9
Conclusions
THE RECONSTRUCTION IN space and time of doses off-site from the release of radioactive material from DOE-managed nuclear facilities involves several steps (refer to Figure 9–1):
-
An analysis of the source term is used to estimate the magnitude of the releases of radionuclides and the periods over which they were released, including episodic releases from nonroutine events.
-
An analysis of the environmental pathways examines the transport of the released radionuclides to identify the concentrations in environmental media such as air, water, and food.
-
An assessment of radiation doses and risks brings together all of the data on releases, transport, and biologic factors to determine doses to persons and the resulting likelihood of disease in those who have been exposed.
-
An examination of epidemiologic considerations is used to evaluate the feasibility and scientific merit of an epidemiologic study.
-
An uncertainty and sensitivity analysis of the parameters and values is used to establish confidence intervals and to identify important factors in the overall analysis of the dose reconstruction.
The way the steps are implemented will vary from site to site, but to achieve maximal scientific rigor and some consistency across sites the committee makes the following recommendations:
-
In dose reconstruction studies, thorough consideration should
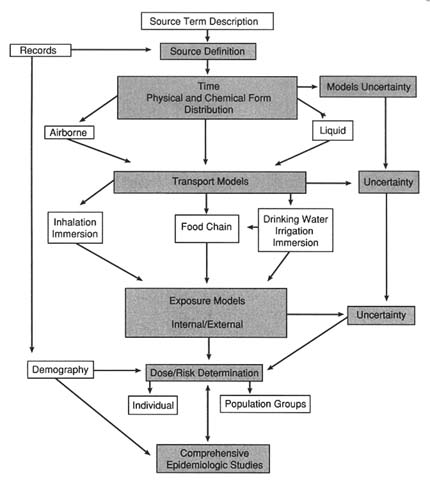
FIGURE 9–1 Dose reconstruction methodology.
-
be given to the collection of representative data, to an evaluation of their representativeness, to quality control, and to public involvement from the time the decision-making process begins, through the release of any results, and after the study concludes in any follow-up activities.
-
An advisory or steering committee should be established at the outset of a dose reconstruction study. This committee should consist of members of the public and knowledgeable scientists who are not associated with the investigators or their sponsors. The meetings of this committee should be open to the public. Along with its steering duties,
-
this committee should be charged with responsibility for establishing an interactive process to communicate the elements and conclusions of the study to the general public.
-
Dose reconstruction studies should begin with a scoping study—a preliminary analysis—to determine whether a comprehensive dose reconstruction study is needed or even possible, either for its own purposes or as the basis for a comprehensive epidemiologic study.
-
All dose reconstruction studies should be reviewed by groups of scientists and public health officials who are not directly involved in the study, either as participants or as advisors, and time and resources should be allocated for resolving discrepancies in the results.
-
There should be coordination between the dosimetric and epidemiologic efforts, which should begin at the outset of the dosimetric study and continue throughout.
-
Premature dissemination of the results should be avoided. Results should not be disclosed until the dose assessment study is complete, has undergone peer review, and has been published. Dissemination of data being considered during the study is appropriate and desirable.
-
A clear understanding of the public's concern should be gained before the study begins. A dose reconstruction study should not proceed until the design is such that it is likely that the results will address the public's concern.
-
Scoping studies should be the primary approach to initiating a source term evaluation. These should be followed, if appropriate, by more comprehensive studies. The scoping study of the source term should seek to generate the data needed to identify the environmental pathways of potential importance and to permit estimation of the concentrations of radionuclides to which the public might have been exposed.
-
To ensure maximum confidence in the source term analysis, proprietary or classified information should be made available for the analysis, or a mechanism should be developed to determine whether such data are essential to the accuracy and consistency of the analysis.
-
The source term should be derived chiefly from available original data in as many different ways as practical. The source term should be confirmed, wherever possible, by comparison with independent environmental monitoring data from another source of information.
-
To ensure completeness and accuracy of the estimated source term, all relevant data should be evaluated. Any gaps in the data should be analyzed carefully for their significance and filled by reconstruction from existing data if appropriate.
-
Episodic events should be documented as separate releases for
-
specific consideration in environmental transport and dose calculations. An event is considered episodic if it lasted for less than 10 days and if the release rate was at least 10 times the average monthly or annual rate.
-
The release quantities provided for use in a comprehensive study of the source term should be complete, unbiased estimates of all amounts and forms of relevant materials released to the environment.
-
Insofar as possible, measurements of environmental radiation or of radionuclides should be used in the environmental pathway analysis. For example, if a single contaminating event has taken place and if measurements have been made (such as external gamma exposure rate or deposition of one or more radionuclides or stable materials), it is often possible to begin the dose reconstruction without the need to model the transport of radionuclides up through this stage. Even if the contamination is chronic, it is often preferable to take suitable soil samples to measure the longer lived components of the contaminant materials (such as 137Cs or 129I) and to infer the deposition of shorter lived components (such as 131I) rather than to depend on atmospheric transport and deposition models, which are much less reliable than are direct measurements.
-
Even if there is an abundant base of monitoring data, mathematical models are usually needed to extrapolate information from situations where measurements have been made to situations where measurements are lacking. Every attempt should be made to validate the predictions of the models against relevant data sets. Caution should be exercised in the use of ''off-the-shelf" computer codes that may have been developed for other purposes such as regulatory analyses.
-
Environmental pathway analyses should include quantitative estimates of uncertainty to indicate the degree of confidence that can be placed in exposure estimates.
-
For accidents, there should be careful scrutiny of any counter-measures, such as removal of contaminated foodstuffs from commerce. Even if contaminated foodstuffs are not removed, people often voluntarily avoid contaminated foodstuffs and change their dietary habits. For routine releases, attention should be paid in the assessment of ingested radionuclides to the movement of foodstuffs into the region of interest because people do not necessarily consume local agricultural products.
-
All exposures from external sources, inhaled radionuclides, and ingested radionuclides should be considered; when certain pathways or other factors suggest that a particular source term or radionuclide will not contribute substantial dose, reports should explain why these sources or specific radionuclides were not considered in the final estimations.
-
Dose assessment should proceed at three levels: preliminary, comprehensive, and individual dose assessment.
-
Acceptable levels of uncertainty should be defined before a decision is made to carry out a detailed study.
-
A readily available set of intake-to-annual dose conversion factors for long-lived radionuclides should be established.
-
Doses should be expressed as effective doses in a preliminary dose assessment and objective criteria should be used to decide whether it is warranted to embark on a full-fledged study.
-
Dose estimates in a comprehensive dose assessment should be expressed as the annual organ absorbed doses from low-LET and high-LET radiations. Estimation of the effective dose may also be helpful.
-
In an individual dose assessment, the doses should be described separately as the annual organ-absorbed doses from low-LET and high-LET radiations.
-
For biologic markers to be useful in dose reconstruction, research will be necessary to
-
measure the stability of persistent biologic markers,
-
define "calibration curves" for low to moderate and chronic exposures,
-
determine the frequency of specific markers in unexposed populations,
-
define the sources of inter-individual variability for various markers, and
-
develop better definitions of marker responses after partial-body (external) or specific organ (internal emitter) exposure.
-
New assays should be developed to address the problems with individual variability in background, with identification of differences in individual susceptibility to radiation genotoxicity, and with the lack of sensitivity for quantifying low radiation exposures so that acute doses greater than 0.1 Gy (10 rad) can be reconstructed.
-
Biologic markers of effect should be used as epidemiologic end points. However, until clear connections are established between the marker and the disease, their use could be misleading rather than illuminating.
-
As the utility of biologic markers becomes established and accepted, the committee recommends that the CDC develop procedural strategies for conducting field studies for both specimen collection and laboratory analyses in the event of an acute release of activity.
-
Dosimetric and epidemiologic scoping studies around the sites of nuclear facilities or accidents should be considered, although the extent of such studies might vary from one site to another based on preliminary evidence about exposures, population sizes, and
-
public concern. These studies should be performed interactively and in parallel, because both are needed to inform a decision about further study of the site or for establishing priorities among sites.
-
Epidemiologic and dosimetric assessments should be closely coordinated. It is important to have epidemiologists involved from the outset of any dose reconstruction activity to ensure that the dosimetric information developed is appropriate for epidemiologic decisions and planning.
-
A full-fledged dose reconstruction and epidemiologic study should be proposed only if the scoping studies show that adverse health effects are likely to be statistically detectable, given the probable dose distribution and size of the exposed population.
-
Studies of health and points for which high-dose studies give no clear evidence of an excess should be avoided, because observing true excesses of these end points is biologically implausible. Such studies tend to waste resources and they are uninformative at best and misleading at worst.
-
A statistical power assessment based on a realistic set of assumptions about the dose distribution, population size, and radiation risk coefficients should be part of the scoping phase.
-
Rules and criteria should be defined for a scoping analysis to determine the desirability of a dose assessment and epidemiologic study. These should include demonstration of the feasibility and plausibility of the study, evidence of an adequate database, demonstration of an adequate range of doses, and appropriate numbers of subjects at the higher end of the dose range to meet the statistical needs of an epidemiologic study.
-
Quantitative criteria should be used to arrive at a credible and cost-effective ranking of sites for study.
-
An iterative procedure should be used for making dose estimates that increase in detail after specific minimum dose criteria are exceeded.






