2
Naturally Occurring Carcinogens and Anticarcinogens in the Diet
This chapter addresses two questions: (1) what is the current state of knowledge regarding the presence and availability of carcinogens and anticarcinogens in the human diet? and (2) how much do we know about the dietary factors that modify carcinogenesis?
Exposure To Naturally Occurring Chemicals
Naturally occurring chemicals present in our food supply can be classified into the following five categories: constitutive naturally occurring substances, derived naturally occurring substances, acquired naturally occurring substances, pass-through naturally occurring substances, and added naturally occurring substances. These are defined in Chapter 1.
Environmental exposures to naturally occurring chemicals occur principally from the food and water we consume (approximately 1-1.5 kg/day of each) and from inspired air (approximately 18 kg/day). While air and water frequently contain at least trace levels of contaminants of human origin, they are seldom a source of naturally occurring substances that raise health concerns, including those about cancer. Among the uncommon exceptions is arsenic. Although it occurs at a few parts per billion (ppb) in most drinking waters, it occurs at the part per million (ppm) level in spring, well, and surface waters in arsenic-rich areas in the United States and in many other countries (Underwood 1973, NRC 1977). Food, however,
is overwhelmingly our major source of exposure to naturally occurring chemicals and is therefore the focus of this report.
The Composition of Foods
Food, of course, is simply what we choose to eat, a choice heavily influenced by availability and culture (Pyke 1968, Jenner 1973, Tannahill 1973, NRC 1975). Although practical experience has taught us to avoid certain plants or animals because eating them results in illness, that experience is limited, largely anecdotal, and incomplete. We usually avoid acute toxicants—those things that make us unpleasantly sick immediately. However, we rarely possess sufficient knowledge about foods that contain naturally occurring toxicants that could cause delayed or chronic effects, including cancer.
In contrast, there are many potentially useful foods we avoid or disregard for reasons of unawareness, aesthetics, religion, culture, or cost. All human diets that sustain life and normal activity must supply at least the minimum quantity of the essential nutrients, including calories. Even given differences in age, body weight, and activity level, the range of those requirements for children and adults is fairly narrow—less than threefold. The range of variation in the foods that supply those nutrients, however, is enormous. Contrast the traditional Eskimo diet, high in animal fat and protein, with the vegetarian diet of the Seventh-Day Adventist or Hindu. Many diets in developing countries are low in animal protein simply because it is too expensive or unavailable. The use of spices and seasonings is often a distinctive cultural mark (Rozin 1973). The foods we choose to eat are merely a fraction of those we could eat. Furthermore, many dietary patterns shift over time, as demonstrated by our current—but recent—broad North American fondness for traditional Italian, Asian, and Latin American foods.
The variety in our modern food supply is due largely to the many
different species of plants we consume. There are estimated to be about 250,000 species of flowering plants, and at least 11,000 are used as foods, spices, or flavoring agents (Tanaka 1976), including vegetables, fruits, and nuts. In some cases, different parts of the same plant are used as food, such as celery stalks, celery seeds, and celery essential oil (derived from the seed). The constituents of each plant part, and hence the expected biological activities, may be entirely different.
The Major Components
In biochemistry and nutrition, it is customary to think of food in terms of its major components. Across the entire U.S. food supply (plant, animal, and microbial), these component classes are, in descending order of concentration, water, carbohydrate, fat, protein, the non-nutrients, and the micronutrients, including minerals and vitamins. On average, carbohydrates supply 46% of our calories, fats supply 42%, and proteins supply 12% (Whistler and Daniel 1985). Of these component classes, proteins are the only primary gene products, i.e., the only class of components (other than DNA and the RNAs) produced directly by the operation of the genetic code of the organism. Minerals are absorbed from the environment, including the diet. All the other component classes are secondary gene products, produced in each organism by the action of the primary gene products, the proteins.
Carbohydrates consist of single or polymerized multiple units of simple sugars, such as glucose or fructose. Glucose, by itself, occurs naturally in foods only to a very limited extent; however, it is the most abundant sugar in the world. Combined chemically with other simple sugars in disaccharides such as sucrose and in starch, a polysaccharide composed solely of glucose, it constitutes about three-fourths of total dietary carbohydrates (Whistler and Daniel 1985). In the American diet, sucrose, fructose, and glucose supply
more than half of carbohydrate calories and starch the remainder. The overall structure of nearly all dietary carbohydrates is remarkably similar—simple sugars in their ring (hemiacetal pyranose) form, linked together in chains. The large differences in their digestibility and functional characteristics lie in the length of these chains, the degree of branching, and in more subtle aspects of structure.
Lipids are a broad group of naturally occurring compounds that typically are freely soluble in organic solvents and nearly insoluble in water. The glycerol esters of fatty acids (triacyl glycerols, also called triglycerides) form up to 99% of the lipids of plant and animal origin and are customarily called fats, or somewhat more precisely, fats and oils (Hawk 1965; Anonymous 1970, 1986; Nawar 1985; NRC 1989b). Fats is the more specific term for those that are solid or semisolid at room temperature and are typically of animal origin, e.g., lard and butter. Oils, such as soy, olive, and corn oils, are liquid at room temperature and are usually of plant origin, although these distinctions have exceptions, e.g., whale oil. Those lipids that are not triacyl glycerols are quantitatively minor but of enormous physiological importance. They include cholesterol, the phospholipids in cell membranes, prostaglandins, and a host of other substances of structural and functional significance (Stryer 1975). Although all triacyl glycerols share the same basic structure, the differences in melting point, oxidative stability, nutritional qualities, and other important characteristics depend on structural aspects, such as fatty acid chain length and degree of unsaturation.
The basic structure of all proteins is that of a polypeptide—a polymer of -aminocarboxylic acids linked by amide bonds. In terminology parallel to that used for the carbohydrates, two amino acids form a dipeptide, and three form a tripeptide. Peptides containing more than three, but fewer than ten amino acids, are often called oligopeptides, and those with ten or more are polypeptides. More than 400 different amino acids occur in nature (Harborne 1993), but only 20 are found in the major food proteins. Of these,
nine are essential in human diets. As will be discussed later, proteins serve several diverse and essential purposes in the organisms that produce them. Although all are polypeptides, the number and sequence of the different amino acids, the nature of the chain—linear, branched, or ring, the additional functional groups (e.g., amino or carboxylic acid) on certain amino acids, and the three-dimensional conformation of the entire molecule determine the physiological role of each protein (Cheftel et al. 1985).
Alcohol, a nutrient only in the sense of a source of calories, is discussed in the section on ''Identifying Potential Human Dietary Carcinogens."
The Minor Components
Minor components of food include the micronutrients (minerals and vitamins), the enzymes that all organisms produce and use as essential catalysts for their own life processes, and the DNA and RNAs that determine the nature of all constituents. In addition, plants and animals, and therefore foods derived from both, contain an almost unlimited variety of largely non-nutrient organic compounds often termed natural products or secondary metabolites. In this report, natural products or secondary metabolites are categorized as constitutive naturally occurring chemicals. Although chemically quite distinct, these chemicals are formed by modification of the same building blocks and biosynthetic pathways that produce carbohydrates, fats, and proteins. Examples of these chemicals are volatile oils, waxes, pigments, alkaloids, sterols, flavonoids, toxins, and hormones. Most plants contain one or a few minor constitutive naturally occurring chemicals of toxicologic or pharmacologic interest. This report intentionally focuses on the minority of these chemicals that are known or are suspected to cause, enhance, or inhibit cancer in humans. However, because of inherent low toxicity or low concentration, the vast majority of these naturally
occurring chemicals are known or can reasonably be presumed not to pose a toxic threat.
Within the higher plants—excluding animals, fungi, and microorganisms—the enormous complexity arises from variations on only a few major biosynthetic pathways, all of which use as their starting materials compounds derived from carbohydrates produced by photosynthesis. In addition to photosynthesis, the principal pathways are
- The shikimic acid pathway, which produces compounds containing benzene rings and related structures (including the three aromatic amino acids—phenylalanine, tyrosine, and tryptophan and a host of secondary metabolites derived from them—numerous quinones, benzoic acid derivatives, lignin, and many other benzenoid compounds)
- The acetate (polyketide) pathway, which adds two carbon atoms at a time and is responsible for fats, waxes, hydrocarbons, certain phenols, and for portions of the structures of many minor constituents
- The isoprenoid pathway, which combines 5-carbon isoprene units (derived from acetate) and is the source of terpenes (e.g., volatile flavor compounds such as menthol and camphor), plant pigments (e.g., carotenes, including Vitamin A), sterols, and rubber
- Protein synthesis, which combines amino acids to produce the primary gene products, proteins, including enzymes
Still further complexity is found in products such as alkaloids that arise from combinations of these pathways.
Complexity and Variability
The identity of the specific constituents in the minor and major components of food—the qualitative composition—is in large part
determined genetically. Environmental factors also affect qualitative composition and influence quantitative composition. Any particular crop is the result of the interplay of genetics and environment. Thus the genetic promise inherent in highly productive and disease-resistant new varieties of rice and wheat—the "green revolution"—cannot be realized without more intensive and better controlled cultural practices, including fertilization and irrigation. For plants, the relevant environmental factors include
- Latitude, which determines hours of daylight
- Climate (long-term temperature and rainfall trends)
- Weather (short-term temperature and rainfall)
- Altitude, which affects temperature independently of latitude, climate, and weather
- Agricultural practices, such as fertilization and irrigation
- Maturity at harvest
- Post-harvest processing
- Soil conditions (e.g., selenium content)
- Storage conditions
For foods of animal origin, the factors are diet, geographic origin, animal husbandry practices, season of harvest or slaughter, and environmental conditions prior to and at harvest or slaughter. All such factors have a major influence on the chemical composition of foods consumed in the diet.
Because of genetic and environmental factors, variation in the quantitative composition of individual foods is often great and can be dramatic. The usual food composition tables provide a useful overall picture, but the average values they contain give little indication of this variation.
Of the major components, water, carbohydrate, and protein vary the least and are typically, though not always, within 20% of the average value. Fat content varies somewhat more, from 50 to 200% of the average value, in foods of both vegetable and animal origin.
This greater variation in fat content reflects genetic and cultural practices, ones that are now changing because of recent appreciation of the role nutrition plays in chronic disease. Trace nutrients, constitutive naturally occurring chemicals, and natural contaminants are subject to much wider variation in foods derived from plants. Table 2-1 presents data representative of these variations in the U.S. diet.
Some foods, e.g., paprika, demonstrate inherently great variability in composition, reflecting variation in plant strain, climate, geographical source, and post-harvest processing. Some constituents, e.g., vitamin C, are highly variable in most of their dietary sources and for the same reasons. When these factors combine, the variability can be extreme: note, for example, the ascorbic acid content of paprika, for which the standard deviation (SD) is nearly equal to the mean. Standard deviations that are large compared with the mean imply that circumstances have combined and led to high production of that particular constituent. In general, variation is greater, i.e., the SD is larger relative to the mean, for
- Plant foods rather than animal foods (animal foods are usually subject to greater genetic control and less environmental influence with the exception of fat content)
- Microconstituents (those present at less than 1%), as opposed to macroconstitutents (those present at more than 1%)
- Plant foods that have a broad genetic base and are produced in many areas (e.g., paprika), as opposed to those that have a narrower genetic base and are produced in a few areas (e.g., California Valencia oranges).
Microconstituents such as selenium vary even more dramatically than those shown in Table 2-1, because of the great variation in the selenium content of soils.
Although the minor constituents account for only small percentages of total composition by weight, they are by far the largest number
Table 2-1 Partial Quantitative Composition for Individual Foods
|
|
Mean Standard Deviationa |
|||||||
|
|
g/100g |
mg/100g |
Retinol Equivalents/100g |
|||||
|
Food |
Carbohydrateb |
Fat |
Protein |
Sodium |
Ascorbic Acid |
Iron |
Thiamin |
Vitamin A |
|
MEAT, FISH |
||||||||
|
Beef, ground, extra lean, raw |
17/4.6 |
19/1.2 |
66/7.6 |
|
2/0.29 |
|
|
|
|
Lamb, shoulder, arm, separable lean, choice, raw |
5.2/0.35 |
20/1.1 |
69/19 |
|
1.7/0.52 |
0.12/0.028 |
|
|
|
Herring, Pacific, raw |
14/5.4 |
16/1.1 |
74/3.5 |
|
|
|
|
|
|
Pork, ham, separable lean, raw |
5.4/1.4 |
20/1 |
55/12 |
|
1/0.31 |
0.88/0.19 |
|
|
|
Tuna, Yellowfin, fresh, raw |
|
|
|
|
0.73/0.26 |
|
|
|
|
Veal, sirloin, separable lean, raw |
2.6/0.30 |
20/2.3 |
80/14 |
|
0.80/0.11 |
0.08/0.02 |
|
|
|
GRAIN |
||||||||
|
Wheat, soft, white |
75.36/-* |
2/0.18 |
11/1.7 |
|
|
5.4/3.6 |
0.41/0.056 |
|
|
|
Mean Standard Deviationa |
|||||||
|
|
g/100g |
mg/100g |
Retinol Equivalents/100g |
|||||
|
Food |
Carbohydrateb |
Fat |
Protein |
Sodium |
Ascorbic Acid |
Iron |
Thiamin |
Vitamin A |
|
FRUITS AND VEGETABLES |
||||||||
|
Beans, snap, raw |
7.14/- |
0.12/0.10 |
1.8/0.50 |
|
|
|
|
670/190 |
|
Broccoli, raw |
5.24/- |
0.35/0.16 |
3/0.51 |
27/10 |
93/8 |
|
|
|
|
Cabbage, raw |
|
|
|
|
32/18 |
|
|
|
|
Cauliflower, raw |
|
|
|
30/17 |
46/22 |
|
|
|
|
Carrots, raw |
10.14/- |
0.19/0.11 |
1/0.14 |
|
|
|
|
28,000/2,000 |
|
Celery, raw |
3.65/- |
0.14/0.083 |
0.75/0.17 |
87/39 |
|
|
|
|
|
Cherries, sour, red, raw |
12.18/- |
|
1/0.21 |
|
|
|
|
1,300/330 |
|
Mangoes, raw |
17/- |
0.27/0.27 |
0.51/0.22 |
|
|
|
0.058/0.030 |
3,900/2,300 |
|
Melon, cantaloupe, raw |
8.4/- |
0.28/0.077 |
0.88/0.35 |
|
|
|
|
3,200/670 |
of individual substances. For example, more than 200 constituents have so far been isolated and identified in orange oil—a simple oil (Maarse and Visscher 1989). Extensive information is available on single classes of such constituents, e.g., alkaloids (Pelletier 1983-1992, Mattocks 1986), terpenoids (Glasby 1982, Connolly and Hill 1991), and flavonoids (Harborne et al. 1975, Harborne 1988). The number of identified constituents of unprocessed food plants is at least 12,200; undoubtedly the actual number is far greater (Farnsworth 1994).
Geography and environment cause a variability in the concentration of these minor non-nutrient constituents, especially the essential oils and alkaloids, at least as great as that of the micronutrients. Salvia officinalis (sage) grows luxuriantly in many temperate areas of the world. Sage from the Dalmatian coast has an oil content of about 2.5% and is the industry standard for defining the characteristic flavor of the herb. However, sage grown in the mid-Atlantic states of the United States has an oil content of about 1.0%, and the flavor quality is variable and not as characteristic. We consume members of the red pepper family (Capsicum annuum or C. frutescens) for their color and flavor, as with paprika, or for color, flavor, and heat (piquancy), as with those used in Tabasco™ sauce. That heat is caused by a family of constituents called capsaicins. The capsaicin content of mild paprika ranges from 0.0002 to 0.0003 percent, that of jalapeño peppers is typically from 0.02 to 0.03 percent, and that of cayenne pepper from 0.2 to 0.3 percent—a thousandfold variation (Hoffman 1994). Ideal environmental conditions for maximum value are often unique to each species or variety and are found only in limited areas. Thus, the spice industry historically is international.
Coevolution, the long-term mechanisms by which environment exerts its effect on composition, is discussed in "The Functional Role of the Components of Food" section. However, short- and long-term events can result in the formation of secondary metabolites, even on plants of identical genetic background. For example,
in the wine industry, the highest concentrations and quality of flavor constituents are often coupled with unfavorable growing conditions and low yields. Even without understanding the interrelationship between environment and biosynthetic mechanisms, it seems obvious that the minor secondary metabolites created are the products of general stress on the plant. Such a conclusion differs from but is consistent with the presumption that these metabolites act as agents for combatting specific predators, pests, or pathogens. Biotechnology techniques have been developed to supplement and extend classical breeding methods, and we now have the opportunity to modify the chemical concentrations in plants (see the section at the end of this chapter).
We complicate the variability of food by processing, especially by cooking, the most widely used and probably the oldest form of processing. We enjoy many foods in the raw state, but most foods must be processed, primarily to delay or prevent spoilage and thus avoid the resultant waste and hazard. Cooking, canning, aseptic packaging, pasteurizing, refrigeration and freezing, dehydration, curing, smoking and salting, and the use of chemical preservatives, fermentation and pickling, and irradiation all preserve food. Moreover, many foods, such as soy beans, must be processed to render them digestible. Others, such as cassava (manioc, Manihot esculenta), a major starch source in the tropics, contain cyanogenic glycosides and must be rendered safe. Neurological damage from chronic cyanide poisoning due to inadequate processing can still be found in central Africa. Processing is also used to eliminate sometimes unwanted constituents, such as caffeine from coffee or tea, or to introduce, increase, or restore desirable constituents, such as iodine in salt, vitamins A and D in milk, and niacin, iron, thiamine, and riboflavin in enriched flour. We process still other foods to make them more acceptable or convenient. Thus, in addition to genetic modification, processing provides a broad set of options for modifying the concentration of constitutive or added naturally occurring chemicals.
Even the simplest processing of the simplest mixtures results in amazing complexity. A single sugar such as glucose, in water solution at neutral or near-neutral pH and at the temperatures used for cooking or sterilization, produces an array of monomeric and dimeric anhydrides, fragments, and second- and third-order reaction products (Davídek et al. 1990). All of these are derived naturally occurring chemicals. Complex raw materials yield far more complex final products. For example, because of its cultural, economic, and commercial importance, coffee aroma has been studied extensively, and more than 1,000 components have been identified (Clarke and Macrae 1985). More might yet be measured by more sophisticated analytical technology. It might not seem desirable to include the consequences of traditional and widely used processing in a definition of naturally occurring, but we must take them into account. We may modify processing in the interest of acceptability, improved nutrition, or safety, just as we have modified genetic composition and cultural practices, but processing is not dispensable. It is inevitable and we must deal with the consequences.
Compared with the huge number of naturally occurring chemicals in food, those of synthetic origin are much fewer. The total number of chemicals—natural and synthetic—added directly or indirectly to food is approximately 6,000, slightly more than half of which are indirect additives used as packaging components or constituents (Hall 1992). The majority of indirect additives are synthetic (see Chapter 3). Of approximately 3,000 intentionally added to food, the great majority are constitutive naturally occurring chemicals. A few are used in high volume. Examples are the major caloric sweeteners such as sucrose (ordinary sugar) and glucose, isolated soy protein used in infant formulas, flavors extracted or distilled from spices, and numerous gums and starches, such as corn or tapioca starch, isolated from one food and used in other foods because of their functional characteristics. Such separated and transferred substances are naturally occurring but can create different patterns of dietary exposure than would otherwise occur.
Because of cost, many constitutive naturally occurring substances are duplicated synthetically, then added to food. As is true of the naturally occurring chemicals, most of the synthetic additives are at microgram or nanogram levels or lower.
Toxicants and Nontoxicants
It was Paracelsus who first observed that ''Everything is poison. There is nothing without poison. Only the dose makes a thing not a poison" (Paracelsus 1564). But to describe everything as poison avoids the practical and vital distinctions we must make in dealing with naturally occurring substances, many of which we consume with far less than conventional margins of safety.
Today, toxicity is defined as the adverse effects produced by chemicals. The nature and extent of the toxic effect depends on the dose of the chemical. For practical reasons, such as those encountered with naturally occurring chemicals, this broad definition must often be reduced to an operational statement. The International Food Biotechnology Council uses a fairly restricted definition (i.e., operational guideline) for toxicant:
The toxic effects that the substance, i.e., the "toxicant," causes in humans, domestic animals or experimental animals either are irreversible (e.g., carcinogenicity, teratogenicity, certain neurotoxicities) or occur with narrow margins of safety, that is, at low multiples (approximately 25 or less) of ordinary exposures (IFBC 1990b).
Using this definition, the report goes on to note that, less than one-tenth of one percent of the total number of food constituents in our current food supply are toxicants. As analytical chemists identify the hundreds of thousands of constituents yet to be found at still lower concentrations, any toxicants to be discovered will have to be potent indeed to be capable of exerting adverse effects at such low concentrations. However, population growth will probably
force the use of new or underused food sources, most of them plants (NRC 1975). In many cases, and especially in developing countries, the populations will not have benefited from the experience that has produced our present western diets. Even in the United States, several people die each year from the toxic constituents in teas made of herbs gathered by amateurs. It is useful to keep in mind that throughout history, plant materials have served as a source of poisons and medicines, as well as food.
Naturally occurring toxicants occur in most plants, in many microorganisms, and also in marine plants and animals. They are, however, essentially absent from the major cereal grains and from farm animals. Clearly, cereal grains have been selectively bred for at least 10,000 years—since the Neolithic Age—and the significant toxicants, e.g., phytic acid, have been bred out or reduced by processing. Naturally occurring contaminants such as mycotoxins are, however, quite common. Domestic animals, under the care of their owners, act as biological screens. Their illnesses have often alerted us to the presence of naturally occurring toxicants. Herd managers are careful to exclude known toxicants from feed and forage, although rare exceptions occur. In some of these exceptions, the naturally occurring toxicant does not affect the domestic animal but does affect the consumer of the animal product. Examples of these pass-through toxicants are the several honey toxins (IFBC 1990c), cicutoxin in water hemlock, and toxol in snakeroot which, when it appears in cow's milk, is suspected of causing the "milk sickness" from which Abraham Lincoln's mother died (Crosby 1969). Pass-through toxicants can be a major concern in seafood, particularly in shellfish. The contamination caused by the "red tide" is the most familiar example, but there are many others, several of which have been identified only in the last few years (Dickey 1989, Hall 1989, Iverson 1989). Much human experience with these toxicants suggests that their potential effects are most likely to become real when the specific foods in which they occur form too large a part of the diet, as in times of food shortages. The varied and balanced
diet needed for good nutrition also provides, through dilution, significant protection from natural toxicants, as well as wider exposure to a range of potential anticarcinogens.
Of the human dietary constituents in our western food supply that can reasonably be called naturally occurring toxicants, only some are now regarded as carcinogens. In this report, the committee has assembled a representative list of chemicals for which there is at least some evidence of carcinogenicity in animals. The list comprises the naturally occurring chemicals, closely related groups of chemicals (such as the aflatoxins), and crude extracts or distillates, such as that from calamus. The committee then selected five chemicals, representative of various categories, for discussion in more detail. Very few naturally occurring chemicals have been tested for their carcinogenic potential, and still fewer have been tested by the standard methods used to determine xenobiotics (see the section on "Dietary Plants and Cancer"). Additional data, particularly if obtained in bioassays using the maximum tolerated dose (MTD), could well increase this number substantially. However, use of the MTD has inherent problems and limitations; these are discussed in chapters 4 and 5.
In addition to carcinogens that occur in the diet, some can be formed endogenously in humans from naturally occurring chemicals that are not toxic at levels found in the diet. N-nitroso compounds, including nitrosamines (discussed more completely in this chapter under N-nitrosodimethylamine and in Chapter 3), illustrate this class of potential human carcinogens. N-nitroso compounds are suspected of being a causal factor in gastric and other cancers (Mirvish 1983). They can be formed endogenously in the stomach by nitrosation of secondary and tertiary amines and other nitrogen compounds that occur naturally in the diet. This reaction also requires nitrite and an acidic environment. Nitrite exposure results from the oxidation of NO produced as the result of inflammatory responses or from reduction of dietary nitrate, the primary source of which is green vegetables (usually about 90%) and drinking water.
The latter is a significant source only if the drinking water nitrate exceeds the EPA limit of 50 mg/L (NRC 1981). Nitrate in vegetables is accompanied by varying amounts of ascorbic acid and polyphenolics, which inhibit the nitrosation reaction. Nitrosamines are also found to a small extent occurring naturally in foods.
It is important to note that the putative naturally occurring carcinogens in our food are not a separate, easily definable class of constituents. They depend on the definitional criteria applied, on what is selected for testing, or on accidental discovery. They are merely a small part of the complexity of food. A critical purpose of this report is to provide a perspective on their importance, in terms of both the range and the size of the threats they present compared with similar threats from the synthetic constituents in our food supply.
The functional Role of the Components of Food
It is useful to review briefly the functional role of naturally occurring constituents in the organisms from which we derive our food. The utility of the major components of food is well known. Water is the solvent and vehicle in which all the biochemical reactions of living organisms take place. Carbohydrates are energy stores and, particularly as cellulose, structural elements in plant foods. Proteins and their simpler relatives, peptides, appear as enzymes, hormones, and structural components, such as muscle. Fats, oils, and other constituents related to them serve primarily as energy stores but also have important functional utility as cell membrane components. Minerals serve a structural purpose, as in bones and teeth, but often play key metabolic roles as well. Examples are the iron complexed in hemoglobin and in the cytochromes, the magnesium in chlorophyll, the cobalt in vitamin B12, and the essential minerals in the metalloproteins.
The role of the numerous, minor plant constituents in the organisms
in which they are found is seldom obvious. However, their roles and the enormous diversity of such natural products can be accounted for by the theory of biochemical coevolution between plants and animals (Fraenkel 1959, Ehrlich and Raven 1964, Feeny 1975, Visser and Minks 1982, Harborne 1993). Simply described, this theory states that a natural product (or group of products), presumably arising from some random mutation, may provide an advantage to the plant by deterring feeding by phytophagous insects, discouraging competitive plants, or by encouraging pollination (and reproduction). If that advantage is significant, further mutations that enhance the plant may confer further advantage and may survive. If the plant is thereby allowed to occupy a new ecological niche, it will flourish there until some mutant form of insect occurs that can feed on the previously protected plant. In this way, plants tend to increase the diversity of plant-eating animals and vice versa.
Consistent with this concept of coevolution, the study of which is often called chemical ecology, are constituents that have a protective role for the plants or animals in which they occur. This is particularly true of the constituents in the component classes called essential (or volatile) oils, the alkaloids, the nonvolatile components of extracts, gums and oleoresins, and the many animal venoms, toxicants, and repellents. Many of these act as pest or predator repellents, pesticides, fungicides, and pathogen inhibitors (ApSimon 1989, Ames et al. 1990, Harborne 1993, Meinwald and Eisner 1995). Some, found particularly in animals, are pheromones—substances released to communicate alarm or sexual availability or to indicate a path to be traveled. Those used as sexual attractants often are unique to the species and are a means of maintaining species isolation. Some constituents appear to be general attractants; others act as competition inhibitors and feeding deterrents. The flowers of many plants that require birds or insects as pollinators use a sweet and often aromatic nectar as an attractant. We find that same nectar attractive in perfumes, as space odorants in
and around our homes, and as a food in the much more concentrated form of honey.
Flower nectars are one of the few examples in which the constituent is used by both humans and the source organisms for a closely similar function. More often, as in spices, plants use a constituent in one role, and humans harvest the same constituent for use in another. Repellents present a different situation. We insist that repellents be pleasant, or no worse than innocuous, to us but noxious to the species to be repelled. Such insistence eliminates many obviously effective repellents, skunk essence, for example.
Similarly, and more significantly, the plant antioxidants such as flavone derivatives, isoflavones, catechins, coumarins, phenylpropanoids, polyfunctional organic acids, phosphatides, tocopherols, ascorbic acid, and the carotenes have clear roles in plants (including dietary plants). They act as reducing compounds, as free radical chain interrupters, as quenchers or inhibitors of the formation of singlet oxygen, and as inactivators of pro-oxidant metals (Simic and Karel 1980, Hudson 1990). Although the plant and animal milieux are quite different, the value to us of antioxidants from each is similar.
Lupines are known in the United States as both wild and cultivated flowering plants. In South America, several species of lupine are used both for domestic animal forage and for human food. Use as food requires careful processing to reduce the levels of naturally occurring quinolizidines which, without such processing, have caused illness and death in both humans and domestic animals. Efforts to breed lupines with lower alkaloid contents have met with success, but these "improved" lupines are susceptible to higher levels of mycotoxin contamination.
"Potato poisoning" was a common occurrence in the 19th and early 20th centuries when potatoes often formed a large part of the diet. Such poisoning was caused by the variable presence of a glycoside, solanine. Even recently, a cultivar of Idaho potatoes had to be taken off the market when it was found to contain toxic levels of the neurotoxin solanine (IFBC 1990b).
When a natural product first appears in a species, it is presumably the result of a random mutational event; however, its perpetuation may at first be for reasons more subtle than those discussed above. Harborne (1993) points out that plants of the legume genus Astragalus have been able to adapt to high selenium soils because they can sequester selenium analogs in nonprotein amino acids, which are structurally different and therefore not incorporated into protein synthesis. A large number of plants, e.g., the genus Prunus, detoxify cyanide by sequestering it in the form of glycosides. Thus, while plants cannot excrete, in the sense of animal physiology, they can sometimes set aside useless or dangerous substances. In such cases, however, the detoxification products also now serve the protective role discussed above. Hölldobler (1995) cites Morgan (1984) in reporting "that the species-specific trail pheromones from the poison glands of myrmicine ants are generally the metabolic byproducts of venom synthesis." Thus, natural products that may originally have been waste products, byproducts, or detoxification products become simply new factors in the process of coevolution. Any or all of these functions may be consistent with the apparent positive correlation of the concentrations of the minor constituents with environmental stress.
Some naturally occurring plant compounds are virtually ubiquitous. Caffeic acid is a metabolic precursor of lignin, a structural polymer found in all land plants. Caffeic acid is also a component of chlorogenic acid, a phenol found widely distributed in fruits and vegetables. D-limonene is particularly characteristic of the orange and other members of the citrus family, but it is also found in more than 75 unrelated species, including allspice, tea, coffee, hops, passion fruit, peppermint, saffron, and vanilla. Alpha- and beta-pinene are major constituents of the oils from the genus Pinus but are also widespread throughout the plant kingdom. The widely distributed anthocyanins, one of several classes of flavonoid pigments, are responsible for the colors of flower petals, ripening fruits, and autumn leaves. Other closely related groups of constituents are found almost entirely in one family, as the glucosinolates
in the genus Brassica (the cabbage family). Still other constituents are uniquely identified with a single species, e.g., cicutoxin in water hemlock or tetrodotoxin in puffer fish. Furthermore, the range of concentrations is as broad as the distribution is diverse. Some instances demonstrate survival value, but others suggest imprecise and slowly evolving systems.
Many plants and nearly all animals, including unicellular ones, have what appears to be a "chemical sense," i.e., they move or grow preferentially in the direction of increasing concentrations of attractive or nutrient substances; they also move away from or do not grow in the direction of or thrive in the presence of increasing concentrations of adverse substances. Thus there is a clear, if general, explanation for the effectiveness and survival value of many constituents. This phenomenon is most apparent in the case of nutrients or, at the opposite extreme, acutely noxious or toxic constituents. It seems reasonable to accept a role for genotoxic constituents if they have the capability of lowering the survival value of subsequent generations of pests or predators. What is far more difficult to imagine, however, is the survival value of a constituent that is an animal or human carcinogen. The typically long induction periods for chemical carcinogenesis could rarely if ever affect the aggressiveness or reproductive effectiveness of a pest or predator. If correct, this then leads to the conclusion that the carcinogenicity—of such concern to us—is merely an incidental aspect of some constituents, functionally unrelated to whatever role they may have in the physiology of the source organism.
This perspective seems further strengthened by the often substantial differences in susceptibility to toxicants, including carcinogens, among different animal species. This report considers elsewhere the difficulties in interspecies comparisons and the problems of comparative risk assessment for carcinogens from different sources and of different potencies. We simply conclude that, as far as we know now, there is a clear survival value for the plant or animal source in many of the naturally occurring toxicants found in food,
but no clear rationale for distinctively carcinogenic constituents as promoters of evolutionary fitness. Carcinogenicity appears to be an incidental aspect—one of many forms of toxicity encountered in the naturally occurring constituents of food.
Certainly, non-nutrient components of foods have the potential to be toxic as well as beneficial, as is discussed below for selected nutrients. It is likely that as purified sources of these chemicals become available, individuals may consume excessive amounts and some people will reach toxic doses. the studies demonstrating toxicity of chemicals that have potential beneficial properties, including nutrients, were conducted using higher doses than those to which people consuming normal diets would be exposed.
Dietary Plants and Cancer
The ability of dietary plant extracts and constituents to induce malignant neoplasms in rodents has not been extensively studied, for several reasons. First, if humans have ingested these plants over the millennia without apparent toxicity, there has been no clear-cut rationale for undertaking such studies. Second, the cost of a two-species, two-sex carcinogenicity study, with the subchronic, metabolic, and analytical work typically needed for proper study design and interpretation, can exceed $2 million per chemical tested. This cost is prohibitive without a compelling rationale for such an effort. Third, traditional food plants are in the public domain. They have no owner or sponsor with a proprietary interest that could justify assuming these high costs.
Moreover, a positive result obtained from testing a plant or crude extract would leave unresolved the question of which constituent, or combination of constituents, produced this result. As discussed earlier, plant composition is complex and highly variable. Useful specifications are sometimes difficult and often impossible, and without them, one cannot be sure what one has tested, or that one
can reproduce the results. Lastly, the great majority of these constitutive naturally occurring chemicals are present at microgram or nanogram levels or lower. Unless there are data suggesting that further study is indicated, there is a high probability that nearly all these chemicals are present in the diet at levels that pose no toxicological significance (see Chapter 5).
As a consequence of all this, except for chili peppers (Capsicum frutescens, C. annuum), calamus (Acorus calamus), black pepper (Piper nigrum), and bracken fern (Pteridium aquilinum), and some edible mushrooms (Agaricus species), very few edible plants or their crude extracts have been shown to be carcinogenic in laboratory animals. Certain plants used as herbal remedies contain carcinogenic pyrrolizidine alkaloids (e.g., lasiocarpine), but are not considered as foods and thus are not included in this discussion. In contrast, animal tests provide some evidence of carcinogenicity for a large number of individual plant constituents (see Appendix A). In many cases, these results were obtained only at or near the MTD. In plants or their crude abstracts, carcinogenic constituents typically are highly diluted by the noncarcinogenic components of the plants and their crude extracts, thus making achievement of an MTD impossible. Because of physical and nutritional limitations, one cannot simply feed more of the plant or crude extract to compensate for the dilution, and also because other constituents, although noncarcinogenic, will often be sufficiently toxic to make the MTD unreachable. In addition, research in this area has been hampered by limited availability of purified plant components and by our limited knowledge about potential interactions between compounds within plants and in humans. Furthermore, studies on toxicity have looked at the effects of plant components in animal systems using the highest doses tolerated, while studies looking for protective properties have generally used somewhat lower doses and have often studied the impact of the agent on the toxicity of a potent chemical or biological agent. These crude approaches certainly result in data that are difficult to use for assessing the risk escalation
or risk reduction resulting from human exposure to these chemicals.
Nevertheless, the literature shows that although the crude extracts of more than 50 species of flowering plants have been tested orally for carcinogenic activity in rodents, only 27 of them were dietary plants. Of these, positive results were seen in only the four noted above. Based on the criteria set forth in the next section, and on the apparent quality of the data, only three of the dietary plants just mentioned might be considered to be carcinogenic. On the other hand, dietary plants which have been reported to inhibit carcinogenesis number approximately 28, and secondary metabolites from dietary plants number approximately 55 out of 65 tested (Farnsworth 1994).
According to current knowledge, the limited number of biosynthetic pathways in higher plants produces only a small number of constitutive naturally occurring chemicals found to be carcinogenic in animals. None of these is a potent carcinogen, comparable to the aflatoxins or the nitrosamines. However, as we have noted, food also contains naturally occurring substances that have been acquired and derived. Among these are some of the most potent animal carcinogens that have been identified.
Naturally Occurring Carcinogens Formed During Processing or Contamination of Food
Mycotoxins
The category of acquired naturally occurring substances includes those known as mycotoxins, resulting from fungal growth on food either in the field or during harvest and storage. Dietary contamination by one or more mycotoxin is common in most parts of the
world, and particularly in hot, humid climates, such as those of Southeast Asia and sub-Saharan Africa (Wogan 1992). Exposure to mycotoxins is a chronic concern worldwide, not only because domestic products from these areas may be contaminated, but also because countries in colder climates import foods from areas where mycotoxin contamination of dietary staples is more frequent and severe (IARC 1993). The presence of one mycotoxin in food generally implies cocontamination by others, because a single fungus can generate several mycotoxins, and also because a food can be contaminated simultaneously by several mycotoxin-producing fungi.
Of several toxigenic species of fungi that can contaminate diet and dietary staples, contamination by two species of Aspergillus, namely A. flavus, and A. parasiticus, both known to produce hepatocarcinogenic aflatoxins, appears ubiquitous. A. flavus produces aflatoxins B1 and B2, whereas A. parasiticus produces aflatoxins B1, B2, G1 and G2 (Pitt et al. 1993). While all four aflatoxins are toxic and believed to be carcinogenic in animals, B1 is the most prevalent and the most potent. Grains, peanuts, tree nuts, and cottonseed meal are among the foods on which aflatoxin-producing fungi commonly grow. Meat, eggs, milk, and other edible products from animals that consume feed contaminated by aflatoxins are additional sources of potential exposure. The relative amounts of aflatoxin B1 on crops such as corn or groundnuts (peanuts), or other grains and cereal products, depends not only on the presence of the toxigenic fungi A. flavus and A. parasiticus , but also on pre and post-harvest conditions (IARC 1993). Levels of aflatoxin in crops can also vary geographically and over time, with the southeastern US frequently referred to as an area where high levels can occur in corn. If a particular corn crop is stressed, for example, by drought or insect attack, it is susceptible to A. flavus growth and hence aflatoxin contamination (U.S. Food and Drug Administration [FDA], Compliance Program Guidance Manual 7106.10). Available data from various parts of the world suggest that the median levels of aflatoxins in corn range from <0.1 to 80 ng/kg, and
that in groundnuts (peanuts) the median levels are always below 26 ng/kg (IARC 1993). The dietary exposure resulting from consumption of aflatoxin-contaminated diets, including milk and milk products from animals that have eaten contaminated feed, ranges widely from about 2.7 (U.S.A.) to 2,027 (Southern Guangxi, China) ng/kg bw/day (IARC 1993).
In view of the ubiquity of Aspergillus in the environment and the possibility that food staples may be contaminated at various stages of production and processing, it is unlikely we can ever completely eliminate exposure to aflatoxin. Regulations now in effect or proposed for many countries generally impose a maximum limit of 50 µg/kg food of aflatoxin B1 or the total of all aflatoxins, and many countries impose far lower limits, e.g., 20 ng/kg in the United States. The carcinogenic properties of the aflatoxins have been extensively investigated, and much of these data are reviewed in somewhat greater detail later in this section.
Epidemiologic studies have provided convincing evidence that dietary consumption of aflatoxins has an etiologic role in hepatocarcinogenesis, and the studies indicate a synergy between chronic viral B (also C) hepatitis and aflatoxin exposure (Ross et al. 1992, Qian et al. 1994). In addition, a synergistic interaction between chronic alcohol consumption and aflatoxin exposure appears to play a role in human hepatocarcinogenesis. This causative role of aflatoxins in human hepatocarcinogenesis has recently been further supported by evidence from molecular epidemiology (see section on aflatoxins). There now can be no doubt that elevated exposure to aflatoxins, and especially to B1, is a major contributor to human liver cancer. Nonetheless, it should be noted that aflatoxin appears to account for a fraction of liver cancer in the United States because of low aflatoxin concentrations in most U.S. foods and low prevalence of hepatitis B virus carrier status (HBsAg+).
Of the other most widely distributed toxigenic fungi, Fusarium moniliforme is a ubiquitous contaminant in corn, and it produces toxins such as fumonisins B1 and B2 and fusarin C. Exposure to
Fusarium toxins appears to play a role in the pathogenesis of esophageal cancer in humans. There are limited data on the levels of these Fusarium toxins in food; thus, it is not possible to estimate reliably the levels of exposure (IARC 1993).
Ochratoxin A, produced predominantly by Aspergillus ochraceus and Penicillium verrucosum, occurs worldwide in many commodities from grains to coffee beans, and it is implicated in urinary tumorigenesis in humans and rodents. Furthermore, barley, wheat, and pork products all appear to be human dietary sources of ochratoxin A (IARC 1993).
Although less well-studied, T2 toxin from Fusarium and other species (Rodericks and Pohland 1981, Watson 1985, Ueno 1987) and the toxins found in Penicillium islandicum Sopp (Ueno 1987) have been reported to be carcinogenic. Beyond these, a large number of toxicants from many species of lower fungi have been reported to cause liver damage in test animals or to be mutagenic in microbiological assays (IFBC 1990). At least some of these would reasonably be expected to be carcinogenic in animals if adequately tested.
Unfortunately, mycotoxins are ubiquitous. They can and must be minimized, but they cannot be eliminated entirely from our diet.
Pyrolytic Products
As indicated earlier, cooking is the oldest and most widely used method of food processing. Cooking alters the chemical structure of the food to be consumed by pyrolysis, rendering it safe from microbial growth. The chemistry of pyrolysis is extremely complex. The pyrolysis products of graphite, 60-carbon aromatic bucky balls, received much attention in the early 1990's; however, the chemistry of this process is simple when compared to the real world process of cooking food (Kroto et al. 1985). Nonetheless, the processes are similar and the chemistry not very well understood. When foods
(or more properly food juices) are subjected to high temperatures, the amino acids, sugars, and other constituents are degraded and recombined to yield a bewildering array of compounds responsible for the aromas and flavors of cooked foods. Many of these compounds (e.g., the pyrazines) constitute the desirable flavor components that we associate with cooking, yet many others, particularly the more complex polycyclic heterocyclic amines, have been shown to be carcinogenic.
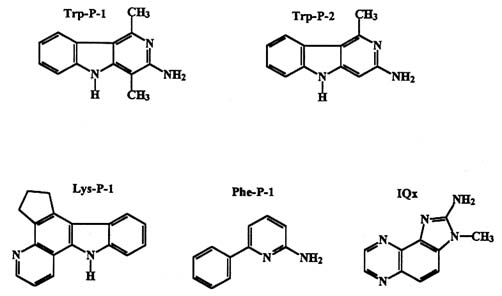
In 1977, Sugimura and coworkers demonstrated that the charred part of a grilled sardine was highly mutagenic in Salmonella typhimurium . After isolation and characterization, the agents responsible were determined to be a variety of polycyclic heterocyclic amines (PHAs). When further investigated, it was shown that any amino acid, when pyrolyzed, would produce its own characteristic set of pyrolysis products. Analysis of these products indicated that as many as 25 mutagenic PHAs may be isolated (Nagao and Sugimura 1993, Sugimura et al. 1994). For instance, tryptophan yields Trp-P-1 and Trp-P-2 (Tryptophan Pyrolysis 1 and 2, respectively) shown in Figure 2-1.
Pyrolysis of many other amino acids yields structurally similar compounds (see Lys-P-1, Phe-P-1, IQx, in Figure 2-1). It is important to keep in mind that these compounds are isolated from single amino acid reactions, and that the mixture of amino acids and other metabolites produces still other, more complex compounds, for instance IQ, MeIQ, and MeIQx. These compounds are among the most potent mutagens yet discovered.
The mutagenicity of these compounds correlates well with their carcinogenicity. When Trp-P-1 is fed to mice at the dosage of 15 mg/kg/day (0.53 mg/day per mouse) it induces hepatocellular tumors in 42% of the animals. In well-charred beef, Trp-P-1 may be present at a concentration of 106 ng/gm beef. Thus, assuming the average rate of consumption of beef to be 188 gm/day, the average human exposure from this source is 285 ng/kg/day, or 19.95 µg for a 70 kg person (Prival 1985). Similarly, when Trp-P-2 was administered
for 112 weeks in the diet at 100 ppm to male and female rats, significant increases in the incidences of neoplastic lesions were found in the liver, urinary bladder, and mammary glands in males and in the mammary gland, hematopoietic system, and clitoral gland of females (Takahashi et al. 1993). While these doses are dependent on cooking conditions and their relevance is a matter of dispute, it is clear that there are several orders of magnitude between the human and rodent experimental doses.
The discovery, identification, analysis, and testing of the PHAs provide a useful example of the progress that can be made, although with much cost in time and effort, in dealing with a complex mixture. The problems involved in dealing with mixtures receive further attention in Chapters 4 and 6.
Cooked sugar was also found to be mutagenic and to promote the growth of colonic microadenomas in rats and mice (Corpet et al. 1990).
Current State of Knowledge of Human Dietary Carcinogens
Identifying Potential Human Dietary Carcinogens
Appendix B presents a list of substances evaluated by the International Agency for Research on Cancer (IARC) and U.S. National Toxicology Program (NTP) on the basis of epidemiologic and animal studies and found to pose some specified degree of carcinogenic risk to humans. Those substances are the primary focus of this report. In addition, Appendix A contains a list of other substances that have been tested for carcinogenicity in animals and found to have some positive evidence of it. Most of these are not included in Appendix B. The list in Appendix A provides a more comprehensive summary of the limited carcinogenicity testing done
so far on naturally occurring chemicals in the food supply and indicates the wide variability in the apparent quality and solidity of the results.
It is not feasible to discuss in-depth each of the substances in either appendix. Instead, the discussion that follows provides some reasonably detailed information on five representative naturally occurring chemicals.
Constitutive Naturally Occurring Carcinogens
Caffeic Acid
Caffeic acid is a constitutive naturally occurring chemical found in higher plants. Of the five chemicals listed in Table 2-2, it is the most widely distributed throughout the plant kingdom, occurring mainly as the three monocaffeoylquinic acid esters, chlorogenic, cryptochlorogenic, and neochlorogenic acid. The chlorogenic acids are apparently hydrolyzed in the stomach to caffeic and quinic acid (Booth et al. 1957, Czok et al. 1974). Caffeic acid also occurs esterified to other hydroxyacids such as malic and tartaric acid and to glucose as a glucoside. It occurs infrequently as a free acid (Herrmann 1989).
The concentration of conjugates of caffeic acid in various fruits and vegetables, expressed in terms of mg/kg fresh weight, has been summarized by IARC (1993). Concentration data summarized by IARC and by Stich (1991) are used in Chapter 5 for making estimates of caffeic acid exposure. Large concentrations of caffeic acid conjugates can be found in apples and lettuce (Herrmann 1989).
There is sufficient evidence in experimental animals for the carcinogenicity of caffeic acid. Administered in the diet, it induced forestomach squamous cell carcinomas in male mice and in male and female rats, and renal tubular cell hyperplasia and adenomas in mice.
Table 2-2 Examples of Carcinogens Naturally Occurring in Normal Human Dietsa
|
|
Degree of Evidence for Carcinogenicityb |
|
|
|
|
|
|
Substance |
Human |
Animal |
Overall Evaluation of Carcinogenicityc |
Nature of Supporting Evidencee |
Extent of Natural Occurrence in Foods |
Referencesf |
|
Constitutive Naturally Occurring Carcinogens |
|
|
|
|
||
|
Caffeic Acid |
ND |
S |
2B [56, 1993] |
Forestomach tumors in male mice, male and female rats; kidney tumors in female mice, male rats. Clastogenic, mutagenic |
Occurs widely in plants as esters of hydroxyacids, such as quinic (e.g., chlorogenic and neochlorogenic acid), tartaric, and malic acid; as glucose ester; and as glucosides; released by hydrolysis |
68, 28, 21, 58, 63, 75, 26, 34, 25, 60, 36, 40 |
|
Urethane |
ND |
S |
2B [7, 1974] |
Lung tumors, lymphomas, hepatomas, and melanomas in rats, mice, and hamsters |
All fermented and yeast-leavened foods; wines, yogurt, soy sauce 1.0-5.0 ppb; sake, liquors 100-500 ppb; ale, beer, bread ≈ 1.0 ppb |
7, 65, 31, 13, 1, 66, 22, 24, 51, 42 |
|
|
Degree of Evidence for Carcinogenicityb |
|
|
|
|
|
|
Substance |
Human |
Animal |
Overall Evaluation of Carcinogenicityc |
Nature of Supporting Evidencee |
Extent of Natural Occurrence in Foods |
Referencesf |
|
Acquired and Pass-Through Naturally Occurring Carcinogen |
|
|
|
|||
|
Aflatoxin B1 |
S |
S |
1 [56, 1993] |
Etiologic role in hepatocar-cinogenesis derived from epidemiologic studies involving dietary consumption. Tumors of liver, colon, kidneys in several animal species. Hepatocellular and/or cholangiocellular liver tumors, including carcinomas in rats, hamsters, monkeys. Renal cell tumors, colon tumors in rats. Liver angiosarcomas of gall bladder and pancreas in monkeys. Altered hepatocytes in rats and hamsters. Clastogenic, mutagenic |
Most prevalent fungal contaminant found in food. Derived from fusarium moniliforne—ubiquitous contaminant in corn. Also found on grains, peanuts, and more rarely, milk |
71, 8, 15, 70, 69, 30, 41, 56, 74, 8, 39, 6, 5, 47, 17, 56, 72, 12, 46, 57, 50, 25, 48, 2, 18, 41 |
|
Derived Naturally Occurring Carcinogens |
|
|
|
|
||
|
PhiP (2-Amino-1-methyl-6-phenyl-imidazo [4,5-b] Pyridine) |
I |
S |
2B [56, 1993] |
Lymphomas in male and female mice; intestinal adenocarcinomas in male rats, mammary adenocarcinomas in female rats. Clastogenic, mutagenic |
Most abundant heterocyclic amine in cooked food, especially fried ground beef, broiled chicken, fried fish. ≈ 20-70 ng/g |
3, 27, 19, 29, 44, 25 |
|
N-Nitrosodimethylamine |
ND |
S |
2A [17, 1978]d |
Liver hamangiosarcomas, hepatocellular carcinomas, kidney, lung tumors in mice; kidney and bile duct tumors in rats, hepatocellular carcinoma, bile duct tumors in hamsters, rabbits, and guinea pigs. Clastogenic, mutagenic |
Cheese, soybean oil, canned fruit, various meat products, bacon, various cured meats, frankfurters, cooked ham, fish, spices for meat curing, apple brandy, other alcoholic beverages, and beer. Concentrations in foodstuff 0-85 ng/g |
38, 52, 74, 4, 59, 64, 65, 62, 11, 16, 14, 35, 49, 10, 32, 45, 53, 33, 76, 20, 61, 54, 25, 43 |
|
a This is not an exhaustive list. Represented chemicals were chosen because they are classified by IARC as carcinogens, represent a range of carcinogenic potencies, and are present in diet for a variety of reasons. b ND, no adequate data; I, inadequate evidence; S, sufficient evidence. For definitions of terms and overall evaluations, see IARC 1993, pp.; 28-29. c Overall evaluation based only on evidence of carcinogenicity in monograph [volume, year]. d Other relevant data, as given in monograph [volume, year], influenced the making of the overall evaluation. e Positive results reported in IARC; routes of administration are oral. f Source of data on occurrence as well as carcinogenicity. |
||||||
|
References |
|||||
|
1 |
Adenis et al. 1968 |
2 |
Aguilar et al. 1994 |
3 |
Alink et al. 1988 |
|
4 |
Argus and Hoch-Ligeti 1961, 1968 |
5 |
Autrup et al. 1987 |
6 |
Bannasch et al. 1985 |
|
7 |
Berenblum & Haran-Ghera 1957 |
8 |
Bulatao-Jayme et al. 1982 |
9 |
Butler et al. 1969 |
|
10 |
Clapp and Toya 1970 |
11 |
Clapp et al. 1968, 1971 |
12 |
Cusumano 1991 |
|
13 |
Della Porta et al. 1963 |
14 |
Den Engelse et al. 1969/1970 |
15 |
Epstein et al. 1969 |
|
16 |
Geil et al. 1968 |
17 |
Gil et al. 1988 |
18 |
Greenblatt et al. 1994 |
|
19 |
Gross 1990 |
20 |
Hadjiolov and Markow 1973 |
21 |
Hirose et al. 1988, 1991, 1992 |
|
22 |
IARC 1974 |
23 |
IARC 1978 |
24 |
IARC 1987 |
|
25 |
IARC 1993 |
26 |
Inoue et al. 1992 |
27 |
Isumi et al. 1989 |
|
28 |
Ito and Hirose 1987 |
29 |
Ito et al. 1991 |
30 |
Kalengayi et al. 1975 |
|
31 |
Klein 1962, 1966 |
32 |
Kowalewski and Todd 1971 |
33 |
Kuwahara et al. 1972 |
|
34 |
Laranjinha et al. 1992, 1994 |
35 |
Le Page and Christie 1969a,b |
36 |
Li and Trush 1994 |
|
37 |
Li et al. 1994 |
38 |
Magee and Barnes 1956, 1959, 1962 |
39 |
Moore et al. 1982 |
|
40 |
Nakayama 1994 |
41 |
Nishizumi et al. 1977 |
42 |
Nomura 1982 |
|
43 |
NTP 1994 |
44 |
Ochiai et al. 1991 |
45 |
Otsuka and Kuwahara 1971 |
|
46 |
Parkin et al. 1991 |
47 |
Peers et al. 1987 |
48 |
Qian et al. 1993 |
|
49 |
Riopelle and Jasmin 1969 |
50 |
Ross et al. 1992 |
51 |
Schmähl 1977 |
|
52 |
Schmähl and Preussmann 1959 |
53 |
Shabad and Savluchinskaya 1971 |
54 |
Shinohara et al. 1976 |
|
55 |
Sieber et al. 1979 |
56 |
Soffritti & McConnell 1988 |
57 |
Srivatanakul et al. 1991 |
|
58 |
Stich 1991, 1992 |
59 |
Takayama and Oota 1963, 1965 |
60 |
Tanaka et al. 1993 |
|
61 |
Taylor et al. 1974 |
62 |
Terracini et al. 1966, 1967, 1969 |
63 |
Toda et al. 1991 |
|
64 |
Tomatis et al. 1964 |
65 |
Toth et al. 1961 |
66 |
Toth and Boreisha 1969 |
|
67 |
Toth et al. 1964 |
68 |
Wattenberg et al. 1980 |
69 |
Wogan et al. 1971 |
|
70 |
Wogan 1969, 1974, 1992 |
71 |
Wogan and Newberne 1967 |
72 |
Yeh et al. 1989 |
|
73 |
Zak et al. 1960 |
74 |
Zawirska and Bednarz 1981 |
75 |
Zhou and Zheng 1991 |
|
76 |
Zwicker et al. 1972 |
|
|
|
|
The relevance to humans of the positive findings in animal studies is uncertain for several reasons: no data are available on the carcinogenicity of caffeic acid in humans; the dose of caffeic acid tested in experimental animals was high; humans do not have a forestomach; and the renal lesions reported in mice and rats were related to toxic lesions. In addition, studying the actions of plant components in isolation can lead to incorrect assumptions about their modes of action and ultimate effects, which represents a specific instance of the general uncertainties about substance interactions. Caffeic acid, which exhibits both carcinogenic and anticarcinogenic activity, appears to be a case in point. In vitro studies indicate that caffeic acid may act either as a pro- or anti oxidant, depending on the experimental conditions. In the presence of free transition metals (e.g., manganese, copper, iron), reactive oxygen species capable of damaging DNA may be formed (Inoue et al. 1992, Li and Trush 1994); however, in their absence, caffeic acid blocks the formation of reactive oxygen species, lipid peroxides, and nitrosamines (Toda et al. 1991; Zhou and Zheng 1991; Stich 1992; Laranjinha et al. 1992, 1994; Li et al. 1994; Nakayama 1994). In vivo studies in which caffeic acid was administered orally in combination with known carcinogens have also yielded seemingly contradictory results with respect to the carcinogenic action of caffeic acid in epithelial tissues. Caffeic acid increased forestomach tumors in rats pretreated with DMBA or MNNG (Hirose et al. 1988, 1991, 1992). However, it inhibited squamous epithelial carcinomas of the rat tongue (Tanaka et al. 1993) and mouse forestomach tumors when administered with benzo[a]pyrene (Wattenberg et al. 1980). The dose range at which caffeic acid has been observed to be protective (500-10,000 ppm) overlaps with the range at which enhancing effects have been seen (5,000-20,000 ppm). A cursory look at the foods in which caffeic acid is present reveals many that are high in fiber, vitamins A, E, C, beta carotene, and numerous other protective compounds that might significantly affect the fate of caffeic acid in the body.
On the basis of studies in animals, IARC (1993) concludes that caffeic acid is possibly carcinogenic to humans and has classified it as a Group 2B carcinogen.
Urethane (Ethyl Carbamate)
Urethane is a derived naturally occurring chemical found in foods produced or modified by fermentation, including alcoholic beverages, bread, soy sauce, yogurt, and olives (IARC 1974). Urethane is also an artifact resulting from treatment of beverages such as wine, beer, orange juice, and some soft drinks with pyrocarbonate, a fungicide that breaks down in the beverage after treatment (Schmähl 1977).
There is clear evidence for the carcinogenicity of urethane in experimental animals (IARC 1987). Urethane has been shown to be carcinogenic in mice, rats, and hamsters after administration by the oral route, producing lung tumors, lymphomas, hepatomas, and melanomas (Berenblum and Haran-Ghera 1957; Toth et al. 1961; Klein 1962, 1966; Della Porta et al. 1963; Adenis et al. 1968; Toth and Boreisha 1969).
The relevance to humans of animal studies of urethane is not known. No case reports or epidemiologic studies in humans of urethane are available, although clearly alcoholic beverages are carcinogenic to humans. On the basis of animal studies, IARC (1987) concludes that urethane is possibly carcinogenic to humans and has classified it as a Group 2B carcinogen.
Acquired Naturally Occurring Carcinogens: Aflatoxin B1
As discussed earlier, the aflatoxins are the most ubiquitous of the fungal toxins, and aflatoxin B1 the most potent and most studied
of all. In addition to being an acquired naturally occurring carcinogen, often found on grains, nuts, and seed meals, it is also a pass-through naturally occurring carcinogen, as it can be found in milk and other edible products from animals that have consumed feed contaminated with aflatoxin.
The carcinogenic properties of aflatoxin B1 have been extensively investigated, and much information has been generated regarding their metabolic activation and mechanisms of action (Wogan 1992). Aflatoxin B1 has been identified by IARC (1993) and NTP (1994) as a carcinogen. There is sufficient evidence to indicate the carcinogenicity of aflatoxin in experimental animals (IARC 1993). Administered in the diet, aflatoxin B1 has been tested for carcinogenicity in many animal species and found to produce tumors primarily of the liver, colon, and kidneys. After oral administration, aflatoxin B1 caused hepatocellular and/or cholangiocellular liver tumors, including carcinomas, in all species tested (including rats, hamsters, and monkeys) except mice (Wogan and Newberne 1967, 1971; Butler et al. 1969; Epstein et al. 1969; Wogan 1969, 1974; Kalengayi et al. 1975; Nishizumi et al. 1977; Sieber et al. 1979; Zawirska and Bednarz 1981; Moore et al. 1982; Bannasch et al. 1985; Soffritti and McConnell 1988). In rats, renal cell tumors were also found but a low incidence of tumors at other sites, including the colon (Wogan and Newberne 1967, Butler et al. 1969, Epstein et al. 1969). In monkeys, liver angiosarcomas of the gall bladder and pancreas developed, in addition to hepatocellular and cholangiocellular carcinomas (Sieber et al. 1979). In rats and hamsters, aflatoxin B1 administered in the diet induced foci of altered hepatocytes, the number and size of which was correlated with later development of hepatocellular adenomas and carcinomas (Wogan and Newberne 1967, Wogan et al. 1971, Kalengayi et al. 1975, Moore et al. 1982, Bannasch et al. 1985, Gil et al. 1988, Soffritti and McConnell 1988).
There is also sufficient evidence to indicate the carcinogenicity of aflatoxin B1 in humans (IARC 1993). Epidemiologic studies
(Autrup et al. 1987, Peers et al. 1987, Yeh et al. 1989) and several case-control studies (Bulatao-Jayme et al. 1982, Cusumano 1991, Parkin et al. 1991, Srivatanakul et al. 1991) have provided convincing evidence that dietary consumption of aflatoxin B1 plays an etiologic role in hepatocarcinogenesis. Two correlation studies—one in Swaziland and one in China—indicate a synergy between chronic viral B (also C) hepatitis and aflatoxin exposure (Peers et al. 1987, Ross et al. 1992, Qian et al. 1993). In addition, a synergistic interaction between chronic alcohol consumption and aflatoxin exposure also appears to play a role in human hepatocarcinogenesis. Furthermore, approximately 55% of the hepatocellular carcinomas from people exposed to aflatoxins contain an AGG to AGT mutation at codon 249 of the p53 tumor suppressor gene (Greenblatt et al. 1994), a mutation that is preferentially induced in cultured human hepatocytes exposed to aflatoxin B1. This fact supports the causative role of aflatoxins in human hepatocarcinogenesis. Less than 4% of liver tumors found in people from developed countries, in which exposure to aflatoxins is relatively low, contain this mutation (Aguilar et al. 1994). Thus, the conventional and molecular epidemiologic studies clearly indicate that aflatoxins are carcinogenic to humans.
On the basis of studies in animals and data in humans, IARC (1993) concludes that aflatoxin B1 is carcinogenic to humans and has classified it as a Group 1 carcinogen.
Derived Naturally Occurring Carcinogens
PhIP (2-Amino-1-Methyl-6-Phenylimidazo[4,5-b]Pyridine)
In investigations of foods for the presence of multiple polycyclic
heterocyclic amines (PHAs), PhIp is usually found to be the most abundant (IARC 1993). PHAs have been isolated from a ordinary human diet cooked simulating household conditions (Alink et al. 1988).
According to IARC (1993), there is sufficient evidence in experimental animals for the carcinogenicity of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP). PhIP was tested for carcinogenicity in one experiment in mice and two experiments in rats by oral administration in the diet. It increased the incidence of lymphomas in mice of each sex (Esumi et al. 1989). In rats, it produced adenocarcinomas of the small and large intestine in males and mammary adenocarcinomas in females (Ito et al. 1991, Ochiai et al. 1991). The relevance of these findings to human health is unknown. No data directly relevant to an evaluation of the carcinogenicity to humans of PhIP were available.
On the basis of these studies, IARC (1993) concludes that PhIP is possibly carcinogenic to humans and has classified it as a Group 2B carcinogen.
N-Nitrosodimethylamine
N-nitrosodimethylamine is a derived naturally occurring carcinogen present in a variety of foods, including cheese, soybean oil, canned fruit, various meat products and cured meats, bacon, frankfurters, ham (cooked), fish and fish products, apple brandy, other alcoholic beverages, and beer. Concentrations in these foodstuffs have been measured to be between 0 and 85 ng/kg. Levels of N-nitrosodimethylamine in various foods from several countries, including the United States, have been reported by IARC (1978).
N-nitrosodimethylamine has been identified by IARC (1978) and NTP (1994) as an animal carcinogen. There is sufficient evidence to indicate the carcinogenicity of N-nitrosodimethylamine in several experimental animal species (IARC 1978). When administered
orally, it induced liver hemangiosarcomas, hepatocellular carcinomas, and kidney and lung tumors in mice (Takayama and Oota 1963, 1965; Terracini et al. 1966; Toth et al. 1964; Clapp et al. 1968, 1971; Den Engelse et al. 1969, 1970; Clapp and Toya 1970; Otsuka and Kuwahara 1971; Shabad and Savluchinskaya 1971; Zwicker et al. 1972). The chemical also induced kidney and bile duct tumors in rats and hepatocellular carcinomas and bile duct tumors in hamsters, rabbits, and guinea pigs when orally administered (Magee and Barnes 1956, 1959, 1962; Schmähl and Preussmann 1959; Zak et al. 1960; Tomatis et al. 1964; Terracini et al. 1967, 1969; Geil et al. 1968; Le Page and Christie 1969a, b; Riopelle and Jasmin 1969; Kowalewski and Todd 1971; Hadjiolov and Markow 1973; Taylor et al. 1974; Shinohara et al. 1976). N-nitrosodimethylamine is also carcinogenic when it is administered prenatally and in single doses. In several of the studies, dose-response relationships were established.
No case reports or epidemiologic studies are available to evaluate the carcinogenicity of N-nitrosodimethylamine in humans. However, similarities in its metabolism by human and rodent tissues have been demonstrated. Therefore, IARC (1978) concludes that N-nitrosodimethylamine ''should be regarded for practical purposes as if it were carcinogenic in humans" and has classified it as a Group 2A carcinogen.
Current State of Knowledge of Human Dietary Anticarcinogens
Considerable evidence suggests that the consumption of fruits and vegetables is important in the prevention of human cancer (NRC 1989a, Birt and Bresnick 1991, Block et al. 1992, Lelloff et al 1994). Fruits and vegetables are associated with reduced rates of several forms of human cancer, including stomach, lung, breast, and colon. These observations, in addition to other beneficial
properties of fruits and vegetables, have encouraged the development of public health campaigns such as the "five-a-day" program designed to increase their intake (NRC 1989a).
A few studies have been conducted in experimental animals on the ability of specific dietary plants to prevent cancer (Birt and Bresnick 1991). Most extensively studied have been members of the cruciferous, allium, and tea families, which were effective in rodents preventing cancers at numerous sites, including esophagus, colon, lung, breast, and skin. There is evidence that some of the inhibition of cancer by fruits and vegetables is due to the essential nutrient vitamins A, C, E, and selenium. There have also been extensive investigations of the particular chemical compounds present in fruits and vegetables, as shown in Appendix C. The results of these investigations suggest that these minor constituents contribute significantly to cancer prevention.
Of particular interest are members of the flavonoid class (apigenin, myricetin, quercetin, robinetin, and rutin), which are widely distributed in foods and which inhibit a range of cancers, such as skin, colon, and lung. Conversely, the most extensively studied flavonoid, quercetin, has also been reported to be a carcinogen under other conditions of feeding (Appendix A). These opposing effects on cancer are not unique to flavonoids. For example, caffeic acid, chlorogenic acid, and eugenol have been observed both to increase and to inhibit the formation of neoplasias. Observations of both positive and negative effects on health have been made for numerous other compounds in foods, such as lectins, phenolic compounds, saponins, and enzyme inhibitors (Thompson 1993). Similarly, synthetic dietary compounds such as butylated hydroxy-anisole (BHA) have been found both to induce and to inhibit carcinogenesis in animals (Ito et al. 1989). For example, forestomach cancer was induced by high doses of a number of antioxidants, including BHA. However, these same antioxidants given at low doses, with more potent carcinogens, were effective in inhibiting cancer at a number of sites (Ito et al. 1989). Further studies are
needed to define the conditions necessary to achieve inhibition of cancer, avoid undesirable effects, and identify mechanisms of action.
A number of plant phenolics, such as genistein and indole-3 carbinol, have received considerable attention in recent years because of their weak estrogenic activity. Such compounds are called phytoestrogens , and it has been hypothesized that these chemicals may block more potent estrogens by binding to estrogen receptors that control gene expression. However, many of the effects of these phenolic compounds do not appear to be related to this binding. It was recently proposed by Safe that some of the potential toxic impact of estrogenic industrial compounds may well be prevented by the large number of phytoestrogens in the diet (Safe, in press).
It is important to note that the results in bioassay testing for carcinogenicity and anticarcinogenicity will probably be skewed by the methods currently used. These methods tend to identify anticarcinogenic properties of chemicals more rapidly than carcinogenic properties, for the following reasons: 1) anticarcinogenicity is typically assayed in the presence of, or following treatment with, a potent carcinogen in a 15-50 week animal bioassay, while to demonstrate potential carcinogenicity, tests require two years with a larger number of animals, and 2) because of the tremendous difference in cost for these two types of studies, it is likely that more chemicals will be studied for their anticarcinogenic effects than for their carcinogenic effects.
There are, moreover, issues of relevance. Compounds are tested for carcinogenicity at high doses to be as certain as possible of detecting weaker effects that would occur rarely and possibly be missed in low-dose testing. However, as noted in Chapter 4, high doses introduce problems of extrapolating properly to low human exposures, even when no other factors render the high-dose effects uninterpretable. High-dose exposure to a potent carcinogen, as described above in animal testing for anticarcinogenic effects, is hardly the typical human situation, and the interpretation of such
high-dose exposures in anticarcinogenicity studies is open to the same criticisms that apply to high-dose testing for carcinogenicity. It would be more realistic to look for the effects of putative anticarcinogens on background tumor rates or on rates in animals with genetic predisposition to certain cancers parallel to human genetic patterns, or on animals fed ad libitum vs. calorie-restricted diets. Biological markers, discussed in Chapter 6, may also prove to be of value here.
Many of the compounds listed in Appendix C are reported to have antioxidant properties. However, there are numerous other mechanisms whereby these dietary components may modify carcinogenesis. These include activating detoxification pathways or deactivating toxification pathways, inhibiting cellular proliferation, affecting hormonal modulation, inhibiting interaction of growth factors with their receptors, etc. Equally important, the impact of any single compound on cancer development will almost certainly be the sum of numerous effects of the chemical, rather than a single biological effect. This complexity will inevitably make it difficult to determine mechanisms of dietary prevention of cancer.
The compounds in Appendix C have been assessed individually in experimental animals, or in cell or tissue culture studies. There is no current evidence that these compounds are effective in cancer prevention in humans. Diets rich in plants or in particular types of plants, however, have been associated with reduced rates of some forms of human cancer.
It is important to note that about 70 constitutive naturally occurring chemicals from dietary plants are reported to possess both mutagenic and antimutagenic and, in some cases antioxidant properties. Most of these fall under the following classes: flavonoids, phenolic acids, phenylpropanoids, coumarins, depsides, cyclitols, isothiocyanates, catechins, simple phenols, monoterpenes, sesquiterpenes, amino acids, and anthraquinones (Farnsworth 1994).
Effect of Dietary Macronutrients on Carcinogenesis
In this and the following section, we discuss the effects of dietary nutrients (macro- and micro-) separately from non-nutrients, because the data available on these two categories are considerably different. In general, nutrients have been extensively studied and attempts have been made to identify optimal ranges of intakes. Studies on the modulation of cancer by nutrients compare low dietary intakes with optimal and high intakes. Few studies assess nutrient-free diets in cancer modulation because of the known adverse health consequences of consuming deficient diets. In contrast, diets that contain non-nutrient additives have been compared with diets that are free of these agents. Since these components, by definition, are not known to be required, their impact on cancer development has been studied by an approach more typical of toxicology than of nutrition.
Dietary macronutrients include carbohydrates, protein, fats, and alcohol (NRC 1989a). With the exception of alcohol, each class consists of a number of substances that are structurally related.
The dietary substances that ordinarily constitute these classes of macronutrients are generally not carcinogenic. However, carcinogens may be generated when foods containing these substances are cooked excessively, as described in the section on pyrolytic compounds (see previous section on pyrolytic mutagens) (Lijinsky and Ross 1967, Sugimura 1985, Felton et al. 1986). Furthermore, macronutrients may influence the development of cancer by acting as enhancers or, in some cases, inhibitors of carcinogenesis. This section is intended to summarize some of the more important, generally accepted findings concerning the effects of dietary macronutrients on cancer development. This subject was previously reviewed by the Committee on Diet and Health (NRC 1989a).
Calories
Reducing calorie intake results in a reduction in cancer. This phenomenon, known as the caloric effect, appears to be the major effect of dietary macronutrients on carcinogenesis in experimental animals.
The caloric effect, discovered early in this century, is one of the most well-documented and generally effective anticarcinogens known for rodents (Pariza and Boutwell 1987). As an example, conducted an experiment in which a group of 50 female BDA mice were fed a mixture of dog chow meal and skimmed milk powder. Supplementation with 1 gm of corn starch in addition to the 2 gm basic feed given to controls increased the spontaneous breast cancer incidence from 0% to 38%. Similar studies conducted in rats reported approximately 20% reduction of lifetime malignant tumor incidence, and many of the tumors observed in the animals whose diets were restricted appeared only when most of the rats fed ad libitum had died. The cancer potency value calculated for the rat study fell between the values derived from the mouse study. These observations suggest that rats and mice might react in a similar manner to excess food and that generalizations to humans may be possible. Haseman and Rao (1992) demonstrated an association in rats between body weight and leukemia, pituitary, and mammary tumors. In male mice an association was also demonstrated between body weight and liver and lung tumors. Hart and Turturro (1994) observed reduced tumor incidences at these sites in calorierestricted animals. These results are consistent with the finding that excess calorie intake by rodents is associated with higher tumor rates for some tissue sites.
There is considerable epidemiologic evidence that the balance between calorie intake and energy expenditure influences the risk of cancer in humans as well (Kritchevsky 1993, Willett 1994). Nevertheless, independent associations between macronutrients and selected cancers have been found in many epidemiologic studies
after controlling for caloric intake (Van't Veer et al. 1990, Willett et al. 1990, Giovannucci et al. 1993b). It appears that the biochemical mechanism of the caloric effect involves changes in hormonal balance (Pariza and Boutwell 1987, Kritchevsky 1993). Specifically, the effect may be mediated by elevated secretion of adrenal hormones such as glucocorticoid hormone and/or dehydroepiandrosterone (Schwartz and Pashko 1994). A considerable body of evidence suggests that glucocorticoids inhibit inflammation, a concomitant of many cytotoxic-regenerative processes associated with enhanced tumorigenesis in some tissues. It is also known that caloric restriction can affect metabolic processes, including enzymes involved in carcinogen activation and inactivation (ILSI 1995). Reduced caloric intake also reduces the rate of cell proliferation or increases the rate of apoptosis (programmed cell death) (Lok et al, 1988, 1990; Grasl-Kraupp et al. 1994; ILSI 1995). Effects on cell proliferation appear to be a particularly significant means of modulating carcinogenesis, including effects on spontaneous tumors in rodents. The effect on apoptosis is particularly prominent in preneoplastic lesions such as hyperplastic nodules in the rat liver (Grasl-Kraupp et al. 1994). A major consequence of inhibiting apoptosis is to increase the number of cells available for replication, in contrast to increasing the rate of proliferation. The resulting effect of either is an increase in the number of DNA replications.
The caloric effect is recognized as markedly influencing the quantitative assessment of carcinogenic potency of chemicals tested by the rodent bioassay (ILSI 1995). Animals fed ad libitum appear to be more responsive when tested in the standard rodent bioassay. In this way, calories play a central role in our evaluation of chemicals and the extrapolation of potency data from rodent studies to humans.
Carbohydrates
The effects of carbohydrates on carcinogenesis have been studied
primarily in relation to their contribution to dietary energy. Recent studies have compared the impact of carbohydrate energy on carcinogenesis with that of fat energy on carcinogenesis (see below).
A more extensively studied macronutrient that is often categorized with carbohydrate is fiber, which is not a single substance but rather a collection of many different carbohydrate-containing materials (NRC 1989a). These include cellulose, hemicellulose, pectin, and lignin. Fiber is not degraded by mammalian digestive enzymes but may be partially metabolized by colonic microflora. Some fibers are water-soluble, others insoluble.
The consumption of fiber-rich foods, including those high in pentoses, is associated with decreased colon cancer risk. However, this relationship may be due, at least in part, to other (nonfiber) components of fruits, vegetables, and grains (NRC 1989a). There is some evidence that dietary fiber may reduce the risk of adenomatous polyps of the colon, which are generally considered to be precursor lesions for colon cancer (Neugut et al. 1993). However, the data from animal studies are not consistent: some studies indicate protection whereas others indicate no effect or even enhanced cancer risk. In general, wheat bran exhibits the most consistent inhibiting effect (NRC 1989a). Recent studies suggest that some of the cancer-prevention effects of dietary fiber may be related to the lignan precursors and other phytoestrogens in whole grain foods rich in fiber (Thompson 1994).
Fat
Previous NRC committees (1982 and 1989) have concluded from epidemiologic studies that of all the dietary factors, fat exhibited the most consistent cancer-enhancing effect. However, a clear consensus on the strength of the relationship between dietary fat per se and cancer risk in humans remains elusive (Pariza and Boutwell 1987, Kritchevsky 1993, Willett 1994). For example, although many case-control studies have found positive associations between
breast cancer and dietary fat intake, most cohort studies have failed to reproduce this finding (NRC 1989a, Willett 1994). Studies of colon cancer and the consumption of fat (including saturated fat) have been more consistent. However, the fat association is at least in part attributable to a strong association with red meat (Giovannucci et al. 1994). Such an association could reflect other carcinogenic constituents in these foods, such as the heterocyclic amines produced during cooking at high temperatures. A similar situation occurs in prostate cancer, which has been associated with high intake of saturated fats, but also with the high consumption of red meats (Kolonel et al. 1988, Giovannucci et al. 1993b, Le Marchand et al. 1994).
In contrast to the results of epidemiologic investigations, animal studies have produced a much more consistent pattern, because the diets of experimental animals can be readily controlled. Recent experimental studies have suggested that dietary fat and energy may interact in some manner to modify cancer. For example, high dietary fat enhanced mammary carcinogenesis in rats only when diets were freely fed (Welsch et al. 1990). Furthermore, when energy from fat and from carbohydrate were compared for their impact on carcinogenesis, calories from fat appeared to be somewhat more effective in enhancing carcinogenesis than calories from carbohydrates (Zhu et al. 1991, Birt et al. 1993). Thus, while at least some of the impact of fat on cancer appears due to its high caloric density, it also appears that certain properties of fat may enhance cancer, independent of energy.
Dietary fat has been extensively studied experimentally as a modulator in animal models of the multistage process of carcinogenesis. The data are strongest for an impact of dietary fat on events involved in cell proliferation and gene expression (NRC 1982).
Rancid fat contains peroxides and aldehyde mutagens that could initiate and/or promote carcinogenesis, but this possibility has not been extensively studied (Ames 1983). It has also been proposed that fatty acid oxidation may be involved in tumor promotion within
the colon (Ames 1983, Carroll 1985, Welsch 1987). Lane et al. (1985) reported that mammary tissue from mice fed diets high in corn oil had less malonaldehyde (a product of lipid peroxidation) than mice fed diets low in corn oil, even though the mice fed the high corn oil diet developed more mammary tumors after 7,12-dimethylbenzathracene was administered. Hence, while in situ lipid peroxidation may be important in enhancing the development of some forms of cancer (e.g., colon cancer), it does not appear to be involved in carcinogenesis at other sites (e.g., breast).
Linoleic Acid
Linoleic acid is the only fatty acid that has been shown unequivocally to enhance carcinogenesis in animal studies. The effect depends on levels of dietary linoleic acid and exhibits a linear dose-response when the concentration is between 1% and 5%. Above 5%, the effect plateaus (Ip et al. 1985). At extremely high levels (>16%) of linoleic acid in the diet, there is a reduction in cell proliferation in the mammary gland. The implications for carcinogenesis in the mammary gland should be explored (Lok et al. 1988, 1990, 1992). Much of the reported effect on experimental carcinogenesis of "fat type" appears to be due to the linoleic acid effect.
Conjugated Linoleic Acid (CLA)
As linoleic acid is the only fatty acid shown unequivocally to enhance carcinogenesis, conjugated linoleic acid (CLA) is the only fatty acid shown unequivocally to inhibit carcinogenesis in experimental animals (Ha et al. 1987, 1990; Ip et al. 1991, 1994). In contrast, the data indicating that omega-3 fatty acids may inhibit carcinogenesis are ambiguous (Pariza 1988), although these fatty acids appear to play a role in reducing the risk of heart disease in
human populations (NRC 1989a). The major dietary sources of CLA are foods derived from ruminant animals, for example dairy products and beef (Chin et al. 1992).
CLA is effective as an anticarcinogen when present in the diet of experimental animals at levels as low as 0.05% to 0.1% (Ip et al. 1994). It appears to act via signal transduction pathways and effects on prostaglandin metabolism. In this regard, it has recently been shown that CLA is a growth factor for rats, possibly because it also modulates the catabolic effects resulting from immune stimulation (Chin et al. 1994). There are similarities in the effects of CLA and the effects of omega-3 fatty acids on mammals and birds; in general, CLA appears to be more potent (Miller et al. 1994).
Bile Acids and Free Fatty Acids
Bile acids and free fatty acids are generated during normal digestion. They are toxic for cells of the colonic mucosa and may potentiate the development of colon cancer. It has been proposed that calcium may inhibit the effect by complexing with bile acids and free fatty acids (Scalmati et al. 1992). Epidemiologic data on the relationship of fecal bile acids and neutral steroids to the risk of colon cancer are inconsistent (Kolonel and Le Marchand 1986).
Protein
The major effect of dietary protein on carcinogenesis appears to be caloric (Clinton et al. 1992), although under conditions where protein is growth-limiting, fewer tumors develop (Topping and Visek 1976). Excessive dietary protein increases colonic ammonia levels; ammonia in turn may enhance the development of chemically induced colonic tumors (Clinton et al. 1988). However, few epidemiologic studies have implicated dietary protein in cancer risk
(NRC 1989a). Some studies show associations of colon and breast cancers with dietary protein, particularly animal protein (Lubin et al. 1986, Potter and McMichael 1986). This association could indirectly reflect high correlations between the intake of protein and fat or protein and red meat in the study populations.
Alcohol
Unlike other dietary macronutrients, alcohol refers to a single substance, ethyl alcohol. However, in epidemiologic investigations, it is generally not possible to isolate the effects of ethanol from those of the many congeners in alcoholic beverages. Excessive alcohol consumption has been linked to increased cancer risk at several sites in humans, particularly when combined with certain other factors including tobacco use, infection with Hepatitis B virus, and poor dietary habits (NRC 1989a). Animal experiments indicate that ethyl alcohol enhances cancer risk when administered in the diet or applied topically in conjunction with another carcinogenic agent. In humans, alcohol ingestion appears to increase the risk of cancer in susceptible individuals, but it does not appear to be a genotoxic carcinogen (Seitz and Simanowski 1988). A variety of mechanisms have been proposed to explain the enhancement of carcinogenesis by alcohol, including effects on cell membranes, DNA structure, and carcinogen metabolism.
Effect of Dietary Micronutrients on Carcinogenesis
As noted earlier, considerable epidemiologic and experimental evidence suggests that a number of micronutrients, including vitamins A, C, E, and selenium, contribute to cancer prevention. Conversely, diets deficient in these micronutrients have been associated
with an increased risk of cancer. These micronutrients are antioxidants and evidence suggests that some of their anticancer effects may be through inhibition of oxidation; however, they may act through other mechanisms.
This section provides an overview of the micronutrients that have been most extensively studied in cancer cause and prevention. Much of the evidence that these micronutrients are important in cancer prevention comes from the association of fruits and vegetables with cancer prevention. It should be noted that supplemental nutrients have not been observed to be as effective as a diet rich in fruits and vegetables (NRC 1989a). It should be further noted that direct evidence for a specific effect of fruits and vegetables, based on intervention studies in humans, has not yet been reported. It is possible that diets rich in fruits and vegetables are associated with reduced cancer rates because of the lower fat and calorie intake associated with such diets. However, studies involving experimental animals, in which fat and energy intake are controlled, suggest that fruits and vegetables have inhibitory properties (Birt and Bresnick 1991). Furthermore, several epidemiologic studies have reported an inverse relationship between the intake of fruits and/or vegetables and specific cancers; the relationship was shown to be independent of fat or energy intake (Macquart-Moulin et al. 1986, Slattery et al. 1988, Hunter et al. 1993, Pohan et al. 1993, Omenn 1995).
Vitamin A
The naturally occurring forms of vitamin A (retinol, retinal, retinoic acid and its carotenoid precursors) have been extensively studied in animals and humans for their efficacy in cancer prevention. The strength of the inverse relationship between intakes of vitamin A (especially its precursors, the carotenoids) and reduced cancer at several sites led to the development of synthetic analogues
of vitamin A which have been extensively studied in animals and recently used in breast cancer patients.
The most convincing evidence that vitamin A and its precursors, the carotenoids, prevent human cancer comes from prospective and retrospective epidemiologic studies associating low intakes of fruits and vegetables with elevated risk of cancer (Ziegler 1991). The data are particularly convincing with respect to lung cancer; however, studies also suggest that vegetable and fruit intake may reduce the risk of cancers at other sites (e.g., the oral cavity, pharynx, larynx, esophagus, colon, rectum, bladder, and cervix) (Ziegler 1991). Investigations suggest that ![]() -carotene is the most effective carotenoid in cancer prevention. However, one study implicates other components of fruits and vegetables in cancer prevention (Le Marchand et al. 1989). In this study of the relationship between the intake of fruits and vegetables and lung cancer risk in humans, results indicated a negative dose-dependent relationship between dietary
-carotene is the most effective carotenoid in cancer prevention. However, one study implicates other components of fruits and vegetables in cancer prevention (Le Marchand et al. 1989). In this study of the relationship between the intake of fruits and vegetables and lung cancer risk in humans, results indicated a negative dose-dependent relationship between dietary ![]() -carotene and lung cancer risk, but no clear association for retinol, vitamin C, folic acid, iron, dietary fiber, or fruits. However, all vegetables showed a stronger inverse relationship with lung cancer risk than did
-carotene and lung cancer risk, but no clear association for retinol, vitamin C, folic acid, iron, dietary fiber, or fruits. However, all vegetables showed a stronger inverse relationship with lung cancer risk than did ![]() -carotene, suggesting that other constituents of vegetables, such as lutein, lycopene, and indoles, may have anticancer activity (Le Marchand et al. 1989).
-carotene, suggesting that other constituents of vegetables, such as lutein, lycopene, and indoles, may have anticancer activity (Le Marchand et al. 1989).
The mechanism(s) of vitamin A inhibition of cancer have been extensively debated. Whatever the mechanism, it appears that the carotenoid precursors of vitamin A are responsible for at least some of its anticancer effects (Bendich and Olson 1989). Hypothesized mechanisms whereby carotenoids may inhibit cancer are diverse and include (1) inhibition of mutagenesis and protection against photo-damage, (2) enhancement of immune system responses, (3) reduction of nuclear damage by carcinogenic agents, (4) protection against neoplastic events in cells, and (5) the quenching of highly reactive singlet oxygen (Bendich and Olson 1989, Krinsky 1991). With respect to these mechanisms, ![]() -carotene has been shown to protect against the mutagenic effects of 8-methoxypsoralen (xanthotoxin)
-carotene has been shown to protect against the mutagenic effects of 8-methoxypsoralen (xanthotoxin)
and ultraviolet-A light in the presence of oxygen but not under anoxic conditions (Krinsky 1991). ![]() -carotene and other carotenoids have also been shown to enhance immune system responses in cells and animals (Bendich and Olson 1989, Krinsky 1991). For example, addition of
-carotene and other carotenoids have also been shown to enhance immune system responses in cells and animals (Bendich and Olson 1989, Krinsky 1991). For example, addition of ![]() -carotene or canthaxanthin to peripheral blood mononuclear cells results in an increase in cells with natural killer markers and with interleukin-2 receptors (Krinsky 1991). A study of the transformation of fibroblasts exposed to 3-methylcholanthrene or to x-rays found that
-carotene or canthaxanthin to peripheral blood mononuclear cells results in an increase in cells with natural killer markers and with interleukin-2 receptors (Krinsky 1991). A study of the transformation of fibroblasts exposed to 3-methylcholanthrene or to x-rays found that ![]() -carotene protected against nuclear damage at physiologic concentrations (Bendich and Olson 1989). Studies have also shown that vitamin A binds to nuclear receptors that are members of the steroid hormone receptor superfamily (Evans 1988).
-carotene protected against nuclear damage at physiologic concentrations (Bendich and Olson 1989). Studies have also shown that vitamin A binds to nuclear receptors that are members of the steroid hormone receptor superfamily (Evans 1988).
In addition, experimental studies with vitamin A-deficient animals indicate an enhancement of lung cancer by 3-methylcholanthrene and of liver and colon cancer by aflatoxin B1. While vitamin A may protect against the development of colon cancer, the presence of vitamin A deficiency inhibited N-methyl-N'-nitro-N-nitrosoguanidine induced colon cancer in animals (Birt 1986). The hypothesis that vitamin A protects against a number of human cancers is based on the similarities observed between morphological changes in vitamin A-deficient tissues and in premalignant lesions. Furthermore, vitamin A has been identified as a mammalian morphogen (Evans 1988).
The development of synthetic analogues of vitamin A was driven by the need for agents possessing its cancer-preventive properties, but without its inherent toxicity. A number of analogues have been developed that are effective in cell culture systems and laboratory animals (Moon 1989). Some of these compounds appear to involve interaction with the nuclear vitamin A receptors to modify gene transcription. Other mechanisms of action may involve induction of apoptosis or induction of growth factors that play a role in regulation of cell proliferation (Roberts and Sporn 1992). Preliminary evidence in human trials suggests that 13-cis-retinoic acid may
prove useful against oral cancer (Borden et al. 1993). However, the toxicity of vitamin A and its analogues still remains a barrier to extensive use.
Ongoing clinical trials with ![]() -carotene in the prevention of several forms of human cancer, including lung, oral cavity, and breast cancer, should help to determine whether the association between fruit and vegetable intake and reduced rates of cancer is due in part to the presence of
-carotene in the prevention of several forms of human cancer, including lung, oral cavity, and breast cancer, should help to determine whether the association between fruit and vegetable intake and reduced rates of cancer is due in part to the presence of ![]() -carotene. In one recent report, neither
-carotene. In one recent report, neither ![]() -carotene nor -tocopherol supplements were effective in preventing lung cancer in Finnish men who were heavy smokers (Heinonen and Albanes 1994). These data may mean that
-carotene nor -tocopherol supplements were effective in preventing lung cancer in Finnish men who were heavy smokers (Heinonen and Albanes 1994). These data may mean that ![]() -carotene and -tocopherol are not the active agents in fruits and vegetables that reduce cancer, or they may indicate that these smokers had a level of damage that could not be corrected by these supplements. Further research will be needed to determine the contribution, if any, of other carotenoids in cancer prevention. It is clear that dietary supplementation with vitamin A for cancer prevention is unlikely at this point because of its inherent toxicity. However, improving our understanding of its mechanism as a morphogen and the role it plays in the induction of differentiation may help us to develop a more effective analogues of vitamin A for future cancer prevention research.
-carotene and -tocopherol are not the active agents in fruits and vegetables that reduce cancer, or they may indicate that these smokers had a level of damage that could not be corrected by these supplements. Further research will be needed to determine the contribution, if any, of other carotenoids in cancer prevention. It is clear that dietary supplementation with vitamin A for cancer prevention is unlikely at this point because of its inherent toxicity. However, improving our understanding of its mechanism as a morphogen and the role it plays in the induction of differentiation may help us to develop a more effective analogues of vitamin A for future cancer prevention research.
Vitamin C (Ascorbic Acid)
Higginson (1966) reported an inverse association between consumption of foods rich in ascorbic acid and the appearance of certain cancers. Since then, a comprehensive review of epidemiologic studies has assessed the role of ascorbic acid in cancer and provided convincing evidence that ascorbic acid, an important component of fruits and vegetables, prevents cancer at a number of sites (Block 1991). An examination of the relationship between gastric cancer and ascorbic acid provides some of the strongest evidence for its
anticancer effects (Mirvish 1983, Block 1991); ascorbic acid status is particularly important in the prevention of gastric cancer in populations with chronic gastric infection (Correa 1994). Cancers of the esophagus, larynx, oral cavity, pancreas, rectum, breast, and cervix are lower in people who consume diets rich in fruits and vegetables (Block 1991). There is also some evidence that inhibition of lung cancer may be related to the ascorbic acid content of these foods (Block 1991). Ascorbic acid was shown to be reduced in smokers, independent of dietary intake. The recommended daily dietary allowance for Vitamin C is 60 mg for adult men and women, while the daily requirement of ascorbic acid is estimated to be 200 mg for smokers (Schectman 1993).
It is believed that ascorbic acid exerts much of its anticarcinogenic effect by inhibiting the formation of N-nitroso compounds in the stomach (Mirvish 1983, Tannenbaum et al. 1991). By reducing nitrite to nitric oxide, ascorbic acid prevents the reaction between nitrite and amines. A reaction between nitrite and amines would result in the formation of nitroso-compounds (Tannenbaum et al. 1991). In addition, it has been demonstrated that sodium ascorbate (22.7 g/kg) and morpholine administered in the diet, along with sodium nitrite in the drinking water, inhibited the formation of N-nitrosomorpholene and liver cancer (Mirvish 1983). The inhibitory role of ascorbic acid in chemically induced carcinogenesis has also been studied in experimental animals at other sites (e.g., skin, trachea, lung, mammary gland, colon, kidney, and urinary bladder) (Birt 1986). Cancers of the skin and colon were reported to be enhanced and inhibited, cancer of the urinary bladder was enhanced, while cancers of the other organs were inhibited. Several mechanisms have been proposed to explain the ability of ascorbic acid to prevent chemically induced carcinogenesis. Mechanisms include its effects as an antioxidant, its role in enhancing cellular immunity, and its role in inhibiting the activation of chemical carcinogens. However, the role of ascorbic acid in chemically induced cancer prevention remains unclear (Block 1991).
Vitamin E (Tocopherols)
Vitamin E consists of a number of related tocopherols. In contrast to the studies of vitamins A and C, epidemiologic studies provide less consistent support for a role of vitamin E in cancer prevention. (Diplock 1991, Stähelin et al. 1991, Garland et al. 1993). The inconsistencies reported in the literature may relate to the poor stability of tocopherol in stored samples. Prospective studies require collection of a large number of samples, and often it is impossible to analyze all the samples at once (Diplock 1991). The 12-year follow-up in the Basel prospective study provided little evidence for an association between vitamin E and cancer at any site (Stähelin et al. 1991). Another prospective study reported that the serum concentration of vitamin E in cancer patients was lower than in controls when the patients were diagnosed within one year of the date of blood collection (Wald et al. 1987). However, these investigators suggested that the low levels of serum vitamin E were a consequence rather than a cause of cancer.
Experimental carcinogenesis studies have provided evidence that vitamin E plays a role in cancer prevention (Birt 1986). Topical administration of vitamin E resulted in inhibition of carcinogenesis when the skin was initially treated with 7-12-dimethylbenz(a)anthracene (DMBA) followed by 12-0-tetradecanoylphorbol-13-acetate. Dietary administration of vitamin E resulted in inhibiting skin cancer induced by dibenzopyrene. In addition, DMBA-produced cancers of the hamster cheek pouch, mouse forestomach, and rat mammary gland were inhibited by oral treatment (gavage or dietary) with vitamin E (Birt 1986). However, consistent effects of vitamin E were not observed in colon carcinogenesis (Birt 1986).
Vitamin E, like ascorbic acid, inhibits nitrosation of amines. Vitamin E, however, is effective in the lipid compartments of cells, while vitamin C acts in aqueous environments (Newberne and Locaiskar 1990). Studies of its ability to quench the superoxide anion indicate that vitamin E, like vitamin A, may contribute to
protection of biological systems from singlet oxygen (Di Mascio et al. 1991). Interestingly, -tocopherol, generally the most abundant tocopherol in the plasma, is also the most effective form of vitamin E in quenching singlet oxygen (Di Mascio et al. 1991). In addition, considerable evidence suggests that vitamin E may inhibit free radicals formed by mitochondria (Ames et al. 1993).
Folic Acid
Folic acid is found abundantly in vegetables and fruits. Because the consumption of such foods has been associated with reduced cancer rates, and because adequate dietary folic acid is required for the regulation of normal gene expression (1994), folic acid has been implicated in cancer prevention. Considerable research has been conducted on the role of methyl deficiency (usually including deficiencies of folic acid, vitamin B-12, choline, and methionine), DNA methylation, and the induction of liver cancer in rodents (Dizik et al. 1991, Cravo et al. 1992). However, it was not clear if the methyl-deficient conditions induced in animals were applicable to human diets, although the prevalence of inadequate folate intakes has been documented (NRC 1989a). A case-control study of colon and rectum cancer was conducted in 1975-1986 (Freudenheim et al. 1991). Cancer patients (428 colon and 372 rectal cancer patients) were matched with controls, and all were interviewed about dietary practices. When data were adjusted for energy intake, odds ratios for rectal cancer patients with the highest folate intakes compared with those with the lowest intakes were 0.3 and 0.5 for men and women, respectively (Freudenheim et al. 1991). Risk of colon cancer was not associated with dietary folate intake. Furthermore, the difference in odds ratio was greatest for men with the highest alcohol intake, suggesting a possible interaction (Freudenheim et al. 1991). Although earlier studies suggested that folate might protect against cervical cancer, recent case-control studies uncovered no such relationship (Ziegler et al. 1990).
The relation between intake of folic acid, methionine and alcohol, and colon cancer was further investigated because hypomethylated DNA was observed in patients with colorectal carcinomas and because folic acid plays a role in DNA methylation (Giovannucci et al. 1993a). This investigation followed women in the Nurses' Health Study and men in the Health Professionals Follow-up Study after a 1-year dietary assessment. Adenomatous polyps were observed in 564 women and in 331 men. Dietary folate was inversely associated with risk of adenoma in women and men, while alcohol intake above 30 gm/day was positively associated with adenoma risk. Dietary methionine was inversely associated with risk of an adenoma one cm or larger (Giovannucci et al. 1993a). These results support the importance of methyl group availability in the prevention of colorectal cancer.
A recent study demonstrates that methyl-deficient diets (deficient in choline, methionine, and folic acid) in rats fed semipurified diets could result in imbalances in deoxynucleotide pools, which are known to produce mutagenic events (James et al. 1992). In addition, hypomethylation of cytosine, cytosine, guanine, guanine (CCGG, a sequence of nucleotides in DNA) sites have been demonstrated in animals with severe methyl deficiency (diets lacking choline, methionine, folic acid, and vitamin B-12). It is known that CCGG sites in genes, such as c-myc, c-fos and c-Ha-ras, are involved in cellular proliferation and cancer (Christman et al. 1993). Furthermore, folate deficiency has been associated with increases in chromosomal breaks (Ames et al. 1995). These observations support the role of folic acid in cancer prevention.
Vitamin D and Calcium
The role of dietary vitamin D and calcium in cancer prevention was first suggested because of the observation that people in increasingly northern latitudes had higher colon cancer mortality rates (Garland and Garland 1980). Such an association could be
due to the impact of ultraviolet light on the synthesis of vitamin D in the skin, and subsequently, on the absorption of dietary calcium. This suggestion was pursued in a 19-year prospective study in Chicago, Illinois, which demonstrated a 50% reduction in colon cancer in men who had a daily intake of 3.75 µg vitamin D and a 75% reduction in men who had a daily intake of > 1200 mg calcium (Garland et al. 1991). Evaluation of the levels of circulating 25-hydroxy vitamin D revealed higher values in controls (67-102 nmol/L) (Garland et al. 1991). A prospective study on women in Iowa further supports the hypothesis that vitamin D and/or calcium protect against colon cancer (Bostick et al. 1993).
An extensive series of experiments was conducted in rodents (Newmark and Lipkin 1992) to assess a diet that mimicked four of the suggested dietary risk factors for colon cancer: high fat and phosphate, and low calcium and vitamin D. Feeding this stress diet for 12 weeks resulted in hyperproliferation of cells in the sigmoid colon. Subsequent experiments demonstrated that increasing the level of dietary calcium could return colonic proliferation to normal values. Studies in human subjects at increased risk for colon cancer similarly found a reduction in hyperproliferation of the colonic epithelium when diets were supplemented with calcium (Newmark and Lipkin 1992). Studies of rats treated with 1,2-dimethylhydrazine (DMH) and fed graded levels of calcium and vitamin D showed that both nutrients reduced DMH-induced colon cancer and altered colonic cell kinetics (Beaty et al. 1993). Comparable studies on mammary carcinogenesis induced by 7,12-dimethylbenz(a)-anthracene (DMBA) suggested that high levels of dietary calcium and vitamin D protect against mammary carcinogenesis, while high levels of dietary phosphate increase susceptibility (Carroll et al. 1991).
Prostate cancer risk was recently reported to be inversely associated with exposure to ultraviolet light and it was hypothesized that this was another cancer related to vitamin D intake (Hanchette and Schwartz 1992). This hypothesis is supported by the presence of
vitamin receptors in the prostate gland (Berger et al. 1988) and by the evidence supporting a role for vitamin D in the regulation of differentiation and gene expression (Minghetti and Norman 1988).
Selenium
The impact of dietary selenium on carcinogenesis has been the subject of considerable controversy. Early observations of selenium toxicity in animals indicated that excessive amounts were associated with the development of neoplastic alterations in the liver. However, under controlled experimental conditions selenium was a potent inhibitor of liver carcinogenesis, an observation that has been extended to a number of experimental models (El-Bayoumy 1991). Over the past 30 years, numerous investigations have probed the role of selenium in cancer epidemiology and in experimental carcinogenesis (El-Bayoumy 1991). There appears to be a particularly narrow range between the intake of dietary selenium that risks deficiency and those levels at which toxicity can occur.
The first investigation of the relationship between selenium and human cancer assessed the connection between forage selenium and cancer mortality (Clark et al. 1991). A strong inverse relationship between regional forage selenium and cancer mortality was observed. This association was re-examined recently, and cancers of the lung, breast, rectum, bladder, esophagus, and corpus uteri were shown to be elevated in areas with low forage selenium. The association between plasma selenium and esophageal cancer was examined in blacks living in rural areas of southern Africa (Jaskiewicz et al. 1988). Blacks living in areas of high esophageal cancer incidence had lower whole blood selenium levels (58-71 ng/ml) than blacks living in areas of low esophageal cancer incidence (114-177 ng/ml) (Jaskiewicz et al. 1988). In addition, the mean level of blood selenium was lower in patients with premalignant or malignant esophageal cytologic changes than in subjects without such lesions.
A prospective cohort study on lung cancer risk and selenium status (measured by toenail selenium) reported a 50% reduction in relative risk for the cancer among individuals with the highest toenail selenium levels (Van den Brandt et al. 1993). Interestingly, the protective effect of selenium against lung cancer was strongest in the individuals with lower intakes of vitamin C or ![]() -carotene (Van den Brandt 1993). Interactions between selenium and other nutrients have also been observed in studies with animals (Birt 1986). Breast cancer risk was not found to be related to plasma selenium in a prospective study on Guernsey Island (Denmark) (Overvad et al. 1991), although numerous experimental studies in animals have shown that selenium treatment inhibits breast cancer (El-Bayoumy 1991). The association between urinary bladder cancer and serum selenium, a-tocopherol, lycopene,
-carotene (Van den Brandt 1993). Interactions between selenium and other nutrients have also been observed in studies with animals (Birt 1986). Breast cancer risk was not found to be related to plasma selenium in a prospective study on Guernsey Island (Denmark) (Overvad et al. 1991), although numerous experimental studies in animals have shown that selenium treatment inhibits breast cancer (El-Bayoumy 1991). The association between urinary bladder cancer and serum selenium, a-tocopherol, lycopene, ![]() -carotene, and retinol was reported in a 12-year follow-up of a prospective study in Washington County, Maryland (Helzlsouer et al. 1989). The results indicated the controls had lower plasma selenium concentrations (Helzlsouer et al. 1989).
-carotene, and retinol was reported in a 12-year follow-up of a prospective study in Washington County, Maryland (Helzlsouer et al. 1989). The results indicated the controls had lower plasma selenium concentrations (Helzlsouer et al. 1989).
Extensive investigations have been conducted on the impact of dietary selenium on carcinogenesis in laboratory animals (El-Bayoumy 1991). Induction of preneoplastic lesions in the liver and of liver carcinogenesis by a number of carcinogens was inhibited by selenium administration by dietary, gavage, intraperitoneal or subcutaneous route (El-Bayoumy 1991). Skin carcinogenesis was generally inhibited by topical and dietary selenium administration (El-Bayoumy 1991), but at high doses of selenium, or high doses of carcinogen, selenium was found to enhance skin carcinogenesis (Birt et al. 1989). Colon carcinogenesis was consistently inhibited in animals administered dietary selenium, but inconsistent effects were observed in the pancreas (El-Bayoumy 1991). Selenium was generally observed to be effective at inhibiting carcinogenesis at doses of 0.2 to 5 ppm in the diet. However, the association between cancer and selenium deficiency in animals has not been clearly demonstrated (El-Bayoumy 1991). Recent investigations are
exploring novel seleno- compounds with increased efficacy in cancer prevention (El-Bayoumy 1991).
Numerous mechanisms have been explored to explain the modulation of carcinogenesis by selenium (Medina 1986, El-Bayoumy 1991). The best characterized function of selenium in mammalian cells is as a component of the seleno- enzyme, glutathione peroxidase. This enzyme is localized in the cytosol and mitochondrial matrix, and it eliminates organic peroxides from the cell (Medina 1986). However, available evidence suggests that the prevention of carcinogenesis by selenium is not related to its function in glutathione peroxidase (Medina 1986). Other seleno- proteins have been identified, but their impact on carcinogenesis is not defined (Medina 1986). There is some evidence that selenium may alter the metabolism of carcinogens or the interaction of chemical carcinogens with DNA, but there is considerable controversy in the literature (Medina 1986). Additional mechanistic studies suggest that selenium may alter cell proliferation and/or immunologic responses (Medina 1986, El-Bayoumy 1991). Further research is needed to understand the mechanisms whereby selenium prevents cancer.
Iron
Considerable controversy has surrounded the role of iron in carcinogenesis (Weinberg 1992), largely because of the policy of fortifying food with iron to prevent anemia. Recent reports have provided some evidence for the impact of iron status on the development of cancer. Results from a prospective study of 41,000 men and women in Finland indicated an elevated risk of colorectal and lung cancer for individuals with transferrin saturation in excess of 60% (Knekt et al. 1994). In contrast, the risk of stomach cancer was inversely related to serum iron and transferrin saturation in those cases occurring during the first 5 years of follow-up (Knekt et al. 1994). Studies conducted in South African populations with
excessive iron intake and high levels of serum iron did not provide evidence of an elevated risk of cancer (Higginson and Oettle 1960). Further investigations are needed to understand the effect, if any, of iron on the development of cancer.
Engineering an Optimal Diet
For thousands of years mankind has manipulated the quality of food to obtain improved flavor, color, odor, productivity, and safety. The work of Gregor Mendel in the last century provided the scientific basis for the discipline of plant breeding used so successfully in this century to improve our food plants. Animal breeders also have selected for superior characteristics in species used as food by man.
Conventional plant breeding is based on the cross breeding of different plants possessing desirable characteristics. Initially, the crossing involved individuals of the same species, but today sexually incompatible species of the same family often can successfully be crossed. In both cases, native DNA of one individual is mixed with the DNA of the second and stably preserved and expressed, producing offspring, some of which will have the characteristics of both parents.
These impressive achievements are now being supplemented and enhanced by numerous techniques described under the general term of biotechnology. Biotechnology can be applied to plants and animals, but plants are enjoying greater attention because of their extensive use as food, their less-complex genetics, and the lack of some of the ethical issues animal biotechnology sometimes raises. Plant genetic engineering is a form of biotechnology in which DNA of defined chemical composition bearing specific genetic information is introduced into the genome of a plant to express a new protein or alter the level of an endogenous gene. In this sense, genetically engineered (transgenic) plants are less randomly changed genetically.
than varieties produced by crossing because the genome of the engineered plant will have been modified by one or at most two or three genes. In traditional plant breeding, many unknown genes are introduced by crossing. The resulting great variability among the offspring requires extensive and time-consuming screening. The full consequences can be known only by detailed physical, chemical, and physiological analysis of the offspring, which is seldom performed. For a more complete discussion of traditional and newer methods of genetic modification, see IFBC (1990a).
The past decade has seen the production of transgenic plants of many crop species (Gasser and Fraley 1989, 1992). Much of the early effort was toward improved agronomic traits such as resistance to herbicides or increased yield. More recently, food plants are being studied to obtain improved quality in storage and transport (tomatoes), tolerance to cold and freezing (strawberries), nutritional improvement (lipids, sugars, amino acids, and proteins), and improved processing properties (Comai 1993). Today, knowledge of plant genetic engineering is sufficiently advanced that any character that is controlled by one or only a few genes probably can be transferred to a food plant. Therefore, the application of plant genetic engineering to the task of removing known carcinogens or increasing the amounts of known anticarcinogens in foods should be expected.
In theory, the removal of a deleterious substance or the increase of a desirable compound could occur in several ways. Thus, the deletion of a carcinogen from a plant food source could occur by using antisense DNA technology to inactivate the gene coding for the enzyme catalyzing the rate-limiting step in biosynthesis of the carcinogenic compound. Alternatively, the amount of a carcinogen might be decreased using gene enhancement to increase the activity of an endogenous enzyme known to convert the carcinogen to the next compound in the normal metabolism of the carcinogen. Introduction of a gene for an enzyme known to detoxify the carcinogen (e.g., a cytochrome P450) would accomplish the same purpose.
The reciprocal approach could be used for increasing the amounts of a desirable anticarcinogen. Thus, the enzyme catalyzing the rate-limiting step in biosynthesis of the anticarcinogen would need to be enhanced, while an enzyme catalyzing the further metabolism of the anticarcinogen should be diminished.
Certain mycotoxins (aflatoxins, fumonisins, and ochratoxins) are produced by fungal infection of plant and animal foods by species of Aspergillus, Fusarium, Penicillium, and Alternaria (CAST 1989, IFBC 1990a). In these cases, it is the infective organism rather than the plant that produces the carcinogen. Because genetic engineering of ubiquitous, phytopathogenic fungi is a daunting, if not impossible task, a secondary approach should be considered. A food plant serving as host provides a source of macro- and micronutrients (sugars, amino acids, vitamins, and growth factors) that are required by the fungus. If the concentrations of nutrients within the host plant can be lowered, the fungus would not be able to grow. Alternatively, plants possess genes that confer resistance to fungi; if those genes can be identified and enhanced, the fungi would be unable to infect the plant and produce the mycotoxin.
All of the genetic engineering described above is dependent on knowledge of the biochemical processes involved in the biosynthesis of the carcinogens, anticarcinogens, and growth factors. That is, the enzymes responsible for a key step in biosynthesis or the first step in catabolism of the compounds of interest need to be available for isolation of the appropriate genes. Regrettably, there is little detailed information on the typically multistep biosynthesis of many of the known carcinogens and anticarcinogens. To decrease the carcinogen directly, another approach might be used. In one recent example, the concentration of a toxic glucosinolate (mustard oil glucoside) was greatly diminished in a commercially significant canola plant, not by inactivating the last step in its biosynthesis, but by diverting the first compound (precursor) in the biosynthetic pathway leading to the glucosinolate. Tryptophan is known to be converted to indole glucosinolates by a sequence of six or seven
reactions, only some of which are known precisely. In other plants, tryptophan can be decarboxylated by the enzyme tryptophan decarboxylase to form tryptamine. When the gene for tryptophan decarboxylase was isolated from the medicinal plant Cantharus roseus and introduced into Brassica napus, the transformed plants were greatly reduced in their content of indole glucosinolates (Chavadej et al. 1994).
It is only a matter of time until a wide variety of bioengineered foods will be available for public consumption. Concerns regarding the safety of genetically engineered food plants have been extensively discussed (Comai 1993, IFBC 1990a, WHO 1991, Kessler et al. 1992, OECD 1992), and most scientists agree that the transformation process introduces no inherently new categories of hazard and that existing procedures for testing and screening, properly employed, are adequate to ensure the safety of the products. The policies regulating genetically engineered foods have also been summarized by Harlander (1993). The basic regulatory principles are found in a statement by the FDA (1992), which indicates that no regulation other than those applied to foods obtained by classical plant breeding are necessary.
The first genetically engineered food to be marketed is the Flavr/Savr™ tomato developed by Calgene, Inc. Its safety has been extensively examined and documented for examination by the FDA (Redenbaugh et al. 1992). As with most other new technologies, public acceptance of this first bioengineered food will doubtless depend on whether the purchaser sees a benefit from the product of the new technology.
Summary And Conclusions
The human diet is enormously complex; it consists of variable mixtures of dietary components. Animal studies indicate that certain dietary components may be carcinogenic, while others may
have anticarcinogenic effects. Indeed, in some instances a single constituent might be carcinogenic and anticarcinogenic under different circumstances.
Nutrients present in the diet contribute to the prevention of cancer. Considerable evidence in human and animal systems suggests that diets rich in a number of vitamins and minerals protect against cancer at a wide variety of sites. Such diets tend to contain an abundance of fruits and vegetables and are also associated with reduced rates of other chronic diseases. Ongoing cancer prevention trials will help to identify the importance of specific nutrients or other constituents and, in some cases, interactions between nutrients. However, until we have this information, it is of the utmost importance to continue recommending that the public consume diets rich in fruits and vegetables but low in fat and calories. The consumption of vitamins and minerals in a moderate, varied, and balanced diet—not as dietary supplements—continues to be one of our best strategies for cancer prevention in people.
References
Adenis, L., A. Demaille, and J. Driessens. 1968. Pouvoir cancérigène de l'uréthane chez le rat Sprague. C.R. Soc. Biol. (Paris) 162:458-461.
Aguilar, F., C.C. Harris, T. Sun, M. Hollstein, and P. Cerutti. 1994. Geographic variation of p53 mutational profile in non-malignant human liver. Science 264:1317-1319.
Alink, G.M., M.G. Knize, N. H. Shen, S. P. Hesse, and J.S. Felton. 1988. Mutagenicity of food pellets from human diets in the Netherlands. Mutat. Res. 206:387-393.
Ames, B.N. 1983. Dietary carcinogens and anticarcinogens. Science 221:1256-1264.
Ames, B.N., L.S. Gold, and W.C., Willett. 1995. The causes and prevention of cancer. Proc. Natl. Acad. Sci. U.S.A. 92:5258-5265.
Ames, B.N., M. Profet, and L.S. Gold. 1990. Dietary pesticides
(99.99% all natural). Proc. Natl. Acad. Sci. U.S.A. 87:7777:7781.
Ames, B.N., M.K. Shigenaga, and T.M. Hagen. 1993. Oxidants, antioxidants, and the degenerative diseases of aging. Proc. Natl. Acad. Sci. U.S.A. 90(17):7915-7922.
Anonymous. 1970. Webster's New World Dictionary of the American Language, 2nd College ed., D.B. Guralnik, ed. New York: World Publishing Co.
Anonymous. 1966. Webster's Third New International Dictionary of the English Language, Unabridged. Chicago, IL: Merriam-Webster. Encyclopedia Britannica.
ApSimon, J. 1989. Phytoalexins as human and animal toxins: recent advances Pp. 229-241 in 1989 Annual Summer Toxicology Forum. Washington, DC: Toxicology Forum.
Argus, M.F., and C. Hoch-Ligeti. 1961. Comparative study of the carcinogenic activity of nitrosamines. J. Nat. Cancer Inst. 27:695.
Autrup, H., T. Seremet, J. Wakhisi, and A. Wasumma. 1987. Aflatoxin exposure measured by urinary excretion of aflatoxin B1 guanine adduct and hepatitis B virus infection in areas with different liver cancer incidence in Kenya. Cancer Res. 47:3430-3433.
Bannasch, P., U. Benner, H. Enzmann, and H.J. Hacker. 1985. Tigroid cell foci and neoplastic nodules in the liver of rats treated with a sigle dose of aflatoxin B1. Carcinogenesis 6:1641-1648.
Beaty, M.M., E.Y. Lee, and H.P. Glauert. 1993. Influence of dietary calcium and vitamin D on colon epithelial cell proliferation and 1,2-dimethylhydrazine-induced colon carcinogenesis in rats fed high fat diets . J. Nutr. 123:144-152.
Bendich, A. and J.A. Olson. 1989. Biological actions of carotenoids. FASEB J. 3:1927-1932.
Berenblum, I., and N. Haran-Ghera. 1957. A quantitative study of the systemic initiating action of urethane (ethyl carbamate) in mouse skin carcinogenesis. Brit. J. Cancer 11:77-84.
Berger, U., P. Wilson, R.A. McClelland, K. Colston, M.R. Haussler, and J.W. Pike. 1988. Immunocytochemical detection of 1,25-dihydroxyvitamin D receptors in normal human tissues. J. Clin. Endocrinol. Metab. 67:607-613.
Birt, D.F. 1986. Update on the effects of vitamins A, C and E and selenium on carcinogenesis. Pp. 311-320 in Proceedings of the
Society for Experimental Biology and Medicine.
Birt, D.F. and E. Bresnick. 1991. Chemoprevention by non-nutrient components of vegetables and fruits. Pp. 221-261 in Human Nutrition: A Comprehensive Treatise, R. Alfin-Slater and D. Kritchevsky, eds. New York: Plenum.
Birt, D.F., P.M. Pour, and J.C. Pelling. 1989. The influence of dietary selenium on colon, pancreas and skin tumorigenesis. Pp. 297-304 in 4th International Symposium on Selenium in Biology and Medicine, A. Wendet, ed.
Birt, D.F., H.J. Pinch, T. Barnett, A. Phan, and K. Dimitroff. 1993. Inhibition of skin tumor promotion by restriction of fat and carbohydrate calories in SENCAR mice. Cancer Res. 53:27-31.
Block, G. 1991. Vitamin C and cancer prevention: The epidemiologic evidence. Am. J. Clin. Nutr. 53:270S-282S.
Block, G., B. Patterson, and A. Subar. 1992. Fruit, vegetables, and cancer prevention: A review of the epidemiological evidence. Nutr. Cancer 18:1-29.
Booth, A.N., O.H. Emerson, F.T. Jones, and F. DeEds. 1957. Urinary metabolites of caffeic and chlorogenic acids. J. Biol. Chem. 229:51-59.
Borden, E.C., R. Lotan, D. Levens, C.W. Young, and S. Waxman. 1993. Differentiation therapy of cancer: Laboratory and clinical investigations. Cancer Res. 53: 4109-4115.
Bostick, R.M., J.D. Potter, T.A. Sellers, D.R. McKenzie, L.H. Kushi, and A.R. Folsom. 1993. Relation of calcium, vitamin D, and dairy food intake to incidence of colon cancer among older women: The Iowa women's health study. Am. J. Epidemiol. 137:1302-1317.
Bulatao-Jayme, J., E.M. Almero, M.C.A. Castro, M.T.R. Jardeleza, and L.A. Salamat. 1982. A case-control dietary study of primary liver cancer risk from aflatoxin exposure. Int J. Epidemiol. 11:112-119.
Butler, W.H., M. Greenblatt, and W. Lijinsky. 1969. Carcionogenesis in rats by aflatoxins B1, G1, and B2. Cancer Res. 29:2206-2211.
Carroll, K.K. 1985. Lipid oxidation and carcinogenesis. Pp. 237-244 in Genetic Toxicology of the Diet, I. Knudsen, ed. New York: Liss. 351 pp.
Carroll, K.K., E.A. Jacobson, L.A. Eckel, and H.L. Newmark. 1991. Calcium and carcinogenesis of the mammary gland. Am. J. Clin.
Nutr. 54(Suppl.):206S-208S.
Chavadej, S., N. Brisson, J.N. McNeil, and V. DeLuca. 1994. Redirection of tryptophan leads to production of low indole glucosinolate canola. Proc. Natl. Acad. Sci. 91:2166-2170.
Cheftel, J.C., J.-L. Cuq, and D. Lorient. 1985. Amino acids, peptides, and proteins. Pp. 245-369 in Food Chemistry, 2nd edition, O.R. Fennema, ed. New York: Marcel Dekker, Inc.
Chin, S.F., W. Liu, J.M. Storkson, Y.L. Ha, and M.W. Pariza. 1992. Dietary sources of conjugated dienoic isomers of linoleic acid, a newly recognized class of anticarcinogens. J. Food Comp. Analysis 5:185-197.
Chin, S.F., J.M. Storkson, K.J. Albright, M.E. Cook, and M.W. Pariza. 1994. Conjugated linoleic acid is a growth factor for rats as shown by enhanced weight gain and improved feed efficiency. J. Nutr. 124:2344-2349.
Christman, J.K., G. Sheikhnejad, M. Dizik, S. Abileah, and E. Wainfan. 1993. Reversibility of changes in nucleic acid methylation and gene expression induced in rat liver by severe dietary methyl deficiency. Carcinogenesis 14:551-557.
Clapp, N.K., A. W. Craig, and R.E. Toya, Sr. 1968. Pulmonary and hepatic oncogenesis during treatment of male RF mice with dimethylnitrosamine. J. Nat. Cancer Inst. 41:1213.
Clapp, N.K., and R.E. Toya, Sr. 1970. Effect of cumulative dose and dose rate on dimethylnitrosamine oncogenesis in RF mice. J. Nat. Cancer Inst. 45:495.
Clark, L.C., K.P. Cantor, and W.H. Allaway. 1991. Selenium in forage crops and cancer mortality in U.S. counties. Arch. Environ. Health 46:37-42.
Clarke, R.J., and R. Macrae, eds. 1985. Coffee, Volume 1, Chemistry. New York: Elsevier Applied Science. 306 pp.
Clinton, S.K., D.G. Bostwick, L.M. Olson, H.J. Manigan, and W.J. Visek. 1988. Effects of ammonium acetate and sodium cholate on N-methyl-N'nitro-N-nitrosoguanidine-induced colon carcinogenesis of rats. Cancer Res. 48:3035-3039.
Clinton, S.K., P.B. Imrey, H.J. Mangian, S. Nandkumar, and W.J. Visek. 1992. The combined effects of dietary fat, protein, and energy intake on azoxymethane-induced intestinal and renal carcinogenesis.
Cancer Res. 52:857-865.
Comai, L. 1993. Impact of plant genetic engineering on foods and nutrition. Annu. Rev. Nutr. 13:191-215.
Connolly, J.D. and R.A. Hill. 1991. Dictionary of terpenoids. Vols. 1-3. London: Chapman and Hall
Corpet, D.E., D. Stamp, A. Medline, S. Minkin, M.C. Archer, and W.R. Bruce. 1990. Promotion of colonic microadenoma growth in mice and rats fed cooked sugar or cooked casein and fat. Cancer Res. 50:6955-6958.
Correa, P. 1991. The epidemiology of gastric cancer. World J. Surg. 15:228-234.
Cravo, M.L., J.B. Mason, and Y. Dayal, M. Hutchinson, D. Smith, J. Selhub, and I.H. Rosenberg. 1992. Folate deficiency enhances the development of colonic neoplasia in dimethylhydrazine-treated rats. Cancer Res. 52:5002-5006.
Crosby, D.G. 1969. Natural toxic background in the food of man and his animals. J. Ag. and Food Chem. 17:(May-June)532-538.
Cusumano, V. 1991. Aflatoxins in sera from patients with lung cancer. Oncology 48:194-195.
Czok, V.G., W. Walter, K. Knoche, and H. Degener. 1974. Reabsorption of chlorogenic acid in the rat.(in German). Zeit. fur Ernahrungswissenschaft 13:108-112.
Davíâdek, J., J. Velâí & Sacute; ek, and J. Pokornây, J., eds. 1990. Chemical Changes During Food Processing. New York: Elsevier. Pp. 100-119.
Della Porta, G., J. Capitano, W. Montipo, and L. Parmi. 1963a. Studio sull' azione cancerogena dell' uretano nel topo. Tumori 49:413-428.
Della Porta, G., J. Capitano, and P. Strambio de Castillia. 1963b. Studies on leukemogenesis in urethan-treated mice. Acta Un. Int. Cancer 29:783-785.
Den Engelse, L., P.A.J. Bentvelzen, and P. Emmelot. 1969/1970. Studies on lung tumours. I. Methylation of deoxyribonucleic acid and tumour formation following administration of dimethylnitrosamine to mice. Chem.-Biol. Interactions 1:394-406.
Di Mascio, P., M.E. Murphy, and H. Sies. 1991. Antioxidant defense systems: the role of carotenoids, tocopherols and thiols. Am. J. Clin.
Nutr. 53:194S-200S.
Dickey, R. 1989. Ciguatera: Toxin production in isolates of Procentrum concavum and P. lima. Pp. 247-252 in 1989 Annual Summer Toxicology Forum. Washington, DC: Toxicology Forum.
Diplock, A.T. 1991. Antioxidant nutrients and disease prevention: An overview. Am. J. Clin. Nutr. 53(Suppl.):189S-193S.
Dizik, M., J.K. Christman, and E. Wainfan. 1991. Alterations in expression and methylation of specific genes in livers of rats fed a cancer promoting methyl-deficient diet. Carcinogenesis 12:1307-1312.
Ehrlich, P.R., and P.H. Raven. 1964. Butterflies and plants: A study in coevolution. Evolution 18:586-608.
El-Bayoumy, K. 1991. The role of selenium in cancer prevention. Pp. 1-15 in Cancer Principles and Practice of Oncology, 4th edition, V. Davita, S. Helman, and S.A. Rosenberg, eds. Philadelphia, PA.
Epstein, S., B. Bartus, and E. Farber. 1969. Renal epithelial neoplasms induced in male Wistar rats by oral aflatoxin B. Cancer Res. 29:1045-1050.
Esumi, H., H. Ohgaki, E. Kohzen, S. Takayama, and T. Sugimura. 1989. Induction of lymphoma in CDF1 mice by the food mutagen, 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine. Jpn. J. Cancer Res. 80:1176-1178.
Evans, R.M. 1988. The steroid and thyroid hormone receptor superfamily. Science 240:889-895.
Farnsworth, N.R. 1994. Data were obtained from the NAPRALERT database on natural products, which covers the natural product literature from at least 1975 through October 1, 1994. NAPRALERT has the capability of restricting searches to plants used as foods and is available on line through the Scientific and Technical Network (STN) of Chemical Abstracts Services. Columbus, Ohio.
FDA (U.S. Food and Drug Administration). 1992. Statement of Policy: Foods derived from new plant varieties. Fed. Regist. 57:22984-23005 (29 May 1992).
Feeny, P. 1975. Coevolution of Animals and Plants: Symposium V, First International Congress of Systematic and Evolutionary Biology.
L.E. Gilbert and P.H. Raven, eds. Austin, TX: University of Texas Press.
Felton, J.S., M.G. Knize, N.H. Shen, B.D. Andersen, L.F. Bjeldanes, and F.T. Hatch. 1986. Identification of the mutagens in cooked beef. Environ. Health Perspec. 67:17-24.
Fraenkel, G.S. 1959. The raison d'être of secondary plant substances. Science 129:1466-1470.
Freudenheim, J.L., S. Graham, J.R. Marshall, B.P. Haughey, S. Cholewinski, and G. Wilkinson. 1991. Folate intake and carcinogenesis of the colon and rectum. Int. J. Epidemiol. 20:368-374.
Garland, C.F., and F.C. Garland. 1980. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int. J. Epidemiol. 9:227-231.
Garland, C.F., F.C. Garland, and E.D. Gorham. 1991. Can colon cancer incidence and death rates be reduced with calcium and vitamin D. Am. J. Clin. Nutr. 54(Suppl.):193S-201S.
Garland, M., W.C. Willett, J.E. Manson, and D.J. Hunter. 1993. Antioxidant micronutrients and breast cancer. J. Am. Coll. Nutr. 12:400-411.
Geil, J.H., R.J. Stenger, R.M. Behki, and W.S. Morgan. 1968. Hepatotoxic and carcinogenic effects of dimethylnitrosamine in low dosage. Light and electron microscopic study. J. Nat. Cancer Inst. 40:713-730.
Gil, R., R. Callaghan, J. Boix, A. Pellin, and A. Llombart-Bosch. 1988. Morphometric and cytophotometric nuclear analysis of altered hepatocyte foci induced by N-nitrosomorpholine (NNM) and aflatoxin B1 (AFB1) in liver of Wistar rats. Virchows Arch. B Cell Pathol. 54:341-349.
Giovannucci, E., E.B. Rimm, G.A. Colditz, M.J. Stampfer, A. Ascherio, C.C. Chute, and W.C. Willett. 1993b. A prospective study of dietary fat and risk of prostate cancer . J. Natl. Cancer Inst. 85:1571-1579.
Giovannucci, E., M.J. Stampfer, G.A. Colditz, E.B. Rimm, D. Trichopoulos, B.A. Rosner, F.E. Speizer, and W.C. Willett. 1993a. Folate, methionine, and alcohol intake and risk of colorectal adenoma. J. Natl. Cancer Inst. 85:875-884.
Giovannucci, E, E.B. Rimm, M.J. Stampfer, G.A. Colditz, A. Ascherio,
and W.C. Willett. 1994. Intake of fat, meat, and fiber in relation to risk of colon cancer in men. Cancer Res. 54:2390–2397.
Glasby, J.S. 1982. Encyclopedia of Terpenoids. Vols. 1 and 2. Chichester, New York: Wiley. 2646 pp.
Grasl–Kraupp, B., W. Bursch, B. Ruttkay–Nedecky, A. Wagner, B. Lauer, and R. Schulte–Hermann. 1994. Food restriction eliminates preneoplastic cells through apoptosis and antagonizes carcinogenesis in rat liver. Proc. Natl. Acad. Sci. USA 91:9995–9999.
Greenblatt, M.S., W.P. Bennett, M. Hollstein, and C.C. Harris. 1994. Mutations in the p53 tumor suppressor gene: Clues to cancer etiology and molecular pathogenesis. Cancer Res. 54:4855–4878.
Ha, Y.L., N.K. Grimm, and M.W. Pariza. 1987. Anticarcinogens from fried ground beef: Heat–altered derivatives of linoleic acid. Carcinogenesis 8:1881–1887.
Ha, Y.L., J. Storkson, and M.W. Pariza. 1990. Inhibition of benzo(a)pyrene–induced mouse forestomach neoplasia by conjugated dienoic derivatives of linoleic acid. Cancer Res. 50:1097–1101.
Hadjiolov, D., and D. Markow. 1973. Fine structure of hemangioendothelial sarcomas in the rat liver induced with N–nitrosodimethylamine. Arch. Geschwulstforsch. 42:120–126.
Hall, R.L. 1992. Food Additives. Pp. 280–283 in McGraw–Hill Encyclopedia of Science and Technology, 7th ed., Vol. 7. New York: McGraw–Hill, Inc.
Hall, S. 1989. Introduction to marine toxins and paralytic shellfish poisons. Pp. 242–246 in 1989 Annual Summer Toxicology Forum. Washington, DC: Toxicology Forum.
Hanchette, C.L., and G.G. Schwartz. 1992. Geographic patterns of prostate cancer mortality: Evidence for a protective effect of ultraviolet radiation. Cancer 70:2861–2869.
Harborne, J.B. 1988. The Flavonoids: Advances in Research Since 1980, vol. 2. London: Chapman and Hall. 900 pp.
Harborne, J.B. 1994. Introduction to Ecological Biochemistry, 4th edition. London: Academic Press. 400 pp.
Harborne, J.B., T.J. Mabry, and H. Mabry. 1975. The Flavonoids. Vols. 1 and 2. London: Chapman and Hall. 1204 pp.
Hart, R.W., and A. Turturro. 1995. Dietary restriction: An update. Pp. 1–12 in Dietary Restriction: Implications for the Design and
Interpretation of Toxicity and Carcinogenicity Studies. R.W. Hart, D.A. Neumann, and R.T. Robertson, eds. Washington, DC: ILSI Press.
Haseman, J.K., and G.N. Rao. 1992. Effects of corn oil, time-related changes, and inter-laboratory variability on tumor occurrence in control Fischer 344 (F344/N) rats . Toxicol. Pathol. 20:52-60.
Hawk, P.B. 1965. Hawk's Physiological Chemistry, 14th edition. B.L. Oser, ed. New York: McGraw-Hill. pp. 112.
Heinonen, O.P. and D. Albanes. 1994. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. N. Engl. J. Med. 330:1029-1035.
Helzlsouer, K.J., G.W. Comstock, and J.S. Morris. 1989. Selenium, lycopene, -tocopherol, -carotene, retinol, and subsequent bladder cancer. Cancer Res. 49:6144-6148.
Herrmann, K. 1989. Occurrence and content of hydroxycinnamic and hydroxybenzoic acid compounds in food. Crit. Rev. Food Sci. Nutr. 28:315-347.
Higginson, J. 1966. Etiological factors in gastrointestinal cancer in man. JNCI 37:527-545.
Higginson, J., and A.G. Oettle. 1960. Cancer incidence in the Bantu and ''Cape Colored" races of South Africa: Report of a cancer survey in the Transvaal (1953-1955. JNCI 24:589-671.
Hirose, M., A. Masuda, S. Fukushims, and N. Ito. 1988. Effects of subsequent antioxidant treatment on 7,12-dimethylbenz[a]anthracene-initiated carcinogenesis of the mammary gland, ear duct and forestomach in Sprague-Dawley rat. Carcinogenesis 9:101-104.
Hirose, M.M. Mutai, S. Takahashi, M. Yamada, S. Fukushima, and N. Ito. 1991. Effects of phenolic antioxidants in low-dose combination on forestomach carcinogenesis in rats pretreated with N-methyl-N'-nitrosoguanidine. Cancer Res. 51:824-827.
Hirose, M., M. Kawabe, M. Shibata, S. Takahashi, S. Okazaki, and N. Ito. 1992. Influence of caffeic acid and other o-dihydroxybenzene derivatives on N-methyl-N'-nitro-N-nitrosoguanidine-initiated rat forestomach carcinogenesis. Carcinogenesis 13:1825-1828.
Hölldobler, B. 1995. The chemistry of social regulation:
Multicomponent signals in ant societies. Proc. Nat. Acad. Sci. 92(1):19-22.
Hudson, B.J.E. 1990. Food Antioxidants. London: Elsevier Applied Science. 317 pp.
Hunter, D.J., J.E. Manson, G.A. Colditz, M.J. Stamper, B. Rosner, C.H. Hennekens, F.E. Speizer, and W.C. Willett. 1993. A prospective study of the intake of vitamins C, E, and A and the risk of breast cancer. N. Engl. J. Med. 329:234-240.
IARC (International Agency for Research on Cancer). 1974. IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man. Some Anti-Thyroid and Related Substances, Nitrofurans, and Industrial Chemicals. Volume 7. Lyon, France: IARC.
IARC (International Agency for Research on Cancer). 1978. Chemicals with Sufficient Evidence of Carcinogenicity in Experimental Animals. IARC Monographs Volumes 1-17. (IARC Intern. Tech. Rep. No. 78/003). Lyon, France: IARC.
IARC (International Agency for Research on Cancer). 1987. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Overall Evaluations of Carcinogenicity: An Updating of IARC Monographs Volumes 1 to 42, Supplement 7. Lyon, France: IARC.
IARC (International Agency for Research on Cancer). 1993. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins. Vol. 56. Lyon France: IARC.
IFBC (International Food Biotechnology Council). 1990a. Biotechnologies and food: Assuring the safety of foods produced by genetic modification. Regul. Toxicol Pharmacol. 12:3(part 2) S1-S196.
IFBC (International Food Biotechnology Council). 1990b. Biotechnologies and food: Assuring the safety of foods produced by genetic modification. Regul. Toxicol Pharmacol. 12:3(part 2) S23.
IFBC (International Food Biotechnology Council). 1990c. Biotechnologies and food: Assuring the safety of foods produced by genetic modification. Regul. Toxicol Pharmacol. 12:3(part 2) S28-S29.
IFBC (International Food Biotechnology Council). 1990d.
Biotechnologies and food: Assuring the safety of foods produced by genetic modification. Regul. Toxicol Pharmacol. 12:3(part 2) S92-93.
ILSI (International Life Sciences Institute). 1995. Dietary Restriction: Implications for the Design and Interpretation of Toxicity and Carcinogenicity Studies. R.W. Hart, D.A. Neumann, and R.T. Robertson, eds. Washington, D.C.: ILSI Press.
Inoue, S., K. Ito, K. Yamamoto, and S. Kawanishi. 1992. Caffeic acid causes metal-dependent damage to cellular and isolated DNA through H2O2 formation. Carcinogenesis 13:1497-502.
Ip, C., C.A. Carter, and M.M. Ip. 1985. Requirement of essential fatty acid for mammary tumorigenesis in the rat. Cancer Res. 45:1997-2001.
Ip, C., S.F. Chin, J.A. Scimeca, and M.W. Pariza. 1991. Mammary cancer prevention by conjugated dienoic derivative of linoleic acid. Cancer Res. 51:6118-6124.
Ip, C., M. Singh, H.J. Thompson, and J.A. Scimeca. 1994. Conjugated linoleic acid suppresses mammary carcinogenesis and proliferative activity of the mammary gland in the rat. Cancer Res. 54:1212-1215.
Ito, N., and M. Hirose. 1987. The role of antioxidants in chemical carcinogenesis. Jpn. J. Cancer Res (Gann) 78:1011-1026.
Ito, N., and M. Hirose. 1989. Antioxidants—Carcinogenic and chemopreventive properties. Adv. Cancer Res. 53:247-302.
Ito, N., R. Hasegawa, M. Sano, S. Tamano, H. Esumi, S. Takayama, and T. Sugimura. 1991. A new colon and mammary carcinogen in cooked food, 2-amino-1-methyl-6-phenylimidazo[4,5-b]-pyridine (PhIP). Carcinogenesis 12:1503-1506.
Iverson, F. 1989. A toxicological perspective on the 1987 domoic acid mussel poisoning incident in Eastern Canada. Pp. 253-268 in 1989 Annual Summer Toxicology Forum. Washington, DC: Toxicology Forum.
James, S.J., D.R. Cross, and B.J. Miller. 1992. Alterations in nucleotide pools in rats fed diets deficient in choline, methionine and/or folic acid. Carcinogenesis 13:2471-2474.
Jaskiewicz, K., W.F.O. Marasas, J.E. Rossouw, F.E. Van Niekerk, and E.W. Heine Tech. 1988. Selenium and other mineral elements in populations at risk for esophageal cancer. Cancer 62:2635-2639.
Jenner, A. 1973. Food: Fact and Folklore. Pp. 1-37. Toronto:
McClelland and Stewart, Ltd.
Kalengayi, M.M.R., G. Ronchi, and V.J. Desmet. 1975. Histochemistry of gamma-gultamyl transpeptidase in rat liver during aflatoxin B1-induced carcinogenesis. J. Natl Cancer Inst. 55:579-588.
Klein, M. 1962. Induction of lymphocytic neoplasms, hepatomas, and other tumors after oral administration of urethan in infant mice. J. Nat. Cancer Inst. 29:1035-1046.
Klein, M. 1966. Influence of age on induction with urethan of hepatomas and other tumors in infant mice. J. Nat. Cancer Inst. 36:1111-1120.
Knekt, P., A. Reunanen, H. Takkunen, A. Aromaa, M. Helivaara, and T. Hakulinen. 1994. Body iron stores and risk of cancer. Int. J. Cancer 56:379-382.
Kolonel, L.N. and L. Le Marchand. 1986. The epidemiology of colon cancer and dietary fat. Pp. 69-91 in Progress in Clinical and Biological Research, Vol. 222: Dietary Fat and Cancer, C. Ip, D. Birt, A.E. Rogers, and C. Mettlin, eds. New York: Alan R. Liss, Inc.
Kolonel, L.N., C.N. Yoshizawa, and J.H. Hankin. 1988. Diet and prostatic cancer: A case-control study in Hawaii. Am. J. Epidemiol. 127:999-1012.
Kowalewski, K., and E.F. Todd. 1971. Carcinoma of the gallbladder induced in hamsters by insertion of cholesterol pellets and feeding dimethylnitrosamine. Proc. Soc. Exp. Biol. Med. (NY) 136:482-486.
Krinsky, N.I. 1991. Effects of carotenoids in cellular and animal systems. Am. J. Clin. Nutr. 53:238S-246S.
Kritchevsky, D. 1993. Undernutrition and chronic disease: Cancer. Proc. Nutr. Soc. 52:39-47.
Kroto, H.W., J.R. Heath, S.C. O'Brien, R.F. Curl, and R.E. Smalley. 1985. C60: Buckminsterfullerene. Nature 318:162-163.
Lane, H.W., J.S. Butel, C. Howard, F. Shepherd, R. Halligan, and D. Medina. 1985. The role of high levels of dietary fat in 7,12-dimethyl-benzanthracene-induced mouse mammary tumorigenesis: Lack of an effect on lipid peroxidation. Carcinogenesis 6:403-407.
Laranjinha, J.A., L.M. Almeida, and V.M. Madeira. 1992. Lipid peroxidation and its inhibition in low density lipoproteins: quenching of cis-parinaric acid fluorescence. Archives Biochem Biophys
297:147-154.
Laranjinha, J.A., L.M. Almeida, and V.M. Madeira. 1994. Reactivity of dietary phenolic acids with peroxyl radicals: antioxidant activity upon low density lipoprotein peroxidation. Biochem Pharmacol. 48:487-94.
Le Marchand, L., C.N. Yoshizawa, L.N. Kolonel, J.H. Hankin, and M.T. Goodman. 1989. Vegetable consumption and lung cancer risk: A population-based case-control study in Hawaii. JNCI 81:1158-1164.
Le Marchand, L., L.N. Kolonel, L.R. Wilkens, B.C. Myers, and T. Hirohata. 1994. Animal fat consumption and prostate cancer: A prospective study in Hawaii. Epidemiol. 5:276-82.
Le Page, R.N., and G.S. Christie. 1969a. Induction of liver tumours in the rabbit by feeding dimethylnitrosamine. Brit. J. Cancer. 23:125.
Le Page, R.N., and G.S. Christie. 1969b. Induction of liver tumours in the guinea pig by feeding dimethylnitrosamine. Pathology 1:49.
Li, Y., and M.A. Trush. 1994. Reactive oxygen-dependent DNA damage resulting from the oxidation of phenolic compounds by a copper-redox cycle mechanism. Cancer Research 54(Suppl 7):1895s-1898s.
Li, P., H.Z. Wang, X.Q. Wang, and Y.N. Wu. 1994. The blocking effect of phenolic acid on N-nitrosomorpholine formation in vitro. Biomed. Environ. Sci. 7:68-78.
Lijinsky, W. and A.E. Ross. 1967. Production of carcinogenic polynuclear hydrocarbons in the cooking of food . Food Cosmet. Toxicol. 5:343-347
Lok, E., E.A. Nera, F. Iverson, F. Scott, Y. So, and D.B. Clayson. 1988. Dietary restriction, cell proliferation, and carcinogenesis: A preliminary study. Cancer Letters 38:249-255.
Lok, E., W.M. Ratnayake, F.W. Scott, R. Mongeau, S. Fernie, E.A. Nera, S. Malcolm, E. McMullen, P. Jee, and D.B. Clayson. 1992. Effect of varying the type of fat in a semi-purified AIN-76A diet on cellular proliferation in the mammary gland and intestinal crypts in female Swiss Webster mice. Carcinogenesis 13(10):1735-1741.
Lok, E., F.W. Scott, R. Mongeau, E.A. Nera, S. Malcolm, and D.B. Clayson. 1990. Calorie restriction and cellular proliferation in various tissues of the female Swiss Webster mouse. Cancer Letters
51:67-73.
Lubin, F., Y. Wax, and B. Modan. 1986. Role of fat, animal protein, and dietary fiber in breast cancer etiology: A case-control study. J. Natl. Cancer Inst. 77:605-612.
Macquart-Moulin, G., E. Riboli, J. Cornee, B. Charnay, P. Berthezene, and N. Day. 1986. Case-control study on colorectal cancer and diet in Marseilles. Int. J. Cancer 38:183-191.
Magee, P.N., and J.M. Barnes. 1956. The production of malignant primary hepatic tumours in the rat by feeding dimethylnitrosamine. Brit. J. Cancer 10:114.
Magee, P.N., and J.M. Barnes. 1959. The experimental production of tumours in the rat by dimethylnitrosamine (N-nitroso-dimethylamine). Acta. Un. Int. Cancer 15:187.
Magee, P.N., and J.M. Barnes. 1962. Induction of kidney tumours in the rat with dimethylnitrosamine (N-nitroso-dimethylamine). J. Path. Bact. 84:19.
Maarse, H., and C.A. Visscher, eds. 1989. Volatile Compounds in Foods. Qualitative and Quantitative Data. The Netherlands: TNOCIVO Food Analysis Institute.
Mattocks, A.R. 1986. Chemistry and toxicology of pyrrolizidine alkaloids. Orlando, FL: Academic Press. 393 pp.
Medina, D. 1986. Selenium and murine mammary tumorigenesis. Pp. 23-41 in Diet, Nutrition, and Cancer: A Critical Evaluation, B.S. Reddy and L.A. Cohen, ed. Boca Raton, FL: CRC Press, Inc.
Meinwald, J., and T. Eisner. 1995. The chemistry of phyletic dominance. Proc. Nat. Acad. Of Sci. 92(1):14-18.
Miller, C.C., Y. Park, M.W. Pariza, and M.E. Cook. 1994. Feeding conjugated linoleic acid to animals partially overcomes catabolic responses due to endotoxin injection. Biochem. Biophys. Res. Commun. 198:1107-1112.
Minghetti, P.P. and A.W. Norman. 1988. 1,25(OH)2-vitamin D3 receptors: Gene regulation and genetic circuitry. FASEB J. 2:3043-3053.
Mirvish, S.S. 1983. The etiology of gastric cancer: Intragastric nitrosamide formation and other theories. J. Natl. Cancer Inst. 71:629-647.
Moon, R.C. 1989. Comparative aspects of carotenoids and retinoids as
chemopreventive agents for cancer. J. Nutr. 119:127-134.
Moore, M.R., H.C. Pitot, E.C. Miller, and J.A. Miller. 1982. Cholangiocellular carcinomas induced in Syrian golden hamsters administered aflatoxin B1 in large doses. J. Natl Cancer Inst. 68:271-278.
Morgan, E.D. 1984. Insect Communication (pp. 169-194), Lewis, T., ed. New York: Academic Press.
Nagao, M., and T. Sugimura. 1993. Carcinogenic factors in food with relevance to colon cancer development. Mutat. Res. 290:43-51.
Nakayama, T. 1994. Suppression of hydroperoxide-induced cytotoxicity by polyphenols. Cancer Research 54(Suppl 7):1991s-1993s.
Nawar, W.W. 1985. Lipids. Pp. 139-244 in Food Chemistry, 2nd edition, O.R. Fennema, ed. New York: Marcel Dekker.
Neugut, A.I., G.C. Garbowski, W.C. Lee, T. Murray, J.W. Nieves, K.A. Forde, M.R. Treat, J.D. Waye, and C. Fenoglio-Preiser. 1993. Dietary risk factors for incidence and recurrence of colorectal adenomatous polyps . Ann. Intern. Med. 118:91-95.
Newberne, P.M., and M. Locniskar. 1990. Roles of micronutrients in cancer prevention: Recent evidence from the laboratory. Prog. Clin. Biol. Res. 346:119-134.
Newmark, H.L. and M. Lipkin. 1992. Calcium, vitamin D, and colon cancer. Cancer Res. 52(Suppl.):2067s-2070s.
Nishizumi, M., R.E. Albert, F.J. Burns, and L. Bilger. 1977. Hepatic cell loss and proliferation induced by N-2-fluorenylacetamide, diethylnitrosamine, and aflatoxin B1 in relation to hepatoma induction. Br. J. Cancer 36:192-197.
NRC (National Research Council). 1975. Underexploited Tropical Plants with Promising Economic Value (pp. 1-8). Washington, DC: National Academy Press.
NRC (National Research Council). 1977. Arsenic. Washington, DC: National Academy Press.
NRC (National Research Council). 1981. The Health Effects of Nitrite, Nitrate, and N-Nitroso Compounds. Washington, DC: National Academy Press.
NRC (National Research Council). 1982. Diet, Nutrition and Cancer. Washington, DC: National Academy Press.
NRC (National Research Council). 1988. Designing foods. Animal Product Options in the Marketplace. Washington, DC: National Academy Press. 367 pp.
NRC (National Research Council). 1989a. Diet and Health: Implications for Reducing Chronic Disease Risk. Food and Nutrition Board, Committee on Diet and Health. Washington, DC: National Academy Press. 749 pp.
NRC (National Research Council). 1989b. Dietary intake and nutritional status: trends and assessment. Pp. 41-84 in Diet and Health: Implications for Reducing Chronic Disease Risk, Food and Nutrition Board, ed. Committee on Diet and Health. Washington, DC: National Academy Press.
NRC (National Research Council). 1989c. Recommended Dietary Allowances, 10th edition. Washington, DC: National Academy Press, p. 44.
NTP (National Toxicology Program). 1994. Seventh Annual Report on Carcinogens 1994-Summary. National Toxicology Program, Research Triangle Park, NC, and Bethesda, MD.
Ochiai, M., K. Ogawa, K. Wakabayashi, T. Sugimura, S. Nagase, H. Esumi, and M. Nagao. 1991. Induction of intestinal adenocarcinomas by 2-amino-1-methyl6-phenylimidazo[4,5-b]pyridine in Nagase analbuminemic rats. Jpn. J. Cancer Res. 82:363-366.
Otsuka, H., and A. Kuwahara. 1971. Hemangiomatous lesions of mice treated with nitrosodimethylamine. Gann 62:147.
Overvad, K., D.Y. Wang, J. Olsen, D.S. Allen, E.B. Thorling, R.D. Bulbrook, and J.L. Hayward. 1991. Selenium in human mammary carcinogenesis: A case-cohort study. Eur. J. Cancer 27:900-902.
Paracelsus (Philippus Theophrastus Aureolus Bobastus von Hohenheim. 1564. Third defense in Septem Defensiones. Arnold Bryckmann. Cologne.
Pariza, M.W. 1988. Dietary fat and cancer risk: Evidence and research needs. Annu. Rev. Nutr. 8:167-183.
Pariza, M.W., and R.K. Boutwell. 1987. Historical perspective: Calories and energy expenditure in carcinogenesis. Am. J. Clin. Nutr. 45(Suppl):151-156.
Parkin, D.M., P. Srivatanakul, M. Khlat, D. Chenvidhya, P. Chotiwan,
S. Insiripong, S., K.A. L'Abbé, and C.P. Wild. 1991. Liver cancer in Thailand. I. A case-control study of cholangiocarcinoma. Int. J. Cancer 48:323-328.
Peers, F.G., X. Bosch, J. Kaldor, C.A. Linsell, and M. Pluijmen. 1987. Aflatoxin exposure, hepatitis B virus infection and liver cancer in Swaziland. Int. J. Cancer 39:545-553.
Pelletier, S.W. 1983-1992. Alkaloids: Chemical and Biological Properties. Vols. 1-6. New York: Wiley.
Pitt, J.I., A.D. Hocking, K. Bhudasamai, B.F. Miscamble, K.A. Wheeler, and P. Tanbook-Ek. 1993. The normal mycoflora of commodities from Thailand. I. Nuts and oilseeds. Int. J. Food Microbiol. 20:211-226.
Pohan, T.E., G.R. Howe, C.M. Friedenrich, M. Jain, and A.B. Miller. 1993. Dietary fiber, vitamins A, C, and E, and risk of breast cancer: A cohort study. Cancer Causes and Control 4:29-37.
Potter, J.D. and A.J. McMichael. 1986. Diet and cancer of the colon and rectum: A case-control study. NJCI 76:557-569.
Prival, M.J. 1985. Carcinogens and mutagens present as natural components of food or induced by cooking. Nutrition and Cancer 6:236-253.
Pyke, M. 1968. Food and Society. London: John Murray. 178 p.
Qian, G.S., R.K. Ross, M.C. Yu, J.M. Yuan, Y.T. Gao, B.E. Henderson, G.N. Wogan, and J.D. Groopman. 1994. A follow-up study of urinary markers of aflatoxin exposure and liver cancer risk in Shanghai, People's Republic of China. Cancer Epidemiol. Biomarkers Prev. 3:3-10.
Ray, J.S., M.L. Harbison, R.M. McClain, and J.I. Goodman. 1994. Alterations in the methylation status and expression of the raf oncogene in phenobarbital-induced and spontaneous B7C3F1 mouse liver tumors. Molecular Carcinogenesis 9:155-166.
Riopelle, J.L., and G. Jasmin. 1969. Nature, classification and nomenclature of kidney tumors induced in the rat by dimethylnitrosamine. J. Nat. Cancer Inst. 42:643.
Roberts, A.B., and M.B. Sporn. 1992. Mechanistic interrelationships between two superfamilies: The steroid/retinoid receptors and transforming growth factor-beta. Cancer Surv. 14:205-220.
Rodricks, J.V., and A.E. Pohland. 1981. Food Hazards of Natural
Origin Pp. 181-237 in Food Safety, H.R. Roberts, ed. New York: Wiley.
Ross, R.K., J.M. Yuan, M.C. Yu, G.N. Wogan, G.S. Qian, J.T. Tu, J.D. Groopman, Y.T. Gao, and B.E. Henderson. 1992. Urinary aflatoxin biomarkers and risk of hepatocellular carcinoma. Lancet 339:943-946.
Rozin, E. 1973. The Flavor Principle Cookbook. New York: Hawthorn Books, Inc.
Safe, S.H. 1995. Environmental and dietary estrogens in human health: Is there a problem. Environmental Health Perspectives 103:346-351.
Scalmati, A., M. Lipkin, and H. Newmark. 1992. Relationships of calcium and vitamin D to colon cancer. Pp. 249-262 in Cancer Chemoprevention, L. Wattenberg, M. Lipkin, C.W. Boone, and G.J. Kelloff, eds. Boca Raton, FL: CRC Press.
Schectman, G. 1993. Estimating ascorbic acid requirements for cigarette smokers. Ann. NY Acad. Sci. 686:335-345.
Schmähl, D., and R. Preussmann. 1959. Cancerogene Wirkung von Nitrosodimethylamin bei Ratten. Naturwissenschaften 46:175.
Schwartz, A.G. and L.L. Pashko. 1994. Role of adrenocortical steroids in mediating cancer-preventive and age-retarding effects of food restriction in laboratory rodents. J. Gerontol. 49:B37-B41.
Seitz, H.K., and U.A. Simanowski. 1988. Alcohol and carcinogenesis. Annu. Rev. Nutr. 8:99-119.
Shabad, L.M., and L.A. Savluchinskaya. 1971. Some results of studying the blastomogenic action of nitrosamines on mice. Biull. Ėksp. Biol. Med. 71:76.
Shinohara, Y., M. Arai, K. Hirao, S. Sugihara, K. Nakanishi, H. Tsunoda, and N. Ito. 1976. Combination effect of citrinin and other chemicals on rat kidney tumorigenesis. Gann 67:147-155.
Sieber, S.M., P. Correa, D.W. Dalgard, and R.H. Adamson. 1979. Induction of osteogenic sarcomas and tumors of the hepatobiliary system in nonhuman primates with aflatoxin B. Cancer Res. 39:4545-4554.
Simic, M.G., and M. Karel. 1980. Autoxidation in food and biological systems. New York: Plenum Press. 659 pp.
Slattery, M.L., A.W. Sorenson, A.W. Mahoney, T.K. French, D.
Kritchevsky, and J.C. Street. 1988. Diet and colon cancer: Assessment of risk by fiber type and food source. J. Natl. Cancer Inst. 80:1474-1480.
Soffritti, M., and E.E. McConnell. 1988. Liver foci formation during aflatoxin B1 carcinogenesis in the rat. Ann. N.Y. Acad. Sci. 534:531-540.
Srivatanakul, P., D. M. Parkin, Y.-Z. Jiang, M. Khlat, U.-T. Kao-Ian, S. Sontipong, and C.P. Wild. 1991a. The role of infection by Opisthorchis viverrini, hepatitis B virus, and aflatoxin exposure in the etiology of liver cancer in Thailand. A correlation study. Cancer 68:2411-2417.
Stähelin, H.B., K.F. Gey, M. Eichholzer, E. Lüdin, F. Bernasconi, J. Thurneysen, and G. Brubacher. 1991. Plasma antioxidant vitamins and subsequent cancer mortality in the 12-year follow-up of the prospective basel study. Am. J. Epidemiol. 133:766-775.
Stich, H.F. 1991. The beneficial and hazardous effects of simple phenolic compounds. Mutat. Res. 259:307-324.
Stich, H.F. 1992. Teas and tea components as inhibitors of carcinogen formation in model systems and man. Preventive Medicine 21:377-84.
Stryer, L. 1975. Biochemistry. San Francisco: W.H. Freeman and Company. 877 p.
Sugimura, T. 1985. Carcinogenicity of mutagenic heterocyclic amines formed during the cooking process. Mutat. Res. 150:33-41.
Sugimura, T., M. Nagao, and K. Wakabayashi. 1994. Heterocyclic amines in cooked foods: Candidates for causation of common cancers . Journal of the National Cancer Institute 86:2-4.
Takahashi, M., K. Toyoda, Y. Aze, K. Furuta, K. Mitsumori, and Y. Hayashi. 1993. The rat urinary bladder as a new target of heterocyclic amine carcinogenicity: Tumor induction by 3-amino-1-methyl-5H-pyrido[4,3-b]indole acetate. Jpn. J. Cancer Res. 84:852-858
Takayama, S., and K. Oota. 1965. Induction of malignant tumours in various strains of mice by oral administration of N-nitrosodimethylamine and N-nitrosodiethylamine. Gann 56:189.
Tanaka, T., T. Kojima, T. Kawamori, A. Wang, M. Suzui, K. Okamoto, and H. Mori. 1993. Inhibition of 4-nitroquinoline-1-oxide-induced
rat tongue carcinogenesis by the naturally occurring plant phenolics caffeic, ellagic, chlorogenic and ferulic acids. Carcinogenesis 14:1321-5.
Tannahill, R. 1973. Food in History. New York: Stein and Day. 448p.
Tannenbaum, S.R., J.S. Wishnok, and C.D. Leaf. 1991. Inhibition of nitrosamine formation by ascorbic acid . Am. J. Clin. Nutr. 53(Suppl.):247S-250S.
Taylor, H.W., W. Lijinsky, P. Nettesheim, and C.M. Snyder. 1974. Alteration of tumor response in rat liver by carbon tetrachloride. Cancer Res. 34:3391-3395.
Terracini, B., P.N. Magee, and J.M. Barnes. 1967. Hepatic pathology in rats on low dietary levels of dimethylnitrosamine. Brit. J. Cancer 21:559.
Terracini, B., G. Palestro, M. Ramella Gigliardi, and R. Montesano. 1966. Carcinogenicity of dimethylnitrosamine in Swiss mice. Brit. J. Cancer 20:871.
Thompson, L.U. 1993. Potential health benefits and problems associated with antinutrients in foods. Food Res. Internatl. 26:131-149.
Thompson, L.U. 1994. Antioxidants and hormone mediated health benefits of whole grains. Crit. Rev. Food Sci. Nutr. 34:473-497.
Toda, S., M. Kumura, and M. Ohnishi. 1991. Effects of phenolcarboxylic acids on superoxide anion and lipid peroxidation induced by superoxide anion. Planta Med. 57:8-10.
Tomatis, L., P.N. Magee, and P. Shubik. 1964. Induction of liver tumors in Syrian golden hamsters by feeding dimethylnitrosamine. J. Nat. Cancer Inst. 33:341.
Topping, D.C. and W.J. Visek. 1976. Nitrogen intake and tumorigenesis in rats injected with 1,2-dimethylhydrazine. J. Nutrit. 106:1583-1590.
Toth, B., and I. Boreisha. 1969. Tumorigenesis with isonicotinic acid hydrazide and urethan in the Syrian golden hamster. Europ. J. Cancer 5:165-171.
Toth, B., G. Della Porta, and P. Shubik. 1961a. The occurrence of malignant lymphomas in urethan-treated Swiss mice. Brit. J. Cancer 15:322-326.
Toth, B., L. Tomatis, and P. Shubik. 1961b. Multipotential carcinogenesis with urethan in the Syrian golden hamster. Cancer Res. 21:1537-1541.
Toth, B., P.N. Magee, and P. Shubik. 1964. Carcinogenesis study with dimethylnitrosamine administered orally to adult and subcutaneously to newborn BALB/c mice. Cancer Res. 24:1712-1721.
Ueno, Y. 1987. Tricothecenes in Food. Pp. 123-147 in Mycotoxins in Food. P. Krogh, ed. Chapter 6. New York: Academic Press.
Underwood, E.J. 1973. Trace elements. Pp. 48-49 in Toxicants Occurring Naturally in Foods. National Research Council. Washington, DC: National Academy Press.
van den Brandt, P.A., R. A. Goldbohm, P. van't Veer, P. Bode, E. Dorant, R.J.J. Hermus, and F. Sturmans. 1993. A prospective cohort study on selenium status and the risk of lung cancer. Cancer Res. 53:4860-4865.
van't Veer, P., F.J. Kok, H.A.M. Brants, T. Ockhuizen, F. Sturmans, and R.J.J. Hermus. 1990. Dietary fat and the risk of breast cancer. Int. J. Epidemiol. 19:12-18.
Visser, J.H., and A.K. Minks, ed. 1982. Proceedings of the Fifth International Symposium on Insect-Plant Relationships. Wageningen, The Netherlands : Pudoc. 464 pp.
Wald, J., S.G. Thompson, J.W. Densem, J. Boreham, and A. Bailey. 1987. Serum vitamin E and subsequent risk of cancer. Br. J. Cancer 56:69-72.
Watson, D.H. 1985. Toxic fungal metabolites in food. CRC Crit. Rev. Food Sci. Nutr. 22:177-198.
Wattenberg, L.W., J.B. Coccia, and L.K.T. Lam. 1980. Inhibitory effects of phenolic compounds on benzo[a]pyrene-induced neoplasia. Cancer Res. 40:2820-2823.
Weinberg, E.D. 1992. Roles of iron in neoplasia. Promotion, prevention, and therapy. Biol. Trace Elem. Res. 34:123-140.
Welsch, C.W. 1987. Enhancement of mammary tumorigenesis by dietary fat: Review of potential mechanisms. Am. J. Clin. Nutr. 45:192-202.
Welsch, C.W., J.L. House, B.L. Herr, S.J. Eliasberg, and M.A. Welsch. 1990. Enhancement of mammary carcinogenesis by high levels of
dietary fat: A phenomenon dependent on ad libitum feeding. JNCI 82:1615-1620.
Whistler, R.L., and J.R. Daniel. 1985. Carbohydrates. Pp. 69-137 in Food Chemistry, 2nd edition, O.R. Fennema, ed. New York: Marcel Dekker, Inc.
Willett, W.C. 1994. Diet and health: What should we eat? Science 264:532-537.
Willett, W.C., M.J. Stampfer, G.A. Colditz, B.A. Rosner, and F.E. Speizer. 1990. Relation of meat, fat, and fiber intake to the risk of colon cancer in a prospective study among women. N. Engl. J. Med. 323:1664-1672.
Wogan, G.N. 1969. Metabolisma and biochemical effects of aflatoxins. Pp. 151-186 in Aflatoxin. Scientific Background, Control, and Implications, L.A. Goldblatt, ed. New York: Academic Press.
Wogan, G.N. 1992. Aflatoxins as risk factors for hepatocellular carcinomas in humans. Cancer Res. 52:2114S-2118S.
Wogan, G.N., and P.M. Newberne. 1967. Dose-response characteristics of aflatoxin B1 carcinogenesis in the rat. Cancer Res. 27:2370-2376.
Wogan, G.N., G.S. Edwards, and P.M. Newberne. 1971. Structure-activity relationships in toxicity and carcinogenicity of aflatoxins and analogs. Cancer Res. 31:1936-1942.
Yeh, F.-S., J.-C. Yu, C.-C. Mo, S. Luo, M.J. Tong, and B.E. Henderson. 1989. Hepatitis B virus, aflatoxins, and hepatocellular carcinoma in southern Guangxi, China. Cancer Res. 49:2506-2509.
Zak, F.G., J.H. Holzner, E.J. Singer, and H. Popper. 1960. Renal and pulmonary tumors in rats fed dimethylnitrosamine. Cancer Res. 20:96.
Zawirska, B., and W. Bednarz. 1981. The particular traits of carcinogenesis induced in Wistar rats by aflatoxin B1. III. Porphyrins and the activity of gamma-glutamyltranspeptidase in primary hepatomas and in their tissue of origin. Neoplasma 28:35-49.
Zhou, Y.-C., and R.-L. Zheng. 1991. Phenolic compounds and an analog as superoxide anion scavengers and antioxidants. Biochem. Pharmacol. 42:1177-1179.
Zhu, P., E. Frei, B. Bunk, M.R. Berger, and D. Schmahl. 1991. Effect
of dietary calorie and fat restriction on mammary tumor growth and hepatic as well as tumor glutathione in rats. Cancer Lett. 57:145-152.
Ziegler, R.G. 1991. Vegetables, fruits, and carotenoids and the risk of cancer. Am. J. Clin. Nutr. 53(Suppl.):251S-259S.
Ziegler, R.G., L.A. Brinton, R.F. Hamman, H.F. Lehman, R.S. Levine, K. Mallin, S.A. Norman, J.F. Rosenthal, A.C. Trumble, and R.N. Hoover. 1990. Diet and the risk of invasive cervical cancer among white women in the United States. American J. Of Epidemiology 132(3):432-445.
Zwicker, G.M., W.W. Carlton, and J. Tuite. 1972. Carcinogenic activity of toxigenic penicillia (Abstract). Lab. Invest. 26:497.