2
Plasma Processing And Low-Energy Plasma Science
Plasma processing technologies are of vital importance to several of the largest manufacturing industries in the world. Foremost among these industries is the electronics industry, in which plasma-based processes are indispensable for the manufacture of very large-scale integrated microelectronic circuits. Plasma processing of materials is also a critical technology in, for example, the aerospace, automotive, steel, biomedical, and toxic waste management industries (Figure 2.1). Most recently, plasma processing technology has been utilized increasingly in the emerging technologies of diamond film and superconducting film growth. Because plasma processing is an integral part of the infrastructure of so many vital American industries, it is important for both the economy and the national security that America maintain a strong leadership role in this technology.
Figure 2.1 Plasma processing is a critical technology in many vital U.S. industries.
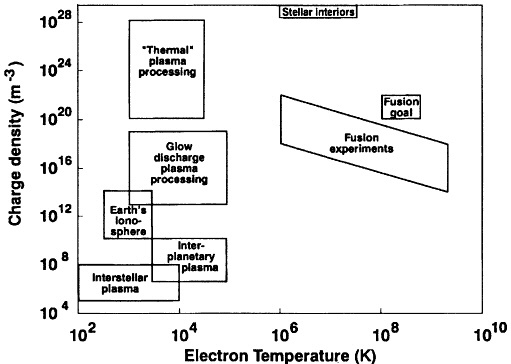
Figure 2.2 Classification of plasmas according to charge density per unit volume and electron temperature in degrees Kelvin. Plasmas used in materials processing cover a large portion of the low-energy density-temperature space.
CLASSIFICATION OF PLASMAS
A plasma is a partially or fully ionized gas containing electrons, ions, and neutral atoms and/or molecules. Plasma science is the study of the nonlinear collective interactions of electrically charged particles with each other, with neutral atoms and molecules, and with electric and magnetic fields. Figure 2.2 illustrates the wide range of electron densities and temperatures that are encountered in nature as well as in the laboratory.
Low-energy plasmas occupy a large portion of the density-temperature plane, with electron densities ranging from 105 to 1028 m-3 and electron temperatures ranging from 100 to 105 Kelvin. Two regimes in the density-temperature plane are characteristic of plasmas used in plasma processing. One of these includes glow discharges, in which the temperatures of electrons and heavy particles are widely disparate. The second includes thermal plasmas, in which electrons and heavy particles are in approximate thermal equilibrium. These plasmas are classical in nature in that the thermal kinetic energy is large in comparison to the average Coulomb interaction energy. Thus, charged particles usually interact weakly with each other, and electron collisions are usually most frequent with neutral atoms and molecules. The degree of ionization may range from a high percentage to less than 1 part per billion. These plasmas are governed by the laws of collective phenomena and atomic physics. Low-temperature plasmas, particularly glow plasmas, are not in thermal equilibrium and exhibit wide differences in electron, neutral, ion, vibrational, and rotational temperatures. Moreover, within each plasma species there are
departures from equilibrium, and Maxwellian energy distributions are seldom observed. Steep gradients between the plasma and solid surfaces or between the plasma and cold gas introduce additional deviations from equilibrium and, in high-pressure plasmas, fluid and kinetic instabilities add to the complexity. Thus, for these conditions the terms thermal and temperature have no strict definition and are only used qualitatively. Because of the myriad interactions that can occur in the volume and at surfaces, the highly nonequilibrium character of these plasmas presents both challenges and opportunities in materials processing.
BRIEF HISTORY OF LOW-ENERGY PLASMA SCIENCE AND TECHNOLOGY
Although ionospheric and, indeed, many astrophysical plasmas are classified as low-energy plasmas, the bulk of plasma science that is currently practiced in this domain is gaseous electronics-based. in its original form, the gas discharge branch of Langmuir's plasma physics, founded in the early part of this century, focused on electron conduction and breakdown in gases, electron emission and other cathode phenomena, and excitation of atomic and molecular species by electron collisions. Applications of the physical principles derived in this new field of science developed rapidly, beginning with the carbon arc as a light source and followed by gas-discharge rectification, high-power switch gear, and welding arcs. The ubiquitous mercury arc lamp followed, as did the more sophisticated fluorescent discharge lamp.
These and related applications have drawn heavily on plasma science in their evolution since World War II and have enlarged the scope of the supporting science to include plasma chemistry, atomic and molecular physics, surface chemistry and physics, optics, high-temperature physics and chemistry, electrical engineering, and computer science. This new science supports many of the world's major industries and is an indispensable part of many military technologies.
-
The use of plasma-based switch gear persists in the now huge power industry, while pulse power switches have been developed for use in high-power radars, discharge laser systems, and, indeed, in the switching of stored electrical power for high-energy plasma experimentation.
-
The early work on discharge light sources has evolved as the core technology in the lighting industry. Here, the earlier carbon and mercury arcs have been supplanted with arcs containing sodium-mercury mixtures as well as rare-earth additives to improve color and efficiency.
-
New lasing species including excimers have been discovered and exploited through the efforts of plasma science investigators, with the result that plasma-based, high-power lasers have formed a new and robust technology.
-
Thermal plasmas have been used for materials processing for many years (see Plasma Processing of Materials, National Academy Press, Washington, D.C., 1985). A multimegawatt thermal plasma process has been used commercially for nitrogen fixation. Thermal plasmas for remelting of metals in arc furnaces and for welding are well known. Development of plasma spray processes has led to significant advances in coating technology, with the aerospace industry being the major beneficiary. Today, plasma spray technology is also used in the automotive industry, the medical implant industry, the petrochemical industry, the cutting tool industry, and the paper industry (see Figure 2.1). Thermal plasmas are widely used in metallurgical refining and waste-processing areas.
-
The newest, and perhaps the largest, technology employing plasma science is the electronics industry(see Chapter 3). Low-pressure plasmas are vital in many plasma etching, deposition, and surface modification steps in the manufacture of nearly all integrated circuits.
-
Beyond this, plasma-enhanced chemical vapor deposition (PECVD) and thermal plasma chemical vapor deposition (TPCVD) are used for high-rate vapor-phase preparation of diamond and superconducting films.
This pervasive use of plasmas in modern technology is a result of the range of parameters that can be accessed and that is inaccessible by any other means. No other medium can provide gas temperatures or energy densities as high as those of plasmas; no other medium can excite atomic and molecular species to radiate as efficiently; no other medium can be arranged to provide comparable transient and nonequilibrium conditions.
In materials processing, opportunity lies in the fact that a highly excited medium can be obtained with no chemical or physical counterpart accessible in the natural environment. Plasmas can thereby alter the normal pathways through which chemical systems evolve from one stable state to another and can thus enable production of novel materials.
The diversity of applications of plasma-based systems used to process materials is matched by the diversity of plasma conditions, geometries, and excitation methods. In just one segment of the applications, PECVD and etching, the gas pressures vary by more than 6 orders of magnitude, and the gas composition includes rare gases and complex molecules. The plasma excitation frequency spans the electromagnetic spectrum from direct current (dc) to microwave. The power loading of the gases ranges from milliwatts per cubic centimeter to tens of kilowatts per cubic centimeter. The substrates may be in direct contact with the plasma, biased relative to the plasma, or placed downstream from the plasma. The practitioners of this composite science must then call on a number of science resources, which include:
-
Plasma electrodynamics and kinetics,
-
Plasma and surface chemistry
-
Basic data for cross sections and rates,
-
Diagnostics of discharge and surface processes,
-
Theory and predictive modeling, and
-
Scaling and process control.
The scientific community has the challenging task of providing the multidisciplinary scientific underpinning that will enable responding to the strong technology pull that exists now and will grow in the future.
INDUSTRIAL APPLICATIONS OF PLASMA PROCESSING OF MATERIALS
The number of industrial applications of plasma-based systems for processing of materials and for surface modification is extensive, and many industries are impacted. Some of these processes and corresponding applications include:
-
Plasma-controlled anisotropic etching in fabrication of microelectronic chips;
-
Plasma deposition of silicon nitride for surface passivation and insulation;
-
Surface oxidation used in fabrication of silicon-based microelectronic circuits;
-
Plasma-enhanced chemical vapor deposition of amorphous silicon films used in solar cells;
-
Plasma-surface treatment for improved film adhesion to polymer surfaces;
-
Plasma nitriding, which is used to harden the surface of steel;
-
Plasma-enhanced chemical vapor deposition and thermal plasma chemical vapor deposition of diamond thin films;
-
Plasma spray deposition of ceramic or metal alloy coatings used for protection against wear or corrosion in aircraft and automotive engines;
-
Plasma spray deposition of clearance control coatings;
-
Plasma melting and refining of alloys;
-
Plasma-assisted manufacture of optical fibers used in communications;
-
Plasma synthesis of ultrapure powders used as ceramic precursors;
-
Plasma spray deposition and thermal plasma chemical vapor deposition of high-temperature superconductors and refractory materials;
-
Plasma welding and cutting; and
-
Plasma sputter deposition of magnetic films for memory devices.
In the electronics industry (covered in greater depth in Chapter 3), plasma-controlled etching of materials provides the essential anisotropic etching for pattern transfer from a developed photoresist to the underlying structure; it is free from the undercutting that is characteristic of the alternative process, wet chemical etching. Such anisotropic etching becomes increasingly critical in the manufacture of progressively higher-density chips. There exists no other viable method to fabricate these structures. Plasma-deposited silicon nitride allows deposition at substrate temperatures between 200 and 350 ºC, much lower than the temperature that would be required for chemical-vapor-deposited silicon nitride. Because of the lower deposition temperature, this material can be deposited after completion of circuit features that would be damaged by a higher processing temperature. Plasma-deposited silicon nitride and silicon dioxide are the dielectric materials of choice for a variety of chip applications, including passivation coating for protection from chemical contamination and from mechanical damage, insulation between metal levels, masks to prevent oxidation or diffusion, and etch masks in multilevel resist structures.
Thermal plasma processing applications range from physical transformation of materials such as bulk melting in metallurgical plasma furnaces, metal cutting and welding, or powder melting during spraying, to driving chemical reactions, as in the reduction of oxides, in the synthesis of ceramic powders or films, or in the destruction of hazardous chemical wastes. Most of these plasma processes could not be replaced by other processes without sacrificing product quality and manufacturing efficiency. Thermal plasma generators have been designed for many diverse applications covering a wide range of operating power levels from less than 1 kilowatt to over 50 megawatts, and using various design principles. For processes of interest to the electronics industry, the rapid deposition of thick films using plasma spraying or thermal plasma chemical vapor deposition (TPCVD) are the processes of principal interest. The aerospace and the medical implant industries have come to rely on plasma spraying as a manufacturing process for a variety of coatings with controlled properties, such as corrosion resistance, porosity, and thermal conductivity. Plasma spraying is currently an $800 million per year business that is highly leveraged by the much larger jet engine and aerospace industries.
Applications of plasma spraying are pervasive in the aerospace gas turbine engine. Some newer engines may contain as many as 5000 components processed with plasma spray coatings. Thermal barter coatings (TBCs) provide the opportunity to use currently available compound materials to increase the operating temperature of turbine components without the use of increased cooling air. TBCs consist primarily of zirconia partially stabilized by yttria or magnesia. The thermal plasma spraying technique is ideally suited for applying multiple-layer TBCs at a reasonable cost. The main components currently being processed by thermal plasma spray techniques include turbine blades and vanes, burner cans, fuel vaporizers, stator vanes, and blade and reheat trough components.
Clearance control coatings (CCCs) generally consist of two components to provide the desired combination of integrity and abradability required to minimize tip clearance in rotating machinery, primarily gas turbine engines. Both metals and ceramics are used as the matrix, while additives include polymers, graphite, and other ceramics or minerals. A correctly designed and applied CCC will maximize efficiency by minimizing the loss between the blade tip and the casing's inner liner. Spray deposition of abradable coatings is especially suited to thermal plasma
processing. The coatings are normally designed with predetermined porosity to enhance component rub characteristics. A limitation of these coatings is erosion resistance, which drops rapidly as porosity increases (porosity levels as high as 20 to 25 percent are common). Current composite abradable coatings for use in the compressor section consist of a porous top layer of a nickel matrix with graphite embedded as the lubricant, an intermediate denser ceramic layer of stabilized zirconia, a second ceramic layer, and a bond coat.
In thermal plasma chemical vapor deposition (TPCVD), deposition occurs with vapor phase precursors at atmospheric pressures. TPCVD has been used to deposit highly textured high-temperature superconductors, polycrystalline as well as homoepitaxial diamond films, and other ceramic films of potential interest to the electronics industry. This technology is on the verge of becoming commercial.
INTERFACE BETWEEN BASIC PLASMA SCIENCE AND APPLICATIONS
The interface between basic plasma science studies and technological applications is continuous. The challenge to industry is to improve system performance and to develop new products or processes. To meet this challenge, managers and support staff as well as engineers and scientists must understand the underlying physics and chemistry of the system, and they must develop the capability to deal with the multidisciplinary and diverse nature of the science. They will require science data bases, fundamental understanding of discharge and chemical processes, and diagnostic tools to make the necessary advances and meet the technological requirements of the future. Data bases needed to develop understanding of most plasma processing systems extend beyond data on homogeneous plasma phenomena to data on the coupling of the plasma with surfaces. Surface processes cause steep spatial gradients in plasma parameters, which greatly complicate data acquisition and system modeling, and alter the chemistry in the plasma. But surface processes are the raison d'etre of plasma processing and distinguish these plasmas from those found, for example, in studies of controlled nuclear fusion.
SCOPE OF THIS REPORT
Consistent with the charge to the panel by the Plasma Science Committee, this report assesses the current state and criticality of low-energy plasma science in materials processing, with emphasis on materials processing in microelectronics. In addition, it identifies key science issues and research needs in plasma physics and chemistry at surfaces. Finally, it presents the panel's recommendations regarding research funding, educational issues, and cooperation among universities, industry, and the national laboratories.
Plasma processing of materials is a broad field encompassing numerous technologies and industries. It is beyond the scope of this report to make meaningful assessments of each of the technologies and industries. In emphasizing the electronics industry, the panel addresses one of the largest industries in the world for which plasma processing is vital. It is also an industry in which leadership by the United States is being lost and an industry whose enabling science and technology are in need of nurturing. Thus Chapter 3 gives a fairly detailed account of the plasma processes used in the fabrication of today's and tomorrow's microelectronic circuits.
Although both glow discharges and thermal plasmas are used in the processing of electronic materials, most applications in fact exploit glow-discharge processing. However, thermal plasmas are used extensively in the deposition of emerging electronic materials such as diamond, silicon carbide, cubic boron nitride, and high-temperature superconductors. Although the panel has noted that the density-temperature parameter space is markedly different for these plasmas, diagnostic techniques, modeling approaches, and fundamental processes are often similar, and
many of the conclusions presented here regarding science issues (Chapter 4) and educational issues (Chapter 5) pertain to both regimes.
This report should be viewed within the context set by the report Materials Science and Engineering for the 1990s (National Academy Press, Washington, D.C., 1989). Most if not all of the recommendations and findings in that report also apply here. Plasma processing is a key enabling technology for materials synthesis and modification. The recommendations made in this report necessarily emphasize the processing of materials for the electronics industry, and, in this sense, its coverage of new materials processing for the aerospace, automotive, and other industries is incomplete. The panel urges further assessment of these areas with respect to the role and criticality of plasma technology.







