APPENDIX E
DEFENSE WASTES-ADDITIONAL INFORMATION
HANFORD DISPOSAL COSTS FOR HIGH-LEVEL WASTE CANISTERS
Costs for disposal of the Hanford vitrified high-level waste canisters can be broken into two elements: canister production at the Hanford site and canister transport to and disposal at the repository. Both of these elements are considered in the brief analysis that follows.
Canister Preparation
A high-level waste (HLW) vitrification plant is planned to be built and operated at the Hanford site. The plant will process wastes from the Hanford HLW tanks after appropriate treatment to produce vitrified waste in canisters for shipment to the HLW repository. A Department of Energy (DOE) report describes three treatment options for waste disposal and estimates the number of canisters to be produced as a result of each option, assuming that ''defense canisters" are used (DOE, 1993a). The options are
|
Option 1 |
sludge wash |
38,000 canisters |
|
Option 2 |
actinide separation |
11,000 canisters |
|
Option 3 |
extensive separations |
2,000 canisters |
The canister considered is a thin-wall (0.375 in.) advanced design with external dimensions of 2 ft. (diameter) by 10 ft. (length). The numbers of canisters are best estimates based on various assumptions about how much material can be removed from the tanks.
The current best estimate available on the capital cost of the Hanford waste vitrification plant is $1.7 billion (Reckman, 1993). The plant as currently designed has a capacity of 372 canisters per year and a design life of 40 years, yielding total plant production of 14,880 canisters if the design production rate is achieved. (European vitrification plants are not meeting design production rates due to melter design and material problems, coupled with melter replacement difficulties. As a result, British Nuclear Fuels has recently undertaken the design and construction of an additional process line for its windscale vitrification plant at Sellafield, U.K.)
Labor figures are not available for the operation of the Hanford plant. However, the Savannah River Site's vitrification plant has a staff of 1,150. Assuming a 1,100-person staff at Hanford, and an average staff wage, including fringe benefits of $45,000 per year, the total labor cost over the life of a plant is $2 billion.
Current limitations and the design status indicate that if option 1, sludge wash, is selected, it will be necessary to build three plants, either in parallel or in series, depending on the time DOE, with the support of Congress, is prepared to take to complete the vitrification process. To a first-order approximation, there does not appear to be any significant economy-of-scale resulting from building three plants, except that plant design cost would not be expected to be duplicated. Therefore, the cost of the sludge wash option and the actinide separation option would appear to be the same per canister, based on a total life-cycle cost per plant of approximately $4 billion and either one or three plants depending on the option.
The extensive separations process offers the prospect that less vitrification plant capacity would be required to produce the reduced number of canisters resulting from the use of that process. For chemical process plants, the plant size should decrease with the cube root of throughput, implying a rough scaling factor of 0.33. Applying that factor to the life-cycle costs for option 2 is probably optimistic, since it surely understates the engineering and operating staff costs. However, it seems to be an adequate approach to a first approximation. Application of the scaling factor to the life-cycle cost for option 2 yields a comparison cost of about $2.3 billion.
The production costs for each of the options, to a first-order approximation based on this simplistic analysis, are as follows:
|
Option 1 |
sludge wash |
production cost, $12 billion |
$316,000 per canister |
|
Option 2 |
actinide separation |
production cost, $4 billion |
$364,000 per canister |
|
Option 3 |
extensive separations |
production cost, $1.8 billion |
$1.80 million per canister |
Canister Disposal
DOE has performed a series of studies on the cost to the government of defense waste transport and storage in the repository. Single-repository and two-repository cases have been considered. This analysis considers only the single-repository case. The most recent available information is DOE's "Preliminary Draft of Cost Estimates for the Hanford Tank Waste Remediation Options Study Cases" (DOE, 1993a). A number of other reports provide detail on the methods by which costs to the government for each option are considered (DOE 1987, 1990a, b, 1993a, b). Elements include costs for transportation, tunneling, borehole drilling, and
emplacement. The analysis provides results that are almost totally dependent on the number of canisters to be emplaced. The disposition cost per canister for each of the three options is
|
Option 1 |
sludge wash |
$185,000 per canister |
|
Option 2 |
actinide separation |
$196,000 per canister |
|
Option 3 |
extensive separations |
$189,000 per canister |
Total per-canister disposal costs, combining the per-canister cost for production and disposal for the three options, are
|
Option 1 (base case) |
$500,000 per canister |
|
Option 2 |
$560,000 per canister |
|
Option 3 |
$2.00 million per canister |
SAVANNAH RIVER SITE PLANS FOR A DEFENSE WASTE PROCESSING FACILITY
Members of the STATS Subcommittee on Separations visited the Savannah River Site in South Carolina in February 1992, and were presented with plans for, and the status of, conversion of existing defense wastes at the site into low-level saltstone for onsite storage and high-level glass in canisters ("glass logs") for eventual shipment to and storage in the first federal repository for HLW. In April 1992, the entire subcommittee was given an overview of these plans at one of the subcommittee meetings. The HLW would consist of about 7,000 canisters about 2 ft. in diameter and 10 ft. long, each with about 3,700 lb. of glass. This would use about 4,000 tons of the federal limitation of 70,000 Mg of heavy metal content on the first repository.
The Defense Waste Processing Facility (DWPF) at the Savannah River Site (SRS) in South Carolina has been under construction for about 10 years and is now nearly completed. The DWPF process appears to represent an acceptable solution to the disposition of defense wastes at SRS. Results from initial DWPF operations should provide important insights for the designers of the Hanford Tank Waste Remediation System.
DWPF Process Description
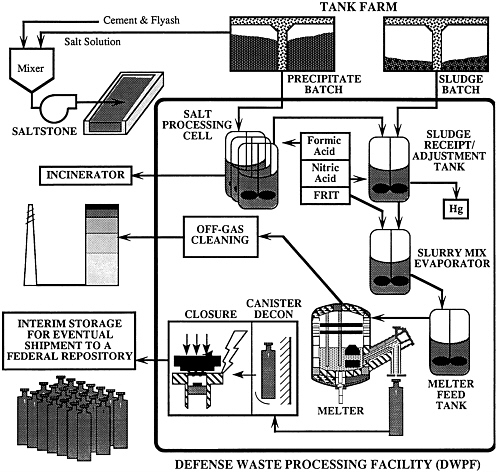
The following DWPF process description was supplied to the subcommittee by Westinghouse Savannah River Company and is believed to represent an up-to-date effective summary of the process1 (Baxter, 1986; Baxter, 1988). A diagram of the process is shown in Figure E-1.
WASTE PROCESSING IN TANK FARMS
The SRS waste is currently stored onsite in carbon steel tanks and exists in three forms: sludge, salt solution, and saltcake (formed through concentration of salt solution [Maher et al., 1981]). The sludge, which makes up approximately 10% of the stored waste by volume, consists primarily of precipitates of the hydroxides of iron, aluminum, and manganese. The salt (saltcake and salt solution) is largely sodium nitrate, sodium aluminate, and sodium hydroxide. The sludge contains most of the radioactivity in the waste, including small amounts of actinides not recovered in the reprocessing plants and most of the fission products, except for 137Cs. The salt fraction contains most (around 95%) of the 137Cs.
The salt (saltcake and salt solution) is decontaminated for disposal as LLW by removing the radionuclides by precipitation and sorption. Dissolved saltcake and salt solutions are pumped to an existing waste tank in the tank farm for pretreatment. A solution of sodium tetraphenylborate is added to form an insoluble double salt with potassium and cesium. At the same time, a small amount of sodium titanate slurry is added to adsorb residual strontium and plutonium in the salt solution.
The precipitate slurry is concentrated by continuous filtration, washed to remove the soluble salt, and pumped to the DWPF. After further processing, the concentrated radionuclides from the salt solution are immobilized as borosilicate glass (see below). The decontaminated salt solution is blended with Portland cement, slag, and fly ash in a separate facility for disposal as LLW (saltstone process).
Sludge waste is also pretreated by alkaline washing in existing waste storage tanks. The object is to dissolve nonradioactive ingredients such as aluminum, so that they may be processed with the salt solution as LLW rather than treated as the more costly HLW glass. High-aluminum sludges are leached with strong caustic to dissolve about 75% of the hydrated aluminum compounds and reduce the volume of this type of sludge by about 50%. All sludges are washed with alkaline solutions to reduce the soluble salt content of the sludge slurry. Current plans are to accumulate about 2,500 m3 of washed sludge slurry and then to use the sludge slurry as batch feed to the DWPF. Each batch of sludge would supply the DWPF for 2 to 3 years of operation.
PRECIPITATE PROCESSING
Within the vitrification building, tetraphenylborate precipitates are processed to remove organic material. About 90% of the phenyl groups of the tetraphenylborate salts are removed through reaction with formic acid and water (precipitate hydrolysis). The products of this reaction are an organic fraction (primarily benzene) and an aqueous product. The aqueous product contains the cesium as soluble formate salts. The aqueous product also contains some boric acid, formic acid, and about 10% of the original phenyl groups in water-soluble forms (such as phenol and phenylboric acid), along with the insoluble radionuclides—loaded titanates. The aqueous product is collected and fed to the sludge receipt and adjustment tank prior to vitrification.
SLUDGE PROCESSING AND ADJUSTMENT
The washed sludge slurry and precipitate hydrolysis product are mixed together in the sludge receipt and adjustment tank. Formic acid is also added to the tank, which reduces any mercury ion present (a minor component of the sludge) to the elemental state. The formic acid also reduces the yield stress of the slurry and provides a reductant to the melt, which minimizes foaming. The mercury is steam distilled from the slurry and is eventually recovered in reusable, metallic form.
WASTE FORM PRODUCTION
The slurry is transferred to the slurry mix evaporator where premelted borosilicate glass frit is added (Boersma, 1984; Weisman et al., 1988). Approximately two-thirds of the necessary frit is fed directly to the evaporator. The remaining one-third of the frit is first used for canister decontamination (frit blasting) and then is added to the evaporator. The frit-waste-formate slurry is concentrated to about 45 wt percent total solids by boiling. This mixture is transferred to the melter feed tank, which provides feed to the melter.
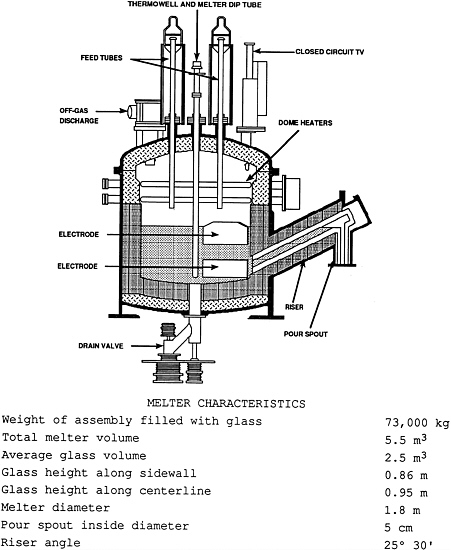
Vitrification of SRS waste is accomplished in a slurry-fed, joule-heated melter (Figure E-2). The feed slurry is introduced from the top of the melter. A crust, or cold cap, is formed on the surface of the melt pool as the water evaporates from the feed slurry and is removed via the off-gas system. Two pairs of diametrically opposed electrodes supply electric power directly to the melt. The nominal glass temperature beneath the cold cap is 1150° C, but varies throughout the melter. The cold cap melts from the bottom and forms the borosilicate waste glass matrix. For a nominal pour rate of 100 kg/h and a nominal glass melt weight of 6,500 kg in the melter, the average residence time in the melter is about 65 hours. The dome of the melter contains four pairs of resistance lid heaters that are used to provide the heat for start-up, as well as any supplemental heat required during glass production.
Glass is normally removed from the bottom of the DWPF melter through a riser and pour spout (Figure E-2). The canister, on the pour turntable, is connected to the melter by a bellows
assembly, which seals the canister connection. Pouring is accomplished by drawing a vacuum on the spout relative to the melter. Canister filling is monitored by both a gamma-and a neutron-level detection system and by a weight system. When the desired level of fill in the canister is achieved, pouring is stopped by equalization of the pressure between the melter and pour spout. After pouring is stopped, venting of the canister to the off-gas system is continued for about 30 minutes to collect any volatile radionuclides (such as 137Cs), which may be emitted from the surface of the glass. The canister is then rotated from beneath the pour spout. The canister is temporarily sealed with a tapered plug that shrink-seals as cooling continues, creating a leaktight seal of better than 2 × 10-4 cc-atm/s of helium.
Frit slurry blasting is used to remove contamination and metal oxides from the canister surface. Cleaning is performed by rotating the canister in an enclosed chamber and using air-injected wet glass-frit blasting on all exposed surfaces. The used frit slurry is sent to the slurry-mix evaporator for melter feed preparation.
Finally, the canister is sealed by welding a plug with a 12.7-cm diameter into the canister nozzle. After decontamination and drying, the temporary seal is pushed down in the canister neck, exposing clean metal for the permanent plug weld. The joint is clamped tightly and made as a resistively heated diffusion bond under pressure by a high amperage electric current. The weld produced is sufficiently leaktight based on experimental evidence, to approximately 1 × 10-9 cc-atm/s of helium and of comparable strength to the base metal to meet the required specification.
INTERIM STORAGE
The filled, decontaminated, and sealed canisters are moved by a shielded transport vehicle and stored in the glass waste storage building. Air is forced across the canisters' surfaces for cooling and to prevent overheating of the building. Although there should be no contamination in the building, the discharged air is passed through high-efficiency particulate filters so that any potential contamination is retained by the circulation system filters.
ULTIMATE STORAGE
The canisters are destined to be eventually shipped by rail to the first federal HLW repository. (Depending on when the repository becomes available, additional interim storage may be required at SRS.) Considerable effort has been spent over the past 5 years on tests to validate this waste form with the Nuclear Regulatory Commission for acceptance at the repository. It is anticipated that this waste will be suitable for acceptance by the agency for ultimate disposal in the first repository.
Comments on the DWPF Process
One problem with the DWPF process that is recognized by SRS personnel is the choice of tetraphenylborate to precipitate 137Cs from supernatant solutions. This choice leads to many different organic compounds arising from thermal degradation. Some, such as benzene, have to be recovered and decontaminated as by-products. Still others have caused plugging problems in pilot-plant equipment. The reason that adsorption of 137Cs on an inorganic zeolite—as was successfully done at West Valley, New York—was not selected at SRS is the resultant significant increase in the number of high-level canisters due to the volume of this zeolite. (This was not a significant consideration at West Valley, because there is only one tank of HLW to dispose of at that site.) However, the added cost burden that would result from zeolite absorption may be less than the costs of dealing with degradation products of tetraphenylborate at SRS. For future plants, it is possible that some alternative option would be superior to either the SRS or West Valley methods of 137Cs collection.
REFERENCES
Federal Register. 1987. Civilian radioactive waste management; calculating nuclear waste fund disposal fees for Department of Energy defense program waste. Federal Register. 52(161). August 20.
U.S. Department of Energy (DOE). 1990a. Report on the Nuclear Waste Fund Fee Adequacy: An Assessment. DOE/RW-0291P. Washington, D.C.: U.S. Department of Energy.
U.S. Department of Energy (DOE). 1990b. Preliminary Estimates of the Total-System Cost for the Restructured Program: An Addendum to the May 1989 Analysis of the Total-System Life Cycle Cost for the Civilian Radioactive Waste Management Program . DOE/RW-0295P. Washington, D.C.: U.S. Department of Energy.
U.S. Department of Energy (DOE). 1993a. Preliminary Draft of Cost Estimates for the Hanford Tank Waste Remediation Options Study Cases. Office of Civilian Radioactive Waste Management, U.S. Department of Energy, Washington, D.C.
U.S. Department of Energy (DOE). 1993b. Preliminary Draft of Cost Estimates for the Hanford Tank Waste Remediation System Study, Two Repository Cases. Program Control and Administration Division, U.S. Department of Energy, Washington, D.C.
SELECTED BIBLIOGRAPHY
Baxter, R. G. 1986. Design and construction of the Defense Waste Processing Facility project at the Savannah River Plant. In Waste Management '86, Vol. 2. Tucson: Arizona Board of Regents.
Baxter, R. G. 1988. Defense Waste Processing Facility Wasteform and Canister Description. DOE report DP-1606, Revision 2.
Boersma, M. D. 1984. Process technology for the vitrification of defense high-level waste at the Savannah River Plant. American Nuclear Society—Fuel Reprocessing and Waste Management Proceedings, 1:131-47.
Maher, R., L. F. Shafranek, J. A. Kelley, and R. W. Zeyfang. 1981. Solidification of the Savannah River Plant high-level waste. American Nuclear Society Trans. 39:228.
Weisman, A. F., L. M. Papouchado, J. R. Knight, and D. L. McIntosh. 1988. High-level waste vitrification at the Savannah River Plant. Pp. 203-210 in R. G. Post, ed., Waste Management '88, Vol. 2. Tucson: Arizona Board of Regents.