8.A INTRODUCTION
Laboratory workers must understand how the facilities designed for chemical laboratories operate. They should be familiar with ventilation systems, environmental controls, fume hoods and other exhaust devices associated with such equipment. In order to reduce accidents, the experimental work should be viewed in the context of the entire laboratory and its facilities, both for safety and efficiency.
Many laboratory operations depend on special utility requirements that have become common, such as "clean" electric power, high voltages, high-volume water or cooling, and special gas services. Larger and/ or special exhaust devices are also increasingly necessary. The large scale of some experiments may require special laboratory configurations and hazard containment measures; thus, these factors need careful consideration during the experimental design stage. Further, enhanced measures to protect laboratory workers from exposure to potential dangers, coupled with steps to conserve energy and minimize waste, have triggered the implementation of sophisticated systems to provide a safe, comfortable, and cost-effective work environment. Individuals who wish to gain the full benefits that these systems offer must know how they work.
Inspection programs are an important component of maintaining both the physical infrastructure of the laboratory and good relationships with environmental health and safety support staff and facility engineering and maintenance staff. These experts can address specific questions about operating equipment that is peculiar to a particular laboratory and can give expert advice before specialized equipment is ordered.
Increasing demands are being placed on laboratory staff to work safely. As experiments have become increasingly elaborate, so has the infrastructure to support them. The laboratory worker should be kept abreast of changes in the working environment, should keep relations with environmental, engineering, and maintenance staff friendly, and should always confer with them about newly acquired equipment. Institutions must make every effort to maximize the quality of training in this regard and provide opportunities for workers to maintain a safe and successful workplace as changes are introduced in the laboratory.
8.B LABORATORY INSPECTION PROGRAMS
Good housekeeping practices supplemented by a program of periodic laboratory inspections will help to keep laboratory facilities and equipment in a safe operating condition. Inspections can safeguard the quality of the institution's laboratory safety program. A variety of inspection protocols may be used, and the organization's management should select and participate in the design of the inspection program appropriate for that institution's unique needs. The program should embrace goals to:
-
maintain laboratory facilities and equipment in a safe operating condition,
-
provide a comfortable and safe working environment for all employees and the public, and
-
ensure that all laboratory procedures and experiments are conducted in a safe and prudent manner.
These goals should be approached with a considerable degree of flexibility. The different types of inspections, the frequency with which they are conducted, and who conducts them should be considered. A discussion of items to inspect and several possible inspection protocols follows, but neither list is all-inclusive.
8.B.1 Items to Include in an Inspection Program
The following list is representative, not exhaustive. Depending on the laboratory and the type of work conducted in it, other items may also be targeted for inspection. A typical maintenance inspection might consider the following potential hazards:
-
Keep water in drain traps, particularly for floor drains or sinks used infrequently. Vapors emitted from dry, unsealed drains may cause an explosive or flammable condition; such vapors are also the most common source of unexplained laboratory odors. Running water into a drain for 20 to 30 seconds is usually sufficient to fill a drain trap.
-
Secure plastic or rubber hose connections. If proper clamps are not used, hoses can slip off fittings, causing serious floods and water damage. Backflow preventers often require periodic testing to comply with local building codes. Locking quick-disconnects are available for locations where lines often need to be disconnected.
-
Routinely check for inadequate or defective wiring. Frayed cords, improper connections, inappropriate use of extension cords, and power and control wires routed in the same path are found most commonly. Circuit breakers and electrical protection devices should also be checked. These problems create the potential for fires and electrocution.
-
Inspect for improper gas tubing, and faulty valve and regulator installations. These types of errors can be difficult to identify but are important because they pose significant hazards in systems where toxic or flammable gases are used. The typical items to inspect
-
for should include incorrect fittings (incompatible materials), installation errors (e.g., excessive tightness), and defective or missing regulators, flow controls, and monitoring devices. Also check for leaks in inert gas systems to avoid the cost for "lost" gases.
-
Check proximity of flammable materials to any potential ignition sources. Open flames and devices that generate sparks should not be near flammables. Pay special attention to devices placed in fume hoods that do not meet National Electrical Safety Code (U.S. DOC, 1993) Division 1, Group C and D explosion-resistance specifications for electrical devices. Stirrers, hot plates, Variacs, heat tape, outlet strips, ovens (all types), refrigerators, flame sources (e.g., flame ionization detectors (FIDs) and atomic absorption spectrometers), and heat guns constitute the majority of devices that do not typically conform to these code requirements (see section 8.C.6.1).
-
Check guards for rotating machinery and heating devices as protective measures for mechanical and thermal hazards. Test safety switches and emergency stops periodically. Inspect setups for unattended operation, shielding of high-pressure and vacuum equipment, and any other equipment hazards. Compliance with OSHA "lock-out/tag-out" regulations (29 CFR 1910.147) also needs to be verified.
-
Periodically test and inspect emergency devices (e.g., safety showers and eyewash stations) and safety equipment (e.g., fire extinguishers, fire blankets, and first aid and spill control kits) to make sure they are functional. Inspectors need to verify that workers are using personal protection and safety equipment appropriately in their day-to-day work.
-
Keep aisles and emergency exits free of obstacles. Unused supplies and equipment should be stored so as to avoid blocking exits.
-
Carefully examine any documentation required by the institution. Such documentation, which should be made readily available, may include experiment plans, training plans and records, chemical and equipment hazard information, operating plans, and an upto-date emergency evacuation plan. An emergency plan should always be prepared for the contingencies of ventilation failure (resulting from power failure, for example) and other emergencies, such as fire or explosion in a chemical fume hood.
8.B.2 Types of Inspection Programs: Who Conducts Them and What They Offer
The easiest inspection program to implement requires laboratory supervisors to inspect their own work space and equipment on a periodic basis. These individuals are the "first line of defense" for a program of safety excellence. They need to note items such as open containers, faulty faucets or valves, frayed wiring, broken apparatus, obstructions on floors or aisles, and unsafe clutter. They also need to follow through to make sure that any problems receive prompt resolution.
General equipment inspection should also be done fairly frequently. For certain types of equipment in constant use, such as gas chromatographs, daily inspections may be appropriate. Other types of equipment may need only weekly or monthly inspection, or inspection prior to use if operated infrequently.
The challenge for any inspection program is to keep laboratory workers continuously vigilant. Workers need positive encouragement to develop the habit of inspection and to adopt the philosophy that good housekeeping and maintenance for their work space protect them and may help them produce better results for their efforts. Incentive programs may stimulate workers to pay closer attention to the condition of their equipment and work space.
Probably the most traditional type of inspection is that conducted by the laboratory supervisor. This form of inspection presents an excellent opportunity to promote a culture of safety and prudence within an organization. The supervisor gains the opportunity to take a close look at the facilities and operations. He or she also can discuss with individual workers issues of interest or concern that may fall outside the scope of the actual inspection. Again, a constructive and positive approach to observed problems and issues will foster an attitude of cooperation and leadership with regard to safety. It can help build and reinforce a culture of teamwork and cooperation that has benefits far beyond protecting the people and physical facilities.
The International Loss Control Institute (ILCI), a private organization contracted to assess safety standards at industrial facilities, recommends a monthly inspection of industrial laboratory facilities and equipment by supervisory personnel. Some institutions may deem this frequency unnecessarily high. Supervisors should probably make inspections no less frequently than once per quarter.
More senior staff should be encouraged to inspect facilities periodically, too. In most cases, these inspections should occur at least annually. They offer the added benefit of providing a senior leader with a good overview of the condition of the facilities, the work conducted in the laboratories, who does what work, and how people feel about their work. In essence, these kinds of inspections supplement the normal practice of "management by walking around"
One of the most effective safety tools a larger institution can employ is periodic peer-level inspections. Usually, the people who fulfill this role work in the organization they serve, but not in the area being surveyed. Individuals may function on an ad hoc basis, or the institution may select specific individuals and confer on them various formal appellations such as ''safety committee member." A peer inspection program has the intrinsic advantage of being perceived as less threatening than other forms of surveys or audits.
Peer inspections depend heavily on the knowledge and commitment of the people who conduct them. Individuals who volunteer or are selected to perform inspections for only a brief period of service may not learn enough about an operation or procedure to observe and comment constructively. On the other hand, people who receive involuntary appointments or who serve too long may not maintain the desired level of diligence. Peer-level inspectors generally perform best if they have some ongoing responsibility with regard to the safety program, such as consulting on experiment designs. They will be more familiar with the work and will conduct the review more effectively.
Having a high-quality peer-level inspection program may reduce the need for frequent inspections by supervisory personnel. However, peer inspections should not replace other inspections completely. Walkthroughs by the organization's leadership demonstrate commitment to the safety programs, which is key to their continuing success.
Another option is to have the organization's environmental health and safety staff conduct inspections. In a smaller organization, these types of inspections may adequately address the ILCI frequency recommendations. In larger institutions, the safety staff may be limited to semiannual or even annual walk-throughs. A more practical use of safety staff for inspections may be to target certain operations or experiments. Or the safety staff could focus on a particular type of inspection, such as safety equipment and systems. Finally, they could perform "audits" to check the work of other inspectors or look specifically at previous problem areas. It is important for the safety staff to address noted deficiencies with appropriate reminders and/ or additional training. Punitive measures should be employed, but only for chronic offenders or deliberate problems that pose a serious potential hazard.
Safety staff are not the only nonlaboratory personnel who should conduct safety inspections. Facility engineers or maintenance personnel may add considerable value to safety inspection programs. It also gives them the opportunity to gain a better perspective on the laboratory work.
Other types of inspections and audits use individuals or groups outside the laboratory organization to conduct the survey. Inspections may be mandatory, such as annual site visits by EPA to hazardous waste generators, or they may be surprise inspections with 24 hours notice or less given prior to the visit. Organizations subject to such inspections must keep their programs and records up-to-date at all times. In fact, all organizations should strive for that goal. Any significant incident or accident within a facility will trigger one or more inspections and investigations by outside organizations. If the underlying safety programs are found to be sound, that factor may help limit negative findings and potential penalties.
Many different types of elective inspections or audits can be conducted by outside experts or organizations. They may inspect a particular facility, piece(s) of equipment, or procedure either during the pre-experiment design phase or during operations.
Tours, walk-throughs, and inspections by outside organizations offer the opportunity to build relationships with governmental agencies and the public. For example, an annual visit by the fire department serving a particular facility will acquaint personnel with the operations and the location of particular hazards. If these individuals are ever called into the facility to handle an emergency, their familiarity with the facility will give them a greater degree of effectiveness. During their walk-through, they may offer comments and suggestions for improvements. A relationship built over a period of time will help make this input positive and constructive.
If a pending operation or facility change may cause public attention and concern, an invitation targeted to specific people or groups may prevent problems. Holding public open houses from time to time can help build a spirit of support and trust. Many opportunities exist to apply this type of open approach to dealing with the public. An organization only needs to consider when to use it and what potential benefits may accrue.
|
An industrial laboratory arranged an on-site visit for representatives from the Sierra Club and other local citizen action groups who had begun to raise objections to the approval of the site's RCRA Part B permit for a hazardous waste incinerator. Concerns about the noise, smoke, and smell from the incinerator were dispelled after the individuals stood right next to it and did not realize what it was and that it was operating at the time! |
8.C LABORATORY VENTILATION
8.C.1 Laboratory Fume Hoods
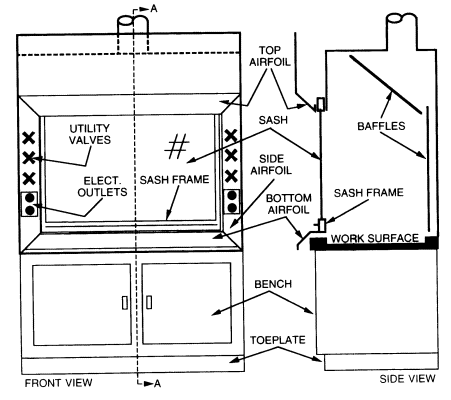
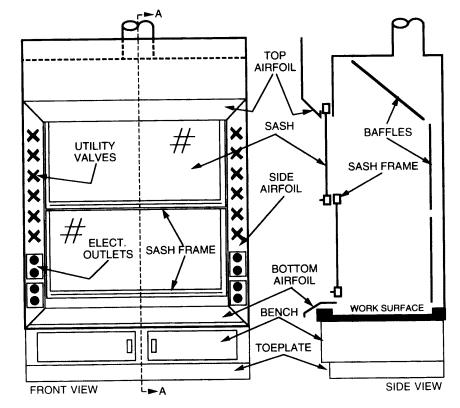
Laboratory fume hoods are the most important components used to protect laboratory workers from exposure to hazardous chemicals and agents used in the laboratory. Functionally, a standard fume hood is a fire-and chemical-resistant enclosure with one opening (face) in the front with a movable window (sash) to allow user access into the interior. Large volumes of air are drawn through the face and out the top to contain and remove contaminants from the laboratory.
8.C.2 Fume Hood Face Velocity
The average velocity of the air drawn through the face of the hood is called the face velocity. It is generally calculated as the total volumetric exhaust flow rate for the hood, divided by the area of the open face, less an adjustment for hood air leakage. The face velocity of a hood greatly influences the ability of the hood to contain hazardous substances, that is, its containment efficiency. Face velocities that are too low or too high will reduce the containment efficiency of a fume hood. In most cases, the recommended face velocity is between 80 and 100 feet per minute (fpm). Face velocities between 100 and 120 fpm may be used for substances of very high toxicity or where outside influences adversely affect hood performance. However, energy costs to operate the fume hood are directly proportional to the face velocity. Face velocities approaching or exceeding 150 fpm should not be used, because they may cause turbulence around the periphery of the sash opening and actually reduce the capture efficiency of the fume hood.
Average face velocity is determined either by measuring individual points across the plane of the sash opening and calculating their average or by measuring the hood volume flow rate with a pitot tube in the exhaust duct and dividing this rate by the open face area. Containment may be verified by using one of the flow visualization techniques found in section 8.C.5 on fume hood testing. Once the acceptable average face velocity, minimum acceptable velocity, and maximum standard deviation of velocities have been determined for a hood, laboratory, facility, or site, they should be incorporated into the laboratory's Chemical Hygiene Plan.
8.C.3 Factors That Affect Fume Hood Performance
Tracer gas containment testing of fume hoods has revealed that air currents impinging on the face of a hood at a velocity exceeding 30 to 50% of the hood face velocity will reduce the containment efficiency of the hood by causing turbulence and interfering with the laminar flow of the air entering the hood. Thirty to fifty percent of a hood face velocity of 100 fpm, for example, is 30 to 50 fpm, which represents a very low velocity that can be produced in many ways. The rate of 20 fpm is considered to be still air because that is the velocity at which most people first begin to sense air movement.
8.C.3.1 Adjacency to Traffic
Most people walk at a velocity of approximately 250 fpm (about 3 miles per hour). Wakes or vortices form behind a person who is walking, and velocities in those vortices exceed 250 fpm. When a person walks in front of an open fume hood, the vortices can overcome the fume hood face velocity and pull contaminants out of the fume hood, into the vortex, and into the laboratory. Therefore, fume hoods should not be located on heavily traveled aisles, and those that are should be kept closed when not in use. Foot traffic near these hoods should be avoided, or special care should be taken.
8.C.3.2 Adjacency to Supply Air Diffusers
Air is supplied continuously to laboratories to replace the air exhausted from the fume hoods and other exhaust sources and to provide ventilation and temperature/humidity control. This air usually enters the laboratory through devices called supply air diffusers located in the ceiling. Velocities that can exceed 800 fpm are frequently encountered at the face of these diffusers. If air currents from these diffusers reach the face of a fume hood before they decay to 30 to 50% of the hood face velocity, they can cause the same effect as air currents produced by a person walking in front of the hood. Normally, the effect is not quite as pronounced as the traffic effect, but it occurs constantly, whereas the traffic effect is transient. Relocating the diffuser, replacing it with another type, or rebalancing the diffuser air volumes in the laboratory can alleviate this problem.
8.C.3.3 Adjacency to Windows and Doors
Exterior windows with movable sashes are not recommended in laboratories. Wind blowing through the windows and high-velocity vortices caused when doors open can strip contaminants out of the fume hoods and interfere with laboratory static pressure controls.
8.C.4 General Safe Operating Procedures for Fume Hoods
In addition to protecting the laboratory worker from toxic or unpleasant agents used in them, fume hoods can provide an effective containment device for accidental spills of chemicals. There should be at least one hood for every two workers in laboratories where most work involves hazardous chemicals, and the hoods should be large enough to provide each worker with at least 2.5 linear feet of working space at the face. If this amount of hood space is not available, other types of local ventilation should be provided, and special care should be exercised to monitor and restrict the use of hazardous substances.
8.C.4.1 Prevention of Intentional Release of Hazardous Substances Into Fume Hoods
Fume hoods should be regarded as backup safety devices that can contain and exhaust toxic, offensive, or flammable materials when the containment of an experiment or procedure fails and vapors or dusts escape from the apparatus being used. Note the following:
-
Just as you should never flush a laboratory waste down the drain, never intentionally send waste up the hood.
-
Instead, fit all apparatus used in hoods with condensers, traps, or scrubbers to contain and collect waste solvents or toxic vapors or dusts.
|
QUICK GUIDE GUIDELINES FOR MAXIMIZING HOOD EFFICIENCY Many factors can compromise the efficiency of a hood operation. Most of these are avoidable; thus, it is important to be aware of all behavior that can, in some way, modify the hood and its capabilities. The following should always be considered when using a hood:
|
-
Make sure that all highly toxic or offensive vapors are scrubbed or adsorbed before the exit gases are released into the hood exhaust system (see section 8.C.8.1 on fume hood scrubbers).
8.C.4.2 Fume Hood Performance Checks
It is necessary to check if the hoods are performing properly. Observe the following guidelines:
-
Evaluate each hood before use and on a regular basis (preferably once a year) to verify that the face velocity meets the criteria specified for it in the laboratory's Chemical Hygiene Plan.
-
Also verify the absence of excessive turbulence (see section 8.C.4.4 below).
-
Make sure that a continuous monitoring device for adequate hood performance is present, and check it every time the hood is used.
(For further information, see section 8.C.5 on testing and verification.)
8.C.4.3 Housekeeping
Laboratory fume hoods and adjacent work areas should be kept clean and free of debris at all times. Solid objects and materials (such as paper) should be kept from entering the exhaust ducts of hoods, because they can lodge in the ducts or fans and adversely affect their operation. Also, the hood will have better airflow across its work surface if there are minimal numbers of bottles, beakers, and laboratory apparatus inside the hood; therefore, it is prudent to keep unnecessary equipment and glassware outside of the hood at all times and store all chemicals in approved storage cans, containers, or cabinets (not in the fume hood). Furthermore, it is best to keep the work space neat and clean in all laboratory operations, particularly those involving the use of hoods, so that any procedure or experiment can be undertaken without the possibility of disturbing, or even destroying, what is being done.
8.C.4.4 Sash Operation
Except when adjustments to the apparatus are being made, the hood should be kept closed, with vertical sashes down and horizontal sashes closed, to help prevent the spread of a fire, spill, or other hazard into the laboratory. Sliding sashes should not be removed from horizontal sliding sash hoods. The face opening of the hood should be kept small to improve the overall performance of the hood. If the face velocity becomes excessive, the facility engineers should make adjustments or corrections.
For hoods without face velocity controls (see section 8.C.6.3.2), the sash should be positioned to produce the recommended face velocity, which often occurs only over a limited range of sash positions. This range should be determined and marked during fume hood testing. For hoods with face velocity controls, it is imperative to keep the sash closed when the hood is not in use.
8.C.4.5 Constant Operation of Fume Hoods
Although turning fume hoods off when not in use saves energy, keeping them on at all times is safer, especially for fume hoods connected directly to a single fan. Because most laboratory facilities are under negative pressure, air may be drawn backward through the nonoperating fan, down the duct, and into the laboratory unless an ultralow-leakage backdraft damper is used in the duct. If the air is cold, it may freeze liquids in the hood. Fume hood ducts are rarely insulated; therefore, condensation and ice may form in cold weather. When the fume hood is turned on again and the duct temperature rises, the ice will melt, and water will run down the ductwork, drip into the hood, and possibly react with chemicals in the hood.
Fume hoods connected to a common exhaust manifold offer an advantage. The main exhaust system will rarely be shut down; hence, positive ventilation is available to each hood on the system at all times. In a constant air volume (CAV) system (see section 8.C.6.3.1), "shutoff" dampers to each hood can be installed, allowing passage of enough air to prevent fumes from leaking out of the fume hoods and into the laboratory when the sash is closed. It is prudent to allow 10 to 20% of the full volume of the hood flow to be drawn through the hood in the off position to prevent excessive corrosion.
8.C.5 Testing and Verification
All fume hoods should be tested, before they leave the manufacturer, by using ASHRAE/ANSI standard 110, Methods of Testing Performance of Laboratory Fume Hoods. The hood should pass the low- and high-volume smoke challenges with no leakage or flow reversals and have a control level of 0.05 parts per million (ppm) or less on the tracer gas test. ASHRAE/ ANSI 110 testing of fume hoods after installation in their final location by trained personnel is highly recommended. The control level of tracer gas for an "as installed" or "as used" test via the ASHRAE/ANSI 110 method should not exceed 0.1 ppm. Periodic per-
formance testing consisting of a face velocity analysis and flow visualization using smoke tubes, bombs, or fog generators should be performed annually. Laboratory workers should request a fume hood performance evaluation any time there is a change in any aspect of the ventilation system. Thus, changes in the total volume of supply air, changes in the locations of supply air diffusers, or the addition of other auxiliary local ventilation devices (e.g., more hoods, vented cabinets, and snorkels) all call for reevaluation of the performance of all hoods in the laboratory.
The ASHRAE/ANSI 110 test is the most practical way to determine fume hood capture efficiency quantitatively. The test includes several components, which may be used together or separately, including face velocity testing, flow visualization, face velocity controller response testing, and tracer gas containment testing. Performance should be evaluated against the design specifications for uniform airflow across the hood face as well as for the total exhaust air volume. Equally important is the evaluation of operator exposure. The first step in the evaluation of hood performance is the use of a smoke tube or similar device to determine that the hood is on and exhausting air. The second step is to measure the velocity of the airflow at the face of the hood. The third step is to determine the uniformity of air delivery to the hood face by making a series of face velocity measurements taken in a grid pattern.
Traditional hand-held instruments are subject to probe movement and positioning errors as well as reading errors owing to the optimistic bias of the investigator. Also, the traditional method yields only a snapshot of the velocity data, and no measure of variation over time is possible. To overcome this limitation, it is recommended that velocity data be taken while using a velocity transducer connected to a data acquisition system and read continuously by a computer for approximately 30 seconds at each traverse point. If the transducer is fixed in place, using a ring stand or similar apparatus, and is properly positioned and oriented, this method can overcome the errors and drawbacks associated with the traditional method. The variation in data for a traverse point can then be used as an indicator of turbulence, an important additional performance indicator that has been almost completely overlooked in the past. If the standard deviation of the average velocity profile at each point exceeds 20% of the mean, or the average standard deviation of velocities at each traverse point (turbulence) exceeds 15% of the mean face velocity, corrections should be made by adjusting the interior hood baffles and, if necessary, by altering the path of the supply air flowing into the room. Most laboratory hoods are equipped with a baffle that has movable slot openings at both the top and the bottom, which should be moved until the airflow is essentially uniform. Larger hoods may require additional slots in the baffle to achieve uniform airflow across the hood face. These adjustments should be made by an experienced laboratory ventilation engineer or technician using proper instrumentation.
The total volume of air exhausted by a hood is the sum of the face volume (average face velocity times face area of the hood) plus air leakage, which averages about 5 to 15% of the face volume. If the hood and the general ventilating system are properly designed, face velocities in the range of 80 to 100 fpm will provide a laminar flow of air over the work surface and sides of the hood. Higher face velocities (150 fpm or more), which exhaust the general laboratory air at a greater rate, both waste energy and are likely to degrade hood performance by creating air turbulence at the hood face and within the hood, causing vapors to spill out into the laboratory.
Because a substantial amount of energy is required to supply tempered supply air to even a small hood, the use of hoods to store bottles of toxic or corrosive chemicals is a very wasteful practice, which can also, as noted above, seriously impair the effectiveness of the hood as a local ventilation device. Thus, it is preferable to provide separate vented cabinets for the storage of toxic or corrosive chemicals. The amount of air exhausted by such cabinets is much less than that exhausted by a properly operating hood. (Also see section 8.C.4.)
Perhaps the most meaningful (but also the most time-consuming and expensive) method for evaluating hood performance is to measure worker exposure while the hood is being used for its intended purpose. By using commercial personal air-sampling devices that can be worn by the hood user, worker exposure (both excursion peak and time-weighted average) can be measured by using standard industrial hygiene techniques. The criterion for evaluating the hood should be the desired performance (i.e., does the hood contain vapors and gases at the desired worker-exposure level?). A sufficient number of measurements should be made to define a statistically significant maximum exposure based on worst-case operating conditions. Direct-reading instruments are available for determining the short-term concentration excursions that may occur in laboratory hood use.
8.C.6 Fume Hood Design and Construction
When specifying a laboratory fume hood for use in a particular activity, the laboratory worker should be
aware of all the design features of the hood. Assistance from an industrial hygienist, ventilation engineer, or laboratory consultant is recommended when deciding to purchase a fume hood.
8.C.6.1 General Design Recommendations
Laboratory fume hoods and the associated exhaust ducts should be constructed of nonflammable materials. They should be equipped with either vertical or horizontal sashes that can be closed. The glass within the sash should be either laminated safety glass that is at least 7/32 inch thick or other equally safe material that will not shatter if there is an explosion within the hood. The utility control valves, electrical receptacles, and other fixtures should be located outside the hood to minimize the need to reach within the hood proper. Other specifications regarding the construction materials, plumbing requirements, and interior design will vary, depending on the intended use of the hood. (See Chapter 6, sections 6.C.1.1 and 6.C.1.2.)
Although hoods are most commonly used to control concentrations of toxic vapors, they can also serve to dilute and exhaust flammable vapors. Although theoretically possible, it is extremely unlikely (even under most worst-case scenarios) that the concentration of flammable vapors will reach the lower explosive limit (LEL) in the exhaust duct. However, somewhere between the source and the exhaust outlet of the hood, the concentration will pass through the upper explosive limit (UEL) and the LEL before being fully diluted at the outlet. Both the hood designer and the user should recognize this hazard and eliminate possible sources of ignition within the hood and its ductwork if there is a potential for explosion. The use of duct sprinklers or other suppression methods in laboratory fume ductwork is not necessary, or desirable, in the vast majority of situations.
8.C.6.2 Special Design Features
There have been two major improvements in fume hood design-airfoils and baffles-since the fume hood was invented. Both features should be included on any new fume hoods that are purchased.
Airfoils built into the fume hood at the bottom and sides of the sash opening significantly reduce boundary turbulence and improve capture performance. All fume hoods purchased should be fitted with airfoils.
When air is drawn through a hood without a baffle (see Figure 8.1), most of the air is drawn through the upper part of the opening, producing an uneven velocity distribution across the face opening. When baffles are installed, the velocity distribution is greatly improved. All fume hoods should have baffles. Adjustable baffles can improve hood performance and are desirable if the adjustments are made by an experienced industrial hygienist, consultant, or hood technician.
8.C.6.3 Fume Hood Airflow Types
The first fume hoods were simply boxes that were open on one side and connected to an exhaust duct. Since they were first introduced, many variations on this basic design have been made. Six of the major variants in fume hood airflow design are listed below with their characteristics. Conventional hoods are the most common and include benchtop, distillation, and walk-in hoods of the constant air volume (CAV), variable air volume (VAV), bypass and non-bypass variety, with or without airfoils. Auxiliary air hoods and ductless fume hoods are not considered "conventional" and are used less often. Laboratory workers should know what kind of hood they are using and what its advantages and limitations are.
8.C.6.3.1
Constant Air Volume (CAV) Hoods
A constant air volume (CAV) fume hood draws a constant exhaust volume through the hood regardless of sash position. Because the volume is constant, the face velocity varies inversely with the sash position. The fume hood volume should be adjusted to achieve the proper face velocity at the desired working height of the sash, and then the hood should be operated at this height. (See section 8.C.4.)
8.C.6.3.2
Variable Air Volume (VAV) Hoods
A variable air volume (VAV) fume hood, also known as a constant velocity hood, is any hood that has been fitted with a face velocity control, which varies the amount of air exhausted from the fume hood in response to the sash opening to maintain a constant face velocity. In addition to providing an acceptable face velocity over a relatively large sash opening (compared to a CAV hood), VAV hoods also provide significant energy savings by reducing the flow rate from the hood when it is closed. These types of hoods are usually of the non-bypass design to reduce air volume (see below).
8.C.6.3.3
Non-Bypass Hoods
A non-bypass hood has only one major opening through which the air may pass into the hood, that is, the sash opening. The airflow pattern of this type of hood is shown in Figure 8.2. A CAV non-bypass hood
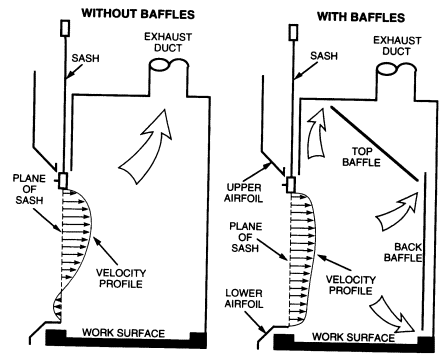
FIGURE 8.1 Effect of baffles on face velocity profile.
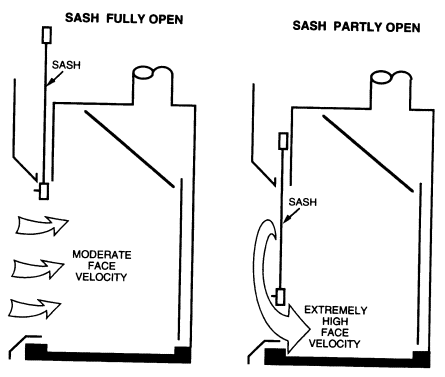
FIGURE 8.2 Non-Bypass fume hood.
has the undesirable characteristic of producing very large face velocities at small sash openings. As the sash is lowered, face velocities may exceed 1,000 fpm near the bottom. Face velocities are limited by the leakage of the hood through cracks and under the airfoil and by the increasing pressure drop as the sash is closed. A common misconception is that the volume of air exhausted by this type of hood decreases when the sash is closed. Although the pressure drop through the hood increases slightly as the sash is closed, no appreciable change in volume occurs. This does not mean that these fume hoods should not be closed when
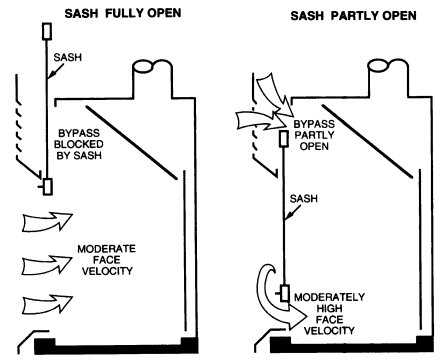
FIGURE 8.3 Bypass fume hood.
not in use, however. A closed hood provides a primary barrier to the spread of a fire or chemical release.
Many workers are reluctant to close their CAV non-bypass hoods because of the increase in air velocity and noise that occurs when the sash is lowered. This high-velocity air jet sweeping over the work surface often disturbs gravimetric measurements, causes undesired cooling of heated vessels and glassware, and can blow sample trays, gloves, and paper towels to the back of the hood, where they may be drawn into the exhaust system. Care should be exercised to prevent materials from entering the exhaust system. They can lodge in the ductwork, reducing airflow to the hood, or can be conveyed through the system and drawn into the exhaust fan and damage the fan or cause sparks. Variable-volume non-bypass hoods avoid these undesirable features by maintaining the velocity at a constant rate except at very low sash positions, where it may increase to a few hundred feet per minute. Hoods with horizontal sashes are usually of the non-bypass variety, because this arrangement is more difficult to connect to the bypass mechanism.
8.C.6.3.4
Bypass Hoods
A bypass fume hood is shown in Figure 8.3. It is similar to the non-bypass design but has an opening above the sash through which air may pass at low sash positions. Because the opening is usually 20 to 30% of the maximum open area of the sash, this hood will still exhibit the increasing velocity characteristic of the non-bypass hood as the sash is lowered. But the face velocity stops increasing as the sash is lowered to the position where the bypass opening is exposed by the falling sash. The terminal face velocity of these types of hoods depends on the bypass area but is usually in the range of 300 to 500 fpm—significantly higher than the recommended operating face velocity. Therefore, the air volume for bypass hoods should also be adjusted to achieve the desired face velocity at the desired sash height, and the hood should be operated at this position. This arrangement is usually found in combination with a vertical sash, because this is the simplest arrangement for opening the bypass. Varieties are available for horizontal sashes, but the bypass mechanisms are complicated and may cause maintenance problems.
8.C.6.3.5
Auxiliary Air Hoods
Quantitative tracer gas testing of many auxiliary air fume hoods has revealed that, even when adjusted properly and with the supply air properly conditioned, significantly higher worker exposure to the materials used in the hood may occur than with conventional (non-auxiliary air) hoods. Auxiliary air hoods should not be purchased for new installations, and existing auxiliary air hoods should be replaced or modified to eliminate the supply air feature of the hood. This feature causes a
disturbance of the velocity profile and leakage of fumes from the hood into the worker's breathing zone.
The auxiliary air fume hood was developed in the 1970s primarily to reduce laboratory energy consumption. It is a combination of a bypass fume hood and a supply air diffuser located at the top of the sash. These hoods were intended to introduce unconditioned or tempered air, as much as 70% of the air exhausted from the hood, directly to the front of the hood. Ideally, this unconditioned air bypasses the laboratory and significantly reduces air conditioning and heating costs in the laboratory. In practice, however, many problems are caused by introducing unconditioned or slightly conditioned air above the sash, all of which may produce a loss of containment.
8.C.6.3.6 Ductless Fume Hoods
Ductless fume hoods are ventilated enclosures that have their own fan, which draws air out of the hood and through filters and ultimately recirculates it into the laboratory. The filters are designed to trap vapors generated in the hood and exhaust "clean" air back into the laboratory. These hoods usually employ activated carbon filters. The collection efficiency of the filters decreases over time. Ductless fume hoods have extremely limited applications and should be used only where the hazard is very low, where the access to the hood and the chemicals used in the hood are carefully controlled, and under the supervision of a laboratory supervisor who is familiar with the serious limitations and potentially hazardous characteristics of these devices. If these limitations cannot be accommodated, then this type of device should not be used.
8.C.7 Fume Hood Configurations
8.C.7.1 Benchtop Hoods
As the name implies, a benchtop fume hood sits on a laboratory bench with the work surface at bench height. These hoods can be of the CAV or VAV variety and can be a bypass or non-bypass design. The sash can be a vertical rising or a horizontal sliding type, or a combination of the two. Normally, the work surface is dished or has a raised lip around the periphery to contain spills in the hood. Sinks in hoods are not recommended because they encourage laboratory workers to dispose of chemicals in them. If they must be used, to drain cooling water from a condenser, for instance, then they should be fitted with a standpipe to prevent chemical spills from entering the drain. The condenser water drain can be run into the standpipe. Spills will be caught in the cupsink by the standpipe for later cleanup and disposal. A typical benchtop fume hood is shown in Figure 8.4.
8.C.7.2 Distillation (Knee-high or Low-boy) Hoods
The distillation hood is similar to the benchtop hood except that the work surface is closer to the floor to allow more vertical space inside the hood for tall apparatus such as distillation columns. A typical distillation hood is shown in Figure 8.5.
8.C.7.3 Walk-in Hoods
A walk-in hood stands on the floor of the laboratory and is used for very tall or large apparatus. The sash type can be either horizontal or double- or triple-hung vertical. These hoods are also usually of the non-bypass type. The word "walk-in" is really a misnomer. One should never actually walk into a fume hood when it is operating and contains hazardous chemicals. Once past the plane of the sash, the worker is inside the hood with the chemicals. If the worker is required to enter the hood during operations where hazardous chemicals are present, personal protective equipment appropriate for the hazard should be worn. This may include respirators, goggles, rubber gloves, boots, suits, and self-contained breathing apparatus. A typical walk-in hood is shown in Figure 8.6.
8.C.7.4 California Hoods and Ventilated Enclosures
The California hood is a ventilated enclosure with a movable sash on more than one side. These hoods can usually be accessed through a horizontal sliding sash from the front and rear. They may also have a sash on the ends. Their configuration precludes the use of baffles and airfoils and therefore may not provide a suitable face velocity distribution across their many openings.
A ventilated enclosure is any site-fabricated hood designed primarily for containing processes such as scale-up or pilot plant equipment. Most do not have baffles or airfoils, and most designs have not had the rigorous testing and design refinement that conventional mass-produced fume hoods enjoy. Both the California hood and the ventilated enclosure are designed primarily to contain, but not capture, fumes like a conventional fume hood. Working at the opening of the devices, even when the plane of the opening has not been broken, may expose the worker to higher concentrations of hazardous materials than if a conventional hood were used.
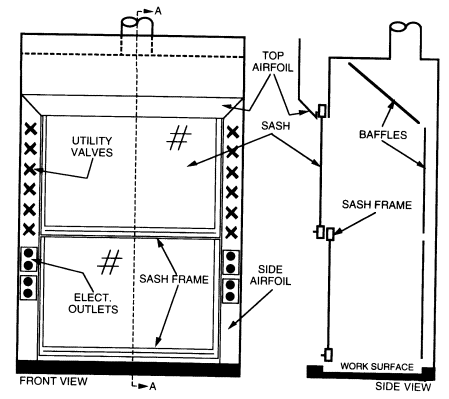
FIGURE 8.6 Walk-in fume hood.
8.C.7.5 Perchloric Acid Hoods
The perchloric acid hood, with its associated ductwork, exhaust fan, and support systems, is designed especially for use with perchloric acid and other materials that can deposit shock-sensitive crystalline materials in the hood and exhaust system. These materials become pyrophoric when they dry or dehydrate (see also Chapter 5, section 5.G.6). Special water spray systems are employed to wash down all interior surfaces of the hood, duct, fan, and stack, and special drains are necessary to handle the effluent from the washdown. The hood liner and work surface are usually stainless steel and are sealed by welding all seams. Perchloric acid hoods have drains in the work surface. Water spray heads are usually installed in the top of the hood, behind the baffles, and in the hood interior. The water spray should be turned on whenever perchloric acid is being heated in the fume hood. Welded or flanged and gasketed fittings to provide air-and water-tight connections are recommended. The ductwork, fan, and stack should be fabricated of plastic, glass, or stainless steel and should be fitted with spray heads approximately every 10 feet on vertical runs and at each change in direction. Horizontal runs should be avoided because they inhibit drain-down, and the spray action is not as effective on the top and sides of the duct. The washdown piping, located outside, should be protected from freezing. A three-way valve on the water supply piping that allows it to drain when not in use is helpful. Care should be used when routing the water lines to prevent the creation of traps that will retain water. Special operating procedures should be written to cover the washdown procedure for these types of hoods. The exhaust from a perchloric acid hood should not be manifolded with that from other types of fume hoods.
8.C.7.6 Radioisotope Hoods
Hoods used for work with radioactive sources or materials should be designed so that they can be decontaminated completely on a regular basis. A usual feature is a one-piece, stainless steel, welded liner with smooth, curved corners, which can be cleaned easily and completely. The superstructure of radioisotope hoods is usually made stronger than that of a conventional hood in order to support lead bricks and other shielding that may be required in the hood. Special treatment of the exhaust from radioisotope hoods may be required by government agencies to prevent the release of radioactive material into the environment. This usually involves the use of high-efficiency particulate air (HEPA) filters (see section 8.C.8.4).
8.C.8 Fume Hood Exhaust Treatment
Until now, treatment of fume hood exhausts has been limited. Because effluent quantities and concentrations are relatively low compared to those of other industrial air emissions sources, their removal is technologically challenging. And the chemistry for a given fume hood effluent can be difficult to predict and may change over time.
Nevertheless, legislation and regulations increasingly recognize that certain materials exhausted by a fume hood may be sufficiently hazardous that they can no longer be expelled directly into the air. Therefore, the practice of removing these materials from hood exhaust streams will become increasingly more prevalent.
8.C.8.1 Fume Hood Scrubbers and Contaminant Removal Systems
A number of technologies are evolving for treating fume hood exhaust by means of fume hood scrubbers and containment removal systems. Whenever possible, experiments involving such materials should be designed so that the toxic materials are collected in traps or scrubbers rather than being released into the hood. If for some reason this is impossible, then HEPA filters are recommended for highly toxic particulates. Liquid scrubbers may also be used to remove particulates, vapors, and gases from the exhaust system. None of these methods, however, is completely effective, and all trade an air pollution problem for a solid or liquid waste disposal problem. Incineration may be the ultimate method for destroying combustible compounds in exhaust air, but adequate temperature and dwell time are required to ensure complete combustion (see section 8.C.9).
Incinerators require considerable capital to build and energy to operate; hence, other methods should be studied before resorting to their use. The optimal system for collecting or destroying toxic materials in exhaust air must be determined on a case-by-case basis. In all cases, such treatment of exhaust air should be considered only if it is not practical to pass the gases or vapors through a scrubber or adsorption train before they enter the exhaust airstream. Also, if an exhaust system treatment device is added to an existing fume hood, the impact on the fan and other exhaust system components must be carefully evaluated. These devices require significant additional energy to overcome the pressure drop they add to the system. (See also Chapter 7, section 7.B.6.1.)
8.C.8.2 Liquid Scrubbers
A fume hood scrubber is a laboratory-scale version of a typical packed-bed liquid scrubber used for industrial air pollution control. Figure 8.7 shows a schematic of a typical fume hood scrubber.
Contaminated air from the fume hood enters the unit and passes through the packed bed, liquid spray section, and mist eliminator, and then into the exhaust system for release up the stack. The air and the scrubbing liquor pass in a countercurrent fashion for efficient gas-liquid contact. The scrubbing liquor is recirculated from the sump and back to the top of the system using a pump. Water-soluble gases, vapors, and aerosols are dissolved into the scrubbing liquor. Particulates are also captured quite effectively by this type of scrubber. Removal efficiencies for most water-soluble acid- and base-laden airstreams are usually between 95 and 98%.
Scrubber units are typically configured vertically and are located next to the fume hood as shown in Figure 8.7. They are also produced in a top mount version, in which the packing, spray manifold, and mist eliminator sections are located on top of the hood and the sump and liquid handling portion are underneath the hood for a compact arrangement taking up no more floor area than the hood itself.
8.C.8.3 Other Gas-Phase Filters
There is another basic type of gas-phase filtration available for fume hoods in addition to liquid scrubbers. These are "inert" adsorbents and chemically active adsorbents. The "inert" variety includes activated carbon, activated alumina, and Molecular Sieves®. These substances typically come in bulk form for use in a deep bed and are available also as cartridges and as panels for use in housings similar to particulate filter housings. They are usually manufactured in the form of beads, but they may take many forms. The beads are porous and have extremely large surface areas with sites onto which gas and vapor molecules are trapped or adsorbed as they pass through. Chemically active adsorbents are simply inert adsorbents impregnated with a strong oxidizer, such as potassium permanganate (purple media), which reacts with and destroys the organic vapors. Although there are other oxidizers targeted to specific compounds, the permanganates are the most popular. Adsorbents can handle hundreds of different compounds, including most volatile organic components (VOCs), but also have an affinity for harmless species such as water vapor.
As the air passes through the adsorbent bed, gases are removed in a section of the bed. (For this discussion, "gas" means gases and vapors.) As the bed loads with gases, and if the adsorbent is not regenerated or replaced, eventually contaminants will break through the end of the bed. After breakthrough occurs, gases will pass through the bed at higher and higher concen-
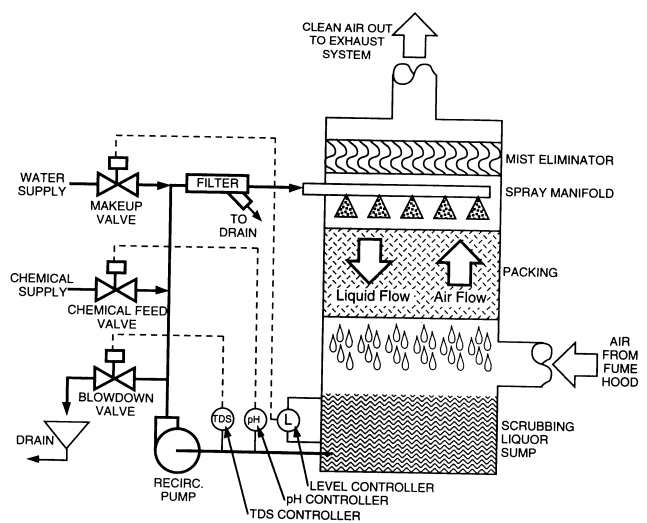
FIGURE 8.7 Typical fume hood scrubber schematic.
trations at a steady state until the upstream and downstream levels are almost identical. To prevent breakthrough, the adsorbent must be either changed or regenerated on a regular basis. Downstream monitoring to detect breakthrough or sampling of the media to determine the remaining capacity of the bed should be performed regularly.
An undesirable characteristic of these types of scrubbers is that if high concentrations of organics or hydrocarbons are carried into the bed, as would occur if a liquid were spilled inside the hood, a large exotherm will occur in the reaction zone of the bed. This exotherm may cause a fire in the scrubber. These devices and other downstream devices such as particulate filters should be located to minimize the effects of a fire, should one occur. Fires can start in these devices at surprisingly low temperatures due to the catalytic action of the adsorbent matrix. Therefore, such devices should be used and operated with care.
8.C.8.4 Particulate Filters
Air from fume hoods and biological safety cabinets in which radioactive or biologically active materials are used should be properly filtered to remove these agents so that they are not released into the atmosphere. Other hazardous particulates may require this type of treatment as well. The most popular method of accomplishing this removal is by using a HEPA filter bank. These HEPA filters trap 99.97% of all particulates greater than 3 microns in diameter. These systems must be specified, purchased, and installed so that the filters can be changed without exposing the worker or the environment to the agents trapped in the filter. Sterilizing the filter bank is prudent before changing filters that may contain etiologic agents.
The "bag-in, bag-out" method of replacing filters is a popular way to prevent worker exposure. This method separates the contaminated filter and housing from the
worker and the environment by using a special plastic barrier "bag" and special procedures to prevent exposure to or release of the hazardous agent.
8.C.9 Thermal Oxidizers and Incinerators
Thermal oxidizers and incinerators are extremely expensive to purchase, install, operate, and maintain. However, they are one of the most effective methods of handling toxic and etiologic agents. The operational aspects of these devices are beyond the scope of this book. Also, their application to fume hoods has historically been rare. When considering this method of pollution control, an expert should be called to assist.
8.C.10 Other Local Exhaust Systems
There are many types of laboratory equipment and apparatus that generate vapors and gases but should not be used in a conventional fume hood. Some examples are gas chromatographs, atomic absorption spectrophotometers, mixers, vacuum pumps, and ovens. If the vapors or gases emitted by this type of equipment are hazardous or noxious, or if it is undesirable to release them into the laboratory because of odor or heat, then they should be contained and removed using local exhaust equipment. Local capture equipment and systems should be designed only by an experienced engineer or industrial hygienist. Also, users of these devices must have proper training, or they may be ineffectively used.
Whether the emission source is a vacuum-pump discharge vent, a gas chromatograph exit port, or the top of a fractional distillation column, the local exhaust requirements are similar. The total airflow should be high enough to transport the volume of gases or vapors being emitted, and the capture velocity should be sufficient to collect the gases or vapors.
Despite limitations, specific ventilation capture systems provide effective control of emissions of toxic vapors or dusts if they are installed and used correctly. A separate, dedicated exhaust system is recommended. The capture system should not be attached to an existing hood duct unless fan capacity is increased and airflow to both hoods is properly balanced. One important consideration is the effect that such added local exhaust systems will have on the ventilation for the rest of the laboratory. Each additional capture hood will be a new exhaust port in the laboratory and will compete with the existing exhaust sources for supply air.
Downdraft ventilation has been used effectively to contain dusts and other dense particulates and high concentrations of heavy vapors that, because of their density, tend to fall. Such systems require special engineering considerations to ensure that the particulates are transported in the airstream. Here again, a ventilation engineer or industrial hygienist should be consulted if this type of system is deemed suitable for a particular laboratory operation.
8.C.10.1 Elephant Trunks
An elephant trunk, or snorkel, is a piece of flexible duct or hose connected to an exhaust system. It cannot effectively capture contaminants that are farther than about one-half a diameter from the end of the hose. Elephant trunks are particularly effective for capturing discharges from gas chromatographs, pipe nipples, and pieces of tubing if the hose is placed directly on top of the discharge with the end of the discharge protruding to the hose. In this case, the volume flow rate of the hose must be at least 110 to 150% of the flow rate of the discharge.
The capture velocity is approximately 8.5% of the face velocity at a distance equal to the diameter of the local exhaust opening. Thus, a 3-inch-diameter snorkel or elephant trunk having a face velocity of 150 fpm will have a capture velocity of only 1 fpm at a distance of 3 inches from the opening. Because the air movement velocity is typically at least 20 fpm, capture of vapors emitted at 3 inches from the snorkel will be incomplete. However, vapors emitted at distances of 2 inches or less from the snorkel opening may be captured completely under these conditions.
8.C.10.2 Slot Hoods
Slot hoods are specially designed industrial ventilation hoods intended to capture contaminants generated according to a specific rate, distance in front of the hood, and release velocity for specific ambient airflow. In general, if designed properly, these hoods are more effective and operate using much less air than either elephant trunks or canopy hoods. In order to be effective, however, the geometry, flow rate, and static pressure must all be correct. Typical slot hoods are shown in Figure 8.8. Each type has different capture characteristics and applications. If the laboratory worker believes that one of these devices is necessary, then a qualified ventilation engineer should be called to design the hood and exhaust system.
8.C.10.3 Canopy Hoods
The canopy hood is not only the most common local exhaust system but also probably the most misunderstood piece of industrial ventilation equipment. It is estimated by industrial ventilation experts that as many as
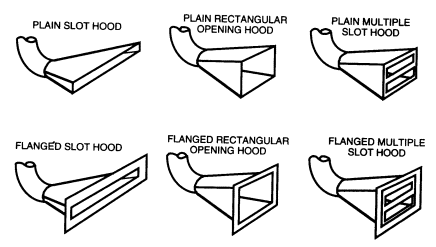
FIGURE 8.8 Typical specific ventilation hoods.
95% of the canopy hoods in use (other than in homes and restaurants) are misapplied and ineffective. The capture range of a canopy hood is extremely limited, and a large volume of air is needed for it to operate effectively. Thus, a canopy hood works best when thermal or buoyant forces exist that will move the contaminant up to the hood capture zone (a few inches below the opening). However, because canopy hoods are generally placed well above a contaminant source so that workers can operate underneath them, they draw contaminants past the worker's breathing zone and then into the exhaust system. If a canopy hood exists in a laboratory, it should be used only for nonhazardous service, such as capturing heated air or water vapor.
8.C.10.4 Flammable-Liquid Storage Cabinets
Flammable and combustible liquids should be stored only in approved flammable-liquid storage cabinets, not in a fume hood, out on the benches, or in a nonapproved storage cabinet.
Whether or not flammable-liquid storage cabinets should be ventilated is a matter for debate. One view is that all such cabinets should be vented by using an approved exhaust system, because it reduces the concentration of flammable vapors below the lower explosive limit inside the cabinet. A properly designed cabinet ventilation system will do this under most circumstances and results in a situation in which no fuel is rich enough in vapor to support combustion. However, there is still liquid in the cabinet and a source of fresh air provided by the ventilation system. All that is needed is an ignition source. The other view is that in most circumstances flammable-liquid storage cabinets should not be ventilated, because the cabinet is designed to extinguish a fire by depriving it of oxygen and ventilation defeats this purpose.
Both opinions are valid, depending on the conditions. Ventilation is prudent when the liquids stored in the cabinet are highly toxic or extremely odoriferous. Particularly odoriferous substances such as mercaptans have such a low odor threshold that even with meticulous housekeeping the odors persist; hence, ventilation may be desired.
If a ventilated flammable-liquid storage cabinet is used under a fume hood, it should not be vented into the fume hood above it. It should have a separate exhaust duct connected to the exhaust system. Fires occur most frequently in fume hoods. Fire from a fume hood may propagate into a flammable-liquid storage cabinet directly vented into the hood.
If a special-flammable storage cabinet ventilation system is installed, it should employ an AMCA-C type spark-resistant fan and an explosion-proof motor. Most fractional horsepower fans commonly used for this purpose do not meet this criterion and should not be used. If the building has a common fume hood exhaust system, then it is appropriate to hook a flammable-storage cabinet up to it if it must be ventilated.
8.C.11 General Laboratory Ventilation and Environmental Control Systems
General ventilation systems control the quantity and quality of the air supplied to and exhausted from the laboratory. The general ventilation system should ensure that the laboratory air is continuously replaced so that concentrations of odoriferous or toxic substances do not increase during the workday and are not recirculated from laboratory to laboratory.
Exhaust systems fall into two main categories: general and specific. General systems serve the laboratory as a whole, and include devices such as fume hoods and snorkels, as codes and good design practices allow.
Specific systems serve isotope hoods, perchloric acid hoods, or other high-hazard sources that require isolation from the general laboratory exhaust systems.
A general ventilation system that gives 6 to 12 room air changes per hour is normally adequate. More airflow may be required to cool laboratories with high internal heat loads, such as those with analytical equipment, or to service laboratories with large specific exhaust system requirements. In all cases, air should flow from the offices, corridors, and support spaces into the laboratories. All air from chemical laboratories should be exhausted outdoors and not recirculated. Thus, the air pressure in chemical laboratories should be negative with respect to the rest of the building unless the laboratory is also a clean room (see section 8.E.2). The outside air intakes for a laboratory building should be in a location that reduces the possibility of re-entrainment of laboratory exhaust or contaminants from other sources such as waste disposal areas and loading docks. (See Chapter 7, section 7.B.6.4, for further information.)
Although the supply system itself provides dilution of toxic gases, vapors, aerosols, and dust, it gives only modest protection, especially if these impurities are released into the laboratory in any significant quantity. Operations that can release these toxins, such as running reactions, heating or evaporating solvents, and transfer of chemicals from one container to another, should normally be performed in a fume hood. Toxic substances should be stored in cabinets fitted with an exhaust device. Likewise, laboratory apparatus that may discharge toxic vapors, such as vacuum pump exhausts, gas chromatograph exit ports, liquid chromatographs, and distillation columns, should vent to an exhaust device such as a snorkel.
The steady increase in the cost of energy in recent years, coupled with a greater awareness of the risks associated with the use of chemicals in the laboratory, has caused a conflict between the desire to minimize the costs of heating, cooling, humidifying, and dehumidifying laboratory air and the need to provide laboratory workers with adequate ventilation. However, cost considerations should never take precedence over ensuring that workers are protected from hazardous concentrations of airborne toxic substances.
8.C.11.1 Supply Systems
Well-designed laboratory air supply systems approach the ideal condition of laminar airflow, directing clean incoming air over laboratory personnel and sweeping contaminated air away from their breathing zone. Ventilation systems with well-designed diffusers that optimize "complete mixing" may also be satisfactory. Usually, several carefully selected supply air diffusers are used in the laboratory. Air entry through perforated ceiling panels may also successfully provide uniform airflow, but proper air distribution above the plenum is required. The plenum, diffuser, or perforated ceiling panels must be kept free of obstructions in order for the supply system to function properly.
8.C.11.2 Constant Air Volume
Constant air volume (CAV) air supply systems are the traditional design standard for laboratories. This method assumes constant exhaust and supply airflow rates through the laboratory. Although such systems are the easiest to design, and in some cases the easiest to operate, they have significant drawbacks due to their high energy consumption and limited flexibility. Classical CAV design assumes that all fume hoods operate 24 hours/day, 7 days/week, and at constant maximum volume. Adding, changing, or removing fume hoods or other exhaust sources for CAV systems requires rebalancing the entire system to accommodate the changes. Most CAV systems in operation today are unbalanced and operate under significant negative pressure. These conditions are caused by the inherent inflexibility of this design type, coupled with the addition of fume hoods not originally planned.
8.C.11.3 Variable Air Volume
Variable air volume (VAV) laboratories are rapidly replacing traditional CAV laboratories as the design standard. These systems are based on fume hoods with face velocity controls. As the users operate the fume hoods, the exhaust volume from the laboratory changes and the supply air volume must adapt to maintain a volume balance and room pressure control. An experienced laboratory ventilation engineer must be consulted to design these systems, because the systems and controls are complex and must be designed, sized, and matched so they operate effectively together.
8.C.12 Exhaust Systems
8.C.12.1 Individual Hood Fans
In traditional exhaust systems, each fume hood has its own exhaust fan. This arrangement has the following disadvantages and advantages: There is no way to dilute the fume hood effluent before release. The possibility of cross-contamination from one fume
hood discharge to another is eliminated. Providing redundancy and emergency power for this arrangement is difficult and expensive; however, a fan failure will affect only one fume hood. The potential to use diversity is limited, as is the potential to use VAV controls. The potential to treat individual fume hood exhaust (as opposed to treating all fume hood exhaust) is excellent. For the same reason, the potential to recover heat from individual fans is almost nonexistent. The maintenance requirement for these systems is considerable, because they contain many pieces of equipment and have many roof penetrations, which can cause leakage problems. The mechanical (shaft) space requirements, initial cost, and operating cost are higher than for alternative systems, such as manifolded systems.
8.C.12.2 Manifolded (Common Header) Systems
For compatible exhaust streams, providing a common, manifolded exhaust system is an attractive design alternative to individual hood fans. This design is chosen increasingly for new laboratory buildings and is compatible with VAV systems. Manifolded systems have the following advantages and disadvantages: The potential for mixing and dilution of high concentrations of contaminants from a single fume hood by the air exhaust from all the other fume hoods on the system is excellent. The cross-contamination potential from one hood to another is minimal. The potential to provide redundancy of exhaust fans and/or provide emergency power to these systems is excellent. Conversely, the effects of a fan failure are widespread and serious; hence, redundancy is required in most cases. The potential to take advantage of VAV diversity and flow variation is also excellent, as is the ability to oversize the system for future expansion and flexibility. The ability to treat individual exhausts is retained by using new in-line liquid scrubber technologies. The maintenance, operating, and initial costs of these systems are all lower than for individual hood fan systems, and these systems require fewer roof penetrations. The heat recovery potential for these systems is maximized by collecting all the exhaust sources into a common duct.
8.C.12.3 Hybrid Exhaust Systems
Certain types of fume hoods and exhaust sources, such as perchloric acid hoods, should not be manifolded with other types of fume hood exhausts. In large buildings where the designer wishes to take advantage of the benefits of manifolded exhaust systems but wishes to isolate a few exhaust streams, a combination, or hybrid, of these two types of systems is usually the most prudent and cost-effective alternative.
8.C.12.4 Exhaust Stacks
Proper stack design and placement are an extremely important aspect of good exhaust system design. Recirculation of contaminated air from the fume hood exhaust system into the fresh air supply of the facility or adjacent facilities may occur if stacks are not provided or if they are not designed properly to force the contaminated exhaust airstream up and into the prevailing wind stream. Stack design should take into account building aerodynamics, local terrain, nearby structures, and local meteorological information. An experienced laboratory consultant or an expert in atmospheric dispersion should be consulted to design exhaust stacks for a laboratory facility.
8.D ROOM PRESSURE CONTROL SYSTEMS
Laboratories and clean rooms usually require that a differential pressure be maintained between them and adjoining nonlaboratory spaces. This requirement may come from code considerations or from the intended use of the space. For example, NFPA Standard 45 states that "laboratory work units and laboratory work areas in which hazardous chemicals are being used shall be maintained at an air-pressure that is negative relative to the corridors or adjacent non-laboratory areas . . ." (NFPA, 1991d). This rule helps to prevent the migration of fire, smoke, and chemical releases from the laboratory space. Laboratories containing radiation hazards or biohazards may also be required by government agencies to maintain a negative pressure in order to contain these hazards. Clean rooms, on the other hand, are normally operated at a positive static pressure to prevent infiltration of particulates. (See sections 8.E.2 and 8.E.3 below for further information.)
8.E SPECIAL SYSTEMS
8.E.1
Glove Boxes
Glove boxes are usually small units that have multiple openings in which arm-length rubber gloves are mounted. The operator works inside the box by using these gloves. Construction materials vary widely, depending on the intended use. Clear plastic is frequently used, because it allows visibility of the work area and is easily cleaned.
Glove boxes generally operate under negative pressure, so that any air leakage is into the box. If the material being used is sufficiently toxic to require the use of an isolation system, the exhaust air will require special treatment before release into the regular exhaust system. These small units have a low airflow; therefore, scrubbing or adsorption (or both) can be easily accomplished.
Some glove boxes operate under positive pressure. These boxes are commonly used for experiments for which protection from atmospheric moisture or oxygen is desired. If positive-pressure glove boxes are used with highly toxic materials, they should be thoroughly tested for leaks before each use. Also, a method to monitor the integrity of the system (such as a shutoff valve or a pressure gauge designed into it) is required.
8.E.2 Clean Rooms
Clean rooms are special laboratories or work spaces in which large volumes of air are supplied through HEPA filters to reduce the particulates present in the room. Several classifications of clean rooms are commonly used. Clean room classifications refer to the number of particles larger than 0.5 microns in size per cubic foot of volume. Unfiltered ambient air has approximately 500,000 to 1,000,000 particles per cubic foot. Certain pharmaceutical, microbiological, optical, and microelectronic facilities require clean rooms of differing classifications from Class 10,000 to Class 10 or lower. Special construction materials and techniques, air handling equipment, filters, garments, and procedures are required, depending on the cleanliness level of the facility. A laboratory consultant or expert in clean room operation should be consulted before a clean room is worked in or built.
8.E.3 Environmental Rooms and Special Testing Laboratories
Environmental rooms, either refrigeration cold rooms or warm rooms, for growth of organisms and cells, are designed and built to be closed air circulation systems. Thus, the release of any toxic substance into these rooms poses potential dangers. Their contained atmosphere creates significant potential for the formation of aerosols and for cross-contamination of research projects. These problems should be controlled by preventing the release of aerosols or gases into the room. Special ventilation systems can be designed, but they will almost always degrade the temperature and humidity stability of the room. Special environmentally controlled cabinets are available to condition or store smaller quantities of materials at a much lower cost than in an environmental room.
Because environmental rooms have contained atmospheres, people who work inside them must be able to escape rapidly. Doors for these rooms should have magnetic latches (preferable) or breakaway handles to allow easy escape. These rooms should have emergency lighting so that a person will not be confined in the dark if the main power fails.
As is the case for other refrigerators, volatile flammable solvents should not be used in cold rooms (see Chapter 6, section 6.C.3.1). The exposed motors for the circulation fans can serve as a source of ignition and initiate an explosion. The use of volatile acids should also be avoided in these rooms, because such acids can corrode the cooling coils in the refrigeration system, which can lead to the development of leaks of refrigerants. Other asphyxiants such as nitrogen gas should also be avoided in enclosed spaces. Oxygen monitors and flammable gas detectors are recommended when the possibility of a low oxygen or flammable atmosphere in the room exists.
8.E.4 Biological Safety Cabinets and Biosafety Facilities
Biological safety cabinets (BSCs) are common containment and protection devices used in laboratories working with biological agents. BSCs and other facilities in which viable organisms are handled require special construction and operating procedures to protect workers and the environment. Conventional laboratory fume hoods should never be used to contain biological hazards. Biosafety in Microbiological and Biomedical Laboratories (U.S. DHHS, 1993) and Biosafety in the Laboratory: Prudent Practices for the Handling and Disposal of Infectious Materials (NRC, 1989) give detailed information on this subject.
8.F MAINTENANCE OF VENTILATION SYSTEMS
Even the best-engineered and most carefully installed ventilation system requires routine maintenance. Blocked or plugged air intakes and exhausts, as well as control system calibration and operation, can alter the performance of the total ventilation system. Filters become loaded, belts loosen, bearings require lubrication, motors need attention, ducts corrode, and minor components fail. These malfunctions, individually or collectively, can affect overall ventilation performance.
Facility-related environmental controls and safety systems, including fume hoods and room pressure con-
trols, fire and smoke alarms, and special alarms and monitors for gases, should thus be inspected and maintained on a regular basis.
Each laboratory should be evaluated periodically for the quality and quantity of its general ventilation and any time a change is made, either to the general ventilation system for the building or to some aspect of local ventilation within the laboratory. The size of a room and its geometry, coupled with the velocity and volume of supply air, determine its air patterns. Airflow paths into and within a room can be determined by observing smoke patterns. Convenient sources of smoke for this purpose are the commercial smoke tubes available from local safety and laboratory supply companies. If the general laboratory ventilation is satisfactory, the movement of supply air from corridors and other diffusers into the laboratory and out through hoods and/or other exhaust sources should be relatively uniform. There should be no areas where air remains static or areas that have unusually high airflow velocities. If stagnant areas are found, a ventilation engineer should be consulted, and appropriate changes should be made to supply or exhaust sources to correct the deficiencies.
The number of air changes per hour within a laboratory can be estimated by dividing the total volume of the laboratory (in cubic feet) by the rate at which exhaust air is removed (in cubic feet per minute). For each exhaust port (e.g., hoods), the product of the face area (in square feet) and the average face velocity (in linear feet per minute) will give the exhaust rate for that source (in cubic feet per minute). The sum of these rates for all exhaust sources in the laboratory yields the total rate at which air is being exhausted from the laboratory. The rate at which air is exhausted from the laboratory facility should equal the rate at which supply air is introduced to the building. Thus, decreasing the flow rate of supply air (perhaps to conserve energy) decreases the number of air changes per hour in the laboratory, the face velocities of the hoods, and the capture velocities of all other local ventilation systems.
Airflows are usually measured with thermal anemometers or velometers. These instruments are available from safety supply companies or laboratory supply houses. The proper calibration and use of these instruments and the evaluation of the data are a separate discipline. An industrial hygienist or a ventilation engineer should be consulted whenever serious ventilation problems are suspected or when decisions on appropriate changes to a ventilation system are needed to achieve a proper balance of supply and exhaust air.
All ventilation systems should have a device that readily permits the user to monitor whether the total system and its essential components are functioning properly. Manometer, pressure gauges, and other devices that measure the static pressure in the air ducts are sometimes used to reduce the need to manually measure airflow. "Telltales" and other similar simple devices can also serve as indicators of airflow. The need for and the type of monitoring device should be determined on a case-by-case basis. If the substance of interest has excellent warning properties and the consequence of overexposure is minimal, the system will need less stringent control than if the substance is highly toxic or has poor warning properties.