2
The GDP Sites: Process, Facilities, Inventories, and Risks
The physical and environmental state of the GDPs and the factors affecting risks subsequent to plant closure are summarized in this chapter.
The Gaseous Diffusion Process
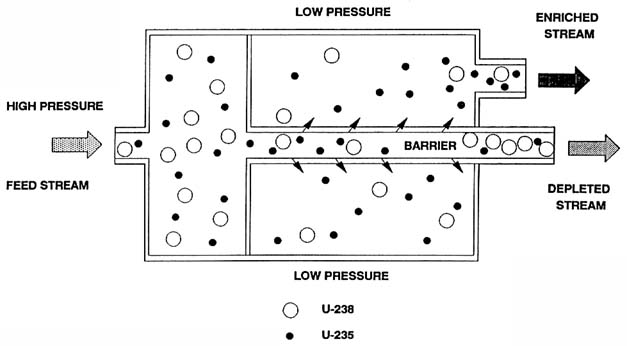
Uranium enrichment can be accomplished in a number of ways, including gaseous diffusion, gaseous separation using centrifugal force, electromagnetic separation, and atomic vapor laser isotope separation. The only method currently in commercial use in the United States is the gaseous diffusion process, which is based on the slight difference in mass between the 235U and 238U (uranium-235 and uranium-238, respectively) isotopes (Glasstone, 1950). Uranium, in the form of UF6 (uranium hexafluoride) gas, flows through a barrier tube with porous walls (see Figure 2-1). Part of the gas (nearly 50 percent) diffuses through the tube wall. The lower molecular weight 235UF6 molecules have a higher molecular velocity and diffuse more readily through the barrier pores. Consequently, the fraction of the gas that passes through is enriched in the 235 U isotope and the gas that does not pass through is slightly depleted in 235U. The efficiency of the enrichment process depends on control of the size of the holes in the barrier.
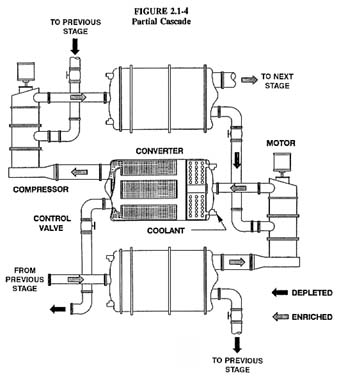
The enrichment obtained in a single diffusion operation is quite small.1 To enrich uranium for use in nuclear power plants and nuclear weapons, this operation must be repeated hundreds to thousands of times by coupling many diffuser units in a series arrangement called a cascade. The basic building block of a cascade is called a stage, which is composed of a converter vessel, gas compressor, motor, control valve, and associated piping (see Figure 2-2). The largest converters in the U.S. GDPs are 3.96 m (13 ft) in diameter by 7.31 m (24 ft) long and contain tightly packed diffusion barrier tubes that perform the isotopic separation and heat exchangers to control system temperature (see Figure 2-3). These large converters are located in the low enrichment section of the cascades; smaller converters are found in the higher enrichment sections. The size of the compressors varies directly with the size of the attached converters. The stages are connected into cells consisting of up to 12 stages, several cells are
connected into units, and several units are connected within a building (see Figure 2-4 and Figure 1-2). The large size and large number of the components require large process buildings in which to house the enrichment equipment. The process buildings contain thousands of enrichment stages with connecting piping, valving, and compressors, and there are additional connections between the process buildings.
Both axial flow and centrifugal compressors are used to compress the UF6 gas to the pressures needed for it to flow through the barrier tubes and from one stage to another. A gas cooler removes about 95 percent of the energy added by the compressor. Cooling is accomplished by a special chlorofluorocarbon (CFC) gas, CFC-114, which is in turn cooled by water. Control valves adjust the gas flow and pressure to optimize stage and cascade performance. Large block valves between successive cells permit isolating stages for maintenance.

FIGURE 2-3 Photograph of a large converter.
SOURCE: DOE (1994a).
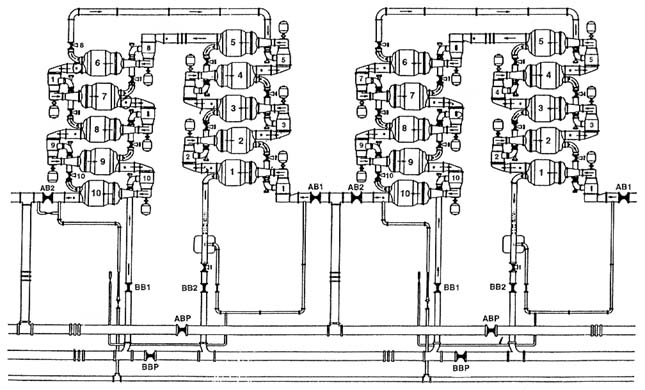
FIGURE 2-4 Arrangement of large converters showing 2 cells with 10 stages each.
SOURCE: DOE (1991a).
Enriched product is withdrawn from product withdrawal stations at the top (high enrichment section) of the main cascade into steel cylinders, allowed to cool to ambient temperature and solidify, and shipped to customers. DUF6 (depleted uranium hexafluoride) is withdrawn at tails withdrawal stations from the bottom (low enrichment section) of the main cascade and stored in 10- to 14-ton cylinders on site in a solid form. Light gases, which are readily separable from the heavier UF6, are removed in a special "purge cascade."
In addition to the process stage equipment, the GDPs require auxiliary systems, such as UF6 feed and withdrawal, electrical power distribution, and cooling towers to dissipate the waste process heat.
Site Descriptions
The GDPs are large facilities, covering hundreds of acres and having many major structures (see Figure 1-3). They are located in relatively remote areas with relatively small
population centers nearby (Table 2-1). The Oak Ridge GDP complex in southeastern Tennessee, sometimes referred to as the K-25 site, 2 is 13 miles west of downtown Oak Ridge and approximately 40 miles northwest of Knoxville; the Portsmouth GDP is in southern Ohio, 4 miles southeast of the town of Piketon, 20 miles north of the city of Portsmouth, and approximately 80 miles south of Columbus; and the Paducah GDP is in western Kentucky, 16 miles west of Paducah, and 135 miles northwest of Nashville, Tennessee. The operating plants at Paducah and Portsmouth are significant employers in their localities.
All three plants are located near water bodies: Poplar Creek runs through the Oak Ridge GDP site and the Clinch River and Watts Bar Lake bound the site; at Portsmouth, the Scioto River, which flows into the Ohio River, is 3 miles to the west; and at the Paducah GDP, the Big and Little Bayou Creeks bound the site and flow into the Ohio River, approximately 3 miles to the north. All the sites encompass large land areas, extensive building floor space, and large structures. At Paducah, the surrounding land is part of the West Kentucky Wildlife Management Area.
In 1989, the Oak Ridge GDP site was given a new mission, which includes environmental restoration (D&D of the facilities and remediation of contaminated soil, water bodies, and buried wastes), waste management (waste storage and incineration), and environmental cleanup technology development. In May 1993, the Oak Ridge GDP site was designated as the Center for Environmental Technology and the Center for Waste Management for the DOE Oak Ridge Operations Office.
In addition to the GDP site, the Oak Ridge government reservation encompasses the Oak Ridge National Laboratory, Y-12 complex, and other DOE activities. Owned by DOE and operated by Lockheed Martin Energy Systems (formerly MMES), the Toxic Substances Control Act of 1979 (TSCA) incinerator is also located at the GDP site. This facility processes hazardous organic wastes that may contain low-enriched uranium and PCBs and has already processed over 7.6 million pounds of such wastes.
There are several contaminated waste areas at the sites. At the Oak Ridge GDP, there are other waste disposal areas near the site that include radioactive, hazardous, and mixed wastes, such as the K-1070-C/D and K-1070-A waste disposal sites. At Portsmouth, buried wastes are located on site, such as in the X-749 low-level radioactive waste landfill, X-749A classified materials disposal facility, and X-701B holding pond (MMES, 1993a). The C-404 radioactive waste burial ground and C-746-S landfill are located at the Paducah site, as are several contaminated and classified scrap metal yards. The sites also have contaminated soils and groundwater, but the study scope excludes attention to remediation of these conditions.
TABLE 2-1 Characteristics of the Uranium Enrichment Facilities
|
GDP Site |
Land Areaa (acres) |
Security Area (acres) |
Number of Buildingsb |
Process Building Floor Areac (million ft2) |
Employees |
Local Populationd |
|
Oak Ridge |
1,500 |
772 |
125 |
10.9 |
3,000 |
27,000e; (68,000) |
|
Paducah |
3,423 |
748 |
161 |
6.4 |
1,850 |
27,000f; (63,000) |
|
Portsmouth |
3,708 |
500 |
109 |
8.2 |
2,600 |
1,700g; (24,000) |
|
a 1 acre = 43,560 square feet. b Not all buildings were included in the cost estimates or are expected to be included in the D&D program. For example, 13 gas centrifuge buildings at Oak Ridge were not included in the cost estimates. As of spring 1995, 82 buildings at Oak Ridge and 8 buildings at Portsmouth have entered the D&D program. The D&D program at Paducah consists of surveillance and maintenance of 15 small facilities that are shut down. c The process buildings account for about 90 percent of the under-roof area in the buildings at the GDPs. There are five process buildings at Oak Ridge (K-25, K-27, K-29, K-31, and K-33), four process buildings at Paducah (C-331, C-333, C-335, C-337), and three process buildings at Portsmouth (X-326, X-330, and X-333). d City and county populations to nearest thousand from Rand McNally (1992). e City of Oak Ridge (Anderson County). f City of Paducah (McCracken County). g City of Piketon (Pike County). SOURCE: Briefings to the committee during site visits to the Oak Ridge, Paducah, and Portsmouth GDPs, and the Ebasco cost estimate (DOE, 1991a, 1993a, 1994; MMES, 1993b). |
||||||
Buildings And Equipment
The process buildings, which house the enrichment cascades, are arranged in an open layout and include overhead cranes and wide access doors for ease of equipment maintenance. These features may also simplify decontamination operations. The process buildings are steel-framed structures with concrete floors and columns and transite (an asbestos-concrete mixture) siding. Much of the concrete floor surfaces have fluorinated, hardened coatings to prevent infiltration of spilled materials. Tie lines between the process buildings transport gaseous UF6 through the cascade. Typical heating, ventilation, and air conditioning systems are present throughout the complex, as are support systems for supplying water, power, lighting, fire protection, instrumentation, and security. The gaseous diffusion process requires a great deal of electrical power. Thousands of capacitors, transformers, and electrical switches are found at the sites, many of which contain PCBs that will require disposal.
Although about 90 percent of the total area under roof at the Oak Ridge GDP site is contained in the five process buildings, numerous support buildings and facilities must also undergo D&D. This is the case at the other two sites as well (DOE, 1991a). These support buildings include decontamination facilities, analytical laboratories, feed and withdrawal stations, and waste management facilities such as incinerators. However, the process buildings account for most of the estimated D&D cost for the GDPs (see Chapter 4). For the Oak Ridge site, the Ebasco cost estimate indicates that the five process buildings account for 92.6 percent of the D&D cost.
An active surveillance and maintenance program exists at all three sites. The current costs for surveillance and maintenance at the Oak Ridge site are close to $40 million per year (Table 2-2). Projections for surveillance and maintenance costs in DOE's Life Cycle Baseline Summary for the D&D of the Oak Ridge GDP amount to about $926 million over the period 1993 to 2019 (DOE, 1993a). A number of the buildings at the three sites have been placed in standby or shutdown mode because of changing production requirements. Most notably, the high enrichment buildings, K-25 and K-27, at the Oak Ridge GDP were shut down in 1964, and the remainder of the GDP was shut down in 1987. Some buildings have been converted to other uses such as waste storage. For example, at the Oak Ridge GDP site, hazardous wastes, low-level radioactive wastes, mixed wastes, and strategic and classified materials are being stored in the ground-level vaults of the K-25, K-31, and K-33 buildings (DOE, 1993b; Gilbert, 1995). Removing these wastes may delay the D&D of the facilities and also engender costs during the D&D, such as cleanup of contamination from waste drums, that should not be ascribed to D&D operations at the GDPs.
The large size of the facilities entails large quantities of various materials, including potentially valuable metal. Converters are generally constructed of nickel-plated steel; their weights range from about 3.3 metric tons (3.7 tons) for the smallest units to 29 metric tons (32 tons) for the largest units. Because there are thousands of these converters, the weight of metals in the process equipment is substantial, in excess of 60,000 metric tons at each site (Lemmon, 1994). Table 2-3 indicates the total amounts of metal expected to result from the D&D of the
TABLE 2-3 Estimated Radioactive Scrap Metal Streams from the D&D of the GDPs (thousands of tons)
|
Category |
Oak Ridge |
Paducah |
Portsmouth |
Total |
|
Ferrous metals/steel |
103.7 |
74.0 |
91.4 |
269.1 |
|
Aluminum/copper |
8.5 |
6.1 |
7.6 |
22.2 |
|
Copper wire tubing/valves |
17.6 |
11.7 |
15.0 |
44.3 |
|
Monel pipe/valvesa |
1.7 |
1.2 |
1.5 |
4.4 |
|
Nickel |
22.1 |
15.9 |
19.8 |
57.8 |
|
Miscellaneous |
123.2 |
81.9 |
105.0 |
310.1 |
|
NOTE: Ferrous metals and steel excludes some structural steel left in place in decontaminated building structures. Miscellaneous includes electrical instrumentation equipment and housings. a Monel is a high-nickel–content alloy containing about 27% copper, 68% nickel, and 2% to 3% iron. SOURCE: DOE (1993b). |
||||
GDPs.3 In addition, about 45,000 metric tons (50,000 tons) of brick and concrete block will result from D&D of the Oak Ridge facility (DOE, 1991a, 1993b). These large quantities of materials also result in the potential for large amounts of low-level radioactive waste from D&D. For example, the Ebasco and TLG cost estimates assumed minimal recycle of material and calculated large but different quantities of low-level radioactive and hazardous wastes under different assumptions (see Appendix J).
DOE pursued gas centrifuge enrichment technology from 1960 to 1985 as an advanced technology to replace gaseous diffusion. Thirteen buildings at the Oak Ridge site and six buildings at Portsmouth house gas centrifuge enrichment facilities that are covered by the D&D program. These facilities are contaminated and contain classified equipment and materials. While these buildings are excluded from the scope of this study, some of this study's findings and recommendations may be applicable to their decommissioning.
Contamination
Most of the data on building contamination reviewed by the committee pertains to the Oak Ridge site. Health physics and other data were reviewed to assess building contamination. Data sufficient for D&D planning needs are not available. (A pilot cell characterization project was planned at Oak Ridge, but it was not carried out.) Environmental audits at Portsmouth and Paducah provided another source of data (Faulkner, 1994b; Faulkner and Dykstra, 1994). Briefings to the committee during its site visits provided additional data.
Radiological Contamination
The main radioactive element to be characterized is uranium, which is both the feed and product of the gaseous diffusion process in the form of UF6. Natural uranium was the most common feed. It consists of the isotopes 238U, 235U, and 234U in trace quantities, and a few relatively short-lived radioactive decay products. Uranium recycled after use in nuclear reactors, primarily from military plutonium production reactors at Hanford and Savannah River, was occasionally fed to the cascades (MMES, 1992). The actinides and fission products that remained in the feed to the plants after uranium recovery, purification, and fluorination were technetium-99 (99Tc), 236U, and traces of plutonium-239 (239Pu) and neptunium-237 (237Np) (see Appendix E, Table E-1).4 Reactor return feeds were shipped directly to Paducah (88 percent) and Oak Ridge (12 percent). Contamination also occurred at the Portsmouth plant. The decay characteristics of the principal radionuclides present at the GDPs are summarized in Table 2-4. (See Appendix E for additional information on radionuclides present at the GDPs.)
TABLE 2-4 Radionuclide Decay Characteristics
|
Radionuclide |
Radioactive Decay Products |
Half-Life (years) |
Radiationa |
|||
|
|
|
gamma |
|
|||
|
Alpha (MeV) |
Beta (maximum MeV) |
(MEV) |
(%)b |
|||
|
238U |
|
4.47 × 109 |
4.2 |
— |
— |
— |
|
|
234 Th |
6.60 × 10-2 |
— |
0.19 |
0.093 |
16 |
|
|
234mPac |
2.22 × 10-6 |
— |
2.29 |
1.001 |
0.9 |
|
234U |
|
2.45 × 105 |
4.7 |
— |
— |
— |
|
235U |
|
7.04 × 108 |
4.4 |
— |
0.185 |
54 |
|
|
231Th |
2.92 × 10-3 |
— |
0.31 |
0.084 |
8 |
|
236U |
|
2.34 × 107 |
4.4 |
— |
— |
— |
|
99Tc |
|
2.14 × 105 |
— |
0.29 |
— |
— |
|
237Np |
|
2.14 × 106 |
4.8 |
— |
0.086 |
14 |
|
|
233Pa |
7.39 × 10-2 |
— |
0.57 |
0.312 |
34 |
|
239Pu |
|
2.44 × 104 |
5.1 |
— |
— |
— |
|
a—= not applicable b % = 100 × (number of gamma/second)/(number of disintegrations/second); only the most intense gamma ray is listed. c m = metastable. |
||||||
Most of the uranium now at the GDPs is the depleted fraction, stored in cylinders on site. After process shutdown, some solid deposits of uranium and its impurities will remain within the cascade. A small fraction is distributed throughout the sites due to leaks, spills, repairs, cascade upgrading, and storage of product and waste. More uranium compounds will undoubtedly be spread by dismantling activities, although great care will be taken to avoid the spread of radioactive or hazardous materials in amounts above that permitted by DOE Orders or federal and state statutes.
The extent of contamination on external surfaces in the facilities, such as floors, walls, structural steel, and exterior surfaces of process equipment and instrumentation, is not well known. The data available from the Oak Ridge GDP (Person, 1995a; DOE, 1991b) show uranium and 99Tc contamination to some degree in all process buildings and in some support buildings (Table 2-5). In areas where the extent of contamination is 90 percent of surface area or greater, both uranium and 99Tc are present. One hundred percent of the interior of the process equipment is assumed to be contaminated.
An extensive nondestructive assay survey for 235U deposits in the process equipment (100,000 individual measurements) has been completed for the 54 units in the K-25 building and
TABLE 2-5 Estimated Percentages of Surface Areas at the Oak Ridge GDP Contaminated with Radioactive Materials
|
Site Facilities |
235U Enrichment |
Building Floor Space |
Building Walls |
Structural Steel |
Nonprocess Equipment |
Process Equipment Housings |
Process Electrical Systems |
Uranium Processing Equipment |
|
K-25 process building (94%) |
20-90 |
15 |
10 |
10 |
Unknown |
90 |
—a |
100 |
|
311-1 Area (6%) |
3-5 |
95 |
90 |
90 |
Unknown |
100 |
—a |
100 |
|
K-27 process building (80%) |
Up to 20 |
15 |
15 |
15 |
Unknown |
90 |
—a |
100 |
|
402-9 Area (20%) |
3-5 |
90 |
80 |
80 |
Unknown |
100 |
—a |
100 |
|
K-29 process building |
2-10 |
90 |
80 |
85 |
Unknown |
NA |
—a |
100 |
|
K-31 process building |
1-4 |
15 |
10 |
10 |
Unknown |
50 |
—a |
100 |
|
K-33 process building |
0.2-2 |
5 |
5 |
5 |
Unknown |
50 |
—a |
100 |
|
Process support buildings |
0.2-5 |
15 |
10 |
10 |
Unknown |
70 |
—a |
100 |
|
Decontamination buildings |
0.2-5 |
95 |
90 |
90 |
Unknown |
100 |
—a |
100 |
|
Water and electrical facilities |
NAb |
—c |
—c |
—c |
Unknown |
NA |
NA |
NA |
|
NOTE: This assessment is based on limited data collected during facility operation; detailed characterization has been delayed until D&D activities are initiated in the buildings. Surfaces are considered contaminated if 5,000 disintegrations per minute (dpm) or more can be detected. Surface contamination can be found from 5,000 dpm/100 cm2 to greater than one million dpm/100 cm2. Most of the contamination is either uranium or 99Tc. Areas 311-1 and 402-9 are the purge cascades for the K-25 and K-27 buildings respectively. Area 311-1 operated until 1977 and 402-9 operated until 1985. a Due to the difficulties in surveying motor and transformer windings, these components have been treated as contaminated in previous maintenance programs. b NA = Not available. c Spot contamination may be found. SOURCE: Faulkner (1994a, b). |
||||||||
the nine units in the K-27 building at the Oak Ridge site (MMES, 1992). The technique used has not been validated and may be subject to considerable error.5 Less detailed surveys were conducted of lower enrichment sections found in the other buildings. The results indicate that there is a large amount of uranium (on the order of tons) present in the process equipment (Table 2-6). Some uranyl fluoride (UO2F2), and uranium fluoride-metal reaction products are found, in a fairly thin coating covering the inside surfaces of the operating equipment. In addition, substantial deposits of UO2F2 occur at many locations, presumably at the sites of past leakage or spills.6 Although 235U is present in the process equipment, the major nuclide present is, of course, 238U. Small concentrations of 234U will also be present and contribute disproportionately to activity of the products. Of the several tons of uranium deposits, much of it is enriched and could present a criticality hazard in the presence of water.7 A number of the stages at the Oak Ridge and Portsmouth GDPs will contain highly enriched uranium as well as moderately enriched uranium having enrichment levels between 5 and 20 percent (Murray, 1994).
Most of the radioactive contamination, both within the process equipment and on exposed areas in the plants, occurs on the surface of materials and should be easily removable. Some, however, is "buried" within the material or painted over. For example, the diffusion barrier, which is made of nickel, contains some uranium within the diffusion pores; concrete may contain radioactive contamination within cracks; and small-diameter tubing may contain internal deposits. For deposits of uranium inside some piping and small-diameter tubing, and for such surfaces as compressor bearings, balding, and valve bodies, contamination may be difficult to access and remove. It would also be difficult to measure levels of contamination in such pieces of equipment to verify compliance with surface contamination standards.
Nonradioactive Contamination
A number of hazardous chemicals and materials, such as PCBs, asbestos, oils, coolants, lead, and other miscellaneous materials must be dealt with in cleaning up the facilities (see Table 2-7). Gaskets used on ventilation ductwork contain PCB oils. Tens of thousands of gaskets are in place at each plant. A Federal Facilities Compliance Agreement requires each plant to have a control and removal program for PCBs. A multimillion dollar program at the Portsmouth and Paducah GDPs has just been completed to install troughs for all PCB gaskets to prevent leaks of PCBs onto the floors. At the Oak Ridge GDP the current plan is to destroy these PCBs during D&D. The committee believes this is a sensible approach that avoids the large and useless expenditure of removing and replacing the PCB-impregnated gaskets, only to remove and destroy the clean replacements after decontamination operations.
|
5 |
The uncertainty currently used for the nondestructive assay technique is ±50 percent for mass and ±20 percent for assay. One goal of the DOE deposit removal program is to assess these uncertainties. |
|
6 |
The cascades operate with UF6, a very reactive form of uranium, which reacts with metal to form and deposit thin wall films of solid UF4 (uranium tetrafluoride). The UF6 also reacts quickly with moisture from in leakage of air to form solid deposits of UO2F2. |
|
7 |
Water acts as a moderator that slows down neutrons and enhances the chance of a 235U nucleus capturing a neutron and fissioning (see Chapter 3 and Appendix G). |
TABLE 2-6 Estimated Uranium Deposits at the Oak Ridge GDP
|
|
Compressors |
Converters |
||||||
|
Building |
Typical Surface Deposition (kgU) |
% Equipment with Large U Deposits (% > kgU) |
Maximum Deposit Measured (kgU) |
Typical Surface Deposition (kgU) |
% Equipment with Large U Deposits (% > kgU) |
Maximum Deposit Measured (kgU) |
Total (kgU) |
|
|
|
K-27 |
0.4 |
NAa |
1 |
1 |
NA |
1 |
1,000 |
|
K-29 |
0.5 |
7% > 2 kgs |
60 |
1 |
5% > 2 kgs |
120 |
5,0000 |
|
|
K-31 |
0.5 |
1% > 5 kgs |
20 |
2 |
9% > 5 kgs |
130 |
6,000 |
|
|
K-33 |
0.5 |
3% > 30 kgs |
100 |
3 |
8% > 30 kgs |
120 |
11,000 |
|
|
Total |
|
|
|
|
|
|
23,000 |
|
|
NOTE: Data for the K-25 building, which housed the enrichment equipment for achieving highly enriched uranium, is not shown because information on amounts of highly enriched uranium is classified. The deposits indicated in the table do not necessarily represent a criticality risk but are shown to indicate the extent to which uranium is still in the equipment. a NA = Not applicable. SOURCE: Faulkner (1994a,b). |
||||||||
TABLE 2-7 Estimates of Key Hazardous Contaminants at the Uranium Enrichment Facilities
|
GDP Site |
Asbestos (ft2)a |
PCBs |
Lubrication Oils (thousand gal) |
Freons®b (thousand lb) |
|
Oak Ridgec |
3,230,000 |
20,000 gaskets; 125 drained transformers; 500 capacitors |
Negligible |
Negligible |
|
Paducah |
3,000,000 |
16,000 gaskets; 73 transformers (285 metric tons of PCBs); 5,084 capacitors; 2,800 metric tons of PCB waste |
500 |
7,100 (in cascade) |
|
Portsmouth |
4,962,000d |
155 transformers (700 metric tons of PCBs); 11,110 capacitors; 2,200 metric tons of PCB waste in storage |
600 |
6,700 (in cascade) 1,100 (in tank car storage) |
|
NOTE: Additional contaminants include 99Tc, 237N, chromated cooling water, mercury (in switches), cadmium, nickel, and various acids. Portsmouth also has 68 million pounds of lithium hydroxide in storage, the disposition of which is not included in the D&D cost estimates. a As transite building siding, ceiling panels, and floor tiles (nonfriable) and as insulation for power cables, cable trays, water liner/heaters, and steam/condensate lines (friable). Significant quantities may also be radiologically contaminated; some may also be PCB contaminated. b Primarily, CFC-114. Most material from the Oak Ridge GDP has been transferred to the Portsmouth and Paducah plants. c Asbestos and PCBs are being removed from Oak Ridge, so estimates of PCBs and asbestos will change over time. For example, removal and disposal of approximately 9,500 PCB-containing capacitors has occurred; 20,000 gallons of lubrication oil has been incinerated; and the Freons® were sent to the Paducah GDP for reuse. d There are also approximately 50 miles of asbestos insulation on pipes, 804,000 linear ft of asbestos cloth on instrument lines, and 10,000 linear ft of asbestos wiring. SOURCE: Briefings to the committee during site visits and the Ebasco cost estimate (DOE, 1991a). |
||||
PCBs are also present in electrical components, such as transformers and capacitors. Where possible, transformers have been or are being drained, cleaned, and refilled with non-PCB oil. Capacitors are being incinerated and new non-PCB capacitors are being installed at the operating plants as required. Removal and disposal of about 9,500 PCB capacitors at the Oak Ridge GDP has occurred. A large number of PCB-contaminated components, including tens of thousands of capacitors, remain to be removed during D&D.
Asbestos is largely present in two forms: as nonfriable transite siding and as potentially friable insulation around piping. Miles of piping are insulated by asbestos products. Removal has already begun at the Oak Ridge GDP. To the extent that buildings are left standing and reused, removal of some transite walls could be avoided. BNFL experience during the D&D of the Capenhurst GDP in the United Kingdom showed that walls can be cleaned of radiological contamination and sealed to control asbestos dust prior to building reuse. All remaining asbestos piping insulation will require removal and disposal in an appropriate landfill. Hundreds of thousands of cubic feet of other materials containing asbestos, such as roof flashing, are also present.
Large quantities of lubricating oils and process coolants (CFCs) are used at the GDPs. At the Oak Ridge GDP, efforts were recently completed to dispose of these materials (DOE, 1993a). For example, the oil was burned at the Oak Ridge TSCA incinerator. The inventory that will remain at the Oak Ridge GDP and be considered a D&D cost is unknown but will likely be small. At the other two plants, alternative non-CFC coolants are being examined to replace existing coolants. USEC will be responsible for the disposal of non-CFC coolants whose disposal costs are expected to be lower than those for CFCs.
Because of the age of the buildings, much of the paint used over the years was lead-based. No estimates of area covered by this paint are available, but it is probably large. The cooling towers contain wood contaminated with chromium, which have been demolished and disposed of appropriately. Miscellaneous chemicals and contaminants include mercury, chromates, cyanides, fluoride compounds, organic solvents, and lead. To deal with lead paint, for example, the TLG cost estimate assumed removal by scabbling (DOE, 1991b). The surface material removed would have to be appropriately disposed of, and some sampling would probably be required. Cleanup of these materials does not represent a large component of the D&D cost estimates.
Storage Of DUF6
DOE has responsibility for the 528,000 metric tons of DUF6 that are stored in approximately 46,000 steel cylinders at the 3 sites. The largest amount is at the Paducah site (Snyder, 1994). Since the Paducah and Portsmouth GDPs are operating plants, they continue to accumulate cylinders containing DUF6. However, the DUF6 produced since the USEC started operating the facilities is the responsibility of the corporation.
Site Differences
While the three GDPs have many similarities, they have differences that can affect D&D cost. The three GDPs differ in the number, size, and physical arrangement of the stages and in the number and type of ancillary structures. Table 2-8 compares the number of stages, cells, and units by equipment size found in the process buildings at the three GDP sites.
The Portsmouth and Paducah GDPs are still operating. Current plans are to remove uranium remaining in the cascades at the end of their operating life through in situ gaseous decontamination using chlorine trifluoride (ClF3), both to remove a significant amount of the uranium deposits and to eliminate nuclear criticality risks for remaining deposits in the cascade. As noted earlier, the removal of uranium deposits representing criticality risks is the responsibility of the USEC. If the in situ gaseous decontamination process is as effective for removal of uranium deposits at the U.S. GDPs as at the Capenhurst GDP, a planned and orderly shutdown that removed uranium deposits should significantly reduce concerns about criticality accidents and special nuclear material accountability during the disassembly and decontamination of cascade components.
In 1992, DOE ceased operations of the high-enrichment cascade at the Portsmouth GDP. The agency has been removing uranium deposits in this cascade using in situ gaseous decontamination, but some uranium deposits will remain. There are plans to test gaseous decontamination at room temperature (the temperature that would exist for this procedure in the nonoperating plant at Oak Ridge) using high concentrations of ClF3.
When the K-25 and K-27 buildings at the Oak Ridge GDP site ceased operations in 1964, and the remainder of the plant shut down in 1985, no systematic in situ gaseous decontamination or mechanical removal was used to remove uranium deposits. As a result, the cascades contain deposits of enriched uranium, some quite large, and represent a potential criticality risk during D&D, especially in the K-25 building, which contains the high-enrichment section of the cascade. For this reason, as well as for control of special nuclear materials, the DOE has initiated a deposit removal program to remove, either mechanically or with low temperature gaseous treatment, highly enriched uranium deposits. The goal of phase I of this program is to remove deposits of more than 500 g in an unfavorable geometry from about 60 components in the Oak Ridge process equipment. The components, however, would not be cleaned to free-release levels. Tests by DOE on the low temperature gaseous decontamination process are aimed at assessing the applicability of the process to the Oak Ridge cascade.
The Paducah GDP differs from Oak Ridge and Portsmouth in that the maximum uranium enrichment level was about 2 percent, although its enrichment level increased to 2.75 percent in 1995. The low enrichment level diminishes the concern with nuclear criticality accidents and the need to safeguard highly enriched uranium. Thus, costs for safeguards, security, and criticality monitoring at the Paducah GDP should be less than for the other plants. The modification of the Paducah GDP to 2.75 percent 235U product may increase somewhat the risk of a criticality accident during D&D.
TABLE 2-8 Cascades and Stages in the Process Buildings at the Three GDP Sites
|
|
|
Converter Equipment Sizea |
|||||
|
GDP |
Item |
25 |
27 |
0 |
00 |
000 |
Total |
|
Oak Ridge |
Stages |
3,018 |
540 |
300 |
600 |
640 |
5,098 |
|
|
Cells |
507 |
90 |
30 |
60 |
80 |
767 |
|
|
Units |
54 |
9 |
3 |
6 |
8 |
80 |
|
|
Buildings |
1 |
1 |
1 |
1 |
1 |
5 |
|
Portsmouth |
Stages |
1,620 |
720 |
600 |
500 |
640 |
4,080 |
|
|
Cells |
140 |
60 |
60 |
50 |
80 |
390 |
|
|
Units |
7 |
3 |
6 |
5 |
8 |
29 |
|
|
Buildings |
0.7 |
0.3 |
0.5 |
0.5 |
1 |
3 |
|
Paducah |
Stages |
0 |
60 |
0 |
800 |
960 |
1,820 |
|
|
Cells |
0 |
10 |
0 |
80 |
120 |
210 |
|
|
Units |
0 |
1 |
0 |
8 |
12 |
21 |
|
|
Buildings |
0 |
1 |
0 |
2 |
2 |
5 |
|
Total |
Stages |
4,638 |
1,320 |
900 |
1,900 |
2,240 |
10,998 |
|
|
Cells |
647 |
160 |
90 |
190 |
280 |
1,367 |
|
|
Units |
61 |
13 |
9 |
19 |
28 |
130 |
|
|
Buildings |
1.7 |
2.3 |
1.5 |
3.5 |
4 |
13 |
|
a The weights of the equipment can be found in Chapter 4, Table 4-8. SOURCE: Faulkner (1994a); Person (1995b). |
|||||||
Risks Subsequent To GDP Closure
Subsequent to closure of a GDP, there may be risks to human health if some pathway allows significant exposure to the hazardous materials within the facilities. It does not appear to the committee that there is any current appreciable risk of exposure of the public to these contaminants (e.g., at the Oak Ridge GDP) because the vast majority of the contamination is contained within the plant systems. As the facilities age, and the containment integrity of the process equipment and buildings begins to fail, the risk of exposure of the public to the contaminants will tend to slowly increase.
Exposure to these hazard sources could develop in a variety of ways. If high levels of loose surface contamination are exposed to the atmosphere, contamination can be resuspended and transported inside and outside the structure. However, very high levels of loose surface contamination would be necessary for this pathway to be significant for public health, and, so far as the committee is aware, this situation is not the case. Human and industrial actions within the buildings could resuspend loose asbestos fibers, leading to airborne concentrations of friable asbestos and asbestos dust. These pathways are primarily of concern to building occupants, and techniques, such as removal, fixation, over painting, or ventilation, are available to control these risks. A major fire in the facilities could give rise to updrafts that send contamination into the atmosphere to be deposited on and off site and could also create toxic combustion products. A major fire could result in exposure of the public to contaminants, although it is likely that the bulk of the contamination would be deposited on site because of the relatively high settling velocities of particulates.
Rainwater, particularly after significant roof and process equipment decay, can transport contaminants into groundwater and surface water supplies. Public exposure could then result from the use of the affected water supplies for drinking, irrigation, or recreation. Atmospheric moisture and natural disasters (such as fires, earthquakes, high wind conditions, or flooding) could also accelerate degradation of the structures now restricting the mobility of contaminants. Failures of water services within the buildings, such as fire protection systems, could also wash contamination down through floor drains and over sills into the external environment. Continued exposure of DUF6 cylinders to the environment could result in corrosive failure of the cylinders, particularly if they have been damaged during previous handling operations (DOE, 1992; DNFSB, 1995). A failed cylinder at ambient temperature could slowly release the DUF6 and any other contamination from within a cylinder, although experience to date indicates that release rates would be very low.
Industrial safety risks from falls and falling objects is increased by degradation of the structural integrity of parts of the buildings, such as roofs, stairways, or handrails. These worker risks are a function of both building degradation and worker activity levels within the building and thus will increase during surveillance and maintenance and D&D activities. Because structural degradation will likely progress as time goes by, these risks will tend to increase over time. Maintenance can reduce these risks by correcting safety problems, although surveillance and maintenance workers would be subject to increased risks as a result of working in the aging facilities.
D&D activity allows other types of exposures, primarily to D&D workers. Cutting, grinding, and scabbling can result in liberation of vapors and particulate matter to the atmosphere. Industrial safety concerns during the extensive moving and lifting operations in dismantling process equipment would be significant, particularly if the structural integrity of the buildings were allowed to deteriorate before the start of D&D. Handling of the DUF6 cylinders could also lead to worker exposures to hydrogen fluoride (HF) in the event of a cylinder rupture. HF is a reaction product generated when UF6 gas comes into contact with moisture-laden air. It is an extremely corrosive vapor, even when diluted in air, and can cause ulceration of the larynx and trachea. Acute exposure to HF vapor, such as from the high-temperature rupture of a cylinder containing UF6, can be fatal. However, HF dissipates relatively quickly, and concentrations at the site boundary from such a rupture could be expected to be minimal,
although they would depend on atmospheric conditions. (See Chapter 7 for more discussion of potential hazards to workers and the public.)
Uranium (like many heavy metals) is nephrotoxic, causing kidney damage if ingested in sufficient quantities (Maynard and Hodge, 1949). The health effects associated with the heavy metal toxicity of uranium may be more significant than its radiological effects, particularly in the case of low specific activity isotopes such as 238U. The health effects of exposure to radioactivity are well documented (NRC, 1990). The radionuclides of concern in the diffusion plant complex emit primarily alpha particles and low-energy beta particles and therefore pose a health risk only if ingested or inhaled. Alpha and beta contamination inside process equipment does not pose an exposure threat to building occupants because the radiation associated with the contaminants is absorbed by the process equipment material. External surface deposits can pose an exposure risk, but occupant protection can be maintained by removal, partial removal, over painting of the deposits, or the use of appropriate respiratory protection. Gamma-emitting radionuclides, such as 235U, can create an external exposure risk, but this radionuclide appears to be present in significant quantities only in small portions of the Oak Ridge plant, for example, primarily in the high-enrichment sections of the process equipment.
PCB compounds are classified as probable human carcinogens (EPA, 1993) based on animal studies. PCB compounds are also toxic to the liver and reproductive system, and dermal exposure to concentrated PCB compounds can cause chloracne (Crine, 1988). Also, although PCB compounds are flame-retardant (which is the reason for their use), they can ignite at very high temperatures, and their combustion products can be extremely toxic. However, PCB compounds are typically composed of a mixture of chlorinated biphenyl isomers, and the physical and toxicological properties of these compounds vary widely.
Inhaled asbestos fibers are classified by the EPA as human carcinogens based upon epidemiological studies of occupationally exposed workers (EPA, 1993). Inhalation of high levels of asbestos fibers can also lead to asbestosis, an occupational hazard to building occupants, if high concentrations of asbestos fibers are allowed to persist and appropriate protection is not used.
Because of the presence of enriched uranium in areas of the facilities, the potential exists for an uncontrolled nuclear criticality event (see Appendix G for a discussion of the causes and consequences of such events). Criticality events create an acute risk to any nearby workers, which is dependent on the magnitude of the neutron burst during the event. These events also lead to the creation of fission products. Criticality risk varies with the degree of enrichment in 235U, the mass of enriched uranium, the presence of a moderator (such as water), and the geometry of the solid uranium mass (see Appendix G). Sufficient masses of enriched uranium to create a criticality risk may be present in deposits internal to process equipment, or may accrue from waterborne transport of soluble uranium (either in the process equipment or during improper aqueous decontamination operations). Within the facilities, water (either rainwater leaking through roofs or breaks in water service mains) may enter process equipment through breaches in the equipment. Such breaches can result from the removal of parts from shutdown process units or from seismic events or collapse of external structures. Finally, the process equipment was not designed to ensure safe geometries for uranium in aqueous solution. It appears that current efforts to remove uranium deposits from the equipment, combined with
procedures to avoid water leaks into process equipment, will be adequate to avoid significant risks of criticality. However, active management and maintenance will be required until the enriched uranium deposits are removed from the process equipment.
Conclusions
In summary, the GDP sites are large facilities with substantial quantities of contaminants. Conclusions pertinent to the D&D include the following:
- Although the presence of radioactive and hazardous materials constrains the approach that can be taken to the D&D of these facilities, the D&D of the GDPs is basically a demolition project.
- The repetitive layout and large numbers of pieces of enrichment equipment, and the open layout of the process buildings including overhead cranes and wide access doors, offer opportunities for simplified D&D activities and for economies of scale in the operations.
- Large quantities of valuable metals are present in the GDPs. These include the nickel barrier membrane, which contains uranium deposits and some 99Tc contamination within its porous structure, and many miles of copper tubing that will have to be stripped of insulation.
- Deposits of enriched uranium compounds are present within the process equipment, have been characterized reasonably well, and are large enough to present a criticality hazard in the presence of a moderator, such as water. The largest deposits occur at the Oak Ridge GDP.
- Some deposits of uranium will be difficult to access in small-diameter tubing and piping, on compressor bearings and balding, and on valve bodies.
- Data indicate that uranium and 99Tc contamination exists to some degree in all process buildings and in some support buildings.
- The risks associated with the GDPs after shutdown are principally from DUF6 cylinders and any remaining enriched uranium. Deteriorating structures will increase the risk. The risks to the surrounding communities are small, but they are not so small that these plants can be abandoned. Risks will increase with time, and D&D will be necessary.
References
Crine, J.P., ed. 1988. Hazards, Decontamination, and Replacement of PCBs: A Comprehensive Guide. New York: Plenum Press.
DNFSB (Defense Nuclear Facilities Safety Board). 1995. Integrity of Uranium Hexafluoride Cylinders. Washington, D.C.: DNFSB. April 27.
DOE (U.S. Department of Energy). 1991a. Environmental Restoration of the Gaseous Diffusion Plants. Technical Summary Document, vol 1. Oak Ridge, Tennessee: Ebasco Environmental for DOE. October.
DOE. 1991b. Preliminary Cost Estimate for D&D of the Gaseous Diffusion Plants. Document S14-25-002. Bridgewater, Connecticut: TLG Engineering for Systematic Management Services for DOE. September.
DOE. 1992. U.S. Department of Energy Independent UF6 Cylinder Assessment Team Draft Preliminary Report. Oak Ridge, Tennessee: DOE Oak Ridge Field Office.
DOE. 1993a. K-25 Site Decontamination and Decommissioning Program Life Cycle Baseline Summary. K/ER/INT-11. Oak Ridge, Tennessee: Martin Marietta Energy Systems for DOE. August.
DOE. 1993b. Oak Ridge K-25 Site Technology Logic Diagram, vol. 1, Technology Evaluation. Oak Ridge, Tennessee: Oak Ridge K-25 Site Office for DOE's Office of Technology Development. February 26.
DOE. 1994. Briefing Book. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, Oak Ridge Uranium Enrichment Facility, Oak Ridge, Tennessee, February 3, 1994 .
EPA (Environmental Protection Agency). 1993. Integrated Risk Information System. Washington, D.C.: EPA. January.
Faulkner, R. 1994a. Decontamination and Decommission of Uranium Enrichment Facilities. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, National Academy of Sciences, Washington, D.C., March 28, 1994.
Faulkner, R. 1994b. Site Characterization. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, National Academy of Sciences, Washington, D.C., June 15, 1994.
Faulkner, R., and J. Dykstra. 1994. Extent and Kinds of Contamination at K-25 Site. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, National Academy of Sciences, Washington, D.C., March 28, 1994.
Gilbert, W. 1995. Waste Management at K-25 Site. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, National Academy of Sciences, Washington, D.C., May 8, 1995.
Glasstone, S. 1950. Sourcebook on Atomic Energy. Princeton, New Jersey: D. Van Nostrant Company, Inc.
Lemmon, W. 1994. Differences Between K-25 and Paducah. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, Portsmouth Uranium Enrichment Facility, Piketon, Ohio, August 22, 1994.
Maynard, E., and H. Hodge. 1949. Studies in the toxicity of various uranium compounds when fed to experimental animals. In: The Pharmacology and Toxicology of Uranium Compounds, vol. 1, C. Voegtlin and H.C. Hodge, eds. New York: McGraw Hill.
MMES (Martin Marietta Energy Systems). 1992. Historical Characterization, K-25 and K-27 Buildings. K/D-605. Oak Ridge, Tennessee: MMES.
MMES, 1993a. Portsmouth Gaseous Diffusion Plant: Environmental Report for 1992. ES/ESH-37. Portsmouth, Ohio: Martin Marietta Energy Systems. September.
MMES. 1993b. Life Cycle Baseline Summary for Surplus Facilities Demolition. POEF-ER-4568, vol. 11, revision 0. Portsmouth Ohio: Martin Marietta Energy Systems. August.
Murray, A. 1994. Review of the Ebasco Cost Estimate and Draft Program Planning. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, National Academy of Sciences, Washington, D.C., June 15, 1994.
NRC (National Research Council). 1990. Health Effects of Exposure to Low Levels of Ionizing Radiation (BEIR V). Washington, D.C.: National Academy Press.
Person, G. 1995a. Personal Communication (letter) from Gary Person, Oak Ridge GDP site, to George Gans, Booz Allen, February 4, 1995, forwarded to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities.
Person, G. 1995b. Personal communication from Gary Person, K-25 Decontamination and Decommissioning (D&D) Group, to Richard Smith, October 24, 1995.
Rand McNally. 1992. Road Atlas, United States, Mexico, Canada. 68th Edition (Rediscover America Edition). Chicago, Illinois: Rand McNally.
Snyder, R. 1994. Ohio Environmental Protection Agency Compliance Status of UF6 Storage. Presented to the Committee on Decontamination and Decommissioning of Uranium Enrichment Facilities, Portsmouth Uranium Enrichment Facility, Piketon, Ohio, August 22, 1994.