1
The Changing Nature of Medical Technology Development
ANNETINE C. GELIJNS AND NATHAN ROSENBERG
It is axiomatic in recent discussions of health care in the United States that its quality and cost have been transformed almost beyond recognition in the years since the end of World War II. A fundamental component of that transformation has been a rapid rate of technological change that has led to the insertion into the health care system of a range of new diagnostic and therapeutic technologies that had no real antecedent in prewar years. The recognition that medical technology is a critical element in the quality as well as the cost of health care has renewed interest in the factors shaping technological change or innovation in medicine. At a more general level, one can observe that medical innovation is stimulated by advances in scientific and engineering knowledge, on the one hand, and by the potential demand for health-improving technology, on the other. The health care reform debate, however, has been dominated by a preoccupation with demand-side factors. In particular, discussions have focused on how changes in demand-side incentives, as precipitated, for instance, by the introduction of global budgets, higher insurance co-payments, and the like, might reduce the cost-ineffective use of existing medical interventions and alter the direction of medical innovation toward the development of cost-reducing technologies. By comparison, the conditions governing the supply of new medical technologies, and their particular characteristics, have been relatively neglected. It is this lacuna that the present volume attempts to address.
At the outset, we need to recognize that policy discussions of technological change have often been impoverished by the notion that innovation is a more or less homogeneous activity, as is implied by the "linear model." The truth, of course, is that innovation is not one thing; it is many things. The processes by
which new technologies are generated differ enormously from one sector of the economy to another. Nothing is more likely to muddy the waters of understanding, and therefore muddy the policymaking process as well, than the notion that we are dealing with a uniform set of relationships when we move from one sector of the economy to the next, carrying with us the assumption that "one size fits all." The beginning of wisdom is the recognition of the extreme diversity of background conditions underlying the innovation process. Arrangements that may work well in semiconductors or the aircraft industry may not work well at all in pharmaceuticals. Indeed, what has worked well in pharmaceuticals may not work at all in the newly emerging biotechnology sector. Moreover, if we consider new clinical procedures or medical devices, the conditions for successful innovation are probably very different from other subsectors of the medical technology realm. Even within these subsectors, there is considerable diversity in this respect. Consider, for example, medical devices. Medical devices encompass a heterogeneous group of products, ranging from low tech, inexpensive devices such as tongue depressors and disposable needles, to sophisticated and expensive modalities such as lithotripters and magnetic resonance imaging (MRI) machines. Obviously, the research required for the invention of, for example, disposable needles is quite different from that required for the invention of the computerized tomography (CT) scanner; the heterogeneity in products, and their research and development (R&D) strategies, is reflected in the manufacturers that produce these products.
If we focus on the conditions governing the supply of new medical technologies, two characteristics emerge that merit specific attention in this respect. First, evidence suggests that medical innovation is becoming increasingly dependent on interdisciplinary research.1 That is, the successful development of a particular technology frequently requires close cooperation among a growing number of individuals with diverse but relevant professional backgrounds. In the case of pharmaceuticals and biologicals, for example, the development of a drug may require cooperation among organic chemists, molecular biologists, immunologists, material scientists, toxicologists, chemical engineers, clinicians, and so on. In the case of medical devices, the interdisciplinary nature of innovation appears even more obvious. The development of devices generally depends on the transfer of scientific and technological advances outside of medicine (e.g., in physics, engineering, and their relevant subfields, such as micro-electronics, materials science, optics, etc.) into medicine. It thus requires the interaction of physicists
and engineers with medical specialists. Moreover, medical innovation may require the involvement of a variety of medical specialties. For example, the evolution of cardiopulmonary bypass was dependent upon a range of professions, including, among others, cardiac surgeons, cardiologists, physiologists, anesthesiologists, and hematologists.
Second, innovation requires not only the crossing of disciplinary boundaries, but it also increasingly involves the crossing of institutional boundaries. In fact, medical innovation depends heavily on interactions between universities, particularly academic medical centers, and industrial firms. Over the past decade, the number and diversity of these university-industry interactions have greatly increased (Blumenthal, 1994).
A review of the literature reveals that there is little insight into how these characteristics affect the efficiency with which research findings are translated into clinical practice. The objective of the present volume therefore is to examine how the interdisciplinary and interinstitutional nature of current medical R&D shapes the rate and direction of medical innovation. In doing so, it focuses particularly on the medical device and biotechnology sector.
ORGANIZATION OF THE PRESENT VOLUME
This volume starts out with a discussion of the increase in public and private spending on medical R&D in the United States. As Bond and Glynn (chapter 2) observe, the decades following World War II have been characterized by an impressive growth in federal funding for biomedical research and medical education. This investment vastly strengthened the position of academic medical centers and stimulated the education of a growing cadre of specialists and subspecialists—defining, and redefining, disciplinary boundaries within medicine. Moreover, in recent years, the government has begun more actively to encourage universities to work with manufacturers to stimulate the transfer of their research findings into medical practice. These efforts have led to a marked increase in university-industry interactions.
The core of the volume then examines how the interdisciplinary and interinstitutional nature of medical R&D has shaped the rate and direction of innovation in several major areas of medical intervention (viz., lasers, minimally invasive surgery, cochlear implants, cardiovascular diagnostics, the development of synthetic insulin, and biotechnology agents in general). These analyses generally take a historical approach to the subject. Innovation, by definition, is not only a process that occurs over time; it is, at bottom, a learning process. Research and development activities are conducted in an environment of limited or, at the least, insufficient information. At the outset, the uncertainty level is high. The learning process of innovation is one in which information is accumulated and uncertainties reduced, commonly over the course of extended time periods. Therefore, if we want to explain why a particular innovation occurred when it did, it is
misleading to refer to the time that elapsed between a scientific breakthrough and an eventual application as a "lag" (Rosenberg, 1982, particularly chapter 3). Galileo was not the inventor of the pendulum clock, even though he did indeed formulate the scientific principles that govern the behavior of the pendulum clock. Similarly, the basic science underlying light transmission was developed by Christiaan Huygens in the seventeenth century, but it would be preposterous to regard him as the inventor of fiber optics. Huygens's scientific breakthrough, a formidable intellectual advance, tells us nothing about why optical fibers were introduced into endoscopy in the 1950s and 1960s rather than the 1850s and 1860s. Other factors need to be invoked to provide the explanation. Those factors would include drastic improvements in the quality, and manufacturing, of optical glass, as well as essential activities at the levels of applied physics and engineering (see Gelijns and Rosenberg, chapter 4, this volume).
The nonhistorical view of innovation, which "knows how the story ends," continually ignores the uncertainties that prevailed at an earlier date, and assumes that important information was already available long before it was in fact acquired, that is, learned (Rosenberg, 1994b). This is partly because most information, once it has been acquired, appears "obvious." The heliocentric view of the universe is, to us, obvious, but it required many centuries of patient observation and cogitation, as well as the availability of more sophisticated instrumentation, before it became obvious. By contrast, the historical approach—by asking about the innovators "what did they know and when did they know it?"—provides information that is indispensable to a full appreciation of the complexity, the creativity, and the time-consuming nature of the innovation process.
Following the analyses of particular areas of medical innovation in chapters 3 through 8, the volume then provides two perspectives on the management of interdisciplinary and interinstitutional R&D. Laubach (chapter 9) discusses how today's rapidly changing health care environment affects the developer-user interface, and the particular challenges this raises for industrial R&D management. Nelson (chapter 10) briefly examines the intertwining of science and technology in medicine. He raises fundamental questions about what is public and what is private in medicine, and what should be patentable and what should not be patentable in this sector. How we address these questions will have profound implications for the transfer of research findings from academia to industry, as well as for the ease of information exchange within the medical profession.
In the remainder of this introductory chapter, we will address some of the findings of the present volume, and highlight the needs for future research in this area.
IMPORTANCE OF UNIVERSITY-INDUSTRY INTERACTIONS
Medical innovation is a particularly interesting area for examining university-industry interactions in the development and commercialization of technology.
Why is this so? One reason is that innovation in this sector provides a rich set of interactions that appear to transcend what might be regarded as the "normal" division of labor between academia and industrial firms.2 Of course, medical schools are involved in basic biomedical research, and the fruits of their efforts may contribute to the development of new technologies by industrial laboratories. The discovery of recombinant deoxyribonucleic acid (DNA) technology by Boyer and Cohen at the University of California, San Francisco, and Stanford University, for example, directly encouraged the industrial development of such biologicals as human insulin, the growth hormones, and tissue plasminogen activator (see Stern, chapter 7). Medical device innovation, by contrast, does not depend nearly as heavily on the exploitation of basic scientific and technological capabilities generated "upstream" within medical schools as does the pharmaceutical or the emerging biotechnology sector. Instead, it relies heavily on the transfer of technological capabilities already generated outside of the medical sector—and indeed more commonly generated in the industrial world rather than the academic world.
Consider, for example, the lithotripter. The concept of using shock waves for medical purposes emerged out of research at the German aircraft manufacturer Dornier, where physicists were investigating the causes of pitting seen on the surface of spacecraft and supersonic airplanes (Gelijns, 1991). They found that a craft's collision with micrometeorites or raindrops at very high speeds created shock waves that had a destructive effect on surface structures. They also accidentally discovered that such shock waves could travel through the human body without harming tissue, but that brittle materials in the body were destroyed. These findings led Dornier, in collaboration with urologists at the University of Munich, to develop the first lithotripter for the treatment of kidney stones.
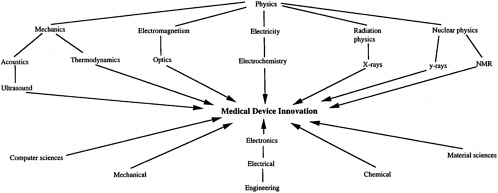
We have deliberately cited an instance of a major new medical technology that had its origin in the aircraft industry in order to highlight how the medical device sector differs from pharmaceuticals and biotechnology. The general point is that this sector looks far outside the realm of biomedical research for new technological capabilities. If one were to look only at the most important contributions to diagnostic technology, one would find that the CT scanner drew heavily on advances in computers and mathematics, ultrasound had its origins in submarine warfare, and MRI originated in the work of experimental physicists exploring the structure of the atom. Thus, medical device innovation is not only inherently interdisciplinary but also outward-looking by nature, and depends on the transfer into medicine of advances in such diverse fields as electronics, optics, computers, and the material sciences (see Figure 1-1).
Whereas, in the context of medical device innovation, medical schools may
not have been the dominant source of basic scientific and technological knowledge, they do nevertheless play a major and multifaceted role in the development of new medical devices (the ''D" of R&D). In academic medical centers, it has been the clinicians rather than basic medical researchers who have most commonly been crucial to the invention of medical devices. They not only identify the clinical need for a new device or for improvements in existing devices but, because of their role as eventual users, they may also be the innovators and builders of the original prototype. Von Hippel and Finkelstein (1979), for example, underlined the importance of these users with regard to the invention of the automated clinical chemical analyzer. The chapters in this volume on endoscopes and cochlear implants (chapters 4 and 5) present similar findings. An exception may be found in the case of nuclear cardiology and echocardiography, where the complexity and multitude of engineering skills required for prototype development were not easily carried out in a clinical research setting (see Finkelstein, Neels, and Bell, chapter 6).
In addition, this volume reveals that in certain cases clinicians may be involved in the manufacturing process. For example, the chapter on minimally invasive surgery (chapter 4) illustrates that the academic/medical community not only developed the first prototype of a flexible gastroenterological endoscope, but that they were also instrumental in teaching the industrial firm how to solve some complicated manufacturing problems—the making of the fiber-optic bundles. This is, to say the least, a drastic departure from what might be regarded as the "normal" division of labor between academics and private industry.
Finally, the academic medical profession plays a critical role during the clinical development, and refinement, process of new medical technology. The medical profession is instrumental and indispensable in evaluating the clinical potential of new devices and drugs during the premarketing development period. As might be expected, feedback at this point may lead to important changes in the design of these products. The development process does not, however, end with the more widespread introduction of a new product into practice. New technologies, in medicine as elsewhere, typically enter the world in a primitive condition. In fact, as we will see, important shortcomings may emerge only after extensive use in practice. Once identified, such shortcomings provide the basis for further modification and improvement. This is illustrated by the history of oral contraceptives. The widespread introduction of the first oral contraceptives into clinical practice during the early 1960s confirmed their high degree of effectiveness, but also revealed that their use increased the risk for thromboembolic disorders. The suspicion, based upon subsequent research, that estrogen might be responsible for such circulatory diseases stimulated manufacturers to reduce estrogen levels and develop low-dose pills, which subsequently led to a dramatic decline in side effects (Gelijns and Rosenberg, 1994). Thus, actual adoption constitutes only the beginning of an often prolonged process in which important redesigning takes place, exploiting the feedback of new information generated by
users. This incremental improvement process occurs not only in industrial laboratories, but also within clinical practice itself. For example, after new medical devices and biologicals are introduced into clinical practice, the medical profession often discovers entirely new indications of use. A case in point is the laser (see Spetz, chapter 3). Originally introduced for use in ophthalmology and dermatology, it is now being used for a wide variety of indications in oncology, thoracic surgery, and gynecology—to name but a few. Thus, there is an important feedback loop by which improvements, as well as new indications, are extracted from the accumulation of clinical experience with novel instruments and biologicals.
In medicine, therefore, it is of the utmost importance for industrial firms to create close interactions with clinicians, often in academic medical centers. At the same time, clinicians have strong incentives to be involved in the development of new technologies. New technologies, of course, are vital for improving clinical practice. In addition, their use may confer prestige and provide professional recognition in the form of publications. Moreover, the internal dynamics of the medical profession—and the hierarchical scientific status of different subspecialties—is such that innovation is a means for enhancing the professional status of particular medical disciplines. Thus, the intertwining of interests of academic medical specialists and segments of the manufacturing industry tends to encourage an ongoing process of innovation. Blume has argued that we should regard this innovation process as taking place not in a market (in which buyers' and sellers' interests are assumed to be opposed), but rather in an interorganizational field (Blume, 1992).
An extremely interesting finding has been the variety of ways in which these university-industry interactions are accomplished, and indeed the extent to which the organization of activities at the university-industry interface has recently been changing in order to accommodate the needs of the innovation process. Traditionally, interorganizational links have hinged strongly on the movement of personnel between academic and industrial settings, which can range from the hiring of graduate students by industrial firms to consultancy agreements with faculty (Klein, 1990; see also chapter 9, this volume). Nowadays these arrangements are still critically important, but new ways of crossing disciplinary and organizational boundaries are becoming apparent. In the medical device sector, for example, the composition of the industrial R&D team is undergoing significant change in that clinicians are being added to these teams. At the same time, product design engineers, as well as marketing specialists, are receiving more clinical training and are spending more time in academic medical centers.
Another indicator of university-industry interactions can be found in the number, and growth, of licensing agreements. The Boyer-Cohen discovery, for example, provided Stanford with a very profitable patent (chapter 9). Over the past decade, the number of patents that universities have filed in the medical sciences has grown markedly, as well as the number of licensing agreements with
industrial firms. Another mechanism for university-industry interactions can be found in research contracts and research agreements. A 12-year, $40 million research agreement, for instance, was recently signed between Harvard Medical School and the Monsanto Company for the development of new interventions for managing cancer (Atkinson, 1994). Such large-scale research agreements are rather unusual. A more common practice these days is that faculty members, and sometimes their parent universities, become involved in the formation of new, start-up companies.
In this respect, small firms can be regarded as transfer mechanisms between traditional university and industrial settings. Small and large companies may play very different roles in the innovation process. In the medical device sector, for example, it has been suggested that small companies—with some notable exceptions—tend to produce the majority of innovations in the early stages of developing a new class of devices, whereas large companies may be more prominent at later stages in the development process (Gelijns and Rosenberg, 1994). Differences in the primary function of small and large device firms, however, should not conceal the complex patterns of relationships that have emerged between the two. Large firms may sponsor research in small firms, often with the intent of acquiring them if their technologies are promising or successful. Furthermore, because recent changes in U.S. payment and regulatory policies have made venture capital funding more difficult, many small firms are seeking alliances with large firms to engage in joint product development, clinical testing, manufacturing, and marketing. Arora and Gambardella (chapter 8) discuss the division of innovative labor in the biotechnology sector and the changing patterns of collaborative alliances between universities, new biotechnology firms, and large pharmaceutical firms.
INTERDISCIPLINARY NATURE OF MEDICAL R&D
We have emphasized the importance of crossing organizational boundaries in the innovation process. In our discussion so far, we have had in mind enterprises such as university research conducted within well-defined academic disciplines on the one hand, and research conducted by industrial firms, large or small, on the other. But there are other boundaries—within these institutions—that may impede the interdisciplinary cooperation and communication necessary for much of today's medical innovation.
In industrial settings, product-related R&D is inevitably interdisciplinary in nature and raises no special challenge to the values and priorities of the organization. Industrial firms are, however, organized into various departments, such as research, product development, manufacturing, and marketing. Management problems have been noted regarding communication and collaboration across these functional activities. For example, Humphrey and Schwartz, at the inaugural meeting of the American Institute for Medical and Biological Engineering in
1992, observed that failure to integrate engineering and manufacturing know-how into the development process of biotechnology drugs prior to phase III clinical trials had locked many final bioprocess designs into nonoptimal processes that did not use leading-edge technology (Humphrey, 1992; Schwartz, 1992).
In the case of academic settings, interdisciplinary R&D may raise some management issues similar to those confronting industrial R&D. Moreover, it may also challenge the basic values and culture of the organization, especially with regard to the pursuit of practical applications of new findings, professional reward structures, and pathways to career advancement. There is little doubt that the organization of the scientific enterprise into disciplines and academic departments has played a major role in the historic success of universities in contributing to the growth of knowledge. Nevertheless, health care problems do not always fall neatly within the boundaries of particular academic disciplines and departments.
Cochlear implants, for example, are essentially interdisciplinary in nature. This has meant that a complex network of different medical specialties were involved in its development. In chapter 5, Blume shows how different patterns of collaboration on cochlear implants evolved, and how these various groups differed considerably in their R&D strategies. In particular, a distinction can be drawn between the more "therapeutically" and the more "experimentally" oriented groups. The therapeutically oriented groups tended to move faster from animal testing to the clinic and collaborated more intensively with industrial firms, whereas the experimental groups entered more slowly into licensing agreements. Blume suggests that the interdisciplinary nature of cochlear implants meant that assembling the necessary skills and competences, securing agreement as to how the value of this new intervention was to be assessed, and establishing what kind of industrial expertise provided commercial advantage was more problematic than in the case of, for example, diagnostic imaging, which is much less interdisciplinary in nature.
The laser and endoscopy chapters (chapters 3 and 4) tell similar stories. The transfer of laparoscopy from gynecology to general surgery, for example, appears to have taken an unnecessarily long time. The authors conclude that "the existing disciplinary boundaries internal to medicine itself may have constituted an even more serious obstacle to innovation than those external to medicine. Put somewhat differently, the most difficult barriers that needed to be crossed were not between medicine and industry, but barriers within medicine itself (p. 93)." Medical schools and university teaching hospitals are increasingly recognizing that, to realize the potential of future medical innovation, they may need to build more sturdy bridges between traditional disciplines.
SOME CONCLUDING OBSERVATIONS
The chapters in this volume have, taken together, opened up a number of
windows onto a complex and uneven terrain where new medical technologies have emerged. But the individual studies also emphasize that acquiring these technologies is not so much like geographic discovery, where one identifies the features of a terrain that has already been shaped by natural forces; rather, it is more like a creative synthesis, in which elements are taken from very different contexts and then combined in a unique way to achieve some specific improvement in medical care.
Medical device innovation appears to differ considerably from innovation in the realm of pharmacology and biotechnology. Although pharmaceutical and biotechnology innovation also draw upon a number of disciplinary specialties, medical device innovation relies much more heavily upon a systematic intellectual trespassing across well-established disciplinary boundaries. Device innovation, as our studies show, not only draws upon a number of disciplines. It also commonly draws upon technological capabilities that have already been developed elsewhere to serve very different human purposes (e.g., lasers, optical fibers, ultrasound, computers), and it reconfigures and reshapes them to serve very different—that is, medical—purposes.
Future research into the determinants of technological change in medicine must probe more deeply, not only into the problems of communications across disciplinary boundaries (including, increasingly, across the boundaries among medical subspecialties). It also needs to examine the conditions for more effective cooperation across institutional boundaries, including, importantly, the impact of federal regulation upon innovation. In considerable measure, this volume identifies those boundaries as lying at the interface between universities and industry. But, in a larger sense, that interface also corresponds to the boundary between the public and private sectors, between sectors that differ drastically in their sources of finance, their incentives, their responsibilities, and their priorities. These studies also suggest that these boundaries have not been rigid but have been redrawn and reshaped, at least occasionally and in certain specific locations. Moreover, as Laubach's chapter suggests, the recent proposals for health reform on the one hand, and the ongoing transformation of the health care delivery system on the other, are also rapidly transforming the environment within which medical innovation must take place. How these transformations are likely to affect both the rate and the direction of medical innovation needs to become a central concern of both the research community and the policymaker.
REFERENCES
Atkinson, S. H. 1994. University-affiliated venture capital funds. Health Affairs 13:159–175.
Blume, S. S. 1992. Insight and Industry: On the Dynamics of Technological Change. Cambridge, Mass.: Massachusetts Institute of Technology Press.
Blumenthal, D. 1994. Growing pains for new academic/industry relationships. Health Affairs 13:176–193.
Gelijns, A. C. 1991. Innovation in Clinical Practice: The Dynamics of Medical Technology Development. Washington, D.C.: National Academy Press.
Gelijns, A. C., and Rosenberg, N. 1994. The dynamics of technological change in medicine. Health Affairs 13:28–46.
Humphrey, A. 1992. Challenges to biotechnology in meeting America's future health needs. Presentation at the inaugural meeting of the American Institute of Medical and Biological Engineering, Washington, D.C.
Klein, J. T. 1990. Innovation and change in organizational relationships: Interdisciplinary contexts. R&D Management 20:97–102.
Reiser, S. J., and Anbar, M. eds. 1984. Machine at the Bedside: Strategies for Using Technology in Patient Care. New York, N.Y.: Cambridge University Press.
Rosenberg, N. 1982. Inside the Black Box. New York, N.Y.: Cambridge University Press.
Rosenberg, N. 1994a. Exploring the Black Box. New York, N.Y.: Cambridge University Press.
Rosenberg, N. 1994b. Uncertainty and technical change. Unpublished manuscript. Palo Alto, Calif.: Stanford University.
Schwartz, J. 1992. The production of protein pharmaceuticals: Past achievements and future challenges. Presentation at the inaugural meeting of the American Institute of Medical and Biological Engineering, Washington, D.C.
von Hippel, E., and Finkelstein, S. 1979. Analysis of innovation in automated clinical chemistry analyzers. Science and Public Policy 6:24–37.