Antigen Specific and Antigen Non-Specific Immunization
Robert Burrell
The word "hypersensitivity" in the multiple chemical hypersensitivity epithet means quite different things to those depending on whether they be toxicologists or immunologists. The term unfortunately prejudices immunologists bemuse hypersensitivity has very precise connotations based on several decades of molecularly oriented studies which concluded that classical immunological reactions are the result of highly specific molecular interactions between precisely defined prosthetic groups on antigens (epitopes) reacting intimately with either specific immunoglobulins induced by prior contact with that antigen or with T lymph-ocyte antigen specific receptors (TCR). Although a certain degree of cross reactivity among different molecules can be expected, such may be explained on the basis of molecular structures having closely identical configurational fits. To suggest that chemically, quite-unrelated molecules cross react in the classical immunological sense is utterly foreign to those steeped in the lore of the enormous number of studies of such pioneers as Landsteiner, Heidleberger, Haurowitz, Campbell, and many other immunochemists. The studies of Landsteiner (1945) on the specificity of serological reactions spanned nearly four decades and one of his innumerable studies is summarized in Fig. 1. His method of diazotizing defined haptens on carrier molecules to obtain antisera and then reacting these antisera with homologous and closely related haptens forms one of the basic foundations of classical immunology.
Later, such reactions were placed on a more quantitative basis by people like Eisen (1990) who used the techniques of physical chemists and enzyme kineticists to study these same kinds of hapten-specific interactions. Through the use of equilibrium dialysis, one can obtain such information as antibody valence and degree of heterogeneity in an antibody population and even the affinity constant of an antigen-antibody reaction (Fig. 2). From these kinds of studies, it soon became evident that antigens and antibodies did not react by forming covalent bonds, but rather were held together by a number of physical forces. These forces (hydrogen bonding, electrostatic, Van der Waals, etc.) in themselves are non-specific and ordinarily are relatively weak, but become quite strong if the two molecules are very close together. Van der Waals forces for example decrease inversely with the seventh power of the distance separating two molecules. But what allows them to approximate themselves to each other so closely is the fine degree of complementary, struc-
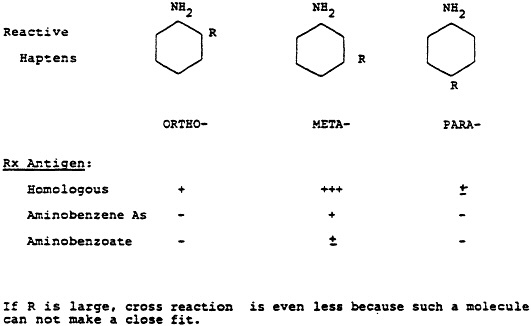
Fig. 1 One of the classical experiments of Landsteiner illustrating the use of antibodies directed to synthetic haptens. In this case, antibodies were prepared against the hapten, meta-aminobenzene sulfonic acid conjugated to an appropriate carrier. Maximum serologic reactivity was obtained only when this antiserum was reacted with the hapten used in its production and not with molecules that were closely related chemically. Further, the specificity even extended to the position on the benzene ring on which the sulfonic group was attached.
tural configuration in the respective molecules. If a closely-related molecule has an additional conformational change, even moving a prosthetic group from a para-to a meta-position (Fig. 1), then that close fit is greatly compromised and there is either no reaction or a very much weaker one.
The availability of myeloma immunoglobulins, for which antigenic reactivity is known, afforded the immunochemist with a homogenous population of antibodies whose light and heavy chain variable domains could be sequenced. Now the exact molecular structure of the antibody as well as the antigen could be known. The amino acid sequence of a polypeptide determines how that peptide will fold three-dimensionally in space and that degree of folding is the antibody characteristic which allows the complementary fit of its homologous antigen. Fig. 3 shows the key amino acids in light and heavy chains that are in most intimate contact with a phosphorylcholine hapten. Essentially, the antigen fits in a very tight cleft formed by the folding of the light and heavy immunoglobulin chains. The lock-and-key analogy used for so many decades to explaining antigen-antibody fit has proven to be remarkably accurate.
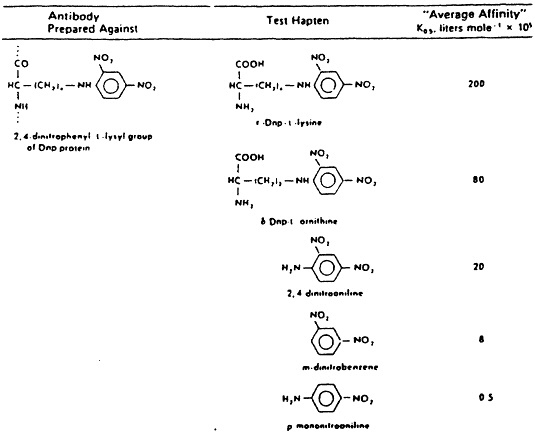
Fig. 2 When serological studies are extended quantitatively, affinity constants can be obtained using equilibrium dialysis methods. Note that the maximum affinity constant occurs only when the antiserum is reacted against the hapten used in its production and that this constant rapidly declines, the further removed the chemically substituted molecules are that are used in reacting against the same antiserum. From H.N. Eisen, General Immunology, 3rd Edition, 1990, Lippincott, Philadelphia, p. 17.
Contemporary molecular immunology is now focussed on the cellular aspects of the immune response, particularly the afferent induction pathways. The pathways involve first contacting the antigen, then processing the antigen so that peptide fragments of the native antigen are presented on the cell membrane surfaces of antigen presenting cells in context with major histocompatibility (MHC) molecules. This presentation is made to T helper lymphocytes who express receptors (TCR) for the presented antigen within this MHC context. Representative TCRs have also been sequenced and their structures can be considered analogous to antibody molecules (Fig. 4). Processed antigen molecules fit within the TCR deft in a manner analogous with the way antigen fits in an imunoglobulin deft. Thus even antigen-lymphocyte interactions are governed by the same kinds of intermolecular constraints that antigens and antibodies are subject to.
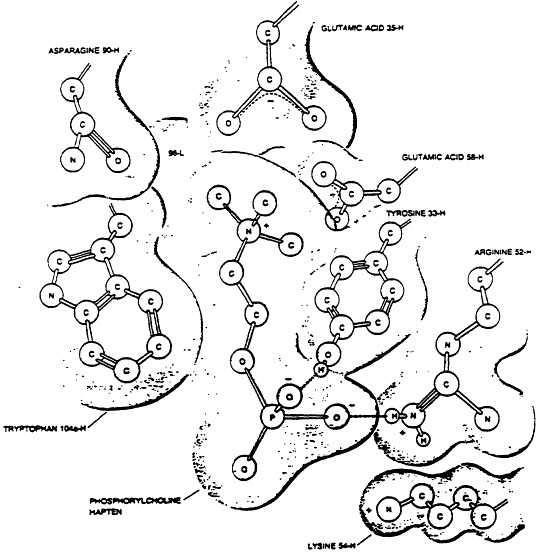
Fig. 3 Model of the binding of the hapten phosphorylcholine to the combining site of the myeloma protein with which it is reactive. The numbers refer to the amino acid position in the heavy (H) or light (L) chain respectively which is in closest approximation with the key polar forces on the haptenic molecule. The way in which the peptide chains of the antibody fold provide for a three-dimensional deft in which the hapten may be attracted in a very close, conformational fit. From Scientific American, 1977, 236(1):58.
But with the tremendous increase in modern knowledge of immunity has come the realizations that 1) the immune system is far more complicated than intimate reactions between closely-fitting, complementary molecules and 2) that the very same immune system
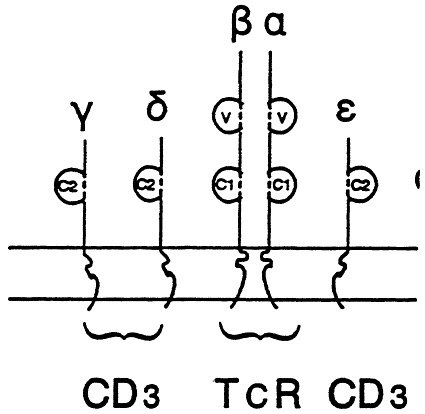
Fig. 4 A stylized model of the T cell receptor (TCR) molecule showing the resemblance to immunglobulin molecule. It is composed of two chains embedded in the lymphocyte membrane. These chains also have variable and constant domains in a manner analogous to immunoglobulins. The CD3 chains are associated in proximity to the TCR and participate in stabilization of the molecular interactions with specific antigen and antigen- presentation markers.
that recognizes specific antigens also can be stimulated non-specifically to carry out its effects independently of antigens. Knowledge emanating from studies of such matters as the plethora of cytokines required for immunological reactions, the many forces involved in priming and translating immune recognition into inflammation, the pharmacology of immune reactions, the ability of some very important immunological cells to recognize foreignness in an antigen-independent manner (e.g., NK cells), and the growing awareness of how the central nervous system and the immune system are inter-connected are a few examples that suggest that the immune system can play an important role in effecting antigen non-specific reactions as well.
Table 1 compares classical, antigen specific immunity with that of antigen non-specific reactions from several aspects. Non-antigen specific reactions are triggered by many kinds of agents, e.g., polysaccharides that activate the alternative complement pathway in the absence of antibody or tumor cells that activate NK cells. Many types of immuno-
TABLE 1
|
Comparison of Antigen Specific and Antigen Non-specific Immune Reaction |
||
|
Feature |
Antigen Specific |
Antigen Non-specific |
|
Stimulus |
Antigen |
Immunomodulator |
|
Response measured |
Antibodies Tc cells |
Mediator production NK cells Pathology |
|
Secondary |
Anamnestic |
Enhanced, Ag non-spedcific |
|
Longevity |
Long, Ag-dependent |
Temporary |
modulators or substances that mimic allergies can fall into this category. Although one usually measures humoral antibody or T cytotoxic cell responses from antigen-driven reactions, increases in mediator production or responsiveness of target tissues are the parameters usually assessed in non-antigen specific reactions. One of the earmarks of classical immune reactions is the anamnestic response, but increased reactivity can also be brought about by a number of non-antigen specific mechanisms as well; the Shwartzman reaction is a good example. Longevity of antigen specific reactions is mostly dependent on dose and presence of antigen as well as the induction of memory cells. Non-antigen specific reactions are often temporary, that is enhanced responsiveness is only present within a short time range at such time when two independent stimuli have been received by the target tissue. At other times, increased hyperreactivity may be more permanent as a result of a major event such as an infection which then renders the subject hyperresponsive at other times years later, especially when another infection or irritation may take place. Table 2 lists several examples of antigen non-specific reactions.
Classical immunology of course is concerned with the effects of antigen on antigen-presenting cells, all classes of lymphocytes, antigen processing, effector regulation and the like, but these same processes are either closely allied or one and the same as the inflammatory mechanisms generated by the induction of immune responses. Even for antigen specific responses, it has been pointed out by Warren and Chedid (1987) that if immunology is defined as the body's reaction to foreign insult, then one must also consider such non-antigen specific, induced responses as the involvement of endothelial cells, activation of the complement and procoagulation cascades, vascular changes, cytokine generation, lysosomal enzyme release, the generation of toxic oxygen radicals, arachidonic acid metabolism, even the effect of interleukin 1 (IL-1) on the hypothalamus, among many others (Table 3).
The most important factor in trying to explain any hypersensitivity reaction is the matter of dealing with why, of all of those exposed to a given agent, only a small subset respond in such a deleterious manner. Classical allergists at least have an explanation largely in the form of antigen-specific IgE or lymphocytes that have been produced.
Explaining individual idiosyncrasies that are not allergy-based is difficult owing to the
TABLE 2
|
Examples of Antigen Non-Specific Reactions |
|
Activation of alternative C pathway |
|
Peptigoglycans |
|
Endotoxin |
|
Thrombin |
|
NK cell recognition |
|
Tumor cells |
|
Virus-infected cells? |
|
Acute phase reactants |
|
C-reactive proteins |
|
Interaction with bacterial polysaccharides |
|
Haptaglobin |
|
Anti-inflammatory factor |
|
Low density lipoprotein |
|
Partial detoxification of bacterial endotoxin |
dearth of studies on other possible explanations. Pharmacologists recognize idiosyncratic reactions to drugs and account for them by a number of physiologic or pharmacologic host factors that determine reactivity to such agents. Those who manifest hyperresponsiveness to low dose levels have a genetic component that is responsible for either how the drug is metabolized or how that person's cellular/molecular mechanisms respond. These pharmacogenetic bases are further complicated by the numerous types of environmental factors that affect gene expression or the occurrence of the clinical manifestation. An individual with an abnormal gene product would never be noticed unless that person becomes exposed to an agent in which there is an interaction with that product In these instances, only a certain subset of the population is going to metabolize the agent in such a way as to result in toxic by-products. When an environmental stimulus is additionally required to express the abnormal gene action, as in the case of cutaneous photosensitization in certain porphyrias, clinical manifestations might be erroneously considered immune in origin, but the interesting thing here is the requirement for two separate signals. In addition to genetic factors, other conditions which are known to alter pharmacologic reactivity to substances are age, presence of concurrent disease, nutritional status, hormone levels, diurnal variation in drug disposition or sensitivity, environmental factors that induce or inhibit drug metabolism, and efficiency of repair mechanisms (Nebert and Weber, 1990).
Although a fair amount is known about pharmacogenetics, much less is know about genetic factors that affect physiologic responsiveness. Some of the host factors that definitely have bases contributing to the way individuals hyperrespond to environmental agents are airway hyperresponsiveness to methyl choline challenge, increased propensity of basophils to release histamine, dermatographism, and hyperreactivity to histamine.
In MCS, not only are we faced with the problem of explaining hyperresponsiveness, we
TABLE 3
|
Examples of Host Responses that Must Also Be Considered Immunological |
|
Endothelial cell reactions |
|
Activation of C cascades in the absence of antibody |
|
Activation of procoagulant activity |
|
Acute phase reactant responses |
|
Aracidonic acid metabolism |
|
NK cell enhancement |
|
Lysosomal enzyme release |
|
Generation of toxic oxygen radicals |
|
IL-1 effect on the hypothalamus |
|
Beta-endorphin activity |
|
Generation of neuropeptides |
have an additional problem of explaining how this hyperresponsiveness is directed towards numerous agents with no obvious molecular relationships in a given individual One of the environmental factors that has been studied more concerning the subsequent development of hyperresponsiveness has been that of association of certain types of infections with triggering development of bronchial asthma particularly in children and to some extent in adults. But even in these instances, other factors are additionally required, e.g., the type of virus musing the infection, the age at which the infection first takes place, the presence of certain constitutional symptoms at the time of infection, genetic predisposition, pre-existing airway hyperreactivity, and sex. Busse (1990) has recently reviewed the possible mechanisms (summarized in Table 4) by which this altered reactivity might occur and among them proposed are that certain viruses, notably respiratory syncitial virus, may enhance IgE synthesis and mediator release from basophils either directly or through the action of interferon from virus-stimulated lymphocytes. The viruses may also interfere with beta-adrenergic function either by directly inhibiting such function, by passively allowing the mast cell to promote more mediator release, or by enhancing cholinergic stimulation of airways smooth muscle. Finally, the virus may desquamate epithelium thus exposing afferent neurons to stimulation and decreasing the amount of an endopeptidase that normally degrades the amount of the peptidergic smooth muscle contractant, substance P. Thus, extrinsic asthma may require 1) previously altered tissue growth/damage from an infectious agent or an irritant, 2) genetic predisposition, and 3) a subsequent trigger later in
TABLE 4
|
Possible Mechanisms to Explain Asthma Predisposing Factors of Respiratory Infections |
|
1. Viruses induce polyclonic IgE antibody responses |
|
Genetic predisposition |
|
Associated with particular viruses, e.g. RSV |
|
Elevated airway histamine |
|
Histamine levels correlate with hypoxia |
|
2. Viruses enhance basophil mediator release/chemotaxis |
|
Can substitute interferon for virus |
|
3. Viruses may diminish beta-adrenergic function |
|
Could establish a permissive situation promoting mast cell mediator release |
|
4. Viruses promote cholinergic-dependent responses |
|
Not in smooth muscle directly, but in inflammatory cell mediator release |
|
5. Viruses affect peptidergic responses |
|
Damage airway epithelium |
|
Affect bronchodilator effects of LPO products |
|
Unopposed constriction of Substance P |
|
Infection inhibits SP degrading enzyme |
|
HYPOTHETICAL SCENARIO |
|
Inflammatory granulocytes, altered by infection |
|
Recruited to the airways |
|
Enhanced mediator release in situ |
|
Leads to airway hyperresponsiveness |
|
Must take place in the generally-predisposed |
life. We might ask with regard to multiple chemical sensitivity, what the role might be of a previous infection or toxic event at a critical point of development in a genetically-predisposed individual such that neurologically controlled effector mechanisms become subsequently more hyperreactive. But again, in trying to explain MCS, one confronts the observations that its signs and symptoms are not referrable to specific shock organs such as bronchial smooth muscle in asthma. Instead, many of the symptoms are so vague, subjective, and rarely subject to measurement.
Another reason certain individuals might be excessively responsive at a given time might be because primary, incidental stimuli may serve to prime inflammatory mediators that temporarily render that individual more responsive to a second, unrelated stimulus. The coincidental exposure to two or more stimuli within a given time may induce synergistically-altered responses whereas if these same two unrelated stimuli were received at
separate times further apart, there would be no abnormal reaction. Much of what we know about this kind of exaggerated responsiveness has been learned from the study of host response to bacterial endotoxin and the prototype of this type of response is the Shwartzman reaction. The endotoxin-induced Shwartzman reaction can be manifest either in a dermal or systemic form-both lead to increased target organ hyperreactivity following exposure to the second stimulus and thus meets the definition of altered reactivity. Neither is antigen specific reaction because the two doses do not need to be antigenically related, the time between sensitization and provocation is too short to induce antigen specific reactivity, and the increased sensitivity does not last more than a few days.
TABLE 5
|
Examples of ''Second Signal'' Mediator Responses |
|
Shwartzman reaction |
|
Adult respiratory distress syndrome |
|
Endotoxin enhanced responsiveness |
|
Infection-asthma |
|
Receptors-cAMP/arachidonic acid metabolites |
|
Immunomodulator/adjuvants |
|
Interferon stimulation |
|
Pharmacogenetic hyperresponsiveness |
It is now felt that this type of temporal, increased reactivity may be due to several mediator systems being turned on to such an extent by various types of priming stimuli that occur prior to exposure to a further stimulant. Much attention has been directed to the idea of endotoxin-initiated "second sign" wherein endotoxin primes target cells for subsequent enhanced reactivity. This is an exceedingly complex area and could involve several different types of systems including calcium ion channel triggering, activation of cyclic nucleotide synthesis, increased toxic oxygen radical formation, and enhanced cytokine production, e.g., TNF and IFN-(Heremans et al., 1990), among others (see Table 5 for examples). This may relate to multiple chemical sensitivity in that if an individual encounters an immunomodulating or pharmacologically-altering agent at near the same time he encounters a triggering substance, the combined effects of mediators from the two stimuli may synergistically cause an exaggerated reaction.
References
Busse, W.W. 1990. Respiratory infections: Their role in airway responsiveness and the pathogenesis of diseases and asthma. J. Allergy Clin. Immunol. 85:671-683.
Eisen, H.N. 1990. General Immunology, pp. 11-18. Philadelphia: J.B. Lippincott.
Heremans, H., J. Vandamme, C. Dillen, R. Dijkmans, and A. Billiau 1990. Interferon-gamma, a mediator of lethal lipopolysaccharide-induced Shwartzman-like shock reactions in mice. J. Exp. Med. 171:1853-1869.
Landsteiner, K. 1945. The Specificity of Serological Reactions. 450 pp. Boston: Harvard University Press.
Nebert, D.W., and W.W. Weber 1990. Pharmacogenetics. In Principles of Drug Action, eds.
W.B. Pratt, and P. Taylor, pp. 469-531. New York: Churchill Livingstone.
Warren, H.S. and Chedid, L.A. 1987. Strategies for the treatment of endotoxemia: Significance of the acute-phase response. Rev. Infect. Dis., 9 (Supp 5), S630-S638.












