CHAPTER TWO—
SUMMARIES OF S&T PROPOSALS AND RADIONUCLIDES RELEASE SCENARIOS IN REPOSITORY
This chapter gives a brief overview of the source and characteristics of radionuclides in spent fuel, summaries of Separation and Transmutation (S&T) proposals being considered for commercial spent-fuel reprocessing and defense high-level waste treatment, and details of the performance of the radionuclides involved in a repository.
SOURCE AND CHARACTERISTICS OF RADIONUCLIDES IN SPENT FUEL
Radioactivity Produced by Reactors
After a nuclear reactor has been run at full power for a few months a large number of new radioactive species are produced. One of the nuclear reactions responsible for this radioactivity production is the neutron, gamma reaction often written (n, γ). In this reaction the target nucleus captures a neutron and emits one or more γ-rays. The target nucleus increases its mass number by one but does not change its atomic number. An example of such a reaction is the capture of a neutron by 238U to produce 239U plus some γ-rays. The product of the (n, γ) may be stable or radioactive. The transuranic isotopes shown in Figure 2-1 exhibit three forms of instability: α-decay, β-decay, and spontaneous fission (SF). None of the nuclides of atomic number greater than 92 are stable. It is also interesting to notice that the (n, γ) reaction can repeat itself and form a series of (n, γ) steps such as exhibited by Pu going from 238Pu to 246Pu. Decay by the emission of an ordinary negative electron called β-decay results in an increase of one in the atomic number. Note that the light-water reactor (LWR) starts with only 235U and 238U in the chemical compound uranium dioxide as fuel, and after a substantial run at power will have produced all 51 of the nuclear species shown in Figure 2-1, although those heavier than 244Cm are present in amounts so small as to be negligible. These 51 species are called the actinides and are of particular interest to the STATS study. The actinides form one important component of the spent fuel produced in LWRs. Since the actinides other than uranium are all produced by nuclear reactions in the fuel, all of them are born inside the uranium dioxide fuel. A second type of reaction that can occur is neutron capture that leads to the fissioning of the target nuclide. In the fission process the capture of a neutron imparts enough energy to the struck nucleus to cause it to break apart into pieces called fission products. During the process, γ-rays and energetic neutrons are given off. Some of the actinides will fission upon capture of a low-energy neutron, e.g., 239Pu; almost all will fission if the incident neutron has an energy above 1 MeV. The products of fission are neutron rich and almost all are unstable against β-decay. These fissions will almost all take place inside the uranium dioxide fuel. There are several hundred different fission products produced by these reactions which cause the spent reactor fuel to be highly radioactive and generate substantial amounts of decay heat. Just after shut down the decay heat from nuclear fuel in the reaction will still produce heat at about 7% of the full power output.
Another source of spent fuel radioactivity will be from a few actinides that decay by spontaneous fission, that is, they undergo fission without a neutron capture or any other source of energy being required. They differ from neutron-induced fission in that neutron-induced fission stops when the chain reaction stops but spontaneous fission continues to decay away at its characteristic half-life. Spontaneous fission neutrons can present shielding problems during reprocessing and fuel fabrication.
Neutron activation products are also generated when nuclear fuel is used to produce power. These are radioactive species formed by neutron bombardment of non-fuel
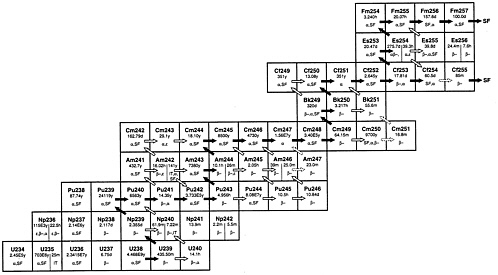
FIGURE 2-1 Neutron capture paths of importance for actinides.
materials such as fuel cladding and other structures inside the reactor. An example of such a case is that trace amounts of nitrogen in the fuel matrix produce 14C, a long-lived activation product of concern in some waste disposal circumstances.
The actinides, fission products, and activation products taken together are the radioactive waste that must be sequestered in some way so that they do not enter the biosphere in unacceptable concentrations. Today, these three radionuclide groups are contained in the uranium dioxide fuel matrix or structural materials of spent fuel assemblies. Almost all of the spent fuel is stored in pools at the reactor sites. Uranium dioxide is a ceramic material that effectively contains radioactive elements under most circumstances. However, storage of spent fuel at the reactor in cooling pools is considered only a temporary solution (less than a century). It is possible and legal to store the spent fuel out of pool up to 60-70 years. After that, a permanent solution will be required. The current government plan is to place the spent fuel, following encapsulation, deep underground in a stable geological formation.
All nuclides have some propensity for absorbing neutrons that changes them into a different species with different nuclear properties. This process is called transmutation. The goal of transmutation for waste management purposes is to convert a long-lived radionuclide that is potentially troublesome at a waste disposal site to a shorter-lived or stable nuclide by exposing the troublesome nuclide to a high flux of neutrons for a sustained time. This has the effect of reducing the long-term toxicity of the waste because most of the waste constituents would then decay to a non-radioactive nuclide in a short time. Since some of the nuclides in the spent fuel will produce unwanted nuclides if bombarded with neutrons, it becomes essential to separate carefully the nuclides before the neutron bombardment so that only those that produce the desired outcome will be included among those exposed to the neutron flux. This is the basic premise of the S&T method of waste disposal. There are several problems that make this more difficult than it might seem. First, in many cases a fraction of the neutron captures produce another long-lived radionuclide, which does little to achieve the transmutation goal stated above. A second problem is that the source of the neutrons is a nuclear reactor of some kind. Thus, as that reactor operates it will produce more of the same nuclides that are being destroyed, so the observed reduction will be the net of destruction minus production.
Toxicity Considerations and Isotopes of Concern
Toxicity Index
There are 200 to 300 different radioactive species generated by the operation of a nuclear reactor, primarily as a
result of neutron capture and neutron-induced fission. The adverse impact of the various radionuclides varies because of the differences in the chemical behavior in the body of, and the radiations emitted by, the radionuclides. To account for differences in the extent to which humans are hypothesized to be damaged by exposure to radionuclides it is common to introduce the concepts of ingestion and inhalation toxicity indices. The former measures the toxicity of exposure if one ingests the waste and the latter if one inhales the waste. The toxicity index is expressed as:
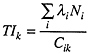
where:
TIk = The volume of water (or air) required to dilute a radionuclide to the maximum permissible concentration for pathway k, m3 of water (or air)/mass or volume of radioactive material.
λiNi = the product of the decay constant and number of atoms of radionuclide i which yields the activity (radioactive decay rate), curies (becquerels).
Cik = Maximum permissible concentration of radionuclide i for pathway k, curies (becquerels)/m3 of water (or air).
The toxicity index is a measure of the potential for adverse radiological impacts to biological organisms associated with exposure to a defined mixture of radionuclides. The toxicity index is not a measure of risk. The risk from a mixture of radionuclides is a function of the potential impacts from exposure as well as the likelihood that exposure will take place. For example, ingestion or inhalation of the contents of a HLW container might produce grave results so the material is considered a highly toxic substance. However, if it is in a well-selected and well-designed repository so that the extent of human exposure is very small it represents a very low risk.
The primary focus of the toxicity of radionuclides in this report is related to waste disposal in a geologic repository. The most common release and exposure mechanisms from a repository involve ground water contacting the waste form followed by slow dissolution, transport of radionuclides to the accessible environment, distribution in the biosphere, and eventual uptake from food and water. As a result, the ingestion toxicity index is of particular importance. In Figure 2-2 the toxicity from ingestion as a function of decay time is shown for a number of radionuclides contained in spent PWR fuel. The activities of the radionuclides in the spent fuel were taken from (Croff, 1980a) and the maximum permissible concentrations were taken from Appendix B, Table 2, Column 2 of 10CFR20 [10CFR20 1991] which are calculated to produce an effective dose equivalent of 50 millirem/yr1.
Isotopes of Principal Concern
Although hundreds of isotopes are present in spent fuel or wastes derived from them only a few of them are important in HLW and spent fuel disposal. A key reason for this is that the NRC regulations in 10CFR60 require that the containment of the waste constituents of a HLW package be substantially complete for a period in the 300-to 1,000-year range after permanent closure of the geologic repository. After 20 half-lives the radioactivity will be reduced by a factor of 220 ~106. We assume this requirement is implemented with a high degree of confidence and materials with half-lives less than 50 years (1,000 yr/20) will have decayed to negligible levels before a significant number of packages failed. Thus, for the purposes of ascertaining the health risk posed by the repository, radionuclides having half-lives less than 50 years are ignored.
By eliminating radionuclides having half-lives of less than 50 years as contributors to the repository health risk, there are two classes of radionuclides that are of concern: medium-lived fission products that substantially affect repository performance and long-lived radionuclides that are produced in significant amounts.
Fission Products. There are two fission products that are a principal concern because of their substantial thermal impact on the repository as opposed to posing a potential health risk: 90Sr and 137Cs. These two radionuclides, which could pose a health risk in an early intrusion scenario, are dominant contributors to the heat released by spent fuel or HLW during at least the first several decades (see Appendix Figure G-5). This heat is a major consideration in repository design and a significant issue in determining the performance of the repository. Cesium-137 is also a major source of penetrating radiation emitted by spent fuel and HLW during the first several decades which is a major consideration in handling these wastes. Some of the properties of these two radionuclides are summarized in Table 2-1. The combination of being medium-lived and having relatively small cross sections makes transmutation impractical for these radionuclides. A number of other methods have been proposed to eliminate this problem and are explained above in the section Impact of S&T on Waste Repository .
Two other fission products are of principal concern because of their potential contribution to health risk: 99Tc and 129I. They are of principal concern because they are long-lived, produced in significant amounts in the fission process (see Table 2-1), generally soluble under geologic conditions, and migrate relatively quickly under common ground water conditions.
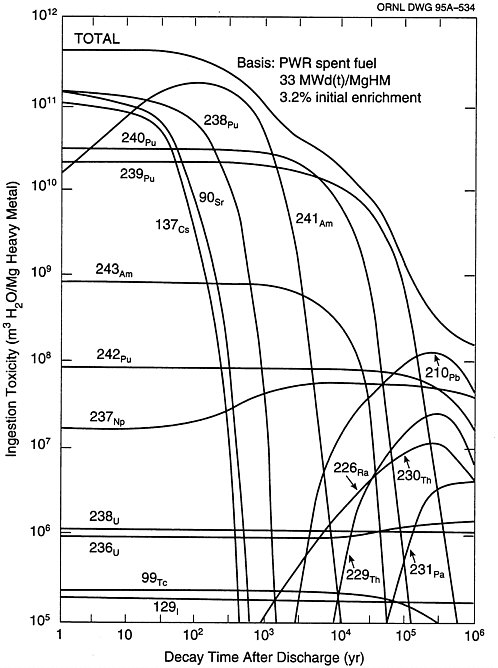
FIGURE 2-2 Toxicity from ingestion as a function of decay time for a number of nuclides in spent LWR fuel. SOURCE: Oak Ridge National Laboratory (1995).
The repository health risk posed by potential 99Tc and 129I releases could be reduced in two ways. First, these radionuclides could be transmuted to innocuous species. This would involve reprocessing spent LWR fuel to recover the radioelements containing these nuclides in relatively pure form, fabricating them into targets, irradiating the targets to destroy part of the radionuclides, and recovering the untransmuted radioelements and recycling them in new targets. The second method of reducing potential releases of these radionuclides is to incorporate them in a waste form specifically tailored to retain them, which would then be
TABLE 2-1 Properties of Selected Fission Products of Interest in Spent Fuel and HLW
|
Nuclide |
Half-Life (years) |
Atoms per Fission Product Pair Produced |
Thermal Cross Section (barns) |
|
99Tc |
2.12 × 105 |
5.77 × 10-2 |
44.4 |
|
129I |
1.7 × 107 |
1.07 × 10-2 |
37.4 |
|
90Sr |
28.1 |
4.43 × 10-2 |
1.34 |
|
137Cs |
30.0 |
6.02 × 10-2 |
0.176 |
sent to the repository for disposal. This would involve reprocessing spent LWR fuel to recover the radionuclides in relatively pure form, combining them with the appropriate materials to yield a high-integrity waste form, packaging the waste form in a suitable container, and disposing of the waste.
Both options would require development of processes to recover the radioelements Tc and I during reprocessing operations. In addition, the transmutation option would require development and qualification of transmutation targets as well as processes for recycling the untransmuted radioelement. The tailored waste form option for Tc and I would require development and qualification of a suitable waste form and package for the repository. Both of these pose significant but probably not insurmountable challenges.
TABLE 2-2 Properties of Actinides in Discharged Uranium Fuela
|
Radionuclide |
Half-Life |
kg |
Ci |
Elemental Boiling Temperature, °Cb |
|
234U |
2.47 × 105 yr |
3.14 |
1.94 × 101 |
|
|
235U |
7.1 × 108 yr |
2.15 × 102 |
4.61 × 10-1 |
|
|
236U |
2.39 × 107 yr |
1.14 × 102 |
7.22 |
|
|
237U |
6.75 days |
9.15 × 10-7 |
7.47 × 101 |
|
|
238U |
4.51 × 109 yr |
2.57 × 104 |
8.56 |
|
|
Total |
|
2.60 × 104 |
α 3.56 × 101 |
4,135 |
|
|
|
|
β 7.47 × 101 |
|
|
237Np |
2.14 × 106 yr |
2.04 × 101 |
1.44 × 101 |
|
|
239Np |
2.35 days |
2.05 × 10-6 |
4.78 × 102 |
|
|
Total |
|
2.04 × 101 |
α 1.44 × 101 |
— |
|
|
|
|
β 4.78 × 102 |
|
|
236Pu |
2.85 yr |
2.51 × 10-4 |
1.34 × 102 |
|
|
238Pu |
86 yr |
5.99 |
1.01 × 105 |
|
|
239Pu |
24,400 yr |
1.44 × 102 |
8.82 × 103 |
|
|
240Pu |
6,580 yr |
5.91 × 101 |
1.30 × 104 |
|
|
241Pu |
13.2 yr |
2.77 × 101 |
2.81 × 106 |
|
|
242Pu |
3.79 × 105 yr |
9.65 |
3.76 × 101 |
|
|
Total |
|
2.46 × 102 |
α 1.23 × 105 |
3,508 |
|
|
|
|
β 2.81 × 106 |
|
|
241Am |
458 yr |
1.32 |
4.53 × 103 |
|
|
242mAm |
141 yr |
1.19 × 10-2 yr |
1.16 × 102 |
|
|
243Am |
7,950 yr |
2.48 |
4.77 × 102 |
|
|
Total |
|
3.81 |
α 5.01 × 103 |
2,880 |
|
|
|
|
β 1.16 × 102 |
|
|
242Cm |
163 days |
1.33 × 10-1 |
4.40 × 105 |
|
|
243Cm |
32 yr |
1.96 × 10-3 |
9.03 × 101 |
|
|
244Cm |
17.6 yr |
9.11 × 10-1 |
7.38 × 104 |
|
|
245Cm |
9,300 yr |
5.54 × 10-2 |
9.79 |
|
|
246Cm |
5,500 yr |
6.23 × 10-3 |
1.92 |
|
|
Total |
|
1.11 |
α 5.14 × 105 |
— |
|
TOTAL |
|
2.63 × 104 |
α 6.42 × 105 |
|
|
|
|
|
β 2.81 × 106 |
|
|
a Uranium-fueled 1,000-MWe PWR, 150 days after discharge. b G.V. Samsonov [S1]. SOURCE: Benedict et al. (1981). |
||||
The Actinides. The important actinides produced in a uranium-fueled reactor are uranium, plutonium, neptunium, americium, and curium. Their half-lives and a calculation of the quantities produced per year in a typical 1,000 MWe PWR are given in Table 2-2. The actinides have a high toxicity index because they are bone seekers when ingested; furthermore, once fixed in the bones, they have a long biological half-life. They irradiate both the bone and the blood-forming cells in the bone marrow, where they may cause both bone cancer and leukemia.
The ingestion toxicity of the actinides of principal concern as a function of time is given in Figure 2-2. For comparison, the ingestion toxicity of the fission products is also shown. As can be seen, the long-term toxicity of spent fuel is dominated by the actinides such as 237Np, 234,236U, and
239,240,242Pu, and the shorter-lived isotopes of thorium, protactinium, radium, lead, etc. that are their progeny. However, the long-term risk is dominated in most scenarios by 129I and 99Tc because, as noted earlier, they are typically soluble and mobile in groundwater pathways. By being relatively insoluble under most conditions, the actinides are not mobile, so despite their high toxicity they contribute very little to the long-term exposure risk in scenarios where groundwater transport is important. There is an exception to this generalization as discussed in Appendix G: the solubility of the actinide neptunium is sensitive to the oxidizing potential as well as pH. Depending on which site properties are used and how infiltration is treated with respect to the availability of oxygen, 237Np, in fact, may dominate the dose in the time frame of around a million years.
In some scenarios, major failure of the repository is postulated. Such failures might be caused by volcanoes, seismic events, or human intrusion, for example. In major-failure scenarios, the actinides might be released by physical damage to the container and the spent fuel. They could then be important contributors to what could be rather high doses to the maximally exposed individual if they are still alive after the more catastrophic events such as a volcano. In such cases, transmutation, which reduces the amount of actinides in the repository, would provide some benefits.
All the actinides will fission in fast-neutron spectrum. A few will also fission in a thermal-neutron spectrum. These latter nuclides include 235U, 239Pu, and 241Pu. Some of the nuclides that do not fission in thermal spectra (e.g., 238U) can be converted into a fissile nuclide by the process:
Nuclides that have this conversion potential property are called fertile. Either a fast or a thermal spectrum can be used to transmute actinides. Because the relative rates of various nuclear reactions differ, the radioactive species in each of the process streams will be different. The amount and type of actinides going to the repository in each case will thus differ, as will the reduction of risk.
Carbon-14. 14C is produced in the atmosphere as a result of a (n,p) reaction on 14N by cosmic-ray-produced neutrons. The total global inventory is about 250 million curies (National Council on Radiation Protection and Measurements, 1985). About 4 million curies of the inventory is in the earth's atmosphere and the atmosphere's annual production rate is about 38,000 curies. 14C is a beta emitter with a half-life of 5,700 years. The global carbon contributes about 1.25 mrem of the natural background dose of about 300 mrem/yr, which includes naturally occurring 222Rn (200 mrem/yr).
14C is produced in nuclear reactors primarily by the (n,p) reaction on 14C, which is present in reactor fuel and structural parts of the reactor as a contaminant. Significant amounts are also produced by (n,α) reactions on 17O that is present in the fuel and by ternary (three-particle) fission. The spent-fuel elements from the once-through fuel cycle to be sent to the repository contain about 1.5 Ci of 14 C per Mg of heavy metal. This means that the inventory of 63,000 MTHM of spent fuel would contain about 95,000 Ci of 14C. Calculations indicate that 14C is the major contributor to the dose from gaseous release in an unsaturated repository like Yucca Mountain. When changes in barometric pressure and/or solar heating cause air to move through the repository, any 14C that has been released may be swept out. Models that predict the movement of 14C from the Yucca Mountain repository find that in a few scenarios the proposed EPA limits for 14C are exceeded. It should be noted that the acceptable risk from 14C is not currently set at a level consistent with other standards. This question is currently under review by a National Research Council committee that has responsibilities to recommend to the EPA the technical bases for HLW and spent-fuel disposal standards for the Yucca Mountain repository.
Nonetheless, it seems clear risk to public health from this release of 14C is small. Even if all the repository's 14C inventory (95,000 Ci) were released, the impact would be relatively small. The global airborne inventory would be increased from 4 to 4.095 MCi, which would raise the average dose per person from 1.25 to 1.28 mrem/yr. (The increase in dose would actually be even smaller than given since only a portion of the total release would remain in the atmosphere.) Some people may receive more than the average dose. However, since the repository release would be in the form of CO2, which is known to rapidly mix in the atmosphere, the maximal dose would still be small. (Maximal and average will both be small, but maximum could be significantly more than average.)
Carbon-14 is essentially untransmutable because its neutron cross section is exceedingly small (about 1 microbarn). If it were to be recovered during spent fuel reprocessing operations, the only possible recourse would be to immobilize it in a specially tailored waste form for repository disposal. However, current practice is to allow it to be released to the atmosphere where, as noted immediately above, it results in a very small dose increase.
The Uranium Problem
A problem that has not received as much attention as it should is the long-term toxicity of mined uranium that is not in the spent fuel. This uranium occurs in two primary materials. The first is uranium mining tailings. Uranium ore typically contains 0.15% to 0.2% uranium mixed with radioactively inert soil and rock. After processing 5% to 10% of the uranium and virtually all of its progeny (e.g.,
230Th, 226Ra) and soil/rock appear in the solid waste from the ore processing facility, which is called tailings. These are typically piled into large mounds which are then stabilized and protected by impermeable caps to retard the release of radioactive radon gas until it decays to a non-volatile radionuclide and prevent the infiltration of water, and large rocks to prevent inadvertent intrusion. If not treated in this way, they can present a major source of public exposure. However, the activity of the tailings decays away with the half-life of 230Th, which is 75,400 years (Croff et al., 1979), and the tailings activity will only decline by a factor of 10 to 20 before the progeny of the very-long-lived uranium isotopes dominate.
The second uranium-bearing material requiring additional attention is tails from uranium enrichment tails. Natural uranium contains about 0.7% 235U whereas LWRs require uranium containing 3-4% 235U. The required 235U concentration is achieved through the uranium enrichment process. However, an inevitable legacy of this process (based on simple mass-balance considerations) is that about 5 kg of depleted uranium remain for each kilogram of enriched uranium product. The 235U concentration in depleted uranium is typically 0.2-0.3%. The United States currently has about 500,000 Mg of depleted uranium stored as UF6 in metal containers above ground, with most being outdoors. Production continues at a significant rate. If these containers lose their integrity they pose a health risk because contact of UF6 with water results in the release of toxic fluorine-bearing compounds. Over the very long times characteristic of uranium half-lives the decay progeny will increase to their previous equilibrium levels, although the concentrations will be about 500 times greater than in the parent ore because of the high uranium concentration. The Defense Nuclear Facilities Safety Board (DNFSB) recently expressed concern over the condition and disposition of the enrichment plant tails. Some of the tails have been used for munitions and radiation shielding, and it is proposed that additional amounts be used for multipurpose waste canisters for spent fuel and to provide fuel for advanced breeder reactors. However, it is clear that the existing inventory plus the continued production rate will far outstrip any likely demand for these materials.
The rate at which mine/mill tailings and enrichment plant tails are generated can be reduced by about 33% if the uranium and plutonium in spent fuel are recovered by reprocessing and recycled back to LWRs. The generation rate of mine/mill tails can be reduced to very low levels and enrichment plant tails to zero by the use of advanced breeder reactors that convert the uranium to fissile material on a self-sustaining basis. however, none of the proposed transmutation systems have any plans to deal with the legacy of enrichment tails, and it does not appear possible for them to do so.
SUMMARY OF S&T PROPOSALS AND TECHNOLOGY STATEMENTS
Light-Water Reactor (LWR)
Of all the concepts investigated, one often overlooked alternative is to use existing or advanced LWRs to provide the neutron source to transmute transuranics (TRUs) as well as selected fission products. An LWR TRU-burner could build on the operational experience with plutonium recycle in commercial LWRs in the United States and especially in other countries. Such transmutation potential could be achieved with the least research and development and be available sooner than any of the other concepts considered. Current LWRs could recycle TRUs with mixed-oxide (MOX) fuel in about a third or more of the reactor cores and eliminate significant quantities of TRUs from the existing spent LWR fuel inventories. LWRs could also transmute existing plutonium inventories from the weapons program.
While the fractional transmutation rates in an LWR are not as high as those proposed for the accelerator transmutation of waste (ATW) concept, current LWRs could eliminate TRUs at a significant rate (comparable to an advanced liquid-metal reactor [ALMR] at 0.65 breeding ratio) and still perform within demonstrated engineering practices for materials interactions and thermal power densities. The LWR thermal neutron spectrum can burn certain long-lived fission products, e.g., technetium, that tend to dominate the radiological hazards of a geologic repository. In burning TRUs, however, thermal reactors tend to build up 240Pu, 241Pu, 242Pu, and higher-mass actinides, such as americium and curium. These affect reactivity in the reactor core and are a complication for reprocessing and recycle fuel fabrication and quality assurance. With full recycle of TRUs, the amounts of 240Pu and 241Am could significantly reduce reactivity. Any reactivity deficiency could be remedied by mixing the recycle TRUs with uranium of moderate enrichment, which would also enhance the capability for burning the long-lived fission products.
An LWR transmutation system could use the most well-known approach to reprocessing based on aqueous separations technology. A new generation of reprocessing plants could be applied to existing spent LWR fuel and could support subsequent application of more advanced transmutation concepts. Also, the reprocessing would allow development of optimized low-water leachable waste forms to enhance the safety of the disposal of untransmuted nuclear waste in a geologic repository.
Advanced Liquid-Metal Reactor (ALMR)
The advanced liquid-metal reactor/integral fast reactor (ALMR/IFR) program had been sponsored by the Depart-
ment of Energy/Nuclear Energy (DOE/NE) during the previous administration and was under joint development by GE and ANL. In 1994 the ALMR/IFR program was cancelled. Limited funds were appropriated for phase out and continuation of research on separations.
The ALMR has been under development for decades as a breeder reactor, but it could be modified to operate as an efficient TRU burner. Its net consumption rate increases as its breeding ratio is reduced from the usual values near unity. At a breeding ratio of 0.65, the ALMR would be comparable to an LWR in net TRU consumption rate. The ALMR, as part of any IFR system, includes pyroprocessing of its spent fuel. This technology has not yet been demonstrated at pilot scale. the possibility of success is intermediate, that is, less than that of the LWR but greater than that of the ATW concepts. An ALMR/IFR system prototype would also require an intermediate time and cost to complete development and full-scale demonstration.
The ALMR fast-neutron spectrum has both advantages and disadvantages for transmutation, compared to the thermal spectrum of an LWR or ATW. The ALMR would produce much less 240Pu, americium, and higher-mass actinides. However, the ALMR would have impractically low rates of transmutation for long-lived fission products, including those that dominate the radiological hazard with a geologic repository. The ALMR presents safety issues peculiar to a fast, liquid metal-cooled critical reactor, in particular, the issue of the sodium void coefficient. The predicted performance appears acceptable for transmutation at breeding ratios near unity. However, considerable experimental and calculational effort at breeding ratios of 0.65 or lower would be necessary to develop a database to support licensing with a reactor core that is fueled for full recycle, as proposed by the ALMR/IFR program, containing higher-mass TRUs and fission products such as the lanthanides.
The choice of an integrated all-pyroprocessing (see Chapter 3 and Appendix D for full details of pyroprocessing and other separation processes) system for an ALMR/IFR is quite a departure from the previously well-studied approach with aqueous separations technology. Pyroprocessing has been used extensively with metallic fuels in the DOE weapons programs and has been demonstrated at laboratory scale for ALMR metallic fuel. However, cost-effective pyroprocessing has not been fully developed for mixed-oxide (MOX) LWR spent fuel, which has a TRU content of only about 1% heavy metal (HM) compared to about 30% for the ALMR metallic fuel. Pyroprocessing requires considerable further development and scale-up with full pilot-plant demonstration, especially for spent LWR fuel application to commercial processing. As a low threshold cost for LWR spent-fuel reprocessing is required for a viable ALMR/IFR burner system, its probability of success as a practical, economic system is lower than that of an ALMR for power production. Indeed, the latter could be started from existing stores of plutonium (or even from highly enriched uranium [HEU]) without reprocessing spent LWR fuel.
Accelerator Transmutation of Nuclear Waste (ATW)
The ATW proposals project exceptionally high fractional rates for transmuting TRUs and long-lived fission products. The proposals aim at reducing the TRUs in waste to such an extent that all waste containing residual TRUs would be Class C or less than Class C, i.e., suitable for shallow land burial. However, the transmutation rates require extraordinary operating conditions in the ATW subcritical reactor and beam target (e.g., a factor of 10 higher thermal neutron flux and 2 1/2 higher power density than in a commercial LWR), posing major engineering and materials challenges. In addition, the ATW would use fluid fuels, an unproven technology that presents problems of reprocessing and reconstituting the fuel and raises operational and safety issues. The on-line reprocessing system has unique requirements for reliability and maintainability, but a detailed concept does not yet exist. Thus, extensive research and development would be required even to ascertain whether an ATW is feasible. Compared to an LWR or ALMR, the ATW concepts have a much lower chance of success as a practical, economic system and would require the longest time and highest cost to complete development and full-scale demonstration.
Major development issues confront the fuel system and structural materials. The ATW concepts differ primarily in the type of fluid fuel employed. In one ATW concept, the fuel is a slurry of tiny waste particles in heavy water circulating in small tubes under high pressure. Nonhomogeneity in the slurry and erosion of tube walls caused the failure of attempts to use this type of approach decades ago in Europe. Also, the ATW would produce more higher actinides than an LWR; the high alpha and neutron activity would degrade the aqueous fluid and reagents and greatly complicate recycle fuel qualification. The more advanced ATW concepts circulate a molten salt with dissolved radionuclides, which would be more radiation resistant. However, this approach suffered corrosion problems in reactors at Oak Ridge National Laboratory (ORNL) in the 1960s and 1970s. Moreover, the structural materials must withstand conditions of neutron flux and fluence beyond engineering experience (e.g., more than 100 displacements per atom) that degrades basic properties and imparts dimensional instabilities.
Intense beams of high-energy protons from an accelerator would be focused on targets such as lithium, tungsten, or molten lead, which would generate high-energy neutrons. These would be thermalized and multiplied in a subscritical
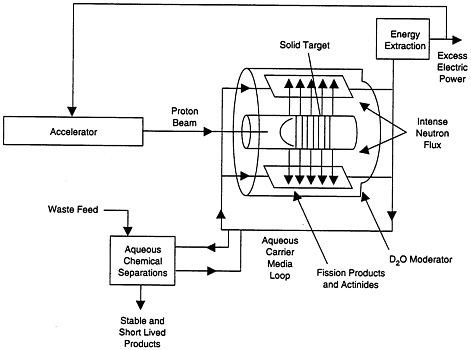
FIGURE 2-3 Reference design for commercial waste transmuter.
reactor to transmute radionuclides. The exceptionally high volumetric fission rate entails high-power densities in the reactor in addition to high thermal power in the beam target. Thus, heat removal is a major development and safety issue. In addition, the possibility of reactivity transients in the ATW may call for reactivity control measures beyond merely shutting off the accelerator as explained in Chapter 4 and Chapter 6. Overall, the safety-related problems are severe enough that licensability of an ATW seems doubtful under present regulatory practices.
It should be noted that the LWR transmutation system envisions using essentially current LWRs with some changes in the fuel composition. As in all transmutation systems, reprocessing would be required. The ALMR system would follow an ANL conceptual design that would employ metal fuel and pyroprocessing. A commercial system like this has never been built and operated; however, experience with similar systems gives confidence that such a system could be designed and built. The ATW is so drastically different in design that there is essentially no experience with such a system. Figure 2-3 shows a schematic of one of the ATW systems. The accelerator is a 1.6-MeV proton machine with a beam current of 0.25 amp. The protons hit a heavy target and produce neutrons. The neutrons are then multiplied by a factor of 20 by a subscritical assembly. The energy dissipated in the target is about 8,000 Mw. The heat is recovered to produce the power for the accelerator, and the excess power is sold to the grid. The material to be transmuted is an aqueous slurry that is processed and fed back into the system. If the designed flux of over 1015 n/cm2-s is achieved, this system would be able to attain much higher burnup rates than either of the other systems (see Appendix F for details).
Separations Technology and Commercial Spent-Fuel Reprocessing
At present, there are the only four industrially demonstrated separations processes applicable either to DOE defense reactor wastes or to meeting the needs of transmutation of TRUs and fission products in spent LWR fuel. The main steps in reprocessing spent LWR fuel are shown in Figure 2-4. These processes are designed primarily for the concentration and purification of plutonium, but only the PUREX process is well established in current worldwide use. In the past, the British have used a solvent extraction process called BUTEX, the French have used ion exchange, and there have been a number of ion exchange processes that have had limited production use in the isolation of minor actinides.
Several potentially applicable separations processes based on new solvents, such as the TRUEX-CMPO (carbonmayl methyl phospine oxide) process, and new ion-exchange materials are in various conceptual or laboratory-scale developmental stages. Such advanced aqueous processes have been proposed to achieve high decontamination factors for TRUs but have not been demonstrated at the full engineering pilot-plant level. Moreover, aqueous technologies would need further development to cope better with the radiation environment accompanying the higher-mass actinides. Among advanced nonaqueous technologies, pyroprocessing has been used extensively in the DOE weap-
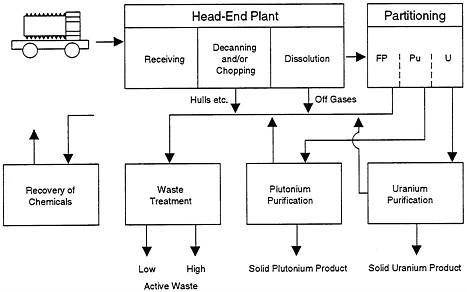
FIGURE 2-4 Main steps in reprocessing spent LWR fuels.
ons programs with metallic fuels (pyroprocessing is described in detail in Chapter 3 and Appendix E). Argonne National Laboratory (ANL) has a pyroprocessing development effort for ALMR spent-fuel reprocessing under way; an extension of that process is being studied by ANL for possible employment in the reprocessing of LWR spent fuel.
The transmutation concepts being proposed require headend treatment of spent LWR fuel to chop and dissolve the fuel, followed by separation of the TRUs and selected fission products. Either aqueous or nonaqueous processes may be used for the initial separations. The well-established PUREX process may be useful for this separations step. For the initial processing of recycle oxide fuels, even PUREX needs further development to overcome difficulties in dissolving MOX.
For the LWR transuranic burner system, an initial PUREX separations process could be followed by an aqueous separations process using an advanced technology, such as a TRUEX-like process, to achieve high decontamination factors for TRUs. An additional specialized stage would be needed to separate technetium, if desired. Development and pilot-scale testing of the latter two processes, plus demonstration of an overall separations system at engineering pilot scale, would be necessary before a full-scale separations system could be designed with high confidence, especially to meet stringent goals for very low overall separations process losses (0.1 % or smaller).
For the metallic fuel used in the ALMR, a number of pyrochemical separations for reactor spent-fuel processing have been demonstrated to a several-kilogram-batch scale with mock fuels. These methods have worked essentially as the basic thermodynamics predicts, with many exhibiting very high separation factors. However, as yet, there has not been extensive development of the equipment and techniques for the multistage cascades needed for the high separation factors entailed in the DOE ALMR/IFR proposal. In addition, ANL proposes pyrochemical processing of spent LWR fuel. The process for the LWR fuel application is less mature than for the metallic ALMR fuel and requires extensive testing, scale-up, and demonstration to achieve a viable ALMR/IFR system.
The separations processes required for the ATW transmutation concept are substantially more difficult than those required for other proposed transmutation concepts (i.e., the ALMR and LWR). This is due to the much more intense radiation fields in the ATW, which destroy many reagents involved, and the low process losses required under these conditions to meet the goals of the ATW proposals. The aqueous concepts that have been suggested are so far beyond demonstrated process experience in such radiation fields as to be highly questionable. In addition, the ATW reprocessing system requires a mechanical chopping subsystem and an industrial-scale TRUEX-like separations process to obtain the TRUs from spent LWR fuel for start-up and make-up.
In addition to separations facilities for spent reactor fuel, recovery plants will have to process the miscellaneous salvage materials generated from the processing of work material, the repair of equipment, the impurities in reagents, and the corrosion products. The recovery processes will be expensive to carry out, and those needed will not be well known until there has been extensive pilot-plant testing. A very high degree of recovery of active elements from these salvage materials will be necessary to meet cycle recoveries needed for low overall process losses.
An additional technical uncertainty arises from the lack of regulatory experience with separations plants. Environmental and safety requirements necessary to meet concerns
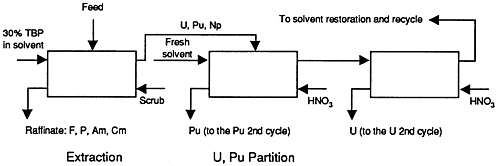
FIGURE 2-5 Flowsheet for the first purification cycle in the PUREX process. SOURCE: Musikas et al., 1992.
identified in the licensing and regulatory process could impose unknown technical obstacles and burdens on design and operation of separations plants as a part of a total separations and transmutation system.
Separations and Transmutation of Plutonium Only
Either an LWR or an ALMR would operate much more efficiently with 239Pu as the recycle fuel than with recycle of all the TRUs contained in the LWR spent fuel. This suggests a waste management concept intermediate between the once-through LWR fuel cycle and the full transmutation of all TRUs and selected fission products, i.e., the accumulated LWR spent fuel could be reprocessed and only the separated plutonium recycled to either an LWR or ALMR. A flowsheet for the first purification cycle in the PUREX process is given in Figure 2-5. The recovered uranium would be packaged for disposal or possibly for recycle to an enrichment facility. In addition, 14C and 129I could be captured, e.g., as off-gasses, and packaged in low-solubility waste forms that could be developed. Such an LWR could also transmute the long-lived 99Tc and 129I fission products if separated and recycled. Of course, the big cost is that of spent LWR fuel reprocessing and the all-remote fabrication of plutonium-bearing recycle fuel. Moreover, the concept opens the door to major public policy issues entailed with the commercial use of plutonium, in particular, its relationship to U.S. nonproliferation policy.
For such plutonium-only transmutation, present-day separations technology using PUREX may be sufficient, with process losses of the order of 1% for plutonium.2 Some of the neptunium might also be recycled if that proved easy to do. The remaining TRUs and the bulk of the fission and activation products would go to high-level radioactive wastes (HLW); this PUREX waste might be contained in a glass waste form as planned for the defense HLW. By also separating certain long-lived fission products, one could possibly devise other improved waste forms, such as for 99Tc and 135Cs. If pyroprocessing were used with an ALMR plutonium burner, other waste forms would need to be developed.
It is more problematical whether such plutonium-only transmutation could capture some of the prospective benefits to waste management claimed for the full transmutation of the TRUs. First, transmuting the plutonium could reduce somewhat the calculated repository hazards from human intrusion scenarios in which waste is brought directly to the surface. However, the effect would depend on the time after repository closure at which the human intrusion event occurred. For postclosure times of about a hundred years or so, 238Pu with an 87.7-year half-life tends to dominate the TRU activity and would be transmuted. However, for postclosure times of a few hundred years to several thousand years, the untransmuted 241Am with a 433-year half-life would dominate the TRU activity. This would be especially true for the older spent fuel in which the 241Pu had already decayed with a 14.4-year half-life to 241Am. For postclosure times of ten thousand years or more, 239Pu and 240Pu would dominate the TRU activity and would be transmuted. Over very long periods of time, when the 239Pu is eliminated, 237Np would dominate the transuranic activity.
In addition, under planned repository conditions, plutonium is quite insoluble in groundwater. Thus, plutonium transmutation would have little direct effect on the important long-term repository risk scenarios in which groundwater dissolves radionuclides and transports them to the biosphere, except by eliminating one of the precursors of 231Pa. Also, removal of the plutonium would reduce the long-term heat load on the repository, although the untransmuted 241Am would still be a major contributor. For the Yucca Mountain repository, however, decay heat is being considered as a means to keep the waste containers dry while increasing the capacity of the repository, so that transmuting the plutonium (or other TRUs) may actually be a disadvantage.
Defense High-Level Waste
Practical separation processes for DOE defense wastes stored in the Hanford tanks appear to be available. The Hanford tank wastes exist as mixtures of aqueous solutions, salt cakes, and sludges. Water washing of the sludge and dissolution of the salt cake will remove most of the 137Cs and some of the 90Sr (the principal radioactive contaminants) from the tanks, facilitating their separation and disposal as HLW and leading to a large reduction in HLW volume.
Additional processing of tank wastes, beyond sludge washing, requires dissolution of the sludge in acid. Subsequent separation of radioactive, and perhaps other, contaminants appears feasible, using processes not yet fully developed or demonstrated. A program of further research and development, ranging from laboratory studies for some proposed separations to pilot-plant development for others, is needed before design of a final separations system. It is expected that there will be appreciable insoluble residues left in some of the tanks after sludge washing. Attempts to remove these residues could seriously damage the tanks.
RADIONUCLIDES AND REPOSITORY PERFORMANCE
LWR Spent Fuel Characterization
The base case for the evaluation of the proposed transmuter systems is the once-through uranium fuel cycle for the LWR, with geologic disposal of the spent fuel. After a few years, the decay of the radionuclides in the spent fuel and the associated heat load on the repository are dominated by two fission products, 137Cs and 90Sr, with half-lives of about 30 years each. For example, in 10-year-old spent pressurized water reactor (PWR) fuel with a nominal burn-up of 33,000 MW-days/MgU at discharge, the 137Cs and 90Sr decays produce about 90 w/MgU and 65 w/MgU, respectively3 (Rawlins et al., 1990). After about 75 years, the heat load begins to be dominated by decay of TRUs, primarily several plutonium isotopes and the americium component of the minor actinides (MA).4 For the reference 10-year-old spent PWR fuel, Table 2-3 lists five TRUs that are important for heat production. In a repository the thermal power integrated from 10 to 1,000 years comes mainly from the following sources: 241Am (including the parent 241 Pu), 44%; 137Cs and 90Sr, 21%; 240Pu, 10%; 238Pu, 7%;
TABLE 2-3 Heat Release from Selected TRUs in Spent PWR Fuel (33,000 MW-days/MgU exposure—10-year decay)
|
Isotope |
Half-Life (years) |
Heat Release (W/MgU) |
|
238Pu |
87.7 |
78.0 |
|
239Pu |
2.411 × 104 |
9.7 |
|
240Pu |
6,560 |
16.5 |
|
241Pu |
14.4 |
2.46a |
|
241Am |
433 |
57.3 |
|
a241Pu is an important source of 241Am by beta decay. SOURCE: Rawlins et al. (1990). |
||
and 239Pu, 7%. After 1,000 years, the relative contributions from 240Pu and 239Pu would continue to grow while those from the other sources diminish.
The maximum attainable temperature of a geologic repository depends on the heat capacity and thermal conductivity of the geologic medium; the age, amount, and density of the spent-fuel loading; and the ventilation practices during operations. For a representative spent-fuel loading planned for the unsaturated tuff at Yucca Mountain, the maximum temperature at the repository center would occur after about 1,000 years and would remain above the boiling point of water for about 2,000 years (Ramspott et al., 1992). If the TRUs were removed, the integrated thermal power from 10 to 1,000 years would decrease by more than a factor of 4. The corresponding repository center temperature would reach the boiling point of water for a brief time and drop below 50°C within a few hundred years (Ramspott et al., 1992).
DOE currently plans to package spent LWR fuel rods in sealed containers for placement in the repository. To satisfy code of NRC regulations 10CFR60, the containers must have high integrity during emplacement and for the decades or so that the repository is being filled, such that the containers could be removed safely if the repository were found to be defective. The NRC regulations also address the integrity of the waste package after the repository is permanently closed and sealed. These regulations require that "… the engineered barrier system shall be designed, assuming anticipated processes and events, so that (A) Containment of HLW within the waste package will be substantially complete for a period to be determined by the Commission and that period must be in the 300 to 1,000-year range after permanent closure of the geologic repository …" (10CFR60.113). The Yucca Mountain program has set a performance goal of 1,000-year integrity for the spent-fuel waste package. From a regulatory point of view, however,
it may not be easy to prove package integrity in a repository for much longer than a few hundred years after closure.5
Estimating Radiological Repository Risk
The health and safety risks associated with HLW in a geologic repository are discussed in Chapter 6. The estimates of such risk focus on the increased radiation dose to which people might be subjected in the future under various scenarios in which radionuclides might escape the repository and reach the accessible biosphere. Such a scenario analysis, using probabilistic risk assessment, plays an important role both in formulating the generic criteria that all repository sites must meet and in evaluating a particular site, such as Yucca Mountain.
Radionuclide Release Scenarios
Radionuclide release scenarios take into account a wide range of possible events. The two time scales for the repository risk, using the 10,000-year EPA time horizon as a divider are:
-
extending for a period of 10,000 year after closure,
-
extending beyond 10,000 years to hundreds of thousands of years.
The half-lives and main sources of various TRUs are listed in Appendix G, Table G-2. For either time period, the three types of radionuclides of importance for repository risk are:
-
plutonium, i.e., 238Pu, 239Pu (which is the principal fissile component in the spent fuel), 240Pu, and 242Pu;
-
minor actinides, i.e., 237Np, 241Am, and 243Am, plus even higher-mass minor actinides in some transmutation schemes; and
-
fission products, mainly 137Cs and 90Sr for a few hundred years, plus long-lived soluble fission products, especially 99Tc (2.1 × 105 yr) and 129I (1.6 × 107 yr) and, to a lesser extent, 135Cs (2.9 × 106 yr).6
Of the scenarios relevant to the long-term repository risk, the most important involve the dissolution of the HLW in groundwater and the transport of the dissolved radionuclides to the accessible biosphere, so-called dissolution-and-migration scenarios. Such events could arise not only with geologic media that are normally saturated with groundwater, but also with initially dry media, if, for example, it is infiltrated with water or when there are long-term changes in the water table. For this class of scenarios, risk estimates show that the isotopes of primary concern are two long-lived, soluble fission products, 99Tc and 129I (Croff et al., 1980). To a lesser extent, long-lived 135Cs could also contribute, but its groundwater transport would be retarded by an estimated factor of 360 due to sorption on rock during the migration (Pigford, 1990).
The TRUs have significant radioactivity during this period, especially the long-lived minor actinide 237Np (2.14 × 106 yr half-life). If reducing conditions exist in a repository the transuranics tend to be relatively insoluble in groundwater and would not be transported to the biosphere in significant quantities.7 For an unsaturated repository with oxidizing conditions, 237Np may be an order of magnitude more soluble than assumed in earlier analysis based on reducing conditions (see Appendix G for details).
Calculations for a granite repository, discussed in Chapter 6 (see section Impact of S&T on Waste Repository) and Appendix G, indicate that the main actinide contributor is not 237Np but rather 231Pa, which grows in spent fuel from the decay of its precursors 235U, 239Pu, and 241Am. For the Yucca Mountain site, there is no reason to think or assume that 231Pa would be any less important for tuff than for granite, given the different conditions of the formations. Thus, 231Pa is likely to be the radionuclide of significance for the dissolution-and-migration scenarios, if the long-lived fission products were greatly reduced.
For these reasons, the various transmutation concepts have been evaluated for the conditions under which they could achieve a reduction of several orders of magnitude in the amounts of 99Tc and 129I, i.e., down to the range at which the TRUs begin to appreciably affect the estimated long-term repository risk.
The second major class of scenarios is that in which the integrity of the repository is compromised, the so-called repository-breach scenarios, which involve bypass of the normal pathways to the biosphere. These scenarios could be caused by, for example, natural events such as earthquakes and volcanism. However, the repository site would be cho-
|
5 |
Sweden is planning a much more elaborate and expensive waste package for the spent fuel, comprising a lead-filled copper container with a wall thickness of 5 to 10 cm, which is highly resistant to corrosion, surrounded by absorbing bentonite clay. The Swedish program anticipates that the integrity of the waste package in a hard-rock repository could be ensured for many thousands, perhaps millions of years (see Chapter 6). |
|
6 |
HLW also contains other long-term fission products of less importance, such as 79Se 93Zr, 107Pd, and 126Sn, plus long-lived activation products such as 59Ni, 93Mo, and 94Nb. Also, 14C may be an important activation product for the special case of the Yucca Mountain repository, as discussed in the text (see also Chapter 6 and Appendix G). |
|
7 |
Decay daughters from uranium (e.g., 226Ra and 231Pa) are also predicted (Svensk Kärnbränslehantering AB [Swedish Nuclear Fuel and Waste Management Co.], 1992\; Statens Kärnkraftinspektion [Swedish Nuclear Power Inspectorate], 1991) to contribute to long-term doses. Transmutation of the uranium parents is not proposed, but reprocessing to accomplish transmutation would reduce the amount of uranium in the repository by a magnitude of the order of the separations process decontamination factor. |
sen to minimize the significance of such natural events (among a variety of other selection criteria). Another potential cause is human intrusion, such as in drilling for water or other extractable resources. Studies are not yet definitive about the significance of human intrusion compared with other release scenarios. One study for a repository in granite indicates that human intrusion scenarios may have significantly less weight in the overall repository risk than dissolution-and-transport scenarios. For example, the risk assessment for Yucca Mountain considers human intrusion scenarios in which radionuclides could be brought to the biosphere in the vicinity of the repository and made accessible over a large area, e.g., by resuspension and wind transport (see Chapter 6 section on Impact of S&T on Waste Repository , and Appendix G). In more complex situations, some repository-breach scenarios can connect with dissolution-and-migration scenarios, for example, the breach of a barrier between the repository and a nearby aquifer, which enables radionuclides to reach the aquifer and be readily transported by groundwater.
For human intrusion scenarios the TRUs could be important in the calculation of repository risk, especially plutonium and americium isotopes (as well as 237Np for the long-term). Thus, this chapter assesses the effectiveness of the various transmutation concepts for reducing the inventory of TRUs. However, such a reduction would not simply produce a proportionate reduction in repository risk for repository-breach scenarios. Transmutation can reduce the amounts of some TRU isotopes while increasing the amounts of others. The net result depends on the scenario and the amounts of the various TRU isotopes remaining after transmutation.
Gaseous release of the radionuclide 14C is currently an issue under discussion for the Yucca Mountain repository (see Chapter 6 section on Impact of S&T on Waste Repository). 14C is produced by neutron absorption in the 14N contained in the uranium dioxide in spent fuel and in the Zircalloy fuel cladding. Although spent fuel does not have a large curie inventory of 14C, the calculations assume that essentially all of it would be released as a gas during a few thousand years under oxidizing conditions such as in the unsaturated tuff at the Yucca Mountain site. The dose to an individual living on the surface would be small, but under the original standard, the calculated probabilistic cumulative release was considered important. If the spent fuel were reprocessed, the 14C could be captured as an off-gas and packaged in a low-release waste form for geologic disposal.
Regulation 10CFR60, together with the U.S. Environmental Protection Agency (EPA) standard for protection of the accessible environment (40CFR191), also put stringent limits on the release rates and the cumulative releases of the radionuclides present in the repository after the containment period, taken here to be 1,000 years. Until recently, the EPA standard governing a geologic repository required compliance with cumulative release limits for categories of radionuclides. To establish the limits, the EPA considered many specific pathways to the accessible biosphere for each of the three principal types of pathways—gaseous transport, groundwater use, and human intrusion—and estimated the amount of each radionuclide that would have to be released to cause the equivalent of 1,000 premature deaths over a 10,000-year period. Such estimates entail assumptions about future populations and have sparked controversy about the practicality and acceptability of the basis of the EPA standard 40CFR191 (see Chapter 6).
In October 1992, Congress exempted the Yucca Mountain repository from the current EPA standard 40CFR191 and directed the EPA to commission a National Research Council study to provide findings and recommendations concerning the technical basis for health-based standards (The Energy Policy Act of 1992—PL 102-486). The National Research Council study is to consider whether a health-based standard based on doses to individual members of the public will provide a reasonable standard for protection of public health and safety. Congress also directed the Nuclear Regulatory Commission (NRC) to modify its technical criteria and requirements to become consistent with the new EPA standards. For either dose basis, however, one can identify the key radionuclides that most affect the dose estimates. The relative effectiveness in reducing the amounts of these key radionuclides can thus provide a useful measure to compare the performance of the various transmutation concepts. Individual dose and risk standards have already been directly incorporated into repository evaluations in other countries. In all cases, the allowable amount of radioactivity that can be introduced into the environment is small. The original EPA standard was consistent with the goal of 1,000 cancer deaths in 10,000 years from 100,000 metric tons uranium (MTU) of spent fuel or equivalent.
Individual dose standards are usually less than background, and in that sense are a fraction of background. However, they are set at a numerical value rather than at a fraction of background.
An Optimized Waste Form
In an S&T system, a waste form optimized for geologic disposal could be used for the HLW remaining after transmutation. Of course, separations by itself could yield waste forms that could, in principle, encompass a material for each constituent radionuclide that maximizes its chemical and physical integrity in the overall waste package. An ideal optimized waste form after separations would be solubility-limited in groundwater for key radionuclides that affect re-
pository risk significantly. For example, AgI has low water solubility and could be used as the waste form for separated 129I to greatly reduce its fractional release rate in a repository.
Indeed, a combined reduction-and-packaging strategy could be considered in which some constituents are transmuted and their remaining HLW optimally repackaged for geologic disposal, together with optimal materials chosen for other separated radionuclides that are not transmuted. Such a strategy would greatly increase the number of options open to a system designer, but puts a premium on the ability to predict waste-form behavior for very long time periods. However, as noted in Chapter 6, Impact of S&T on Waste Repository, there is no evidence from the assessment to date of repository risk that S&T systems and/or optimized waste forms are actually needed to improve nuclear waste disposal relative to the once-through LWR fuel cycle.
REFERENCES
Croff, A.G., R.L. Haese, and N.B. Gove. 1979. Updated Decay and Photon Libraries for the ORIGEN Code, Oak Ridge National Laboratory Report ORNL/TM-6055.
Croff, A. G., J. O. Bloemeke, and B. C. Finney. 1980. Actinide Partitioning-Transmutation Program Final Report. I. Overall Assessment. ORNL-5566. Oak Ridge, Tenn.: Oak Ridge National Laboratory.
National Council on Radiation Protection and Measurements. 1985. Carbon-14 in the Environment. Report No. 81. Bethesda, Md.: NCRP.
Oak Ridge National Laboratory. 1995. ORNL DWG 95A-534.
Pigford, T. H. 1990. Actinide Burning and Waste Disposal. An Invited Review for the MIT International Conference on the Next Generation of Nuclear Power Technology. UCB-NE-4176. Berkeley: University of California.
Ramspott, L. D., J. Choi, W. Halsey, A. Pasternak, T. Cotton, J. Burns, A. McCabe, W. Colglazier, and W. W. L. Lee. 1992. Impacts of New Developments in Partitioning and Transmutation on the Disposal of High-Level Nuclear Waste in a Mined Geologic Repository. UCRL ID-109203. Livermore, Calif.: Lawrence Livermore National Laboratory.
Rawlins, J. A., S. E. Binney, C. H. Bloomster, H. R. Brager, C. A. Burgess, W. J. Gruber, G. F. Howden, A. J. Naser, L. G. Niccoli, A. W. Prichard, G. W. Reddick, W. W. Shulz, J. P. Sloughter, J. L. Swanson, J. W. Thornton, C. N. Wilson, D. E. Wood. 1990. CURE: Clean Use of Reactor Energy. Report WHC-EP-0268. Richland, Wash.: Westinghouse Hanford Co.
Statens Kärnkraftinspektion (Swedish Nuclear Power Inspectorate). 1991. SKI Project-90. SKI Technical Report 91:23. Vol. 2. Stockholm: Statens Kärnkraftinspektion.
Svensk Kärnbränslehantering AB (Swedish Nuclear Fuel and Waste Management Co.). 1992. Final Disposal of Spent Nuclear Fuel. Importance of the Bedrock for Safety. SKB Technical Report 92-20. Stockholm: Svensk Kärnbränslehantering.
















