6
Personnel Issues in Human Genetics
Historically, genetic tests have been administered and interpreted by highly trained health professionals working in academic health settings, usually with a strong genetics research and service record. In the future, however, genetic tests will become available for a growing variety of monogenic and complex diseases and for susceptibility to more common disorders such as breast, colon, and other cancers. Testing on such a broad scale will necessarily move us beyond the models of service delivery and professional roles that have characterized genetic testing and screening in the past. Increasingly, genetic tests will be offered and interpreted within the context of the mainstream of medicine in primary care practice—including pediatrics, obstetrics, internal medicine, and family practice in a variety of individual and group practice settings.
This exciting and challenging prospect for the future involves a large pool of potential personnel for genetic testing, screening, education, and counseling, but will they be prepared to play this role? How will primary care practitioners be trained to provide these services appropriately and to understand the complexities and limitations of genetic tests? How will they be trained to provide the nondirective counseling that is absolutely essential in reproductive decisions and in testing for disorders for which there is no effective treatment? Once trained, how will primary care practitioners keep up with the exponential growth in knowledge about the role of genetics in health and about genetic tests?
This chapter presents an overview of available data on specialized genetics personnel and primary care practitioners, as well as information on their training in and knowledge of genetics. The chapter also examines the personnel implications arising from the trends in genetic testing and screening discussed elsewhere
in the report. The committee considered whether there will be enough adequately trained health professionals in the future to handle the potential volume, diversity, and complexity of genetic tests, and to perform specific functions in genetic services, such as laboratory testing, taking family history, diagnosis, education, counseling, technical support, and research.1 This chapter addresses the separate issues of specialized genetics education for specialists and genetics education for generalists (primary care practitioners), recognizing that both will be needed as more genetic tests become available. The following section focuses on current and future supplies of genetic specialists and their certification. The latter half of the chapter addresses, more broadly, the issues surrounding genetics education in medical school and general practice, and the potential role of other health professionals in providing genetics services. The chapter concludes with recommendations intended to help prepare the nation for changes likely to be brought about by widespread genetic testing. These recommendations include suggestions for research to better inform policy makers as they prepare for that future.
GENETIC SPECIALISTS
Many types of professionals provide specialized genetic services: physicians, Ph.D. clinical geneticists, genetic counselors, nurses, and social workers. Other individuals trained as research scientists are involved in genetics research. A large medical center that provides genetic services and conducts research is likely to employ individuals at all levels—master's, Ph.D., and M.D. Smaller private or community-based hospitals are likely to employ master's-level genetic counselors or nurses and physicians trained in genetics. Until recently, Ph.D. geneticists and genetic counselors were not able to see patients without the oversight of an M.D., but changes in the certification status of these individuals may change their roles in the clinical setting (see below). Although available data indicate that the numbers of individuals graduating from human genetics training programs are increasing, it is not clear that this increase is occurring at the rate necessary to ensure adequate and appropriate levels of support for genetic services in the future.
Furthermore, the geographic distribution of genetic specialists will be critical in ensuring access to individuals needing genetic services. Currently, genetics professionals tend to be clustered in the Northeast and on the West Coast, as well as in the Chicago area. A survey of genetic counselors and nurses working in genetics showed a heavy concentration of counselors in five states, with 43 percent of respondents located in California, Illinois, New Jersey, New York, and Pennsylvania (OTA, 1992b). This uneven distribution of scarce genetic practitioners is even more limiting given the specialized expertise of many genetic centers in a relatively small number of genetic disorders. As a result, families must often travel long distances to receive specialized genetic services for a particular genetic disorder.
Reimbursement policies regarding genetic testing also have a significant effect on personnel issues. Currently, genetic counselors cannot be reimbursed di-
rectly; genetic counseling must be authorized and billed by a physician for insurance reimbursement. If these reimbursement practices continue, it is not clear whether full genetics services will be provided as needed, including essential education and counseling. Because primary care practitioners are able to obtain reimbursement for genetic consultation as a type of office visit, patients might be more inclined to seek care through this route than through a route that could involve out-of-pocket expenses (see Chapter 7).
Some of the controversy about personnel needs stems from uncertainty about who will be providing genetic services in the future. For example, one study estimated that a minimum of 651,000 counseling hours would be required annually if the maximum estimate of 6 to 8 million preconceptional couples are screened for cystic fibrosis (CF) carrier status (Wilfond and Fost, 1990). Given the current number of practicing genetic counselors in the United States today, this translates to 17 weeks per year from each genetic counselor just to serve CF-related clients. This study concluded that CF screening could not be offered solely through specialized genetics centers; it is likely that this and genetic tests of significance in making reproductive decisions, such as carrier screening, will be offered increasingly by obstetrician-gynecologists.
Background Data on Genetics Professionals
As early as 1985—prior to the initiation of the Human Genome Project—concerns were raised about the availability of clinical genetics personnel. In a survey of 476 programs providing genetic services in the United States, 195 programs supplied data on predoctoral and postdoctoral trainees (Finley et al., 1987). A total of 524 students were enrolled in medical genetics training in the 195 training programs; of these, 224 (43 percent) were postdoctoral candidates, 193 (37 percent) were doctoral candidates, and 107 (20 percent) were master's candidates. The study also asked about the number of job vacancies, both current and anticipated, for the next five years: in 1985, there were 150 vacancies, of which 36 percent were for cytogenetic technicians, 27 percent for M.D. clinical geneticists, and 19 percent for genetic counselors. Over the period 1986 through 1990, more than 600 vacancies were anticipated by these 195 programs (Finley et al., 1987).
The American Society of Human Genetics (ASHG)—a nonprofit professional society founded in 1948—serves as the primary scientific and professional society for all human geneticists in North America. It routinely surveys its membership as well as the graduate and postgraduate training programs from which its members come. Its 1989 survey gathered data on its approximately 4,000 members (Garver and Lent, 1990). Membership distribution according to highest degree obtained is 44 percent Ph.D.'s, 29 percent M.D.'s, 20 percent master's, and 7 percent M.D.Ph.D.'s. Graduate degree distributions appear to be changing (Table 6-1).
In terms of current enrollment in human genetics training programs, 64 percent of the students are in Ph.D. programs (both predoctoral and postdoctoral), 22
TABLE 6-1 Graduates from Human Genetics Training Programs, 1984-1992
|
Degree |
1984-1985 |
1986-1987 |
1988-1989 |
1991-1992 |
|
Masters, Genetic Counseling |
147 |
156 |
168 |
155 |
|
Other Masters |
97 |
62 |
81 |
118 |
|
Subtotal |
234 |
218 |
249 |
273 |
|
Doctoral, Ph.D. in Human Genetics |
155 |
129 |
132 |
185 |
|
Postdoctoral |
|
|
|
|
|
M.D. |
108 |
134 |
159 |
157 |
|
Ph.D. |
81 |
88 |
122 |
185 |
|
M.D./Ph.D. |
3 |
1 |
6 |
7 |
|
Subtotal |
192 |
223 |
287 |
349 |
|
Grand total |
581 |
570 |
662 |
807 |
|
SOURCES: Riccardi and Smith, 1986; Friedman and Riccardi, 1988; Murray and Toriello, 1990; Blitzer, 1992. |
||||
percent are in master's-level programs, and 14 percent are in M.D. or M.D-Ph.D. programs (Blitzer, 1992). Although there has been steady growth in the number of graduates of human genetics training programs since 1985 (807 individuals completed training in 1992, compared with 581 in 1985), postdoctoral fellows account for approximately 74 percent of the growth (Riccardi and Smith, 1986; Murray and Toriello, 1990). Many of these programs are not oriented to delivering clinical genetics services, particularly preparation in the behavioral sciences essential for genetic counseling. Also, since most Ph.D.-level geneticists enter into research- or laboratory-oriented activities, the growing number of Ph.D. postdoctoral students appears to indicate a trend toward research or laboratory careers.
From limited available data, it appears that only a relatively small percentage of people attaining these doctorates enters medical genetics, although some of these Ph.D. geneticists are also likely to work in clinical laboratories. Since the majority of genetic testing is currently being done in research laboratories, some of this pool of Ph.D. geneticists is likely to be involved in human genetics and genetic testing and screening as part of research programs; however, most Ph.D.'s will not be providing traditional genetics services or practicing in traditional medical genetics settings. Since they do not have the clinical training required for some aspects of medical genetics, Ph.D.'s have often been discouraged from entering medical genetics.
Individuals with M.S. degrees from established programs in genetic counseling are more likely to pursue traditional clinical genetics service careers. However, the number of graduates with an M.D. or M.S. has remained relatively stable, with only slight increases in the numbers of physicians specializing in genetics.
Of those M.D.'s who do complete their training in genetics, most are in pedi-
atrics (59 percent), followed by internal medicine (12 percent), obstetrics (10 percent), and ''other" (16 percent). As genetic tests for presymptomatic and predispositional assessment become more widely available, therefore, there may not be enough physicians trained to provide the necessary specialized genetics services, including the education and genetic counseling that will be essential if and when more widespread genetic testing and screening develop in the future. This will require close attention over the next three to five years.
Another growing area of concern involves the effects of market forces on the training and career paths of genetics professionals. Anecdotal evidence suggests that commercial laboratories are drawing personnel away from academic laboratories. This poses problems for the future training of genetics professionals, since commercial facilities are less likely to provide advanced clinical training than academic centers. For example, the committee heard reports that commercial laboratories recently have begun to buy genetic testing laboratories in academic institutions and to discontinue fellowships and other advanced genetics training in those laboratories.
Although there is a need for trained genetics personnel for research and laboratory testing, the readily available funding for research training appears to be leading more genetics students to enter career paths leading to potential research careers rather than to clinical genetics careers.
Training Programs
The total number of human genetics training programs increased slightly between 1984 and 1992, from 99 to 111 (see Table 6-2) (Riccardi and Smith, 1986; Friedman and Riccardi, 1988; Murray and Toriello, 1990; Blitzer, 1992). Approximately 40 percent of human genetics graduates have come from 10 percent of the human genetics training programs. The American Board of Medical Genetics (ABMG) accredits most U.S. human genetics training programs, although it accredits only the clinical training sites of the master's-level genetic counseling programs. ABMG certification by subspecialty is shown in Figures 6-1 and 6-2 and Table 6-3.
TABLE 6-2 Number of North American Human Genetics Training Programs
|
Programs |
1984-1985 |
1986-1987 |
1988-1989 |
1990-1991 |
1992-1993 |
|
Total listings |
99 |
106 |
120 |
120 |
111 |
|
Degree-granting |
68 |
66 |
83 |
83 |
69 |
|
Nondegree-granting |
31 |
40 |
37 |
37 |
42 |
|
SOURCES: Riccardi and Smith, 1986; Friedman and Riccardi, 1988; Murray and Toriello, 1990; Blitzer, 1992 |
|||||
TABLE 6-3 ABMG Certification by Year and Subspecialty Area
|
|
No. of Diplomates per Exam Year |
|
|
|||
|
Subspecialty |
1981 |
1984 |
1987 |
1990 |
Total No. of Certificates |
% of Diplomatesa |
|
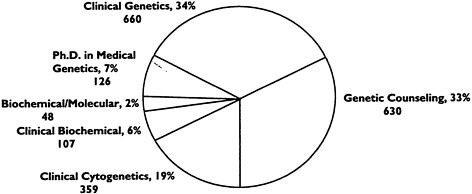
Clinical biochemical |
57 |
24 |
26 |
48b |
155 |
9 |
|
Clinical cytogenetics |
125 |
71 |
100 |
63 |
359 |
22 |
|
Genetic counseling |
169 |
143 |
177 |
141 |
630 |
38 |
|
Clinical genetics |
286 |
127 |
111 |
136 |
660 |
40 |
|
Ph.D. medical genetics |
56 |
31 |
26 |
13 |
126 |
8 |
|
Total |
693 |
396 |
440 |
401 |
1,930 |
|
|
a Percentage of total number of diplomates (N = 1,639); certification in more than one subspecialty possible. b Certified as biochemical-molecular geneticists (1990 only); since 1993 a separate subspecialty exam in molecular genetics has been available. SOURCE: Records of the American Board of Medical Genetics (1991). |
||||||
The curriculum of doctoral or postdoctoral training in human genetics has not been extensively reviewed. However, as part of the process of accreditation for their clinical training through ABMG, human genetics training programs must submit extensive data on their programs (see Box 6-1).
Master's-Level Genetic Counselors
The master's-level genetic counselor is a relatively new addition to the human genetics community. There are approximately 1,000 master's-level genetic counselors practicing in the United States, 100 times more than the 10 first graduated in 1971. To date, genetic counselors have been certified by the ABMG (currently 68 percent are certified), and their training has both reflected and shaped the requirements of that board.
Genetic counselors formed their own professional organization in 1979, the National Society of Genetic Counselors (NSGC). Current membership includes more than 1,000 individuals working in the United States and several foreign countries. More than 80 percent of the members are in clinical practice, with most working in a university medical center or a private hospital (OTA, 1992b; Uhlmann, 1992).
The curriculum of master's-level genetic counseling training programs has evolved over time and is a balance of medical genetics, practical and theoretical counseling, and behavioral sciences. During the 1970s, a series of meetings were held to discuss the role and educational needs of the genetic associate or genetic counselor (Genetics Associates, 1979). Then, in 1989, a conference held in Asilomar reevaluated recommendations for the minimum program curriculum of master's-level training in genetic counseling (Walker et al., 1990). The recommenda-
|
BOX 6-1 ABMG Accreditation Requirements The American Board of Medical Genetics (ABMG) conducts an extensive review of doctoral and postdoctoral training in programs applying for its accreditation. Its application process includes:
|
tions were based in part on requirements for certification through the ABMG. The need for and desirability of training beyond the master's level in genetic counseling (i.e., doctoral study) was also discussed along with alternatives to master's level training to overcome a projected shortage of genetic counseling personnel. A 1989 survey of NSGC members indicated that just over half of those responding saw a need for a doctoral (Ph.D.) degree in the field of genetic counseling, while 30 percent were undecided (Gaupman et al., 1991). To date, there is one program (University of Pittsburgh) offering a doctoral degree in genetic counseling.
The Asilomar conference recommended a minimum curriculum at the master's level that includes seven specific didactic course work content areas, as well as a broad scope of clinical experience; these include
-
principles and application of human genetics and related sciences;
-
principles and practice of clinical and medical genetics;
-
genetic laboratory methods;
-
theory and application of interviewing and counseling;
-
social, ethical, legal, and cultural issues;
-
health care delivery systems and principles of public health; and
-
teaching.
The scope of clinical experience includes participation as primary genetic counselor in at least 50 cases, in three or more clinic settings (e.g., general genetics, prenatal diagnosis, specialty diseases); supervision by a geneticist or ABMG-certified genetic counselor; and demonstration of competence in the skills of genetic counseling.
Experience in several additional areas was deemed desirable, but not mandatory, for minimum clinical experience, including service delivery, screening programs, cross-cultural issues, community and professional education, and clinic administration. Master's-level genetic counselors receive specialized multidisciplinary training and experience to prepare them for counseling related to a wide variety of genetic disorders, including late-onset adult disorders, as well as birth defects. The committee strongly recommends the incorporation of these essential areas into all genetic counseling curricula, particularly as new programs develop.
Over the past 20 years, master's-level graduate programs in genetic counseling have increased to 14 in the United States, and one in Canada (see Table 6-4). Combined, they produce approximately 81 graduates each year (77 in the United States and 4 in Canada), but 30 percent of all graduates in a given year come from one training program, Sarah Lawrence College (New York), the oldest program in the United States.
TABLE 6-4 Master's-Level Programs in Genetic Counseling in the United States and Canada
|
School |
Average No. of Graduates per Year |
|
Howard University |
3 |
|
Medical College of Virginia |
1 |
|
Northwestern University |
4 |
|
Sarah Lawrence College |
23 |
|
University of California, Berkeley |
8 |
|
University of California, Irvine |
3 |
|
University of Cincinnati |
2 |
|
University of Colorado |
5 |
|
University of Michigan |
4 |
|
University of Minnesota |
3 |
|
University of Pittsburgh |
12 |
|
University of South Carolina |
3 |
|
University of Texas, Houston |
1 |
|
University of Wisconsin, Madison |
5 |
|
McGill University, Canada |
4 |
|
Total |
81 |
|
SOURCE: Riccardi and Smith, 1986; Friedman and Riccardi, 1988; Murray and Toriello, 1990; Blitzer, 1992. |
|
|
BOX 6-2 Genetic Counseling Personnel in New York State A recent, limited pilot survey was conducted by Zinberg and Greendale (1991) to assess whether a shortage of genetic counselors existed in New York State in 1990. In their study, 57 comprehensive and specific genetic disease centers were asked about their level of genetic counselor staffing. A total of 103 ABMG-certified or board-eligible genetic counselors were employed at the 57 centers. Ninety-four percent of these were graduates from an NSGC-recognized genetic counseling training program. This study also examined the overall attrition rate and found it to be very high. In the previous two years, 56 genetic counselors had been hired, the majority of whom were to fill 34 positions left vacant in New York by staff departures. Recognizing the need for additional genetic counselors, the state of New York developed two new initiatives to meet personnel demands. First, a new certificate program is offered by the Division of Medical and Molecular Genetics at Mt. Sinai Medical Center to prepare individuals with master's degrees in related fields for ABMG certification in genetic counseling. Second, as part of their request for funding for genetic services, New York genetics providers may now obtain partial support, in the form of salary, stipend, or scholarship, for a graduate student enrolled in a master's-level program in genetic counseling. Once graduated, the student would be committed to work at the sponsoring institution for a period of three years. |
Training support for master's-level genetic counselors has been minimal. The U.S. Department of Health and Human Services (DHHS) provides no financial support for the training of genetic counselors or for improving genetics education in medical schools (Holtzman, 1989). However, the Maternal and Child Health Program of DHHS's Health Resources and Services Administration provides support to the Council of Regional Networks for Genetic Services (CORN) for some continuing professional education programs for physicians and postdoctoral students, but not for master's-level counselors. The same explosion of new genetic information affects genetic counseling, and continuing education is just as essential for genetic counselors (NSGC, 1991; Gettig, 1992).
Although an average of 77 graduates entered the nation's work force annually from 1984 to 1992, the demand for genetic counselors continues to exceed the available supply. According to the NSGC executive office, from 1988 to 1991 there were at least 35 unfilled genetic counseling positions listed with its Jobs Hotline at any given time. The total number of unfilled positions nationwide is unknown. However, an attempt to answer this question for the state of New York suggests that the number is high (see Box 6-2). Finally, there appears to be some attrition of existing master's-level genetic counselors into full-time administration or away from the profession altogether. Not nearly enough is known at this time
about how many master's-level genetic counselors are now or will be needed, since so many uncertainties exist about the nature and scope of their role in the future.
Non-Master's-Level Counselors
Research to examine a "train-the-trainer" model for increasing the availability of genetic counseling in the United States is being supported by the Ethical, Legal, and Social Implications (ELSI) Program of the National Center for Human Genome Research at the National Institutes of Health (NIH). As the profession has developed, master's-level counselors have begun taking on the role of trainers of other health professionals. In some clinical settings, master's-level genetic counselors are training non-master's-level individuals to meet the demand for patient education related to a single diagnostic category of disease. In other settings, non-master's-level individuals assist genetic counselors in overcoming cultural, linguistic, geographic, or economic barriers.
Individuals who assist genetic counselors, often called "single-gene counselors," "single-disorder counselors," or ''non-master's-level counselors," do not have the same training as master's-level genetic counselors and have not been eligible for ABMG certification. With the growth of genetic services and increasing demands on the time and resources of traditionally trained counselors, the use of these individuals has stimulated debate. A number of programs have been developed to train non-master's-level educators or counselors (see Box 6-3).
Advocates for the use of single-disorder counselors cite the limited number of practicing genetic counselors and the increasing development of genetic testing as reasons to support this type of training. Single-disorder counselors could also improve the quality of service in underserved, culturally diverse populations that are disproportionately affected by a particular genetic disease (OTA, 1992a).
Those opposed to single-disorder counselors express concern about what they view as a lack of genetics and genetic counseling training. There is also concern about whether single-disorder counselors have a broad enough view of clinical genetics to identify complex and obscure risks of other genetic disorders in their patients. Since taking a family history often exposes previously unknown or undiagnosed genetic disorders or predispositions, individuals who focus on one category of disease might not recognize the need to further investigate peripheral information.
An NSGC task force made the following recommendations to the society:
-
acknowledge the current and predicted personnel needs for genetic counselors, as well as the shortage of master's-level genetic counselors;
-
recognize the existing use of non-master's-level counselors and the benefits they offer;
-
educate NSGC membership regarding the potential use of these individuals;
|
BOX 6-3 Single-Disorder Educator-Counselor Programs Genetic counselors have been involved in developing and conducting training programs in several states. In California and Massachusetts, established programs lasting four days to one month, respectively, provide training to become a sickle cell educator-counselor or a hemoglobin trait counselor. Students are trained and supervised by master's-level genetic counselors. In addition to training, the California program also provides certification for sickle cell educators-counselors. The sickle cell educators must attend a four-day training course and pass a final certification examination. The sickle cell counselor must meet these same requirements plus attend an additional two-day counseling course and counseling practicum. San Francisco General Hospital utilizes genetic aides to provide patient advocacy and education, and to act as liaisons between non-English-speaking patients and the genetic counselor. These bilingual or multilingual individuals are trained and supervised by genetic counselors, who continue to serve as case managers. A similar genetic aide program also exists at the University of California at San Diego. To date, the numbers of trained genetic aides remain relatively small. However, with the increasing Southeast Asian population in California, the need for multilingual individuals to provide state-supported genetic services is increasing. Actual job titles of such individuals vary according to their role or function and include, but are not limited to, single-gene counselor, single-disorder counselor, hemoglobin trait counselor, sickle cell educator, sickle cell counselor, genetic counseling aide, genetics educator, and genetic interpreter. |
-
support the use of non-master's-level counselors in specific settings where genetic counselors can be involved in training, evaluating, and supervising these individuals; and
-
establish a committee to collaborate with other organizations.
Certification and Accreditation of Genetics Specialists
Genetics professionals who are physicians are licensed by the states as physicians. Certification procedures for specialties are voluntary but might be a requirement for employment or reimbursement in some settings. Genetic counselors and Ph.D. geneticists are not licensed by states, but until 1992 were certified by the ABMG (see Box 6-4). Ph.D. geneticists continue to be certified by the ABMG.
As of the 1990 exam cycle, 1,639 total diplomates had been certified. A breakdown of the number of diplomates by year and subspecialty is shown in Table 6-3. The percentage of certificates awarded according to subspecialty area is depicted in Figure 6-1.
|
BOX 6-4 Certification by the American Board of Medical Genetics The ABMG was incorporated in 1980 at the request of the American Society of Human Genetics to provide accreditation of training programs and certification of individuals in the United States who provide medical genetics service. Until 1992, certification was available for the following subspecialties: clinical geneticist, Ph.D. medical geneticist, genetic counselor, clinical biochemical geneticist, and clinical cytogeneticist. Four exam cycles have occurred: 1981, 1984, 1987, and 1990. During the 1990 cycle, dual certification as a clinical biochemical/molecular geneticist was available; independent certification as a clinical molecular geneticist has been available since 1993. Eligibility to sit for ABMG examinations requires application and credential review. Eligibility criteria include, but are not limited to, training in a required set of clinical experiences and verification of training in an ABMG-accredited clinical genetics training program. For the subspecialties of M.D. clinical geneticist, Ph.D. medical geneticist, clinical biochemical geneticist, clinical cytogeneticist, and clinical molecular geneticist, a minimum of two years of postdoctoral training experience through appropriate ABMG-accredited training programs is required. The genetic counselor subspecialty requires at least one year of clinical experience in one or more ABMG-accredited genetic counseling training sites, although the master's-level genetic counseling programs per se are not accredited by ABMG as they are for other subspecialties. In Canada, an equivalent certifying body to the U.S. ABMG, the Canadian College of Medical Geneticists, also provides certification in clinical genetics, Ph.D. medical genetics, cytogenetics, biochemical genetics, and molecular genetics, but not for genetic counselors. |
The status of certification changed in 1992 when the American Board of Medical Specialties (ABMS) agreed to admit the American Board of Medical Genetics to its ranks, the first such admission since 1979. ABMS has never admitted a board that certifies non-doctoral-level individuals and had, to this date, only admitted one other board that certified doctoral-level specialists who were not physicians. In order to gain admission, the ABMG had to agree not to certify master's-level genetic counselors in the 1993 examination.
Acceptance of both M.D.'s and Ph.D.'s in the ABMS was considered a victory by some, as was the admission to ABMS, which had previously not recognized medical genetics as a bona fide medical specialty. This precedent also paves the way for the newly formed American College of Medical Genetics (ACMG) to seek recognition by the American Medical Association (AMA) and to be granted a seat in the AMA House of Delegates, which is essential for the development of additional current procedural terminology (CPT) codes for genetic procedures (used in billing and insurance reimbursement policy)(see Chapter 7). Others are concerned that this medical subspecialization may further isolate medical genetics
from general medicine at a time when it most needs to be integrated into routine medical education and practice, and that it is motivated to improve revenues for genetic services. For the first time, Ph.D. clinical geneticists will be able to set fees, be reimbursed by insurance companies, and not be under the supervision of an M.D. Genetic counselors who were not already certified by the ABMG have established an American Board of Genetic Counseling.
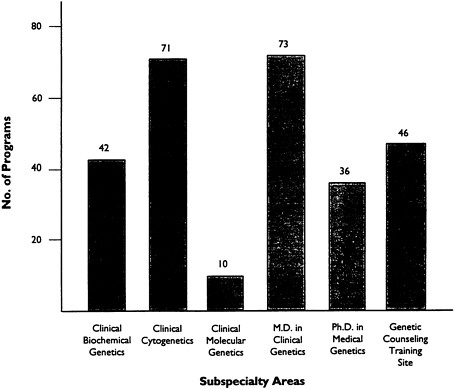
Beginning in 1993, the ABMG will provide accreditation of U.S. clinical genetics training programs for all six subspecialties; prior to 1990, molecular genetics accreditation was not available. Accreditation of Canadian training programs is available through the Canadian College of Medical Genetics (CCMG) in the same areas, except genetic counseling. As of May 1991, 81 programs had received accreditation through the ABMG in at least one of the six subspecialty areas (Table 6-4). The CCMG has accredited eight training programs. A breakdown of the training programs accredited by ABMG and CCMG is shown in Table 6-5.
At present, there is no national certification or standard for training, supervision, or job responsibilities for genetics service providers without graduate training. The state of California provides both training and certification for sickle cell educators and sickle cell counselors. Certification is based on completion of the California training program requirements and passing the certification examination. Other states are looking at this program as a model. In addition, a proposal to initiate a national effort to provide training accreditation and certification of sickle cell counselors-educators was endorsed by CORN at its fall 1990 meeting, but this proposal has not yet been implemented.
Related Genetics Certification and Training: Cytogenetics
In 1981, the Association of Cytogenetic Technologists (ACT), in conjunction with the National Certification Agency of Medical Laboratory Personnel (NCAMLP), developed and administered a technologist certification examination in cyto
TABLE 6-5 Accredited Clinical Genetics Training Programs by Subspecialty Area
|
Subspecialty |
ABMG |
CCMG |
Total |
|
Clinical biochemical genetics |
42 |
44 |
6 |
|
Clinical cytogenetics |
71 |
6 |
77 |
|
Clinical molecular genetics |
10 |
3 |
13 |
|
M.D. clinical genetics |
73 |
8 |
81 |
|
Ph.D. medical genetics |
36 |
3 |
39 |
|
Genetic counseling training site |
46 |
NA |
46 |
|
SOURCES: ABMG Executive Office, personal communication, May 1991, Murray and Toriello, 1990 |
|||
genetics. More than 1,500 individuals have been certified. Recertification is also available, either by accumulation of continuing education units or by retaking and passing the certification examination.
The majority of cytogenetic technologists have university degrees in scientific fields. Historically, however, cytogenetic training has been acquired on the job. With rapid developments in the field of cytogenetics in recent years, there has been an increasing demand for trained cytogenetic technologists. Training programs in cytogenetic technology were established in 1980 to meet this demand.
The annual number of graduates has been approximately 60 to 80 (1987 data). Job surveys conducted by ACT in cooperation with other groups document a demand that exceeds the supply of qualified people in clinical cytogenetics. Since 1987, more than 400 jobs have been available each year, with only 60 to 80 graduates. The current number of graduates is anticipated to meet only 10 to 15 percent of the human resource needs of clinical cytogenetic laboratories in the future (Fatemi and Gasparini, 1990).
GENETICS INSTRUCTION IN MEDICAL SCHOOLS
Increasingly, primary care physicians will be called on to interpret tests results, relay this information to the patient in an accurate and sensitive manner, and deal with the sometimes profound impact such information can have on the patient's psychological well-being. Attention to genetics education in the nation's medical schools has not increased to meet this growing demand (see Box 6-5). The emergence of a variety of genetic indicators of the more common multifactorial conditions, such as diabetes and heart disease, means that genetics will increasingly touch nearly every discipline in medicine. Yet perhaps the persistent notion that genetic diseases are inevitable and untreatable has delayed more rapid integration of genetics into medical school curricula (Motulsky, 1983; Davidson and Childs, 1987).
Few studies have examined physicians' knowledge of genetics or their awareness of the availability of tests for genetic disorders (Naylor, 1975; Lemkus et al., 1978; Kapur et al., 1983; Firth and Lindenbaum, 1992; Hofman et al., 1993). Recent data show that there is a significant upward trend in total genetics knowledge as a function of year of graduation from medical school. In fact, year of graduation is the most powerful predictor of knowledge (Hofman et al., 1993). Having a required genetics course in medical school was also a significant predictor of genetics knowledge.
The level of instruction in genetics in medical schools has been a source of concern and recommendation for nearly 20 years (NAS, 1975; Childs et al., 1981; Riccardi and Schmickel, 1988; Graham et al., 1989). The 1975 National Academy of Sciences report reported on a survey that included questions on past medical education and identification of potential barriers to genetic screening by physicians (pediatricians, internists, and obstetrician-gynecologists). Nearly 75
|
BOX 6-5 Reflections on the Inclusion of Genetics in Medical Education By Barton Childs One way of discussing the assimilation of genetics into medical education is to ask how genetic risks should be presented in the curriculum. Should it be in practical ways: for example how to calculate risks and impart them to actual patients and their relatives? Or should genetic risks be presented first as inherent in the way nature deals with the perpetuation of the species? Posing the two approaches in this manner highlights the tension in medical education between training and education; tension that exists because the exigency of medicine tends to give primacy to training. Education embodies the principles and ideas of disease and health, whereas training represents exercises in problem solving and dealing with disease whether or not its mysteries can be solved. There is a widespread and profound uneasiness in the minds of medical educators, a sense that something has gone wrong even when so much about medical practice has gone right. Two aspects of this concern stand out; a) technology seems to have gotten itself interposed between the doctor and the patient so that students are perhaps less likely to be brought up in an atmosphere of deep humane concern for the feelings and fears of sick people than once they were; and b) the volume of knowledge to be absorbed is enormous and sometimes results in a lack of coherence for the student. The challenge for medical education is how to proceed from the facts of reductionism, with its arcane language of acronyms, letters and numbers, through the many levels of integration by which these fundamental units coalesce into the systems that inform the unity of the organism. Genetics courses given to first and second year students should serve two missions; to teach the rudiments of genetics to students with variable background, and at the same time to make clear that there is such a thing as medical genetics as a separate entity. There is always the risk that the latter will be over emphasized, or that the students will draw the conclusion for themselves that the main application of genetics is to a medical specialty called medical genetics. Since most entering students today have been exposed to the basics of mendelism, a shift could be effected from emphasis on the facts of genetics to the ideas of genetics, to the preparation of the mind to receive at a later time the quite different ideas of practice. This would necessarily move emphasis from mendelizing disorders to multifactorial conditions which comprise most of medicine. SOURCE: Childs, 1992. |
percent of responding physicians reported that no courses in genetics had been available during their medical training; even among those in practice less than six years, only half reported that such courses had been available to them. This survey also identified substantial differences among specialties in the perceived frequency of genetic defects; pediatricians and obstetricians believed genetic diseases to be more common than did family practitioners. The conclusion of the 1975
report was that "the medical profession as represented by the three subspecialties studied is not as a whole ready to accept the importance of genetic disease and of screening for it at the present time." However, the readiness of physicians could be increased "if physicians had greater knowledge of genetics, deeper appreciation of the impact of untreated genetic disease on families, and more direct experience with genetic disease" (NAS, 1975, p. 164).
Another survey in 1977 examined the level and nature of genetics instruction in U.S. medical schools (Childs et al., 1981) (see Box 6-6). Nearly two-thirds of the schools reported required courses in genetics, but most courses were entirely lectures, with discussion groups in only one-third and laboratory hours in less than one-quarter. Most teaching of genetics was offered in schools with a department or division of genetics, and most teaching faculty members were pediatricians. Very little teaching of genetics occurred in departments of preventive, community, or family medicine.
In 1985, Riccardi and Schmickel (1988) conducted a less elaborate survey of all medical schools in the United States, Canada, and Puerto Rico. Even seven years later, there had been little improvement in the number of course hours devoted to genetics. They found that nearly half (47 percent) of medical schools had nonexistent or inadequate human genetics teaching, with only 21 percent having good or excellent teaching in this area.
In response to the results of this 1985 survey and the 1984 report Physicians for the Twenty-First Century (AAMC, 1984), a multidisciplinary task force of members of the American Society of Human Genetics (Graham et al., 1989) was convened to examine the challenges of teaching human genetics in medical schools. The group agreed on a set of core content areas that should be understood by medical students by the time of graduation (see Box 6-6).
Finally, the Association of American Medical Colleges (AAMC) maintains a data base on courses offered at all U.S. medical schools. A review of curricula for 1992 showed that more medical schools are including some human and medical genetics in their curriculum in required courses (AAMC, 1991).
Some progress has been made in increasing physicians' knowledge of genetics and genetic testing and their ability to take good family histories, particularly in specialties that involve more genetic tests (e.g., pediatrics and obstetrics) and among more recent medical graduates. Nevertheless, not enough progress has been made—certainly not enough to prepare physicians-in-training for the increasing requirements for genetic testing, education, and counseling projected for the future. More research is needed on ways for medical education to begin to incorporate a genetic point of view throughout its curriculum, but especially in the critical clinical years, and on the knowledge of genetics and skills needed for genetics education and genetic counseling among all of these professional groups so that proper reforms can be implemented.
The Council on Resident Education in Obstetrics and Gynecology (CREOG) has also published standards for minimal skills that an obstetrics-gynecology res-
|
BOX 6-6 Genetics Instruction in Medical Schools 1977: A survey was conducted in 1977 (Childs et al., 1981) to determine the level and nature of genetics instruction in U.S. medical schools (100 responders). It found that
1985: Riccardi and Schmickel (1988) conducted a less elaborate survey of all medical schools in the United States, Canada, and Puerto Rico (119 out of 140 schools responding). A review of 79 course schedules, 41 syllabuses, and 40 sets of examination questions revealed the following:
1989: In response to the results of the 1985 survey, a multidisciplinary task force of members of the American Society of Human Genetics was convened to examine the challenges of teaching human genetics in medical schools (Graham et al., 1989). The group agreed that the following content areas should be understood by medical students before graduation:
|
|
1991: The Association of American Medical Colleges maintains a data base on courses offered at all U.S. medical schools. A review of curricula for 1992 shows that in terms of courses required, more medical schools are including human and medical genetics in their curriculum (AAMC, 1991). During 1991-1992, 79 of 126 medical schools (63 percent) required a human or medical genetics course. Of these, 70 percent required the course in the first year and 30 percent required it in the second year. The number of hours spent in the course ranged from 4 to 74, with the average course entailing 30 hours. |
ident should possess in dealing with prenatal genetic counseling. The resident was expected to be able to discuss the principles of and give examples for Mendelian inheritance, define multifactorial inheritance, understand and be able to discuss chromosomal anomalies, obtain a genetic history and construct a family history chart, know the indications for amniocentesis, discuss the principles of cell biochemical analysis for the detection of inborn errors of metabolism, discuss maternal serum alpha-fetoprotein (MSAFP) screening, and discuss the value of population screening for genetic disorders as well as the prerequisites for such a screening program (CREOG, 1984). In June 1992, CREOG issued its revised core curriculum, including requirements for genetic counseling, which includes much more detailed and extensive knowledge and skills in genetics required for obstetrics-gynecology residents (CREOG, 1992) (see Box 6-7).
The Association of Professors of Gynecology and Obstetrics (APGO) proposed similar standards in its 1992 Guide to Basic Science Prerequisites to a Clerkship in Obstetrics and Gynecology to assist basic science departments of medical schools with developing essential curricular materials (APGO, 1992). The recommended proficiencies essential to genetics are listed in Table 6-6.
Continuing Medical Education
Continuing education credits for physicians wanting to learn more about medical genetics have been in existence for some time. Many medical centers offer courses, as do specialized centers such as the Jackson Memorial Laboratories in Bar Harbor, Maine, and the annual March of Dimes birth defects meetings. Such courses could also help to attract human and molecular geneticists into clinical genetics. The effects of such training on changes in clinical genetics practice are not known.
Physicians can also voluntarily engage in self-assessment activities in specific disciplines. For example, the American College of Physicians (ACP) offers a Medical Knowledge Self-Assessment Program in Genetic and Molecular Medi-
|
BOX 6-7 CREOG Standards for Training of Ob-Gyn Residents
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 6-6 Basic Science Prerequisites in Genetics for a Clerkship in Obstetrics and Gynecology
|
I. Define the following basic genetic terms and mechanisms |
|
A. The structure of the nucleic acids |
|
B. Cell division |
|
C. Chromosomal abnormalities |
|
D. Molecular genetics |
|
E. Linkage, crossing over, and chromosome mapping |
|
F. Diagnostic techniques |
|
II. Describe the manifestations and mechanisms for detection of abnormal inheritance |
|
A. Single-gene inheritance |
|
B. Sex chromosomal anomalies (risk, characteristics) |
|
C. Autosomal anomalies |
|
III. Describe laboratory and special studies |
|
A. Tissue culture techniques |
|
B. Karyotype construction and interpretation |
|
C. DNA testing |
|
SOURCE: Association of Professors of Gynecology and Obstetrics, 1992. |
cine, among other subjects. This home-study program can also provide continuing medical education credits for participating physicians.
In addition to continuing medical education, informal learning occurs in contacts between genetics specialists and referring physicians. This is particularly true for physicians frequently exposed to genetic conditions, such as obstetricians and pediatricians, who tend to score higher on tests of genetic knowledge than do physicians with little clinical exposure (Hofman et al., 1993). The clinical ''need to know" may be the best motivator for genetics education. As more genetic tests are available, the need to know will inevitably grow.
Professional Statements, Guidance, and Proficiencies
Many medical and scientific organizations issue standards and guidelines for practice regarding drugs and procedures, including genetic tests and procedures that might serve an educational purpose. For some physicians, these guidelines and directives might be influential in their decision making regarding incorporation of new tests and procedures. Concerns about protection from professional liability may play an important role in the development of professional standards and guidelines, and may influence the speed with which genetic tests are adopted into clinical practice.
The American Society of Human Genetics and the American College of Obstetricians and Gynecologists (ACOG) have issued several statements in the past
decade recommending particular courses of action (or inaction) regarding genetic tests and procedures. For example, the ASHG (1992) has issued statements regarding cystic fibrosis carrier screening, and MSAFP screening (see also Garver, 1989; Caskey et al., 1990).
In 1986, ACOG suggested that obstetricians discuss the availability of the MSAFP test with their pregnant patients, but recommended against routine use of MSAFP screening in all pregnancies; ACOG issued a medical liability "alert" recommending that obstetricians offer every pregnant woman screening for elevated MSAFP to detect increased risk of certain genetic disorders (ACOG, 1985, 1986, 1991).
In 1987, ACOG issued guidelines for the use of antenatal diagnosis of genetic disorders. This technical bulletin (ACOG, 1987) serves as an educational aid to obstetrician-gynecologists and contains information on taking a genetic history, indications for prenatal genetic studies, technical considerations for amniocentesis, chorionic villus sampling, fetal visualization, fetoscopic tissue sampling, and molecular genetics in prenatal genetics. The standards require that physicians obtain the following information in evaluating genetic risk: (1) advanced parental age; (2) previous offspring with a chromosomal aberration; (3) chromosomal abnormality in either parent, particularly a translocation; (4) family history of a sexlinked condition; (5) family history of an inborn errors of metabolism; (6) family history of a neural tube defect; (7) family history of hemoglobinopathies; and/or (8) ancestry, indicating risk for Tay-Sachs disease, beta-thalassemia, or alphathalassemia.
OTHER HEALTH PROFESSIONALS
Other professional groups could (and sometimes do) provide essential genetic counseling and support services. Other health professionals now playing or likely to play a critical role in providing genetic services include nurses, social workers, and public health workers. Key issues concern the training of other professionals to provide essential genetic services and how their services will be overseen.
Nurses in Genetics
There are nearly 2 million registered professional nurses in the United States, many involved in maternal and child health nursing, providing a unique potential to contribute to the effective delivery of genetic services. In some settings—such as community, occupational, or school health—nurses may be the only link with the health care system (Forsman, 1988; Jones, 1988). Thus, nurses can assist in the identification, education, counseling, and follow-up of patients (Fibison, 1983; Jones, 1988; Thomson, 1992). Yet although nurses can be a valuable part of genetics services, to date they are a largely untapped resource (Forsman, 1988).
For nearly 30 years, it has been suggested that human genetics be included in the nursing curriculum (Brantl and Esslinger, 1962). A 1980 workshop on education in genetics for nurses and social workers developed academic criteria; didactic course work and clinical experience requirements for training as a master'slevel clinical nurse specialist in genetics were developed in graduate programs in schools of nursing, were developed (Forsman and Bishop, 1981) (see Box 6-8). Candidates must meet the same academic admission requirements defined by the National League for Nursing accredited maternal child nursing program with a clinical nurse specialist option.
Despite this attention to the importance of genetics in nursing education, a 1984 survey of nursing instructors revealed that most schools dedicated less than 10 hours to genetics instruction, with little clinical experience (Forsman, 1988). Only four of the 200 universities in the United States that offer graduate degrees in nursing have established programs providing a master's-level genetics major (Forsman, 1988). A small number of nurses, particularly those in maternal and child health nursing, have been certified in genetic counseling by the ABMG (Forsman, 1988; OTA, 1992b). More than 100 nurses are employed in genetics, according to the International Society of Nurses in Genetics (ISONG).
Social Workers in Genetics
Social workers can play an important role in genetics services delivery, particularly in underserved communities. Social workers also have contact with many clients with genetic conditions in medical settings, such as facilities for high-risk infants, or pediatric, neurological, endocrine, and other specialty clinics. Other social work settings that might require some genetics expertise include family planning services, adoption and child welfare agencies, child guidance clinics, public health programs, agencies serving the mentally retarded and their families, and departments that serve developmentally and physically disabled clients (Schild and Black, 1984).
A 1980 conference (see Box 6-8) recommended that all social workers be provided a working knowledge of genetic diseases, their etiology, and consequences. In 1986, the Council on Social Work Education published a guide to genetic content for graduate social work education (Rauch, 1986). Nevertheless, only 9 of almost 100 accredited social work graduate programs in the United States offer special courses on genetic topics (Friedman and Blitzer, 1991). A course in genetics is offered to nurses and social workers in Washington, D.C. (see Box 6-8).
Public Health
Similarly, public education in genetics requires increased commitment at the public health level (also see discussion in Chapter 5). This requires educating
|
BOX 6-8 Education in Genetics for Nurses and Social Workers NURSING In 1980, a workshop "Education in Genetics for Nurses and Social Workers" was sponsored by the Office of Maternal and Child Health to explore training and recommend service needs for nurses and social workers in genetics. The conference recommended that all nurses be provided a working knowledge of genetic diseases, their etiology, and consequences, and developed academic criteria, a list of didactic course work in human and medical genetics, and clinical experience required for training as an MSN clinical nurse specialist in genetics (Forsman and Bishop, 1981). Training occurs in graduate programs in schools of nursing. Candidates must meet the same academic admission requirements defined by the National League for Nursing accredited maternal child nursing program with a clinical nurse specialist option. Didactic course work in both human and clinical medical genetics is required, including:
Although genetics is generally a part of the nursing school curriculum, but that offer graduate degrees in nursing, only four have established programs providing a master's-level major in genetics (Forsman, 1988). again, programs vary (Forsman, 1988). Of the 200 universities in the United States A course in genetics is offered to nurses and social workers in Washington, D.C., and is funded by the Genetics Services Branch, Division of Maternal and Child Health, Office of Research and Training, Health Resources and Services Administration, in the Department of Health and Human Services. The course introduces the fundamental concepts of human genetics and provides for learning the skills of screening and identification, referral, case management, and health education. A small number of nurses, particularly those in maternal and child health nursing, have specialized in genetics in order to sit for the genetic counseling examination given by the ABMG (Forsman, 1988; OTA, 1992b; Thomson, 1992). According to ISONG, more than 100 nurses are employed in genetics; 48 percent of its members have a master's degree and 11 percent doctoral degrees; 43 percent have achieved certification in genetics or a nursing specialty; 37 percent have had 10 or more years of experience in genetics; and 85 percent have had 10 or more years of experience in nursing. SOCIAL WORK This conference also recommended a working knowledge of genetics for all social workers, including an understanding of the etiology and implications of genetic disorders. The Council on Social Work Education published a guide to genetics education for social workers (Rauch, 1986) |
public health professionals about pertinent issues related to medical genetics and changing the attitudes and staffing patterns of key state agencies (Cunningham and Kizer, 1990; Davis, 1990). Yet a survey of curricula at member schools of the Association of Schools of Public Health indicated a decrease in the number of schools offering human genetics as a major area of study (Friedman and Blitzer, 1991). Few schools of public health offer genetics as part of their curriculum, and in none is it required (Schull and Hanis, 1990). The limited attention given to genetics in schools of public health is troubling. Genetic testing will become an increasingly important aspect of health and social policy as more tests are developed. Some genetic testing programs—especially newborn screening—directly involve public health agencies.
FINDINGS AND RECOMMENDATIONS
As the availability of genetic tests increases and testing becomes more commonplace, it is likely that genetic testing will follow the path of other technological innovations in health care, and will be ordered and interpreted by primary care physicians, including pediatricians, obstetrician-gynecologists, internists, and general practitioners. There seems to be no prospect, in the foreseeable future, of having enough highly specialized genetics personnel to handle all genetic testing, including essential genetics education and genetic counseling. Although the number of individuals being certified in clinical genetics has been increasing, it is not clear that this is occurring quickly enough to ensure adequate and appropriate levels of support for greatly expanded genetic testing, education, and counseling in the future. Indeed, much of the manpower increase has gone to research, rather than to clinical genetics.
As a result, the role of the genetic specialist as the primary provider of genetic services is likely to change in the future. Genetic specialists will be called upon to play an important and expanded role in three areas: (1) continuing to provide genetic testing and counseling for disorders with the most complex interpretations and implications; (2) training and continuing education for other professionals in genetics and genetic counseling (Waples et al., 1988); and (3) seeing people in need of specialized genetic services referred by many more professionals (see discussions in Chapters 2 and 4). As genetic tests penetrate increasingly into the repertoire of primary care providers, it is likely that genetic specialists—be they master's-level genetic counselors, Ph.D.'s, or physicians—will continue to receive referrals from primary care providers of individuals with positive test results or psychosocial concerns that require more intensive follow-up. With training from genetic specialists, other health professionals, such as nurses, nurse practitioners, social workers, psychologists, and physicians, could be integrated into the existing genetics network.
Two issues should be addressed in terms of professional education in order for new genetic tests to be assimilated into medical practice in an appropriate
manner. First, the committee recommends efforts to train more genetics professionals with the skills needed for adequate and accurate education, diagnosis, and counseling to (1) expand the pool of teachers in human genetics; and (2) meet the expected increase in referrals from primary care practitioners. This means increasing support (and funding, as appropriate) for the following:
-
Training of master's-level genetic counselors. The committee recommends that, at least, the current number of genetic counseling graduates be maintained annually. This area will require close consideration over the next few years as genetic testing, education, and counseling services expand. In addition, the committee recommends funds for the training of master's-level genetic counselors, including stipends to attract minority students to this field. Because of the rapid development of new knowledge in genetic testing, the committee also recommends the development of formal continuing education programs for genetic counselors.
-
Development and evaluation of programs for single-disorder educators-counselors. Programs should use innovative methods and personnel including the use of single-disorder educator-counselors drawn from the populations they are intended to serve. Evaluation should focus particularly on the types of methods or personnel necessary to provide specific kinds of genetic education and counseling, settings in which these services should be provided, and the training and support required if these innovative approaches are to be successful.
The committee recommends that genetic counseling and education be considered essential components of the standard of care, not only for specialized genetics personnel, but also for physicians, particularly primary care practitioners, who offer and interpret genetic tests. The committee endorses the core content areas identified by the ASHG task force, particularly the inclusion of ethics. However, the committee strongly recommends the addition of attention to patient education and counseling in genetics to this list of core subjects that should be understood by medical students by the time of graduation, and that national board examinations and specialty examinations for primary care specialists include more questions on genetics. The committee also commends CREOG and APGO for establishing standards and basic proficiencies for genetic counseling, and it recommends that other specialties follow their example.
The committee strongly recommends the expansion of continuing medical education programs in clinical genetics, including those geared to the primary care practitioner, and the development of additional continuing medical education programs to improve the knowledge and skills of currently practicing physicians in genetic testing, education, and counseling. This should include physician fellowships and postdoctoral support for training in clinical genetics, not only support for research training and research careers; postdoctoral education in human genetics in the form of intensive two-week courses or summer courses; and predoctoral graduate students in genetics and molecular bi-
ology to take appropriate human genetics classes intended to increase the understanding of medical genetics (such as those offered by medical school faculties).
To ensure that genetic tests are administered in an appropriate manner, the committee recommends
-
reform of the education of physicians and other health professionals who are not trained as geneticists to include increased attention to basic genetics;
-
in particular, training of medical students to deal with the sensitivities of genetics education and the need for nondirectiveness, especially in counseling about reproductive options and about disorders for which no treatment exists;
-
reform of medical education should begin to incorporate a genetic point of view throughout its curriculum to enable physicians to recognize that human variability exists in the pathogenesis of human disease;
-
training for genetics professionals and others offering or referring for genetic testing in the ethical, legal, and social issues surrounding genetic diagnosis, testing, and screening;
-
training for laboratory personnel in order to understand the complexities of genetic testing so as to adequately interpret tests with a knowledge of test limitations and a sensitivity to the social issues surrounding genetic testing;
-
more research on knowledge of genetics and skills needed for genetics education and genetic counseling among all of the professional groups discussed in this report so that proper reforms can be implemented, and
-
establishment of basic proficiencies in genetics for physicians, nurses, and social workers.
The committee also recommends the development of formal continuing education and training for genetic counselors and other professionals . Geneticists should also take a leadership role in genetics education for the public. Other health care professionals should also participate in programs intended to increase public awareness and education about genetics.
The committee believes that with proper training, the integration of other health professionals such as nurses, nurse practitioners, social workers, psychologists, and primary care physicians, into the existing genetics services network will supplement the time and skills of the traditional genetic counselor. Consequently, the committee strongly recommends expanded undergraduate and graduate training of nurses and social workers in the special requirements of genetics, genetics education, and genetic counseling. The committee recommends that public health professionals have training to ensure that they understand the underlying science of genetics and genetic testing, as well as the ethical, legal, and social issues outlined in this report.
The committee recommends the recruitment of more minorities for training programs in all aspects of clinical genetics. This will be especially important in providing culturally sensitive and appropriate genetic testing, education, and counseling services in the future when so-called minority groups will comprise a majority of the population of the United States (see Chapter 4).
The committee also recommends the development and evaluation of innovative on-line computer and interactive computer systems to disseminate the latest information on genetic disorders and on recommendations and guidelines for genetic testing. This is one way to improve the quality of genetic testing, education, and counseling services in the future (e.g., through the program of the National Library of Medicine (NLM) and the American College of Physicians for ACP members to have on-line access to the resources of the NLM). The committee believes that the NLM is an excellent mechanism for providing access to critical information on genetics and recommended criteria for genetic testing, genetic counseling, and follow-up care to members of ACP and other interested professional groups. The ELSI program should coordinate with professional genetics organizations and the NLM to develop such a genetics education and dissemination program for interested health professionals.
NOTE
REFERENCES
Acton, R., et al. 1989. Use of self-administered family history of disease instruments to predict individuals at risk for cardiovascular diseases, hypertension and diabetes. American Journal of Human Genetics 45(S):A275.
American College of Obstetricians and Gynecologists (ACOG). 1985. Professional Liability Implications of AFP Testing (Liability Alert). May. Washington, D.C.
American College of Obstetricians and Gynecologists (ACOG). 1986. Prenatal Detection of Neural Tube Defects. ACOG Technical Bulletin No. 99. December 1986 (replaced Technical Bulletin No. 67. October 1982). Washington, D.C.
American College of Obstetricians and Gynecologists (ACOG). 1987. Antenatal Diagnosis of Genetic Disorders. ACOG Technical Bulletin No. 108. September 1987 (replaced No. 34, January 1976). Washington, D.C.
American College of Obstetricians and Gynecologists (ACOG). 1991. Alpha-Fetoprotein. ACOG Technical Bulletin No. 154. April. Washington, D.C.
American Society of Human Genetics (ASHG). 1992. Statement of the American Society of Human Genetics on cystic fibrosis carrier screening. American Journal of Human Genetics S1:1443-1444.
Association of American Medical Colleges (AAMC). 1984. General professional education of the physician: Physicians for the twenty-first century. Journal of Medical Education 59:1-208.
Association of American Medical Colleges (AAMC). 1991. 1991-92 AAMC Curriculum Directory. Washington, D.C.
Association of Professors of Gynecology and Obstetrics (APGO). 1992. Guide to Basic Science Prerequisites for a Clerkship in Obstetrics and Gynecology. Washington, D.C.
Blitzer, M. 1992. Guide to North American Graduate and Postgraduate Training Programs in Human Genetics, 1992-93 (2nd Ed.). Bethesda, Md.: American Society of Human Genetics.
Brantl, V., and Esslinger, P. 1962. Genetic implications for the nursing curriculum. Nursing Forum (Spring 1):90-100.
Caskey, T., et al. 1990. The American Society of Human Genetics statement on cystic fibrosis screening. American Journal of Human Genetics 46:393.
Childs, B. 1992 (published in 1994). Genetics and medical education. In Fullarton, J. (ed.) Proceedings of the Committee on Assessing Genetic Risks. Washington, D.C.: National Academy Press.
Childs, B., et al. 1981. Human genetics teaching in U.S. medical schools. American Journal of Human Genetics 33:1-10.
Cole, J., et al. 1978. Genetic family history questionnaire. Journal of Medical Genetics 15:10-18.
Council on Resident Education in Obstetrics and Gynecology (CREOG). 1984. American College of Obstetrics and Gynecology. Washington, D.C.
Council on Resident Education in Obstetrics and Gynecology (CREOG). 1992. American College of Obstetrics and Gynecology. Washington, D.C.
Cunningham, G., and Kizer, K. 1990. Maternal serum alpha-fetoprotein screening activities of state health agencies: A survey. American Journal of Human Genetics 47:899-903.
Davidson, R., and Childs, B. 1987. Perspectives in the teaching of human genetics. In Harris, H., and Hirschhorn, K. (eds.) Advances in Human Genetics 16. New York: Plenum Press.
Davis, J. 1990. Invited editorial: State-sponsored maternal serum alpha-fetoprotein activities: Current issues in genetics and public health. American Journal of Human Genetics 47:896-898.
Fatemi, C., and Gasparini, R. 1990. 1990 salary statistics for cytogenetic technologists and supervisors throughout the United States. Applied Cytotechnology 16(4):85-90.
Fibison, W. 1983. The nursing role in the delivery of genetic services. Issues in Health Care of Women 4:1-15.
Finley, W., et al. 1987. Letter to the editor: Survey of medical genetics personnel. American Journal of Human Genetics 40(4): 374-377.
Firth, H., and Lindenbaum, R. 1992. U.K. clinicians' knowledge of and attitudes to the prenatal diagnosis of single gene disorders. Journal of Medical Genetics 29:20-23.
Forsman, I. 1988. Education of nurses in genetics. American Journal of Human Genetics 43:552-558.
Forsman, I., and Bishop, K. 1981. Education in Genetics: Nurses and Social Workers: Proceedings of a Workshop. HHS Pub. No. 81-51 120A. U.S. Department of Health and Human Services. Rockville, Md.
Friedman, J., and Blitzer, M. 1991. ASHG/NSGC activities related to education: Workshop on human genetics education. American Journal of Human Genetics 49:1127-1128.
Friedman, J., and Riccardi, V. (eds.). 1988. Guide to North American Graduate and Postgraduate Training Programs in Human Genetics (2nd Ed.). Rockville, Md.: American Society of Human Genetics.
Garver, K. 1989. Update on MSAFP policy statement from the American Society of Human Genetics. American Journal of Human Genetics 45:332-334.
Garver, K., and Lent, K. 1990. American Society of Human Genetics membership survey results, 1989. American Journal of Human Genetics 47:345-348.
Gasparini, R., et al. 1988. Undergraduate and postgraduate training programs for cytogenetic technologists. American Journal of Human Genetics 42: 200-203.
Gaupman, K., et al. 1991. ASHG/NSGC activities related to education: The doctoral degree in genetic counseling: Attitudes of genetic counselors. American Journal of Human Genetics 49:488-493.
Genetics Associates: Their, Training, Role and Function (Conference Proceedings). 1979. Williamsburg, Va.: U.S. Department of Health, Education, and Welfare.
Gettig, E. 1992 (published in 1994). Recommendations on minimum criteria for genetic counseling. In Fullarton, J. (ed.) Proceedings of the Committee on Assessing Genetic Risks. Washington, D.C.: National Academy Press.
Graham, J., et al. 1989. Report of the task force on teaching human genetics in North American medical schools. American Journal of Human Genetics 44:161-165.
Hofman, K., et al. 1993. Physicians' knowledge of genetics and genetic tests. Academic Medicine 68(8):625-631.
Holtzman, N. 1989. Proceed with Caution: Predicting Genetic Risks in the Recombinant DNA Era. Baltimore, Md.: The Johns Hopkins University Press.
Holtzman, N. 1991. The interpretation of laboratory results: The paradoxical effect of medical training. Journal of Clinical Ethics 2(4):1-2.
Hunt, S., et al. 1986. A comparison of positive family history definitions for defining risk of future disease. Journal of Chronic Disease 39:809-821.
Jones, S. 1988. Decision making in clinical genetics: Ethical implications for perinatal practice. Journal of Perinatal and Neonatal Nursing 1(3): 11-23.
Kapur, S., et al. 1983. Medical practice and genetics in the mid-Michigan area. Journal of Medical Education 58: 186-193.
Lemkus, S., et al. 1978. Genetic and congenital disorders: Knowledge and attitudes of the public, nurses, and medical practitioners in South Africa. South African Medical Journal 53:491-494.
Motulsky, A. 1983. Role of medical genetics in United States academic medicine. In Bowers, J., and King, E. (eds.) Academic Medicine: Present and Future. North Tarrytown, N.Y.: Rockefeller Archive Center.
Murray, J., and Toriello, H. (eds.). 1990. Guide to North American Graduate and Postgraduate Training Programs in Human Genetics (3rd Ed.). American Society of Human Genetics. Bethesda, Md.
Napier, J., et al. 1972. Limitations of morbidity and mortality data from family histories: A report from the Tecumseh community health study. American Journal of Public Health 62:30-35.
National Academy of Sciences. 1975. Genetic Screening: Programs, Principles, and Research. Washington, D.C.: National Academy of Sciences.
National Society of Genetic Counselors (NSGC). 1991. Code of Ethics. Chicago, Ill. Naylor, E. 1975. Genetic screening and genetic counseling: Knowledge, attitudes, and practices in two groups of family planning professionals. Social Biology 22:304-314.
Office of Technology Assessment (OTA). 1992a. U.S. Congress. Cystic Fibrosis and DNA Tests: Implications of Carrier Screening. OTA-BA-532. Washington, D.C.: U.S. Government Printing Office.
Office of Technology Assessment (OTA). 1992b. U.S. Congress. Genetic Counseling and Cystic Fibrosis Carrier Screening: Results of a Survey. OTA-BP-BA-97. Washington, D.C.: U.S. Government Printing Office.
Pew Health Professions Commission. 1991. Healthy America: Practitioners for 2005: An Agenda for Action for U.S. Health Professional Schools. Durham, N.C.
Rauch, F. 1986. Guide to Graduate Social Work Education in Genetics. New York: Council on Social Work Education.
Riccardi, V., and Schmickel, R. 1988. Human genetics as a component of medical school curricula: A report of the American Society of Human Genetics. American Journal of Human Genetics 42:639-643.
Riccardi, V., and Smith, A.C.M. (eds.). 1986. Guide to Human Genetics Training Programs in North America (1st Edition). Bethesda, Md.: American Society of Human Genetics.
Robert Wood Johnson Foundation. 1991. Environment for Learning: An Interim Report of the Robert Wood Johnson Foundation Commission on Medical Education: The Sciences of Medical Practice. Philadelphia, Pa.
Schild, S., and Black, R. 1984. Social Work and Genetics: A Guide for Practice. New York: Hawthorne Press.
Schull, W., and Hanis, C. 1990. Genetics and public health in the 1990s. Annual Review of Public Health 11:105-125.
Singer, E. 1991. Public attitudes toward genetic testing. In Population Research and Policy Review 10:235-255.
Thomson, E. 1992 (published in 1994). Issues in genetic counseling: The nursing perspective. In Fullarton, J. (ed.) Proceedings of the Committee on Assessing Genetic Risks. Washington, D.C.: National Academy Press.
Uhlmann, W.R. 1992. Professional status survey results. Perspectives in Genetic Counseling, Suppl. 14(2):7-10, Summer.
Walker, A., et al. 1990. Report of the 1989 Asilomar Meeting on education in genetic counseling. American Journal of Human Genetics 46:1223-1230.
Waples, C., et al. 1988. Resources for Genetic Disorders. Genetics Applications: A Health Perspective. Lawrence, Kans.: Learner Managed Designs.
Wilfond, B., and Fost, N. 1990. The cystic fibrosis gene: Medical and social implications for heterozygote detection. Journal of the American Medical Association 263:2777-2783.
Williams, R., et al. 1988. Health family trees: A tool for finding and helping young family members of coronary and cancer prone pedigrees in Texas and Utah. American Journal of Public Health 1878:1283-1286.
Zinberg, R., and Greendale, K. 1991. Do we have a shortage of genetic counselors in New York State? The Newsletter of the Genetics Network of the Empire State 3(3): 1.