The Chemistry of Eavesdropping, Alarm, and Deceit
MARK K. STOWE, TED C. J. TURLINGS, JOHN H. LOUGHRIN, W. JOE LEWIS, AND JAMES H. TUMLINSON
A diverse multitude of arthropods hunt other arthropods as food for themselves or for their progeny. It is becoming increasingly obvious that in many of these systems, chemical signals, or "semiochemicals" (1), are crucial to the hunters' success. These semiochemicals can function in a variety of ways to bring hunters and quarry together. In the simplest systems, arthropod predators or parasitoids are attracted to their prey or hosts by semiochemicals called "kairomones" (2) emitted by the victims. Often the semiochemicals used by parasitoids or predators to locate their hosts or prey function as different types of signals in other communication systems. Thus, there is an overlapping of chemical signals with the hunters intercepting messages ''intended for" a different receiver. For example, many parasitoids exploit the pheromonal signals of their hosts during foraging. This strategy, which Vinson (3) has termed "chemical espionage" (see also ref. 4), is effective only when pheromones indicate the location of the life stage of the host that is the
Mark Stowe is a research associate in the Department of Zoology at the University of Florida, Gainesville. Ted Turlings is leader of the Chemical and Behavioral Ecology Research Team in the Institute of Plant Sciences/Applied Entomology at the Swiss Federal Institute of Technology, Zurich. John Loughrin is a postdoctoral scholar in the Department of Entomology at the University of Kentucky, Lexington. W. Joe Lewis is a research entomologist in the Insect Biology and Population Management Research Laboratory of the U.S. Department of Agriculture, Tifton, Georgia. James Tumlinson is research leader in the Insect Attractants, Behavior and Basic Biology Research Laboratory of the U.S. Department of Agriculture, Gainesville, Florida.
target of the parasitoid. When the target life stage does not reveal itself by long-distance pheromonal signals, predators and parasitoids have been forced to adopt other strategies. In some systems parasitoids or predators locate herbivorous prey by exploiting plant signals induced by the herbivores (5-8). Thus, both the plants and the predators or parasitoids benefit from this interaction. In contrast to the foraging predators, some arthropods remain stationary and emit mimetic pheromone signals to attract and capture their prey (9).
During the last decade we have been investigating the chemically mediated foraging behavior of beneficial entomophagous arthropods in an effort to elucidate the factors that guide them to their hosts or prey. Our ultimate goal is to be able to manipulate and control these organisms to increase their effectiveness as biological control agents and thus reduce our dependence on pesticides for control of insect pests in agriculture. As we and our colleagues have learned more about these systems, we have found them to be quite complex in many instances. We have also found a surprising diversity of mechanisms by which these systems operate. Here we briefly survey three categories of chemically mediated predatorprey relationships which we have arbitrarily termed "eavesdropping, alarm, and deceit." Recent reviews (8-13) describe many of these systems in more detail.
EAVESDROPPING
Pheromones, by definition, are chemical signals between two members of the same species. In most instances sex pheromones are highly specific, attracting members of the same species only and not those of closely related species. However, it appears that many predatory and parasitic arthropods are able to intercept the sex pheromone signals of their prey or hosts. Bedard in 1965 (as cited in ref. 14) first reported the attraction of a parasitic wasp, the pteromalid Tomicobia tibialis Ashmead, to volatiles produced by males of the bark beetle Ips paraconfusus (Le Cônte) boring in ponderosa pine. While the identity of the volatile kairomone in this case has not been determined, it is very likely the sex or aggregating pheromone produced by the beetles. In analogous studies several bark beetle predators have been captured in traps baited with synthetic components of the pheromones of Ips and Dendroctonus species (14). Also, several hymenopterous parasites of the elm bark beetle, Scolytus multistriatus (Marsham), are attracted to components of its pheromone, multistriatin, 4-methyl-3-heptanol, and cubebene and combinations thereof (15).
Corn earworm moth, Helicoverpa zea, females emit a blend of hexadecanal and (Z)-7-, (Z)-9-, and (Z)-11-hexadecenal (16) that is a highly
specific attractant for H. zea males. However, we discovered that field application of synthetic H. zea sex pheromone significantly increased rates of parasitization of H. zea eggs by naturally occurring female wasps belonging to the chalcid genus Trichogramma (17). Similarly, Noldus and van Lenteren (18) found that Trichogramma evanescens Westwood females respond to sex pheromones emitted by females of Pieris brassicae L. and Mamestra brassicae L. in a laboratory olfactometer. These findings came as a surprise because female moths release sex pheromones at night, while the wasps are only active during the day. Subsequent studies support the hypothesis that wasps are able to detect sex pheromone scent which has adsorbed onto the leaf surfaces near calling female moths at night and which is still being released by the leaves the following day. Noldus et al. (19) exposed leaves to calling female moths and then tested wasps for their response to exposed and unexposed leaves. Wasps showed a significantly greater response to exposed leaves for as long as 24 hr after exposure.
Lingering sex pheromone scents are useful clues to the general location of moth eggs. Female noctuid moths call in the presence of host plants and usually lay eggs nearby. Because the pheromone scent emanates from a source (leaves) that is only an approximate indication of the location of the target (moth eggs), the wasps (in contrast to male moths) do not fly upwind in pheromone plumes. Instead they respond to pheromone odor by landing on nearby surfaces and then rely on visual and short-range chemical cues to find the eggs themselves.
At present only Trichogramma species and the scelionid egg parasitoid Telenomus remus (20) are known to respond to moth sex pheromones. However, this phenomenon could be much more widespread and overlooked because these wasps apparently do not fly to point sources and are not caught in pheromone traps.
A different eavesdropping system can be found in the relationship between true bugs (Heteroptera) and some of their parasites and parasitoids (reviewed in ref. 21). The male bugs emit sex pheromones to attract females. In the process they also attract diverse parasitic flies and wasps. Unlike the Trichogramma wasps that attack moth eggs, these flies and wasps are attracted to point sources of synthetic pheromone compounds. Sticky traps baited with the synthetic sex pheromone [(E)-2-hexenal, benzyl alcohol, linalool, terpenen-4-ol, a-terpineol, and piperitol] (21) of the spined soldier bug Podisus maculiventris caught more than 17,800 parasitoids in three seasons—more than 5 times the number of female bugs captured. Two species of tachinid flies, Euclytia flava and Hemyda aurata, are highly attracted to the soldier bug pheromones and lay eggs, primarily on adult males. Female soldier bugs escape parasitization, except during mating, and consequently have about 25% as many
tachinid eggs on their bodies as do males. Interestingly, a "cheater" male strategy has apparently evolved. Soldier bug males are attracted by pheromone produced by other males and wait in close proximity to the pheromone-emitting males while not emitting pheromone themselves. Thus, these "cheater" males attempt to intercept attracted females without being parasitized.
Tiny ceratopogonid flies are also attracted to the pheromone released by calling male bugs. They puncture the bugs' pronotum and engorge on blood, apparently without disturbing the bugs. Some of the ceratopogonid flies are known to be extreme generalists and may be capable of eavesdropping on a wide range of pheromones (21).
Females of the wasp Telonomus calvus parasitize the eggs of P. maculiventris and Podisus fretus. They wait in the vicinity of male bugs releasing pheromone and become phoretic on female bugs that mate with the males (22, 23). T. calvus requires eggs less than 12 hr old to successfully develop. Thus, by riding on the female until she oviposits, the female wasps are assured of finding fresh eggs before they are discovered by competing parasitoids or predators (21).
All of the long-range kairomones attractive to parasitoids that have been identified thus far are sex pheromones of the hosts. However, we are probably aware of only a small fraction of the predators and parasites that are eavesdropping on the pheromonal communications of their prey or hosts. While the evolution of individuals that are as inconspicuous as possible to their enemies is favored, it is impossible for a species to completely avoid emitting chemical signals. Thus, pheromones that are important to reproduction or other vital functions, and are good indicators of the presence of a species, are available for predators or parasitoids to exploit.
ALARM
One of the most interesting systems that we have studied involves interactions of herbivorous larvae, the plants on which they feed, and parasitic wasps that attack the larvae. On the one hand, the larvae have evolved to become as inconspicuous as possible to avoid parasitization and predation. However, they must feed to survive and in feeding they damage the plant, and in doing so induce a reaction from the plant. What is most surprising is the plant's reaction. In addition to passively releasing volatile chemicals from their damaged tissues, plants under attack actively produce and release volatile compounds from undamaged as well as damaged tissues. This suggests a plant defensive mechanism to repel invaders since many of the volatiles released by damaged plants have been shown to be insect repellents. However, the wasps are clearly
not repelled by these volatile odors released by damaged plants, but, on the contrary, exploit them to find their hosts. Thus, both the plants and the wasps may benefit.
The first indication of the active role of plants in producing volatile chemicals to attract the natural enemies of their herbivorous attackers was found by Dicke, Sabelis, and coworkers (7, 24) in their studies of predatory mites that prey on plant-feeding mites. They found that when herbivorous spider mites feed on lima bean leaves, the plant releases a blend of volatiles that attracts predatory mites. The blend produced differs between plant species and varies depending on the species of spider mite that is attacking the plant. The blends even differ between plant cultivars infested with the same spider mite species, and the predatory mites can detect these differences (25, 26). Artificially damaged leaves are not attractive to the predatory mites.
Behavioral studies in our laboratories on host foraging of Cotesia marginiventris, a parasitoid of larvae of several species of noctuid moths, indicated that plants damaged by hosts were the most important source of volatile attractants for the female wasps (6, 27). Removal of hosts and host products, including feces, slightly diminished the attractiveness of the plants, but neither hosts nor feces were as attractive as the damaged plants alone. Collection and analysis of volatiles produced by corn seedlings fed on by beet armyworm, Spodoptera exigua, larvae overnight showed that the corn was producing both short chain compounds, like the 6-carbon aldehydes, alcohols, and esters normally associated with green leafy odors, as well as indole and several terpenes and sesquiterpenes (28) (Figure 1). However, when larvae were allowed to feed on fresh seedlings for only 2 hr, during which time volatiles were collected and analyzed, only the green leafy compounds were found. Subsequent tests showed that the plants only begin producing the terpenes several hours after damage. Furthermore, the green leafy volatiles were only produced during active caterpillar feeding. When larvae were removed from the damaged plants, the release of the low molecular weight compounds decreased immediately. In contrast, 16 hr later collection of volatiles from those damaged plants yielded large amounts of the indole, terpenes, and sesquiterpenes. Evidently, there is a delay in the plant's response to herbivore damage, which indicates changes in the plant's physiology and biochemistry. The actual mechanisms are still unknown, but it is obvious that rather than just passively emitting compounds from an open wound, the plant is actively releasing chemicals in response to herbivore feeding.
Artificially damaged plants do not emit large amounts of the terpenoids and indole emitted by the caterpillar damaged plants (Figure 1). However, application of caterpillar spit or regurgitant to the wound of
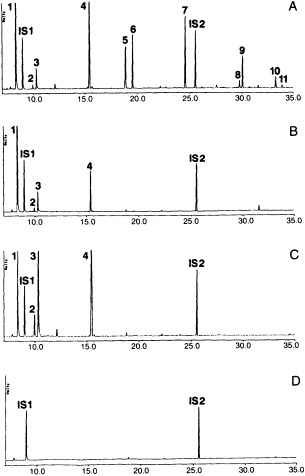
FIGURE 1 Gas chromatographic profiles of volatiles released by corn seedlings subjected to different damage treatments (from ref. 29). (A) Old damage: seedlings plus feeding beet armyworm caterpillars, where the caterpillars have been feeding on the seedlings overnight. (B) Fresh damage: seedlings plus feeding caterpillars (seedlings were undamaged at the start of volatile collection). (C) Artificially damaged fresh seedlings. (D) Undamaged seedlings. All collections lasted 2 hr. Peaks: 1, (Z)-3-hexenal; 2, (E)-2-hexenal; 3, (Z)-3-hexen-1-ol; 4, (Z)-3-hexen-1-yl acetate; 5, linalool; 6, (3E)-4,8-dimethyl-1,3,7-nonatriene; 7, indole; 8, a-trans-bergamotene; 9, (E)-ß-farnesene; 10, (E)-Nerolidol; 11, (3E,7E)-4,8,12-trimethyl-1,3,7,11-tridecatetraene. Internal standards were n-octane (IS1) and n-nonyl acetate (IS2), each representing 1000 ng. Compounds were analyzed on a 50-meter methyl silicone capillary column. Ordinate is in volts; abscissa is in minutes of retention time.
artificially damaged leaves results in release of volatiles equal in quantity to those released by plants that have actually been fed on by caterpillars. Application of caterpillar spit to undamaged leaves does not induce volatile emission (30). Therefore, a substance in the caterpillar spit induces the wounded plants to begin making and releasing volatile chemicals. Moreover, choice tests in the wind tunnel revealed that artificially damaged plants with spit were as attractive to the wasps as the plants with real caterpillar damage. Without spit, the artificially damaged plants are much less attractive.
We also found that the response of the plant to the caterpillar spit is systemic (31). Thus, not only the damaged leaves but the entire plant produces and releases volatile compounds when one or more leaves are attacked by caterpillars. Dicke et al. (7) had earlier found a similar effect in that undamaged leaves of a spider mite-injured plant attracted predatory mites. This systemic effect could be very significant in terms of enabling the natural enemies to locate their victims. It makes the plant under attack stand out from its neighbors and act as a beacon to foraging natural enemies.
While a wasp may use the chemical cues released by damaged plants to find its cryptic herbivorous hosts, the chemical signals will vary considerably if its hosts are feeding on different plant species. In modern agriculture with large plantings of a single species, this may not be a problem. However for parasitoids like Microplitis croceipes, whose hosts feed on a wide variety of plant species, the different plants will send out completely different signals. For example, cotton, cowpea, or soybean each produce a unique blend of volatile chemicals when fed on by corn earworm caterpillars. Furthermore, the composition of the volatiles can even differ when the hosts feed on different parts of the same plant (12). The volatiles released from damaged flowers or flower buds may differ considerably from those released from damaged leaves of the same plant. When a parasitoid attacks caterpillars of several different species of moths, as does C. marginiventris, the picture gets even more complicated. The same plant may release two different blends when fed on by two different species of caterpillars. Thus, feeding fall armyworm larvae induce corn seedlings to produce a distinctly different blend than do beet armyworm larvae, but both are still suitable hosts for C. marginiventris (29).
Obviously plant volatiles induced by herbivore feeding will be quite variable among all the combinations and permutations of herbivore species, plant species, plant parts, and growth stages. Thus, the predator or parasitoid is faced with the formidable task of finding its prey or host in many different habitats with a great variety of odors. Under these conditions it is not surprising that the hunters rely heavily on learning (8,
12, 32, 33), and their response to plant volatile mixtures is reinforced by hunting success.
Two recent studies in our laboratories indicate that the rate of emission of insect herbivore-induced volatiles from corn and cotton varies over the course of the day (T.C.J.T., A. Manukian, R. R. Heath, and J.H.T., unpublished data; ref. 52). Release of the induced volatiles followed a diurnal cycle, with peak emission occurring during the photophase, which corresponds to the period during which parasitoids and predators normally forage. These studies and others that indicate variations in volatile emission with different growth stages and different parts of the plants indicate that considerable caution should be exerted when plant volatile emissions are studied. Particularly when plant-arthropod interactions are being investigated it is important to consider the growth stage and the part of the plant involved in the interactions as well as the time of the day that the interactions occur.
Whether the release of volatiles by the plant has evolved to attract the natural enemies of the herbivores or whether these carnivores only exploit a plant defense mechanism aimed at the herbivore is currently under discussion. It is possible that the increase in volatile emission by plants under attack by herbivores is connected to physiological changes in the plant, which increase the level of toxins and/or antifeedants in plant tissues and deter further herbivore. In corn, leaves damaged by lepidopterous larvae become less palatable to the feeding larvae. This parallels the increase in volatile release by larvae-damaged corn leaves (34). Also, cotton varieties that release the greatest quantities of volatiles when leaves are damaged by larvae are the least palatable to those larvae (J.H.L., A. Manukian, R. R. Heath, T.C.J.T., and J.H.T., unpublished data). If these phenomena are general, it may prove difficult to determine whether plants have evolved signals to attract natural enemies of the herbivores, or whether predators and parasitoids merely exploit a plant defense mechanism to find their herbivorous prey.
Price (35) predicted that future research will reveal widespread use of plant volatiles by herbivore predators, but it has only been looked for in a few systems. Mealybug infestations of cassava induce unidentified changes in infested and uninfested leaves which make both types of leaves more attractive to encyrtid wasp parasitoids (36). This system indicates that sucking as well as chewing insects can induce systemic changes in plant volatiles. Although theoretically less likely (8), some generalist predators appear to make some use of plant volatiles to locate prey. Yellow jacket wasps in the Vespula vulgaris species group are attracted to mixtures of (E)-2-hexenal with either a-terpineol or linaloolall ubiquitous components of volatiles from plants under attack by herbivores (37, 38).
DECEIT
The predators discussed up to this point search for prey by using their ability to perceive certain chemical clues. Some unusual predators have evolved the ability to attract their prey with scents that mimic the odor of a valuable resource (see reviews of chemical mimicry in refs. 9 and 39). Several groups of spiders lure male insect prey with scents that mimic the sex pheromone scents of females of the prey species (see reviews in refs. 9, 13, 40, and 41). To the best of our knowledge, these spiders are the only predators that mimic sex pheromones. However, the spiders share some similarities with the diverse orchids which mimic insect sex pheromones to lure pollinators (9, 42, 43) and with the predatory fireflies, which practice elaborate mimicry of visual sexual signals to lure their prey: heterospecific male fireflies (44).
While most spiders are generalist predators, the "bolas spiders" in the araneid subfamily Mastophorinae feed as adults almost exclusively on male moths (see reviews in refs. 9, 13, and 41). Hunting spiders construct a simplified web which includes a short dangling line ending in a drop of glue (the "bolas"), which is hurled at prey. Moths approach the spiders from down-wind, flying in a zig-zag, apparently anemotactic, fashion until the moth is within a short distance of the spider. Spiders can only capture moths that come within the range of the bolas (typically only three or four spider body lengths). Starting with the first published report of this behavior in 1903 (45), observers have suggested that the spiders mimicked the scent of female moths.
The research necessary to support this hypothesis has been hindered by the fact that the spiders are rarely encountered-they are nocturnal, cryptic, and appear to exist at very low population levels in most areas. These spiders are also difficult to work with in the field, where they move frequently, and in the laboratory, where they rarely engage in normal hunting behavior. Nonetheless, in recent years, field tests have shown that Mastophora species attract moths from five families (9, 13, 41, 46). At one field site in southern California where spiders survive the winter and hunting adults can be found throughout the year, Mastophora cornigera spiders caught at least 15 moth species (M.K.S. and W. Icenogle, unpublished data). The pheromone chemistry of eight of these moth species is known from chemical analysis of glands or volatiles or from field screening.
Identification of compounds in volatiles collected from hunting M. cornigera revealed three common components of moth sex pheromone blends: (Z)-9-tetradecenal, (Z)-9-tetradecenyl acetate, and (Z)-11-hexadecenal [while there was insufficient material for mass spectrometry, gas chromatographic retention time evidence suggests that (Z)-11-hexadece-
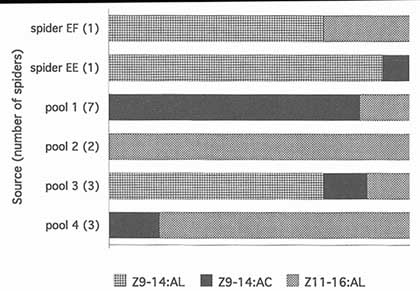
FIGURE 2 Relative proportions of three identified components of M. cornigera spider volatiles (47). The variation in the ratios suggests that spiders change the moth-attracting blends they produce. Z11-16:AL, (Z)-11-hexadecenal; Z9-14:AC, (Z)-9-tetradecenyl acetate; Z9-14:AL, (Z)-9-tetradecenal.
nyl acetate was also present] (47). These compounds are pheromone components for four M. cornigera prey species. In moths they are produced by the same biosynthetic pathway (48).
Volatiles collected from two single M. cornigera individuals and pools of collections from up to seven individuals contained quite different proportions of components (Figure 2). This preliminary evidence suggests that blends vary between individuals or that single individuals change blend composition over time. Field evidence also suggests that spiders vary their blends, since the spiders' prey includes many species that appear to be pheromonally incompatible—compounds that are necessary components in attractive blends for one or more moth species make blends unattractive for other species (Figure 3).
It is likely that the spectrum of compounds produced by all Mastophora species includes a wider range of compounds than those found in this study. Several Mastophora species catch male moths that are known to respond to pheromone compounds in the even-carbon number aldehyde/ acetate/alcohol chemical class, as well as males of other species that are known to respond to pheromone compounds in the odd-carbon number hydrocarbon chemical class (9, 13).
Recent field tests have examined the prey-attracting ability of Masto-
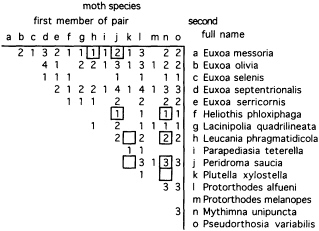
FIGURE 3 Pairwise combinations of moths caught by a population of spiders (M. cornigera) at one site in California (Winchester, CA) and the number of spider individuals catching both species in a pair [from a study of six spiders whose prey was recorded for up to 1.5 years over a span of 5 years (M.K.S. and W. Icenogle, unpublished data)]. Cells surrounded by a border indicate pairs of moths that seem to be pheromonally incompatible—i.e., compounds that are necessary components in attractive blends for one moth species make blends unattractive for the other species (47).
phora spiderlings as well as spiderlings and adults of two other spider genera (Kaira and Phoroncidia) that spin simplified webs (reviewed in refs. 9 and 13; see also ref. 49). These controlled tests demonstrate that in all three genera, adult spiders attract moths, and spiderlings attract various families of nematocerous flies. Preliminary chemical analysis suggests that adult Phoroncidia spiders produce known moth sex pheromone compounds (M.K.S. and J.H.T., unpublished data). These three spider genera are currently placed in unrelated subfamilies in the superfamily Araneoidea. Phylogenetic studies based on behavioral and morphological characters support this classification and the idea that the genera evolved the ability to attract prey independently (J. Coddington and N. Scharff, personal communication). (See references in ref. 9 for evidence of sexpheromone mimicry in other groups of spiders.)
FUTURE DIRECTIONS
Many fundamental questions remain to be investigated in the study of these systems. How do reception and perception of pheromone molecules by ''eavesdroppers" compare to the same processes in the prey? For
example, male moths generally respond to only a range of pheromone component ratios that centers around the ratio of components produced by females (reviewed in ref. 50); do the wasps that eavesdrop on female moth sex pheromones have the same ratio specificity? How does this differ between specialist and generalist parasitoids? Does learning modify the responses of eavesdropping parasitoids and predators as it does in the parasitoids and predators that respond to plant "alarm" odors? In plants damaged by herbivorous arthropods, what are the mechanisms involved in the release of volatile signals? (See refs. 51, 52.) What are the factors in insect spit that plants respond to? How do hormones or other chemical signals bring about the systemic response of the entire plant to localized herbivore damage? How does the mechanism of plant response vary between major plant groups? (For speculation concerning the evolution of the eavesdropping and alarm systems, see refs. 8 and 10.) In the prey-attracting spiders, where are the volatile-producing glands? How does the biosynthesis of spider compounds compare to the biosynthesis of pheromone compounds by their prey? Do all three spider groups vary their prey-attracting blends? Is this variation controlled by learning, influenced by seasonal cues, or due to genetic differences between individuals? (For speculation concerning the evolution of these systems, see ref. 9.)
Much of the interest in these complex chemically mediated relationships lies in the potential agricultural application of research results. Future work on "eavesdropping"- and "alarm"-based relationships may lead to more effective use of parasitoids for biological control. Semiochemicals may be useful for attracting parasitoids or predators into a crop or increasing the amount of time they spend searching for hosts or prey in a field. New crop varieties may be developed that emit greater amounts of "alarm" compounds when attacked by herbivorous pests and thus are more effective in recruiting natural enemies of their attackers. Spiders that practice deceit represent an unexploited chemical library of compounds that may be useful in pheromone-based monitoring and control of crop pests. Analysis of spider volatiles might provide the first insight into the sex pheromone chemistry of those numerous prey taxa whose chemistry has never been studied or might reveal the existence of attractive sex pheromone analogs. The eventual applications arising from research in this area may be as unanticipated and exciting as many of the research results obtained so far.
SUMMARY
Arthropods that prey on or parasitize other arthropods frequently employ those chemical cues that reliably indicate the presence of their
prey or hosts. Eavesdropping on the sex pheromone signals emitted to attract mates allows many predators and parasitoids to find and attack adult insects. The sex pheromones are also useful signals for egg parasitoids since eggs are frequently deposited on nearby plants soon after mating. When the larval stages of insects or other arthropods are the targets, a different foraging strategy is employed. The larvae are often chemically inconspicuous, but when they feed on plants the injured plants respond by producing and releasing defensive chemicals. These plant chemicals may also serve as "alarm signals" that are exploited by predators and parasitoids to locate their victims. There is considerable evidence that the volatile "alarm signals" are induced by interactions of substances from the herbivore with the damaged plant tissue. A very different strategy is employed by several groups of spiders that remain stationary and send out chemical signals that attract prey. Some of these spiders prey exclusively on male moths. They attract the males by emitting chemicals identical to the sex pheromones emitted by female moths. These few examples indicate the diversity of foraging strategies of arthropod predators and parasitoids. It is likely that many other interesting chemically mediated interactions between arthropod hunters and their victims remain to be discovered. Increased understanding of these systems will enable us to capitalize on natural interactions to develop more ecologically sound, environmentally safe methods for biological control of insect pests of agriculture.
REFERENCES
1. Law, J. H. & Regnier, F. E. (1971) Annu. Rev. Biochem. 40, 533-548.
2. Nordlund, D. A. (1981) in Semiochemicals: Their Role in Pest Control, eds. Nordlund, D. A., Jones, R. L. & Lewis, W. J. (Wiley, New York), pp. 13-23.
3. Vinson, S. B. (1984) in Chemical Ecology of Insects, eds. Bell, W. J. & Cardé, R. T. (Sinauer, Sunderland, MA), pp. 111-124.
4. Noldus, L. P. J. J. (1989) Ph.D. dissertation (Wageningen Agricultural Univ., Wageningen, The Netherlands).
5. Nordlund, D.A., Lewis, W. J. & Altieri, M.A. (1988) in Novel Aspects of Insect-Plant Interactions, eds. Barbosa, P. & Letourneau, D. K. (Wiley, New York), pp. 65-90.
6. Turlings, T. C. J., Tumlinson, J. H. & Lewis, W. J. (1990) Science 250, 1251-1253.
7. Dicke, M., Sabelis, M. W., Takabayashi, J., Bruin, J. & Posthumus, M. A. (1990) J. Chem. Ecol. 16, 3091-3118.
8. Vet, L. E. M. & Dicke, M. (1992) Annu. Rev. Entomol. 37, 141-172.
9. Stowe, M. K. (1988) in Chemical Mediation of Coevolution, ed. Spencer, K. (Academic, San Diego), pp. 513-580.
10. Dicke, M. & Sabelis, M. W. (1993) in Insect Chemical Ecology: An Evolutionary Approach, eds. Roitberg, K. & Isman, M. B. (Chapman & Hall, New York), pp. 122-155.
11. Tumlinson, J. H., Turlings, T.C.J. & Lewis, W.J. (1992) Agric. Zool. Rev. 5, 221-252.
12. Turlings, T. C. J., Wäckers, F. L., Vet, L. E. M., Lewis, W. J. & Tumlinson, J. H. (1993) in Insect Learning: Ecological and Evolutionary Perspectives, eds. Lewis, A. C. & Papaj, D. R. (Chapman & Hall, New York ), pp. 51-78.
13. Yeargan, K. V. (1994) Annu. Rev. Entomol. 39, 81-99.
14. Wood, D. L. (1982) Annu. Rev. Entomol. 27, 411-446.
15. Kennedy, B. H. (1984) J. Chem. Ecol. 10, 373-385.
16. Klun, J. K., Plimmer, J. R., Bierl-Leonhardt, B. A., Sparks, A. N., Primiani, M., Chapman, O. L., Lee, G. H. & Lepone, G. (1980) J. Chem. Ecol. 6, 165-175.
17. Lewis, W. J., Nordlund, D. A., Gueldner, R. C., Teal, P. E. A. & Tumlinson, J. H. (1982) J. Chem. Ecol. 8, 1323-1331.
18. Noldus, L. P. J. J. & van Lenteren, J. C. (1985) J. Chem. Ecol. 11, 781-791.
19. Noldus, L. P. J. J., Potting, R. P. J. & Barendregt, H. E. (1991) Physiol. Entomol. 16, 329-344.
20. Nordlund, D. A., Lewis, W. J. & Gueldner, R. C. (1983) J. Chem. Ecol. 9, 695-701.
21. Aldrich, J. R. (1995) in Chemical Ecology of Insects II, eds. Cardé, R. T. & Bell, W. J. (Chapman & Hall, New York), pp. 318-363.
22. Aldrich, J. R. (1985) in Semiochemistry: Flavors and Pheromones, eds. Acree, T. E. & Soderlund, D. M. (de Gruyter, Berlin), pp. 95-119.
23. Orr, D. B., Russin, J. S. & Boethel, D. J. (1986) Can. Entomol. 118, 1063-1072.
24. Dicke, M. & Sabelis, M. W. (1988) Neth. J. Zool. 38, 148-165.
25. Takabayashi, J., Dicke, M. & Posthumus, M. A. (1991) Chemoecology 2, 1-6.
26. Takabayashi, J., Dicke, M. & Posthumus, M. A. (1991) Phytochemistry 30, 1459-1462.
27. Turlings, T. C. J., Tumlinson, J. H., Eller, F. J. & Lewis, W. J. (1991) Entomol. Exp. Appl. 58, 75-82.
28. Turlings, T. C. J., Tumlinson, J. H., Heath, R. H., Proveaux, A. T. & Doolittle, R. E. (1991) J. Chem. Ecol. 17, 2235-2251.
29. Turlings, T. C. J. (1990) Ph.D. dissertation (Univ. of Florida, Gainesville).
30. Turlings, T. C. J., McCall, P. J., Alborn, A. T. & Tumlinson, J. H. (1993) J. Chem. Ecol. 19, 411-425.
31. Turlings, T.C.J. & Tumlinson, J.H. (1992) Proc. Natl. Acad. Sci. USA 89, 8399-8402.
32. Lewis, W. J. & Tumlinson, J. H. (1988) Nature (London) 331, 257-259.
33. Lewis, W. J., Vet, L. E. M., Tumlinson, J. H., van Lenteren, J. C. & Papaj, D. R. (1990) Environ. Entomol. 19, 1183-1193.
34. Turlings, T. C. J. & Tumlinson, J. H. (1991) Fla. Entomol. 74, 42-50.
35. Price, P. W. (1981) in Semiochemicals: Their Role in Pest Control, eds. Norlund, D. A., Jones, R. L. & Lewis, W. J. (Wiley, New York), pp. 251-279.
36. Nadel, H. & van Alphen, J. J. M. (1987) Entomol. Exp. Appl. 45 , 181-186.
37. Aldrich, J. R., Kochansky, J. P. & Sexton, J. D. (1985) Experientia 41, 420-422.
38. Aldrich, J. R., Lusby, W. R. & Kochansky, J. P. (1986) Experientia 42, 583-585.
39. Dettner, K. & Liepert, C. (1994) Annu. Rev. Entomol. 39, 129-154.
40. Eberhard, W. G. (1980) Psyche 87, 143-169.
41. Stowe, M.K. (1986) in Spiders Webs: Behavior and Evolution, ed. Shear, W. (Stanford Univ. Press, Stanford, CA), pp. 101-131.
42. Kullenberg, B. (1961) Zool. Bidr. Uppsala 34, 1-330.
43. Borg-Karlson, A. K. (1990) Phytochemistry 29, 1359-1387.
44. Lloyd, J. E. (1984) in Oxford Survey of Evolutionary Biology, eds. Dawkins, R. & Ridley, M. (Oxford Univ. Press, Oxford), pp. 48-84.
45. Hutchinson, C. (1903) Sci. Am. 10, 172.
46. Yeargan, K. V. (1988) Oecologia 74, 524-530.
47. Stowe, M. K., Tumlinson, J. H. & Heath, R. R. (1987) Science 236, 964-967.
48. Bjostad, L. B. & Roelofs, W. L. (1983) Science 220, 1387-1389.
49. Eberhard, W. G. (1981) J. Arachnol. 9, 229-232.
50. Phelan, P. L. (1993) in Insect Chemical Ecology: An Evolutionary Approach, eds. Roitberg, K. & Isman, M. B. (Chapman & Hall, New York), pp. 265-314.
51. Croft, K. P. C., Jüttner, F. & Slusarenko, A. J. (1993) Plant Physiol. 101, 13-24.
52. Loughrin, J. H., Manukian, A. Heath, R. R., Turlings, T. C. J. & Tumlinson, J. H. (1994) Proc. Natl. Acad. Sci. USA 91, 11836-11840.
















