermochemical Decomposition Cycle." Int. J. Hydrogen Energy, 9, 187 (1984).
20. Aochi, A., Tadokoro, T., Yoshida, K., Kameyama, H., Nobue, M., and Yamaguchi, T. "Economical and Technical Evaluation of UT-3 Thermochemical Hydrogen Production Process for an Industrial Scale Plant." Int. J. Hydrogen Energy, 14, 421 (1989).
21. Kameyama, H., Tomino, Y., Sato, T., Amir, R., Orihara, A., Aihara, M., and Yoshida, K. "Process Simulation of MASCOT Plant Using the UT-3 Thermochemical Cycle for Hydrogen Production." Int. H. Hydrogen Energy, 14, 323 (1989).
22. Yoshida, K., Kameyama, H., Aochi, T., Nobue, M., Aihara, M., Amir, R., Kondo, H., Sato, T., Tadokoro, Y., Yamaguchi, T., and Sakai, N. "A Simulation Study of the UT-3 Thermochemical Hydrogen Production Process." Int. J. Hydrogen Energy, 15, 171 (1990).
23. Aihara, M., Umida, H., Tsutsumi, A., and Yoshida, K. "Kinetic Study of UT-3 Thermochemical Hydrogen Production Process." Int. J. Hydrogen Energy, 15, 7 (1990).











