Appendix A
Contributed Manuscripts
A1
EFFECTOR AND MEMORY T CELL RESPONSES TO COMMENSAL BACTERIA1
Yasmine Belkaid,2Nicolas Bouladoux,2and Timothy W. Hand2
Barrier surfaces are home to a vast population of commensal organisms that together encode millions of proteins, each of them possessing several potential foreign antigens. Regulation of immune responses to this enormous antigenic load represents a tremendous challenge for the immune system. Tissues exposed to commensals have developed elaborate systems of regulation including specialized populations of resident lymphocytes that maintain barrier function and limit potential responses to commensal antigens. However, in settings of infection and inflammation these regulatory mechanisms are compromised and specific effector responses against commensal bacteria can develop. This review discusses the circumstances controlling the fate of commensal specific T cells and how dysregulation of these responses could lead to severe pathological outcomes.
________________
1 Reprinted with permission from Yasmine Belkaid. Originally published as Belkaid, Y. et al. 2013. Effector and memory T cell responses to commensal bacteria. Trends in Immunology 34:6. doi: 10.1016/j.it.2013.03.003.0.
2 Mucosal Immunology Section, Laboratory of Parasitic Diseases, National Institute of Allergy and Infectious Diseases, NIH, Bethesda 20892, USA.
Commensal Microbiota Shape T Cell Resident Homeostasis
The epithelial surfaces in the body act as a scaffold to sustain diverse communities of commensal organisms that include bacteria, archaea, fungi, protozoa, and viruses (Eckberg et al., 2003; Foulongne et al., 2012; Grice and Segre, 2012; Iliev et al., 2012; Reyes et al., 20102). With an estimated composition of 100 trillion cells, commensals outnumber host cells by at least a factor of 10 and encode 100-fold more genes than the host genome (Ley et al., 2006). All barrier sites, including the genital mucosa, skin, airways, and gut are constitutively colonized by highly diverse and site-specific flora. The gastrointestinal (GI) tract represents the most abundant commensal niche with a population of more than 1,000 individual strains that contain some 3 million unique genes (Clemente et al., 2012; Qin et al., 2010). Although commensalism is defined in ecology as a relationship in which only one party benefits while the other is neutral, many of the bacteria of the GI tract are better described as mutualists, adding tremendous enzymatic and protective capability to the host while taking advantage of the nutrient-rich environment that the host provides (Backhed et al., 2005; Costello et al., 2012). In particular, commensal bacteria can prevent colonization by pathogenic organisms (Stecher and Hardt, 2011) and control many aspects of host physiology, not the least of which are lymphocytes of the immune system.
T cells are found in large numbers lining barrier surfaces where they are tasked with surveillance against infection while maintaining diplomatic relations with the resident commensal microbiota (Hooper et al., 2012). As a result, CD4 T cells at these surfaces can adopt multiple inter-related fates associated with the expression of characteristic cytokines and transcription factors (Murphy and Stockinger, 2010; Zhou et al., 2008). Under steady-state conditions, the GI tract and gut-associated lymphoid tissue (GALT) is dominated by interleukin (IL)-17A-producing T helper (Th)17 cells, interferon (IFN)-γ-producing Th1 cells, and forkhead box (Fox)p3+ regulatory T (Treg) cells (Hall et al., 2008; Maynard and Weaver, 2009). The balance between these populations is tightly controlled by the cytokine milieu, which at barrier surfaces is in part dependent upon dietary elements and the microbiota (Bettelli et al., 2006; Hall et al., 2011; Hu et al., 2011; Konkel and Chan, 2011). Th17 cells are largely limited to barrier surfaces and have been an area of particular interest in the study of mucosal immunology. The protective role of IL-17A is associated with its capacity to induce neutrophil granulopoiesis by stimulating epithelial cells to secrete granulocyte colony-stimulating factor (G-CSF) and drive the recruitment of neutrophils by local stromal cells (Fossiez et al., 1996; Korn et al., 2009). Additionally, via their capacity to also produce both IL-22 and IL-17A, Th17 cells can bolster innate epithelial defense mechanisms and reinforce tight junctions (Ishigame et al., 2009; Sonnenberg et al., 2011, 2012; Zheng et al., 2008). The function of mucosal Th1 cells under steady-state conditions remains unclear, but we might speculate that these cells also contribute to the promotion of various aspects of innate mucosal responses. Treg cells also represent a prominent population of
resident cells at barrier sites. Treg cells are required for the maintenance of tolerance to both self-antigens and innocuous antigens derived from food, commensal bacteria, and other environmental sources (Bilate and Lafaille, 2012). Treg cells that line the GI tract can arise from the thymus or be induced locally in response to oral antigen, a process required for the acquisition of oral tolerance (Coombes et al., 2007; Sun et al., 2007).
The differentiation of T cells at barrier sites into each of these different fates has been associated with the presence of signals derived from the commensal microbiota (Atarashi et al., 2011; Gaboriau-Routhiau et al., 2009; Ivanov et al., 2009; Mazmanian et al., 2008). Notably, the capacity of T cells to produce IL-17A and IFN-γ is severely compromised in the absence of commensals in both the gut and skin (Hall et al., 2008; Ivanov et al., 2009; Naik et al., 2012). Germ-free mice also tend to harbor higher frequencies of Th2 cells and this too can be reversed by colonization of the mice with a single species of commensal bacteria (Mazmanian et al., 2005). In the GI tract, microbial products such as bacterial DNA or defined group of bacteria such as segmented filamentous bacteria (SFBs) can play a dominant role in the promotion of steady-state GI-resident Th1 and Th17 cells (Hall et al., 2008; Ivanov et al., 2009). In the skin, reconstitution of germ-free mice with Staphylococcus epidermidis restores dermal IL-17A (Naik et al., 2012). The frequencies, origin, and activation of Foxp3+ Treg cells in the skin and GI tract are also influenced by the microbiota. In the skin for instance, the absence of commensals is associated with enhanced frequencies of Treg cells (Naik et al., 2012). By contrast, in the GI tract, Bacillus fragilis via expression of polysaccharide A can expand IL-10-producing CD4+ T cells and Treg cells at the expense of the differentiation of Th17 cells (Mazmanian et al., 2008; Ochoa-Reparaz et al., 2010; O’Mahony et al., 2008; Round et al., 2011). Bacteria of the Clostridium cluster XIV species also promote Treg cell accumulation in the colon, but not small intestine via an increased capacity to promote transforming growth factor (TGF)-β (Atarashi et al., 2011). The gut microbiota can also set the tone of immune responses at distal sites during infection and in some cases contribute to the induction of autoimmune disorders (Abt et al., 2012; Ichinohe et al., 2011; Ivanov et al., 2009; Molloy et al., 2012; Wu et al., 2010).
Antigen-Specific Responses to Commensal Organisms at Steady State
Independent of differentiation state and function, a critical question associated with T cells residing at barrier sites is their antigen specificity. The microbiota encodes millions of proteins, each of them expressing several potential foreign antigens directly associated with inflammatory pathogen-associated molecular patterns. This enormous antigenic load represents a tremendous challenge for barrier immunity as unwanted responses against these antigens could lead to severe pathological consequences. A central strategy utilized by the mucosal immune system to maintain its homeostatic relation with the microbiota is to limit
contact between luminal microorganisms and the epithelial cell surface. This is accomplished by the establishment of a structural and immunological barrier resulting from the combined action of mucus, IgA, and antimicrobial proteins (Hooper et al., 2012) (Figure A1-1A). Further, commensals can promote their own containment and thus mucosal tissue homeostasis by enhancing various aspects of this physical and immunological shield (Brandl et al., 2008; Cong et al., 2009; Vaishnava et al., 2011).
The physical segregation between the host and microbiota is not absolute, and the GI mucosal barrier are prevented from systemic traffic and confined to the GALT by the mucosal firewall (Macpherson et al., 2009; Macpherson and Uhr, 2004). In the GALT, active mechanisms of tolerance regulate T cell responses, and as a result, the GI tract represents a privileged site for the induction of tolerance (Figure A1-1A). The regulatory nature of the GI tract is perhaps best demonstrated by experiments examining oral tolerance to food (Weiner et al., 2011). This phenomenon has long been recognized in both rodent models and humans and is clinically important, because dysregulated T cell responses against food-derived antigens are associated with both celiac disease and GI allergies. Experiments have shown that under steady-state conditions, the predominant antigen-presenting cells of food-derived antigens are dendritic cells (DCs) that express the integrin CD103 (Coombes et al., 2007; Sun et al., 2007). CD103+ DCs drive the differentiation of Foxp3+ Treg cells via their capacity to produce TGF-β and to convert retinol to retinoic acid (RA) (Benson et al., 2007; Coombes et al., 2007; Denning et al., 2011; Mucida et al., 2007, 2009; Sun et al., 2007). Further CD103+ DCs can also induce the expression of gut homing receptors on immune cells, including Treg cells, favoring their migration to the GI tract (Mora et al., 2006). Commensal antigens are likely encountered in the context of these tolerogenic responses, thus, it has been proposed that the GI tract also promotes the induction of Treg cells specific for commensal antigens (Figure A1-1A). Indeed, some types of commensal bacteria seem to support the differentiation and maintenance of colonic Treg cells, although whether these Treg cells are specific to commensal-derived antigens is not known (Atarashi et al., 2011; Round and Mazmanian, 2010). T cell receptor transgenic mice made specific to commensal-derived antigens can differentiate to become Treg cells after transfer to lymphopenic hosts (Feng et al., 2011; Lathrop et al., 2011), and Foxp3+Treg cells specific for commensals have been identified in the GI tract of healthy mice (Lathrop et al., 2011). Aside from the direct maintenance of T cell tolerance, commensal-specific Treg cells may reinforce the mucosal firewall. Treg cells present in the Peyer’s patches of the small intestine have been shown to promote class-switching to IgA in an antigen-specific manner (Cong et al., 2009; Tsuji et al., 2009). IgA can directly modulate expression of commensal antigens and mucosal association, therefore, this implies that Treg cells may play multiple and complementary roles in controlling the host relation with the microbiota (Peterson et al., 2007; Suzuki et al., 2004). Commensal-specific
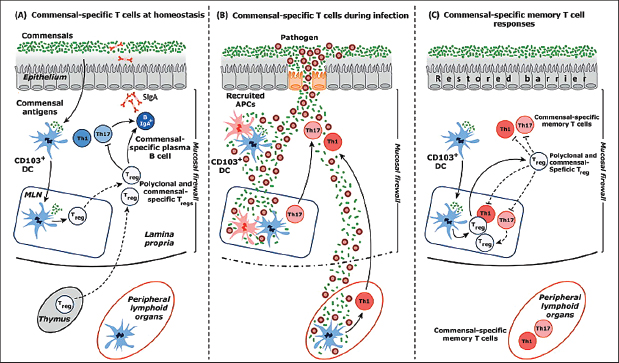
FIGURE A1-1 Commensal-specific T cells at homeostasis and during infection. (A) At homeostasis. Commensal and food-derived antigens are presented to T cells by CD103+ T cells that have trafficked to the mesenteric lymph node (MLN) from the lamina propria. Presentation by these dendritic cells (DCs) to commensal-specific T cells may lead to the differentiation of commensal-specific T regulatory (Treg) cells. Commensal-specific Treg cells traffic to the lamina propria and Peyer’s patches, where they, along with polyclonal Treg cells, can regulate effector T cell responses and induce class switching and IgA production from resident commensal-specific B cells, reinforcing the commensal barrier. Critically, the combination of the epithelial barrier, mucus layer, IgA and regulatory DCs and T cells comprises the “mucosal firewall,” which limits the passage of commensal and food-derived antigens to the gut-associated lymphoid tissue (GALT), preventing untoward activation and pathology. (B) During GI infection. Infection via the intestinal barrier can cause inflammation and tissue damage. This pathology can disrupt the mucosal firewall and allows for systemic translocation of commensal organisms and their associated antigens. During times of translocation, the host immune system becomes unable to discriminate between commensal and pathogen-derived antigens and therefore commensal-specific T cell responses mimic responses to the invasive pathogen. (C) After clearance of infection, the intestinal barrier reforms and the mucosal firewall is restored perhaps preventing the chronic activation of commensal-specific memory T cells. Treg cells may regulate commensal-specific responses either directly or via the modulation of resident DCs. Abbreviation: APCs, antigen-presenting cells.
T cells transferred into lymphopenic hosts can also differentiate into Th1 and Th17 cells (Feng et al., 2011), but Th1, Th17, and Treg cells resident in the GI tract are not necessarily specific to commensal-derived antigens (Geuking et al., 2011; Lochner et al., 2011). For example, Th17 cells develop in the absence of cognate antigen in mice expressing a single T cell receptor (TCR) (Geuking et al., 2011; Lochner et al., 2011). Furthermore, Treg cells can be found in the GI tract of germ-free mice, and albeit significantly reduced, Th17 and Th1 cells are also present in the absence of live commensals (Ivanov et al., 2008). It is worth noting that the diet of germ-free mice contains microbial products, including antigens, that can provide surrogate signals to the ones normally provided by the flora and may be responsible for the presence of these cells without live commensals (Hill et al., 2010). Alternatively, T cell differentiation and gut homing may occur at a low level independently of signals derived from the commensal flora. Although these data support the notion that the flora promotes the induction and/or maintenance of T cells in the GI tract independent of antigen specificity, they do not exclude that a significant portion of mucosal T cells recognize commensal antigens and the specificity of Th1, Th17, or Treg cells that reside at barrier sites under physiological settings remains an important open question.
Environmentally Induced Shifts in the Microbiota
Active regulation of immune tolerance to the commensal microbiota is a lifelong process, because, in contrast to the host genome, the commensal microbiota is not fixed. Recent studies have shown that, although the core metagenome of the gut microbiota is stable, species composition and consequently antigenic composition of the flora vary over time in response to a variety of factors such as diet, sanitation, infection, and antibiotic use (Dethlefsen and Relman, 2011; Lozupone et al., 2012). Therefore, the adaptive immune system of the GI mucosa must be flexible and constantly reset itself to take into account novel commensal antigens. Changes in the microbiota are particularly pervasive during GI infection and inflammation where the commensal microbiota becomes dominated by commensals with enhanced invasive properties (Chow et al., 2011). For instance, the γ-proteobacteria class of bacteria is particularly selected for growth during a diverse set of inflammatory conditions that ranges from Crohn’s disease (CD), colon cancer, and type 2 diabetes in humans to Toxoplasma gondii infection and inflammatory bowel disease (IBD) induction in mice (Arthur et al., 2012; Craven et al., 2012; Garrett et al., 2010; Heimesaat et al., 2006; Lupp et al., 2007; Rolhion and Darfeuille-Michaud, 2007; Qin et al., 2012). Because the γ-proteobacteria family is mostly composed of genera that are opportunistic or obligate pathogens, this effect could be due to the unique ability of these organisms to colonize the inflamed GI tract. Indeed, Escherichia coli that dominates the GI tract in T. gondii infection is skewed toward inflammatory and invasive strains that can contribute to the pathological process during inflammatory responses and
infection (Benson et al., 2009; Craven et al., 2012; Egan et al., 2011; Heimesaat et al., 2006). This phenomenon is not limited to γ-proteobacteria because many other clinically relevant species, such as Enterococcus faecalis and Clostridium difficile, can bloom during bouts of GI dysbiosis (Britton and Young, 2012; Ubeda et al., 2010). How the immune system deals with these opportunistic residents and in particular whether tolerance to antigen associated with these bacteria is maintained during inflammation are of central importance to our understanding of mucosal inflammatory disorders.
Environmentally Induced Breach of the Mucosal Firewall
At homeostasis and in highly controlled experimental settings, the mucosal firewall ensures that bacterial translocation is strictly limited to the intestinal tissue and associated lymphoid structures (Macpherson and Uhr, 2004). In mouse models, this system has been proven extraordinarily robust because only complete deficiency of key innate and adaptive mucosal immune mechanisms leads to systemic commensal specific antibody responses (Slack et al., 2009). Accordingly, in mice raised under pathogen-free conditions, commensal-specific T cells present outside the mucosa remain naïve despite the presence of commensal antigens in the GI tract (Cong et al., 2009; Hand et al., 2012). However, controlled environments that lack pathogens completely represent a highly artificial setting to understand tissue homeostasis and local immune responses. Indeed, we are now beginning to appreciate that under physiological conditions, systemic translocation of commensals and microbial products beyond the mucosa and GALT is a more common occurrence than initially postulated. Translocation of bacterial products to the systemic circulation has been associated clinically with a diverse set of circumstances such as alcohol abuse and cirrhosis, chronic nonsteroidal anti-inflammatory drug (NSAID) use, malnutrition, chronic inflammation, extreme exercise regimens, and in particular infections (Arakawa et al., 2012; Berklery et al., 2005; Hughes et al., 2009; Purohit et al., 2008; Nieman et al., 2006; Wong, 2012; Zimmermann et al., 2012). In several murine models of GI infections, such as T. gondii and Yersinia pseudotuberculosis, immunopathology can induce the translocation of commensal bacteria (Hand et al., 2012; Heimesaat et al., 2006; Meinzer et al., 2012). Chronic GI barrier dysfunction is also observed during HIV infection in humans and in simian immunodeficiency virus (SIV) infection of rhesus macaques (Brenchley et al., 2006; Brenchley and Douek, 2012; Estes et al., 2010). Barrier sites including the gut, skin, and airways are primary sites for infections. It is estimated that a child will suffer 10–15 diarrheal episodes on average before the age of 5 years; all of them potentially associated with transient commensal translocation (Kosek et al., 2003; Vernacchio et al., 2006), which if added together with common skin and lung infections, provides ample opportunity for exposure of the immune system to commensal antigens under inflammatory conditions.
Commensal-Specific T Cell Responses During Infection and Inflammation
Infections represent highly volatile situations for the mucosal immune system, because pathogens and commensals share the same inflamed environment. The potential danger of this situation is illustrated by studies that suggest that oral tolerance to newly introduced food antigens breaks down during acute GI infection (Oldenhove et al., 2009; Severance et al., 2012). Recent evidence suggests that in a similar manner, tolerance to commensal-derived antigens may be lost during acute infections. In a mouse model of T. gondii infection, Treg cells are lost, commensals translocate, and the immune system becomes unable to discriminate between commensals and pathogen-derived antigens (Hand et al., 2012) (Figure A1-1B). During this highly Th1 polarized infection, commensal-specific T cells also develop as Th1 cells according to cues provided by the inflammatory milieu, rather than Th17 cells or Treg cells, as previous studies have associated with commensal-driven responses (Atarashi et al., 2011; Ivanov et al., 2009). The idea that innate inflammatory cues drive the fate of commensal-specific T cells rather than their specificity is further supported by the observation that physical breakdown of the intestinal barrier via the administration of dextran sodium sulfate (DSS) resulted in the activation of commensal-specific T cells that differentiated toward the Th17 fate (Hand et al., 2012). Thus, the immune response to GI pathogens is associated with parallel responses against commensal-derived antigens that develop according to the inflammatory milieu. The commensal antigen followed in that study is expressed by the Clostridium cluster XIV class of bacteria that is known to live in the mucus layer of the intestinal mucosa and is a dominant antigen in CD (Atarashi et al., 2011; Lodes et al., 2004). It will be important to determine with future studies whether these responses are directed against all commensal antigens or are limited to the most prevalent or accessible antigens.
Commensal-Specific Memory T Cell Responses
One remarkable feature of all barrier sites is their ability to repair efficiently after inflammation or breach. In the GI tract, this implies that after acute tissue damage and transiently increased exposure to commensals, physical segregation between the flora and the immune system is rapidly restored. In the absence of chronic exposure to antigen, activated lymphocytes can survive long term as memory populations capable of rapid reactivation and proliferation. Indeed, following GI infection with T. gondii, commensal-specific CD4 T cells can persist long term in both the GI tract and secondary lymphoid tissue and maintain the ability to become activated, express Th1 inflammatory cytokines, and proliferate upon secondary encounter with their cognate antigen (Hand et al., 2012).
Much like pathogen-specific CD4 T cells and in contrast to virus-specific CD8 memory T cells, commensal-specific memory T cells declined steadily over time (Hand et al., 2012; Homann et al., 2001; Pepper et al., 2009). CD4 T cells
carry out the complex task of discriminating pathogenic organisms from benign organisms in the face of a constantly changing environment; therefore, perhaps development of CD4 memory reflects this necessity for flexibility. An evolving pool of specificities within the Treg and CD4 effector compartment may allow for the maintenance of tolerance and barrier function in the context of fluctuating commensal populations and intermittent infection. Such flexible repertoire has been proposed for mucosal IgA responses that lack the memory characteristics associated with CD8 T cells and are able to respond to flux in the commensal microbiota composition. Indeed, established IgA-producing clones are outcompeted by novel antibacterial responses, allowing the mucosal immune system to respond to a constantly changing microbiota (Hapfelmeier et al., 2010).
The physiological consequence of long-term CD4+ T cell memory against commensals remains to be addressed. Due to the extraordinary antigenic diversity of the host microbiota at all body surfaces and the prevalence of infections, a significant fraction of memory cells are expected to be commensal specific and could develop over time in response to successive infections and/or various barrier breaches (Figure A1-2). In support of this hypothesis, healthy human serum contains antibodies specific to skin and intestinal microbiota (Zimmermann et al., 2012). Thus, primary exposure to a pathogen in the skin, lung and GI tract is likely to occur in the context of a much broader recall response against commensal bacteria. One possible consequence of these responses may be the induction of heterologous memory wherein antigen-specific responses against previously encountered commensal bacteria could drive the rapid production of inflammatory cytokines, leading to increased protection against secondary infection and associated translocation of commensal bacteria (Figure A1-2). Colon-resident Th17 cells have been shown to contribute to early protection against enteric pathogens, although whether these cells are specific to commensal antigens is not clear (Geddes et al., 2011). Further contributing to the possibility of heterologous protection against infection by commensal-specific T cells, a recent study has suggested that CD4 T cell clones that are cross-reactive to commensals and viruses are common in healthy patients (Su et al., 2013). By contrast, aberrant accumulation of commensal-specific T cells under defined settings may lead to several pathogenic consequences, such as IBD and psoriasis (Sartor, 2006). Exploration of the antigen specificity of the memory cell compartment of lymphocytes residing at all barrier sites would inform us of the potential impact of these commensal-specific T cell responses on tissue physiology and subsequent pathologies. It would be of particular interest to address how responses to conserved bacterial antigens across barrier surfaces affect local and systemic tissue responses over time.
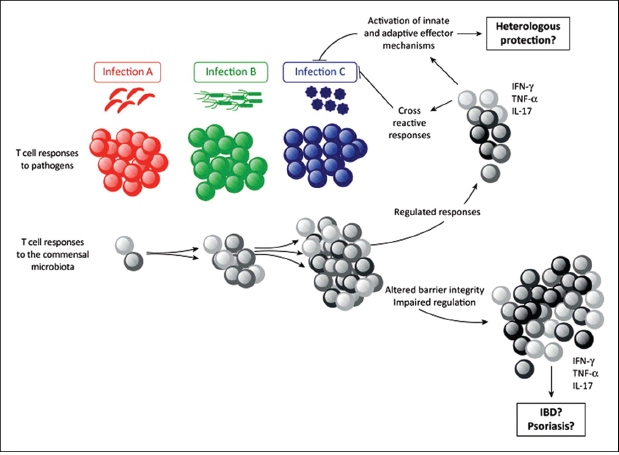
FIGURE A1-2 Potential consequences of commensal-specific memory T cells. During infection at barrier sites (gut, skin, airway, and genital mucosa), immune responses against the invading agent can be associated with specific T cell responses against a large number of coincident commensal antigens. These commensal-specific effector T cell responses can persist as memory cells that upon subsequent infection will be recalled as secondary commensal-specific effectors, alongside the priming of a novel immune response to the invasive pathogen. Therefore, each infection at barrier surfaces represents an additional opportunity for the reactivation of commensal-specific T cells. Given the extraordinary number of commensal antigens, these responses may represent a significant proportion of memory T cells. If properly controlled, commensal-specific effector/memory cells could contribute to protection against infections by promoting innate and adaptive effector mechanisms that assist in the clearance of the pathogen. Further, commensal memory responses could be protective due to cross-reactivity with pathogen-derived antigens. By contrast, in situations where commensal-specific T cells become dysregulated due to impaired regulatory pathways and/or barrier function, these T cells could drive chronic pathology such as inflammatory bowel disease or psoriasis.
Commensal-Specific T Cells and GI Disorders
IBD refers to a group of chronic inflammatory disorders affecting the GI tract (Kaser et al., 2010). There are two main clinical forms of IBD: CD, which can affect any part of the GI tract, and ulcerative colitis (UC), in which pathology is restricted to the colonic mucosa (Kaser et al., 2010). The etiology of these
disorders is complex and believed to be the consequence of genetic factors, the host immune system, and environmental factors such as the microbiota (Maloy and Powrie, 2011). Individual genomewide association studies have revealed that a large number of risk factors are associated with active immune responses and altered barrier function (Anderson et al., 2011; Barrett et al., 2008; Franke et al., 2010; Jostins et al., 2012; McGovern et al., 2010). In light of recent findings, commensal-specific T cells may represent an important component of the disease. In mouse models of colitis, it is well known that the commensal microbiota is necessary for the induction of disease via activation of both innate and adaptive immune mechanisms. Commensal-specific CD4 T cells have been identified in murine models of colitis and in some models are required to drive disease (Kim et al., 2007; Kullberg et al., 2002; Yoshida et al., 2002). For example, immortalized commensal-specific CD4 T cell clones derived from colitis-prone mice are capable of transferring disease to wild type mice (Cong et al., 1998). Activated T cells can also induce colitis in nucleotide-binding oligomerization domain-containing protein 2 NOD2-deficient mice in response to antigens carried by commensal bacteria (Watanabe et al., 2006). Studies comparing TCR transgenic cells in the recombination activating gene/severe combined immunodeficiency (Rag/SCID) model of colitis indicate that although commensal specificity is unnecessary for the proliferation and accumulation of CD4 T cells at mucosal sites, cognate antigen responses are required for the induction of colitis (Feng et al., 2010). Additionally, germ-free IL-10 knockout mice monocolonized with either E. coli or Ent. faecalis have been shown to develop CD4 T cell responses against the colonizing bacteria (Kim et al., 2007). Furthermore, feeding IL-10 knockout mice a diet high in milk fat is associated with increased Th1 T cell responses to a particular bacteria, Bilophila wadsworthia, although whether these are strictly antigen specific remains unclear (Devkota et al., 2012). A single genus of bacteria, Helicobacter, is sufficient for the induction of colitis in both the IL-10 and Rag/SCID models of colitis, and T cells specific to Helicobacter are present and sufficient for disease in animal models of IBD (Kullberg et al., 2001, 2003; Powrie and Uhlig, 2004). Finally, in mice in which both IL-10 and TGFβ signaling is deficient in T cells, spontaneous colitis occurs that is directly dependent upon the presence of bacteria of the genus Bacteroides (Bloom et al., 2011). One key point taken from these studies is that multiple bacterial types comprising several distantly related bacterial phyla can promote colitis, possibly via the induction of specific CD4 T cell responses, supporting the idea that IBD may develop as a consequence of a broad loss of tolerance to the commensal microbiota (Sartor, 2006). In support of this hypothesis, higher titers of commensal-specific antibodies are found in the serum of CD patients compared to healthy donors, and the measurement of antibody responses to a panel of seemingly unrelated commensal-derived antigens is commonly used as a diagnostic method for IBD (Iskandar and Ciorba, 2012; Zimmermann et al., 2012). However, commensal-specific responses are observed in healthy individuals, suggesting that on its own, immunity to commensals is not
sufficient for the induction of diseases (Haas et al., 2011; Sartor, 2006). Thus, under normal settings, effector and memory responses against commensals that have been induced by infections or injuries are likely held in check by the combined effect of the mucosal firewall and active mechanisms of tolerance. IBD, however, could be the result of the environmental activation of commensal-specific T cells in the context of a genetic predisposition for intestinal pathology and in particular defects in repair and immune regulation. This multiple hit mechanism of disease induction is supported by data that show mucosal viral infection, the commensal microbiota, and diminished Paneth cell function due to reduced expression of autophagy-related protein 16 L1 (ATG16L1), converge to increase disease severity in experimental colitis (Cadwell et al., 2010). However, despite clear connections between commensal-specific T cell responses and IBD, more remains to be done to understand how immunity to commensals could be causative in disease and how commensal-specific effector T cells are regulated under homeostatic conditions (Figure A1-1C).
Concluding Remarks
Adaptive immunity, as defined by the presence of lymphocytes with rearranged antigen receptors of near infinite specificity, is a characteristic of organisms that carry complex populations of microbial symbionts upon their mucosal surfaces. One might speculate that the coevolution between the adaptive immune system and commensal microbiota was primarily driven by the difficulty of maintaining and controlling such a complex relationship. However, barrier surfaces are not static and are often perturbed by environmental or infectious challenges, causing changes to the commensal microbiota and increasing tissue permeability. In Westernized countries, increased use of antibiotics, reduced worm infections, and drastic changes in nutrition have imposed massive changes in our relations with these organisms. Our understanding of commensal–immune interactions under these highly fluctuating circumstances is still in its infancy and much remains to be understood about commensal-specific responses and their consequences for human health.
Acknowledgments
This work was supported by the Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health. We would like to thank the members of the Belkaid laboratory for helpful discussions and in particular Drs. A. Poholek, E. Wohlfert, and S. Naik for critical reading.
References
Abt, M.C. et al. (2012) Commensal bacteria calibrate the activation threshold of innate antiviral immunity. Immunity 37, 158–170
Anderson, C.A. et al. (2011) Meta-analysis identifies 29 additional ulcerative colitis risk loci, increasing the number of confirmed associations to 47. Nat. Genet. 43, 246–252
Arakawa, T. et al. (2012) Small intestinal injury caused by NSAIDs/ aspirin: finding new from old. Curr. Med. Chem. 19, 77–81
Arthur, J.C. et al. (2012) Intestinal inflammation targets cancer-inducing activity of the microbiota. Science 338, 120–123
Atarashi, K. et al. (2011) Induction of colonic regulatory T cells by indigenous Clostridium species. Science 331, 337–341
Backhed, F. et al. (2005) Host-bacterial mutualism in the human intestine. Science 307, 1915–1920
Barrett, J.C. et al. (2008) Genome-wide association defines more than 30 distinct susceptibility loci for Crohn’s disease. Nat. Genet. 40, 955–962
Benson, A. et al. (2009) Gut commensal bacteria direct a protective immune response against Toxoplasma gondii. Cell Host Microbe 6, 187–196
Benson, M.J. et al. (2007) All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J. Exp. Med. 204, 1765–1774
Berkley, J.A. et al. (2005) Bacteremia among children admitted to a rural hospital in Kenya. N. Engl. J. Med. 352, 39–47
Bettelli, E. et al. (2006) Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238
Bilate, A.M. and Lafaille, J.J. (2012) Induced CD4+Foxp3+ regulatory T cells in immune tolerance. Annu. Rev. Immunol. 30, 733–758
Bloom, S.M. et al. (2011) Commensal Bacteroides species induce colitis in host-genotype-specific fashion in a mouse model of inflammatory bowel disease. Cell Host Microbe 9, 390–403
Brandl, K. et al. (2008) Vancomycin-resistant enterococci exploit antibiotic-induced innate immune deficits. Nature 455, 804–807
Brenchley, J.M. and Douek, D.C. (2012) Microbial translocation across the GI tract. Annu. Rev. Immunol. 30, 149–173
Brenchley, J.M. et al. (2006) Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat. Med. 12, 1365–1371
Britton, R.A. and Young, V.B. (2012) Interaction between the intestinal microbiota and host in Clostridium difficile colonization resistance. Trends Microbiol. 20, 313–319
Cadwell, K. et al. (2010) Virus-plus-susceptibility gene interaction determines Crohn’s disease gene Atg16L1 phenotypes in intestine. Cell 141, 1135–1145
Chow, J. et al. (2011) Pathobionts of the gastrointestinal microbiota and inflammatory disease. Curr. Opin. Immunol. 23, 473–480
Clemente, J.C. et al. (2012) The impact of the gut microbiota on human health: an integrative view. Cell 148, 1258–1270
Cong, Y. et al. (1998) CD4+ T cells reactive to enteric bacterial antigens in spontaneously colitic C3H/HeJBir mice: increased T helper cell type 1 response and ability to transfer disease. J. Exp. Med. 187, 855–864
Cong, Y. et al. (2009) A dominant, coordinated T regulatory cell-IgA response to the intestinal microbiota. Proc. Natl. Acad. Sci. U.S.A. 106, 19256–19261
Coombes, J.L. et al. (2007) A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF- {beta}- and retinoic acid-dependent mechanism. J. Exp. Med. 204, 1757–1764
Costello, E.K. et al. (2012) The application of ecological theory toward an understanding of the human microbiome. Science 336, 1255–1262
Craven, M. et al. (2012) Inflammation drives dysbiosis and bacterial invasion in murine models of ileal Crohn’s disease. PLoS ONE 7, e41594
Denning, T.L. et al. (2011) Functional specializations of intestinal dendritic cell and macrophage subsets that control Th17 and regulatory T cell responses are dependent on the T cell/APC ratio, source of mouse strain, and regional localization. J. Immunol. 187, 733–747
Dethlefsen, L. and Relman, D.A. (2011) Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl. Acad. Sci. U.S.A. 108 (Suppl. 1), 4554–4561
Devkota, S. et al. (2012) Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice. Nature 487, 104–108
Eckburg, P.B. et al. (2003) Archaea and their potential role in human disease. Infect. Immun. 71, 591–596
Egan, C.E. et al. (2011) Synergy between intraepithelial lymphocytes and lamina propria T cells drives intestinal inflammation during infection. Mucosal Immunol. 4, 658–670
Estes, J.D. et al. (2010) Damaged intestinal epithelial integrity linked to microbial translocation in pathogenic simian immunodeficiency virus infections. PLoS Pathog. 6, e1001052
Feng, T. et al. (2010) Microbiota innate stimulation is a prerequisite for T cell spontaneous proliferation and induction of experimental colitis. J. Exp. Med. 207, 1321–1332
Feng, T. et al. (2011) Interleukin-12 converts Foxp3+ regulatory T cells to interferon-gammaproducing Foxp3+ T cells that inhibit colitis. Gastroenterology 140, 2031–2043
Fossiez, F. et al. (1996) T cell interleukin-17 induces stromal cells to produce proinflammatory and hematopoietic cytokines. J. Exp. Med. 183, 2593–2603
Foulongne, V. et al. (2012) Human skin microbiota: high diversity of DNA viruses identified on the human skin by high throughput sequencing. PLoS ONE 7, e38499
Franke, A. et al. (2010) Genome-wide meta-analysis increases to 71 the number of confirmed Crohn’s disease susceptibility loci. Nat. Genet. 42, 1118–1125
Gaboriau-Routhiau, V. et al. (2009) The key role of segmented filamentous bacteria in the coordinated maturation of gut helper T cell responses. Immunity 31, 677–689
Garrett, W.S. et al. (2010) Enterobacteriaceae act in concert with the gut microbiota to induce spontaneous and maternally transmitted colitis. Cell Host Microbe 8, 292–300
Geddes, K. et al. (2011) Identification of an innate T helper type 17 response to intestinal bacterial pathogens. Nat. Med. 17, 837–844
Geuking, M.B. et al. (2011) Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunity 34, 794–806
Grice, E.A. and Segre, J.A. (2012) The human microbiome: our second genome. Annu. Rev. Genomics Hum. Genet. 13, 151–170
Haas, A. et al. (2011) Systemic antibody responses to gut commensal bacteria during chronic HIV-1 infection. Gut 60, 1506–1519
Hall, J.A. et al. (2008) Commensal DNA limits regulatory T cell conversion and is a natural adjuvant of intestinal immune responses. Immunity 29, 637–649
Hall, J.A. et al. (2011) Essential role for retinoic acid in the promotion of CD4(+) T cell effector responses via retinoic acid receptor alpha. Immunity 34, 435–447
Hand, T.W. et al. (2012) Acute gastrointestinal infection induces long-lived microbiota-specific T cell responses. Science 337, 1553–1556
Hapfelmeier, S. et al. (2010) Reversible microbial colonization of germ-free mice reveals the dynamics of IgA immune responses. Science 328, 1705–1709
Heimesaat, M.M. et al. (2006) Gram-negative bacteria aggravate murine small intestinal Th1-type immunopathology following oral infection with Toxoplasma gondii. J. Immunol. 177, 8785–8795
Hill, D.A. et al. (2010) Metagenomic analyses reveal antibiotic-induced temporal and spatial changes in intestinal microbiota with associated alterations in immune cell homeostasis. Mucosal Immunol. 3, 148–158
Homann, D. et al. (2001) Differential regulation of antiviral T-cell immunity results in stable CD8+ but declining CD4+ T-cell memory. Nat. Med. 7, 913–919
Hooper, L.V. et al. (2012) Interactions between the microbiota and the immune system. Science 336, 1268–1273
Hu, W. et al. (2011) Priming microenvironments dictate cytokine requirements for T helper 17 cell lineage commitment. Immunity 35, 1010–1022
Hughes, S.M. et al. (2009) Dendritic cell anergy results from endotoxemia in severe malnutrition. J. Immunol. 183, 2818–2826
Ichinohe, T. et al. (2011) Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc. Natl. Acad. Sci. U.S.A. 108, 5354–5359
Iliev, I.D. et al. (2012) Interactions between commensal fungi and the C-type lectin receptor Dectin-1 influence colitis. Science 336, 1314–1317
Ishigame, H. et al. (2009) Differential roles of interleukin-17A and - 17F in host defense against mucoepithelial bacterial infection and allergic responses. Immunity 30, 108–119
Iskandar, H.N. and Ciorba, M.A. (2012) Biomarkers in inflammatory bowel disease: current practices and recent advances. Transl. Res. 159, 313–325
Ivanov, I.I. et al. (2008) Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe 4, 337–349
Ivanov, I.I. et al. (2009) Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498
Jostins, L. et al. (2012) Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 491, 119–124
Kaser, A. et al. (2010) Inflammatory bowel disease. Annu. Rev. Immunol. 28, 573–621
Kim, S.C. et al. (2007) Dual-association of gnotobiotic IL-10-/- mice with 2 nonpathogenic commensal bacteria induces aggressive pancolitis. Inflamm. Bowel Dis. 13, 1457–1466
Konkel, J.E. and Chen, W. (2011) Balancing acts: the role of TGF-beta in the mucosal immune system. Trends Mol. Med. 17, 668–676
Korn, T. et al. (2009) IL-17 and Th17 Cells. Annu. Rev. Immunol. 27, 485–517
Kosek, M. et al. (2003) The global burden of diarrhoeal disease, as estimated from studies published between 1992 and 2000. Bull. World Health Organ. 81, 197–204
Kullberg, M.C. et al. (2001) Helicobacter hepaticus-induced colitis in interleukin-10-deficient mice: cytokine requirements for the induction and maintenance of intestinal inflammation. Infect. Immun. 69, 4232–4241
Kullberg, M.C. et al. (2002) Bacteria-triggered CD4(+) T regulatory cells suppress Helicobacter hepaticus-induced colitis. J. Exp. Med. 196, 505–515
Kullberg, M.C. et al. (2003) Induction of colitis by a CD4+ T cell clone specific for a bacterial epitope. Proc. Natl. Acad. Sci. U.S.A. 100, 15830–15835
Lathrop, S.K. et al. (2011) Peripheral education of the immune system by colonic commensal microbiota. Nature 478, 250–254
Ley, R.E. et al. (2006) Ecological and evolutionary forces shaping microbial diversity in the human intestine. Cell 124, 837–848
Lochner, M. et al. (2011) Restricted microbiota and absence of cognate TCR antigen leads to an unbalanced generation of Th17 cells. J. Immunol. 186, 1531–1537
Lodes, M.J. et al. (2004) Bacterial flagellin is a dominant antigen in Crohn disease. J. Clin. Invest. 113, 1296–1306
Lozupone, C.A. et al. (2012) Diversity, stability and resilience of the human gut microbiota. Nature 489, 220–230
Lupp, C. et al. (2007) Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe 2, 119–129
Macpherson, A.J. and Uhr, T. (2004) Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 303, 1662–1665
Macpherson, A.J. et al. (2009) The mucosal firewalls against commensal intestinal microbes. Semin. Immunopathol. 31, 145–149
Maloy, K.J. and Powrie, F. (2011) Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature 474, 298–306
Maynard, C.L. and Weaver, C.T. (2009) Intestinal effector T cells in health and disease. Immunity 31, 389–400
Mazmanian, S.K. et al. (2005) An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122, 107–118
Mazmanian, S.K. et al. (2008) A microbial symbiosis factor prevents intestinal inflammatory disease. Nature 453, 620–625
McGovern, D.P. et al. (2010) Genome-wide association identifies multiple ulcerative colitis susceptibility loci. Nat. Genet. 42, 332–337
Meinzer, U. et al. (2012) Yersinia pseudotuberculosis effector YopJ subverts the Nod2/RICK/TAK1 pathway and activates caspase-1 to induce intestinal barrier dysfunction. Cell Host Microbe 11, 337–351
Molloy, M.J. et al. (2012) Intestinal microbiota: shaping local and systemic immune responses. Semin. Immunol. 24, 58–66
Mora, J.R. et al. (2006) Generation of gut-homing IgA-secreting B cells by intestinal dendritic cells. Science 314, 1157–1160
Mucida, D. et al. (2007) Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260
Mucida, D. et al. (2009) Retinoic acid can directly promote TGF-beta-mediated Foxp3(+) Treg cell conversion of naive T cells. Immunity 30, 471–473
Murphy, K.M. and Stockinger, B. (2010) Effector T cell plasticity: flexibility in the face of changing circumstances. Nat. Immunol. 11, 674–680
Naik, S. et al. (2012) Compartmentalized control of skin immunity by resident commensals. Science 337, 1115–1119
Nieman, D.C. et al. (2006) Ibuprofen use, endotoxemia, inflammation, and plasma cytokines during ultramarathon competition. Brain Behav. Immun. 20, 578–584
O’Mahony, C. et al. (2008) Commensal-induced regulatory T cells mediate protection against pathogen-stimulated NF-kappaB activation. PLoS Pathog. 4, e1000112
Ochoa-Reparaz, J. et al. (2010) Central nervous system demyelinating disease protection by the human commensal Bacteroides fragilis depends on polysaccharide A expression. J. Immunol. 185, 4101–4108
Oldenhove, G. et al. (2009) Decrease of Foxp3+ Treg cell number and acquisition of effector cell phenotype during lethal infection. Immunity 31, 772–786
Pepper, M. et al. (2009) Different routes of bacterial infection induce long-lived TH1 memory cells and short-lived TH17 cells. Nat. Immunol. 11, 83–89
Peterson, D.A. et al. (2007) IgA response to symbiotic bacteria as a mediator of gut homeostasis. Cell Host Microbe 2, 328–339
Powrie, F. and Uhlig, H. (2004) Animal models of intestinal inflammation: clues to the pathogenesis of inflammatory bowel disease. Novartis Found. Symp. 263, 164–174
Purohit, V. et al. (2008) Alcohol, intestinal bacterial growth, intestinal permeability to endotoxin, and medical consequences: summary of a symposium. Alcohol 42, 349–361
Qin, J. et al. (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65
Qin, J. et al. (2012) A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60
Reyes, A. et al. (2010) Viruses in the faecal microbiota of monozygotic twins and their mothers. Nature 466, 334–338
Rolhion, N. and Darfeuille-Michaud, A. (2007) Adherent-invasive Escherichia coli in inflammatory bowel disease. Inflamm. Bowel Dis. 13, 1277–1283
Round, J.L. and Mazmanian, S.K. (2010) Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl. Acad. Sci. U.S.A. 107, 12204–12209
Round, J.L. et al. (2011) The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science 332, 974–977
Sartor, R.B. (2006) Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis. Nat. Clin. Pract. Gastroenterol. Hepatol. 3, 390–407
Severance, E.G. et al. (2012) Anti-gluten immune response following Toxoplasma gondii infection in mice. PLoS ONE 7, e50991
Slack, E. et al. (2009) Innate and adaptive immunity cooperate flexibly to maintain host-microbiota mutualism. Science 325, 617–620
Sonnenberg, G.F. et al. (2011) Border patrol: regulation of immunity, inflammation and tissue homeostasis at barrier surfaces by IL-22. Nat. Immunol. 12, 383–390
Sonnenberg, G.F. et al. (2012) Innate lymphoid cells promote anatomical containment of lymphoid-resident commensal bacteria. Science 336, 1321–1325
Stecher, B. and Hardt, W.D. (2011) Mechanisms controlling pathogen colonization of the gut. Curr. Opin. Microbiol. 14, 82–91
Su, L.F. et al. (2013) Virus-specific CD4(+) memory-phenotype T cells are abundant in unexposed adults. Immunity 38, 373–383
Sun, C.M. et al. (2007) Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 204, 1775–1785
Suzuki, K. et al. (2004) Aberrant expansion of segmented filamentous bacteria in IgA-deficient gut. Proc. Natl. Acad. Sci. U.S.A. 101, 1981–1986
Tsuji, M. et al. (2009) Preferential generation of follicular B helper T cells from Foxp3+ T cells in gut Peyer’s patches. Science 323, 1488–1492
Ubeda, C. et al. (2010) Vancomycin-resistant Enterococcus domination of intestinal microbiota is enabled by antibiotic treatment in mice and precedes bloodstream invasion in humans. J. Clin. Invest. 120, 4332–4341
Vaishnava, S. et al. (2011) The antibacterial lectin RegIIIgamma promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258
Vernacchio, L. et al. (2006) Diarrhea in American infants and young children in the community setting: incidence, clinical presentation and microbiology. Pediatr. Infect. Dis. J. 25, 2–7
Watanabe, T. et al. (2006) Nucleotide binding oligomerization domain 2 deficiency leads to dysregulated TLR2 signaling and induction of antigen-specific colitis. Immunity 25, 473–485
Weiner, H.L. et al. (2011) Oral tolerance. Immunol. Rev. 241, 241–259
Wong, F. (2012) Recent advances in our understanding of hepatorenal syndrome. Nat. Rev. Gastroenterol. Hepatol. 9, 382–391
Wu, H.J. et al. (2010) Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 32, 815–827
Yoshida, M. et al. (2002) Differential localization of colitogenic Th1 and Th2 cells monospecific to a microflora-associated antigen in mice. Gastroenterology 123, 1949–1961
Zheng, Y. et al. (2008) Interleukin-22 mediates early host defense against attaching and effacing bacterial pathogens. Nat. Med. 14, 282–289
Zhou, L. et al. (2008) TGF-beta-induced Foxp3 inhibits T(H)17 cell differentiation by antagonizing RORgammat function. Nature 453, 236–240
Zimmermann, K. et al. (2012) Systemic antibody responses to gut microbes in health and disease. Gut Microbes 3, 42–47

















