B12 Toluene
Hector D. Garcia, Ph.D.
Johnson Space Center Toxicology Group
Biomedical Operations and Research Branch
Houston, Texas
Physical and Chemical Properties
Toluene is a clear, colorless, non-corrosive, flammable liquid with a sweet, pungent, "aromatic" odor. Values reported for the odor threshold range from 0.2 to 16 ppm (Sandmeyer, 1981).
|
Synonyms: |
Antisal, phenyl methane, methacide, methyl benzene, methylbenzol, NCI-C07272, tolueen, toluen, toluol, toluolo, tolu-sol, UN 1294 |
|
Formula: |
C7H8 |
|
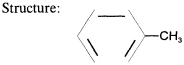
|
|
|
CAS number: |
108-88-3 |
|
Molecular weight: |
92.14 |
|
Boiling point: |
110.62°C |
|
Melting point: |
-95°C |
|
Liquid density: |
0.8869 |
|
Vapor pressure: |
36.7 mm Hg at 30°C |
|
Solubility: |
Insoluble in water Very soluble in alcohol and ether |
|
Conversion factors at 25°C, 1 atm: |
1 ppm = 3.77 mg/m3 1 mg/m3 = 0.265 ppm |
Occurrence and Use
Toluene has been measured in urban air at 0.01-0.05 ppm, probably stemming from production facilities, automobile and coke-oven emissions, gasoline evaporation, and cigarette smoke, and can occur in human respiratory air in smokers and nonsmokers (Sandmeyer, 1981). It is used extensively as a component of gasoline, as a solvent in the chemical, rubber, paint, and drug industries, as a thinner for inks, perfumes and dyes, and as a nonclinical thermometer liquid and suspension solution for navigational instruments (Sandmeyer, 1981). Intentional inhalation of toluene vapors from glue was popular among some youth during the last few decades because of toluene's effects on the central nervous system (CNS). Toluene has been detected in spacecraft air in numerous missions at levels of up to 64 ppm.
Pharmacokinetics and Metabolism
Absorption
The major route of absorption of toluene is by inhalation. During inhalation, arterial blood concentrations of toluene in humans reach 60% of maximum in 10-15 min (Benignus, 198 1a). A linear relationship was found between toluene concentrations in alveolar air and arterial blood in human subjects exposed to toluene at 100, 300, 500, and 714 ppm for 20 min per concentration (Gamberale and Hultengren, 1972). Reports of uptake rates (to 95% of asymptote) in humans exposed at up to 500 ppm vary from 10 to 80 min (Gamberale and Hultengren, 1972; Veulemans and Masschelein, 1978; Benignus, 1981a). Exercise increases the rate of uptake (Benignus, 1981a). Retention, including from cigarette smoke, was 86-96% (Sandmeyer, 1981). In mice, however, Peterson and Bruckner (1978) reported that the arterial blood concentration did not approach maximum values until about 2 h after the onset of exposure to toluene at 4000 ppm and was still rising slightly after 3 h.
Distribution
Inhaled toluene is distributed widely throughout the body, most rap-
idly into highly vascularized tissues, with fatty tissues acting as reservoirs. During exposure, the concentrations of toluene are highest in the liver, then the brain and blood (Bruckner and Peterson, 1981a).
Excretion
The majority of inhaled toluene is exhaled unchanged. Toluene concentrations fall off rapidly after cessation of exposure, dropping to 30% of maximum in humans in 40 min and near zero levels in 4 h when subjects are at rest (Benignus et al., 1981). In rats, toluene blood concentrations fall to 50% of maximum about 60 min after termination of exposure (Benignus, 1981b). In humans, about 80-85% of total toluene is excreted in the urine as conjugates of benzoic acid. Of this, about 80% is hippuric acid (the glycine conjugate of benzoic acid) and 20% is benzoylglucuronide (the glucuronic acid conjugate of benzoic acid). In 23 male volunteers inhaling toluene vapor for 3 h or 7 h (with a break of 1 h), the excretion of urinary hippuric acid over an 18h period was equivalent to 68% of the toluene absorbed (Ogata et al., 1971). In humans exposed to toluene at up to 200 ppm, the total amount of hippuric acid excreted was proportional to the total exposure (ppm x h) (Ogata et al., 1971).
Metabolism
Toluene is metabolized in several ways, but mainly it is converted by oxidation of the methyl group to benzoic acid via benzyl alcohol and benzaldehyde in the microsomes of the liver parenchymal cells (Benignus et al., 1981). Benzoic acid is conjugated with glycine to form hippuric acid (80%) and with glucuronic acid to form benzoylglucuronide (20%), which are excreted in the urine (Benignus et al., 1981). Possible ethnic differences in toluene metabolism were reported for Chinese, Turkish, and Japanese solvent workers; the male Japanese excreted almost twice as much hippuric acid as the male Chinese under similar exposure conditions, although the difference was less marked between female Chinese and Japanese workers, and there were no differences in the excretion of o-cresol (Inoue et al., 1986).
Toxicity Summary
Acute and Short-Term Toxicity
Lethality
The LC50 in Fischer rats for a 60-min inhalation exposure to toluene vapors is 26,700 ppm, and the LC100 for a 60-min inhalation exposure is about 40,000 ppm (Pryor et al., 1978). During inhalation studies in which continuous exposure and fixed concentrations of toluene vapor were used, the LC50 was a function of both the concentration of toluene and the duration of exposure. A group of pregnant rats (n = 9) exposed to toluene at 400 ppm continuously for 8 d had a 28% mortality, but no mortality was seen at 266 ppm (n = 10) (Hudák and Ungváry, 1978). A later publication from the same laboratory indicated that rats could be exposed to toluene at 950 ppm for 48 h, starting on d 10 of pregnancy without significant maternal mortality (Ungváry et al., 1982).
Irritation and CNS Effects
In 16 volunteers, 21-32 y old, exposure to toluene at 40 ppm for 6 h has been shown to be a no-observed-adverse-effect level (NOAEL) for irritation and CNS effects, including vigilance, visual perception, psychomotor functions, and higher cortical functions (Andersen et al., 1983). In the same study, 100 ppm was found to cause slight irritation of eyes and nose and increased headaches, dizziness, and a feeling of intoxication; in another study (Gamberale and Hultengren, 1972), 100 ppm was found to be a NOAEL for impairment of reaction time.
In a Danish study, 20 printers previously occupationally exposed for 9-25 y to a mixture of solvents containing toluene at 0-20% and 22 naive control subjects were exposed to toluene at 100 ppm for 6 h. All 42 exposed subjects complained of low air quality, strong odor, fatigue, sleepiness, a feeling of intoxication, and irritation of the eyes, nose, and throat. The exposed naive control subjects showed a statistically significant 1 % decrease in manual dexterity, decreased color discrimination, and slightly decreased accuracy in visual perception compared with unexposed naive controls (Bælum et al., 1985).
Male rats and mice exposed to toluene at 12,000 ppm for 5 min exhibited marked depression, but recovered fully after breathing fresh air for 10 min (Bruckner and Peterson, 1981a). A study by Guillot et al. (1982) reported that a 1-h exposure to liquid toluene using several protocols irritated rabbits' eyes, but the irritation was reduced to slight irritation if the eyes were rinsed 30 s after instillation.
Subchronic and Chronic Toxicity
Histopathology, Clinical Chemistry, and Hematology
A well-executed chronic inhalation study in F344 rats performed by the Chemical Industry Institute of Toxicology in 1980 failed to show any adverse effects at the doses tested (Gibson et al., 1983). Groups of 120 male and 120 female F344 rats were exposed to toluene at 30, 100, or 300 ppm (> 99.98% pure), 6 h/d, 5 d/w, for 24 mo. An unexposed group of 120 male and 120 female rats served as a control. Clinical chemistry, hematology, and urinalysis tests were conducted at 18 and 24 mo. All parameters measured at the termination of the study were normal except for a dose-related reduction in hematocrit values in females exposed to toluene at 100 and 300 ppm. Forty-one tissues from each animal (5-76 animals per group) sacrificed at 6, 12, 18, and 24 mo were examined grossly and histopathologically. The authors considered the highest dose of 300 ppm to be a NOAEL.
Hepatotoxicity and Nephrotoxicity
Evidence for liver and kidney toxicity by toluene is weak and inconsistent. Rats exposed to toluene at 5000 ppm for 7 h/d for 25 exposures showed reversible increases in kidney and liver weights (Von Oettingen, 1942). Mice exposed at 40,000 ppm for 3 h/d for 40 exposures and rats exposed at 1000 ppm for 6 h/d for 65 exposures showed no evidence of kidney or liver damage. Clinical case reports on humans who have either accidentally or intentionally been exposed to long durations or high levels or both of toluene also show reversible kidney and liver pathology symptoms, but the reports are not consistent in their findings of pathology (Benignus, 1981a).
CNS Effects
Chronic inhalation abuse of pure toluene produces irreversible cerebellar, brain-stem, and pyramidal-tract dysfunction (Spencer and Schaumberg, 1985), but comparable changes have not been found in solvent workers (n = 43) occupationally exposed for 22 y (SD = 7.4) to toluene at a mean concentration of 117 ppm (Bælum et al., 1985; Juntunen et al., 1985).
Rats exposed to toluene at 1000 ppm, 6 h/d, 5 d/w, for 30 d exhibited a small but significant alteration in brain function (flash-evoked potentials) 18-26 h after the last exposure (Dyer et al., 1984).
Lethality
In an extensive study conducted by the National Toxicology Program (NTP) on nonpregnant animals, mice (n = 120) and rats (n = 100) exposed to toluene at 1200 ppm for 2 y had no significant differences in survival compared with unexposed controls (Huff, 1990).
Carcinogenicity
In a 1989 NTP study (Huff, 1990), groups of 60 male and 60 female F344 rats were exposed to toluene at 0, 600, or 1200 ppm and 60 male and 60 female B6C3F1 mice were exposed at 0, 120, 600, or 1200 ppm for 6.5 h/d, 5 d/w, for 2 y. At 15 mo, an interim sacrifice revealed an increased incidence and severity of non-neoplastic lesions of the nasal cavity of exposed rats. Minimal hyperplasia of the bronchial epithelium was seen in 4 of 10 female mice at 1200 ppm. There were also effects on the olfactory and respiratory epithelia of exposed rats. There was no evidence of carcinogenicity for any group of animals in this study.
Genotoxicity
Chromosome studies on peripheral blood lymphocytes of 34 workers of a rotogravure plant found no significant differences between toluene-
exposed workers and a group of 34 matched controls (Forni et al., 1971). A similar study found no increases in the frequencies of sister chromatid exchanges (SCEs) or chromosomal aberrations (CAs) (Maki-Paakkanen et al., 1980). No increases in SCEs or CAs were found in vitro in toluene-treated human lymphocytes (Gerner-Smidt and Friedrich, 1978). Toluene was found to be nonmutagenic in the Ames assay (Bos et al., 1981). Toluene was found to be a potent mitotic arrestant in the grasshopper embryo system, but it did not induce an accumulation of colchicine-like mitoses (Liang et al., 1983).
Reproductive Toxicity
Wives (n = 28) of men with high or frequent occupational exposure to toluene had an increased odds ratio of spontaneous abortion compared with 29 referents (controls), but there was no association with congenital malformations (Taskinen et al., 1989). No increases were seen for intermediate or low or rare exposures.
A study examining occupational exposure to chemicals in the workplace compared 301 working women who had recently given birth to a child with congenital defects with 301 matched working women whose most recent child was born normal (McDonald et al., 1987). The authors' analysis indicated that, of nine categories of chemical exposures examined, only in those women exposed to aromatic solvents, primarily toluene, were there a suspicious excess number of cases of defects. Six mothers of children with congenital defects were exposed to toluene and two of these were also exposed to other solvents. The authors state that the statistical significance of this result (Z = 1.77; one sided p ≈ 0.04) might be an overestimate.
Developmental Toxicity
There are conflicting reports of the teratogenicity of toluene. Exposure to toluene at 399 ppm for 24 h/d during various portions of pregnancy was not teratogenic to rats, but there was considerable retardation of fetal development (Hudák and Ungváry, 1978). In mice, however, exposure at 400 ppm from gestation days 6-15 indicated teratogenicity (a significant shift in the rib profile) but no fetotoxicity (Courtney et
al., 1986). Continuous exposure at 266 ppm from gestation days 6-15 caused spontaneous abortion in rabbits, while maternal-weight gain decreased (Ungváry and Tátrai, 1985). Toluene was found to be negative in the Chernoff/Kavlock developmental toxicity screen in mice (Taskinen et al., 1989).
The offspring of pregnant mice given toluene at 400 ppm in drinking water and maintained at that concentration after weaning showed a decreased habituation or open-field activity at 35 d of age and depressed rotorod performance at 45-55 d of age, but no change in surface-righting response (Kostas and Hotchin, 1981).
Interaction with Other Chemicals
Toluene and trichloroethylene have been shown to noncompetitively inhibit each other's metabolism in rats. Trichloroethylene suppresses the urinary excretion of hippuric acid, a main metabolite of toluene, and toluene reduces the amount of urinary total trichloro-compounds, the metabolites of trichloroethylene (Ikeda, 1974). Ikeda also reported suppression of benzene and styrene oxidation in vivo by co-administration of toluene in rats (Ikeda et al., 1972). Benzene and toluene mutually inhibit each other's metabolism, with toluene more effectively inhibiting benzene metabolism than the reverse (Purcell et al., 1990). A drastic increase in toluene concentration in the blood of rats was reported after combined inhalation with acetone (Freundt and Schneider 1986). Inhalation of toluene at 954 ppm by pregnant rats combined with acetylsalicylic acid at 500 mg/kg resulted in increased maternal and fetal toxicity and teratogenicity (Tátrai et al., 1979).
Simultaneous exposure of human volunteers to toluene and ethanol has been shown to reduce the urinary excretion of hippuric acid and o-cresol, metabolites of toluene, to less than half of the value for exposure to toluene alone (Døssing et al., 1984).
TABLE 12-1 Toxicity Summary
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
7-112 |
Occupational (rotogravure) |
Human (n = 32) |
NOAEL for SCE and chromosome aberrations |
Maki-Paakkanen et al., 1980 |
|
40 |
6 h/d, 4 d |
Human (n = 16), ages 21-32 y |
NOAEL for irritation of eyes and nose, lung function, nasal mucus flow, visual perception, vigilance, psychomotor functions, higher cortical functions |
Andersen et al., 1983 |
|
100 |
6.5 h |
Human (n= 41), ages 29-50 y |
Strong odor, fatigue, sleepiness, inebriation; irritation of eyes, nose, and throat; decreased manual dexterity, decreased color discrimination; decreased accuracy in visual perception |
Bælum et al., 1985 |
|
100 |
6 h/d. 4 d |
Human (n = 16), ages 21-32 |
Odor; irritation of eyes and nose; NOAEL for lung function, nasal mucus flow, visual perception, vigilance, psychomotor functions, higher cortical functions |
Andersen et al., 1983 |
|
117 |
Occupational. 22 y |
Human (n = 43) |
NOAEL for neurological functions, neuroanatomical changes, and neurophysiology |
Juntunen et al., 1985 |
|
''High'' |
Occupational. frequent |
Human (n = 301) |
Congenital defects in children |
McDonald et al., 1987 |
|
70 |
18 h |
Monkey (n = 1) |
Reduced shock avoidance |
Weiss et al., 1979 |
|
100 |
6 h/d, 4 d |
Human (n = 12), ages 20-35 |
NOAEL for impairment of reaction time |
Gamberale et al., 1972 |
|
300 |
6 h/d, 4 d |
Human (n = 12), ages 20-35 |
Impairment of reaction time |
Gamberale et al., 1972 |
|
300 |
6 h/d, 5 d/w, 24 mo |
Rat |
NOAEL for histopathology |
Gibson et al., 1983 |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
400 |
4 h |
Pigeon |
Increased rate of key pecking |
Weiss et al., 1979 |
|
500 |
6 h/d, 5 d/w, 6 mo |
Rat |
Increased brain-cell volume; altered neurotransmitter levels |
Ladefoged et al., 1991 |
|
1000 |
1 h |
Monkey |
Self-administration of toluene (toluene "sniffing") |
Weiss et al., 1979 |
|
1000 |
8 h/d, 3 mo |
Rat (n = 8) |
No change in catecholamine content of sympathetic neurons and adrenal medulla |
Alho et al., 1986 |
|
1000 |
6 h/d, 5 d/w, 30 d |
Rat |
Altered latencies for flash-evoked potentials (CNS) |
Dyer et al., 1984 |
|
1200 |
6.5 h/d, 5 d/w, 2 y |
Rat |
NOAEL for lethality |
Huff, 1990 |
|
2000 |
48 h, continuous |
Rat (n = 8) |
Reduction in catecholamine content of sympathetic neurons |
Alho et al., 1986 |
TABLE 12-2 Exposure Limits Set by Other Organizations
TABLE 12-3 Spacecraft Maximum Allowable Concentrations
|
Duration |
ppm |
mg/m3 |
Target Toxicity |
|
1 h |
16 |
60 |
Neurotoxicity |
|
24 h |
16 |
60 |
Neurotoxicity |
|
7 da |
16 |
60 |
Neurotoxicity, irritation |
|
30 d |
16 |
60 |
Neurotoxicity, irritation |
|
180 d |
16 |
60 |
Neurotoxicity, irritation |
|
a Temporary 7-d SMAC was set at 20 ppm. |
|||
Rationale for Acceptable Concentrations
The SMAC values listed above were set based on the lowest acceptable concentration (AC) for any adverse effect at each exposure duration using guidelines established jointly by the National Research Council and NASA (NRC, 1992). The evidence and logic used to determine
the ACs for each adverse effect and exposure duration are documented below.
Irritation
A NOAEL of 40-ppm toluene vapor for irritation of the eyes and nose during a 6-h exposure was established in 16 young male volunteers. Because irritation is dependent on concentration but not on exposure duration, the ACs for all exposure durations from 7 d to 180 d were based on the 40-ppm NOAEL, adjusting for the low number of subjects by a factor equal to one-tenth the square root of the number of subjects tested:
7-d, 30-d, and 180-d ACs based on irritation
= NOAEL x 1/small n factor
= 40 ppm x (square root of 16)/10
= 16 ppm.
Some irritation is acceptable for short-term SMACs; therefore, the irritancy ACs for 1 h and 24 h were set equal to the LOAEL:
1-h and 24-h ACs based on irritation
= LOAEL
= 100 ppm.
Neurotoxicity
A variety of CNS effects in humans have been reported in the literature, and ACs were calculated separately for each set of end points or experiment. These ACs were not adjusted for duration of exposure because the CNS depressant effects of toluene have been shown to be reversible within a few minutes of cessation of exposure (Bruckner and Peterson, 1981b), except possibly after chronic exposure to high concentrations, indicating that the effects are dependent on blood concentrations but not exposure duration. Pharmacokinetic experiments have shown that arterial blood concentrations reach 60% of maximum in 10-
15 min and decrease rapidly after cessation of exposure (Benignus et al., 1981).
Although there are a number of reports in the literature that have examined the effects on humans of exposure to toluene vapors at concentrations in the range of the threshold limit value (TLV) of 100 ppm, the ACs have been set using the study that yields the lowest values of the ACs: In 16 young subjects, exposure at 10 or 40 ppm for 6 h did not result in any adverse effects for any of the end points examined (nasal mucus flow, lung function, subjective response, and psychometric performance), but exposure at 100 ppm produced irritation of eyes and nose and borderline significance in reduced performance in a battery of eight psychometric tests for visual perception, vigilance, psychomotor functions and higher cortical functions (Andersen et al., 1983). Acceptable concentrations for vigilance, visual perception, psychomotor functions, higher cortical functions, headache, dizziness, and a feeling of inebriation were calculated. The ACs for 7 d, 30 d, and 180 d were based on the 40-ppm NOAEL, adjusting for the number of subjects.
7-d, 30-d, and 180-d ACs based on CNS effects
= NOAEL x 1/small n factor
= 40 ppm x (square root of 16)/10
= 16 ppm.
Although headaches, irritation, and insignificant decrements in psychometric tests would be acceptable for short-term contingency exposures, dizziness would not be acceptable, even for brief exposures during contingency operations. Thus, the ACs for 1 h and 24 h were also based on the 40-ppm NOAEL, adjusting for the number of subjects.
1-h and 24-h ACs based on CNS effects
= NOAEL x 1/small n factor
= 40 ppm x (square root of 16)/10
= 16 ppm.
ACs for clinical, neuropsychological, or autonomic nervous system effects that persist after long-term occupational exposure were calculated on the basis of the 117-ppm NOAEL, correcting it for the number of subjects.
180-d AC based on neurotoxicity
= NOAEL x 1/small n factor
= 117 ppm x (square root of 43)/10
= 77 ppm.
ACs for exposure periods shorter than 180 d were not based on these persistent effects because the effects involve anatomical lesions produced after long-term exposures rather than a blood-concentration-dependent functional impairment.
High-frequency hearing loss and appropriate cochlear changes were found in weanling rats exposed to toluene at 1200-1400 ppm, 14 h/d, 7 d/w, for 5 w (Rebert et al., 1982; Pryor and Rebert, 1984). In adult animals, however, no solvent-induced alterations in the structure of brain or peripheral nerves were seen after exposure at 1500 ppm, 6 h/d, 5 d/w, for 6 mo (Spencer et al., 1985). Thus, this finding was not used to set an AC.
One study (Dyer et al., 1984) reported that a 30-d intermittent exposure of rats to toluene at 1000 ppm led to subtle changes in brain-wave activity, which was measured a day after exposure. This report was not used in setting an AC for neurotoxicity for the following reasons: First, the time course for this effect was not studied, and, second, the brain-wave changes were not correlated with any functional deficit. Similarly, the study of mice (Kostas and Hotchin, 1981) exposed to toluene in the womb through 35 d of age at a concentration of 400 ppm in drinking water indicated a CNS effect that could not easily be correlated with effects in humans (that is, decreased habituation or open-field activity at 35 d of age and depressed rotorod performance at 45-55 d of age).
Reproductive Toxicity
There are inconclusive reports concerning toluene's reproductive toxicity. In rabbits, continuous exposure at 266 ppm for gestation days 615 caused a decrease in maternal-weight gain and a slight, but not statistically significant, increase in spontaneous abortion (Ungváry and Tátrai, 1985). A study of spontaneous abortion rates in the wives (n = 28) of men who were occupationally exposed to toluene suggested an
increased risk for spontaneous abortion, but the exposure concentrations could not be quantitated (Taskinen et al., 1989). Thus, no AC could be set for reproductive toxicity.
Hepatotoxicity and Nephrotoxicity
Even at high but unmeasured concentrations and long exposure durations in humans (such as chronic glue sniffers), liver and kidney toxicity is variable and reversible. Subchronic intermittent exposure of rats to toluene at 5000 ppm produced reversible weight gains in the liver and kidneys, which is not considered an adverse effect. Thus, an AC was not set for hepatotoxicity and nephrotoxicity.
Lethality
A NOAEL of 1200-ppm toluene was seen in a 2-y exposure of mice (n = 120) and rats (n = 100), 6.5 h/d, 5 d/w (equivalent to 140 d continuous exposure). Thus, applying Haber's rule for 180-d exposures (but not increasing the AC for exposures shorter than the experimental exposure) and using a species extrapolation factor of 10, the 30-d and 180-d ACs are the following:
30-d AC based on lethality
= NOAEL x 1/species factor
= 1200 ppm x 1/10
= 120 ppm.
180-d AC based on lethality
= NOAEL x time adjustment x 1/species factor
= 1200 ppm x (140 d/180 d) x 1/10
= 93 ppm (rounded to 90 ppm).
ACs for shorter exposures are not justified from this data set.
For 1-h exposures, the 1-h rat LC50 of 27,000 ppm was reduced by factors of 10 for species extrapolation and 10 for estimating an LCo from an LC50.
1-h AC based on lethality
= LC50 x 1/species factor x 1/LC50 to LC0 factor
= 27,000 ppm x 1/10 x 1/10
= 270 ppm.
Spaceflight Effects
The toxicity of toluene is not expected to be altered by the conditions of spaceflight.
Recommendations
The molecular mechanisms of action of toluene in producing its CNS effects and other toxic effects are poorly understood. Multi-species studies (including humans) are needed to elucidate the role of metabolites in the pharmacokinetics of toluene toxicity and offer results for comparison between species.
The development and validation of neurobehavioral tests, which measure effects relevant to the ability of humans to function competently in space or on earth, are needed to quantitate the effects of toluene exposure. Studies using such validated tests should include measurements at several vapor concentrations and with and without exercise at light and heavy workloads.
Although the data would not be applicable to spaceflight under current NASA guidelines, which prohibit spaceflight by pregnant astronauts, more data are needed on the developmental and reproductive toxicity of inhaled toluene, particularly with regard to teratogenicity and spontaneous abortion. Although some data are available on tests conducted in rats and mice that support suggestive results from epidemiological studies in female workers, these data are not compelling and require replication and extension.
TABLE 12-4 End Points and Acceptable Concentrations
|
|
Uncertainty Factors |
|
|||||||||
|
End Point |
Exposure Data |
Species and Reference |
NOAEL |
Time |
Species |
Spaceflight |
Acceptable Concentrations, ppm |
||||
|
1 h |
24 h |
7 d |
30 d |
180 d |
|||||||
|
Irritation Eye and nose |
NOAEL at 40 ppm, 6 h |
Human (n = 16) (Andersen et al., 1983) |
1 |
1 |
1 |
100 |
100 |
16 |
16 |
16 |
|
|
Neurotoxicity Headache, dizziness, inebriation, visual perception, vigilance, psychomotor functions, higher cortical functions |
NOAEL at 40 ppm, 6 h |
Human(n = 16) (Andersen et al., 1983) |
1 |
1 |
1 |
16 |
16 |
16 |
16 |
16 |
|
|
Clinical, neurophysiological, neurological, and autonomic function effects |
NOAEL at 117 ppm, 8 h/d, 5 d/w, 22 y |
Human (n = 43) (Juntunen et al., 1985) |
1 |
— |
1 |
1 |
— |
— |
— |
— |
77 |
|
Lethality |
NOAEL at 1200 ppm, 6.5 h/d, 5d/w, 2 y |
Rat (Huff, 1990) |
10 |
HRb |
10 |
1 |
— |
— |
— |
120 |
90 |
|
|
LC50 at 27,000 ppm, 1 h |
Rat (Pryor, 1978) |
10 |
— |
10 |
1 |
270 |
— |
— |
— |
— |
|
SMAC |
|
16 |
16 |
16 |
16 |
16 |
|||||
|
a Haber's rule was not used to extrapolate from the experimental exposure duration because both irritancy and neurotoxicity are dependent on concentration but not on exposure duration. b HR = Haber's rule. |
|||||||||||
References
Alho, H., H. Tahti, J. Koistinaho, and A. Hervonen. 1986. The effect of toluene inhalation exposure on catecholamine content of rat sympathetic neurons. Med. Biol. 64:285-288.
Andersen, I., G.R. Lundqvist, L. Mølhave, O.F. Pedersen, D.F. Proctor, M. Væth, and D.P. Wyon. 1983. Human response to controlled levels of toluene in six-hour exposures. Scand. J. Work Environ. Health 9:405-418.
Bælum, J., I. Anderson, G.R. Lundqvist, L. Mølhave, O.F. Pedersen, M. Væth, and D.P. Wyon. 1985. Response of solvent-exposed printers and unexposed controls to six-hour toluene exposure. Scand. J. Work Environ. Health 11:271-280.
Benignus, V.A. 1981a. Health effects of toluene: A review. Neurotoxicology 2:526-588.
Benignus, V.A. 1981b. Neurobehavioral effects of toluene: A review. Neurobehav. Toxicol. Teratol. 3:407-415.
Benignus, V.A., K.E. Muller, C.N. Barton, and J.A. Bittikofer. 1981. Toluene levels in blood and brain of rats during and after respiratory exposure. Toxicol. Appl. Pharmacol. 61:326-334.
Bos, R.P., R.M. Brouns, R. Van Doorn, J.L. Theuws, and P.T. Henderson. 1981. Non-mutagenicity of toluene, o-xylene, m-xylene, p-xylene, o-methylbenzylalcohol and o-methylbenzylsulfate in the Ames assay. Mutat. Res. 88:273-279.
Bruckner, J.V., and R.G. Peterson. 1981a. Evaluation of toluene and acetone inhalant abuse. I. Pharmacology and pharmacodynamics. Toxicol. Appl. Pharmacol. 61:27-38.
Bruckner, J.V., and R.G. Peterson. 1981b. Evaluation of toluene and acetone inhalant abuse. II. Model development and toxicology. Toxicol. Appl. Pharmacol. 61:302-312.
Courtney, K.D., J.E. Andrews, J. Springer, M. Menache, T. Williams, L. Dalley, and J.A. Graham. 1986. A perinatal study of toluene in CD-1 mice. Fundam. Appl. Toxicol. 6:145-154.
Døssing, M., J. Bælum, S. Hansen, and G. Lundqvist. 1984. Effects of ethanol, cimetidine, and propanolol on toluene metabolism in man. Int. Arch. Occup. Environ. Health 54:309-315.
Dyer, R.S., K.E. Muller, R. Janssen, C.N. Barton, W.K. Boyes, and V.A. Benignus. 1984. Neurophysiological effects of 30 day chronic
exposure to toluene in rats. Neurobehav. Toxicol. Teratol. 6:363-368.
Forni, A., E. Pacifico, and A. Limonta. 1971. Chromosome studies in workers exposed to benzene or toluene or both . Arch. Environ. Health 22:373-378.
Freundt, K.J., and J.-C. Schneider. 1986. Drastic increase of the m-xylene or toluene concentrations in blood of rats after combined inhalation with acetone. In 27th Spring Meeting of the Deutsche Pharmakologische Gesellschaft [German Pharmacological Society], Mainz, West Germany, March 11-14, 1986.
Gamberale, F., and M. Hultengren. 1972. Toluene exposure. II. Psychophysiological functions. Work Environ. Health 9:131-139.
Gerner-Smidt, P., and U. Friedrich. 1978. The mutagenic effect of benzene, toluene, and xylene studied by the SCE technique. Mutat. Res. 58:313-316.
Gibson, J.E., and J.F. Hardisty. 1983. Chronic toxicity and oncogenicity bioassay of inhaled toluene in Fischer-344 rats. Fundam. Appl. Toxicol. 3:315-319.
Guillot, J.P., J.F. Gonnet, C. Clement, L. Caillard, and R. Truhaut. 1982. Evaluation of the ocular-irritation potential of 56 compounds. Food Chem. Toxicol. 20:573-582.
Hudák, A., and G. Ungváry. 1978. Embryotoxic effects of benzene and its methyl derivatives: toluene, xylene. Toxicology 11:55-63.
Huff, J. 1990. NTP Technical Report on the Toxicology and Carcinogenesis Studies of Toluene (CAS No. 108-88-3) in F344/N Rats and B6C3F1 Mice (Inhalation Studies). NTP TR 371. National Institutes of Health, National Toxicology Program, Research Triangle Park, N.C.
Ikeda, M. 1974. Reciprocal metabolic inhibition of toluene and trichloroethylene in vivo and in vitro. Int. Arch. Arbeitsmed. 33:125-130.
Ikeda, M., H. Otsuji, and T. Imamura. 1972. In vivo suppression of benzene and styrene oxidation by co-administered toluene in rats and effects of phenobarbital. Xenobiotica 2:101-106.
Inoue, O., K. Seiji, T. Watanabe, M. Kasahara, J. Nakatsuka, S. Yin, G. Li, S. Cai, C. Jin, and M. Ikeda. 1986. Possible ethnic difference in toluene metabolism: A comparative study among Chinese, Turkish, and Japanese solvent workers. Toxicol. Lett. 34:167-174.
Juntunen, J., E. Matikainen, M. Antti-Poika, H. Souranta, and M. Valle. 1985. Nervous system effects of long-term occupational exposure to toluene. Acta Neurol. Scand. 75:512-517.
Kostas, J., and J. Hotchin. 1981. Behavioral effects of low-level perinatal exposure to toluene in mice. Neurobehav. Toxicol. Teratol. 3:467-469.
Ladefoged, O., P. Strange, A. Moeller, H.R. Lam, G. Oestergaard, J. Larsen, and P. Arlien-Soeborg. 1991. Irreversible effects in rats of toluene (inhalation) exposure for six months. Pharmacol. Toxicol. (Copenhagen) 68:384-390.
Liang, J.C., T.C. Hsu, and J.E. Henry. 1983. Cytogenetic assays for mitotic poisons. The grasshopper embryo system for volatile liquids. Mutat. Res. 113:467-479.
Maki-Paakkanen, J., K. Husgafvel-Pursiainen, P.L. Kalliomäki, J. Tuominen, and M. Sorsa. 1980. Toluene-exposed workers and chromosome aberrations. J. Toxicol. Environ. Health 6:775-781.
McDonald, J.C., J. Lavoie, R. Cote, and A.D. McDonald. 1987. Chemical exposures at work in early pregnancy and congenital defect: a case-referent study. Br. J. Ind. Med. 44:527-533.
NRC. 1992. Guidelines for Developing Spacecraft Maximum Allowable Concentrations for Space Station Contaminants. Washington, D.C.: National Academy Press.
Ogata, M., Y. Takatsuka, K. Tomokuni, and K. Muroi. 1971. Excretion of hippuric acid and m- or p-methylhippuric acid in the urine of persons exposed to vapours of toluene and m- or p-xylene in a exposure chamber and in workshops, with specific reference to repeated exposures. Br. J. Ind. Med. 28:382-385.
Peterson, R., and J. Bruckner. 1978. Animal model for studying the toxicological and pharmacological effects resulting from exposure to volatile substances. Final Report. Contract No. 271-75-3067. National Institute on Drug Abuse, Rockville, Md.
Pryor, G.T., and C.S. Rebert. 1984. Hearing loss in rats caused by inhalation of toluene, xylenes, and styrene. Abstract No. 315 in Arbetarskyddsverket. International Conference on Organic Solvent Toxicity, Stockholm, Sweden.
Pryor, G., H.R. Howd, R. Malik, R. Jensen, C. Rebert. 1978. Biomedical studies on the effects of abused inhalant mixtures. Annual Progress Report 2, Contract No. 271-77-3402. National Institute on Drug Abuse, Rockville, Md.
Purcell, K.J., G.H. Cason, M.L. Gargas, M.E. Anderson, and C. C. Travis. 1990. In vivo metabolic interaction of benzene and toluene. Toxicol. Lett. 52:141-152.
Rebert, C.S., S.S. Sorensen, R.A. Howd, and G.T. Pryor. 1982. Toluene-induced hearing loss in rats evidenced by the brain stem auditory-evoked response. Pp. 314-319 in The Toxicology of Petroleum Hydrocarbons. H.N. MacFarland, C.E. Holdsworth, J.A. Macgregor, R.W. Call, and M.L. Kane, eds. American Petroleum Institute, Washington, D.C.
Sandmeyer, E.E. 1981. Aromatic hydrocarbons. Pp. 3283-3291 in Patty's Industrial Hygiene and Toxicology, Vol. 2B, G.D. Clayton and F.E. Clayton, eds. New York: John Wiley & Sons.
Spencer, P.S., and H.H. Schaumberg. 1985. Organic solvent neurotoxicity. Facts and research needs. Scand. J. Work Environ. Health 11:53-60.
Taskinen, H., A. Anttila, M.L. Lindbohm, M. Sallman, and K. Hemminki. 1989. Spontaneous abortions and congenital malformations among the wives of men occupationally exposed to organic solvents. Scand. J. Work Environ. Health 15:345-352.
Tátrai, E., A. Hudák, and G. Ungváry. 1979. [Experimental model for the study of the teratogenic interaction of chemical agents and drugs (toluene and acetylsalicylic acid)]. Morphol. Igazsagugyi Orv. Sz. 19:299-304.
Ungváry, G., and E. Tátrai. 1985. On the embryotoxic effects of benzene and its alkyl derivatives in mice, rats, and rabbits. Arch. Toxicol. 8:425-430.
Ungváry, G., E. Tátrai, M. Lorincz, and G. Barcza. 1982. Joint embryotoxic effect of toluene, an extensively used industrial chemical substance, and acetylsalicylic acid (aspirin). Munkavedelem 28:25-31.
Veulemans, J., and R. Masschelein. 1978. Experimental human exposure to toluene: Factors influencing the individual respiratory uptake and elimination. Int. Arch. Occup. Environ. Health 42:91-103.
Von Oettingen, W., P. Neal, D. Donahue, J. Svirbely, H. Baernstein, A. Monaco, P. Valaer, and J. Mitchell. 1942. The toxicity and potential dangers of toluene, with special reference to its maximum permissible concentration. U.S. Public Health Service Bull. 279:1-50.
Weiss, B., R.W. Wood, and D.A. Macys. 1979. Behavioral toxicology of carbon disulfide and toluene . Environ. Health Perspect. 30: 39-45.





















