Introduction
The flow of chemicals through the ocean system has a fundamental impact on global processes, including the Earth's energy balance. For example, the ocean holds approximately 60 times as much dissolved carbon dioxide species (TCO2) as are present in atmospheric carbon dioxide. Perturbations in the flow of carbon dioxide through the ocean-atmosphere system are closely linked to climate change during glacial periods (Lorius et al., 1990). Human perturbation of the carbon cycle, which has resulted in a 30% increase in atmospheric carbon dioxide since 1850, has the potential to produce even greater climate changes than were experienced over the last glacial cycle (Ramanathan, 1988).
Chemical distributions in the ocean are also strongly influenced by human activities on local scales, particularly in the coastal ocean. Nutrient enrichment in the New York Bight and Chesapeake Bay has led to anoxia in the water column and significant impacts on marine resources (Officer et al., 1984; Falkowski et al., 1980). New measurement technologies will be required to achieve an adequate understanding of the cycling of carbon and other biologically important chemicals in the coastal ocean. Coastal areas present special difficulties in understanding because of the complexity of coastal processes and their greater variability in both space and time.
In many cases, the impact of human disturbances cannot be predicted because a fundamental understanding of natural chemical cycles is lacking, due to insufficient information on the distributions of chemical components in the ocean. For example, we do not yet understand in adequate detail what processes control primary production (photosynthetic production of
biomass) in many areas of the ocean. Such knowledge is critical because primary production in the ocean acts as a "biological pump" to move carbon dioxide from the ocean surface layer, where it exchanges with the atmosphere, to the deep ocean. Future scenarios for greenhouse warming are impossible to evaluate until we understand how this "biological pump" operates. The oceanic absorption of fossil fuel CO2 is difficult to observe and understand due to the large and variable background of the natural carbon cycle (Tans et al., 1990), which is itself subject to climatic perturbation.
Primary production in the ocean is controlled by major nutrients, such as nitrate and phosphate, but also by certain trace metals. Dissolved iron was hypothesized (over 50 years ago) to be a key nutrient limiting primary production rates in the sea. However, credible data for the concentration of dissolved iron in seawater have only become available in the last 8 years. Iron is present in surface seawater at concentrations less than 0.5 nanomole per kilogram. These low concentrations of dissolved iron suggest that it is, in fact, a nutrient that can limit primary production in the ocean (Martin et al., 1989). The role of iron in limiting productivity of the ocean can be resolved only when measurements of dissolved iron at concentrations below 1 nanomole per kilogram become routine. There is evidence that other trace metals could also control phytoplankton growth.
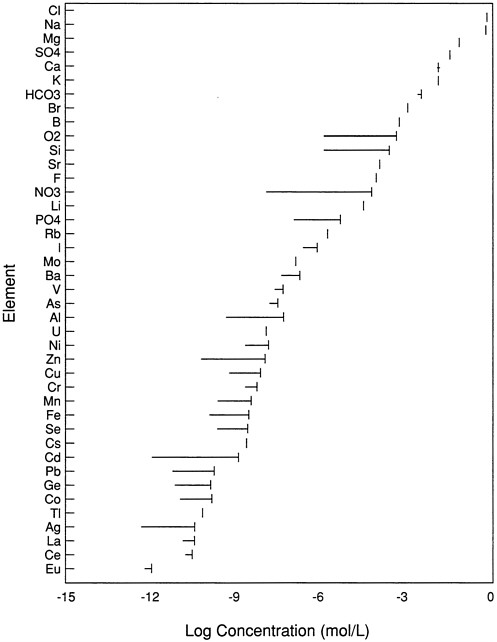
Chemical analyses of seawater present unique difficulties. These problems are not limited to iron. In fact, with the exception of the major ions (Na+, K+, Mg2+, Ca2+, Cl-, SO42-, HCO3-) found in seawater, most other elements are present at trace concentrations (Figure 1). The biologically limiting compounds are almost exclusively present at concentrations less than a few micromoles per kilogram seawater, posing significant challenges to their measurement. Their concentrations must be determined in the presence of more than 1 million-fold excess of major ions. Trace metal measurements typically require the sample to be concentrated greatly before analysis by even the most sensitive techniques. Contamination from ships and sample containers is a ubiquitous problem, and extreme care must be taken to protect sample integrity. Determination of the chemical speciation of elements at these concentrations is even more difficult. For example, copper, which is present in surface seawater at concentrations of less than 1 nanomole per kilogram, is bound by an unidentified ligand with a high specificity for the metal (Coale and Bruland, 1988). This ligand is present at concentrations on the order of 1 nanomole per kilogram and appears to regulate the free copper activity.
The interest in coordinated international studies of the global environment has increased since it has become apparent that humankind is affecting the composition of the atmosphere and ocean on a global scale. One proposal that has been accepted by the International Oceanographic Commission is for a global ocean observing system (GOOS). GOOS, as presently
envisioned, consists of components for climate monitoring, assessment, and prediction; monitoring and assessment of marine living resources; coastal zone management and development; assessment and prediction of the health of the ocean; and marine meteorological and oceanographic services. The successful implementation of several of these components will depend on development and deployment of chemical sensors that are not yet available for long-term use in the ocean.
Collection of data over long periods of time often is required to resolve chemical changes in the ocean because there is a great deal of natural variability in chemical concentrations over time. The natural cycles of biologically active chemicals in the surface layer of the ocean are characterized by large seasonal changes in concentration. Variability in concentration can also occur on a daily cycle because of the respiration, photosynthesis, and nutrient uptake of plants in response to light and dark. Interannual changes in chemical distributions also result from processes such as El Niño that occur only a few times each decade. These temporal variations of different frequencies are often superimposed on slow concentration changes due to human activity. For example, lead concentrations in the surface waters of the Atlantic Ocean appear to have decreased about twofold from 1979 to 1984 to values near 100 picomoles per kilogram, as a result of the phasing out of leaded gasoline in North America (Boyle et al., 1986). The decrease in lead concentrations is obscured, however, by the large variability in the Atlantic Ocean produced by movement of the Gulf Stream and the water masses it sheds. This trend could only be identified by comparing concurrent measurements of the isotope 210Pb, which is a natural radioactive decay product of uranium, and stable lead (primarily 207Pb), which has an anthropogenic source.
World ocean circulation can be pictured as flow along the "oceanic conveyor" from its source in the North Atlantic through the ocean depths to the North Pacific via the Antarctic Circumpolar Current, where it returns along the surface, through the Indian Ocean to its source in the North Atlantic (Broecker, 1991). About 1000 to 1500 years are required for sea-water to complete this cycle. During this time, chemicals that are released from sinking organic matter through decomposition by microorganisms accumulate in the water, while those that are scavenged onto sinking particles are depleted. Concentrations must often be measured with high precision to detect the chemical changes that occur with time in the ocean. The concentration of total carbon dioxide, TCO2 = [CO2] + [H2CO3] + [HCO3-] + [CO32-], increases by about 20% from an initial value near 2 millimolar as seawater follows this path from the surface to the deep ocean and back to the surface, due primarily to respiration. If TCO2 and oxygen are measured with a precision and accuracy of better than 0.1% and 1%, respectively, it is possible to detect the change in TCO2 due to the increase of carbon dioxide
in the atmosphere since the Industrial Revolution. Such measurements have been used to reconstruct the past composition of the atmosphere (Brewer, 1978). Measurements of tracer chemicals such as chlorofluorocarbons, tritium, and 14C, largely introduced to the ocean by human activities, are used to determine the rates at which reactions occur as seawater completes the global circulation pattern.
There are large gaps in our fundamental knowledge of organic compounds dissolved in seawater, and the concentrations of dissolved organic carbon and nitrogen are still matters of debate. The molecular identity of little of the pool of organic carbon dissolved in the ocean is known with any certainty. There is almost no knowledge of the temporal variability of individual organic compounds, their spatial distributions, or their role in sequestering atmospheric carbon or other chemical species. This information is essential if the role of the oceans in global processes is to be understood. Many organic compounds are directly measurable at natural concentrations found in the ocean by using techniques such as derivitization of amino acids and by measuring reducing sugars by pulsed amperometric detection. Identification of other specific components of the oceanic carbon pool will be possible with improved measurement capabilities, leading to a better understanding of chemical and biological processes in the ocean.
NEED FOR NEW APPLICATIONS OF ANALYTICAL CHEMISTRY TO OCEANOGRAPHY
The objective of this report is to review the potential application of methods of analytical chemistry to study the ocean carbon cycle and processes—physical, chemical, and biological—that affect carbon fluxes. The committee sought to identify state-of-the-art measurement technologies that could produce significant progress in our understanding of oceanic bio-geochemical cycles. These promising analytical technologies are reviewed briefly. The report focuses on analytical methods that could operate continuously on mid-ocean moorings, drifting buoys, or commercial ships making regular ocean passages. Continuous measurements are critical to understand the spatial and temporal variability of processes that are fast and discontinuous, such as gas exchange, primary production, and bacterial uptake of dissolved material, as well as the processes that control the variability. Laboratory-based measurements of discrete samples are also considered for cases in which chemical measurement technologies have not advanced to the stage of continuous in situ or shipboard use.
The status of measurement technologies for ocean chemicals is being reviewed for several reasons. First, presently employed technologies for chemical measurements are inadequate for global system science. Many of the ocean's physical and biological parameters, such as surface temperature,
light transmission, up-and down-welling irradiance, and chlorophyll fluorescence, can be measured frequently from satellites or open-ocean moorings over long periods of time. These measurements have led to rapid advances in our understanding of the oceanographic processes that control these variables. However, remote sensing of chemical parameters is limited. In most cases, chemists can study dissolved chemical distributions only when research vessels are present on station to collect water samples. For instance, at a typical sampling site, one might determine the vertical distribution of an analyte in the water column by acquiring a dozen or more samples in Niskin bottles from the surface to depths of kilometers. Even using a sophisticated water collecting device called a rosette, collecting the samples at one site can take hours. In other cases, one can immerse a pump at a preset depth to bring water to the surface for analysis, but this method cannot be used for sampling water from great depths. Dependence on ship-based sampling leads to severe undersampling in both time and space.
Undersampling is a particularly strong limitation on large-scale studies of globally significant processes, such as the air-sea transfer of carbon dioxide and the factors regulating oceanic primary production. The seasonal variability in parameters such as the partial pressure of carbon dioxide (pCO2) in surface seawater is often an order of magnitude greater than the changes that might be expected due to a doubling of atmospheric carbon dioxide. Solving the undersampling problem is essential to our ability to predict the impact of increasing carbon dioxide on future climate.
Second, chemical measurements over global scales represent highly challenging problems that can be addressed effectively by analytical chemists and oceanographers. Rapid advances in microelectronics, detection techniques, and data collection and analysis have solved many of the problems associated with chemical analysis and have opened the frontier on new methods of chemical sensing. New types of selective chemical reactions are available through advances in biotechnology. The materials sciences have provided new technologies for the production of microelectrodes, micropumps, fiber optics, and novel polymers, all key components for sensing chemicals dissolved in the ocean. Developments in these areas are being prompted by the growing requirements of clinical chemistry, environmental monitoring, and industrial process control.
The need for a new generation of chemical observing systems for the ocean is widely recognized. Global science initiatives including the Joint Global Ocean Flux Study (GOFS, 1984) and the Ridge Inter-Disciplinary Global Experiments (RIDGE, 1989) have identified developments in chemical sensing that are required to meet their scientific objectives. At least five recent program announcements from federal agencies—the National Science Foundation, the Office of Naval Research, the Department of Energy,
and the National Oceanic and Atmospheric Administration—have called for the development of new sensor technology.
New chemical technologies are needed for measurements on a local scale as well. Each year $133,000,000 is spent for ocean monitoring programs required to protect the marine environment (NRC, 1989), so that improvement in techniques could have a substantial impact on the cost of measurements that contribute to this monitoring cost. Monitoring results must be incorporated into the environmental management process in a timely manner. However, this is not possible if there are long time delays between sampling and availability of data or there are insufficient numbers of measurements to interpret the processes that are occurring. There are not enough ships, scientists, technicians, or funds to support the ship-based measurements that would be required to collect these data. Consequently, alternative methods must be sought.
The shortcomings of existing methods will be surmounted only by using new approaches. This report discusses and identifies new and emerging measurement technologies from the field of analytical chemistry that could be applied to chemical and biological oceanography, ranging from those that must be confined to land-based laboratories to methods that can be used in shipboard laboratories, ship-tethered profilers, and long-term in situ sensors. The ultimate implementation of new measurement technologies will be in the form of in situ measurements. The ocean is constantly changing; consequently, measurements of rates are essential to understanding ocean processes. Techniques that provide continuous monitoring capability are to be preferred to those that require discrete samples.
The committee attempted to project the availability of new technologies up to 15 years into the future. This longer-range view is consistent with the 10-year development cycle needed for introducing new instrumentation into the ocean measurement field as described by Wunsch (1989). The slow pace of introducing new techniques is due primarily to the extreme requirements of the ocean system. There are other nonenvironmental limiting factors, such as the lack of testing facilities, that could be changed to speed instrument development. Of course, it is likely that, within 15 years, unforeseen scientific developments and technical breakthroughs will make new measurement opportunities available.
A final objective of this report is to highlight the exciting opportunities afforded by this interdisciplinary endeavor and to encourage greater cooperation between analytical chemists and ocean scientists. Several past efforts have been aimed at bringing together practitioners of the diverging fields of marine chemistry and analytical chemistry (Goldberg, 1975, 1988). Graedel et al. (1988) noted that developments are needed in two types of analytical capabilities—real-time data acquisition and instruments for labo-
ratory use—for studying the air-sea interface. They listed a number of analytes for which surface ocean time series measurements would be useful: pCO2, pH, O2, argon, a fluorescence proxy for chlorophyll, dimethyl sulfide, methyl halides, and isotopes of major nutrient elements. Martin et al. (1988) described a number of analytical techniques that could be applied to studies of the seawater-sediment interface and chemical processes at the ocean bottom. These included electrochemical and fluorescent techniques, combined with new specific chemistries. Potential new methods of concentration and fractionation of dissolved organic material were also described. The development of chemical measurement technologies for ocean sciences requires cooperative efforts by scientists from these two fields and communication between them. The ocean chemistry community is a small one, whereas the analytical chemistry community represents one of the largest subdisciplines of chemistry. Organization of departments, professional societies, and scientific journals have the tendency to impede interactions among fields as different as ocean science and analytical chemistry. Thus, scientific culture slows progress at the interface between these fields. Progress will continue to be slow until appropriate mechanisms and incentives can be instituted to encourage interactions between scientists in these two disciplines, and to accommodate interdisciplinary work. If analytical chemists are made aware of the exciting opportunities available in the field of ocean science, they are more likely to choose an ocean measurement problem and adapt their analytical techniques to it. By fostering interdisciplinary research efforts, the development of ocean measurement technology should progress more rapidly.