APPENDIX C
BASE CASE ONCE-THROUGH URANIUM FUEL CYCLE FOR LIGHT-WATER REACTOR WITH GEOLOGICAL REPOSITORY*
This is the flowsheet for the fueling of a 1,000 MWe light-water reactor (LWR) for 1 year of operation with a 80% capacity factor. A once-through fuel cycle is assumed. The two columns on the left show the amount of uranium at the output of each step in metric tons (1 T = 1,000 kg) and the percent enrichment of 235U. The right-hand column shows the percentage of uranium lost in the process. The technical data used in this fuel cycle are taken from the textbook Nuclear Chemical Engineering by Benedict, Pigford, and Levi, 2nd edition, McGraw Hill, New York, 1981.

|
 |
|
|
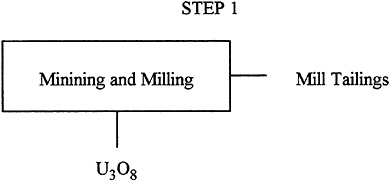
Description: |
In this step, the mined ore is processed to concentrate the uranium. A combination of methods is used including leaching, precipitation, solvent extraction, and ion exchange. The output stream is U3O8. The main byproduct stream is mill tailings that contain a small fraction of the initial uranium and all its initial radioactive daughter products. |
|
Input: |
The input is the mined ore and the amount depends on the grade of the ore, typically around 0.15–0.2%. |
|
Output: |
228 T of U3O8. |
|
Environment: |
The principal impact is due to disposal of mill tailings that can be dispersed by rain and wind and produce a significant dose to the public. |
|
Siting Issues: |
The ore processing activity can be noisy and dusty. It is usually carried out near the mine and removed from population. |
|
Public Health: |
This process is a predominant contributor to occupational dose and dose to the public. |
|
 |
|
|
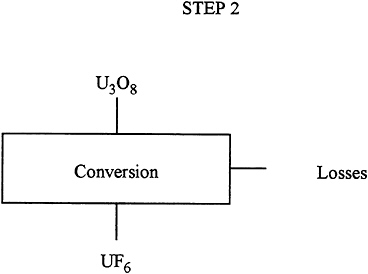
Description: |
In this step, the U3O8 is chemically processed to convert it into UF6, a gas that is the input for the gaseous diffusion process. |
|
Input: |
228 T of U3O8. |
|
Output: |
284 T of UF6 |
|
Environment: |
There is very little impact, and it is mostly associated with uranium losses. |
|
Siting Issues: |
This process is like many chemical plants. It would be restricted to an industrial zone. Uranium separated from its daughter products is only weakly radioactive. |
|
Public Health: |
This process would be a small contributor to the cycle's public health effects. |
|
 |
|
|
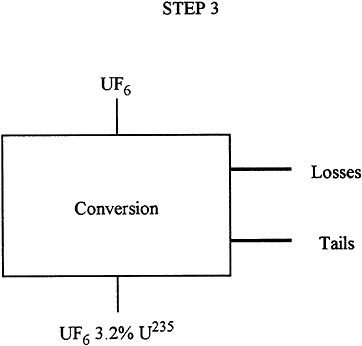
Description: |
In Step 3, the natural uranium UF6 is put through the enrichment plant to increase the percentage of 235U from 0.71 to 3.2 at a tails assay of 0.3%. |
|
Input: |
284 T UF6 (200 T uranium 0.71% 235U). |
|
Output: |
40.1 T of UF6 product (27.2 T uranium 3.2% 235U). 242.4 T UF6 tails (164 T uranium 0.3% 235U). |
|
Losses: |
0.5% (1 Ton natural uranium). |
|
Environment: |
The enrichment plant is very large and uses moderate-to-large amounts of electricity (depending on the technology). Uranium losses lead to a small amount of environmental degradation. The tails stream constitutes a major output stream, the disposition of which has not been decided. |
|
Siting Issues: |
A large nonurban site is needed. |
|
Public Health: |
There are some occupational health issues—small public health concerns. |
|
 |
|
|
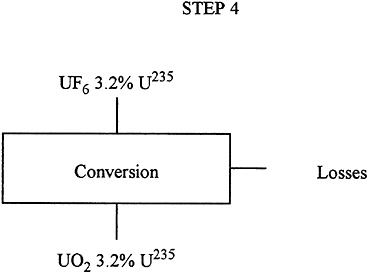
Description: |
The enriched UF6 must now be converted to UO2, which is the material used in reactor fuel elements. This is a chemical process. |
|
Input: |
27.2 T uranium 3.2% 235U as UF6. |
|
Output: |
27.1 T uranium 3.2% 235U as UO2. |
|
Losses: |
0.5% uranium. |
|
Environment: |
There is very little impact, and it is mostly associated with uranium losses. |
|
Siting Issues: |
The process is like many chemical plants. It would be restricted to an industrial zone. Uranium separated from its daughter products is only weakly radioactive. |
|
Public Health: |
This process would be a small contributor to the cycle's public health effects. |
|
 |
|
|
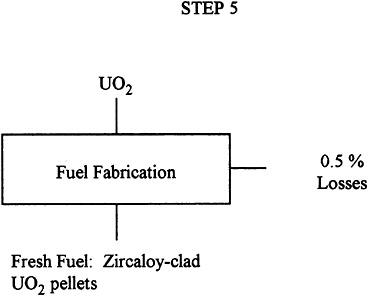
Description: |
In this step, the UO2 is compacted into pellets (0.3 to 0.4 inches diameter x 0.5 inches long), and these pellets are sutured and placed into Zircaloy tubes about 12 feet long. These tubes are assembled in square arrays of about 17×17 tubes per fuel element. |
|
Input: |
27.1 T uranium 3.2% 235U as UO2. |
|
Output: |
26.9 T uranium 3.2% 235U as UO2. |
|
Losses: |
0.5% uranium. |
|
Environment: |
There is very little impact, and it is mostly associated with uranium losses. |
|
Siting Issues: |
The process is like many chemical plants. It would be restricted to an industrial zone. Uranium separated from its daughter products is only weakly radioactive. |
|
Public Health: |
This process would be a small contributor to the cycle's public health effects. |
|
Other Issues: |
Uranium in this step is well below the enrichment useful for nuclear weapons production. |
|
 |
|
|
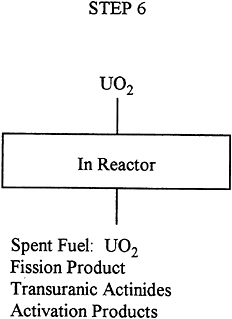
Description: |
The fabricated fuel is put in the reactor and used to sustain the chain reaction. It is typically irivaluated for about 33,000MWd per T of uranium. The enrichment at the end of the run of 1,100 days is about 0.83% 235U. |
|
Input: |
26.9 T uranium 3.2% 235U as UO2. |
|
Output: |
25.9 T uranium 0.83% 235U as UO2. |
|
By-products: |
See enclosed tables giving concentrations of major radionuclides in spent pressurized water reactor fuel (Appendix A. 1, ORNL/TM-7431) in terms of curies, watts, and ingestion hazard as a function of time. Tritium concentrations are about 22,000 Ci/yr (818 Ci/MTHM). About two-thirds is from ternary fission. About one-third is from an assumed 2 ppm of lithium in the fuel, but this number is speculative. See attached sheet for the 14C inventories in the fuel and their distribution (ORNL/TM-6051). |
|
Environment: |
During operation, all reactors release small amounts of radioactivity, including tritium, noble gasses, and 14C. |
|
Siting Issues: |
Tend to be distinctive, visible, and controversial, primarily because of potential accidents. |
|
Public Health: |
Routine releases are generally small, but the worst of major accidents could result in substantial offsite doses. |
|
Proliferation: |
UO2 input fuel has an enrichment of 3.2 235U that is well below that required for use in nuclear explosives. The output contains 0.246 T of plutonium, which is mixed with the highly radioactive fission products, so it is considered to be self-protected. |
|
 |
|
|
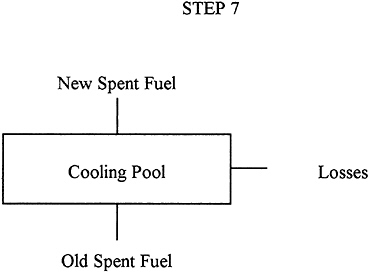
Description: |
The fuel assemblies are unloaded from the reactor and stored in the reactor cooling pool to remove decay heat and provide shielding from radiation. In the United States, reprocessing of commercial fuel is not yet economic, so reprocessing is not being done. Thus, all commercial fuel at power plants is being stored in pools at the reactor site. Some fuel has remained stored in this way for over 20 years. |
|
Input: |
Spent-fuel assemblies discharged from the reactor. |
|
Output: |
Spent fuel, typically decayed for many years |
|
Environment: |
A small fraction of rods will begin to leak while in storage. Since they are under water, most leaked radioactivity will be trapped in the water. Constant monitoring will catch leaks shortly after they occur. Once identified, leaking fuel assemblies are placed in special containers maintained for this purpose. |
|
Public Health: |
There is some exposure of workers at facility. There is very little impact on the public from this operation. |
|
Losses: |
Very small. |
|
 |
|
|
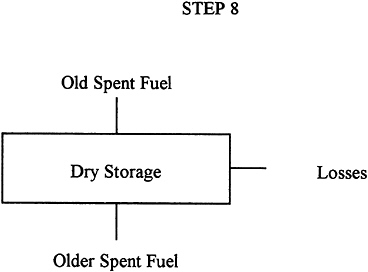
Description: |
Once all storage pools are full, a number of utilities are proposing to store them in special shielded storage casks on or near the surface. The elements would typically have been in cooling pools for many years and much of their radioactivity would have been reduced by decay. The casks would be designed to conduct the small amount of heat still being generated. Presumably, the fuel would be quite safe in a storage cask until it was shipped to a geologic repository. |
|
Input: |
Spent-fuel elements after significant cooling in pool. |
|
Output: |
Spent-fuel elements on their way to a repository or monitored retrievable storage facility. |
|
Environment: |
A small number of leaks might develop, but if casks are well sealed, very little loss to environment would be expected. |
|
Public Health: |
There is some exposure of workers at facility. There is very little impact on the public from this operation. |
|
Losses: |
Very small. |
|
 |
|
|
Description: |
In this step, the spent fuel would be sent to the repository for final emplacement. At this point, they would be removed from the cask and possibly repackaged into a tighter configuration, or they might be sequestered in their casks. If repackaging is done, some occupational exposure would be expected along with some small release of radioactivity. Once emplaced, the container is designed to survive 1,000 years, and so no leakage from the repository should occur before that time. At times greater than 1,000 years, the container is assumed to have failed and the leakage rate is then controlled by the rate of leaching and migration to the repository boundary. |
|
Input: |
Spent fuel. |
|
Output: |
None. Permanent emplacement. |
|
Environment: |
Gradual inflow of water, waste-form degradation, radionuclide dissolution, and transport is assumed. Releases are predicted to be extremely small and to occur over extremely long times. Intrusion is possible (e.g., drilling through a waste package). |
|
Public Health: |
Impacts are predicted to be extremely small, except to an intruder. |
|
Losses: |
Loss rate is controlled by leaching and migration (inventory of radioactivity 1,000 years after emplacement). |












