Page 89
Chapter 5
Single Photon Emission Computed Tomography
5.1 Introduction
Single photon emission computed tomography (SPECT) is a medical imaging technique that is based on conventional nuclear medicine imaging and tomographic reconstruction methods. The images reflect functional information about patients similar to that obtained with positron emission tomography (PET). Both SPECT and PET (see Chapter 6) give information based on the spatial concentration of injected radiopharmaceuticals, in contrast to the other medical imaging modalities used for clinical diagnostic purposes.
Figure 5.1 shows the imaging process and the components of a typical nuclear medicine imaging system. A radioactive-labeled pharmaceutical (radiopharmaceutical) is administered to a patient. Depending on the

Figure 5.1. The imaging process and the components of a
typical nuclear medicine imaging system.
Page 90
biodistribution properties of the radiopharmaceutical, it is taken up by different organs and/or tissue types. Most radiopharmaceuticals used in nuclear medicine and SPECT are labeled with radionuclides that emit g-ray photons. Typically, a scintillation camera system is used as the imaging device. The scintillation camera consists of a lead collimator that allows photons traveling in given directions to pass through a large-area scintillator (commonly NaI(Tl) crystal) that converts the energy of g -ray photons to lower-energy photons which are in turn converted to electric signals by photomultiplier tubes (PMTs). The signals from an array of PMTs are processed by electronic circuitry to provide information about the position at which a photon interacts with the crystal. The scintillation camera provides a two-dimensional projection image of the three-dimensional radioactivity distribution or radiopharmaceutical uptake within the patient.
SPECT takes conventional two-dimensional nuclear medicine images acquired at different views around the patient and provides an estimate of the three-dimensional radioactivity distribution using methods of image reconstruction from multiple projections. SPECT differs from x-ray computed tomography (CT) in that the radiation source is within instead of outside the patient. The goal of SPECT is to determine accurately the three-dimensional radioactivity distribution resulting from the radiopharmaceutical uptake inside the patient (instead of the attenuation coefficient distribution from different tissues as obtained from x-ray CT). SPECT utilizes radiopharmaceuticals that are common in nuclear medicine clinics, rather than those that emit positrons with subsequent generation of two 511-keV annihilation photons as is the case with PET. SPECT requires instrumentation and image reconstruction methods that differ from those used in other medical imaging modalities.
The amount of radiopharmaceutical that can be administered is limited by the allowable dose of radiation to the patient. This requirement results in a limited number of photons that can be used for imaging. Also, the acceptance angle or geometric response of the collimator further limits the fraction of photons that are acceptable for the projection data. The collimator can be designed to allow detection of more photons, but increased detection efficiency usually can be achieved only with a concurrent loss of spatial resolution. A major goal of SPECT instrumentation development is to increase the detection efficiency while at the same time improving the spatial resolution of the imaging system, goals that are pursued by adding more detectors around the patient.
The SPECT imaging process imposes unique difficulties and challenges in
Page 91
image reconstruction. For example, before exiting the patient, many of the g-ray photons experience photoelectric interactions that cause absorption of photons, and many thus experience Compton scattering, which changes the direction and energy of the original photons. When conventional reconstruction techniques (e.g., x-ray CT algorithms) are used in SPECT, the reconstructed images are severely affected by statistical noise fluctuations, poor spatial resolution, low contrast, and inaccurate quantitative information.
Following is a brief discussion of the basic principles of SPECT imaging and the current status of SPECT instrumentation and image reconstruction methods. Emphasis is placed on the physics, mathematics, and engineering aspects of SPECT, and future trends and potential areas of further investigation are discussed. In combination with new radiopharmaceuticals and clinical applications, these developments could ultimately improve patient care.
5.2 Physical and Instrumentation Factors
That Affect SPECT Images
Attenuation from photoelectric absorption and Compton scattering is the major factor that affects the quantitative accuracy and quality of SPECT images. The degree of attenuation is determined by (1) the path length between the source and the edge of the attenuating material, and (2) the linear attenuation coefficient, which is a function of photon energy and the amount and types of materials contained in the attenuating medium. For example, the attenuation coefficient for the 140-keV photons (from the commonly used isotope Tc-99m) in water or soft tissue is 0.15 cm-1. For the 140-keV photons, this attenuation coefficient gives rise to a half-valued-layer—the thickness of material that attenuates half of the incident photons—of 4.5 cm in water. The attenuation effect is further complicated by the fact that different regions of the body have different attenuation coefficients. In particular, the non-uniform attenuation distribution in the thorax is a major problem in cardiac SPECT image reconstructions.
Photons that have been scattered before reaching the radiation detector give erroneous spatial information about the origin of the radioactive source, and a significant fraction of the photons detected in SPECT have been scattered: typical ratios of scattered to unscattered photons are about 20-30% in brain SPECT studies and about 30-40% in cardiac and body
Page 92
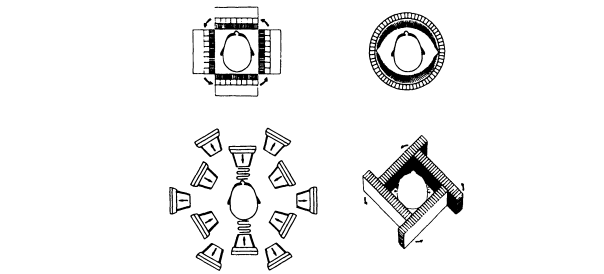
Figure 5.2. Simple schematic showing the development over time of SPECT systems.
SPECT studies for 140-keV photons. Overall, the resolution of contemporary SPECT systems is about 7 mm. As this measurement is about the same as the thickness of many organ subregions (e.g., in the brain, breast, kidney, or bones), the resulting images are significantly blurred. This relatively low resolution affects image quality as well as quantitative accuracy.
5.3 SPECT Instrumentation
5.3.1 SPECT System Designs
The development of SPECT systems can be summarized by the schematic diagram shown in Figure 5.2. The systems are designed to acquire projection data from multiple views around the patient. In general, SPECT instrumentation can be divided into three categories: those that use (1) arrays of multiple scintillation detectors, (2) one or more scintillation cameras, or (3) hybrid scintillation detectors combining the first two approaches.
Figure 5.2 (upper left) shows the first multiple-detector SPECT system design developed in the 1960s and consisting of four banks of linear arrays of detectors. Multidetector SPECT systems provide the capability of high counting rates but are restricted to single or a limited number of image slices.
Page 93
The SPECT system designs found in most commercial SPECT systems are based on a single scintillation camera or on multiple rotating scintillation cameras. The scintillation cameras allow acquisition of two-dimensional projection data that can be reconstructed to form multiple reconstructed image slices. Although the counting rate of the camera-based SPECT systems is generally lower than that of multiple detector-based SPECT systems, the data from camera-based SPECT systems are usually adequate for most clinical studies. Multiple-camera-based SPECT systems increase the number of cameras surrounding the patient to provide higher detection efficiency (Fig. 5.3). The three-camera design allows 360° sampling in periods as short as 5 s.
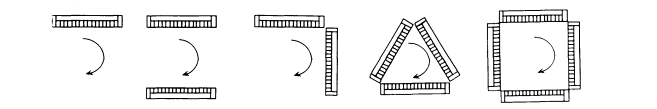
Figure 5.3. Different arrangements of multiple-camera-based SPECT systems, which increase the number of cameras surrounding the patient to provide higher detection efficiency.
A unique hybrid brain SPECT system design is being developed at the University of Arizona. As shown in Figure 5.4, the system consists of a stationary array of small modular cameras (10 x 10 cm2) arranged on a half hemispherical surface surrounding the patient's head. A set of pinholes on a hemispherical lead collimator allows formation of projection images from multiple views that can be reconstructed directly to form a threedimensional reconstructed image. Since no rotation motion is required to collect the complete projection data set, the system is capable of acquiring fast dynamic three-dimensional SPECT data.
5.3.2 Special Collimators
As shown in Figure 5.1, the most common collimator design used in nuclear medicine and SPECT consists of parallel holes. For the parallel-hole collimator design, the detection efficiency can be increased at the expense of a concurrent degradation of spatial resolution. This trade-off between detection efficiency and spatial resolution is a major factor in the development
Page 94
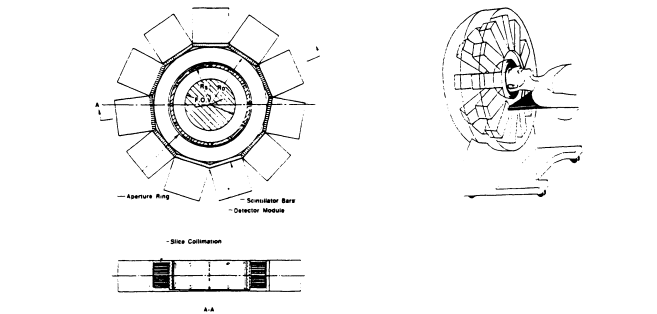
Figure 5.4. A hybrid brain SPECT design being developed at the University of Arizona, consisting of a stationary array of small modular cameras (10x 10 cm2) arranged on a half hemispherical surface surrounding the patient's head. A set of pinholes on a hemispherical lead collimator allows formation of projection images from multiple views that can be reconstructed directly to form a three-dimensional reconstructed image. Since no rotation motion is required to collect the complete projection data set, the system is capable of acquiring fast dynamic three-dimensional SPECT data.
of SPECT systems and in the improvement of SPECT image quality. The goal of special collimator designs in SPECT is to decrease the severity of this trade-off as compared to the parallel-hole design.
Two collimator designs used in SPECT that improve efficiency with no resolution penalties are the fan-beam and cone-beam geometries (Fig. 5.5). The gain in detection efficiency is obtained by the increased solid angle of acceptance. Typically, the fan-beam and cone-beam collimators provide about 1.5 and 2 times the detection efficiency of a parallel-hole collimator with the same spatial resolution. However, the geometries of the converging-hole collimator designs require special reconstruction algorithms and data acquisition strategies.
Page 95
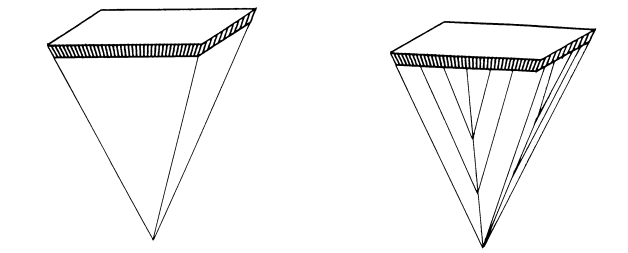
Figure 5.5. Fan-beam (left) and cone-beam (right) collimator designs, which improve
efficiency with no resolution penalties.
5.3.3 New Radiation Detector Technologies
For over 3 decades the scintillation camera has been the most popular imaging device of nuclear medicine. During this time its performance has been thoroughly studied and optimized, and further improvement will likely be dependent on the development of solid-state detector arrays. Current scintillation cameras have about 3-mm intrinsic spatial resolution and 10% energy resolution at 140 keV, numbers that have remained essentially unchanged for over a decade. Since the spatial resolution of a commonly used general-purpose collimator is on the order of 7 to 15 mm, the 3-mm intrinsic resolution of the camera has been regarded as adequate. However, new detector and collimator strategies such as those embodied in the design shown in Figure 5.4 give encouragement that improved resolution can be achieved. Simulations from the research group at the University of Arizona have shown that a final tomographic resolution of around 2 mm is possible if detectors with very large areas and small pixels can be produced.
Semiconductor detectors or combinations of scintillators and solid state photon detectors offer good prospects for significant improvements in spatial and energy resolution. In the past, silicon and germanium detectors were investigated for use in conventional nuclear medicine imaging. However, their use was limited due to their lower stopping power, high material cost,
Page 96
and the requirement to operate at low temperature. However, new room-temperature semiconductors with high stopping power are now available, offering numerous options for new detector designs. For example, one exciting new material, cadmium zinc telluride, now yields a room-temperature energy resolution of about 3-4% at 140 keV. These new semiconductor detectors combined with optimized imaging geometries offer the prospect of major advances in the performance of SPECT imaging systems. The limit in useful resolution depends ultimately on the allowable patient dose of radionuclide and the solid angle effectively spanned by the detection system.
The sensitivity of SPECT is much less than that of PET because of the necessity of using lead channels or collimators, which limit the solid angle subtended by the tomograph. A general expression for sensitivity S is
![]()
where A is the area of detector material seen by each point in the object being scanned, Îis the efficiency of the detector, n = 1 for SPECT and 2 for PET, g is the attenuation factor (typically 0.2 to 0.6), and r is the radius of the tomograph. One can show that the ratio of sensitivity between PET and SPECT is about 150 divided by the resolution; thus, for a resolution of 7.5 mm, the ratio is 20.
Innovations suggested to improve the sensitivity of SPECT include not only methods of increasing A through collimator-detector arrangements, but also the use of methods for tracking the trajectory of each photon. The latter include Compton scattering angle determinations from energy discrimination.
5.4 SPECT Image Reconstruction
5.4.1 The SPECT Reconstruction Problem
In SPECT, the goal of image reconstruction is to determine accurately the three-dimensional distribution of administered radiopharmaceutical in the patient. Assume an ideal situation in which the emission photons do not experience attenuation and scatter in the patient and the collimator-detector has perfect spatial resolution without blurring effects in the measured data. A naive approach to the SPECT reconstruction is to consider the projections p(t, q) of radiopharmaceutical activity as the simple Radon transform, which
Page 97
for a two-dimensional distribution f(x,y) is
![]()
where t is the position on the projection array, q is a particular projection angle, and c is the gain factor that transforms radioactivity concentration to detected signals. The SPECT reconstruction problem is different from the classical problem of image reconstruction from projections because each photon is attenuated and scattered by the material between its source and the detector.
When attenuation is taken into consideration, the two-dimensional attenuated Radon transform can be written as
![]()
where a(u, v) is the two-dimensional attenuation coefficient distribution and ![]() is the attenuation factor for photons that originate from (x,y), travel along the direction perpendicular to the detector array, and are detected by the collimator-detector. See also section 14.1.2.
is the attenuation factor for photons that originate from (x,y), travel along the direction perpendicular to the detector array, and are detected by the collimator-detector. See also section 14.1.2.
However, in realistic situations, the projection data in SPECT are often measured in two dimensions and are affected by the three-dimensional effects of attenuation and scatter of photons in the patient's body and the three-dimensional spatial resolution response of the collimator-detector. When these three-dimensional effects are taken into consideration, the measured projection data are given by a more complicated attenuated Radon transform,
![]()
where t = (x,y) is a point on the two-dimensional projection image p(t,q) at viewing angle q, and f (r) and a(u) are the three-dimensional radioactivity and attenuation distributions in the patient, respectively. At each point on the projection image, unattenuated and scattered photons that fall within the field of view of the collimator-detector are detected. The inner integral of equation 5.3 considers attenuation of these photons through the patient with a three-dimensional attenuation distribution a(u). The combined geometric collimator-detector and scatter response are represented by a three-dimensional response function, h(s,w ;r), which is a function of the
Page 98
position r at which the photon originates in the patient and its distance from the collimator-detector. The function is non-zero within a solid angle defined by W.
5.4.2 SPECT Image Reconstruction Methods
The SPECT reconstruction problem without attenuation expressed in equation 5.1 estimates f(x, y) by using conventional techniques for reconstructing images from projections. The most commonly used techniques are based on the Fourier projection theorem. These techniques apply a ramp-shaped filter to the Fourier transform of the projections (equation 5.1) and then backproject the inverse transform. The methods are generally known as filtered backprojection of filtered projection or, equivalently, convolution methods. The shape of the filter depends on the amount of noise suppression desired. (More detailed descriptions of these image reconstruction techniques are found in Chapter 14.) When used in routine clinical SPECT image reconstruction, these reconstructions ignore the effects of attenuation, scatter, and the collimator-detector response. Consequently, SPECT images obtained with these reconstruction techniques are quantitatively inaccurate. More importantly, these images have artifacts and distortion, resulting in poor image quality. However, analytical solutions to the inverse Radon transform expressed in equations 5.2 and 5.3 have not been found for the general case of non-uniform attenuation. Hence, special reconstruction methods are needed to estimate accurately the true three-dimensional radiopharmaceutical distribution in the patient, and research in this area has been a major focus in the development of SPECT in recent years. In particular, iterative reconstruction algorithms have been used to provide estimates of the solutions of the SPECT reconstruction problem expressed in equations 5.2 and 5.3.
In general, quantitative SPECT image reconstruction methods consist of two major components: (1) algorithms for image reconstruction from projections, and (2) techniques that compensate for image-degrading effects such as attenuation, scatter, and collimator-detector response. Often, the image reconstruction algorithm is inseparable from the compensation technique, resulting in SPECT reconstruction methods that are not found in other tomographic imaging modalities.
Iterative Reconstruction Algorithms A typical iterative reconstruc-
tion algorithm starts with an initial estimate of the object source distribution. Projection data are generated from the initial estimate using a projec-
Page 99
tion matrix that models the imaging process. The calculated projection data are compared with the measured data at the same projection angles and their differences are determined. With application of a given statistical criterion, the differences are used to update the initial image estimate. A new set of calculated projection data is regenerated from the new image estimate and compared with the measured projection data. The procedure is repeated until the differences between the calculated and measured projection data are smaller than a preselected small value. Statistical criteria that have been used in formulating iterative reconstruction algorithms include the minimum mean squares error (MMSE), weighted least squares (WLS), maximum entropy (ME), maximum likelihood (ML), and maximum a posteriori (MAP) approaches. Iterative algorithms that have been used in estimating the reconstructed images include the Gauss-Seidel (GS), conjugate gradient (CG), and expectation maximization (EM) algorithms (see section 14.1.2). Iterative reconstruction methods differ in their assumptions about the statistical properties of the measured data, convergence rates, and noise characteristics. These properties are important considerations for assessing the applicability of particular reconstruction methods in SPECT.
Interest in the applications of iterative reconstruction algorithms was initially propelled by the need to compensate for non-uniform attenuation in the imaging of heart muscle with radiopharmaceuticals, which illuminate relative perfusion. More recently, iterative reconstruction algorithms have been applied to compensate for the spatially variant system response function (collimator-detector response) and scatter. These compensations are achieved by modeling the imaging process in the projection and backprojection operations during the iterative steps. A major drawback of the iterative techniques is the long processing time involved. However, rapid advances in computer technology and in software implementation of these algorithms have significantly reduced the computation times. For example, the reconstruction time for 30 iterations of the iterative ML-EM algorithm with non-uniform attenuation compensation is 4 minutes for 32 64 x 64 image slices using a state-of-the-art DEC Alpha 3000 workstation. Continued development in computer hardware, algorithms, and codes will bring quantitative SPECT reconstruction methods to clinical use in the near future.
Compensation Methods
Methods of compensating for attenuation can be grouped into two categories: (1) methods that assume that the attenuation coefficient is uniform over the body region, and (2) methods for non-
Page 100
uniform distributions of attenuation coefficients. The assumption of uniform attenuation can be applied to SPECT imaging of the head and of the abdomen region. If uniform attenuation is assumed, i.e., a(x,y) = constant, analytical solution of the inverse attenuated Radon transform given in equation 5.2 can be derived, and the radioactivity distribution can be estimated accurately.
In cardiac SPECT studies, iterative reconstruction methods have been used to compensate for the non-uniform attenuation distribution that arises from the heterogeneity of the chest region. Transmission CT methods using an external radiation source in the SPECT instrument can map the attenuation coefficient distribution in a patient. The attenuation coefficient distribution is then incorporated into the iterative reconstruction algorithms to achieve accurate compensation for the attenuation effects. The resultant SPECT images show improved quantitative accuracy and fewer image artifacts and distortions.
Methods of compensating for scatter can be grouped into two general approaches. In the first approach, scattered photons are considered to carry no useful imaging information. The compensation techniques involve estimating the scatter component and subtracting it from the measured data or from the reconstructed images to obtain scatter-free reconstructed images. Various techniques have been developed, including those using two or more energy windows during acquisition. The compensation methods based on subtraction of scattered photons provide SPECT images with increased contrast and improved quantitation. However, the methods tend to increase noise in the subtracted images.
In the second approach, the scattered photons are utilized in estimating the true radioactivity distribution. When the scatter component is not subtracted, the compensated images are less noisy than those obtained with the first approach. In one method, an average scatter response function is combined with the geometric response of the collimator-detector to form the total response of the imaging system. The total response function is then used to generate a restoration filter that approximately compensates for the geometric and scatter response. Another class of scatter compensation methods determines the exact scatter response function and incorporates it into iterative reconstruction algorithms. Since the exact scatter response function depends on the physiology of a particular patient as well as on the characteristics of the imaging system, it is nonstationary and asymmetric in shape. The determination and implementation of these scatter compensation methods require extensive computations. However, efforts are being
Page 101
made to parameterize the scatter response function and to optimize its implementation in the iterative reconstruction methods to achieve a substantial reduction in processing time.
By assuming an average and stationary geometric collimator-detector response function, restoration filters can be used to provide partial and approximate compensation for the effects of a collimator-detector. Examples are the Metz and Wiener filters. An analytical method has also been developed to compensate for the spatially variant geometric collimator-detector response under the assumption that the response function has a special mathematical form. In addition, the exact collimator-detector response function can be incorporated into iterative reconstruction methods for accurate compensation of scattering.
Three-Dimensional Reconstruction Methods for Special Collimator Designs
As discussed in the previous section, fan-beam and conebeam collimators have been used in SPECT to improve the trade-off between detection efficiency and spatial resolution as compared to the conventional parallel-hole design. Due to the special geometries of such converging-hole collimators (others include astigmatic and variable focal collimators), special three-dimensional reconstruction methods and acquisition strategies are required.
For example, the typical cone-beam collimator shown in Figure 5.5 provides an example of a fully three-dimensional SPECT system configuration where the paths of the detected photons lie on lines passing through the collimator focal point and cannot be grouped into two-dimensional transverse planes. The set of cone-beam projections taken from a 360° circular motion of the detector does not provide all the necessary samples for a complete three-dimensional reconstruction. As a result, reconstruction from this geometry constitutes a three-dimensional ''limited-angle reconstruction" problem, and the reconstructed images have artifacts and distortions as a result. Special three-dimensional reconstruction methods and data acquisition strategies are being developed to overcome this problem. One approach is to use interpolation techniques to fill part of the missing region and thus reduce artifacts due to missing data. Another approach is to gather a complete set of cone-beam projections using specially designed orbits. To ensure a complete projection data set, the path in space taken by the focal point of the cone-beam collimator must intersect every plane crossing the reconstructed image space by having rotation orbits that are not constrained to lie in a
Page 102
plane. Optimizing detector motion, while satisfying the orbit requirements and avoiding data truncation, is an area of current research in SPECT.
5.5 Research Opportunities
Promising research and development directions related to innovative detector technologies include the following:
· Investigation of new room-temperature semiconductor materials, combination of scintillators and solid-state detectors, and readout electronics for use in single photon imaging systems; and
· Development of digital scintillation cameras with high counting rates and improved imaging capabilities, including improved stability, uniformity, energy resolution, and intrinsic resolution.
In the area of better SPECT system configurations, research is recommended for the following:
· Development of new SPECT system configurations for improved detection efficiency, spatial resolution, and ease of operation; and
· Development of new collimator designs for improved trade-off between spatial resolution and detection efficiency.
The most promising research directions related to image reconstruction and mathematical approaches include the following:
· Development of analytical solutions for the SPECT reconstruction problem that include the effects of non-uniform attenuation, spatially varying scatter, and collimator-detector response;
· Development of fast and stable iterative reconstruction methods that incorporate non-uniform attenuation, spatially varying scatter, and collimator-detector response for accurate SPECT image reconstruction; and
· Development of new three-dimensional reconstruction methods and data acquisition strategies for converging-beam SPECT.
Page 103
5.6 Suggested Reading
1. Barrett, H.H., Perspectives on SPECT, Proc. SPIE 671 (1986), 178183.
2. Budinger, T.F., Gullberg, G.T., and Huesman, R.H., Emission computed tomography, in Image Reconstruction from Projections: Implementation and Applications, G.T. Herman, ed., Springer-Verlag, New York, 1979, 147-246.
3. Gullberg, G.T., Zeng G.L., Datz, F.L., Christian, P.E., Tung, C.-H., and Morgan, H.T., Review of convergent beam tomography in single photon emission computed tomography, Phys. Med. Biol. 37 (1992), 507-534.
4. Jaszczak, R.J., and Tsui, B.M.W., Single photon emission computed tomography, in Principles of Nuclear Medicine, H.N. Wagner, Jr., ed., 2nd Edition, W.B. Saunders Company, Philadelphia, 1995.
5. Shepp, L.A., and Vardi, Y., Maximum likelihood reconstruction for emission tomography, IEEE Trans. Med. Imaging MI-1 (1982), 113122.
6. Tretiak, O.J., and Metz, C.E., The exponential Radon transform, SIAM J. Appl. Math. 9 (1980), 341-354.
7. Tsui, B.M.W., Frey, E.C., Zhao, X-D., Lalush, D.S., Johnston, R.E., and McCartney, W.H., The importance and implementation of accurate 3D compensation methods for quantitative SPECT, Phys. Med. Biol. 39 (1994), 509-530.
8. Tsui, B.M.W., Zhao, X-D., Frey, E.C., and McCartney, W.H., Quantitative single-photon emission computed tomography: Basics and clinical considerations, Semin. Nucl. Med. XXIV (1994), 38-65.
















