4—
CAPTIVE BREEDING OF THE 'ALALA
Through research and experimentation, biologists have developed several techniques to preserve endangered species (Temple, 1977b). One such technique–captive propagation—has found increasing acceptance as a valid conservation strategy and has been applied to various vertebrate species within the last decade (Cade, 1988; Conway, 1988). Captive breeding has been used principally to avoid extinction of some species, such as the California Condor (Gymnogyps californianus) (Snyder and Snyder, 1989), black-footed ferret (Mustela nigripes) (Thorne and Williams, 1988), Guam Rail (Rallus owstoni), and Micronesian Kingfisher (Halcyon c. cinnamomina) (Witteman et al., 1990). In most instances, however, it has been used to complement conventional field research and management efforts to preserve habitat and restore threatened populations. For example, in the case of the Peregrine Falcon (Falco peregrinus), captive breeding and reintroduction enabled the restoration of breeding populations in large portions of the former range at a rate far exceeding that which would have occurred through natural colonization (Cade, 1986a,b, 1988).
When wild populations are small, fragmented, and therefore prone to extinction, captive populations can serve several important roles in recovery efforts (Carpenter and Derrickson, 1981; Derrickson and Snyder, 1992). First, and most important, they can provide a safety net to minimize the probability of extinction while preserving genetic variation through controlled pedigree breeding. Second, if captive breeding can be achieved and sustained, captive populations can provide stock for release to the wild. Such releases can be extremely important in correcting sex-ratio imbalances, infusing genetic variation, introducing new behavior, re-establishing extirpated populations, and establishing new populations in natural or altered habitats. In many instances, breeding biology can be manipulated in captivity so that reproductive output can be increased substantially over that obtainable in the wild. Third, captive populations can facilitate research studies on biological, behavioral, or ecological characteristics that are essential for recovery but that are difficult or impossible to address in the wild, owing to small population sizes or environmental conditions. Finally, captive populations and their associated reintroduction programs can provide a foundation for establishing subsidiary programs aimed at habitat preservation, public education, and conservation training (cf. Durrell and Mallinson, 1987; Kleiman, 1989; Butler 1992).
Origins and Facilities of the 'Alala Captive-Breeding Program
The precipitous decline of the wild 'Alala populations in the late 1960s prompted state and federal biologists to endorse the establishment of a captive-breeding program for the species by removing injured or sick birds from the wild. The immediate objectives of the program, like those of similar programs initiated at about the same time for other endangered species, were to avoid extinction in case wild populations were extirpated, to enable research on behavioral and reproductive requirements, and to produce offspring that could be returned to the wild to augment the existing wild populations. The last of those objectives was emphasized in the 'Alala Recovery Plan developed later (Burr et al., 1982).
Two fledglings exhibiting pox lesions were retrieved from adjacent nesting territories on the Hualalai Ranch in June 1970 and were held at the research aviary in Hawai'i Volcanoes National Park. Both were transferred to the endangered-species breeding facilities at the Patuxent Wildlife Research Center in Maryland in August 1970 after treatment and apparent recovery from pox. One of the two died soon after arrival at Patuxent, and the other died, apparently of circulatory problems, on October 1, 1975 (Banko and Banko, 1980). Three additional wild fledglings were salvaged in 1973; two were found on the ground, and the third was discovered to have a heavy infestation of northern fowl mites. All three were successfully raised at the Hawai'i Volcanoes National Park's research aviary and later transferred to the state of Hawai'i's endangered-species breeding facility at Pohakuloa in early 1976 (Banko and Banko, 1980). Between 1977 and 1981, seven more 'Alala were removed from the wild and transferred to the Pohakuloa facility for captive breeding.
The Pohakuloa facility, on the leeward slope of Mauna Kea at an elevation of about 1,950 m, was established in 1949 for the propagation of the Hawaiian Goose, or Nene (Branta sandvicensis). By 1976, when the state acquired responsibility for the 'Alala breeding program from the federal government, two additional endangered species were being propagated at Pohakuloa—the Hawaiian Duck (Anas wyvilliana) and the Laysan Duck (Anas laysanensis). Although considerable success was achieved with the three waterfowl species, the breeding of 'Alala proved much more difficult, despite periodic consultations with aviculturists and zoo personnel. In 1985, as a result of poor 'Alala reproduction and the recognition that propagation efforts would probably be expanded to include other endangered Hawaiian forest birds, the state asked Stanley Temple, of the University of Wisconsin, to explore the potential for developing a new facility at a more suitable location. He suggested that the 'Alala be moved from Pohakuloa for several reasons, including substandard facilities, personnel and predator problems, prevailing climatic conditions, and the periodic disturbance caused by military training activities at the nearby U.S. Army Pohakuloa Training Facility. After reviewing several alternative sites in the state, Temple recommended that the former Olinda Honor Prison Camp, near the town of Makawao on Maui, be renovated as a new propagation facility, which would have the advantages of pre-existing buildings, suitable climate, and relative isolation.
At an elevation of 1,500 m on the windward slope of Haleakala, the 45-acre site is characterized by a cool, wet climate typical of mid-elevation rain-forest habitat. Federal and state funds and in-kind construction assistance from the U.S. Army were used to renovate the facility. Initial work at Olinda focused on the construction of two large subdivided aviaries for the 'Alala, the remodeling and renovation of buildings for an interim veterinary clinic and staff quarters, and the installation of water and emergency power equipment.
The 'Alala pens were completed in July 1986, and the nine 'Alala in the captive flock at Pohakuloa were transferred to Olinda in late November that year. Site renovation and development have continued since 1986, with the addition of waterfowl breeding and rearing complexes, infectious and noninfectious quarantine facilities, security fencing, and miscellaneous renovations of the existing buildings. All the 'Alala breeding pens are occupied, and only a single juvenile enclosure is unoccupied. Although up to six additional birds could be accommodated by dividing some of the existing enclosures, those modifications would not be suitable for breeding (F. Duvall, pers. comm., 1991, 1992). Plans are now under way to construct additional 'Alala and forest-bird pens and to expand the existing Nene breeding complex in 1992.
Three full-time staff are employed at Olinda: a director-aviculturist and two animal technicians. Three additional staff members include an avicultural assistant with a 1-year contractual appointment and two laborers with temporary limited-term appointments. Veterinary services, both emergency and routine, are provided by Ben Okimoto of the Honolulu Zoo on O'ahu; previously, these services were handled by a local veterinarian, Renata Gassman-Duvall, through a contract with the Hawai'i Department of Land and Natural Resources.
Staff at both Pohakuloa and Olinda have sought the assistance of a variety of aviculturists and institutions through the years. The closest ties have been with staff at the San Diego Zoo. In September 1990, Cynthia Kuehler, a zoologist with the Zoological Society of San Diego and an advisor to this committee, conducted a review of the 'Alala breeding program and made a number of husbandry and management recommendations to improve the health and productivity of the captive flock, many of which have been implemented. She also reviewed and summarized the existing records and prepared a studbook for the captive population, which has greatly facilitated the demographic and genetic analyses discussed below. A copy of the studbook, listing hypothetical founders and all living and dead 'Alala in the captive population, is provided in Appendix B.
In March 1991, laparoscopic examinations were conducted on juvenile and nonreproductive adult 'Alala at Olinda by San Diego Zoo veterinarian Donald Janssen and animal health technician Kim Williams to confirm the birds' sexes and evaluate their general health.
Demographics and Genetics
The studbook lists a total of 23 'Alala, including three hypothetical founders, 14 wild-caught birds brought into captivity from 1965 to 1983, and six captive-hatched birds. No wild-caught birds have entered the population since 1983, and the population has remained small because of both adult mortality and low reproduction (see Appendix B). Specific causes of mortality have included shipment stress (one case), pericarditis/splenitis (one), toxemia (one), hemochromatosis (three), yolk peritonitis (two), and unknown causes (one). Six pairings, involving four females and four males, resulted in the production of eggs during the period 1979–1991, but only four pairs, involving three females and three males, have produced offspring (Figure 4.1, Table 4.1, Table 4.2).
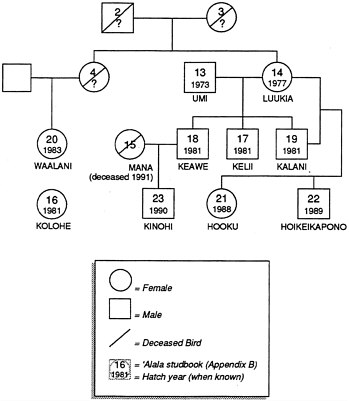
Figure 4.1 Representation of pedigree of captive 'Alala.
Table 4.1 Pairings at Pohakuloa (1979–1986) and Olinda (1987–1991) Facilities
|
Pairings |
Year |
No. of eggs |
Fledgling survival |
|
|
F |
M |
|
|
|
|
Luukia |
Umi |
1979 |
3 |
0 |
|
|
|
1980 |
2 |
0 |
|
|
|
1981 |
8 |
3 (Kalani, Keawe, Kelii) |
|
|
|
1986 |
1 |
0 |
|
Hiialo |
Kalani |
1984 |
10 |
0 |
|
|
|
1985 |
4 |
0 |
|
|
|
1986 |
2 |
0 |
|
|
|
1987 |
2 |
0 |
|
|
|
1988 |
10 |
1 (Hooku) |
|
Mana |
Kelii |
1984 |
2 |
0 |
|
|
|
1985 |
2 |
0 |
|
Mana |
Keawe |
1988 |
3 |
0 |
|
|
|
1988 |
3 false-lay clutches |
0 |
|
|
|
1990 |
5 |
1 (Kinohi) |
|
|
|
1991 |
3 |
0 |
|
Luukia |
Kalani |
1989 |
8 |
1 (Hoikei) |
|
|
|
1990 |
8 |
0 |
|
|
|
1991 |
2 |
0 |
|
Waalani |
Umi |
1991 |
3 |
0 |
|
|
|
Total |
81 |
6 |
Table 4.2 Summary of 'Alala pairings in the captive flocka. In monogamous species of birds, successful reproduction requires physiological synchronization and sexual and social compatibility.
|
Female |
Pairing year(s) |
Mate |
Comments |
|
Waalani |
1987–1991 |
Kelii |
Late nest-building, 1987–1990; male broke egg 1991, no relaying; separated because of aggression |
|
|
1991 |
Umi |
Compatible, but nest-building only |
|
Hina |
1977-1978 |
Umi |
Productive, chicks and eggs disappeared; female died August 7, 1978, of toxemia |
|
Hooku |
1991 |
Keawe |
Placed together in August after death of Mana; appear compatible |
ADDITIONAL NOTES REGARDING CHICK REARING:
-
Three siblings raised at the Honolulu Zoo were kept together until August 1981, when they were transferred to Pohakuloa facility and placed in separate enclosures at Pohakuloa.
-
At Olinda facility young have been raised individually and then placed together as fledglings until approximately 1-year old (when intraspecific aggression occurs); they are then separated and housed individually.
Of the nine birds transferred from Pohakuloa to Olinda in November 1986, six were wild-caught birds originating in three locations along a 75-km stretch of the Kona coast: Hualalai (Hiialo, Luukia, and Waalani), Yee Hop-Alika Homestead (Umi), and Honaunau (Mana and Kolohe). The genetic relationships among these birds are unknown, but it should be noted that all breeding females have been derived from only two areas about 35 km apart, and several from locations adjoining the McCandless Ranch. Although the breeding female Mana died in 1991, she is still genetically represented in the population by a single offspring produced with the male Keawe. The 10-year-old female Kolohe (a potential founder) was behaviorally imprinted on humans at an early age and has not yet reproduced.
Of the 10 surviving birds (four females and six males) held at the Olinda facility, six were hatched and raised in captivity (Kelii, Keawe, Kalani, Hooku, Kinohi, and Hoikei), and four were hatched in the wild (Umi, Luukia, Waalani, and Kolohe). As would be expected, the age structure of the captive population is unstable, and the pedigree (Figure 4.1) is relatively shallow.
Because one of the primary goals of endangered-species propagation programs is the conservation of extant genetic variation, the 'Alala pedigree has been analyzed with the Single Population Animal Records Keeping System (SPARKS) studbook package developed by the International Species Inventory System (1990) to calculate founder contributions, inbreeding coefficients, and mean kinship values for the living population. As shown in Figure 4.2, fractional founder contributions within the living population are highly variable. Three of the founders (2, 3, and Umi) are overrepresented, two founders (P4 and Mana) are underrepresented, and one founder (Kolohe) has not contributed at all. Because such unequal founder contributions will lead to lower levels of genetic variation in later generations, current breeding strategies should have as a short-term goal reducing the disparity in founder contributions (cf. Lacy, 1989; Haig et al., 1990; Ballou, 1991) and maximizing the retention of genetic variation. Analysis of the current pedigree suggests that the living population retains only about 75% of the genetic diversity present in the founders, but this would increase to slightly over 88% if Kolohe produced offspring, and possibly even higher yet if additional founders were added to the captive population.
Assuming that all the original founders of the captive population were unrelated, two birds (female Hooku and male Hoikei) have inbreeding coefficients of 0.25, and the remaining birds have inbreeding coefficients of 0. Inbreeding coefficients of potential offspring from all potential and actual pairings in the captive population are shown in Table 4.3. Two of the four current pairs will produce offspring with inbreeding coefficients of 0.25, thereby causing a reduction in overall genetic variation within the population. To manage the population so as to achieve target founder contributions and maximize retained heterozygosity, it would be advisable to pair males and females that have minimum mean kinship values (Table 4.4) (Ballou, 1991). To achieve this goal, pairings on this basis would be: Umi x Waalani, Luukia x Kinohi, Keawe x Hooku, and Kelii x Kolohe. It should be noted that three of these recommended pairings (i.e., Umi x Waalani; Keawe x Hooku; Kelii x Kolohe) are presently being attempted at Olinda (Table 4.4) and thus these male-female pairs appear compatible (Table 4.2). The fourth recommended pairing (i.e., Luukia x Kinohi) has never been tried. Considering that this pairing would require the separation of the breeding pair Luukia x Kelani, and that the upcoming breeding season is fast approaching, the re-pairing of Luukia with Kinohi should probably be delayed until after the 1992 breeding season.
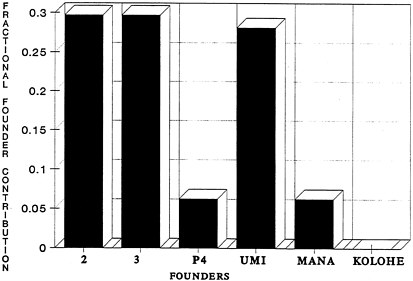
Figure 4.2 Founder Contributions, 'Alala. Studbook numbers beginning with P (see Appendix B) indicate wild or unknown founders that mated with studbook # (without the P) to produce captive-bred offspring.
Table 4.3 Possible pairings and inbreeding coefficients
|
|
Male |
|
|
|
|
|
|
|
Umi |
Kelii |
Keawe |
Kalani |
Hoikei |
Kinohi |
|
Female |
|
|
|
|
|
|
|
Luukia |
0.00 |
0.25 |
0.25 |
0.25a |
0.375 |
0.125 |
|
Kolohe |
0.00 |
0.00a |
0.00 |
0.00 |
0.00 |
0.00 |
|
Waalani |
0.00a |
0.0625 |
0.0625 |
0.0625 |
0.0937 |
0.0312 |
|
Hooku |
0.1250 |
0.250 |
0.25a |
0.375 |
0.375 |
0.125 |
|
NOTE: Males - horizontal Females - vertical Current pair. -a |
||||||
Table 4.4 Mean kinship of living birds to living nonfounders.
|
Name |
Sex |
Mean Kinship |
|
Umi |
M |
0.1406 |
|
Luukia |
F |
0.2812 |
|
Kolohe |
F |
0.0000 |
|
Kelii |
M |
0.2422 |
|
Kelani |
M |
0.2734 |
|
Keawe |
M |
0.2578 |
|
Hooku |
F |
0.3086 |
|
Hoikei |
M |
0.3086 |
|
Kinohi |
M |
0.1758 |
|
Waalani |
F |
0.1289 |
Because the amount of genetic variation that can be retained in a captive population over time is primarily a function of the size of the initial founder population, the rate of population growth to carrying capacity, and the generation length (Soulé et al., 1986; Hedrick and Miller, 1992), it is clear that the captive population needs to be expanded as rapidly as possible. Using the CAPACITY program developed by Jon Ballou of the National Zoological Park, we have examined the population sizes required to maintain extant levels of genetic heterozygosity for various lengths of time. Our results suggest that even when relatively optimistic model parameters are assigned (population growth rate (per year) = 1.5, generation length (years) = 6.0, Ne/N ratio = 0.5, and heterozygosity to date = 88%), the population will have to be expanded to about 130 individuals to retain 75% of the existing heterozygosity over a period of about 50 years. This would essentially require a captive population containing approximately 40 breeding pairs. We believe that this number should be adopted as an interim population objective, while recognizing that this number could potentially be reduced somewhat under certain management strategies, such as the integrated management of both the wild and captive populations (see the discussion of options for management in Chapter 6).
Inbreeding Depression
It has been postulated that the poor success obtained in breeding 'Alala in captivity results from inbreeding depression, and several lines of evidence support this hypothesis. First, as mentioned above, all the captive founders were taken from a relatively small area at a time when the population was declining precipitously; therefore, the assumption that all founders are genetically unrelated might be false. Second, as discussed in Chapter 3, inbreeding is known to affect embryo viability, egg hatchability, and chick survivorship. Between 1979 and 1991, only eight (24%) chicks were hatched from 33 unbroken, fertile eggs, and only six of the eight
(75%) survived. The pattern closely approximates that observed in inbred Pink Pigeons (Jones et al., 1989) and Hawaiian Geese (Kear and Berger, 1980), except that chick survivorship in the 'Alala is considerably higher than that recorded in either of these species. Embryo deaths caused by inbreeding typically occur in the early and later stages of incubation (Romanoff, 1972), and this pattern of mortality has been claimed for the 'Alala. We have reviewed information provided by several sources, but are unable to conclude that such a pattern exists, owing to inaccuracies, discrepancies and omissions in the records, the relatively small sample of eggs, and our inability to rule out other potential sources of embryo death, such as bacterial infections, vitamin deficiencies, and mishandling of eggs (cf. Kuehler and Good, 1990). We do note, however, that the highest embryo mortality has occurred in a pair (Keawe x Mana) whose offspring have the highest inbreeding coefficient (0.25) of all chicks produced to date. It is also interesting to note that this female (Mana) has produced the largest number of abnormal eggs.
Finally, analyses of DNA ''fingerprints" of seven captive 'Alala suggest that genetic variation in the captive population is very low, as evidenced by high band-sharing coefficients (Duvall et al., 1991; Fleischer and Tarr, pers. comm., 1992). We do not know the degree to which wild 'Alala would have shared DNA-fingerprint bands when the population was larger, although this is currently being examined by removing DNA from museum specimens (Fleischer and Tarr, pers. comm., 1992). However, several other corvids have been examined recently (Figure 4.3), and the results support a hypothesis of elevated inbreeding among the captive 'Alala. J. Quinn (pers. comm., 1992) used Jeffreys 33.15 probe on a large sample of Florida Scrub Jays and found only 20% bandsharing among unrelated jays. Similarly low band-sharing occurred among unrelated American Crows (Fleischer and Tarr, pers. comm., 1992) and Common Ravens (P. Rabenold, pers. comm, 1992). In contrast, apparently unrelated 'Alala in captivity shared 60% of their bands using both Jeffreys 33.15 and M13 probes (Figure 4.3). This finding reinforces the necessity of both expanding the captive population as rapidly as possible, and adopting breeding strategies that minimize the rate of inbreeding within the captive population.
Brock (1991) recently compared band-sharing in Hispaniolan Parrots (Amazona ventralis) and the highly endangered Puerto Rican Parrot (A. vittata). She found that the average band-sharing coefficient was higher for unrelated Puerto Rican Parrots than for unrelated Hispaniolan Parrots and that, among mated pairs of captive Puerto Rican Parrots, reproductive success declined as band-sharing coefficients increased.
It must be pointed out that some of the captive 'Alala presumed to be "unrelated" (Figure 4.1) could have shared parents or grandparents. Furthermore, it is possible that small effective population sizes and high variance in lifetime reproduction led to reduced genetic variation (hence high average band-sharing) long before the population declines of 'Alala during this century. A high degree of band-sharing and apparent inbreeding among the captive 'Alala today, therefore, does not necessarily reflect an abnormally elevated level of inbreeding among the remnant wild population of the 1970s and 1980s.
We have no information on whether or not reproduction or survival of the wild birds during those years was affected by inbreeding. We also cannot be certain to what degree the existing level of inbreeding in the captive flock is affecting their ability to survive or reproduce.
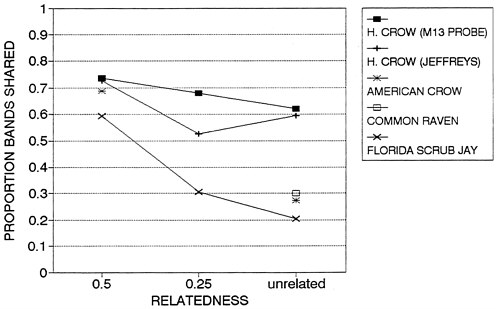
Figure 4.3 Comparison of DNA fingerprint band-sharing coefficients among 7 captive 'Alala (using 2 different probes) and wild populations of three other corvids (all using Jeffreys 33.15 probe). Unpublished Florida Scrub Jay results (N = 16 closely-related, 6 moderately-related, and 56 unrelated pair-wise comparisons) provided to the committee by J. Quinn, J.W. Fitzpatrick, and G.E. Woolfenden (pers. comm., 1992).
Animal Husbandry and Management
The husbandry and management of the captive 'Alala at Olinda are inadequate, and there is room for modification and improvement of existing methods and procedures. There is a strong tendency to become locked into invariable patterns when working with endangered species, and there is always room for experimentation. The old adage "nothing ventured, nothing gained" applies in aviculture as in other vocations, and creativity and experimentation should always be encouraged. Continual review of the avicultural literature and communication with colleagues are essential for developing or applying new techniques and procedures. To date, 39 species of corvids, including nine Corvus species, have been bred successfully in captivity (Table 4.5).
Many 'Alala eggs have been broken by the parents, and this source of egg loss has been a substantial drain on potential production. There is also some evidence that chicks were cannibalized by the parents at Pohakuloa. Egg-breaking and cannibalism, especially by males, have long been known in the breeding of captive corvids and have been documented in a wide variety of species, e.g., Eurasian Jay (Delacour, 1936), Red-Billed Blue Magpie (Richards, 1976), Southern Tree Pie (Roots, 1970), Crested Jay (C. Pickett, pers. comm., 1992), Chough (Edworthy, 1972), and Rook (Richards, 1976). In discussing corvid propagation, Shelton (1989) identified egg-eating and cannibalism as an important, but solvable, problem. With two species—the Green Magpie and the Magpie Jay—Shelton simply removed the males from the breeding enclosure as soon as the first egg of the clutch was laid. That procedure works well, because females in most, if not all, corvid species perform all the incubation duties. Also, inasmuch as captive females need only go to a food pan for their own food and that of the nestlings, neither eggs nor nestlings suffer from neglect. Relocating the male out of sight and, if possible, out of hearing range of the female has worked well at the Houston Zoo (L. C. Shelton, pers. comm., 1991, 1992).
The same technique should be attempted with pairs of 'Alala where egg-breaking has been documented and might provide an additional advantage of allowing natural incubation by the female for a period of several days. Natural incubation of eggs for 5–7 days has been shown in a number of species to enhance egg hatchability greatly, and such short-term incubation does not usually interfere with efforts to recycle females for additional laying. Repeated relaying has been documented in a variety of corvids, including the 'Alala. Because the development of effective procedures for full-term artificial incubation will continue to be hampered by the inadequate number of eggs laid each year, initial incubation by the female is strongly recommended. Corvids in most zoos and private collections are allowed to incubate and raise their own young, and with experience they do so efficiently. Consideration should also be given to allowing selected pairs to incubate and hatch their second or third clutches and rear the young. Such reproductive experience can act to strengthen the pair bond and increase future reproductive performance (cf. Derrickson and Carpenter, 1987).
Table 4.5 Captive Breeding of Corvidae
|
Genus and speciesa |
Common Name |
No. Successful Breeding Locations, 1961–1991 |
Multiple Generations |
Referencesb |
|
Platylophus |
||||
|
galericulatus |
Crested Jay |
1–3 |
No |
ISIS |
|
Cyanocitta |
||||
|
cristata |
Blue Jay |
4–6 |
No |
IZY |
|
stelleri |
Steller's Jay |
4–6 |
No |
Risdon (1960); Gibson (1980); ISIS |
|
Aphelocoma |
||||
|
coerulescens |
Scrub Jay |
1–3 |
No |
Partridge (1966); Webber and Cox (1987) |
|
Cissilopha |
||||
|
melanocyanea |
Bushy-Crested Jay |
1–3 |
No |
IZY |
|
sanblasiana |
San Blas Jay |
>6 |
No |
IZY; ISIS |
|
beecheii |
Purplish-Backed Jay |
1–3 |
Yes |
Todd (1980); IZY; ISIS |
|
Cyanocorax |
||||
|
caeruleus |
Azure Jay |
1–3 |
No |
IZY |
|
cristatellus |
Curl-Crested Jay |
1–3 |
No |
IZY |
|
chrysops |
Plush-Crested Jay |
>6 |
Yes |
IZY; ISIS; Coles (1980) |
|
mysticalis |
White-Tailed Jay |
1–3 |
No |
ISIS |
|
yncas |
Green Jay |
>6 |
Yes |
Roles (1971); IZY; ISIS |
|
Calocitta |
||||
|
formosa |
Magpie Jay |
4–6 |
Yes |
IZY; ISIS |
|
Garrulus |
||||
|
glandarius |
Eurasian Jay |
4–6 |
No |
IZY |
|
lanceolatus |
Lanceolated Jay |
1–3 |
No |
Goodwin (1954) |
|
lidthii |
Purple Jay |
1–3 |
No |
Oliver (1964); IZY |
|
Genus and speciesa |
Common Name |
No. Successful Breeding Locations, 1961–1991 |
Multiple Generations |
Referencesb |
|
Urocissa |
||||
|
caerulea |
Taiwan Blue Magpie |
4 |
No |
IZY; ISIS |
|
flavirostris |
Yellow-Billed Blue Magpie |
1–3 |
No |
IZY; ISIS |
|
erythrorhyncha |
Red-Billed Blue Magpie |
>6 |
Yes |
IZY; ISIS, Richards (1976) |
|
Cissa |
||||
|
chinensis |
Green Magpie |
>6 |
No |
IZY; ISIS; Coles (1978) |
|
thalassina |
Short-Tailed Magpie |
1–3 |
No |
ISIS |
|
Cyanopica |
||||
|
cyana |
Azure-Winged Magpie |
>6 |
Yes |
IZY; ISIS; Wayre (1970a) |
|
Dendrocitta |
||||
|
vagabunda |
Rufous Treepie |
4–6 |
No |
IZY; ISIS |
|
leucogastra |
Southern Treepie |
1–3 |
No |
Roots (1970); IZY |
|
frontalis |
Collared Treepie |
1–3 |
No |
IZY |
|
Crypsirina |
||||
|
temia |
Racket-Tailed Treepie |
1–3 |
No |
IZY; ISIS |
|
Pica |
||||
|
pica |
Magpie |
>6 |
Yes |
IZY; ISIS |
|
Nucifraga |
||||
|
caryocatactes |
Nutcracker |
1–3 |
Yes |
IZY |
|
Genus and speciesa |
Common Name |
No. Successful Breeding Locations, 1961–1991 |
Multiple Generations |
Referencesb |
|
Pyrrhocorax |
||||
|
pyrrhocorax |
Chough |
>6 |
Yes |
IZY; ISIS; Edworthy (1972) |
|
graculus |
Alpine Chough |
>6 |
Yes |
Wayre (1970b); IZY |
|
Corvus |
||||
|
monedula |
Jackdaw |
>6 |
Yes |
IZY |
|
frugilegus |
Rook |
4– |
No |
IZY; Richards (1973) |
|
brachyrhynchos |
Common (American) Crow |
1–3 |
No |
IZY |
|
corone |
Carrion (Hooded) Crow |
4–6 |
No |
IZY |
|
alba |
Pied Crow |
1–3 |
No |
IZY; ISIS |
|
hawaiiensis |
Hawaiian Crow |
1–3 |
Yes |
IZY |
|
ruficollis |
Brown-Necked Raven |
1–3 |
No |
IZY |
|
corax |
Raven |
>6 |
Yes |
IZY; Gwinner (1965); Wolinski (1989) |
|
albicollis |
White-Necked Raven |
1–3 |
No |
ISIS |
|
a Taxonomy follows Morony et al. (1975). b IZY, International Zoo Yearbook, Vols. 1–29 (1959–1987); ISIS, International Species Inventory System, Species Distribution Reports 1984–1991; L.C. Shelton (pers. comm., 1991, 1992). |
||||
Usnik (1990) suggested that attendants' time spent around the birds during the day during feeding and cleaning of cages should be increased over the normal pattern and noted that this appeared not to affect the birds. We wholeheartedly concur. Increased contact with the birds will condition them to the human presence and in the long-run will make unexpected periods of contact much less stressful. Sanitation in the enclosure would also be improved substantially.
Because 'Alala are territorial, we believe that visual barriers should be used to prevent adjacent pairs from seeing each other directly. In many species, when males can see each other but are thwarted from physical pursuit or contact, females can become the victims of redirected aggression. The use of visual barriers would be best applied initially to pairs that have had a history of sexual or social incompatibility or that have exhibited male aggression directed at the female. Consideration should also be given to increasing the complexity and naturalness of the breeding and juvenile enclosures by providing more plantings, perching material, and nesting baskets. The latter could be placed appropriately to accommodate remote monitoring.
At present, the captive 'Alala receive a rather complicated diet consisting of at least eight food types (e.g., native fruits, commercial exotic fruit, nectar, corn, dry Hi-Pro dog kibble, low-iron bird of paradise pellets, two 1-day-old chicks or a single rat pup, mealworms, and a blended egg "omelet"). This type of diet can pose a number of problems. First, when offered such a variety, many birds will consume only those items that they prefer, and this selectivity can lead to nutritional deficiencies. Such selective feeding has been noted in the captive 'Alala (F. Duvall, pers. comm., 1991, 1992) although its nutritional impact remains unknown. Second, dominant males can monopolize or consume items essential for the female. Third, a diet that includes chicks and rat pups can expose the birds to gram-negative bacteria, which can adversely affect reproduction. Because crows frequently cache food before feeding, the possibility of bacterial contamination is increased.
Our final concern is related to the dietary amounts of heme iron to which the birds can be exposed: three captive 'Alala have succumbed to iron storage disease—Kekau (Patuxent, 1978), Iola-Elau (Pohakuloa, 1979), and Imia (Pohakuloa, 1981). Although the etiology of this disease is poorly understood (Fairbanks et al., 1971; Lowenstein and Petrak, 1978; Davies, 1979; Taylor, 1985; Kincaid and Stoskopf, 1987), it is clear that many fruit-eating species—such as birds of paradise, mynahs, cotingas, and toucans—and others are susceptible to it when maintained on captive diets high in heme iron and acidic fruits. Both chicks and rat pups fed at Olinda are high in heme iron. Serious consideration should be given to simplifying the diet greatly—reducing it to one or two principal items that are palatable, are nutritionally balanced, and contain nonheme iron, and then supplementing this base diet with available native fruits. Use of such a simplified diet would also reduce cost and preparation time.
Staffing and Administration
As a result of interviews with current and former Olinda staff members and the review of internal documents and memoranda provided by USFWS and the Hawai'i Department of Land and Natural Resources, the committee concludes that there have been substantial administrative and personnel problems at Olinda and that staffing and staff training requirements need to be increased. It is clear to the committee that the programmatic problems and deficiencies are recognized by the responsible administrators in these two agencies and that appropriate remedial actions have been initiated or are under review. The committee believes, however, that these problems must be addressed and fully resolved as soon as possible.
The proposed expansion of the Olinda facility to include propagation efforts for three more species of endangered Hawaiian forest birds will obviously require additional staff if the operation is to run effectively 7 days per week. At a minimum, the personnel added to the existing staff should include a facility administrator-curator, a full-time veterinarian, another avicultural assistant-lead keeper, and two animal caretakers.
While the 'Alala facilities at Olinda clearly need to be expanded in the immediate future, breeding facilities should also be developed at an additional, second site (possibly even a third site) on the Hawaiian islands to minimize the possibility of significant losses resulting from disease outbreaks or other potential catastrophes. Because many captive breeding programs for endangered species have suffered severe setbacks as a result of disease outbreaks (see Derrickson and Snyder 1992 for examples and discussion), division of the captive flock and the adoption of strict quarantine procedures and protocols must be considered high priorities.
Additional specific recommendations for the improvement of the captive-breeding program are presented in Chapter 7.


















