SESSION IV
Fetal Tissue Transplantation
Plasticity and Commitment in the Developing Mammalian Brain
SUSAN MCCONNELL
Stanford University, Stanford, California
The mammalian brain is composed of a huge variety of nerve cells, or neurons, which arise from common progenitor cells of an embryonic structure called the neural tube. The hollow space inside the neural tube eventually becomes the ventricles of the brain; progenitor cells line the inner, ventricular, surface of the tube. In the fully formed brain, a multilayered sheet of neurons, the cerebral cortex, covers the cerebral hemispheres. Some regions of the cortex are involved with movement and sensation, but other are involved with higher-order analysis and reasoning. Thus, the cortex is responsible for our highest cognitive and perceptual processes. Studies of the development of the cerebral cortex are helping to explain how neurons achieve their individual identities and may shed light on the mechanisms of fetal brain injury.
The cortex is particularly amenable to this kind of investigation because its neurons are organized into six distinct layers, each containing specific types of neurons that make highly specialized axonal connections (Figure 4). The upper layers of cortical neurons typically send their axons to other cortical cells, and neurons in the lower levels communicate with neurons located beneath the cortex in deeper regions of the brain.
Progenitor cells give birth to daughter cells through the process of mitosis, in which they double their chromosomal package and then split in half. In the forming of the cerebral cortex, the first cells to be born and migrate out from the ventricular surface are destined for the deepest layers of the cortex, layers five and six. They travel along special, elongated cells, called radial glial fibers,
which are transiently present during development and serve as a guide wire for the early migrations of cortical neurons. Neurons born later migrate past the early settlers to the top of the cortical plate. Because the different layers are generated in a precise sequence, a cortical neuron's "birthday" provides a remarkably accurate tool for predicting its ultimate position and connections.
Two explanations have been put forth to account for the correlation between cell birthday and ultimate destiny. One possibility is that a neuron's fate is determined in advance according to a kind of internal clock in its progenitor cell. An alternative view is that neurons within a given layer adopt their identities in response to environmental influences, which are likely to be chemical in nature and could occur before or during the cells' migration.
In animal experiments designed to determine the developmental potential of cortical neurons, cell groups containing progenitor cells were removed from developing rat brains at specific stages and transplanted into brains at different stages of development. For example, cells normally destined to become layer-six neurons were transplanted, before migration, into an environment in which the deep layers had already been generated and upper-layer neurons were in the process of migrating. What resulted was that the progenitor cells transplanted at
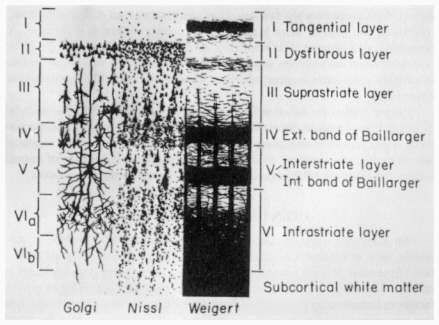
Figure 4
Cell layers and fiber arrangement of the human cerebral cortex. SOURCE: Carpenter, M.B., Core Text of Neuroanatomy, Third Edition, Williams and Wilkins, 1985. Reprinted with permission.
the point in the cell cycle just before mitosis migrated to their normal destination, as dictated by their birthday. Cells transplanted while still in the early phase of the cell cycle changed their normal fate and migrated to the cortical layer dictated by the brain into which they had been transplanted. Thus, the influence of the environment was a potent force in cell differentiation, but only if the cells are present just before mitosis. Further, these influences affect only those cells that have not divided yet, but they have no effect on migrating cells.
This scheme is complicated by the results of experiments suggesting that there is an asymmetric division in mitosis in the generation of cortical neurons. That is, instead of the two daughter cells migrating to their appropriate levels, it seems that sometimes only one daughter cell migrates and the other returns to an early stage in the cell cycle and is capable of dividing again. Further, this cell could later give rise to neurons in other cortical layers. The implication of these findings is that the message from the environment, acting on the mother cell in the early stages of the cell cycle, can be erased later by the daughter cell that does not migrate and returns to the cell cycle.
One proposed mechanism for asymmetric division was that the mother cell splits in a plane parallel to the ventricular surface, thus leaving one cell still adherent, or moored, to the ventricular surface while the other cell migrates away. Another idea was that there might be observable structural, or morphological, differences between two daughter cells that behaved differently. It was possible to test both of these hypotheses by directly observing dividing cells, containing a fluorescent dye, through a specially equipped microscope. The cells were observed to divide in a plane perpendicular to the ventricular surface in a highly predictable manner, and they showed no noticeable morphological differences.
Current studies are aimed at finding the molecular mechanisms that underlie the regulation of cortical development. These mechanisms may have important implications for neural transplant studies. There are, however, many congenital defects typified by abnormal cortical development, and a knowledge of normal cortical development is key to understanding how such perturbations occur.
OPEN DISCUSSION PERIOD
An interesting question concerned what was known about the role of substances such as hormones or growth factors in cortical cell mitosis and migration. At present, it is not possible to tell an upper-layer from a deep-layer cell in culture. Therefore, in order to be able to assess the effects of different growth factors or hormones on progenitor cells and their subsequent differentiation, it is first necessary to find markers for the different cell layer types. Also discussed was whether plasticity is possible in every cell cycle. It is suspected that late progenitors lose the ability to produce deeper-layer cells, and experiments to test that are under way.
Noting that the effects of lead in retarding cognitive development are still unexplained, a participant suggested that the culture system described in which a slice of developing cortex is maintained in vitro might provide an excellent assay for observing the influence of toxic substances on early stages of cortical neurogenesis and migration.
Stem-Cell Transplantation in Human Brain Disease
RONALD MCKAY
National Institute of Neurological Disorder and Stroke, Bethesda, Maryland
The central problem of modern neurobiology is the molecular complexity of the nervous system. For all the proteins active in the nervous system there is generally a large family of genes, as well as a whole series of steps in the expression of the genes, that increases the functional diversity of the proteins. One path toward developing a strategy for understanding neuronal complexity is to focus initial attention on stem cells, the multipotential cells from which neurons form during fetal development.
Neuroepithelial stem cells, which line the walls of the embryonic neural tube (the neuroepithelium), generate all the neurons and glial cells of the central nervous system. Many proteins thought to contribute to driving differentiation of these and other body cells are expressed only in highly specific subsets of cells: for example, certain classes of neurons or specific types of endocrine, skin, or muscle cells. A protein called nestin is expressed in all precursor cells throughout the embryonic nervous system (and in early muscle cells) that can give rise to neurons or to the supporting cells of the brain called glia. Nestin expression is turned off abruptly in the nervous system when the cells become postmitotic, functionally committed neurons (or glia). The portion of the nestin gene that controls its expression in nervous system precursor cells differs from the part that controls muscle expression. That fact, along with nestin's ubiquity in the nervous system, makes it an ideal protein for experimental manipulations of neuroepithelial stem cells.
Neural stem cells in culture continue to express nestin for long periods. To preserve them for study, the cells are immortalized through a technique using viruses that carry oncogenes, genes that give rise to cancer. One of the qualities of cancer cells is that they can function indefinitely. Cells into which these viruses are introduced can be manipulated in culture so that the oncogenes lose their cancer-inducing capability while still bestowing immortality.
Further manipulation of the culture conditions could induce the stem cells to differentiate into neurons and glia in vitro, but only very inefficiently. By contrast, when they were implanted in the well-studied hippocampus region of brains of newly born rats, they differentiated with great efficiency. The key question,
however, was, Would they display the characteristics of hippocampal neurons? To answer this question, it was necessary to demonstrate that the transplanted cells took on the structure and position of some type of hippocampal neuron. By labeling the stem cells with a radioactive marker prior to transplantation, it was found that the transplanted cells differentiated into the type of neuron called granule cells in the hippocampus. Much is known about these granule cells including their precise connections with other areas of the brain and the fact that they contain specific, identifiable proteins. In addition, they respond in a highly predictable manner to the neurotransmitter glutamate or to compounds that behave like glutamate (called agonists). These responses include up-regulation of a distinctive oncogene and changes in the sensitivity of the cells' electrophysiological responses under specific types of stimulation (called long-term potentiation). The transplanted, differentiated cells were found over a long series of experiments to exhibit all these normal characteristics of hippocampal granule cells.
These findings in animals demonstrate that embryonic neural precursor cells can be preserved in culture for long periods of time yet still be capable of differentiating when placed into a recipient brain. Transplantation of embryonic cells into adult brains raises the possibility of providing a useful therapy for neurode-generative diseases, many of which are intractable and impose terrible personal, social, and economic costs. Experimentally, not only can cultured stem cells integrate with a recipient brain, they can be genetically manipulated before transplantation to allow a more precise analysis of gene function in the normal and diseased state. Moreover, there are data to support the idea that this approach need not be confined to the central nervous system.
Applications of these techniques, however, must await extensive further research including extending the investigations to primates. Peter Medawar, a pioneering contributor to understanding of the immune system, noted that the idea of transplantation always elicits a kind of social taboo, but that the important question is, Does it work? With regard to stem cells the answer appears to be yes.
OPEN DISCUSSION PERIOD
One participant wondered whether the strategy described for genetic manipulation of stem cells could be used to produce disease mutations for investigational purposes. Although there is considerable debate about how to do this, and different approaches are being tried, there seems no reason to presume that overwhelming technical difficulties would prevent such an application.
Another question concerned genetic changes in immortalized cell lines that might influence the neuronal functions studied. The differences among primary cells, immortalized cells, and transformed (cancerous) cells constitute an important issue. Immortalized cells may not turn out to be the appropriate ones for treating disease.
A final question was raised about age limits for the recipients of transplanted stem cells. Although the accepted dogma is that conditions do not exist in the adult brain for neuronal differentiation, ongoing research by a variety of laboratories suggests that the efficiency of differentiation of transplanted cells decreases with age, eventually reaching a very low level. It is an area, nevertheless, that is under active investigation.
Fetal Tissue Transplantation for Patients with Parkinson's Disease
RICHARD ROBBINS
Yale University School of Medicine, New Haven, Connecticut
Upwards of a million people in the United States have Parkinson's disease, a progressively degenerative condition affecting movement. The tremor and motor-control loss that characterize it result from the death of a group of brain cells that produce the neurotransmitter dopamine. What causes the cells to die—whether a virus, an environmental toxin, or another agent—is not known.
Therapy using L-dopa, the precursor of dopamine, alleviates the symptoms of parkinsonism for five to ten years, after which patients frequently develop resistance to it. Other approaches have been tried, including slow-release dopamine implants and transplants of the patient's own adrenal cells, which had to be stopped because of adverse side effects. Experimental work in this disease progressed slowly for decades, because there was no good animal model of the disease. That situation was changed almost 10 years ago by a tragic accident. An illegally produced "designer" drug containing a contaminant, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), was taken by a number of young healthy individuals, who then developed very severe parkinsonism. Once the cause was established and MPTP was shown to produce the same effect in animals, a badly needed animal model was suddenly available. The work of the Yale-Rochester private implantation group at St. Kitt's clearly demonstrated that fetal dopaminergic tissue could innervate MPTP-damaged brain regions and reverse the neurological defects. The idea of transplanting fetal dopaminergic cells into Parkinson's patients grew out of this work.
Thirteen Parkinson's patients have received fetal tissue transplants in Yale University's Neural Transplant Program. Because of the long-standing ban on federal funding of fetal tissue transplantation, the program has been privately funded. The tissue used in the program is collected from elective abortions. The women who consent to its donation are in no way influenced in their decision to terminate pregnancy; the request for donation is made only after the women have already signed consent forms for the abortion. The normal abortion procedure is also not changed in any way. Donors cannot specify who will receive the tissue, no money is exchanged, and no one involved profits financially.
The neurons in the donor tissue, usually from a seven- or eight-week fetus, are still immature, captured soon after they have committed to becoming dopaminergic cells, but before they have fully differentiated. The tissue is microdissected by a neuroanatomist into small blocks that contain supporting glial cells. These cells are believed to secrete growth factors important to the development and differentiation of dopamine neurons. Cryopreservation is used to store the tissue, because about six weeks of testing are needed to ensure that the tissue is free of infectious agents. The dopamine content of the cells is also evaluated. Storage also permits surgery to be scheduled at a time of optimal health of the patients.
When a patient is ready for surgery, an atlas of his or her brain is created by means of magnetic resonance imaging to ensure safety and accuracy in the placement of the implant. The procedure, which takes about six hours, is done under local anesthesia and the patient usually goes home the following day. All of the patients undergo intensive preoperative and follow-up neurological and psychological evaluation. Their status is tracked, along with that of a nonoperated control group, in a ''blind'' manner so that the examiners do not know which patient has received a transplant and which has not. Improvement in motor function generally begins several months after surgery and continues for up to two years.
Although not cured of their disease, these patients perform better than they did under optimal medication before implantation, and they perform still better if they have medication, in reduced dosages, after surgery.
Continuing research is directed toward questions such as the following that are critical to the progress of implantation therapy for Parkinson's disease:
-
What is the optimal age of fetal tissue for implantation?
-
How much of the tissue should be placed—and where?
-
What are the most sensitive noninvasive measurements of graft viability?
-
Which cells in the graft are responsible for improvement?
-
Should growth factors be added to the grafts?
-
How would graft rejection be detected?
-
How much immunosuppression should be used? For how long?
-
Are outcomes among transplant centers comparable?
-
Will there be enough fetal tissue for sufficient trials?
-
Should procedures to collect tissue be modified?
-
Should dopamine cells be genetically engineered for controlled proliferation?
Dopamine neurons arise in a part of the midbrain called the substantia nigra and communicate with the striatum, further forward in the brain, specifically with the caudate and putamen areas. The events in cell differentiation are being studied in cultured substantia nigra neurons and glial cells for insights into why cells die in Parkinson's patients. The effects of growth factors are being examined to see if they can be used to increase survival of dopamine cells in culture.
Tests have been set up to look for dopaminergic toxins or other possible toxic factors in patients' caudate tissue.
A major problem for research and treatment is the difficulty of obtaining healthy fetal tissue. Treating a few patients per year will not make a significant dent in the large population of Parkinson's sufferers. One possible solution is to create an immortalized dopaminergic cell line. Because this may involve viral vectors or oncogenes, methods for preventing the cells from turning cancerous must be ascertained. The cells must also be able to release dopamine in a regulated manner, because too much dopamine is as bad as too little.
Despite its present limitations, fetal tissue transplantation has been shown to restore neurologic function in patients with severe parkinsonism. Research to verify the clinical improvements and to define the factors that are directly responsible for the improvements must proceed. The lifting of the ban on fetal tissue research will hopefully enable research on this disease to go forward more quickly.
Improving Fetal Neuronal Graft Technology Through the Use of Nonhuman Primates to Meet the Needs of Human Therapeutic Applications for Parkinson's Disease
JOHN R. SLADEK, JR.
Neuroscience Institute, Chicago Medical School, Chicago, Illinois
Twenty years of study using rodent models and, more recently, nonhuman primates have demonstrated that grafts of embryonic nerve tissue implanted in the brain alleviate the symptoms of experimentally induced parkinsonism. These studies were based on the elegant work of a number of researchers who had investigated the viability and physiology of transplanted tissues. The development of the primate model of parkinsonism and successful neural grafting, reported in 1985, opened the way to clinical trials with patients with Parkinson's disease. Results to date, although very promising, have pointed up the need for more refined methods of graft tissue selection and preparation to achieve greater graft-cell viability and growth. Continuing animal studies are providing new data that, when used in comparative studies with human cells, should lead to better understanding and treatment.
One of the questions that has been explored recently in studies with rhesus monkeys is the optimal age of fetal donor cells for transplantation. The use of ultrasound in a primate breeding colony has allowed access to embryos at progressive stages of development. Tissues from different stages have been grafted into recipient animals, and samples of the graft retrieved later for examination. Computer programs have been adapted to extrapolate information from a sample to create a three-dimensional reconstruction of the graft and measure the percentage of transplanted cells that survive.
These experiments have yielded a number of valuable results. Very impor-
tantly, the grafts contained all of the cell types normally found in the region of the brain in which dopamine cells arise, and they were filled with growing axons, the cell fibers from which dopamine is released (Figure 5). Most significantly, the studies revealed that there is a very brief window of time during neurogenesis when developing cells are best suited for survival in a graft. Translating that information from the monkey to the human gestational period has not yet been done, in large part because of the prohibition on fetal tissue research.
Dopamine neurons, arising in the substantia nigra region of the midbrain, normally send out their axons across the brain to target cells in the striatum. In both laboratory and clinical trials, grafts have been implanted directly into the striatum. Because the normal system is thus short-circuited, there was uncertainty as to whether factors in the natural environment that are missing in the transplant environment are important to dopamine cell control. To look into this, tissue from both the substantia nigra and the striatum of the same donor embryo were grafted, about 2 mm apart, in a host brain. The nigral cells sent their axons preferentially to the donor striatum. Moreover, some of the damaged brains appeared to regenerate in a pattern similar to normal development in response to the graft.
Other co-graft experiments showed for the first time that long-distance targeting, from one side of the brain to the other, could be effected. This has suggested that growth-factor signals in the embryonic striatum are so strong that they might be used as stepping stones by growing axons. By grafting dopamine cells into the host substantia nigra, their natural location, and placing growth-factor packets along the route to the striatum, it may be possible to reconstruct the natural pathway.
Data from animal and human cells need to be compared. Experiments grafting human cells into nonhuman primates are necessary to compare long-distance cell targeting and circuit reconstruction. The optimal human donor-cell age needs to be determined, as does the optimal age for a graft recipient. Experiments in aged monkeys are necessary to learn when best to intervene in the course of disease, a critical question. For example, work from our laboratory suggests that L-dopa, especially long-term therapy, may itself reduce the effectiveness of dopamine released from the graft. The comparative survival in grafts of cryopreserved tissue, freshly dissected tissue, and short-term cultured cells also needs to be assessed. Resolving these issues is essential to providing graft-cell yield that can best compensate for the progressive loss of dopamine neurons in Parkinson's disease.
OPEN DISCUSSION PERIOD
The comments and questions that followed these presentations underscored how much remains to be learned about fetal-cell therapy for Parkinson's disease. One participant asked if there was any evidence in patients in the Yale trials as to whether the effects of transplants are due to the formation of axonal connections
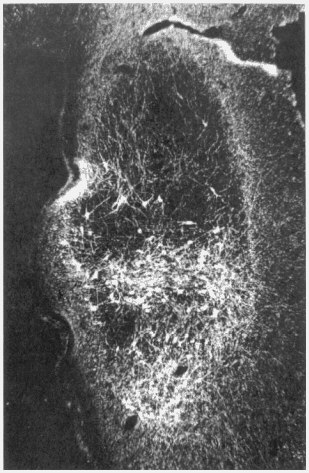
Figure 5
Transplantation of embryonic neural tissue can result in the survival of thousands of neurons of a specific transmitter type as illustrated in this photomicrograph. Selection of tissue samples from the optimal stage of neural development has yielded over 22,000 surviving dopamine neurons following grafting into a nonhuman primate model of Parkinson's disease. This represents about one-third the normal adult complement of dopamine neurons derived from the mesencephalon, a critical brain region in Parkinson's disease. These neurons also were characterized by the presence of extensive outgrowth of axons and dendrites into the host brain, which suggests the opportunity for functional integration between grafted and host tissue. SOURCE: Sladek, J.R., Redmond, D.E., Roth, R.H., et al. "Fetal Dopamine Cell Survival After Transplantation Is Dramatically Improved at a Critical Donor Gestational Age in Non-Human Primates," Experimental Neurology, 122:22, 1993. Reprinted, with permission, from Academic Press, Inc.
or to diffuse release of dopamine. The answer is not known. It may be that the glial cells implanted with dopamine neurons make growth factors, which in turn encourage other dopamine neuron systems to grow. The implant may also increase sensitivity to L-dopa, among other possibilities. Another question concerned how much fetal material is needed for a human transplant. The amount currently used is about equivalent to two fetal midbrains, but fetuses are rarely retrieved intact, so only a fragment of midbrain is usually available.
Among questions generated by the presentation of animal research data was whether adult host cells would form reciprocal connections with fetal cells grafted into the midbrain. The question of neuron-target interactions will require detailed experimental reconstruction and analysis at both ends of the system. A participant asked if there was any evidence that co-grafting provides better recovery. Although the monkey studies are still in the process of analysis, rodent models appear to improve more rapidly after co-grafting.
Information was requested about human transplantation therapy programs and results worldwide. In addition to the Yale program and one at the University of Colorado in this country, there are programs in Sweden, Cuba, England, and Poland. During the fetal tissue research ban in the United States, an American neurosurgeon took patients to China for implantation. Specific data on results are beginning to be collected. Patients in a joint American and Swedish program have been described as showing the most marked improvement. Interestingly in terms of recipient age, however, these patients are all young people who developed drug-induced parkinsonism from use of MPTP. It is important to remember the differing etiologies and the fact that MPTP-induced parkinsonism is not progressive.
Forces That Mold the Antibody Repertoire: Lessons from Fetal Lymphocytes
ROGER PERLMUTTER
University of Washington, Seattle, Washington
The fundamental property of the immune system is its ability to recognize the entire universe of potentially pathogenic foreign invaders, such as viruses and bacteria. Recognition is achieved by a virtually unlimited repertoire of receptor molecules on white blood cells, or lymphocytes. Lymphocytes express specific receptors in response to specific molecules on invaders (antigens) that mark them as foreign. This process also accounts for rejection of grafts of nonself tissue. Remarkably, immune receptors are encoded by a relatively small number of genes. They achieve their diversity through a complex feat of genetic recombination.
Lymphocyte receptors are formed from pairs of protein chains. Each chain contains a constant region and a variable region. The variable regions are encoded in gene segments capable of vast numbers of recombinatorial rearrangements.
Superimposed on this system are other processes that enhance receptor flexibility. The system's extreme heterogeneity has made it difficult to study the way in which the immune repertoire is generated. Studies of fetal lymphocytes, conducted over several decades, have provided a useful entrée into the system.
It would be logical to assume that, because their gene rearrangements are random, lymphocyte types arise randomly, but in fact, they develop at specific times during an animal's life. This process has been studied in greatest detail in the mouse, where a whole hierarchy exists for the acquisition of lymphocyte receptors that bind particular antigens. To investigate this process in human development, the fetal liver, which contains arising B lymphocytes, has been examined at different stages of growth.
Antibodies are released by B lymphocytes and function as immune receptors dispatched by the cells into the body's fluids. Research has revealed that of the 200 variable-region gene segments that function in the adult, only about 12 or 13 are active in the fetus. Studies of developing T lymphocytes have shown that the organization of their receptor genes looks very much like the pattern of limited diversity and characteristic developmental order seen in antibodies.
Interestingly, fetal antibody gene segments are very similar in humans and mice. At the level of the nucleotide (the building block of DNA) sequences, the segments are 92 percent similar. This close relationship has facilitated comparative research to begin to determine what is so special about the variable gene segments that are expressed early in life. One thing that has been discovered about them is that they will bind self-components, something ordinarily prohibited in immune response. Immune tolerance to self-antigens is essential to survival. Fetal antibodies include many that appear identical to antibodies responsible for autoimmune disease, such as rheumatoid arthritis, in which immune cells attack other cells of their own body.
What is emerging from this research is confirmation that a tremendous amount can be learned by starting with a relatively limited repertoire as a foundation on which to build understanding of the complex progression that leads to the immensely larger immune receptor repertoire. It is important, however, to examine these issues in human fetal lymphocytes.
Isolation of Candidate Human Stem Cells Using SCID-hu Mice Implanted with Human Fetal Tissue
IRVING WEISSMAN1
Stanford University School of Medicine, Stanford, California
Hematopoiesis is the process through which all the different types of blood cells are formed, from red blood cells, to T and B lymphocytes, to a number of different types of so-called white blood cells. Given the multitude of human
diseases that affect these cells, it is understandable that this process is the subject of intense study. All the blood cells formed in hematopoiesis derive from primitive stem cells, and by definition, a stem cell must have two properties: the ability to give rise to every kind of blood cell and to more stem cells.
A search of several years' duration was conducted to isolate true stem cells—cells not yet committed to a particular destiny—in the bone marrow cell populations of experimental mice. Sorting methods involved the use of monoclonal antibodies that, when fully catalogued and mapped, can home in on specific types of cells in a mix by recognition of distinct cell-surface antigens (markers). The search showed only 1 in every 2,000 marrow cells to be free of markers that would indicate they were type-committed.
To make sure that the marker-free cells had full stem-cell potential, they were isolated and injected into mice that had been irradiated to kill their existing marrow cells. When such lethally irradiated mice are injected with unsorted donor marrow, it takes about 200,000 cells to save them; it required only 85 to 100 of the isolated cells to do the same job.
Further experiments showed that a single stem cell could give rise to 100 million blood cells of all types, including many hundreds or thousands of new stem cells. When progeny stem cells from the original cell were collected from the first recipient mouse and subsequently injected into irradiated animals, all of the animals' marrow became fully reconstituted. This finding also meant that one could now study the full developmental repertoire of a single stem cell.
Growth factors have important effects on both primitive and mature blood cells. One very useful effect is to cause stem cells to migrate from the bone marrow into the peripheral blood. By applying these factors, stem cells can then be harvested from the peripheral blood for clinical use—a procedure vastly more simple than harvesting from bone marrow. Thus, this procedure will undoubtedly provide the most enriched and utilized source for marrow or hematopoietic transplants in the future.
T lymphocytes arise in the thymus and move to target organs such as the skin and other epithelial areas of the body where they serve specific functions. The order of appearance of these targeted T cells in the fetus follows a developmental timetable. Experiments have shown that the ability to make skin-homing cells, for example, switches on and off only during a specific and limited time window after which the production of skin-homing cells stops. It has also been shown that if an adult stem cell is inserted in the fetus, it will give rise only to adult-programmed types of T cells. These findings suggest the significance of the age of the donor stem cells for any future fetal therapy and imply that donor cells should be the same age as the recipient.
The study of human hematopoietic stem cells began by harvesting cells from human fetal livers (like bone marrow, a rich source of such cells in the fetus) and growing them in culture to identify the different types. Stem cells were found that could generate all the necessary blood cell types in culture, but it
was unknown what would happen in vivo. Although the needed experiments could not be done in humans, an invaluable alternative was found in a mutant mouse strain that exhibits severe combined immunodeficiency (SCID). SCID mice cannot make T cells or B cells and are, therefore, tolerant of foreign tissue. Human fetal thymic grafts implanted in SCID mice produce human T cells tolerant of both the mouse and their own human tissue, but fully reactive to other tissue. These grafts, as well as transplanted human fetal bone marrow, will reconstitute all the human blood cell types.
From a clinical standpoint, the development of the SCID-human (SCID-hu) model and the study of human stem cells make a number of strategies possible. For example, it has been found that stem cells from a human being with a certain type of leukemia, which lack the genetic change associated with leukemia, give rise to normal, healthy blood cells. Purified stem cells could then be harvested from a patient with leukemia and used to repopulate the bone marrow after the diseased cells were destroyed by irradiation. Other cancers that may be responsive to such treatment include myeloma and lymphoma. Some diseases that affect blood cells are inherited, such as severe combined immunodeficiency disease and thalassemia. If stem cells from the siblings of affected patients could be isolated, they could then be transplanted successfully because such cells do not give rise to graft-versus-host disease. In cases where suitable sibling cells are not available, it may be possible in the future to put corrective genes directly into the patient's own stem cells. A potential that has aroused great excitement is stem-cell treatment for AIDS.
Human immunodeficiency virus (HIV) attacks and cripples the immune system. Its port of entry is a subset of T cells designated CD4. A number of studies indicate that HIV does not penetrate stem cells. If that is true, it may be possible to insert into the stem cells of AIDS patients genes that encode mechanisms to stop viral replication or the pathogenic effects of HIV on CD4 cells. Attempts to make such gene constructs have begun and the SCID-hu model is valuable in these efforts also.
In the process of mapping thymic development in the SCID-hu mouse, it was discovered that the very first progeny cell in the human thymus (unlike the mouse) is the CD4 cell. HIV injected into the SCID-hu mouse infects these human CD4 cells, which means that SCID-hu can be used as a model to study viral infection. SCID-hu can also be used to study the development of many different tissue types. For example, fetal lungs or bone can be implanted successfully in SCID mice and provide cells for study. In addition, to study genetic diseases it may be possible to implant fetal tissue that is known through early diagnostic testing to be affected by some type of genetic disease.
Another stem-cell potential derives from a phenomenon that has been known for a long time: namely, that in an irradiated animal in which bone marrow has been replaced with donor marrow, any organ can be grafted from that same donor and not be rejected by the host. In recent experiments studying how this
might affect autoimmune diseases, stem cells from a strain of mice that do not get diabetes were implanted into mice that invariably develop diabetes. Stem-cell implants were done, after lethal or sublethal irradiation, at a time when the recipients' lymphocytes were collecting in the pancreas, indicating the onset of autoimmune attack that would eventually destroy the insulin-producing pancreatic cells. The transplanted mice never developed diabetes.
Xenogeneic Transplantation: The Use of Animals for Organ Donors
DENISE FAUSTMAN
Massachusetts General Hospital, Charlestown, Massachusetts
There is a long and growing list of human disorders for which cell or organ transplantation promises more effective therapy. The problem is that the human tissue available for transplant, now or in the future, cannot begin to meet the need. Removing the barriers to xenogeneic (cross-species) transplantation could open the way to organ banks with an unlimited supply of replacement cells and organs.
Xenogeneic tissue has advantages and disadvantages compared to human tissue—some obvious, some less obvious, some potential. Among the advantages, in addition to being plentiful, animal donors often lack, or could be genetically modified to lose, susceptibility to human disabilities or pathogens, such as HIV. The disadvantages include differences in size and structure, as well as critical metabolic incompatibilities that must be avoided or compensated for. Animal studies have shown, for example, that although the ability to secrete insulin is intrinsic to all pancreatic islet cells, the cells regulate blood sugar differently across species. Thus, a diabetic human given goat islet cells could possibly go into hypoglycemic coma, because the goat cells respond to glucose levels appropriate for that species and secrete more insulin than is appropriate for humans.
For liver transplants, there are also hemodynamic problems. In the recent experiment in which a baboon liver was implanted in a human subject, the baboon was chosen because its cardiac output is similar to that of humans and it is resistant to AIDS (from which the patient was suffering), but the clotting factors produced by the baboon liver differed significantly and resulted in severe clotting abnormalities in the recipient.
The primary barrier to the clinical reality of xenografts is that the recipient mounts a much more pronounced rejection response to nonhuman donor tissues. The strategy currently used in all clinical transplant protocols to circumvent immunogenic graft rejection is immunosuppression. Although the graft may take, the patient, stripped of immune defenses, succumbs easily to massive infection. A solution to this problem may lie in research currently under way, based on new information about the molecular mechanisms that regulate the T lymphocytes, especially those known as killer cells that attack tissue transplants.
A killer T cell attacks a foreign graft in a series of steps that begin when the T cell recognizes and sticks to a binding site on the target cell. The binding triggers a cascade of events, including activation of the T cell's receptor, that culminate in the T cells' drilling a hole in the target cell and killing it. In an experimental approach that works in reverse of immunosuppression, instead of inactivating T cells the donor tissue is altered in such a way that its surface antigens are hidden from the recipient's T cells. This has been done by taking antibodies to the key antigen of the recipient cells and separating the binding part of the antibody from its "handle," or the part to which T killer cells stick in the initiation step of donor cell destruction. These separated antibody fragments were shown in tissue culture experiments, and eventually in transplantation experiments, to mask the antigenic sites of the donor tissue and protect it from the host's immune system.
In the initial experiments, monoclonal antibodies were used to engage the donor cell binding sites, thereby making them unavailable for binding by T cells. Human islet cells, so disguised, were implanted in mice and survived up to 200 days without T cell attack. In subsequent experiments, another approach was tried, in which the genes for the donor cell binding sites were inactivated or knocked out. With both masking and knockout strategies, researchers have now created masked donor heart, liver, kidney, and insulin-secreting cells. There are no clear answers yet as to whether surface masking or genetic engineering protects donor tissue in the long-term. There is renewal with time of the antigenic cell-surface proteins that are not masked and are therefore vulnerable, although this renewal seems slower than normal. However, there are also hints in some tissue types, possibly due to slow renewal, that the recipient's immune system may become tolerant to these cell-surface markers.
Many barriers to xenogeneic grafts must still be overcome, but these experiments have shown that it is possible to design new methods to manipulate cells. Xenogeneic transplant therapy offers the chance to enhance and broaden treatment options for many diseases and, in some cases, to provide permanent cures. We may one day be treating problems of cholesterol metabolism and hemophilias with xenogeneic liver cells or bone marrow stem cells, diabetes with xenogeneic insulin-secreting cells, and cardiac disease with pig heart transplantation.
















