Executive Summary
Since it appeared in the early 1980s, the acquired immune deficiency syndrome (AIDS), and the human immunodeficiency virus (HIV) that causes it, have wrought alarming physical and social devastation around the world. The number of people afflicted by the illness has increased markedly, and the range of communities affected has expanded. In the United States, while AIDS was first considered a disease of gay men, the reach of the epidemic now has expanded to injection drug users and their sexual partners, heterosexual partners of non-drug-using HIV-infected persons, infants born to mothers infected with HIV, and hemophiliacs and others exposed to blood products or blood transfusion.
The number of AIDS cases reported in the United States has grown rapidly, surpassing 100,000 in 1988, 200,000 in 1990, and 300,000 in 1992. The rate of increase is alarming—the first 100,000 reported cases occurred within an eight-year period, while the second 100,000 cases occurred in only a two-year period (CDC, 1992a). By the end of 1992, AIDS had become the leading cause of death for men between the ages of 25 and 44 and the fourth leading cause of death for women in that age group. By the end of 1992, over 200,000 people in the United States had died of complications related to AIDS (CDC, 1994).
Despite extensive efforts to develop effective treatments as well as a vaccine for HIV/AIDS, a fully effective treatment, cure, vaccine,
or other medical intervention appears to be years away. In conjunction with such developments, efforts to prevent the transmission of HIV through the cessation of behaviors that contribute to it must be expanded. This requires a commitment to understanding and intervening in human behavior.
This report surveys the AIDS research programs of the National Institute on Alcohol Abuse and Alcoholism (NIAAA), National Institute on Drug Abuse (NIDA), and National Institute of Mental Health (NIMH). Much of the AIDS research supported by these institutes is dedicated to developing and implementing (and to a lesser extent, evaluating) HIV preventive interventions in different populations. These interventions should be driven by basic behavioral and social science research on the determinants of behavior and behavior change; and to a great extent they are. Yet much of this basic research is still in its early stages, its development inhibited by a political climate during the first decade of the epidemic that made it difficult, and on some occasions impossible, to conduct research on the very behaviors in question: drug use and sex. For example, although numerous scientific and policy reports called for a federally sponsored, national survey of sexual behavior to help determine the nature and level of risk for HIV transmission in the general population, federal and congressional restrictions did not allow it.
The absence of information about the actual sexual behaviors in which people are engaging has hampered AIDS prevention efforts during this period. Now, although the current climate is more supportive of such research, basic behavioral and social science research is having to catch up. Moreover, much-needed efforts to integrate this research with basic biomedical research to obtain a more complete understanding of the complex factors that contribute to the transmission, disease progression, and prevention of HIV/AIDS among different people are still just beginning.
Part I of this report presents an overview of findings from relevant research to date. The committee felt it was important to elucidate the status of knowledge about the substance abuse and mental health aspects of HIV/AIDS first, as a backdrop to its assessment of the specific research programs of NIAAA, NIDA, and NIMH, which appears in Part II. This was thought to be a constructive approach, as it provides a baseline for assessing contributions, gaps, and future directions for AIDS research at the institutes.
Chapter 1 provides a general introduction to the report and its organization. Chapter 2 reviews the extent of knowledge about the range and contexts of sexual and substance-using behaviors by which HIV is transmitted. Although attempts to conduct large-scale projects on the specific behaviors involved have been hampered, significant contributions to the knowledge base have come from individual studies using the methods of qualitative, social science research and behavioral epidemiology. Chapter 3 reviews what is known about the neurobiological, psychological, and social determinants of HIV risk behavior and examines the application of that basic knowledge to interventions directed at changing behavior in positive ways.
While preventing the initial transmission of HIV is the top priority from a public health perspective, it is equally important to develop effective treatment interventions for those people who are already infected. This requires knowledge about disease progression and how it might differ among individuals and populations. With respect to mental health and substance abuse, a key issue is the relationship between HIV, the brain, and behavior. This is explored in Chapter 4.
After highlighting some of the significant developments and outstanding gaps in AIDS-related behavior, mental health, and substance abuse research, the report moves in Part II to a discussion of the specific AIDS programs of NIAAA, NIDA, and NIMH. Chapter 5 describes the general context within which these programs have been operating. The most salient elements of that context are: (1) the recent reorganization of the Alcohol, Drug Abuse, and Mental Health Administration (ADAMHA); (2) the new budget and program authority of the Office of AIDS Research (OAR) at the National Institutes of Health (NIH); (3) the budget process; and (4) the grant review process. Chapter 6 contains the committee's assessment of the content and management of the institutes' AIDS programs themselves, focusing on balance among scientific approaches, the use of various funding mechanisms to support research, and the overall funding picture. Finally, Chapter 7 explores the relationship between the AIDS research programs of NIAAA, NIDA, and NIMH and the services programs formerly at ADAMHA and now at SAMHSA (and elsewhere in the Public Health Service).
PART I
RESEARCH FINDINGS AND OPPORTUNITIES
UNDERSTANDING HIV TRANSMISSION
Although the general categories of risk behavior are well known, the specific acts responsible for HIV transmission—use of injection drugs and sexual practices, for example—have not been adequately researched or discussed because of their sensitive nature. Yet, in order to develop effective prevention and treatment strategies, it is crucial to understand the specific behaviors involved. As described in Chapter 2, knowledge gained to date from the biological, epidemiological, psychological, and social sciences—much of it supported by NIAAA, NIDA, and NIMH—has helped elucidate the specific behaviors responsible for transmitting HIV. However, much remains to be learned. In particular, the prevalence and spread of those behaviors within certain communities is unknown.
SEX, DRUG USE, AND HIV TRANSMISSION
As with any sexually transmitted disease, the transmission of HIV is highly dependent on the number of partners one has and the specific types of sexual practices in which one engages. Anal intercourse carries the highest risk for HIV acquisition, followed by penile-vaginal and oral-genital intercourse. These sexual behaviors can place anyone at for HIV, regardless of his or her sexual orientation or gender. However, having a large number of sexual partners, and having a concurrent or prior infection (such as those associated with syphilis, gonorrhea, anal and genital warts, and herpes), may increase one's susceptibility to infection (CDC, 1987).
With respect to drug use, the sharing of hypodermic needles, syringes, and other injection paraphernalia is the most likely route of HIV transmission. The vector is the exchange of the blood of the previous user that is lodged in the paraphernalia. Certain methods of injection drug use are believed to be more risky than others, but any method that involves contact with potentially infected blood is dangerous (Grund, Kaplan, and Adriaans, 1989; Grund et al., 1990; Inciardi, 1990a; Inciardi and Page, 1991; Jose et al., 1993).
Much high-risk sexual and drug-using behavior in urban locales takes place in specific settings, such as ''shooting galleries" and
"crack houses." Although these settings have not yet been systematically studied, emerging ethnographic research has revealed some of the particular practices that contribute to the potential for HIV transmission among patrons (Marmor et al., 1987; Murphy and Waldorf, 1991; Schoenbaum et al., 1989). This research suggests that these dangerous settings would be particularly promising sites in which to prevent HIV transmission. A better understanding of the character and dynamics of such settings is required to develop fully effective preventive interventions.
In order to understand the potential course of the epidemic, behavioral epidemiologists have focused on determining the likelihood that infected individuals in one group will have contact with uninfected individuals in another; identifying the contexts that influence risk taking; investigating whether people in high-and low-risk groups are adopting recommended preventive behaviors; and examining how the spread of HIV infection will be altered by changing patterns in concurrent epidemics of drug use and infectious diseases.
In Chapter 2, after reviewing the status of knowledge on HIV transmission through sexual and drug-using behaviors, the committee makes several recommendations:
-
The committee recommends that a national survey be undertaken to determine the prevalence and correlates of HIV risk-taking behavior. NIAAA, NIDA, and NIMH should collaborate to sponsor such a survey.
-
The committee recommends that NIAAA, NIDA, and NIMH support studies of the social, psychological, and biological determinants of HIV risk-taking behavior using a variety of methods complementing the national survey. These studies would allow an understanding of the detailed mechanisms of such behavior, the social dynamics within which they occur, the differing conceptualization and terminology in specific communities, and the role of concurrent health events.
-
The committee recommends that NIAAA, NIDA, and NIMH develop studies of the highrisk settings, such as shooting galleries and crack houses, that may contribute to epidemic spread and implement prevention efforts in those settings.
-
The committee recommends that NIAAA, NIDA, and NIMH support research that integrates basic biological, epidemiological,
-
psychological, and social research to better understand transmission of HIV through sex and drug use.
-
The committee recommends that NIAAA, NIDA, and NIMH support research on AIDS risk and behavior change among seriously mentally ill men and women and people with other cognitive impairments, including those not in psychiatric treatment. Such research should be conducted in a range of geographic locations.
-
The committee recommends that the Public Health Service coordinate interagency efforts to monitor and respond to concurrent epidemics (such as drug use, violence, and infectious diseases) that will alter the course of the HIV epidemic.
UNDERSTANDING THE DETERMINANTS OF HIV RISK BEHAVIOR
Human behavior is determined by multiple factors in the individual and the environment, ranging from the micro-level molecular biological to the macro-level social and environmental. These factors often interact in mutually reciprocal relationships such that one factor both influences and is influenced by another.
Much has been learned from research on the psychosocial determinants of AIDS-related sexual and drug-using behaviors. However, research on the brain biology of sexuality and drug addiction has rarely been integrated into these studies, even though it may be critical for understanding and preventing highrisk behavior.
NEUROBIOLOGICAL DETERMINANTS OF RISK BEHAVIOR
While impressive strides have been made in the area of sexuality, the brain, and behavior, much remains to be learned. Characterization of sexual dimorphism at the genomic, molecular, cellular, behavioral, and social levels is still in its early stages. In the absence of this basic information, the very existence of a biology of highrisk sexual behaviors—a central missing element in basic biomedical and neurobiological AIDS research—cannot be approached.
Some outstanding issues include: identifying the neurochemical molecular substrates, if any, associated with sexual risk taking; determining how insights from the studies of the neurobiology of sexuality would relate to highrisk sexual behavior and to sexually transmitted diseases, including AIDS; and determining how society might best integrate the study of the biology of sexuality
and sexual risk taking into the broader context of sexuality, sexual behavior, and sexually transmitted diseases.
Understanding the biological basis of drug addiction is an important link to understanding drug abuse behaviors, and unlike the biology of sexuality has been the object of a great deal of research. Recent studies on the cellular and molecular basis of dependence and tolerance suggest that the processes are separate and distinct, and are mediated by different brain systems. The actual anatomical circuits in the brain that participate in the addictive process have been identified in some detail, although the story is not yet complete. The neuroanatomic and molecular bases of the withdrawal syndrome are less clearly understood.
PSYCHOSOCIAL DETERMINANTS OF RISK BEHAVIOR
Theoretical models, primarily from psychology, have been used either to predict risk behavior or to predict behavior change and, less frequently, maintenance of positive behavior change. Such models focus on the individual's perception of susceptibility, perception of benefits, constraints, and intentions to behave in particular ways (Fishbein et al., 1991). The concept of self-efficacy—the individual's belief that he or she can effectively carry out a desired behavior in a particular setting—is central to most models.
Despite their conceptual contributions, current theoretical models are limited in their ability to predict risk behavior for two main reasons. First, with respect to sexual behavior, the models are based on the assumption that sexual encounters are regulated by self-formulated plans of action, and that individuals are acting in an intentional and volitional manner when engaging in sexual activity. However, sexual behavior is often impulsive and, at least in part, physiologically motivated.
Second, the dominant theoretical models of behavior do not easily accommodate contextual personal and sociocultural variables such as gender, race/ethnicity, culture, and class. For example, gender roles and cultural values and norms influence the behavior of women and men and the nature of the relationships in which sexual activity occurs. Unsafe sexual practices often are not the result of a deficit of knowledge, motivation, or skill, but instead have meaning within a given personal and sociocultural context. A great deal of work remains to be done to better integrate theories of gender and culture with models of behavior change.
Additionally, models designed to explain or predict risk behavior
tend to focus only on one level of analysis—the individual—without regard for other levels, such as the culture and community to which an individual belongs. This focus on the individual has obscured the influence of social factors.
SOCIAL SCIENCE PERSPECTIVES ON BEHAVIOR AND BEHAVIOR CHANGE
Social science perspectives, which only recently have been applied broadly to AIDS research, have the potential for productively refocusing the investigation of AIDS to reveal the complex, yet powerful linkages between individuals and their social structure, and to suggest how specific social changes can inspire individual changes (Adam, 1992; Friedman, Des Jarlais, and Ward, 1994).
Several levels of social arrangements can affect behaviors related to the transmission of HIV—ranging from couples to social networks to the community to society as a whole. At the broadest level, social conditions, such as the lack of universal access to health care, racial and ethnic discrimination, unemployment, and a lack of public monies to promote AIDS prevention, contribute to a social context in which HIV transmission is prone to occur.
The smallest unit of social interaction that may influence one's participation in risk-taking behavior is the dyad—the two-person relationship. However, social influence does not begin and end with a partner. The scope and character of one's broader social network—the array of individuals upon whom one relies for support, who serve as reference groups, and who establish group standards of conduct (social norms) and sanction behavior—are central to understanding the behavior that puts one at risk for HIV infection (Klovdahl, 1985; Klovdahl et al., 1994; Neaigus et al., 1994). It is at this level that the operation of social norms can best be observed and understood, and where intervention to change norms may be most feasible.
Social networks and the norms that govern the behavior of their members exist within communities, including those defined by culture. Sensitivity to the values and historical experiences of cultural communities is essential for designing and implementing appropriate AIDS interventions. At the same time, cultural values may be at cross-purposes with public health interests, as is the case with the opposition among many racial/ethnic community leaders to needle exchange programs in areas with significant drug-using populations (Bayer, 1994). How the various interests of different communities and of members within them are best
mediated in the context of HIV/AIDS is an important area of further study.
INTERVENTIONS TO CHANGE BEHAVIOR
Theoretical models and constructs from the psychological and social sciences, notwithstanding their limitations, have been applied to the design of preventive interventions and behavior change strategies in an attempt to prevent further transmission of HIV. AIDS prevention intervention research typically focuses on identifying and modifying behaviors—usually those related to drug use and sex—known to be associated with HIV infection and targets both uninfected and infected persons in a range of populations and settings. Although most studies target individuals, some recently have begun to focus on the community as the target for intervention, recognizing the importance of socially created norms as determinants of behavior.
AIDS intervention studies employ a range of methodologies. Although experimental studies that randomize subjects into control and experimental groups have been considered to be the "gold standard," most studies do not adhere to this rigid design because of the difficulties associated with maintaining experimental conditions in the settings involved. Moreover, in some cases the randomized controlled trial is simply not appropriate for the question under study (Oakley, 1989). Rather, simple pre-post comparison studies are the most common means of assessing the effects of prevention programs, and specific changes (such as increased condom use) and mediating variables (such as demonstrated self-efficacy) have been the standard outcome measures for determining whether or not a particular intervention has been successful.
Even though many intervention studies have demonstrated behavior change, they may be limited in a few ways: (1) most rely solely on self-reported data; (2) for the most part they have not yet demonstrated long-term behavior change (beyond 6 months); (3) it is not yet known whether they work with populations outside of their target groups; (4) many interventions may not be cost-effective to implement on a larger scale; and (5) with few exceptions, they do not measure HIV transmission and do not necessarily indicate that HIV infection has been averted.
The mixed results of interventions informed by basic psychological and social research suggests that much remains to be learned—including how the psychological and social dimensions of behavior interact with the biological to encourage or prevent risky behavior
and to initiate and maintain positive behavior change. Cross-disciplinary research in this regard will play an important role in improving the design, application, and evaluation of HIV prevention interventions.
After reviewing relevant neurobiological, psychological, and social research supported by NIAAA, NIDA, and NIMH in Chapter 3, the committee makes the following set of recommendations:
-
The committee recommends that NIAAA, NIDA, and NIMH expand basic research on the biology of sexuality as it potentially relates to highrisk sexual behaviors. This might include research on the central nervous system (CNS) sexual systems that mediate sexual behaviors, the CNS neural systems underlying sexual behavior, and the molecular genetics of sexual behaviors.
-
The committee recommends that NIAAA, NIDA, and NIMH expand research on the biology of substance abuse to provide additional knowledge for approaching highrisk behaviors. This might include research to define structure-activity relationships in the function of dopamine systems; the role of noradrenergic systems and molecular mechanisms in the components of addiction (including euphoria, tolerance, sensitization, and withdrawal); the role of opiate peptide receptor subtypes in components of the addiction-abuse syndrome; as well as research to identify mechanisms of cocaine addiction.
-
The committee recommends that, where appropriate, NIAAA, NIDA, and NIMH coordinate their efforts with other relevant federal agencies (e.g., other NIH institutes, the National Science Foundation) that are also attempting to integrate biological, behavioral, and social research to define highrisk behaviors.
-
The committee recommends that NIAAA, NIDA, and NIMH support AIDS research that integrates theories of gender (identity, development, and dynamics) and behavior change models.
-
The committee recommends that NIAAA, NIDA, and NIMH expand the research effort examining social and structural factors (such as class, race/ethnicity, gender relations, and community) that increase risk for AIDS, affect progression of disease, and provide points of intervention. This
-
might require research that takes as the unit of analysis the social context and relationship (e.g., dyads, families, communities) in which HIV occurs—as opposed to the individual at risk of or who has HIV.
-
The committee recommends that NIAAA, NIDA, and NIMH, in conjunction with other NIH institutes, develop new and existing woman-controlled HIV/STD prevention methods (e.g., female condoms and microbicides) and examine the social and behavioral issues related to their use.
-
The committee recommends that NIAAA, NIDA, and NIMH support basic and applied research on the maintenance of behavior change, for example, risky sexual behavior and alcohol and other drug-using behavior, including the prevention of relapse. (The committee notes that this has been recommended in previous NRC reports—AIDS: The Second Decade, 1990; AIDS, Sexual Behavior, and Intravenous Drug Use, 1989—but has not been attended to adequately.)
-
The committee recommends that NIAAA, NIDA, and NIMH expand funding for HIV intervention research initiatives, particularly those that: (1) have rigorous evaluation components; (2) investigate motivations, intentions, and barriers in addition to behavior change; (3) include outcome measures in addition to behavior change, such as HIV seroprevalence, STD rates, and pregnancy rates; and (4) target a full range of racial/ethnic, gender, and cultural groups for the purpose of assessing between-group differences.
-
The committee recommends that NIAAA, NIDA, and NIMH support research that estimates the number of HIV infections averted by current prevention efforts and that includes cost estimates for these efforts.
DISEASE PROGRESSION AND INTERVENTION
Although every effort must be made to prevent new transmission of HIV, it is equally important to diagnose, treat, and care adequately for people who already are infected. Research on the pathogenesis and disease progression of HIV/AIDS relevant to mental health and substance abuse has developed quickly over the past decade. This research, which ranges from basic molecular and cellular biology to clinical pathology to social psychology, has provided some important clues about the relationship between
HIV, the brain, and behavior, and most importantly, about their interactive nature. Chief interactions discussed in Chapter 4 include: (1) HIV infection of the brain and the effects it has on the central nervous system; (2) effects of interactions among HIV infection, substance use, and mental illness, and the unique medical care and treatment issues associated with such interactions; and (3) the relationship between psychosocial factors and HIV infection for individuals with the disease, as well as for their loved ones and caregivers.
THE RELATIONSHIP BETWEEN HIV AND THE CENTRAL NERVOUS SYSTEM
HIV-associated conditions affecting both the central nervous system (CNS) and the peripheral nervous system (PNS) are common and result in considerable morbidity and mortality. Some conditions are secondary complications of HIV infection, resulting from opportunistic infections or systemic organ dysfunction that follows from immune deficiency induced by HIV infection. Among these are cerebral toxoplasmosis, primary CNS lymphoma, cryptococcal meningitis, and progressive multifocal leuko-encephalopathy.
Other conditions likely result directly from HIV infection itself. The pathogenesis of these primary complications is not yet clearly understood, but they are thought to be sequelae of interactions among HIV, the immune system, and various components of the nervous system. These conditions include, at the early stage of HIV infection, mild meningitis with headache. As the infection progresses, patients may experience "aseptic" meningitis, peripheral neuropathy, and, most significantly, a set of afflictions collectively known as AIDS dementia complex (ADC).
In its mild form, ADC syndrome slows intellectual processing, blunts concentration and impairs rapid and fine motor control. When more severe, it can dull the personality and cause truly devastating dementia that reduces the patient to a shell of his or her former self, impairs walking, and, eventually, leaves the victim bedridden, incontinent, and mute (Navia et al., 1986; Navia, Jordan, and Price, 1986; Price and Sidtis, 1992). ADC can be a source of protracted and severe disability, modifying and markedly diminishing the quality of remaining life, reducing enjoyment of work and daily life, depriving the patient of social and intellectual pleasures, and requiring emotionally and financially costly care.
The core features of the ADC have now been well characterized,
and a descriptive staging system, useful to provide a common vocabulary for both clinical practice and clinical investigation, has been developed (see Price, 1994; Sidtis, 1994). However, the character of HIV infection of the CNS is not yet fully understood. Changes in the virus, with respect to both predominant cell tropism and virulence may be important. A number of studies have shown early invasion of the CNS and early local host immune responses in the cerebrospinal fluid (CSF). A critical question remaining to be answered is whether the virus then persists in the CNS in latent form or as an indolent infection that is asymptomatic. If it can remain latent, it is important to know what cells might harbor the proviral DNA: microglia, astrocytes, or other CNS cells. Pathological observations to date suggest that the virus indirectly injures the brain rather than directly killing or infecting nerve cells. This distinguishes HIV from other viral infections of the brain, such as those caused by poliovirus or herpes simplex virus.
Understanding the pathogenic mechanisms has great potential importance for treating ADC patients not only using methods that interfere with the virus, but also strategies directed at interrupting some of its toxic processes. Indeed, these considerations underlie some of the approaches now being taken to treat ADC, including treatment protocols using nimodipine, a calcium channel blocker that can prevent gp 120-induced neuronal death in vitro, and pentoxyphilline, an antagonist of Tumor Necrosis Factor.
Research on ADC and CNS HIV infection has biological importance not only for understanding the nature and course of HIV infection, but also more generally for suggesting mechanisms involved in other infectious, immunological, and neurodegenerative diseases. The early penetration of the blood-brain barrier by HIV, the local immune response detected in the spinal fluid, and the subsequent active replication of HIV in the brain late in infection hold clues regarding the CNS ecology of HIV.
INTERACTIONS AMONG HIV, SUBSTANCE USE, AND MENTAL ILLNESS
The second type of interaction among brain, behavior, and HIV relates to the unique issues associated with multiple diagnoses, that is diagnosis of any combination of HIV, drug or alcohol abuse, and mental illness.
Drinking alcohol to excess has been shown to cause damage to the immune system (Kruger and Jerrells, 1992). It therefore seems reasonable to assume that alcohol consumption in significant amounts
might also make an individual more vulnerable to HIV infection and facilitate the transition from HIV infection to AIDS. Alcohol may increase the susceptibility of phagocytes to initial infection, impairing their ability to eliminate the virus in the early stages of infection, and thus increasing their potential to function as reservoirs for HIV (Kruger and Jerrells, 1992).
Alcohol also reduces the number of T-cells in the spleen, lymph nodes, thymus, and blood (Jerrells, Smith, and Eckardt, 1990; Saad and Jerrells, 1991). This loss of cells in the immune system may accelerate the onset of clinical manifestations among HIV-infected people, but research on this issue is inconclusive. Although it is clear that excessive alcohol consumption can impair cell-mediated immunity, this has not translated into accelerated clinical progression of HIV to AIDS in reports to date (Isaki and Gordis, 1993; Kaslow et al., 1989). The precise relationship between alcohol use and HIV/AIDS remains to be elucidated.
Treatment of drug users who are at risk for or already infected with HIV is complicated by many factors: the fact that the clinical course of HIV infection may have special characteristics among drug injectors, the existence of complex medical and psychosocial comorbidities, and the often tenuous relationship between drug users and the health care system. Although a significant body of research has been conducted on clinical aspects of HIV disease and its management among drug users, a number of key research areas require further examination. These include: charting changes in the spectrum of HIV disease over time, such as, the emergence or disappearance of AIDS-related illnesses in this group as a result of medical interventions or other differences; further epidemiologic and clinical study of tuberculosis among HIV-infected drug users; and clarifying further the clinical expression and outcome of hepatitis C infection in co-infected groups (Donahue et al., 1991; Haverkos and Lange, 1990; Novick et al., 1988; van den Hoek et al., 1990).
Pharmacokinetic and other pharmacologic studies involving drugs of abuse and medications used for the treatment of HIV-related disease, as well as prescribed opioids such as methadone used to treat opiate addiction, will help determine whether interactions have potential clinical significance. With the increasing number of medications continually being added to the standard therapeutic regimens of HIV-infected patients, it will be even more important in the future to assess these agents pharmacologically in relation to psychoactive drugs, both prescribed and over-the-counter drugs. Because most of the therapies used for HIV infection and related conditions (e.g., tuberculosis) require long-term medical
regimens, often involving multiple medications, the levels of adherence to medical care among drug users in a variety of treatment settings must be examined. Finally, because drug injectors as a group are likely to have a high lifetime prevalence of depression, anxiety, personality disorder, and other psychiatric diagnoses (Batki, 1990a; Rounsaville et al., 1991), the comorbidity of drug abuse and psychiatric disorders must be studied further. On this point, a research and clinical agenda that brings together mental health services, drug abuse treatment, and medical care for HIV should be pursued. This will frequently require crossing categorical boundaries that separate research and services programs.
The convergence of psychiatric disorders, substance abuse, and HIV among those already infected who are also severely mentally ill raises difficult questions about appropriate treatment, similar to those related to injection drug users. For example, possible toxic reactions between antipsychotic and anti-HIV medications, and perhaps even antinarcotic medications, require further investigation. Moreover, little if anything is yet known about HIV disease progression among the seriously mentally ill. Given what has already been learned about the interactions of the virus and the CNS, it is possible that there are unique manifestations among people already suffering brain disorders. Yet, this area remains remarkably understudied.
THE RELATIONSHIP BETWEEN PSYCHOSOCIAL FACTORS AND HIV INFECTION
The final type of brain, behavior, and HIV interaction addressed in Chapter 4 is the relationship between HIV infection and certain psychiatric disorders, behavioral states, or reactions. For example, concern about HIV infection can lead to anxiety or depression. The stress of coping with the illness is not confined to the person infected, but also encompasses those who care for him or her. While not directly related to HIV infection per se, there are both diagnostic and therapeutic implications in the management of HIV infection.
The bidirectional relationship between psychosocial factors and HIV infection may influence disease progression and the ability to effectively treat related symptoms. These phenomena primarily have been informed by two types of research: psychoneuroimmunology and psychosocial research on coping and caregiving.
In the context of HIV/AIDS, psychoneuroimmunology is the study of how mental states might modify immune defenses and even viral replication within the immune system. Depression
and other mental states may have an impact on the immune system through the hypothalamic-pituitary-adrenal axis, the autonomic nervous system, and other pathways. Stress and other nervous system perturbations may alter immune function in both animal models and humans. The complex interactions among HIV infection, immune function, and mental states have been a major theme of AIDS-related research, particularly at NIMH. Although much has been learned about the effects of HIV on psychosocial factors, less is known about the effects of psychosocial factors on HIV.
A number of studies have looked at the psychiatric implications of HIV/AIDS, but they have not been able to determine whether psychiatric disorder is an effect of HIV or existed prior to HIV. For example, among gay men, psychiatric morbidity rates are relatively high regardless of HIV serostatus. This may be due to the fact that these men have to deal with a host of psychological issues related to being gay and their consequent status in society (Atkinson et al., 1988; Tross et al., 1987; Williams et al., 1991). This finding suggests the need to understand how the context of HIV affects psychiatric symptoms among gay men, that is, what disclosure and recognition of both being gay and having HIV may mean.
After learning that they are HIV positive, most people employ strategies of coping and social support that vary at different stages of the disease. Studies of asymptomatic HIV-positive people suggest that ''avoidant coping"—screening out the negative implications and focusing instead on the positive—does not protect them from emotional distress (Joseph et al., 1990; Nicholson and Long, 1990; Rabkin et al., 1990; Storosum, Van Den Boom, and Beauzekom, 1990). Cognitive coping strategies, however, such as positive reinterpretation, sense of control over events, and positive changes in daily life, seem to promote psychological well-being throughout the course of the disease (Hart et al., 1990; Rabkin et al., 1991; Storosum, Van Den Boom, and Beauzekom, 1990).
To date, nearly all of the research on coping—including that on bereavement after the death of someone with AIDS—has focused on gay men. Much remains to be learned about how people from various gender, racial/ethnic, and cultural groups cope with their own and others' HIV status, including not only general psychological strategies, but also motivation or resistance to adherence to medical treatment and its implications for disease progression.
People who provide care to those with HIV/AIDS also experience its psychological impact. Most informal AIDS caregivers
(relatives, lovers, and friends) are forced to learn a range of technical and emotional skills on the job, and many experience dysphoria—an emotional state of depression, anxiety, and restlessness (Folkman, Chesney, and Christopher-Richards, 1994).
Although stress experienced by formal HIV caregivers is reported in professional and lay articles, research has not included systematic documentation of the incidence and prevalence of physical, psychological, occupational, or interpersonal symptoms or disorders in health care professionals who devote a substantial amount of their clinical activities to patients with HIV illness (Silverman, 1993). Anecdotal reports of various symptoms, such as AIDS-related nightmares and psychological numbing, together with other symptoms related to stress and depression, suggest that some caregivers might be experiencing a form of posttraumatic stress disorder. However, according to Silverman (1993), only one published psychiatry article has addressed this possibility (Horstman and McKusick, 1986).
The psychological and physical implications of AIDS caregiving for health professionals undoubtedly is affected by their attitude toward AIDS. Surveys among nurses, physicians, dentists, social workers, psychiatrists, and health profession students are notable for revealing a consistent aversion among caregivers to the HIV/AIDS disease, to patients and their lifestyles, and to caregiving work itself, much of it based on fear of contagion (Silverman, 1993). A related issue is the psychological and social consequences (including occupational stress) of dealing with HIV/AIDS among outreach workers and service providers (Broadhead and Fox, 1993).
After reviewing the literature on disease progression and intervention in Chapter 4, the committee makes a number of recommendations:
4.1 The committee recommends that NIMH continue research on the pathogenesis of HIV infection of the brain, including the factors controlling virus replication such as local immune defenses and changes in the viral genome determining neuropathogenicity.
4.2 The committee recommends that NIMH continue research on the pathobiology of nervous system injury underlying the AIDS dementia complex, including the morphological, biochemical, and molecular basis of neuronal dysfunction related to viral and cellular gene expression.
4.3 The committee recommends that NIMH support collaborative
studies of the prevention and treatment of the AIDS dementia complex and other central nervous system complications of HIV infection.
4.4 The committee recommends that NIAAA, NIDA, and NIMH continue supporting research on the development of animal models for examining the basic neurochemical and behavioral changes associated with HIV/AIDS.
4.5 The committee recommends that NIAAA, NIDA, and NIMH expand research on the natural history of HIV infection among various populations, including injection drug users, other substance abusers, and the seriously mentally ill.
4.6 The committee recommends that NIAAA, NIDA, and NIMH support research on the relationship between the medical consequences of substance abuse and related behaviors and the clinical expression of HIV among the drug-using population.
4.7 The committee recommends that NIAAA, NIDA, and NIMH support research at the intersection of AIDS treatment and clinical care for substance abusers, addressing, for example, how to treat HIV/AIDS in mental health and substance abuse treatment programs.
4.8 The committee recommends that NIAAA, NIDA, and NIMH collaborate on research to investigate interactions among psychotropic (including illicit) drugs, antinarcotic and antipsychotic medications, and drugs used to treat HIV and opportunistic infections—for example, the effect of methadone on zidovudine (AZT)—including research on toxicities that might develop through such drug interactions.
4.9 The committee recommends that NIAAA, NIDA, and NIMH support more research on the neuropharmacology of anti-HIV medication.
4.10 The committee recommends that all relevant NIH institutes eliminate systemic barriers to the inclusion of injection drug users and other substance abusers in AIDS and AIDS-related clinical trials.
4.11 The committee recommends that NIAAA, NIDA, and NIMH support research on the utilization of health resources by people with AIDS, including substance abuse and mental health treatment programs. This research might include studies
of the extent and manner in which the medical system acts (intentionally or not) to exclude drug users from care.
4.12 The committee recommends that NIAAA, NIDA, and NIMH support research on the relationship between adherence to HIV/AIDS medical treatment and disease progression among individuals from diverse gender, racial/ethnic, and cultural groups.
4.13 The committee recommends that NIAAA, NIDA, and NIMH support research that integrates substance abuse and mental health treatment; in particular, demonstration projects for integrated multidisciplinary treatment systems that include mental health.
4.14 The committee recommends that NIMH support research on positive as well as negative consequences of HIV, for example, how people with AIDS and their caregivers maintain positive coping strategies in the face of the disease.
4.15 The committee recommends that NIAAA, NIDA, and NIMH support research on how families (broadly defined to include persons who consider themselves to be family through mutual commitment) from diverse racial/ethnic, socioeconomic, and sexual orientation backgrounds cope with the reality of having family members who are infected with HIV or have AIDS. Special attention should be given to patterns and consequences of caregiving in such families.
PART II
MANAGING THE AIDS RESEARCH PROGRAMS AT NIAAA, NIDA, AND NIMH
THE CONTEXT OF AIDS PROGRAMS AT NIAAA, NIDA, AND NIMH
In order to analyze the AIDS research programs of NIAAA, NIDA, and NIMH, one must understand the larger context in which they have been operating. The most significant elements for this study are: (1) the passage of the ADAMHA Reorganization Act of 1992 (PL 102–321), which separated ADAMHA's research and services entities into two different agencies (NIH and SAMHSA respectively); and (2) the NIH Revitalization Act of 1993 (PL 103–43), which assigned the NIH Office of AIDS Research (OAR)—housed in the office of the NIH director—new budgetary authority over
the AIDS programs of all NIH institutes. Other factors, such as the overall federal budget process and procedures for program planning and grant review, affect the nature of the AIDS programs of NIAAA, NIDA, and NIMH. Chapter 5 discusses these contextual factors.
EFFECTS OF THE ADAMHA REORGANIZATION AND NIH REAUTHORIZATION
Overall, the reorganization appears to have had only a limited effect on the research programs of the three institutes to date. Their organizational structure and staffing have been left largely intact, and their review processes remain the same as they were at ADAMHA for the period FY 1993 through FY 1996, as mandated by the reorganization legislation.
ADAMHA never had an official, authorized, functioning AIDS office. Because the Department of Health and Human Services (HHS) was considering reorganization, ADAMHA created an acting AIDS office rather than a permanent one. This office was headed by a part-time acting coordinator, who also directed the NIMH AIDS office. Each institute at ADAMHA devised its own way to manage its AIDS program: NIAAA had only one or two staff acting as AIDS coordinators on a part-time basis; NIDA had an official AIDS coordinator located within one of its regular divisions; and NIMH had a formal Office on AIDS Programs with a full-time director. These organizational structures were maintained after reorganization. Only three of the 24 programs that were transferred from ADAMHA to SAMHSA as a result of the reorganization are directly related to AIDS: AIDS health care worker training (moved from NIMH to CMHS); AIDS health care worker training/AIDS hotline (moved from NIDA to CSAT); and AIDS service delivery demonstrations (also moved from NIDA to CSAT).
Simultaneous to the ADAMHA reorganization, NIH underwent changes in its leadership, and under the NIH reauthorization bill passed in 1993 developed a much more centrally managed AIDS research effort as new and greater authority over the AIDS budget was given to OAR. The bill requires a comprehensive plan for the expenditures of appropriations, and authorizes an emergency discretionary fund for the director of OAR. According to the legislation, the plan must: (1) provide for the conduct and support of all AIDS activities at NIH; (2) prioritize the various AIDS activities, which are required to have objectives, measures, and a time frame; (3) ensure that the budget is allocated accordingly; (4) be updated
annually; (5) ensure that approval of specific projects and ongoing operation remain with the individual institute, center, and division directors; and (6) include a full range of research (including basic, applied, intramural, extramural, investigator-initiated, NIH-directed, and behavioral and social science research). This comprehensive plan is meant to be the basis for developing the annual budget requests for AIDS research.
Beginning in FY 1995, when AIDS funds are appropriated to NIH by Congress they will go directly to the director of OAR. The director will allocate to the individual institutes, centers, and divisions all of the funds received in accordance with the previously approved comprehensive plan for expenditures and appropriations.
THE BUDGET PROCESS
The specific funding of ADAMHA and NIH AIDS activities over time has taken place within the larger context of the overall federal budget process and has been directly affected by it. Appreciating the length and complexity of the budget process is critical for understanding the problems faced by rapidly changing and expanding research areas such as AIDS and by organizations that are in the process of restructuring, as were ADAMHA and NIH during the course of this study.
At any one point in time, an institute (whether at ADAMHA or NIH) must consider its research program in relation to three separate budgets. For example, at the end of calendar year 1993, an institute would have been in the midst of utilizing its current funds (which were allocated through September 30, 1994), presenting and defending its next year's (FY 95) budget to reviewers ranging from HHS and the Office of Management and Budget (OMB) to various congressional committees and interest groups, and planning the subsequent year's (FY 96) budget request.
NIAAA, NIDA, and NIMH joined in the FY 1994 NIH budget process after the major internal HHS decisions already had been made about the NIH AIDS budget, although they still were affected by those decisions. In FY 1994, as in all preceding years, once the institute received its budget allocation, the determination of the specific projects to fund was mostly a function of the grant review process.
THE GRANT REVIEW PROCESS
The ADAMHA Reorganization Act provided that the ADAMHA peer review systems, advisory councils, and scientific advisory committees would remain in effect through FY 1996 (ending September 30, 1996).
In most ways, the review procedures of the former ADAMHA research institutes are similar to the procedures at NIH. ADAMHA, like NIH, used a dual review system that separated technical and scientific assessment of projects from subsequent policy decisions concerning programmatic, scientific areas in which projects would be supported. This dual technical and programmatic review has been retained at the institutes. The scientific review process is kept separate from funding in part to ensure that program officials are not involved in making determinations on the scientific merit of research applications.
The first level of grant review is conducted by technical experts, largely from outside the federal government, and is designed to evaluate competing applications based on scientific and technical merit. The second level of review is conducted by advisory councils to assess the quality of the first-level review and to offer recommendations based on the relevance of the research to the institute's mission. The recommendations of both levels of review are advisory to the federal government and the final funding decisions reside with the institute director. Funding decisions are based not only on scientific merit and policy consideration, but may also consider administration policy, funding availability, and other factors.
Although the ADAMHA Reorganization Act mandated that the review processes (both at the first and second levels) be maintained through 1996, it is not yet clear how AIDS research applications at the three institutes will be reviewed when this period ends.
Given the composition of the NIH-wide AIDS study sections, some have expressed concern that applications related to the biobehavioral and social-behavioral research foci of NIAAA, NIDA, and NIMH—that is, the cross-disciplinary focus—will not fare well should the three institutes be subject to the overall NIH review process. While it is too soon to determine if this concern is well founded, the committee is aware that such concern is widespread among the institute program staff and the external research community.
It is the perception of many people, including some members of the committee and researchers interviewed for the study who have
sat on study sections, that the current NIH review and scoring procedures are inherently prejudicial to innovative and collaborative proposals. Review panel members are likely to regress the scores that they give to proposals involving disciplines other than their own. They often are not in a position to know what constitutes an "extremely good" proposal in another discipline. As a result, a single-discipline proposal will be less competitive to the extent that it is judged by specialists from other disciplines. A cross-disciplinary proposal is almost guaranteed to contain elements that will be unfamiliar to each reviewer.
These problems can be addressed either by working within the existing review procedures or by changing them. Within the system, one possibility is to weight ratings by the disciplinary competence of the raters or rescale scores so that they are standardized within different categories. More fundamental changes might include convening special review panels, affording votes to ad hoc reviewers, and dedicating funds to unconventional proposals (so that they compete only amongst themselves). Implementing these proposals would require considering the appropriateness of existing review staff for creating the appropriate panels and procedures. It also would have to be in accord with the scientific principles of evaluation. As this report was being written, the current NIH director established a series of inter-NIH panels to examine the ways in which the peer review process could be streamlined and more innovative projects rewarded.
From its overview of funding and administration of AIDS research, presented in Chapter 5, the committee makes two recommendations:
5.1 The committee recommends that NIAAA, NIDA, and NIMH develop new programs and grant review procedures to encourage and facilitate innovative, collaborative, and cross-disciplinary proposals.
5.2 The committee recommends that the NIH task force charged with streamlining peer review consider alternative scoring schemes that would favor cross-disciplinary and innovative research proposals.
RESEARCH FUNDING, PROGRAMS, AND PRIORITIES AT NIAAA, NIDA, AND NIMH
The specific AIDS programs and priorities of NIAAA, NIDA, and NIMH are analyzed in Chapter 6. The committee was asked
to assess the adequacy of the response of these three institutes to the AIDS epidemic by evaluating the scope and content of their AIDS research program activities and the balance between biomedical and behavioral research. The committee conducted a grant-by-grant analysis of all AIDS research projects funded by NIAAA, NIDA, and NIMH from FY 1983—the first year of AIDS funding—through FY 1992—the most recent year for which complete data were available. In addition, the committee reviewed a range of documents and plans produced by the institutes that describe their AIDS programs and priorities and interviewed a number of researchers, federal agency staff, and public advocates.
When the committee began to address the balance between biomedical and behavioral AIDS research at NIAAA, NIDA, and NIMH early in the study, it quickly realized that the categories were too limiting, because the definitions of "biomedical" and "behavioral" were not clear and because counterpoising these two categories masked the true level of cross-disciplinary research supported by the institutes. As the committee began to review the research portfolios grant by grant, it became apparent that labeling a project as either biomedical or behavioral was misleading, because significant portions of many projects included both.
As a consequence, the committee developed its own simple matrix, using four domains of science: biomedical/biobehavioral, epidemiological, psychosocial, and social-structural. These domains were cross-cut by two types of research: basic and applied. "Biomedical/biobehavioral" research focuses on improving knowledge about basic biological mechanisms and processes, disease pathogenesis, and clinical issues related to progression and treatment of HIV/AIDS. "Epidemiological" research focuses on transmission of HIV and the natural history of infection and disease progression. This category also includes biostatistical research to develop and refine mathematical modeling techniques for improved forecasting of HIV seroprevalence. ''Psychosocial" research includes efforts to understand psychological determinants of behavior and behavior change and to develop and evaluate preventive interventions, the effect of psychosocial variables on disease progression, and the impact of HIV/AIDS on behavior and psychological functioning. "Social-structural" research examines the social context in which HIV/AIDS is transmitted and experienced, by focusing on relationships, families, communities, institutions, and cultures rather than on individuals. Social-structural research includes research on health services, evaluation, and operations. "Basic" research studies the basic mechanisms underlying biological,
neurological, behavioral, and social processes and outcomes, and includes theoretical work. "Applied" research encompasses projects that test interventions.
The committee constructed an electronic database from abstracts of funded, extramural grants at NIAAA, NIDA, and NIMH from FY 1983 to FY 1992. Each AIDS grant was either single-coded with one of the four science categories or, where appropriate, multicoded in two or more of the science categories. These latter were considered by the committee to be cross-disciplinary research. In addition, each grant was coded as either basic or applied. Grants also were coded according to their funding mechanism.
A significant proportion of AIDS research at NIAAA, NIDA, and NIMH is cross-disciplinary, according to the committee's coding scheme. The committee's analysis also discovered that most extramural AIDS research grants funded by NIAAA, NIDA, and NIMH involve basic research.
Although NIAAA, NIDA, and NIMH have utilized all mechanisms for supporting extramural AIDS research, they have done so in different proportions. For example, the majority of NIAAA's extramural research grants and dollars have been committed to investigator-initiated grants (R01s); NIMH AIDS extramural funding has been largely committed to R01s and research centers (P50s); and NIDA's extramural program has employed the widest variety of mechanisms, particularly by committing resources to research demonstration projects (R18s).
When compared to non-AIDS research, a greater proportion of AIDS research at NIAAA, NIDA, and NIMH has been directed in some way by the institutes. The level of directed research in the AIDS programs—as evidenced by program announcements (PAs), requests for applications (RFAs), and the funding of core and center grants—suggests active leadership among the institute staff in encouraging investigations of specific AIDS-related topics. This kind of leadership may be more necessary in a new research field, such as AIDS, when it becomes important to recruit investigators who have been establishing careers in other, related areas of science.
NIAAA
Because of the small size of its AIDS program, NIAAA has never established an Office of AIDS and has never had any fulltime AIDS staff. Currently, NIAAA has two "part-time" coordinators—one for social and behavioral research and one for biomedical
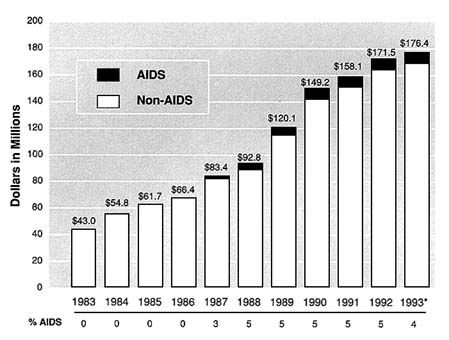
FIGURE S.1 NIAAA Expenditures (AIDS/Non-AIDS), 1983-1993. * Estimate. Source: NIAAA Budget Office.
research. AIDS funding at NIAAA increased from $2.4 million in FY 1987 (when the institute began its AIDS program) to $8 million in FY 1993, while total NIAAA funding (AIDS and non-AIDS) grew from $43 million to $176 million in the same period (Figure S.1).
Since NIAAA initiated its AIDS program, it has devoted much of its extramural and intramural research efforts to biomedical issues. NIAAA's broad research goal in this area is to elucidate how alcohol alters the immune system in ways that may compromise host defense against HIV. By 1990, NIAAA's research portfolio shifted so that half of the research grants had a biomedical component, half had a psychosocial component, and approximately one-fourth had an epidemiological component. None of the NIAAA research grants included a social-structural component until 1992.
NIDA
Although NIDA has had an AIDS coordinator since the mid-1980s, the institute only formally established an Office of AIDS
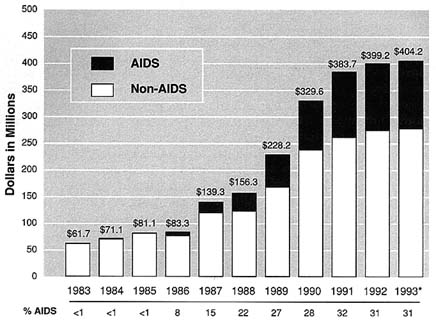
FIGURE S.2 NIDA Expenditures (AIDS/Non-AIDS), 1983-1993. * Estimate.Source: NIDA Budget Office.
in FY 1993, as required by the ADAMHA Reorganization Act. NIDA's AIDS program is designed to be decentralized and it appears that the office has a fairly limited coordinating role. NIDA's Office of AIDS has no budgetary or oversight authority for the wide range of AIDS activities. The current director of the Office of AIDS is also the acting director of the Division of Clinical Research. Although AIDS made up less than 1 percent of the total NIDA budget in FY 1983 (when NIDA's AIDS program began), it now constitutes approximately one-third ($127 million) of NIDA's budget, making it the third largest AIDS budget among the NIH institutes (Figure S.2). NIDA's AIDS portfolio has included a significant proportion of research demonstration projects (R18s) and cooperative agreements (U01s), in addition to traditional investigator-initiated grants (R01s). The bulk of NIDA's AIDS budget, however, remains allocated to research project grants (RPGs).
During the first decade of AIDS research funding at NIDA, a significant proportion of AIDS grants were biomedical and psychosocial, according to the committee's coding. Beginning in 1987, behavioral
interventions aimed at reducing highrisk drug-using and sexual behaviors became a significant focus of the AIDS program. Some also included a social-structural component.
NIDA-supported biomedical research focuses on: the interrelationships among HIV infection and effects of drugs of abuse on the immune, neuroendocrine, and central nervous systems; vertical transmission of HIV (pediatric AIDS projects); nonreusable syringe development; medications development to create new pharmacological agents for treating substance abuse and addictive disorders; and treatment to reduce drug use and related risky behaviors.
Epidemiological research supported by NIDA monitors HIV infection and risk behaviors among injection drug users through surveillance efforts that are an integral part of various program initiatives, including natural history studies, community-based outreach studies, and research on the sex-for-crack phenomenon.
NIDA supports basic psychosocial research on the determinants of AIDS risk behaviors (including sexual and drug-using behaviors) and applied research on AIDS preventive interventions, in particular, community-based AIDS programs to get people into drug treatment to reduce their AIDS risk behaviors. Social-structural research supported by NIDA investigates community and relationship factors that influence behaviors among drug users and their affiliates. Although NIDA has not yet been able to support such research, the institute's five-year strategic plan also emphasizes the need for research to examine HIV risk behaviors over time and under different social circumstances using multidimensional, longitudinal studies.
One of NIDA's main priorities for AIDS extramural and intramural research includes strategies for increasing the effectiveness of drug abuse treatment. Although treatment research has always existed as a priority for NIDA, the AIDS epidemic has heightened its importance. Drug addiction often involves physical and psychiatric health problems in addition to environmental and social conditions that may contribute to addiction; therefore, effective treatment requires a comprehensive approach. Indeed, because treatment research utilizes both pharmacologic and behavioral approaches, it is an area that was not easily categorized into one of the four scientific domains identified by the committee matrix. In fact, what makes these NIDA-funded research projects so unique is their emphasis on employing various combinations of pharmacologic, behavioral, and social approaches to improve the effectiveness of addiction treatment.
NIMH
Unlike NIDA and NIAAA, the AIDS research program at NIMH has for some time been managed by a central AIDS office, which develops the AIDS plan (with input from outside consultants and the various divisions and offices in NIMH), develops the annual AIDS budget, and directs the AIDS extramural program. The total NIMH budget increased from $191.5 to $560.5 million between FY 1983 and FY 1992. The AIDS budget increased from $200,000 to $76.2 million in the same period, making it the fourth largest AIDS budget among the NIH institutes. AIDS accounted for less than 1 percent of the total NIMH budget from FY 1983 through FY 1985 and rose to 14 percent by FY 1990 (Figure S.3).
NIMH funding generally has been committed to traditional investigator-initiated grants (RO1s) and research center grants (P50s). NIMH has supported more AIDS training grants than the other two institutes. With respect to representation across fields, the most significant single domain of extramural research at NIMH,
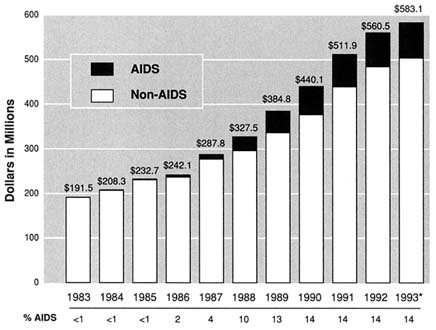
FIGURE S.3 NIMH Expenditures (AIDS/Non-AIDS), 1983–1993. *Estimate. Source: NIMH Budget Office.
according to the committee coding scheme, is psychosocial, followed closely by biomedical. In fact, the NIMH AIDS portfolio is relatively well-balanced between the two areas. Psychosocial research grants at NIMH include basic research on psychological mechanisms that underlie behavioral outcomes (including such topics as stress and coping processes and decision-making processes), behavioral and psychological responses to HIV testing and HIV status, and stress and coping among caregivers. Applied psychosocial research grants include behavioral interventions for HIV prevention. NIMH also supports behavioral epidemiology research to identify specific populations at risk for HIV/AIDS and to understand the specific behaviors of various populations. Biomedical/biobehavioral grants at NIMH examine the effects of HIV/AIDS on CNS function, the neuropsychological, neuropsychiatric, and neurological sequelae of HIV infection on the CNS, and the biological interface of stress and immune function (psychoneuroimmunology).
Many NIMH research grants have both biomedical and psychosocial research components. Applied biomedical/psychosocial grants include: behavioral interventions for HIV prevention that incorporate outcome measures such as HIV seropositivity rates and/or sexually transmitted disease (STD) rates in addition to behavioral outcome measures; and treatments and interventions for AIDS-related depression and emotional distress. Basic biomedical/psychosocial grants include: natural history and descriptive studies examining the neuropsychological and neurobehavioral sequelae of HIV infection and research examining the relationship between psychosocial factors and the immune system generally and/or with HIV disease progression specifically (in humans and nonhuman primates).
NIMH's few grants with a social-structural component are all basic research and focus on understanding the role of social relationships and social networks in shaping an individual's HIV risk-related behavior.
COLLABORATIVE PROJECTS
NIAAA, NIDA, and NIMH have collaborated on a number of AIDS research activities. Perhaps most significant are the jointly sponsored multidisciplinary extramural AIDS research centers, first initiated in 1986. Most funding for the centers has come from NIMH, but NIDA contributed some support to three centers from 1986 through 1991. In many instances, individual researchers
affiliated with the AIDS research centers also are supported with additional R01s from NIAAA, NIDA, or NIMH. Currently, NIMH is funding five AIDS research centers, each with a different focus, ranging from the basic cellular mechanisms underlying ADC to basic and applied research on HIV risk behaviors.
THE NIH CONTEXT
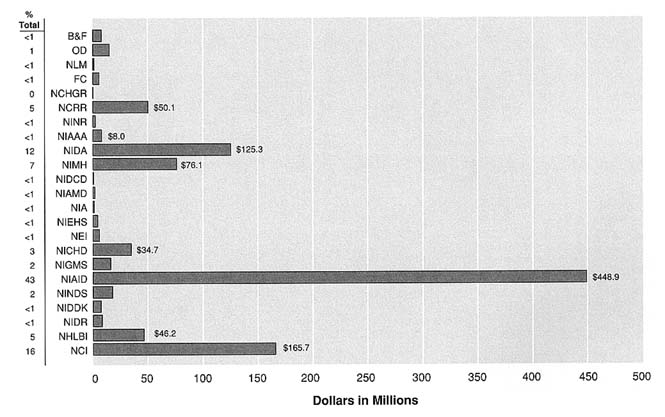
Total NIH funding increased more than 100 percent between 1983 and 1993 (from $4.3 billion to $10.3 billion). While growth in AIDS research in the same period appears to be explosive by absolute measures, it reflects the requirement to respond rapidly to a new disease with major public health implications. Seven NIH institutes (including NIDA and NIMH) together received the vast majority of AIDS funding during this period: 90 percent of the total AIDS budget for FY 1992 and over 90 percent of the cumulative total since 1983 (Figure S.4).
While NIAAA, NIDA, and NIMH represent a significant portion of the NIH AIDS budget, their funds are distributed quite differently from most of the other institutes. In FY 1992, NIAAA, NIDA, and NIMH together comprised approximately 20 percent of the total NIH AIDS budget (NIAAA was a relatively minor portion of this). According to estimates for FY 1993, the three institutes continued to account for 11 percent of the total NIH budget and 20 percent of the AIDS budget. Yet they funded more than 97 percent of NIH's AIDS-related behavioral research, 64 percent of surveillance, 54 percent of neuroscience and neuropsychiatric research at NIH, and over 30 percent of health services research and research training (as defined by categories used by the Public Health Service). On the other hand, NIAAA, NIDA, and NIMH account for less than one percent of the funding for therapeutic agents and 12 percent of the funding for biomedical research, two categories that together represent nearly 60 percent of the total NIH AIDS budget. It is clear that NIAAA, NIDA, and NIMH play a critical role in the overall NIH AIDS research agenda and in its biobehavioral and behavioral agenda in particular.
After reviewing the content and management of the AIDS research portfolios of the NIAAA, NIDA, and NIMH in Chapter 6, the committee makes several recommendations:
6.1 The committee recommends that NIAAA, NIDA, and NIMH each establish a position for a full-time AIDS coordinator. The coordinator should be provided appropriate resources
to develop and coordinate the institute's AIDS programs in cooperation with division and branch staff. The coordinator also should be linked to the NIH Office of AIDS Research.
6.2 The committee encourages OAR to serve as a catalyst for cross-institute and cross-PHS agency research collaborations through its funding authority and leadership role.
6.3 The committee recommends that the OAR leadership include competence in biomedical, neuroscience, behavioral, and social science perspectives.
6.4 The committee recommends that NIAAA, NIDA, and NIMH ensure the maintenance of the behavioral and social science research programs of the three institutes within the NIH context. The committee supports the recommendation of the National Commission on AIDS (1993b) to expand research in the following behavioral and social science research perspectives: behavioral epidemiology; cognitive science; cultural and ethnographic studies; intervention research; mental health research; behavioral aspects of technological interventions; and organizational studies. The committee adds to that list cost-effectiveness research and evaluation research.
6.5 The committee notes that, of all the types of AIDS research at NIAAA, NIDA, and NIMH, social science research is the most underfunded. The committee therefore recommends that the three institutes develop new initiatives to support research on the role of social, cultural, and structural factors in HIV/AIDS transmission, prevention, and intervention.
6.6 The committee recommends that, given the prominent role of drug injection in HIV transmission and given the considerable evidence that has been assembled over the past several years regarding the efficacy of needle exchange, the U.S. government remove current restrictions barring federal funding for needle exchange programs, promote services-oriented research to help implement such programs where warranted, and evaluate these programs with an eye toward maximizing their preventive impact.
6.7 The committee recommends that drug abuse treatment research at NIDA be continued to support the design and evaluation of innovative and cross-disciplinary drug abuse
treatment strategies, including collaborative efforts with SAMHSA. These strategies should include those targeted to highrisk populations, such as drug-involved offenders, prisoners, women and crack-cocaine users. The committee urges NIDA to pay particular attention to developing treatment strategies for crack-cocaine.
6.8 The committee recommends that NIAAA, NIDA, and NIMH restore support for research demonstration projects, using a mechanism similar to the R18 that facilitates cooperation between the NIH research institute and the relevant PHS services agency or agencies.
6.9 The committee recommends that an effort be made to coordinate between institutes that have overlapping AIDS research programs (for example, HIV and CNS function at NIMH and NINDS) by collaborating in the program development, review, and funding processes.
6.10 Given the disproportionate impact of the epidemic on men, African Americans, and Hispanics/Latinos, it is important to understand the sociocultural-specific factors—including gender, race/ethnicity, and class—that play a role in the behavioral aspects of AIDS. Therefore, the committee recommends that NIAAA, NIDA, and NIMH, with input from appropriate experts, develop a mechanism for collecting and reporting data on the gender, race/ethnicity, and socioeconomic status (class) of study populations in projects supported by the institutes. Such data collection and reporting should be guided by clear articulation of the role of these variables in the epidemic.
LINKAGES BETWEEN RESEARCH AND SERVICES
With respect to AIDS prevention and intervention, research findings must be disseminated to the field as quickly and as effectively as possible. At the same time, service providers often are in a unique position to discover new, researchable questions. How the two worlds of researchers and service providers interact is of great concern to all those involved in AIDS activities and is addressed in Chapter 7. To facilitate the exchange of ideas, federal agencies charged with missions for research and services must overcome differences and develop strategies for effective coordination and communication.
AIDS PROGRAMS AT SAMHSA
The integration of substance abuse and mental health services into the general health care system is the primary goal of the SAMHSA AIDS office, which was created in 1992 by the ADAMHA Reorganization Act. Currently, SAMHSA has an associate administrator for AIDS who is located in the office of the SAMHSA administrator. The associate administrator works with the three center offices on AIDS. These, too, were established by the ADAMHA reorganization bill, and are responsible for ensuring that HIV/AIDS issues are addressed and integrated into the overall programs of the center.
SAMHSA's total budget (comparable to the ADAMHA services programs' budget before FY 1993) increased from $794.7 million in FY 1988 to a $2.1 billion appropriation in FY 1993. During the same time period, specifically identified AIDS funding decreased from $42.5 million to $28 million (falling from 5 percent of the total budget to 1 percent). To some degree, the small proportion of the SAMHSA budget devoted to AIDS is a legacy of the ADAMHA years, during which the vast majority of AIDS funding at the agency was allocated to research activities (Figure S.5). These research activities are now located at NIH.
As of FY 1994, only four programs are recognized in SAMHSA's formal budget as AIDS-related. Two of these programs—which constitute the bulk of the agency's specific HIV/AIDS funds—are located in CSAT (formerly OTI). These are the Demonstrations and Training Program and the Treatment Improvement Demonstration Program, also known as the Comprehensive Community Treatment Program (CCTP). The CMHS AIDS program includes Training and AIDS Training, which began at NIMH in FY 1990, and a new AIDS Mental Health Services Demonstration Program, which was initiated in FY 1994. CSAP (formerly OSAP) had no AIDS program until FY 1994, when it proposed providing supplements to the Prevention Demonstrations for HighRisk Youth Program to fund outreach and risk reduction activities related to HIV/AIDS, including skills building, pre-post HIV test counseling, outreach to resistant populations, and services to those who have lost people to AIDS.
COLLABORATIONS BETWEEN RESEARCH AND SERVICES
A major concern about the ADAMHA Reorganization Act of 1992 (see Chapter 5) was how it would effect the relationship
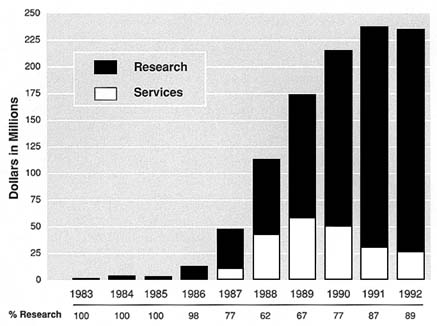
FIGURE S.5 ADAMHA AIDS Budget Authority, 1983-1992. Note: ''Research" includes former ADAMHA research activities currently with NIH (NIAAA, NIDA, NIMH, Office of Director, Buildings and Facilities); "Services" include former ADAMHA services activities currently with SAMHSA. Source: SAMHSA Budget Office.
between research and services, with regard to the rapid, bidirectional transmission of information, that is, the transfer of research findings "from bench to bedside" and observations and concerns from practitioners to the research community. Those concerned about the splitting of the research and services programs of ADAMHA expressed fears that such separation could disrupt the linkages that existed within a single agency and make the process of "technology transfer" more difficult. Those favoring the reorganization argued that separating the programs might well enhance relationships and thus improve linkages.
The problem of a lack of collaboration between research and services communities exists beyond the federal agencies, and is based in great part on different professional cultures. The two salient features of the research culture are the drive for "knowledge for knowledge's sake" and the existence of a reward system based on amount and type of publications. The pursuit of knowledge involves the employment of a scientific method that emphasizes
conducting controlled experiments, limiting randomness of outcomes, and producing findings that can be replicated by other scientists.
In contrast, the services world is driven by a desire to provide direct services to as many people as possible. Although the methods of service delivery are planned and evaluated, their internal integrity is often less important than the number of clients served. And while extensive reporting is often required by funding sources, this too is seen as less important to the mission of service providers than actually rendering the services—indeed, it is often seen as a distraction. Furthermore, unlike researchers, service providers are trained to be subjective, to identify with and to advocate for their clients. These fundamental differences in culture and orientation between researchers and service providers result in significant barriers to collaboration.
This situation is experienced at the federal level among agencies charged respectively with supporting research and services programs and is evidenced by the relatively limited collaborations between the AIDS research programs at NIAAA, NIDA, and NIMH and the AIDS services programs of SAMHSA, Centers for Disease Control and Prevention (CDC), and the Health Resources and Services Administration (HRSA), and the broader service-providing community. Even information exchange is limited. For example, each year the three research institutes sponsor many research exchange meetings, including research planning meetings, technical review meetings, workshops, and symposia. However, most of these meetings are designed to bring researchers together and very few reach beyond the research community to include service providers.
All but one (the CMHS demonstrations) of the examples of collaborations between research and services noted in Chapter 7 predate the reorganization of ADAMHA. Moreover, according to institute and agency staff, these collaborations came about as a result of informal communications between individual staff. No formal mechanism existed—or yet exists—to encourage interagency cooperation. Although within ADAMHA there had been tension between the research and services entities, some fear that the organizational separation of those entities will make collaboration even more difficult. In addition to cultural differences, legal and regulatory barriers also hinder collaboration between the institutes now at NIH and the services entities at SAMHSA (as well as HRSA and CDC).
COORDINATION OF AIDS ACTIVITIES WITHIN THE PUBLIC HEALTH SERVICE AND THE DEPARTMENT OF HEALTH AND HUMAN SERVICES
The possibility of collaborative activities between NIAAA, NIDA, and NIMH and SAMHSA, CDC, and HRSA is framed within the larger context of PHS and HHS AIDS activities. Coordination of PHS programs has resided for the past number of years with the National AIDS Program Office (NAPO), and cross-agency coordination has been the function of the Federal Coordinating Committee on AIDS, chaired by PHS. In 1993, however, the Clinton administration elevated the status of federal AIDS coordination to a White House position with the appointment of a National AIDS Policy Coordinator responsible for all federal AIDS policy and program. The activities of the newly titled Office of National AIDS Policy (ONAP) and the structures of organization and coordination within PHS and across federal agencies were still being determined at the time this report was written.
Having reviewed in Chapter 7 the scope of collaborations and exchanges between the AIDS research programs of NIAAA, NIDA, and NIMH and the services programs of SAMHSA and other PHS entities, the committee makes the following recommendations:
7.1 The committee recommends that NIAAA, NIDA, and NIMH ensure adequate follow-up time and money in their AIDS intervention research grants to accelerate information dissemination activities, including technical assistance.
7.2 The committee recommends that formal mechanisms be developed within the NIH institutes and other PHS agencies to foster linkages between AIDS research and services.
7.3 The committee recommends that NIAAA, NIDA, and NIMH sponsor regular research exchange meetings with services agencies and service providers, including local, regional, and national meetings.
7.4 The committee recommends that the Assistant Secretary for Health of HHS and the Director of the Office of National AIDS Policy continue to develop a specific strategic plan for interagency cooperation and coordination among PHS AIDS activities, including an implementation plan.