2
Stem Cell Therapies—Knowns and Unknowns
A variety of stem cell–based products are being proffered to patients for the treatment of a wide spectrum of disease, though many of these therapies have not undergone extensive testing of their effectiveness and safety (for a summary of highlighted points, see Box 2-1). Individual speakers at the workshop provided broad overviews of the process for developing stem cell–based therapies and the current state of clinical offerings. Much of the excitement surrounding the field is being driven by hype, some participants said. Yet important research is also being conducted that could lead to highly beneficial medical advances.
- Objective outcome measures would be useful for assessing the effectiveness of stem cell therapies. (Wagner)
- Marketing claims by clinics offering stem cell therapies are not necessarily supported by clinical evidence in the scientific literature and may be misleading to patients. (Caulfield)
- Unproven stem cell treatments can harm patients by leading to complications such as tumors, meningitis, or even death; however, patients who have no other treatment options may be willing to take these risks if there is a slight chance of success. (Caulfield)
- Improved communication of clear information about treatment benefits and risks as well as about the value of safety regulations would be useful for patients. (Caulfield)
STEM CELLS AS REGULATED THERAPEUTICS
There are a variety of diseases for which conventional medicine offers no available treatment options, said John Wagner, professor of pediatrics and director of the Division of Hematology–Oncology and Blood and Marrow Transplantation at the University of Minnesota. When patients are desperate, they may turn to experimental or unproven treatments because the therapies afford them hope.
Wagner described his experiences treating patients with epidermolysis bullosa as an example of the process and challenges for developing a stem cell therapy. Epidermolysis bullosa is a mutilating disease of the skin and mucosa that results in shortened survival, a life filled with pain, the inability to perform normal daily tasks, and death due to infection or cancer (Fine et al., 2008; Tolar and Wagner, 2012). As Wagner stated, these patients “would do anything at all for any price. It does not matter. [Any treatment could be made up] and they would be there tomorrow.” When Wagner and his colleagues were organizing initial clinical trials to test a potential treatment for the disease, he even had patients offering $1 million to be included because they were so desperate.
Developing a stem cell therapy that can be offered to the public usually begins with an idea that has typically been conceived in a laboratory. For Wagner and his colleagues, they hypothesized that stem cells might be used to repair skin in a mouse model of epidermolysis bullosa. They evaluated the effect of treating these mice with different stem cell populations and found a population of bone marrow cells that resulted in various improvements, such as longer survival times, the absence of blisters, and the appearance of anchoring fibrils, which are missing in disease-affected individuals because of a genetic mutation (Tolar et al., 2009). Even though only three of the treated mice survived, the FDA approved moving forward to clinical trials in humans because of the severity of the disease and the lack of alternative treatment options.
Wagner established the clinical trial under the auspices of an oversight committee which performed data safety and monitoring and reviewed each patient’s eligibility. Seven patients received stem cells in the first trial, and, according to Wagner, the treatment produced an “extraordinary” improvement.
Still, the medical community was skeptical of the results. Wagner and his colleagues therefore enrolled more patients, extended the monitoring period, and developed more objective measures for outcomes. Electron microscopy was used to demonstrate that anchoring fibrils were
present post-treatment. In patients whose hands had never formed fingers, it was demonstrated that fingers began to grow after the stem cell transplantation. In addition, Wagner and his colleagues built a device to conduct a blister test that measured the response of a patient’s skin to an external stimulus. This device in particular made the reports of improvement more objective and increased overall acceptance in the medical community.
Each step forward in moving a treatment toward approval requires regulatory review and additional expense, Wagner observed. Progressing from basic research to clinical research and then to Phase I, II, and III trials “is a laborious, long, expensive process.” The numbers of patients involved in each successive trial gets larger, as does the expense.
The strategic priorities of the FDA’s Center for Biologics Evaluation and Research are to enhance the nation’s public health preparedness, improve public health globally, improve the quality and safety of biological products, enhance the translation of innovative science and technology into products for patients, advance regulatory science, and strengthen human resources and performance (CBER, 2011). Ensuring safety, however, requires more than just monitoring clinical trials. The FDA must also oversee the manufacturing of products, which includes equipment monitoring, materials management, inventory management, document control, label control, facility control, quality assurance, and other activities. For example, stem cells have to be certified to be free of endotoxin, bacteria, and fungal elements and have certain surface markers before they can be released. These behind-the-scenes activities can be “a big deal,” Wagner said, “particularly if you are a small university laboratory.”
Therapies Under Development
At the time of the workshop, very few stem cell therapies had received approval from a regulatory oversight body, Wagner said. In North America, the FDA had approved Hemacord®, an umbilical cord blood product developed by the New York Blood Center, while Prochymal, which was developed by Osiris Therapeutics to treat acute graft-versus-host disease in children, had received market authorization from HealthCanada.1
__________________
1In addition to Hemacord®, four other hematopoietic progenitor cell products have been approved by the FDA. For a full list of approved cell therapy products see http://www.fda.gov/BiologicsBloodVaccines/CellularGeneTherapyProducts/Approved.Products/default. htm (accessed April 4, 2014).
The majority of stem cell therapies, Wagner said, have been approved outside the United States, with a disproportionate number occurring in South Korea. However, many of these are not really stem cell therapies, Wagner said. Instead, they use allogeneic amniotic membrane, autologous cultured epidermal cells and chondrocytes, or allogeneic bone. Clinical trial numbers indicate that a much broader developmental effort may be under way. A search of the website ClinicalTrials.gov for the term “stem cells” at the time of the workshop found 4,831 studies, though Wagner noted that not all of these were really using stem cells.
CLINICAL OFFERINGS USING STEM CELLS
The issue that the stem cell community is most concerned about is the marketing and sale of stem cell–based therapies that have not been shown to be effective or safe (Caulfield et al., 2012), said Timothy Caulfield, Canada Research Chair in Health Law and Policy and professor in the Faculty of Law and the School of Public Health at the University of Alberta. Desperate patients are searching the Internet and being directed to clinics that look legitimate and offer treatments for virtually everything—Alzheimer’s disease, autism, aging, immune deficiencies, liver disease, cancer, diabetes, hair loss, multiple sclerosis (MS), spinal cord injury, and stroke. “This is a marker of quackery, when you have a therapy that allegedly can cure anything,” said Caulfield.
A 2008 study found that the claims being made by stem cell purveyors were not supported by published clinical evidence (Lau et al., 2008), and a follow-up study found that marketing practices remained unchanged over the intervening time even though the issue of potentially fraudulent treatments had received increasing attention (Ogbogu et al., 2013). In some respects, Caulfield said, the claims had actually gotten worse, with Internet advertisements saying that the treatments were efficacious, routine, and low risk.
Many of these websites use what Wagner characterized as “debate tactics.” For example, they question the motives of an opponent by claiming that the FDA and the pharmaceutical industry are colluding. They cite irrelevant facts or logics, such as claiming that cord blood can cure spinal cord injury because of its effectiveness against leukemia, Caulfield said, or draw on the opinions of unqualified individuals. They play on long-held fantasies and fears, such as implying that a good parent would do anything possible for their child, and they imply that people
who do not pursue a miraculous cure have given up or succumbed to hopelessness.
However, press accounts of stem cell therapies and the clinics in which they are delivered are, with a few notable exceptions, overwhelming positive (see Figure 2-1). Headlines such as “Stem cell miracle gives gift of sight to toddler”2 make stem cells seem cutting edge, exciting, and legitimate, said Caulfield. Feature stories in magazines, the promise of miracle cures, and promotions by athletes and other celebrities have fed the enthusiasm and hype for this field.
Currently, the words “stem cell” and “regeneration” are being used as marketing tools to play on the excitement that surrounds the field. They are terms that have cultural traction and can influence how people think about an offered cure, Caulfield said. “I call it ‘scienceploitation.’
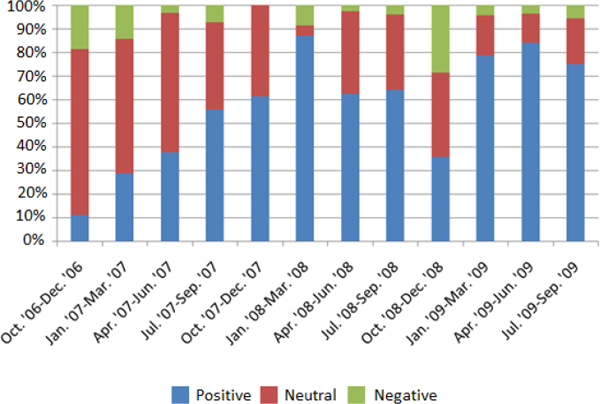
FIGURE 2-1 The tone of news media reports that covered stem cell therapies has been largely positive.
SOURCE: Timothy Caulfield, IOM, NAS, and ISSCR workshop presentation on November 18, 2013. Adapted by permission from Macmillan Publishers Ltd: Nature Biotechnology (Zarzeczny et al., 2010), copyright 2010.
__________________
2See http://www.mirror.co.uk/news/uk-news/stem-cell-miracle-gives-gift-380706 (accessed March 6, 2014).
It happened with electricity. It happened with magnetism. It happened with radioactivity. . . . Any time there is a scientific development that is exciting and sexy, people use it to profit.” One way to deal with this would be to pick some clear examples where therapies do not work and make definitive statements in those areas, Caulfield said.
The Number of Patients Seeking Stem Cell Treatments and the Effects on Those Patients
It is very difficult to determine the number of patients who are receiving stem cell treatments, either in absolute terms or compared to other forms of treatments that patients may be seeking in overseas clinics, Caulfield said. There are currently no registries in which this information is recorded, noted Alan Petersen, professor of sociology in the School of Political and Social Inquiry at Monash University in Melbourne, Australia. The field thus relies on indirect information, which makes it difficult to obtain quantifiable data. For example, Caulfield said, one clinic in China has claimed to have seen more than 5,000 patients between 2005 and 2009, but determining the veracity of this claim is difficult. Moreover, patients often do not want their doctors or their patient communities to know that they have sought out these treatments. However, Caulfield said, “if you believe [the claims], the numbers are big.”
The home countries of patients are difficult to ascertain, but some evidence suggests that the majority of patients are traveling from developed countries to developing countries, Petersen said. Stem cell clinics are a global phenomenon, Caulfield said, with clinics spread around the world (Regenberg et al., 2009). A survey of 224 patients, most of whom were from the United States, Canada, and the United Kingdom, found that about 60 percent were male and about 44 percent were minors (Zarzeczny et al., 2010). Forty-five percent of these patients traveled to clinics in China, with Mexico, Germany, the Dominican Republic, and India the next most common destinations. The most common conditions being treated were optical disorders and blindness, paralysis, multiple sclerosis, cerebral palsy, and brain injuries or damage, Caulfield said. A separate analysis of the websites advertising stem cell treatments and the blogs of patients who receive treatments at these clinics also concluded that the clinics are primarily located in Asian countries, particularly China and India, but also found Central America and Caribbean countries to be major destinations, Petersen said (Levine and Wolf, 2012). China has been a hotbed for stem cell tourism, added I. Glenn Cohen, assistant pro-
fessor at Harvard Law School. More than 200 institutions in China are offering treatments. Russia also has hundreds of unlicensed clinics that are offering stem cell treatments, and many of these clinics employ practitioners without medical qualifications, he said.
In addition, some stem cell providers are offering treatments in developed countries by exploiting regulatory loopholes, Petersen said. For example, some patients in his study were discovering through the Internet that trials were being conducted in the United States, and when they talked with the scientists they were told that they could be treated outside the trial.
Receiving stem cell treatments has resulted in harm to patients, both Caulfield and Petersen reported. Complications have included tumor growth, meningitis, and even death (Amariglio et al., 2009; Bohgaki et al., 2007; Mendpara et al., 2002; Thirabanjasak et al., 2010). One woman had bone fragments growing in her eye after a cosmetic procedure (Jabr, 2012). There is financial harm as well, with an individual treatment costing between $20,000 and $50,000, not including travel, time away from work and family, and potential long-term physical harms. More broadly, harms to patients have eroded public trust and the legitimacy of the field and have created great challenges for the clinical trial and regulatory process, Caulfield said.
Caulfield’s colleagues and others have done an analysis of online blogging by patients which has revealed some of the reasons people pursue such therapies (Ryan et al., 2010). (Chapter 3 examines these reasons in more detail.) Though the evidence is anecdotal, and some of the blogs may be originating from the clinics themselves, many of the themes heard among patients quickly surface in the blog posts (Rachul, 2011):
- “While China is not our first choice, it is our only choice for now. We will go anywhere and do anything to give our children a chance at life.”
- “I may die anyway, but at least I died fighting. This is an experiment. What did I have to lose?”
- “We need therapies in our own cities and towns. It is deplorable and inhumane that we must put ourselves in harm’s way, pushing our already weakened bodies to the point of exhaustion, to seek stem cell therapy halfway around the world.”
People are going to these clinics because they feel that they have no other choice, said Caulfield, and that they cannot afford to wait for stem cell therapies to be approved through traditional regulatory processes.
Overseas clinicians are offering hope, Wagner said, despite their obvious financial conflicts of interest. They seek to create connections with patients who feel victimized by the current system. They offer patients control over their lives so they can choose the therapies they want. Patients go directly to the therapy and bypass their physicians, no matter what scientists or clinicians say.
Caulfield and his colleagues recently finished an analysis of more than 175 websites from scientific societies, stem cell research groups, and patient disease societies examining how they try to educate their communities about stem cell tourism (Master et al., 2013). Many of these sites are trying to do a good job, he said. But very few define stem cell tourism, talk about the evidence and the risk, or discuss what the patients should be seeking. They also do not explain why the regulatory process exists and why it is valuable, which is something that needs to be communicated, Caulfield said. As a participant noted, a vehicle is needed to provide patients with access to better information so that they can educate themselves. However, cautioned Ralph Cicerone, president of the NAS, it has been noted that “simply providing information to patients does not overcome their desperate need for some intervention.”
This issue is going to become even more challenging as stem cell therapies become more widespread, Caulfield said. Regulators will have more difficulty saying that treatments are ineffective or that action should wait until the results of clinical trials are available, especially when the therapy is being offered elsewhere in the world. Bolder statements need to be made about what the science says about a potential treatment. The problematic aspects of the field need to be addressed because clinical applications are proliferating, Caulfield said. “We have to get this right now in order to deal with the issue in the future.”








