1
A Review of Current Knowledge
In 1992, the Committee on Low-Frequency Sound and Marine Mammals was established under the auspices of the Ocean Studies Board of the National Research Council. The committee was charged with (1) reviewing the current state of knowledge and ongoing research on the effects of low-frequency [1 to 1,000 hertz (Hz)] sound on marine mammals, (2) advising the sponsors about the effects of low-frequency sound on marine mammals, and (3) considering the tradeoffs between the benefits of underwater sound as a research tool and the possibility of its having harmful effects to marine mammals.
The report is organized as follows. Chapter 1 discusses current knowledge with respect to changing levels of sound in the ocean and some possible effects on marine mammals. It then describes the state of knowledge regarding components that must be determined to evaluate a potential source of noise interference affecting marine mammals. The committee then discusses the gaps in knowledge, including those on auditory sensitivity and behavioral responsiveness of marine mammals, that would require research before predictions can be made of the effects of low-frequency sound on marine mammals. Chapter 1 closes with a discussion of the difficulties related to a sound criterion that is being applied to underwater noise.
Citing an urgent need for additional research, the committee takes up the question of the regulation of research on marine mammals and related topics under the Marine Mammal Protection Act of 1972.
Chapter 2 reviews the permitting process as it now stands, and presents the committee's recommendations for change in the regulatory structure in order to facilitate needed research.
Chapter 3 outlines the research that would provide some of the missing information needed for a better understanding of the effects of low-frequency sound on marine mammals and their prey. The chapter is divided into sections on Behavior of Marine Mammals in the Wild, Structure and Function of the Auditory System, Effects of Low-frequency Sounds on the Food Chain, and Development and Application of Measurement Techniques.
Appendix B of this report provides an introduction to marine mammals, including tables listing the species by their scientific classification. The tables indicate the species for which audiometric information is available.
Changes in Ocean Sounds and Potential Effects on Marine Mammals
Noise is widely acknowledged to be an environmental pollutant for humans and many other terrestrial species, and it is no doubt a pollutant for marine animals as well. All animals, whether they are terrestrial or aquatic, have evolved to live in particular ecological niches. The physical characteristics of these niches have established various constraints on the perceptual systems and communication abilities of these organisms. Consequently, the hearing abilities of marine mammals1 undoubtedly evolved to deal with the ambient noise levels in the aquatic environment prior to the industrial age.
Terrestrial mammals depend on sound to analyze and interact with their environment and to communicate among themselves. Marine mammals, including cetaceans (various whales, porpoises, and dolphins) and pinnipeds (seals, sea lions, and walruses), are probably even more dependent on sound and hearing than terrestrial mammals are. For example, it is known that dolphins use the echoes from the high-frequency sounds they produce in order to navigate and to locate prey. They use moderate-frequency sounds to maintain contact among individuals. Baleen whales are known to produce low-frequency sounds that propagate over long distances. Although the
exact functions of most of these sounds are not yet known, they clearly play an important role in the life of the species.
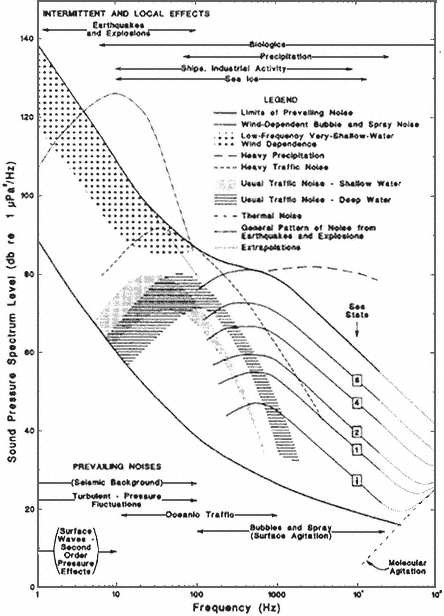
The natural sounds of the ocean—produced by wind, waves, precipitation, natural seismic events, and creatures that live in the sea—create a noisy environment (Wenz, 1962; Tavolga, 1967). There are many sources of sound, and sound travels efficiently in water. In recent times, the background sound level has increased considerably because humans have developed a number of highly intense sound sources (Ross, 1976; Urick, 1983, 1986). Underwater sound produced by numerous ocean-going vessels is one of the more intense human-made intrusions (see Figure 1). Supertankers produce spectrum levels that equal or exceed 200 decibels (dB) [reference (re) 1 micropascal (μPa) at 1 meter (m)—water standard2] in the very low (<10 Hz) region of the spectrum (Cybulski, 1977; Leggat et al., 1981). Explosive and nonexplosive seismic sounds used in geological exploration for oil and gas, as well as sounds from dredging, drilling, and marine construction, also contribute to the sonic burden of the sea (Richardson et al., 1991). Surface vessels and submarines employ active sonar, an apparatus that uses sonic or ultrasonic waves to locate submerged objects and in the process introduce brief, high-intensity pulses that sometimes propagate great distances. Acoustic oceanographers use intense sounds, especially low-frequency sounds, to study the physical properties of the ocean (Spindel and Worcester, 1990; Baggeroer and Munk, 1992; Worcester et al., 1993). Such human-made sounds combine with the natural sounds of the ocean, and elevate the ambient noise level, mainly in the frequency region below 1,000 Hz (Figure 1).
Major changes in human-made noise, such as the change resulting from the advent of supertankers, can be assumed to affect the ability of marine mammals to communicate and to receive information about their environment. Such noise may interfere with or mask the sounds used and produced by these animals and thereby interfere with their natural behavior.
Higher levels of human-made sounds can cause obvious disruptions: they may frighten, annoy, or distract the animals and lead to physiological and behavioral disturbances. They can cause reactions that might include disruption of marine mammals' normal activities
|
2 |
The reader should be aware that different reference pressures are used to specify sound levels in air and in water (see Appendix A for additional information). When decibel numbers are given to specify sound levels in the text of this report, the reference number, together with the designation ''water standard'' or "air standard," is also given. |
and, in some cases, short-or long-term displacement from areas important for feeding or reproduction. They may also disturb the species such as fishes, squids, and crustaceans upon which the marine mammals prey.
At still higher levels, human-made noise may cause temporary or permanent hearing impairment in marine mammals. Such impairment would have the potential to diminish the individual's chances for survival.
For some years, there has been growing general concern about the potential effects of human-made underwater sounds on marine mammal communication (e.g., Payne and Webb, 1971; Reeves, 1977; Myrberg, 1978; Acoustical Society of America, 1981). During the 1980s, a number of more specific studies were undertaken to determine the reactions of several cetacean species and, to a lesser degree, pinnipeds to noise from oil industry operations, shipping, tourist activities, and rocket launches (reviewed in Richardson et al., 1991). Recently, concern has also arisen about the potential effects of strong low-frequency sounds used by acoustic oceanographers, underwater acousticians, and operational naval activities (Mulroy, 1991; Simmonds and Lopez-Jurado, 1991).
Evaluating Noise Interference
For a sound signal to be detected by a marine mammal or its prey, the signal must contain enough energy to exceed the ambient (background) level of sounds near the animal at frequencies near the frequency of the signal. This effective signal-to-noise ratio is determined by four components: (1) the source level of the signal; (2) the transmission loss in the sea—the decrease in intensity as sound travels to the animal; (3) the level of other sounds near the animal (ambient noise level); and (4) the auditory characteristics of the receiver, the hearing organ. Any potential source of noise interference must be evaluated by determining these four components of the source-path-receiver model. Specific information is often known or can be obtained more easily about the first component, the source level of the signal, in comparison to the other three components, which are more technically complex. The other three components are discussed briefly below.
Transmission Loss of Sound in the Ocean
The second component, the transmission loss of sound in the ocean, is a complicated matter but one that is reasonably well understood,
both in theory and in practice (Urick, 1982, 1983). Studying the transmission of sound in the sea has been a major research area of modern oceanography and of the world's naval forces. The transmission loss problem is complicated by the presence of gradients of temperature, pressure, and salinity (gradients that cause variation in the sound speed of the medium) and by the variable effects of the sea bottom and surface on sound transmission. These phenomena result in refractions and reflections of sound waves. Despite these many factors, reasonable approximations exist that permit prediction of sound levels in many underwater situations.
Ambient Noise Level of the Sea
The third component of the source-path-receiver model, the ambient noise level of the sea, has also been extensively studied (see Figure 1 and Urick, 1986). Ambient noise levels have been of considerable interest both to naval forces and to acoustic oceanographers. These levels and the details of their spectra and variation over time establish the conditions for detecting extraneous signals either by a physical receiver or by any biological organism. Extensive data and models concerning both acoustic propagation and the ambient noise level can be used to help evaluate the potential impact on marine mammals of sound from any human-made source.
Acoustic Characteristics of Marine Mammal Hearing Organs
The component of the source-path-receiver model about which the least is known is the fourth component—the acoustic characteristics of the receiver, the hearing organ. At present, very little is known about the detection and interpretation of low-frequency sounds by the animals that appear to use such signals. To understand the significance of this lack of knowledge, it is useful to think of a sensory system—in this case, hearing—as a window that allows the brain to gather information about a particular aspect of the physical environment. That is, the sense of hearing in any animal is sensitive to a limited range of sound frequencies, and outside this range the animal is unaffected by any stimulus, no matter how intense. With respect to the auditory sensitivities of marine mammals, the range of frequencies by which these animals are affected appears to vary among, as well as within , the three different orders of Mammalia to which they belong (see Appendix B for the classification of marine mammals in the three orders). As described in more detail below, the order Cetacea includes dolphins and the smaller toothed whales, whose
auditory sensitivities are greatest at very high frequencies. Available data for all toothed whales that have been measured for auditory sensitivity are reasonably consistent. However, for another group of cetaceans whose hearing is undoubtedly tuned to low frequencies—the baleen whales—there are virtually no quantitative data on auditory sensitivity.
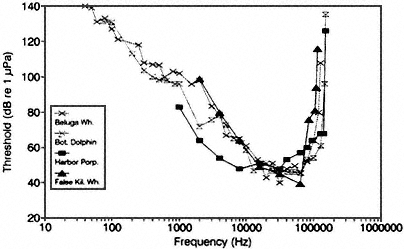
Audiograms have been obtained for several dolphins and smaller toothed whales. Sensitivity of hearing is best at about 20,000 Hz, with about 80 dB less sensitivity at 100 Hz for two species—the beluga whale and bottlenose dolphin—for which data are available (see Figure 2A). For reasons described below, the auditory sensitivities of these smaller cetaceans are probably quite different from those of the baleen or larger toothed whales, whose auditory sensitivities have never been assessed quantitatively. Also, even for the relatively well-studied toothed whales, audiograms have been obtained for only a few species, and in most cases for only one or two individuals per species. However, different individuals of a single species can have quite different hearing capabilities (see, e.g., Hall and Johnson, 1971; vs. Bain et al., 1993; Ridgway and Carder, 1993).
It is thought that baleen whales hear very low-frequency sounds because their vocalizations contain considerable low-frequency energy. Some of the dominant calls of the blue and fin whales are in the frequency range between 12 and 25 Hz, and their source level is about 177 dB re 1 μPa—water standard. The anatomical nature of baleen whale hearing organs also suggests that they are sensitive to low-frequency sounds (Fleischer, 1976; Ketten, 1991). Certainly there are obvious anatomical differences between the hearing organs of the baleen and the toothed whales (Ketten, 1991).
The strongest quantitative evidence of the auditory sensitivity of baleen whales comes from field observations in which human-made sounds in the low-frequency regions caused two species of baleen whales to alter their migration paths or to avoid certain areas (Malme et al., 1983, 1984, 1988; Richardson et al., 1986, 1990; Richardson and Malme, 1993; Dahlheim and Ljungblad, 1990). Recordings from a variety of sources were used in these studies, including continuous sounds such as those from oil drilling and production platforms, as well as impulsive sounds from air guns. The broadband levels of low-frequency sounds causing avoidance by about 50 percent of the gray and bowhead whales in the area of the observations occurred when the received levels were around 115 to 120 dB re 1 μa—water standard for the continuous sounds and about 160 to 170 dB for the pulsed sounds. These received levels are overall levels, and the sources produced energy over a few octaves. The peaks of the broadband
A. Audiograms of Selected Odontocetes
B. Underwater Audiograms of Pinnipeds
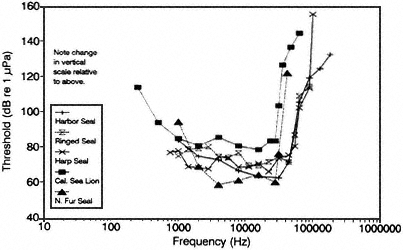
Figure 2 Underwater audiograms of selected toothed whales (A) and pinnipeds (B). Source: Adapted from Richardson et al. (1991): (A) beluga (White et al., 1978; Awbrey et al., 1988; and Johnson et al., 1989); bottlenose dolphin (Johnson, 1968); harbor porpoise (Anderson, 1970); false killer whale (Thomas et al., 1988); (B) harbor seal (Møhl, 1968); ringed seal (Terhune and Ronald, 1975); harp seal (Terhune and Ronald, 1972); California sea lion (Schusterman et al., 1972); northern fur seal (Moore and Schusterman, 1987), courtesy of LGL Ltd.
spectra were in the general region of 100 Hz. The hearing sensitivities of baleen whales are not known over any specific frequency region but are clearly different from those of toothed whales. In the low-frequency region, it is uncertain that the toothed whale could even detect continuous sounds with received levels near 115 dB re 1 μPa—water standard. The implications of this paucity of audiometric information include profound uncertainty about the interfering effect of any potential sound source on baleen whales. For example, depending on this animal's auditory sensitivity, the effect of loud low-frequency sound could conceivably range between potential hearing damage and gradual deafness for the entire species—and eventual extinction—or practically no discernible impact. Such uncertainty prevents the committee from reaching any but the most general conclusions in this regard.
Among pinnipeds, which include, for example, seals, sea lions, and walruses, underwater audiograms are also available for several species, but there are very few data on underwater hearing sensitivity at frequencies below 1,000 Hz (see Figure 2B). Based on the slopes of the audiograms at the lowest frequencies measured, it appears that some pinnipeds may have better hearing sensitivity at low frequencies than do the two odontocetes, the beluga whale and bottle-nose dolphin, for which low-frequency audiograms are available. There are no published data for elephant seals and few for walruses (Kastelein et al., in press). Elephant seals are known to dive deeply and are of special interest because they may dive deep enough to reach the sound channel 3 and thus be exposed to higher levels of sound (in Chapter 3, see the subsection on Replication of Data in the section on Structure and Function of the Auditory System). There are essentially no data about the behavioral reactions of pinnipeds as a function of the received level of human-made underwater sounds (Richardson et al., 1991). More is known about the low-frequency hearing abilities of some pinnipeds in air and about behavioral responses of certain pinnipeds to airborne sounds. The in-air information, however, cannot be applied directly to the underwater situation.
Although there has been considerable work on the anatomy of the auditory apparatus of manatees (e.g., Fischer, 1988; Ketten et al., 1992) and several relevant studies are under way, there is very little information about the hearing abilities of marine mammals in the
order Sirenia, which includes manatees and dugongs. The evoked-potential data that are available for the manatee do not extend below 1,000 Hz (Bullock et al., 1980, 1982; Popov and Supin, 1990). Recent behavioral tests indicate some detection of sound as low as 15 Hz and as high as 4 to 6 kilohertz (kHz), with the greatest sensitivity in the 6–20 kHz region (Gerstein et al., 1993). Sirenian vocalizations are known to be in the 1–8 Khz range (Nishiwaki and Marsh, 1985; Hartman, 1979). There is little information on the behavioral responsiveness of sirenians in relation to underwater sound level, although work on this topic has also begun (Weigle et al., 1993).
The committee is not aware of any specific data on the hearing sensitivity of sea otters or polar bears, both of which are considered marine mammals. There is little information on the reactions of these mammals to underwater sound (see, however, Riedman, 1984).
The scarcity of information about the auditory sensitivity of marine mammal hearing is coupled with equal uncertainty on the structural and behavioral levels. With respect to structure, for example, how does sound actually get to the inner ears of these animals? Are the pathways different for different marine mammal groups, and are they different in a given species for different sound frequencies? In terms of behavior, if human-made sounds are interfering with marine mammals, how do the animals respond to the sounds, and do they habituate when the sound is repeated? Much greater knowledge is also needed about the role of acoustical information and hearing in the behavior of such mammals in their natural (undisturbed) habitats. Only with such baseline information can it be determined that a response caused by an intrusive sound is normal.
The Need for Additional Research
There have been some observational or experimental studies and numerous anecdotal reports about the responses of marine mammals to sound. Extensive reviews and evaluations of this literature can be found in Richardson et al. (1991) and Reeves (1992). Missing from most of these studies, however, is information on the level of the sound exposure: typically neither the source level nor the received level was measured. Even in cases where the level of the sound at the source was known, there were often uncertainties in calculating the received level near the animal. Direct measurements or reliable estimates of received levels of sound are difficult or impossible to obtain during many observational studies and usually were not obtained even when it might have been possible. There is need for planned experiments in which the received level of the sound and the behav-
ior of the animal can be studied together. Such investigations would probably be logistically complex and would require scientific permits. Chapter 2 discusses the permit issue, and Chapter 3 describes the kinds of studies needed.
As discussed in the preceding section, the lack of knowledge about the auditory sensitivity and behavioral responsiveness of marine mammals makes it impossible to predict how low-frequency sound may affect them. The lack of such knowledge, which must serve as the basis for identifying and determining the effects of particular underwater sounds on particular species, precludes the committee from being able to obtain a reasonable estimate of costs on which to base advice about the potential trade-offs between "the benefits of underwater sound as a research tool" and "the possibility of harmful effects to marine mammals." Research on marine mammals and their major prey is urgently needed to permit an adequate response to this question. The most immediate research needs are outlined in Chapter 3 of this report.
It is the belief of this committee that an accelerated program of scientific studies of the acoustic effects of low-frequency sound on marine mammals and their prey (including the studies described in Chapter 3) should be undertaken. These studies should be designed to provide the information needed to direct policies that will provide long-term protection to the species.
Origins and Drawbacks of the "120-dB Criterion"
The phrase "120-dB criterion" refers to a level of sound that has been identified informally as a level above which acoustic effects on marine mammals might occur. In reviewing plans for activities that produce underwater noise, it appears to the committee that the National Marine Fisheries Service of the U.S. Department of Commerce considers that marine mammals exposed to broadband received levels above 120 dB (re 1 μPa—water standard) might be affected by the sounds. 4 Almost all sound sources used in acoustical oceanography or for operational Navy purposes have a source level far above 120 dB (re 1 μPa at 1 m—water standard), as do some motorized boats. Depending on the source level, frequency, and local propagation condi-
tions, the received level of sound from these three sources will exceed 120 dB within a distance that could range from a few meters or tens of meters for a weak source up to many kilometers for a stronger one. Thus, if the 120-dB criterion were to be applied consistently, almost any source of human-made underwater noise—including every powerboat—might be subject to regulatory scrutiny.
The 120-dB criterion arises primarily from two series of field studies. One series involved gray whales migrating along the coast of California and summering (spending the summer season) in the Bering Sea (Malme et al., 1983, 1984, 1988). The other was a series of studies of migrating and summering bowhead whales in the Beaufort Sea (Richardson et al., 1985, 1986, 1990; Ljungblad et al., 1988; Richardson and Malme, 1993). Unlike most other field studies on marine mammals, these two series of studies provided estimates of the sound exposure level in the vicinity of the animals while their behavior was being observed. Both series demonstrated that a variety of broadband continuous sound stimuli with spectra peaking in the frequency region of 100 to 300 Hz caused a detectable change in the behavior of some animals. The received level of continuous sound that caused a reaction in about half the animals was about 120 dB (re 1 μPa—water standard). There was considerable variation, however, with some animals reacting at lower levels and some not reacting at considerably higher levels.
The 120-dB figure has been applied, at times, to other types of sound and to other species of marine mammals without regard to the frequency spectrum or temporal pattern of the sound or to differences in the auditory sensitivity of the different groups of marine mammals. These variables are undoubtedly important in determining whether the 120-dB figure is appropriate for any given situation. For example, the temporal pattern of exposure was found to be very important for both the gray whale and the bowhead whale. The average pulse pressure level of a series of brief impulses had to be 30 to 50 dB more intense (150 to 170 dB re 1 μPa—water standard) to produce the same change in the animal's behavior as did a steady-state or continuous sound at 120 dB (Malme et al., 1984; Richardson et al., 1986; Ljungblad et al., 1988). In contrast, bowhead whales sometimes react to increasing noise levels from an approaching boat when the broadband level is well below 120 dB (Miles et al., 1987:225ff; Richardson and Malme, 1993). Thus, the actual threshold of reaction can range from well below to well above 120 dB, depending on circumstances.
As is true of most field observations, many different interpretations of these results can be offered. For example, according to the
studies mentioned, the change in behavior of the migrating gray whales was minor and brief, involving a slight deflection in the migratory path. One can argue that the animals simply detected a potential obstruction and made a relatively mild deflection in their course to avoid the obstacle. Certainly the energy expended in their response was minimal. Energetic effects were obviously greater for migrating bowhead whales. They apparently avoided an icebreaker-supported drillship by 10 to 30 kilometers (km) (LGL and Greeneridge, 1987; Brewer et al., 1993). Additionally, in the case of the withdrawal of bowheads from feeding areas, the action was observed when a novel stimulus was introduced (Richardson et al., 1990; Richardson and Malme, 1993). Such withdrawal behavior might or might not quickly habituate if the sound were repeated, but that study was not able to obtain information about habituation. Furthermore, only two species of whales were involved, and the results of the same experiment would very likely be different for other species. Because of their apparent lack of sensitivity at these low frequencies, some toothed whales, for example, may not detect sound at the levels that affected the gray whales and bowhead whales (see Figure 2A).
That the 120-dB number is considered to be such an important regulatory criterion is testimony only to the paucity of our knowledge about marine mammals.
In trying to protect human beings from the harmful effects of noise, the United States has adopted no absolute standard. It is known at what levels sound elicits the response of ''highly annoyed'' on questionnaires, but there are no national standards on an upper limit to such exposure. Noise is recognized as a source of stress, and we know that physiological changes can occur when subjects are exposed to certain noise levels. There is little consensus as to either the level of noise exposure that produces either harmful extra-auditory effects in humans or the dose-response relation over the long term. The one national U.S. standard with respect to human exposure to noise is the 90-dBA level (re 20 μPa—air standard) adopted by the U.S. Department of Labor's Occupational Safety and Health Administration (OSHA). It establishes that in the workplace if the exposure exceeds 90 DBA (air standard), then some kind of hearing protection program must be initiated. The 90-DBA level is about 100 dB above the level at which such a sound might first be detectable. If we assume the same dynamic range (100 dB) for whales as for humans and that 120 dB represents a detection level, then the upper limit for marine mammals might be set at 220 dB re 1 μPa (water standard).
In humans, the upper limit in the OSHA standard was determined largely on the basis of extensive scientific experiments involv-
ing induced temporary hearing loss. Thus, for humans, temporary threshold shifts provided the basic data for the adoption of the primary noise standard in the United States. The limits were subsequently confirmed in epidemiological studies of permanent hearing loss. No data on temporary threshold shifts (that is, the occurrence of temporary hearing loss) in marine mammals have been published to date.
Human annoyance to human made noise is widely variable, depending on the individual, the situation, and the characteristics of the noise. People are frequently annoyed by noise at levels well below those that introduce temporary or permanent hearing loss. The significance of human annoyance, however real, has proven difficult to evaluate. The biological significance of disturbance reactions by marine mammals will probably be even harder to determine.
References
Acoustical Society of America. 1981. San Diego workshop on the interaction between man-made noise and vibration and arctic marine wildlife. Rep. from Acoust. Soc. Am. for Alaska Eskimo Whaling Comm., Barrow, AK. 84 pp.
Andersen, S. 1970. Auditory sensitivity of the harbour porpoise Phocoena phocoena. Invest. Cetacea 2:255–259.
Awbrey, F.T., J.A. Thomas, and R.A. Kastelein. 1988. Low-frequency underwater hearing sensitivity in belugas, Delphinapterus leucas. J. Acoust. Soc. Am. 84(6):2273–2275.
Baggeroer, A., and W. Munk. 1992. The Heard Island Feasibility Test. Phys. Today 45(9):22–30.
Bain, D.E., B. Kriete, and M.E. Dahlheim. 1993. Hearing abilities of killer whales (Orcinus orca). J. Acoust. Soc. Am. 94(3, Part 2):1829.
Brewer, K.D., M.L. Gallagher, P.R. Regos, P.E. Isert, and J.D. Hall. 1993. ARCO Alaska Inc. Kuvlum #1 exploration prospect/ Site specific monitoring program final report. Rep. from Coastal and Offshore Pacific Corp., Walnut Creek, CA, for ARCO Alaska Inc. and the Nat. Mar. Fish. Serv., Anchorage AK. 80 pp.
Bullock, T.H., D.P. Domning, and R.C. Best. 1980. Evoked brain potentials demonstrate hearing in a manatee (Trichechus inunguis). J. Mammal. 61(1):130–133.
Bullock, T.H., T.J. O'Shea, and M.C. McClune. 1982. Auditory evoked potentials in the West Indian manatee (Sirenia: Trichechus manatus ). J. Comp. Physiol. 148A(4):547–554.
Cybulski, J. 1977. Probable origin of measured supertanker radiated noise spectra. In: Oceans 77 Conference Record, Inst. Electrical and Electronic Eng., New York, NY. pp. 15C-1 to 15C-8.
Dahlheim, M.E., and D.K. Ljungblad. 1990. Preliminary hearing study on gray whales (Eschrichtius robustus) in the field. In: J.A. Thomas and R.A. Kastelein (eds.), Sensory Abilities of Cetaceans/Laboratory and Field Evidence. Plenum, New York. pp. 335–346.
Fischer, M.S. 1988. Zur Anatomie des Gehörorganes der Seekuh (Trichechus manatus L.), (Mammalia: Sirenia). Z. Söugetierk. 53:365–379.
Fleischer, G. 1976. Hearing in extinct cetaceans as determined by cochlear structure. J. Paleontol. 50(1):133–152.
Gerstein, E.R., L.A. Gerstein, S.E. Forsythe, and J.E. Blue. 1993. Underwater audiogram of a West Indian manatee (Trichechus manatus). Abstr. 10th Bien. Conf. Biol. Mar. Mamm., Galveston, TX, Nov. 1993:53, 130 pp.
Hall, J.D., and C.S. Johnson. 1971. Auditory thresholds of a killer whale Orcinus orca Linnaeus. J. Acoust. Soc. Am. 51:515–517.
Hartman, D.S. 1979. Ecology and behavior of the manatee (Trichechus manatus). In: Florida Spec. Publ. Am. Soc. Mammal. 5, 153 pp.
Johnson, C.S. 1968. Relation between absolute hearing threshold and duration-of-tone pulses in the bottlenosed porpoise. J. Acoust. Soc. Am. 43(4):757–763.
Johnson, S.R., J.J. Burns, C.I. Malme, and R.A. Davis. 1989. Synthesis of information on the effects of noise and disturbance on major haulout concentrations of Bering Sea pinnipeds. OCS Study MMS 88–0092. Rep. from LGL Alaska Res. Assoc. Inc., Anchorage, AK, for U.S. Minerals Manage Serv., Anchorage, AK. 267 pp. NTIS PB89-191373.
Kastelein, R.A., C.L. van Ligtenberg, I. Gjertz, and W.C. Verboom. In press. Free field hearing tests on wild Atlantic Walruses (Odobenus rosmarus) in air. Aquat. Mammals 19(3).
Ketten, D.R. 1991. The marine mammal ear: specializations for aquatic audition and echolocation . In: D.B. Webster, R.R. Fay, and A.N. Popper (eds.), Evolutionary Biology of Hearing. Springer-Verlag, Berlin. pp. 717–750.
Ketten, D.R., D.K. Odell, and D.P. Domning. 1992. Structure, function, and adaptation of the manatee ear. In: J.A. Thomas, R.A. Kastelein, and A.Y. Supin (eds.), Marine Mammal Sensory Systems. Plenum, New York. pp. 77–95.
Leggat, L.J., H.M. Merklinger, and J.L. Kennedy. 1981. LNG carrier underwater noise study for Baffin Bay. In: N.M. Peterson (ed.), The Question of Sound from Ice-breaker Operations: The Proceedings of a Workshop. Arctic Pilot Proj., Petro-Canada, Calgary, Alb. pp. 115–155.
LGL and Greeneridge. 1987. Responses of bowhead whales to an offshore drilling operation in the Alaskan Beaufort Sea, autumn 1986. Rep. from LGL Ltd., King City Ont., and Greeneridge Sciences, Inc., Santa Barbara, CA, for Shell Western E & P. Inc., Anchorage, AK. 371 pp.
Ljungblad, D.K., B. Würsig, S.L. Swartz, and J.M. Keene. 1988. Observations on the behavioral responses of bowhead whales (Balaena mysticetus) to active geophysical vessels in the Alaskan Beaufort Sea. Arctic 41(3):183–194.
Malme, C.I., P.R. Miles, C.W. Clark, P. Tyack, and J.E. Bird. 1983. Investigations of the potential effects of underwater noise from petroleum industry activities on migrating gray whale behavior. BBN Rep. 5366. Rep. from Bolt Beranek & Newman Inc., Cambridge, MA, for U.S. Minerals Manage. Serv., Anchorage, AK. Var. pag. NTIS PB86-174174.
Malme, C.I., P.R. Miles, C.W. Clark, P. Tyack, and J.E. Bird. 1984. Investigations of the potential effects of underwater noise from petroleum industry activities on migrating gray whale behavior/Phase II: January 1984 migration. BBN Rep. 5586. Rep. from Bolt Beranek & Newman Inc., Cambridge, MA, for U.S. Minerals Manage. Serv., Anchorage, AK. NTIS PB86-218377.
Malme, C.I., B. Würsig, J.E. Bird, and P. Tyack. 1988. Observations of feeding gray whale responses to controlled industrial noise exposure. In: W.M. Sackinger, M.O. Jeffries, J.L. Imm, and S.D. Treacy (eds.), Port and Ocean Engineering Under Arctic Conditions, vol. II. Geophysical Inst., Univ. of Alaska, Fairbanks, AK. pp. 55–73.
Miles, P.R., C.I. Malme, and W.J. Richardson. 1987. Prediction of drilling site-specific interaction of industrial acoustic stimuli and endangered whales in the Alaskan Beaufort Sea. BBN Rep. 6509; OCS Study MMS 87–0084. Rep. from BBN Laboratories Inc., Cambridge, MA, and LGL Ltd., King City, Ont., for U.S. Minerals Manage. Serv., Anchorage, AK. 341 pp. NTIS PB88-158498.
Møhl, B. 1968. Auditory sensitivity of the common seal in air and water. J. Aud. Res. 8:27–38.
Moore, P.W.B., and R.J. Schusterman. 1987. Audiometric assessment of northern fur seals, Callorhinus ursinus. Mar. Mamm. Sci. 3(1):31–53.
Mulroy, M.J. 1991. Munk's experiment. Science 253:118–119.
Myrberg, A.A., Jr. 1978. Ocean noise and the behavior of marine animals: Relationships and implications. In: J.L. Fletcher and R.-G. Busnel (eds.), Effects of Noise on Wildlife. Academic Press, New York. pp. 169–208.
Nishiwaki, M., and H. Marsh. 1985. Dugong. In: S.H. Ridgway and R.J. Harrison (eds.), Handbook of Marine Mammals. Vol. III, The Sirenians and Baleen Whales. Academic Press , London. pp. 1–31.
Payne, R., and D. Webb. 1971. Orientation by means of long range acoustic signaling in baleen whales. Ann. N.Y. Acad. Sci. 188:110–141.
Popov, V., and A. Supin. 1990. Electrophysiological studies of hearing in some cetaceans and a manatee. In: J.A. Thomas and R.A. Kastelein (eds.), Sensory Abilities of Cetaceans/Laboratory and Field Evidence. Plenum, New York. pp. 405–415.
Reeves, R.R. 1977. The problem of gray whale (Eschrichtius robustus ) harassment: At the breeding lagoons and during migration. U.S. Mar. Mamm. Comm. Rep. MMC-76/06. NTIS PB-272506. 60 pp.
Reeves, R.R. 1992. Whale responses to anthropogenic sounds: a literature review. Sci. & Res. Ser. 47. N.Z. Dep. Conserv., Wellington, New Zealand. 47 pp.
Richardson, W.J., and C.I. Malme. 1993. Man-made noise and behavioral responses. In: J.J. Burns, J.J. Montague, and C.J. Cowles (eds.), The Bowhead Whale. Spec. Publ. 2, Soc. Mar. Mamm., Lawrence, KS. pp. 631–700.
Richardson, W.J., M.A. Fraker, B. Würsig, and R.S. Wells. 1985. Behaviour of bowhead whales Balaena mysticetus summering in the Beaufort Sea: reactions to industrial activities. Biol. Conserv. 32(3):195–230.
Richardson, W.J., B. Würsig, and C.R. Greene, Jr. 1986. Reactions of bowhead whales, Balaena mysticetus, to seismic exploration in the Canadian Beaufort Sea. J. Acoust. Soc. Am. 79(4):1117–1128.
Richardson, W.J., B. Würsig, and C.R. Greene, Jr. 1990. Reactions of bowhead whales, Balaena mysticetus, to drilling and dredging noise in the Canadian Beaufort Sea. Mar. Environ. Res. 29(2):135–160.
Richardson, W.J., C.R. Greene, Jr., C.I. Malme, and D.H. Thomson. 1991. Effects of noise on marine mammals. OCS Study MMS 90–0093; LGL Rep. TA834-1. Rep. from LGL Ecol. Res. Assoc. Inc., Bryan, TX, for U.S. Minerals Manage. Serv., Atlantic OCS Reg., Hemdon, VA. NTIS PB91-168914. 462 pp.
Ridgway, S.H., and D.A. Carder. 1993. High-frequency hearing loss in old (25+ years old) male dolphins. J. Acoust. Soc. Am. 94(3, Part 2):1830.
Riedman, M.L. 1984. Effects of sounds associated with petroleum industry activities on the behavior of sea otters in California. Appendix D . In: Malme et al. (1984). NTIS PB86-218377. Var. pag.
Ross, D. 1976. Mechanics of Underwater Noise. Pergamon, New York. 375 pp.
Schusterman, R.J., R.F. Balliet, and J. Nixon. 1972. Underwater audiogram of the California sea lion by the conditioned vocalization technique. J. Exp. Anal. Behav. 17(3):339–350.
Simmonds, M.P., and L.F. Lopez-Jurado. 1991. Whales and the military. Nature 351:448.
Spindel, R.C., and P.F. Worcester. 1990. Ocean acoustic tomography. Sci. Am. 263(4):94–99.
Tavolga, W.N. 1967. Noisy chorus of the sea. Natural History 76:20–27.
Terhune, J.M., and K. Ronald. 1972. The harp seal, Pagophilus groenlandicus (Erxleben, 1777). III. The underwater audiogram. Can. J. Zool. 50:565–569.
Terhune, J.M., and K. Ronald. 1975. Underwater hearing sensitivity of two ringed seals (Pusa hispida. Can. J. Zool. 53:227–231.
Thomas, J., N. Chun, W. Au, and K. Pugh. 1988. Underwater audiogram of a false killer whale (Pseudorca crassidens). J. Acoust. Soc. Am. 84(3):936–940.
Urick, R.J. 1982. Sound Propagation in the Sea. Peninsula Publishing, Los Altos, CA. Var. pag.
Urick, R.J. 1983. Principles of Underwater Sound, 3rd ed. McGraw-Hill, New York. 423 pp.
Urick, R.J. 1986. Ambient Noise in the Sea. Peninsula Publishing, Los Altos, CA. Var. pag.
Weigle, B.L., I.E. Beeler-Wright and J.A. Huff. 1993. Responses of manatees to an approaching boat: a pilot study. Abstr. 10th Bien. Conf. Biol. Mar. Mamm., Galveston, TX, Nov. 1993:111. 130 pp.
Wenz, G.M. 1962. Acoustic ambient noise in the ocean: Spectra and sources. J. Acoust. Soc. Am. 34(12):1936–1956.
White, M.J., Jr., J. Norris, D. Ljungblad, K. Baron, and G. di Sciara. 1978. Auditory thresholds of two Beluga whales (Delphinapterus leucas ). Hubbs/Sea World Res. Inst. Tech. Rep. 78–109 for Naval Ocean Systems Center , San Diego, CA. 35 pp.
Worcester, P.F., J.F. Lynch, W.M.L. Morawitz, R. Pawlowicz, P.J. Sutton, B.D. Cornuelle, O.M. Johannessen, W.H. Munk, W.B. Owens, R. Shuchman, and R.C. Spindel. 1993. Evolution of the large-scale temperature field in the Greenland Sea during 1988–1989 from tomographic measurements. Geophys. Res. Lett. 20(20):2211–2214 (22 Oct. 1993).