B2 Benzene
John T. James, Ph.D., and Harold L. Kaplan, Ph.D.
Johnson Space Center Toxicology Group
Biomedical Operations and Research Branch
Houston, Texas
Physical and Chemical Properties
Benzene is a clear, colorless, highly flammable liquid with an odor characteristic of aromatic hydrocarbons (Sandmeyer, 1981; ATSDR, 1989). The odor threshold is 4-5 ppm (ATSDR, 1989).
|
Synonym: |
Benzol |
|
Chemical structure: |
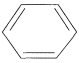 |
|
Formula: |
C6H6 |
|
CAS number: |
71-43-2 |
|
Molecular weight: |
78.11 |
|
Boiling point: |
80.1 °C |
|
Melting point: |
5.5°C |
|
Specific gravity: |
0.88 (20°C) |
|
Vapor pressure: |
95 torr (25°C) |
|
Solubility: |
Slightly soluble in water, very soluble in organic solvents |
|
Conversion factors at 25°C, 1 atm: |
1 ppm = 3.26 mg/m3 1 mg/m3 = 0.31 ppm |
Occurrence and Use
Benzene is a natural constituent of crude oil, coal tar, and other fossil fuels (Sandmeyer, 1981). Most of the millions of gallons of benzene that are used in the United States each year are produced by petroleum refining. A major use of benzene is as a component in gasoline, particularly in unleaded fuels, because of its antiknock properties (ATSDR, 1989). Its content in gasoline is estimated to range from 1% to 2% in the United States and up to 5% in European countries. Large quantities are also used to synthesize chemicals for the manufacture of various plastics, resins, elastomers, dyes, and pesticides (Marcus, 1987). Minimal amounts are now used as a solvent in paints, cements, adhesives, and paint removers. Sources of atmospheric contamination include fugitive emissions from gasoline handling, thermal degradation of plastics, solid waste gasification, and tobacco smoke (Sandmeyer, 1981; Marcus, 1987).
Benzene has been detected in approximately 10% of recent air samples in the space-shuttle cabin and in Spacelab at concentrations of 0.01-0.1 mg/m3 (James et al., 1994). Benzene has not been used as a payload or system chemical aboard the space shuttle; hence, the low concentrations observed are due to materials out-gassing. Benzene has been found as a pyrolysis product of electronic components identical to ones that failed in data-display units aboard STS-35 (J. Boyd, NASA, unpublished data).
Pharmacokinetics and Metabolism
Absorption
In humans, benzene vapor is rapidly absorbed by the lungs in amounts equivalent to about 50% of the doses inhaled over several hours of exposure to concentrations of 50-100 ppm (Nomiyama and Nomiyama, 1974a,b; Sato and Nakajima, 1979; R. Snyder et al., 1981; IARC, 1982). In men and women exposed to 52-62 ppm for 4 h, respiratory uptake was 47%, with little difference between the sexes (Nomiyama and Nomiyama, 1974a,b; IARC, 1982). Absorption was greatest during the first 5 min of exposure and reached a constant level
between 15 min (Srbova et al., 1950) and 3 h (Nomiyama and Nomiyama, 1974a,b; IARC, 1982). Respiratory retention (the difference between respiratory uptake and excretion) was estimated as 30% of the inhaled dose (Nomiyama and Nomiyama, 1974a,b; IARC, 1982).
Benzene can also be absorbed through the skin, but the rate of absorption is lower than that for inhalation exposure (ATSDR, 1989). It has been calculated that an adult working in ambient air containing benzene at 10 ppm would absorb 7.5 µL/h from inhalation versus 1.5 µL/h from whole-body dermal absorption (Blank and McAuliffe, 1985). Absorption of benzene vapor by animals also is rapid, but retention of absorbed benzene might be affected by exposure concentration. In rats and mice, the percentage of inhaled vapor that was retained decreased from 33% to 15% during a 6-h exposure and from 50% to 10%, respectively, as the concentration was increased from approximately 10 ppm to 1000 ppm (Sabourin et al., 1987).
Distribution
Once benzene is absorbed into the blood, it is rapidly distributed to tissues; the relative uptake is dependent on the perfusion rate of tissues (ATSDR, 1989). Because of its high lipophilicity, benzene tends to accumulate in fatty tissues. In experimental human exposures, lower blood concentrations and slower elimination in females than in males were attributed primarily to relatively higher fat content of females (Sato et al., 1975). Tissue levels of benzene in accidental- or intentional-exposure victims are variable but generally indicate higher concentrations in brain, fat, and liver (Winek et al., 1967; Winek and Collom, 1971). About 60% of the absorbed benzene was found in bone marrow, adipose tissue, and liver of humans exposed to unspecified concentrations (Duvoir et al., 1946).
Distribution of benzene in animals also is rapid; the relative uptake and accumulation in tissues appear to be dependent on perfusion rate and lipid content (Schrenk et al., 1941; Ghantous and Danielsson, 1986). Following a 10-min inhalation exposure of mice, benzene was present in well-perfused tissues, such as liver and kidney, and in lipid-rich tissues, such as brain and fat (Ghantous and Danielsson, 1986). In rats exposed to 500 ppm, steady-state concentrations were highest in
fat, bone marrow, and blood (15:5.5:1 ratio) and lower in kidney, lung, liver, brain, and spleen (Rickett et al., 1979). Female rats and male rats with large body fat content stored benzene longer and eliminated it more slowly than lean animals (Sato et al., 1974).
Excretion
Following inhalation, benzene is eliminated from the body by humans and animals in unchanged form in the exhaled air and in metabolized form in the urine (ATSDR, 1989). Estimates of the fraction of absorbed benzene excreted in the expired air of humans range from 12% to 50% (Srbova et al., 1950; Teisinger et al., 1952; Nomiyama and Nomiyama, 1974a,b; IARC, 1982). The respiratory elimination is described as triphasic-an initial fast component having a half-life of 0.9 h and two slower components having half-lives of 3 and 15 h, respectively (Nomiyama and Nomiyama, 1974a,b; IARC, 1982). No differences in respiratory elimination were observed between men and women (Nomiyama and Nomiyama, 1974a,b; IARC, 1982).
In rats exposed to 500 ppm for 6 h, a biphasic pattern of respiratory elimination of benzene was observed, with half-lives of 0.7 and 13.1 h (Rickert et al., 1979). Respiratory elimination might be increased as a result of saturation of metabolic pathways by high doses of benzene. At lower concentrations (10-130 ppm), less than 6% of inhaled 14C-benzene was exhaled by rats and mice, whereas at concentrations of 260 and 870 ppm (rats) and 990 ppm (mice), exhaled radioactivity increased from 11% to 48% (Sabourin et al., 1987).
Metabolism
The metabolism of benzene is complex and not completely elucidated. It is well established that most of the absorbed benzene is metabolized through a variety of major and minor pathways in humans and animals and excreted as metabolites in the urine (Snyder, 1987). The major site of metabolism is the liver, although mixed-function oxidases that catalyze the oxidation of benzene also occur in bone marrow, the target organ of benzene toxicity (Snyder, 1987). Benzene is
metabolized mostly by oxidation to the major metabolites—phenol, catechol, and hydroquinone, which are excreted in the urine as sulfate and glucuronide conjugates (Snyder, 1987). Many minor metabolites also are formed, of which phenylmercapturic acid and trans, trans-muconic acid are the most important.
In the initial metabolic step, benzene is thought to be oxidized by hepatic microsomal mixed-function oxidase to a reactive intermediate, benzene oxide (Erexson et al., 1985; Yardley-Jones et al., 1991). Most of the benzene oxide rearranges spontaneously to phenol. Phenol is mostly conjugated and excreted as sulfate ester and glucuronide, but some can be further oxidized to catechol and hydroquinone (Erexson et al., 1985; ATSDR, 1989). The latter compound spontaneously oxidizes to form 1,4-benzoquinone. The remaining benzene oxide can be conjugated with glutathione to produce phenylmercapturic acid, which is excreted in the urine, or it can be converted to benzene glycol (ATSDR, 1989; Erexson et al., 1985). The latter compound can undergo dehydrogenation to form catechol or further oxidation and ring breakage to produce trans, trans-muconic acid. Most of the catechol is conjugated and excreted, but a small amount is oxidized to the trihydroxybenzene 1,2,4-benzenetriol.
In humans, the major urinary metabolite of benzene is phenol (Teisinger et al., 1952). Most of the phenol is excreted as sulfate ester (Teisinger et al., 1952), but significant amounts of glucuronide can be formed, especially after exposure to high concentrations of benzene (Sherwood, 1972). In an inhalation study with human subjects, 28.8% of the absorbed benzene was excreted in the urine as phenol, 2.9% as catechol, and 1.1 % as hydroquinone (Teisinger et al., 1952). Urinary excretion was highest within the first 24 h following exposure and was essentially complete within 48 h.
In workers exposed for 7 h to benzene at 1-76 ppm, the correlation between exposure concentration and urinary phenol excretion was 0.891 (Inoue et al., 1986). A urinary phenol concentration of 75 mg/L indicates an 8-h (time-weighted average) exposure at 10 ppm (NIOSH, 1974) and a concentration of 100 mg/L indicates an 8-h exposure at 25 ppm (Sandmeyer, 1981). The ACGIH biological exposure index (BEI) for benzene exposure is a urinary concentration of phenol at 50 mg/g of creatinine at the end of a work shift, but ACGIH notes that phenol is usually present in unexposed individuals and also might result from ex-
posure to other chemicals (ACGIH, 1991). Recently, S-phenylmercapturic acid in urine (Stommel et al., 1989) and benzene hemoglobin adducts in blood (Sun et al, 1990) were evaluated as possible biomarkers of benzene exposure.
In a study in which radiolabeled benzene was administered to rabbits by oral intubation, 43 % of the radioactivity was recovered as exhaled, unmetabolized benzene and 1.5% was recovered as carbon dioxide (Parke and Williams, 1953). Urinary metabolites (representing 35% of the dose) were mainly in the form of phenolic sulfates and glucuronides and included phenol (23%), hydroquinone (4.8%), catechol (2.2%), trans, trans-muconic acid (1.3%), phenylmercapturic acid (0.5%), and 1,2,4-trihydroxybenzene (0.3%). This same general profile was also found in rats (Cornish and Ryan, 1965), mice (Longacre et al., 1981), and cats and dogs (Oehme, 1969).
Benzene metabolism appears to be qualitatively similar but quantitatively different among species. Mice metabolized the largest fraction of benzene (67%) to hydroquinone conjugates and muconic acid metabolites, followed by monkeys (31%), rats (17%), and chimpanzees (14%) (Sabourin et al., 1992). Urinary metabolite data from workers exposed for about 7 h to benzene at 50 ppm suggest that the metabolism of benzene to hydroquinone compounds in humans is quantitatively comparable to that in mice, whereas the metabolism to muconic acid is comparable to that in rats and one-third of that in mice (Henderson et al., 1992).
Metabolic pathways leading to putative toxic metabolites, such as hydroquinone and muconic acid metabolites, have been designated ''toxification pathways'' in contrast to "detoxification pathways," which lead to the less toxic metabolites, such as phenyl conjugates and phenylmercapturic acid products (Henderson et al., 1992). In mice, rats, and monkeys, the toxification pathways appear to be low-capacity, high-affinity pathways that become saturated at relatively low concentrations, resulting in proportionately less formation of hydroquinone and muconic acid at higher concentrations (Henderson et al., 1992).
Stimulation or inhibition of hepatic mixed-function oxidase activity by benzene, other chemicals, or dietary factors might alter the rate of metabolism of benzene. Exposure of mice to benzene enhanced the in vitro metabolism of benzene by hepatic microsomes from these animals, but exposure to phenolic metabolites did not (Dean, 1978; Gonasun et
al., 1973). In contrast, repeated inhalation exposure of mice and rats to benzene at 600 ppm for 6 h/d, 5 d/w, for 3 w had minimal effects on urinary metabolite profiles (Sabourin et al., 1990). Ethanol ingestion as well as food deprivation and carbohydrate restriction enhanced the metabolism of benzene in rats (Sato and Nakajima, 1985).
Three physiologically based pharmacokinetic (PBPK) models were recently proposed to describe the pharmacokinetics and metabolism of benzene in animals (Medinsky et al., 1989; Woodruff et al., 1989; Travis et al., 1990). The models are herein arbitrarily referred to by the name of the first author as the Medinsky model (Medinsky et al., 1989), the Travis model (Travis et al., 1990), and the Woodruff model (Woodruff et al., 1989). The Medinsky model was adjusted to data obtained experimentally with mice and rats (Medinsky et al., 1989), the Travis model with data on mice, rats, and humans (Travis et al., 1990), and the Woodruff model with data on rats (Woodruff et al., 1989). The models have similar structures but differ in the parameter values used for the same species. In a comparison and evaluation of the models, the investigators concluded that PBPK models are useful for investigating the mechanism of toxicity of benzene but not for risk assessment of cancer (Bois et al., 1991).
Toxicity Summary
Acute and Short-Term Toxicity
Neurotoxicity
In humans, acute inhalation of benzene produces CNS effects, including euphoria, giddiness, nausea and drowsiness at lower concentrations, and ataxia, narcosis, delirium, convulsions, unconsciousness, and even death at high concentrations (Sandmeyer, 1981). Recovery is usually rapid, but, in some cases, symptoms have persisted for weeks.
The symptoms, and their severity, vary with concentration and duration of exposure. It is estimated (without supporting data) that exposure at 25 ppm for 8 h has no effects; 50-150 ppm for 5 h produces headache, lassitude, and weakness, symptoms that are exaggerated at 500 ppm; 3000 ppm for 0.5-1.0 h can be tolerated; 7500 ppm for 30 min is
dangerous to life; and 19,000-20,000 ppm can be fatal in 5-10 min (Gerarde, 1962; Von Oettingen, 1940). The rapid development of CNS effects, including death in some cases, suggests that benzene, not a metabolite, is responsible for the acute toxicity and that the cause of sudden death is asphyxiation, respiratory arrest, CNS depression, or cardiac arrhythmia (Sandmeyer, 1981).
Acute inhalation exposure to benzene also causes CNS effects in animals. In mice, exposure at 2200 ppm produced narcosis, and at 4600 ppm and 11,800 ppm produced narcosis in 51 min and 8 min, respectively (Von Oettingen, 1940). At 11,800 ppm, deaths occurred in 38295 min, and at 24,000 ppm, deaths occurred in 50 min. About 4000 ppm was the narcotic concentration threshold for laboratory animals, and more than 10,000 ppm was fatal after several hours of exposure (Leong, 1977). At 35,000-45,000 ppm, anesthesia occurred in about 4 min, with excitation and tremors after 5 min, loss of pupillary reflexes after 6.5 min, involuntary blinking after 15 min, and death after 22-71 min (Carpenter, et al., 1944).
Lethality data of rats confirm the low potential for benzene to cause death via inhalation. A 4-h exposure at 16,000 ppm resulted in the deaths of four of six rats (Smyth et al., 1962). An LC50 value of 13,700 ppm was determined for a 4-h exposure of rats (Drew and Fouts, 1974). Respiratory paralysis followed by ventricular fibrillation was observed in male rats exposed to lethal concentrations (Sandmeyer, 1981).
A limited number of animal studies measured electroencephalographic and behavioral changes to investigate the CNS effects of benzene. In cats, exposure to benzene at a concentration of 12,000 ppm for 10 min caused restlessness, rapid respiration, and head nodding, accompanied by hypersynchronous amygdaloid EEG activity (Contreras et al., 1979). Ataxia and postural collapse occurred when concentrations were increased to 52,000 ppm. With repeated daily 10-min exposures, a 3-Hz spike-wave activity in the gyrus cinguli developed and generalized tonic-clonic seizures developed after a sensitization period. Behavioral disturbances, characterized by increased milk-licking, were evident in C57BL mice after the first week of exposure to 100 or 300 ppm (Dempster et al., 1984). Less sensitive parameters, home-cage food intake and hind-limb grip strength, were reduced at 1000 and 3000 ppm, but not at 100 or 300 ppm, even when exposure durations were adjusted to yield a minimum Ct (concentration x time) product of 3000 ppm.
Cardiac Sensitization
Acute inhalation of high concentrations of benzene by cats and monkeys induced ventricular dysrhythmias, which were abolished by removal of the adrenals and the stellate ganglia, and were restored by injections of epinephrine (Nahum and Hoff, 1934). The effects were attributed to the sensitization of the myocardium to epinephrine by benzene.
In Wistar rats, previous inhalation of benzene at 3000 and 7000 ppm, but not at 1500 ppm, for 15 min increased the number of ectopic ventricular beats induced by coronary ligation or intravenous administration of aconitine (Magos et al., 1990). With an increased dose of aconitine, ventricular fibrillation developed rapidly at 7000 ppm, and progressed to asystole and death after 16 min.
Hematotoxicity and Immunotoxicity
Although benzene-induced hematotoxicity and immunotoxicity are generally associated with prolonged exposure, abnormal hematological parameters have been observed in some workers exposed to low concentrations for short periods (ATSDR, 1989). These observations are consistent with the results of animal studies showing hematological changes after short-term, and even acute, exposures. After an 8-h inhalation exposure of mice to 4680 ppm, a significant depletion of bone-marrow colony-forming cells was evident in an in vitro cell culture (Uyeki et al., 1977). In mice, continuous exposure at 100 ppm for 2 d produced leukocytopenia (Gill et al., 1980) and a 1-w exposure (6 h/d, 5 d/w) at 300 ppm decreased peripheral blood erythrocyte and lymphocyte counts (Snyder et al., 1978). Continuous exposure of NMRI mice at a concentration of 21 ppm for 4-10 d significantly decreased cellularity (number of nucleated cells) and colony-forming granulopoietic stem cells (CFU-C) in tibia bone marrow (Toft et al., 1982). Intermittent exposure (8 h/d, 5 d/w) for 2 w at 21 ppm reduced the number of CFU-C cells.
In female Wistar rats exposed 8 h/d for 7 d, peripheral leukocyte counts were depressed significantly after exposures at 50-300 ppm but not at 20 ppm (Li et al., 1986). Leukocyte alkaline phosphatase (LAP) concentrations were significantly increased at 300 ppm, marginally increased at 100 ppm, and not affected at 20 or 50 ppm.
Short-term exposures of animals at low concentrations might produce hematological changes that can affect immune-associated processes. In male C57BL mice exposed at 10 ppm for 6 h/d for 6 d, femoral lipopolysaccharide (LPS)-induced B-lymphocyte-colony-forming ability was significantly depressed, but total numbers of B lymphocytes were not (Rozen et al., 1984). At 30 ppm, splenic phytohemagglutinin (PHA)-induced blastogenesis was significantly depressed, but there was no concomitant significant depression in numbers of T lymphocytes. After six exposures at 300 ppm, mitogen-induced proliferation of bone marrow and splenic B and T lymphocytes was depressed, and numbers of T lymphocytes in thymus and spleen were reduced (Rozen and Snyder, 1985).
Genotoxicity
Benzene is not mutagenic in most in vitro test systems, including both Salmonella typhimurium (five strains) and Saccharomyces cerevisiae with and without metabolic activation, Drosophila melanogaster, mouse lymphoma cells, various human, mouse, and Chinese hamster cells, and others (ATSDR, 1989; Marcus, 1987). In vitro studies of chromosomal aberrations and other genotoxic effects of benzene yielded positive, negative, or mixed results, depending on the end point and test system. Positive results were obtained in studies of DNA binding in rabbit bone marrow and rat liver mitoblasts; negative results were obtained in studies of DNA breaks in rat hepatocytes, Chinese hamster V79 cells, and mouse L5178Y cells; and mixed results were obtained in studies of chromosomal aberrations and sister chromatid exchange (SCE) in human lymphocytes (ATSDR, 1989). Benzene did not increase SCE frequency in human lymphocytes stimulated by phytohemagglutinin (Morimoto and Wolff, 1980) or in human lymphocytes incubated without rat liver S-9 (Morimoto, 1983). Delaying addition of benzene, however, to 24 h after mitogen stimulation produced significant concentration-related increases in SCE frequency, decreases in mitotic indices, and inhibition of cell-cycle kinetics without S-9 (Erexson et al., 1985).
In contrast to in vitro results, benzene-induced cytogenetic effects,
including chromosomal and chromatid aberrations, SCE, and micronuclei, were consistently found in in vivo animal studies (ATSDR, 1989). Acute inhalation studies have shown cytogenetic effects in animals, even at low exposure concentrations. Exposure of mice at 10 ppm for 6 h induced SCE in peripheral blood lymphocytes and bone marrow as well as micronuclei in bone-marrow polychromatic erythrocytes (Erexson et al., 1985). Exposure of DBA/2 mice at 3100 ppm for 4 h significantly increased SCE frequency in bone-marrow cells in both sexes and inhibited marrow cellular proliferation in males only, but did not affect the frequency of chromosomal aberrations (Tice et al., 1980).
Subchronic and Chronic Toxicity
Prolonged inhalation of benzene by humans can result in CNS, hematotoxic, myelotoxic, immunotoxic, genotoxic, or carcinogenic effects. These effects are well established for chronic exposure, but less is known about some of these effects as a result of subchronic exposure. Limited information exists on the potential of benzene to cause adverse effects on reproductive function and pre-and postnatal development in humans.
Neurotoxicity
Involvement of the CNS might be an important effect of chronic inhalation exposure of humans and animals to benzene, but it can be masked by other more-visible effects (Sandmeyer, 1981). Workers exposed even to low concentrations (e.g., 50 ppm) reported symptoms of headaches, dizziness, fatigue, anorexia, dyspnea, and visual disturbances (Sandmeyer, 1981). Some workers also exhibited signs of CNS lesions, such as abnormal caloric labyrinth irritability and impairment of hearing. Although there are reports of polyneuritis associated with exposure to benzene, other chemicals were also involved (Sandmeyer, 1981). Exposure of rats for 5.5 mo to 20 ppm resulted in a delay in conditioned reflex response time; however, the effect was not seen at 4 ppm (Novikov, 1956).
Hematotoxicity and Myelotoxicity
The effects of benzene on the hematopoietic system have been known for many years. Prolonged exposure causes hypoplasia and depressed function of bone marrow, resulting in leukopenia, anemia, or thrombocytopenia (Sandmeyer, 1981; ATSDR, 1989). With continued exposure, bone-marrow aplasia results in pancytopenia and aplastic anemia; bone-marrow aplasia might progress and develop into myelogenous leukemia or other types of leukemia. These are not distinct diseases but rather are a continuum of changes reflecting the severity of damage to the bone marrow.
The cytopenias, which can occur as a group or in various combinations, can manifest themselves as specific adverse health effects (ATSDR, 1989). For example, thrombocytopenia induces capillary fragility, petechiae, and hemorrhage, which might result in death. Decreased circulating granulocytes decrease defenses against infection, which might have been responsible for deaths of some benzene-exposed individuals. Also, lymphocytopenia and eosinophilia, which might be related to impaired immune function, were observed in some workers.
A high prevalence and wide range of hematological responses to benzene are evident in the numerous epidemiological studies and case reports of occupationally exposed workers. Of 332 rotogravure workers exposed to benzene at 11-1060 ppm for 6-60 mo, 23 had severe cytopenia (23 of 23, leukopenia; 15 of 23, erythropenia; 18 of 23, thrombocytopenia) (Goldwater, 1941). In a rubber factory, 25 of 1104 workers exposed at up to 500 ppm (100 ppm average) developed severe pancytopenia (9 of the 25 were hospitalized), and 83 others had mild hematological disorders (Wilson, 1942). The relationship between pancytopenia, preleukemia and acute leukemia was reviewed by Goldstein (1977). Pancytopenia also was diagnosed in 6 of 217 apparently healthy shoe-factory workers exposed at 30-210 ppm for 3 mo to 17 y; 45 others had some hematological abnormalities (Aksoy et al., 1971). There also are numerous reports of aplastic anemia in occupationally exposed workers (Aksoy et al., 1972; Vigliani and Forni, 1976). Of 32 cases of aplastic anemia among workers exposed at 150-650 ppm for 4 mo to 15 y, there were eight deaths due to thrombocytopenic hemorrhage and infection (Aksoy et al., 1972). In a 10-y followup of 216 workers in a study of 282 workers, four had persistent cytopenias and one died of aplastic
anemia 9 y after cessation of exposure (Guberan and Kocher, 1971). The exposure level associated with development of noncarcinogenic hematological effects of benzene has not been established (ATSDR, 1989). A threshold of about 10 ppm for cytopenia was suggested on the basis of observations of minimal hematotoxicity in workers exposed at 20 ppm (Chang, 1972).
There is evidence that benzene-induced pancytopenia or aplastic anemia is associated with the later development of leukemia (ATSDR, 1989). In 44 patients with pancytopenia (exposure at 150-650 ppm for 4 mo to 15 y), six developed leukemia within 6 y of followup (Aksoy and Erdem, 1978). Leukemia also occurred in workers with aplastic anemia either during exposure to high concentrations or shortly after cessation of exposure; however, in a few cases the latency period was long (Aksoy et al., 1976; Aksoy, 1978). Benzene-induced leukemia is discussed in more detail in the section on carcinogenicity.
The hematotoxic effects observed in humans have been reproduced experimentally in animals; however, the response depends on species, strain, sex, and intermittent vs. continuous exposure, in addition to exposure concentration and duration (ATSDR, 1989).
Exposure of rats to benzene at concentrations of 831, 65, or 61 ppm produced a significant leukopenia within 2 to 4 w, and a less severe leukopenia after 5-8 w at concentrations of 47 or 44 ppm (Deichmann et al., 1963). Leukocyte counts were not affected by 31 ppm for 4 mo, 29 ppm for 3 mo, or 15 ppm for 7 mo. In CD-1 mice, 300 ppm for 6 h/d, 5 d/w for 13 w increased mean cell volume and mean cell hemoglobin and decreased hematocrit, hemoglobin, erythrocyte, leukocyte, and platelet counts and percentage of lymphocytes (Ward et al., 1985). Histopathological changes, including bone-marrow hypoplasia, lymphoid depletion in lymph nodes and tissue, and increased splenic extra-medullary hematopoiesis, were more prevalent and severe in males than females. No effects were evident at 1, 10, or 30 ppm.
Sprague-Dawley rats were less severely affected by the same exposure regimen than mice. The rats exhibited no effects at 1, 10, or 30 ppm and significant decreases only in leukocyte counts and percentage of lymphocytes at a concentration of 300 ppm (Ward et al., 1985). The only histological lesion was slightly decreased femoral-marrow cellularity. Repeated inhalation exposures at 80-85 ppm for 136 exposures of rats, 175 exposures of rabbits, and 193 exposures of guinea pigs in-
duced leukopenia, increased spleen weights, and histopathological changes in bone marrow in the rats, guinea pigs, or rabbits (Wolf et al., 1956).
Several animal studies indicate that benzene induces its hematotoxicity by acting on early progenitor cells in the bone marrow and spleen. In CD-1 mice, 103 ppm and higher exposures for 6 h/d for 5 d reduced marrow and spleen cellularity and decreased granulocyte macrophage colony-forming units (GM-CFU-C) in spleen but not in marrow (Green et al., 1981a). At concentrations of 302 ppm for 26 w, marrow and spleen cellularity, colony-forming units in spleen (CFU-S), and marrow GM-CFU-S were decreased. Depression of CFU-S also was reported in C57BL mice exposed at 400 ppm for 6 h/d intermittently for 9 d or consecutively for 11 d (Harigaya et al., 1981). Bone-marrow cellularity and pluripotential stem cells were significantly reduced in C57BL mice exposed for 2 w at 100 ppm, but not at 10 or 25 ppm (Cronkite et al., 1985). At 300 ppm, 2 w were required for recovery of stem-cell numbers after 2- or 4-w exposures, and 25 w were required for recovery to 92% of control values after a 16-w exposure. Peripheral blood lymphocyte counts were not affected at 10 ppm (2 w), but exhibited a dose-related decrease at 25-400 ppm.
Other investigators also observed depletions in pluripotential stem-cell numbers (Gill et al., 1980) and reductions in granulocyte and macrophage progenitor cells (C.A. Snyder et al., 1981) in mice. In studies of the erythroid cell line, repeated exposures to benzene at 10 ppm reduced the number of progenitor red blood cells, i.e., erythroid colony-forming units (CFU-E) in mice (Baarson et al., 1984; Valle-Paul and Snyder, 1986). The effects of benzene at concentrations of 100, 300, and 900 ppm (6 h/d, 5 d/w, for up to 16 w) on hematopoietic stem-cell compartments were investigated in a series of studies with female BDF1 mice (Seidel et al., 1989a,b, 1990). The CFU-E was the most sensitive compartment, showing significant concentration-dependent decreases and a marked decrease at 100 ppm as early as 1 w after the start of exposure (Seidel et al., 1990). The BFU-E (burst-forming units), CFU-S, and CFU-C compartments of progenitor cells showed dose-related decreases at 300 and 900 ppm. Recovery of stem-cell compartments was slow, requiring 73-185 d after exposure at 300 ppm.
Potential mechanisms for the development of pancytopenia and its variants in humans and animals exposed to benzene include destruction
of bone-marrow stem cells, impairment of the differentiation of these cells, and destruction of more mature hematopoietic cell precursors and circulating cells (Goldstein, 1977). Numerous studies have shown that benzene-induced bone-marrow depression is the result of inhibitory effects on proliferation, maturation, or replication of pluripotential stem cells or early proliferating committed cells in either the erythroid or myeloid lines (ATSDR, 1989). Several molecular mechanisms have been proposed to explain the hematotoxicity and myelotoxicity, as well as other toxicities, of benzene. These include suppression of RNA and DNA synthesis, alkylation of cellular sulfhydryl groups, disruption of the cell cycle, oxygen activation (or free radical formation), and covalent building of benzene metabolites to cellular macromolecules (ATSDR, 1989). These mechanisms will be reviewed in greater detail in the section on carcinogenicity.
Although the exact mechanism is unknown, it is generally accepted that benzene must be metabolized before its toxic effects (other than neurotoxicity and cardiac sensitization) are manifest (ATSDR, 1989). Evidence for a primary role for benzene metabolites in inducing myelo-suppression and hematotoxicity is provided by studies showing that agents that alter benzene metabolism modify its toxicity. Partial hepatectomy alleviated benzene-induced depression of erythropoiesis, and decreased urinary levels of benzene metabolites and covalent binding of reactive metabolites to bone-marrow protein (Sammett et al., 1979). The apparent decrease in toxicity provided by phenobarbital and Arochlor-1254 was attributed to increased detoxifying metabolism of benzene in the liver (Ikeda and Ohtsuji, 1971; ATSDR, 1989); the decrease provided by toluene and aminotriazole was attributed to inhibition of metabolism resulting in a decreased rate of toxic metabolite formation (Hirokawa and Nomiyama, 1962). On the other hand, ethanol ingestion generally increases benzene-induced hematotoxicity, possibly by increasing the rate of formation of toxic metabolites (Driscoll and Snyder, 1984).
Also supporting a causative role of benzene metabolites in bone-marrow suppression and hematotoxicity are studies showing the accumulation of metabolites in bone marrow. Although benzene can be metabolized in bone marrow, mixed-function oxidase activity appears to be insufficient to account for the high concentrations of phenol, hydroquinone, and catechol in bone marrow (ATSDR, 1989). It is postulated,
therefore, that one or more metabolites formed in the liver are transported to the bone marrow where they accumulate and produce a metabolic impairment expressed as bone-marrow depression (Marcus, 1987). Finally, metabolites of benzene, including benzene oxide, hydroquinone, catechol, and trans, trans-muconaldehyde (a precursor of muconic acid), have been shown to be hematotoxic to animals (Marcus, 1987; ATSDR, 1989).
Immunotoxicity
It has long been suspected that benzene might adversely affect human immune functions. Studies in the early 1900s demonstrated an increased susceptibility of benzene-treated rabbits to tuberculosis and pneumonia (Marcus, 1987). Later, depression of lymphocytes and increased susceptibility to infection became increasingly associated with exposure of workers to benzene. With advances in immunology, alterations in serum immunoglobulin and complement levels were detected in occupationally exposed workers (Marcus, 1987). In 35 painters exposed to benzene, along with toluene and xylene, at concentrations of 3.4-48 ppm, serum IgG and IgA levels were significantly decreased compared with controls, and IgM levels were increased (Lange et al., 1973). Leukocyte agglutinins also were increased in some workers, leading to the suggestion that benzene might cause an allergic blood dyscrasia in some individuals. Other findings in workers, including eosinophilia and leukocyte agglutination associated with granulocytopenia, also suggest that autoimmunity or allergy is responsible for benzene-induced effects on immune function (Goldstein, 1977). Furthermore, auto-immune phenomena and reticulosis were implicated in the pathogenesis of bone-marrow disease.
In C57BL mice, 300-ppm exposures for 6 h/d for 6, 30, or 115 d reduced mitogen-induced proliferation of bone-marrow and splenic B and T lymphocytes and markedly reduced the number of B lymphocytes in bone marrow and spleen and the number of T lymphocytes in thymus and spleen (Rozen and Snyder, 1985). Increased bone-marrow cellularity and numbers of thymic T cells between the 6th and 30th exposure suggested a compensating proliferative response. Thymic lymphoma was observed after 115 exposures. Significant suppression of the pri-
mary antibody response to fluid (FTT) and adsorbed (APTT) tetanus toxoid was observed in Swiss albino mice exposed at 200 ppm for 6 h/d for 10-20 d but was not observed at 50 ppm (Stoner et al., 1981). At concentrations of 400 ppm for 5, 12, or 22 exposures, the primary antibody response to FTT was reduced by 74-89% and that to APTT by 8%, 36%, and 85%, respectively. The secondary antibody response was unaffected at 50, 200, or 400 ppm.
In a study of cell-mediated immunity, host resistance to Listeria monocytogenes was measured in mice exposed to benzene for either 5 d prior to infection (pre-exposure regimen) or for 5 d prior to and 7 d after infection (continuous regimen) (Rosenthal and Snyder, 1985). The pre-exposure regimen at 300 ppm increased splenic bacterial counts (730% of controls) on day 4 but had no effect at 10, 30, or 100 ppm. With the continuous-exposure regimen, bacterial counts were increased at 30, 100, and 300 ppm to 490%, 750%, and 720% of controls, respectively, and were unaffected at 10 ppm. On day 7, bacterial counts were not increased by either regimen, indicating recovery of the immune response. At concentrations of 30 ppm and higher, a concentration-dependent decrease in T and B lymphocytes was observed, with B lymphocytes showing a greater decrease. Tumor resistance, another parameter of cell-related immunity, also is adversely affected by benzene. In male C57BL mice exposed at 100 ppm for 6 h/d, 5 d/w, for 20 exposures and then injected with cells from a virus-induced tumor, 90% developed lethal tumors, compared with 30% of controls (Rosenthal and Snyder, 1986).
Several metabolites of benzene are suspect in benzene's immunotoxicity, but the identity of the causative agent or agents and the mechanism of action have not been established. Catechol, hydroquinone, benzoquinone, and 1,2,4-benzenetriol are cytotoxic to spleen cells, reduce the number of progenitor cells from the spleen and bone marrow, or suppress T- and B-lymphocyte mitogen responses (Irons and Neptun, 1980; Pfeifer and Irons, 1981). Suppression of cell growth and function in the lymphoid system, as in the bone marrow, correlates with the concentration of hydroquinone and catechol, which accumulate in lymphoid tissue following exposure to benzene (Greenlee et al., 1981; Wierda and Irons, 1982; Irons et al., 1982). Also, hydroquinone, benzoquinone, phenol, and catechol suppress microtubule assembly and progenitor cells (Kalf et al., 1987). Inhibition of microtubule function might
result in suppression of phytohemagglutinin-stimulated lymphocyte activation; the inactivation correlates with the ability of the metabolites to undergo sulfhydryl-dependent autoxidation (Irons and Neptun, 1980; Pfeifer and Irons, 1981). It has been suggested that hydroquinone or its terminal oxidation product, p-benzoquinone, might be responsible for these effects (Irons and Neptun, 1980).
Mutagenicity and Genotoxicity
Evidence that benzene is genotoxic to humans comes from epidemiological studies of occupationally exposed workers. These studies show that workers with benzene-induced blood disorders consistently exhibited an increased prevalence of chromosomal aberrations; in workers who were without overt signs of toxicity or were exposed to benzene at low concentrations, the results were less consistent (ATSDR, 1989).
There are extensive reviews of epidemiological studies and case reports of benzene-induced chromosomal aberrations in workers (Snyder et al., 1977; White et al., 1980; Van Raalte and Grasso, 1982; Dean, 1985). A significantly higher number of lymphocytes with unstable chromosomal aberrations were found in 20 men (many with neutropenia) exposed to benzene for 1-20 y than were found in unexposed controls (1.4% exposed vs. 0.6% controls) (Tough and Brown, 1965). In rotogravure workers exposed at 125-532 ppm for 1-22 y, unstable and stable chromosomal aberrations in lymphocytes were significantly increased, compared with controls (Forni et al., 1971a). Similar results were reported in 25 persons (13 men and 12 women), even after recovery from hemopathy (Forni et al., 1971b).
Even at low concentrations, chromosomal aberrations were increased; for example, increases were found in 52 workers exposed at < 10 ppm (estimated time-weighted average exposure 2.1 ppm) for 5 y (Picciano, 1979) and in 22 healthy workers exposed at 13 ppm for 11 y (Sarto et al., 1984). In other studies, significant increases were not found when exposures were <25 ppm (Austin et al., 1988). Some investigators reported increases in the frequency of chromosomal damage and of sister chromatid exchange (SCE) in peripheral blood lymphocytes of workers exposed to concentrations as low as 1 ppm (Dean, 1985). Others failed to detect a statistically significant increase in SCE frequency in
workers exposed to higher concentrations (Watanabe et al., 1980; Clarke et al., 1984; Sarto et al., 1984).
Exposure of CD-1 mice for 22 h/d, 7 d/w for 6 w at 0.04 ppm or 0.01 ppm increased the frequencies of spleen lymphocytes with mutations at the hypoxanthine-guanine phosphoribosyl transferase (hrpt) locus and the frequencies of chromosomal aberrations (chromatid breaks) (Au et al., 1991; Ward et al., 1992). Reduced effects at 1 ppm were attributed possibly to increased glutathione-S-transferase levels at higher doses, resulting in more detoxification metabolites and less putative toxic compounds (Ward et al., 1992). Female mice were found to be more sensitive than males in the Au et al. (1991) and the Ward et al. (1992) studies, but in studies using high doses, male mice were more sensitive than female mice (Uyeki et al., 1977; Choy et al., 1985). Exposure of mice (DBA/2, B6C3F1, and C57BL mice) at 300 ppm for 6 h/d, 5 d/w (regimen 1) or 3 d/w (regimen 2) for 13 w induced a highly significant increase in the frequency of micronucleated polychromatic erythrocytes (Luke et al., 1988a). The magnitude of the increase was strain-specific, with DBA/2 greater than C57BL, and C57BL equal to B6C3F1 mice, but independent of exposure regimen and, except for B6C3F1 mice, independent of exposure duration. In DBA/2 mice, this genotoxic injury to the bone marrow was accompanied by a decreased percentage of polychromatic erythrocytes, indicating depression of erythropoiesis (Luke et al., 1988b).
Carcinogenicity
Epidemiological studies and case reports provide convincing evidence of the carcinogenic (leukemogenic) effects of benzene inhalation (Vigliani, 1976; Infante et al., 1977; Ott et al., 1978; Rinsky et al., 1981; Maltoni et al., 1989). The first epidemiological study of benzene, published in 1974, reported a leukemia incidence during 1967-1973 of 13/100,000 among 28,500 Turkish shoe workers exposed to benzene at concentrations of 150-650 ppm for 4 mo to 15 y (Aksoy et al., 1974). This incidence was significantly higher than the estimated 6/100,000 for the general population, and the incidence decreased after use of benzene was discontinued in 1969 (Aksoy, 1980). A mortality study and continued followup studies of rubber-industry workers ex-
posed at 10-100 ppm for up to 10 y or more reported excessive mortality from myelogenous leukemia and reported a direct correlation between benzene exposure and other forms of leukemia (Infante et al., 1977; Infante, 1978; Rinsky et al., 1981, 1987). Epidemiological studies or case reports of chemical workers or other workers suggested a direct or possible correlation between exposure and excess mortalities from, or the development of, one or more forms of leukemia (Aksoy, 1978; Infante, 1978; Bond et al., 1986; Rinsky et al., 1987). Some of these studies, however, were criticized for inappropriate sampling techniques, exposure determinations, mortality standards, and experimental design (Van Raalte and Grasso, 1982).
IARC (1982), EPA (1989), NIOSH (1977), and others (ATSDR, 1989) concluded that benzene is carcinogenic to humans and is associated with an increased incidence of myelogenous leukemia. The data, however, do not establish an association with other types of leukemia. Several risk-assessment models were developed to estimate the probability of developing leukemia from a particular exposure level, utilizing data from the major epidemiological studies. A number of these assessments, including the Crump and Allen, the Rinsky et al. (and variations), and the White, Infante, and Chu assessments, were critically reviewed (Brett et al., 1989). The models yield widely varying risk estimates, depending on the particular model, the data selected for the model, and the exposure assumptions. In 1987, OSHA established an 8-h permissible exposure limit (PEL) of 1.0 ppm for benzene, relying on the Crump and Allen linear risk assessment, which was based on combined data from three high-quality epidemiological studies (Brett et al., 1989). The assessment projects a risk of 10 excess leukemia deaths per 1000 workers as a result of a 45-y occupational exposure to benzene at 1 ppm.
In 1990, the ACGIH proposed revision of its threshold limit value (TLV) for benzene from 10 ppm to 0.1 ppm, with a skin notation and designation as an Al carcinogen (confirmed human carcinogen) (ACGIH, 1991). The ACGIH based its proposal on: (1) leukemia risk assessments emphasizing NIOSH case control data; (2) exposure levels in a cohort mortality study of leukemia in chemical workers; and (3) exposure levels associated with chromosomal breakage. Recently, the proposed lower limit was defended on the basis of the need to consider effects other than leukemia, chromosomal aberrations in workers at low
exposure levels, and increased toxicity from dermal and intermittent exposures (Infante, 1992).
In animals, benzene by inhalation or gavage is a multipotent carcinogen, capable of producing a variety of neoplasms at several sites. Elevated incidences of tumors are reported in the Zymbal gland, oral cavity, preputial gland, harderian gland, liver, mammary gland, lungs, and ovaries, in addition to lymphomas and leukemias (Huff et al., 1988; Maltoni et al., 1989). Many attempts to induce leukemia in animals yielded negative or debatable results because of difficulties in establishing a suitable animal model (ATSDR, 1989). In an early study of 40 C57BL/6 mice, occurrences of leukemia (1), thymic lymphoma (6), plasmacytoma (1), and bone-marrow hyperplasia (13) were reported after exposures at 300 ppm for 6 h/d, 5 d/w, for life, as compared with the occurrence of two lymphomas (nonthymic) in controls (Snyder et al., 1980). A similar exposure of AKR mice caused bone-marrow hypoplasia, but did not alter the incidence or induction time of viral-induced lymphomas common in this strain.
In subsequent studies by the same laboratory, myelogenous leukemia occurred in 1 of 40 CD-1 mice exposed at 100 ppm and in 2 of 40 exposed at 300 ppm for 6 h/d, 5 d/w for life (Goldstein et al., 1982); it also occurred in 1 of 40 Sprague-Dawley rats similarly exposed at 100 ppm (Snyder et al., 1984). Liver tumors (4 in 40 rats) and Zymbal gland carcinomas (2 in 40 rats) also were observed. The investigators suggested a causative role for benzene because spontaneous myelogenous leukemia had not been observed in these strains. However, the results might be of questionable significance because of the small numbers of animals and marginally increased incidences (Maltoni et al., 1989).
More convincing evidence of the leukemogenicity of benzene was provided by a major series of studies in which animals were exposed 6 h/d, 5 d/w, for 16 w to approximate the average exposure duration of workers (15% of lifetime). In C57BL/6 mice, exposure at 300 ppm resulted in a highly significant increase in lymphoma-leukemia, from 0% (0/90) in controls to 8% (8/90) (Cronkite et al., 1984). The exposure produced a definite pattern of lymphoma and mortality, with the first wave of deaths at 330-390 d of age primarily due to thymic lymphoma and the second wave beginning at 570 d of age due to nonthymic lymphoma and solid tumors. In a recent study from the same labo-
ratory, exposure at 300 ppm 6 h/d, 5 d/w, for 16 w significantly increased the incidence of myelogenous leukemia from 0% in controls to 19.3% in male CBA/Ca mice and of nonhematopoietic nonhepatic neoplasms (Zymbal, harderian, mammary) from 21.7% to 52.6% in males and from 35.0% to 79.6% in females (Cronkite et al., 1989). A similar exposure at 100 ppm did not affect the incidence of myelogenous neoplasms, but did increase the incidence of nonhematopoietic nonhepatic tumors from 20.0% to 44.7% in males.
The Bologna Institute of Oncology conducted several chronic exposure studies in which benzene was administered by inhalation or gavage to various strains of rats and mice (Maltoni et al., 1989). Sprague-Dawley rats were exposed by inhalation either at 200 ppm for 15 w or at 200 ppm for 19 w followed by 300 ppm for 85 w (total of 104 w). Although extensive data from the studies were presented, the data do not appear to be complete for all tumor sites. Positive results were summarized as an increase or marginal increase in the incidence of the various tumor types or sites without statistical analysis. In the Sprague-Dawley rat, inhalation of benzene was reported to be ''associated'' with an increase in the incidence of total malignant tumors and carcinomas of the Zymbal glands and oral cavity and with a marginal increase in the incidence of hepatomas and carcinomas of the nasal cavities and mammary gland (Maltoni et al., 1989). At an exposure of 200 ppm, 4-7 h/d, 5 d/w, for 19 w, followed by an exposure at 300 ppm, 7 h/d, 5 d/w, for 85 w, with exposure started in embryonal life, the incidence of malignant tumors increased from 17.3% in male and female controls to 43.6%, Zymbal gland carcinomas increased from 0.7% to 10.0%, and hepatomas increased from 0.3% to 6.4%. In females, mammary gland tumors increased from 5.4% in controls to 13.8%. The investigators concluded that the carcinogenic effects of benzene increased with increasing doses (daily dose and length of treatment) and that the carcinogenic effect is enhanced when exposure is started during embryonal life (Maltoni et al., 1989).
Many mechanisms have been suggested for the carcinogenicity of benzene. One involves modification by benzene or its metabolites of "immune surveillance," thereby allowing development of unusual cellular species that cause leukemia and other neoplasms in humans (Leong, 1977). Another suggestion is that benzene or its metabolites might act as a promoter, rather than an initiator, by forcing compensatory hema-
topoiesis (regenerative hyperplasia), with the resultant appearance of preleukemic and leukemogenic clones from stem cells exposed to leukemogenic-initiating agents prior to benzene exposure (Harigaya et al., 1981).
One of the favored mechanisms involves the covalent binding of benzene metabolites to cellular macromolecules. Covalent binding to DNA was observed in the livers of rats exposed to benzene vapor (Lutz and Schlatter, 1977). In mice, radiolabeled metabolites were covalently bound to liver, bone marrow, kidney, spleen, blood, and fat; the label was bound to nucleic acids of hematopoietic cells and to nucleic acids and other macromolecules of mitochondria (Gill and Ahmed, 1981). Also, bioactivation of benzene by mitochrondia caused adduct formation with DNA and inhibited the ability of RNA polymerase to transcribe the genome (Kalf et al., 1982).
A recent review of the molecular pathology of benzene points out that its carcinogenic, hematotoxic, cytotoxic, and genotoxic effects are the consequences of highly complex, interactive biological processes (Yardley-Jones et al., 1991). Possible processes include increased production of hydroxyl radicals, generation of oxygen radicals, depletion of endogenous glutathione, activation of protein kinase c (an enzyme involved in cell transformation and tumor promotion), covalent binding to glutathione, protein and other cellular macromolecules, DNA damage, and induction of micronuclei.
Reproductive Toxicity
Studies of the effects of benzene on reproductive functions in humans are limited in number and scope but suggest a possible association between chronic exposure and adverse effects in females. In a study of 30 employed women with symptoms of benzene toxicity (indicative of higher exposure levels than currently allowed), 12 had menstrual disorders (Vara and Kinnunen, 1946). There were two spontaneous abortions and no births during employment, even though no contraceptive measures were taken by the 12 women, 10 of whom were married. Gynecological examinations revealed that the scanty menstruations in five of the women were due to hypoplasia of the ovaries.
Two European studies reported menstrual disturbances (heavy
bleeding) in workers exposed to benzene at concentrations of 31 ppm (Michon, 1965) and ovarian hypofunction in workers in another factory (Pushkina et al., 1968). In another European study of 360 gluing operators, all of whom were women who were exposed to petroleum (containing benzene) and chlorinated hydrocarbons both dermally and by inhalation, no significant difference in fertility between exposed workers and unexposed controls was found, but spontaneous abortion and premature birth increased (Mukhametova and Vozovaya, 1972).
Inhalation studies with animals have demonstrated adverse effects of benzene on the reproductive systems of both sexes, but particularly of males. In CD-1 mice exposed at 1, 10, 30, or 300 ppm, 6 h/d, 5 d/w, for 13 w, exposure at 300 ppm resulted in histopathological changes to the testes and ovaries (Ward et al., 1985). Changes to the testes included atrophy and degeneration, decreases in spermatozoa, and moderate increases in abnormal sperm forms. Pathological changes to the ovaries were less severe and consisted of bilateral cysts. In chronic exposure studies of rabbits and guinea pigs, slight increases in average testicular weight occurred in guinea pigs at 88 ppm 7-8 h/d, 5 d/w, for up to 6 mo and slight histopathological changes to the testes (degeneration of the germinal epithelium) occurred in rabbits at 80 ppm (Wolf et al., 1956).
Developmental Toxicity
There is little information on the developmental toxicity of benzene in humans. It is known, however, that benzene crosses the human placenta and is present in cord blood in amounts equal to those in maternal blood (Dowty et al., 1976).
A few case reports and epidemiological studies of benzene-exposed pregnant workers are available in the literature. The results generally are mixed or inconclusive and do not provide direct evidence of the developmental toxicity or the teratogenicity of benzene. One study reports on two infants with no evidence of chromosomal alterations delivered from a worker who had severe pancytopenia and increased chromosomal aberrations and who had been exposed to benzene during her entire pregnancy (Forni et al., 1971b). In another study, an increased frequency of chromatid and isochromatid breaks and SCE was
found in lymphocytes from 14 children of female workers exposed to benzene during pregnancy; however, the workers were also exposed to other organic solvents (Funes-Cravioto et al., 1977). Other epidemiological studies of pregnant women occupationally exposed to undefined organic solvents or living near waste dumps contaminated with benzene and other carcinogens found no evidence of developmental toxicity or teratogenicity (ATSDR, 1989).
Numerous inhalation studies have shown that benzene is embryotoxic and fetotoxic in animals, as evidenced by increased incidences of resorption, reduced fetal weight, skeletal variation, and altered fetal hematopoiesis (ATSDR, 1989). However, no studies have shown benzene to be teratogenic or embryolethal in animals, even at concentrations that are toxic (reduced weight gain) to the mother. Exposure to benzene vapor adversely affected pregnant rabbits and rats at concentrations above 100 ppm (Green et al., 1978). Maternal toxicity was evidenced by a decrease in maternal-weight gain; the decrease was accompanied by retarded fetal growth. Some investigators found no increase in resorption in rats exposed at 100, 300, or 2200 ppm (Green et al., 1978), or at 10, 50, or 500 ppm (Murray et al., 1979), but others reported increased resorption in rodents, mostly at concentrations above 150 ppm (ATSDR, 1989). Exposures of mice at 500 ppm 7 h/d on gestation days 6-15 (Murray et al., 1979) and at 156 or 313 ppm 24 h/d or 4 h/d on gestation days 6-15 and exposures of rabbits at 313 ppm 24 h/d (Ungvary and Tatrai, 1985) resulted in growth retardation and increased skeletal variants in fetuses, but no malformations. In rats, concentrations of 50-2200 ppm caused decreased fetal weight, but numbers of skeletal variants increased significantly at 125 ppm and higher (Green et al., 1978). No pregnancies were reported in 10 female rats exposed to benzene at 210 ppm for 10-15 d and then joined by two unexposed males (Gofmekler, 1968). Changes in the weights of body organs were also reported at lower exposures, but the data are difficult to interpret because of a lack of any dose-response relationship. Hematopoietic alterations were reported in the fetuses and offspring of pregnant Swiss-Webster mice exposed by inhalation to benzene. At concentrations of 5, 10, or 20 ppm 6 h/d on gestation days 6-15, the number of erythroid colony-forming cells of progeny was markedly decreased, and at 10 and 20 ppm, granulocytic colony-forming cells also were reduced (Keller and Snyder, 1986).
Interaction with Other Chemicals
Ethanol has been shown to consistently increase the hematotoxicity of benzene in animals. In mice, decreased blood cell counts and bone-marrow cellularity induced by benzene were further reduced by ethanol administration (Seidel et al., 1990). The administration of ethanol increased the depression of hematopoietic progenitor cells, CFU-E, BFU-E, and CFU-C, induced by exposure to benzene at 300 or 900 ppm in BDF1 mice (Seidel et al., 1990).
TABLE 2-1 Toxicity Summary
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
<10 |
5 y |
Human (workers) (n= 52) |
Increased chromosomal aberrations |
Picciano, 1979 |
|
10-100 |
Up to 10 y |
Human (workers) (large n) |
Excessive mortality from myelogenous leukemia in followup studies |
Infante et al., 1977; Infante, 1978; Rinsky et al., 1981, 1987 |
|
11-1069 |
6-60 mo |
Human (workers) (n = 332) |
Severe cytopenia in 23 workers |
Goldwater, 1941 |
|
13 |
11 y |
Human (workers) (n = 22) |
Increased chromosomal aberrations |
Sarto et al., 1984 |
|
25-20,000 |
5 min to 8 h |
Human (estimates) |
No effects at 25 ppm, 8 h; headache, lassitude, weakness at 50-150 ppm, 5 h; effects tolerated at 3000 ppm, 0.5-1.0 h; dangerous to life at 7500 ppm, 0.5 h; can be fatal at 20,000 ppm, 5 or 10 min |
Von Oettingen, 1940 |
|
30-210 |
3 mo to 17 y |
Human (workers) (n = 217) |
Pancytopenia in 6; 45 others with hematological abnormalities |
Aksoy et al., 1971 |
|
3.4-49 |
N.S. |
Human (workers) (n = 35) |
Serum IgG and IgA decreased, IgM increased; leukocyte agglutinins increased in some; other solvent exposures were involved |
Lange et al., 1973 |
|
Up to 500 (100. average) |
N.S. |
Human (workers) (n = 1104) |
Severe pancytopenia in 25, hematological disorders in 83 others |
Wilson, 1942 |
|
125-532 |
1-22 y |
Human (workers) |
Increased unstable and stable chromosomal aberrations in lymphocytes |
Forni et al., 1971a |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
150-650 (maximum) |
4 mo-15 y |
Human (workers) (n = 28,500) |
Pancytopenia in 44, aplastic anemia in 32 with 8 deaths due to thrombocytopenic hemorrhage and infection; 6 developed leukemia within 6 y of followup |
Aksoy et al., 1972; Aksoy and Erdem, 1978 |
|
0.04, 0.01, 1.0 |
22 h/d, 7 d/w, 6 w |
Mouse (CD-1) |
Increased no. of spleen lymphocytes with mutations at hrpt locus and of chromatid breaks (effects reduced at 1.0 ppm) |
Au et al., 1991; Ward et al., 1992 |
|
1, 10, 30, 300 |
6 h/d, 5 d/w, 13 w |
Rat (Sprague-Dawley) |
No effects at 1, 10, 30 ppm; at 300 ppm, decrease in WBCs, percentage of lymphocytes, and decreased marrow cellularity |
Ward et al., 1985 |
|
1, 10, 30 |
6 h/d, 5 d/w, 13 w |
Mouse (CD-1) |
No histopathology to testes or ovaries |
Ward et al., 1985 |
|
300 |
6 h/d, 5 d/w, 13 w |
Mouse (CD-1) |
Atrophy and degeneration of testes, decrease in spermatozoa, moderate increase in abnormal sperm; bilateral cysts in ovaries |
Ward et al., 1985 |
|
1, 10, 30 |
6 h/d, 5 d/w, 13 w |
Mouse (CD-1) |
No hematological changes |
Ward et al., 1985 |
|
300 |
6 h/d, 5 d/w, 13 w |
Mouse (CD-1) |
Increase in mean cell volume, Hb; decrease in hematocrit, Hb, RBC, WBC, platelet counts, percentage of lymphocytes; histopathology to bone marrow, lymphoid tissue, and spleen |
Ward et al., 1985 |
|
4, 20 |
5.5 mo |
Rat |
Conditioned reflex response time delayed at 20 ppm but not affected at 4 ppm |
Novikov, 1956 |
|
5, 10, 20 |
6 h/d on d 6-16 of pregnancy |
Mouse (Swiss Webster) |
No. of CFU-E markedly reduced in progeny; at 10 and 20 ppm, granulocytic colony-forming cells also reduced |
Keller and Snyder, 1986 |
(Table continued from previous page)
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
10-28 |
4-6 h |
Mouse |
Induced SCE in peripheral blood lymphocytes, bone marrow, and micronuclei in bone-marrow polychromatic erythrocytes |
Erexson et al., 1985 |
|
10, 25, 100, 300, 400 |
6 h/d, 5 d/w, up to 16 w |
Mouse (C57BL) |
Bone-marrow cellularity and pluripotential stem cells reduced at 100 ppm (2 w) but not at 10 and 25 ppm; peripheral blood lymphocytes not affected at 10 ppm but reduced at 25-400 ppm |
Cronkite et al., 1985 |
|
10, 30 |
6 h/d, 6 d |
Mouse (male C57BL) |
Femoral LPS-induced B-lymphocyte colony-forming ability depressed at 10 ppm; splenic PHA-induced blastogenesis depressed at 30 ppm |
Rozen et al., 1984 |
|
10, 30, 100, 300 |
6 h/d, 5 d before infection (pre-exposure regimen) or 5 d before and 7 d after infection (continuous regimen) |
Mouse |
Pre-exposure regimen had no effect at 10, 30, 100 ppm, but 300 ppm increased splenic bacterial counts (Listeria monocytogenes) 730% on d 4; continuous regimen had no effect at 10 ppm, but increased counts at 100 and 300 ppm |
Rosenthal and Snyder, 1985 |
|
20, 50, 100, 300 |
8 h/d, 7 d |
Rat (female Wistar) |
Peripheral leukocytes depressed at all concentrations except 20 ppm; leukocyte AP levels increased at 100 and 300 ppm and unaffected at 20 and 50 ppm. |
Li et al., 1986 |
|
21 |
4-10 d continuous or 8 h/d, 5 d/w, 2 w |
Mouse (NMRI) |
Continuous 4-10 d exposures decreased bone-marrow cellularity and no. of CFU-C; 2-w intermittent exposures decreased CFU-C; no such effects found in 8-w exposures at 10 ppm. |
Toft et al., 1982 |
|
44, 47, 61, 65, 831 |
Variable |
Rat |
Leukopenia within 2-4 w at 61, 65, and 831 ppm and after 5-8 w at 44 and 47 ppm. |
Deichmann et al., 1963 |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
50, 200, 400 |
6 h/d, 10-20 d |
Mouse (Swiss albino) |
Suppression of primary antibody response to fluid (FTT) and adsorbed (APTT) tetanus toxoid at 200 ppm (10-20 d), at 400 ppm (5, 12, or 22 d), but not at 50 ppm; secondary response unaffected at 50, 200, and 400 ppm |
Stoner et al., 1981 |
|
50-2200 |
N.S. |
Rat |
Decreased fetal weight; significant increase in no. of skeletal variants at 125 ppm and above |
Green et al., 1978 |
|
80-88 |
7-8 h/d, 5 d/w, 136, 175, 193 exposed |
Rat, rabbit, guinea pig |
Leukopenia, increased spleen weight, histopathology to bone marrow in rat (136 exposed), rabbit (175 exposed), and guinea pig (193 exposed) |
Wolf et al., 1956 |
|
80-88 |
7-8 h/d, 5 d/w, 6 mo |
Rabbit, guinea pig |
Slight degeneration of germinal epithelium in rabbit; slight increase in testicular weight in guinea pig |
Wolf et al., 1956 |
|
100 |
2 d, continuous |
Mouse |
Leukocytopenia |
Gill et al., 1980 |
|
100 |
6 h/d, 5 d/w, 20 exposed |
Mouse (male C57BL/6) |
Injection of viral tumor cells induced 90% incidence of lethal tumors vs. 30% in controls |
Rosenthal and Snyder, 1986 |
|
100 |
6 h/d, 5 d/w, lifetime |
Rat (Sprague-Dawley) |
Myelogenous leukemia (1/40), liver tumors (4/40), Zymbal gland carcinomas (2/40). |
Snyder et al., 1984 |
|
100 |
6 h/d, 5 d/w, 16 w |
Mouse (CBA/Ca) |
Increased nonhematopoietic nonhepatic tumors from 20.0% in controls to 44.7% in males; no effect on myelogenous leukemia |
Cronkite et al., 1989 |
|
300 |
6 h/d, 5 d/w, w |
16 Mouse (CBA/Ca) |
Significantly increased myelogenous leukemia from 0% in controls to 19.3% in males and of nonhematopoietic nonhepatic tumors from 21.7% to 52.6% in males and from 35.0 to 79.6% in females |
Cronkite et al., 1989 |
|
100, 300 |
6 h/d 5 d/w, lifetime |
Mouse (CD-1) |
Myelogenous leukemia in 1/40 at 100 ppm and in 2/40 at 300 ppm |
Goldstein et al., 1982 |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
100, 300, 900 |
6 h/d, 5 d/w up to 16 w |
Mouse (female BDF1) |
Marked decrease in CFU-E at 100 ppm after 1 w; BFU-E, CFU-S, CFU-C decreased at 300 and 900 ppm |
Seidel et al., 1990 |
|
100,300, 1000, 3000 |
6 h/d, 5 d or Ct = 3000 ppm d |
Mouse (C57BL) |
Increased milk-licking after first wk of exposure at 100 ppm or 300 ppm; food intake, hind-limb grip strength reduced at 1000 and 3000 ppm |
Dempster et al., 1984 |
|
103, 302 |
6 h/d, 5 d/w, up to 26 w |
Mouse (CD-1) |
Marrow and spleen cellularity; GM-CFU-C in spleen reduced at 103 and 302 ppm for 5 d; at 302 ppm for 26 w; CFU-S and GM-CFU-S also reduced |
Harigaya et al., 1981 |
|
156, 313 |
4 h/d, 3 d or 24 h/d, on d 6-15 of pregnancy |
Mouse |
Growth retardation and increased skeletal variants in fetuses but no malformations |
Ungvary and Tatrai, 1985 |
|
200, 300 |
200 for 4-7 h/d, -5 d/w, 19 w 300 for 7 h/d, 5 d/w, 85 w; exposure stated in embryonal life |
Rat (Sprague-Dawley |
Increase in males and females in total malignant tumors from 17.3% (controls) to 43.6%; Zymbal gland carcinomas from 0.7% (controls) to 10.0%; hepatomas from 0.3% (controls) to 6.4%; in females; mammary gland tumors from 5.4% (controls) to 13.8% |
Maltoni et al., 1989 |
|
210 |
continuous, 10-15 d before mating and 3 w after mating |
Rat (female) |
No litters |
Gofmekler, 1968 |
|
300 |
6 h/d, 5 d |
Mouse |
Decreased peripheral blood erythrocytes, lymphocytes |
Snyder et al., 1978 |
|
300 |
6 h/d, 6, 30, or 115 d |
Mouse (male C57BL) |
Reduced mitogen-induced proliferation of bone-marrow and splenic B lymphocytes and splenic T cells; reduced number of B lymphocytes in bone marrow and spleen and of T lymphocytes in thymus and spleen; thymic lymphoma after 115 exposures |
Rozen and Snyder, |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
300 |
6 h/d, 3 d/w or 5 d/w, 13 w |
Mouse (DBA/2, B6C3F1, C57BL) |
Increased frequency of MN-PCE in all strains; in DBA/2, decrease in percent of PCE. |
Luke et al., 1988a,b |
|
300 |
6 h/d, 5 d/w, 16 w |
Mouse (C57BL/6) |
Increased in lymphoma-leukemia from 0/90 (controls) to 8/90 (exposed) |
Cronkite et al., 1984 |
|
300 |
6 h/d, 5 d/w, lifetime |
Mouse (C57BL/6, AKR) |
Leukemia (1/40), thymic lymphoma (6/40), plasmacytoma (1/40), bone-marrow hyperplasia (13/40 vs. nonthymic lymphoma (2/40 control) in C57BL/6; in AKR, bone-marrow hypoplasia, but lymphomas not affected |
Snyder et al., 1980 |
|
300, 900 |
6 h/d, 5 d, followed by 2-w nonexposure before repeat exposure |
Mouse (male CD-1, C57BL) |
Behavior not affected on nonexposure days; on exposure days, eating, grooming more frequent, less inactivity; effects more marked at 300 than 900 ppm |
Evans et al., 1981 |
|
313 |
24 h/d, d 6-15 of pregnancy |
Rabbit |
Growth retardation and increased skeletal variants in fetuses, but no malformations |
Ungvary and Tatrai, 1985 |
|
400 |
6 h/d, intermittently for 9 d or consecutively for 11 d |
Mouse (C57BL) |
Depression of CFU-S |
Harigaya et al., 1981 |
|
1500, 3000, 7000 |
15 min |
Rat (Wistar) |
Increased ectopic ventricular beats induced by coronary ligation or i.v. aconitine at 3000 and 7000 ppm, but not at 1500 ppm |
Magos et al., 1990 |
TABLE 2-2 Exposure Limits Set by Other Organizations
TABLE 2-3 Spacecraft Maximum Allowable Concentrations
|
Duration |
ppm |
mg/m3 |
Target Toxicity |
|
1 h |
10 |
35 |
Immune system |
|
24 h |
3 |
10 |
Immune system |
|
7 d |
0.5 |
1.5 |
Immune system |
|
30 d |
0.1 |
0.3 |
Immune system |
|
180 d |
0.07 |
0.2 |
Immune system (leukemia) |
Rationale for Acceptable Concentrations
Because a large database exists on benzene's toxicity to animals and humans, multiple toxic effects must be considered in setting safe exposure concentrations. The immediate effects of benzene exposure are thought to be due to benzene itself, whereas the delayed effects are caused by toxic metabolites reaching target cells, particularly in the bone marrow. The most important threshold-type effect induced by benzene itself is CNS depression; however, mucosal irritation and cardiac effects also will be considered briefly. The clinical diseases associated with myelotoxicity are caused by benzene metabolites that injure cells in
the bone marrow; the weight of evidence indicates that these effects are more severe with either increasing benzene concentration or increasing exposure time (cumulative-type effects).
Species Extrapolation
Based on work by Sabourin et al. (1992), it appears that mice are the best model for studying the effects induced by benzene metabolites. Analyses of urinary metabolites indicated that both humans and mice have a higher propensity to metabolize benzene to its toxic metabolites, muconic acid and hydroquinone, than do rats, monkeys, or chimpanzees. The ratio of hydroquinone or muconic acid to phenol in the urine of mice was 80% and 300%, respectively, as compared with concentrations in human urine (Henderson et al., 1992). Mice are also the most sensitive rodent species to metabolite-mediated effects, but do not seem to show leukemogenic changes similar to those found in industrial workers (see below). The usual species extrapolation factor of 10 will not be used because that factor compensates for species differences in both metabolism and target-tissue susceptibility. Although the comparative tissue susceptibility of mice and humans is unknown, mice seem to produce more toxic metabolites; hence, a species factor of 3 will be used to extrapolate metabolite-induced effects in mice to human estimates. A factor of 10 will still be used for CNS effects thought to be caused directly by benzene itself. This factor is chosen because of potential differences in tissue susceptibility and in the reduced sensitivity of CNS tests in animals compared with the sensitivity of CNS tests in humans (e.g., performance decrements).
Benzene-Induced Toxicity
The acute irritancy and cardiac arrhythmogenic effects of benzene occur only at concentrations that induce significant CNS effects. In rodents, aconitine-induced cardiac arrhythmias were induced by preadministration of benzene at a few thousand parts per million for 15 min; however, narcotic effects were pronounced in the animals exposed to benzene (Magos et al., 1990). Likewise, in monkeys and cats, cardiac
rhythm disturbances occurred primarily during induction of narcosis (Nahum and Hoff, 1934). Protection against CNS effects should protect against any sort of cardiac sensitization similar to that observed in the experimental models. Benzene apparently is not very irritating; humans have been reported to tolerate a concentration of 3000 ppm for up to 1 h (Flury, 1928), whereas CNS effects (headache, lassitude, and weariness) are estimated to occur in the range of 50-150 ppm (Gerarde, 1960). Concentrations low enough to protect against CNS effects will protect against irritation from benzene vapor.
Metabolite-Induced Toxicity
During longer exposures that are below the CNS threshold, the metabolites of benzene may induce several toxic effects that are mediated through damage to stem cells (and other cells) in the bone marrow. Each toxic effect will be analyzed separately; they include hematological, immunological, and neoplastic effects. The first two effects are particularly important because spaceflight causes loss of red-blood-cell mass and reduced immune function in some cells.
Benzene has also been reported to cause reproductive toxicity, developmental toxicity, and genotoxicity. High (but unknown) concentrations of benzene apparently caused menstrual disorders in 12 of 30 female workers exhibiting other signs of benzene toxicity (Vara and Kinnunen, 1946); however, other studies in humans have not confirmed this finding (Barlow and Sullivan, 1982). Animal studies have shown ovarian and testicular changes in mice after subchronic exposure at 300 ppm, but nonreproductive effects (e.g., hematological) occurred much earlier in the same study (Ward et al., 1985). Benzene concentrations low enough to protect against myelogenic toxicity also are not likely to produce any reproductive effects. No convincing human or animal data exist to show that inhalation exposures to benzene cause developmental abnormalities except at concentrations that are toxic to the mother. Benzene is clearly a genetic toxicant; however, the clinically recognized effects that it causes (hematotoxicity, immunotoxicity, and leukemia) are largely mediated through genetic mechanisms; hence, the genotoxic effects of benzene will not be analyzed separately.
Nervous System Effects
The early literature contains reports on the CNS effects induced by inhaling high concentrations of benzene for brief periods (Flury, 1928; Von Oettingen, 1940). Unfortunately, these human data are not sufficiently specific in terms of exposure concentrations, number of test subjects, and sensitivity of end points to be useful in setting acceptable concentrations for benzene. Animal studies typically focus on the myelogenic effects; however, Demster et al. (1984) reported that hind-limb grip strength was diminished 12% in mice exposed for 6 hr to benzene at 1000 ppm, but ten 6-hr exposures at 300 ppm produced no significant effect. Using 300 ppm as the no-observed-adverse-effect level (NOAEL) and applying a species extrapolation factor of 10, gave an acceptable concentration (AC) for benzene exposure of 30 ppm. This value seems to be below the estimated threshold (50-150 ppm) for induction of CNS effects in humans; therefore, it was adopted for all exposure times from 1 hr to 180 d. (See Table 2-5 at the end of this section.)
Hematological Effects
The hematological effects of benzene (e.g., hemorrhage of skin and mucous membranes and anemia) have been known for almost a century; however, quantitative human data on benzene's ability to induce these effects are not available. Most human studies have focused on the leukemogenic properties of benzene (considered below). Suitable data from three studies of mice were used to estimate safe human exposure limits to protect against hematological effects. For reasons cited above, the species extrapolation factor was 3; however, because a 10% loss in red-blood-cell mass is typical during spaceflight, a spaceflight factor of 3 was also applied. From the study by Dempster et al. (1984), two 6-h exposures at 300 ppm did not induce hematological effects. The human limits were calculated as follows:
AC (1 h) = 300 ppm x 1/3 x 1/3 = 33 ppm.
AC (24 h) = 300 ppm x 1/3 x 1/3 x 12/24 = 16 ppm.
Extrapolation to longer exposures is not reasonable from these data; for longer exposures, the data of Green et al. (1981b) were used. They found that 50 6-h exposures at 10 ppm did not induce hematological effects. This information was used to estimate human exposure limits as follows:
AC (7 d) = 10 ppm x 1/3 x 1/3 = 1.1 ppm.
AC (30 d) = 10 ppm x 1/3 x 1/3 x 50 x 6/720 = 0.5 ppm.
Extrapolation to 180 d was not necessary because data from Toft et al. (1982) show that an 8-w continuous exposure at 10 ppm caused no measurable changes in bone-marrow cellularity. From this 56-d exposure, the human limit was estimated as follows:
AC (180 d) = 10 ppm x 1/3 x 1/3 x 56/180 = 0.3 ppm.
This estimate was only slightly below the 30-d AC, suggesting that concentrations in the few tenths of a parts-per-million range approach a no effect level even when exposures are very long.
Immunological Effects
Dempster et al. (1984) reported that five 6-h exposures to benzene at a concentration of 100 ppm induced a 30% reduction in circulating lymphocytes in mice. No significant change was detected after a single 6-h exposure; hence, the NOAEL was 100 ppm for 6 h. From this finding, the short-term ACs were derived as follows:
AC (1 h) = 100 ppm x 1/3 x 1/3 = 11 ppm.
AC (24 h) = 100 ppm x 1/3 x 1/3 x 6/24 = 3 ppm.
Since the immunological effects, which are similar (or greater) in mice and humans, were presumably induced by benzene's toxic metabolites, the species factor was only 3. Likewise, it was concluded that a spaceflight factor of 3 was appropriate because of the numerous reports of spaceflight effects on immune function in rats and, to a lesser extent, in astronauts (Lesnyak et al., 1993; Taylor, 1993). At landing, shuttle
astronauts show depression in blast-cell transformation, changes in cytokine function, and depression of peripheral T-inducer, T-cytotoxic, and natural-killer cells (Taylor, 1993). The changes noted in animals include altered lymphocyte blastogenesis, cytokine function, killer-cell activity, and colony-stimulating factors (Lesnyak et al., 1993).
Rosenthal and Snyder (1985) showed that 12 6-h exposures (72 h total) to benzene at 10 ppm did not increase the susceptibility of mice to infection by Listeria monocytogenes. This observation was used to calculate ACs for 7 d and 30 d of exposure as follows:
AC (7 d) = 10 x 1/3 x 1/3 x 72/168 = 0.5 ppm.
AC (30 d) = 10 ppm x 1/3 x 1/3 x 72/720 = 0.1 ppm.
These were the lowest of the immunotoxicity ACs and were also the lowest for any toxic effect known to be caused by benzene.
Data were available to set 7-d and 30-d ACs from Green et al. (1981b). They found that mice exposed to benzene at 9.6 ppm for 6 h/d for 50 d (300 h = 12.5 d total) showed no changes in peripheral blood or bone marrow. Although splenic cellularity and weight changed, these changes are considered adaptive rather than adverse effects, because the direction of splenic changes in heavily exposed mice was opposite to the changes noted in mice exposed at 9.6 ppm. This information was used to calculate ACs as follows:
AC (7 d) = 9.6 ppm x 1/3 x 1/3 = 1.1 ppm.
AC (30 d) = 9.6 ppm x 1/3 x 1/3 x 300/720 = 0.4 ppm.
No long-term exposure data are available on the immunological effects of benzene exposure; however, Haber's rule can be used to extrapolate a value of 0.07 ppm for continuous exposures of 180 d.
Risk of Leukemia
The risk of death from leukemia induced by occupational and environmental exposure to benzene vapor has been estimated by many investigators. Most estimates have been based on positive epidemiological findings from relatively high industrial exposures; however, other
estimates have been based on ''detection limits'' of negative epidemiological studies or extrapolation from animal data. Numerous controversial issues affect the outcome of the risk estimates. These may be categorized as follows:
- The spectrum of neoplasms that can be ascribed to benzene exposure.
- The concentrations found in industrial workers exposed at high concentrations.
- The most appropriate model to extrapolate risk from high- to low-concentration exposures.
- The most appropriate control population from which to judge excess leukemia risk.
The only point of agreement among investigators seems to be that cumulative benzene exposures above 300 ppm-y induce a significant increase in the occurrence of leukemia in workers. This observation is based on the results of a thorough epidemiological study of 1165 workers exposed at two industrial sites in Ohio (Infante et al., 1977). In that study, standard mortality ratios (SMRs) were not statistically above 100 except for the group with an exposure at the highest concentration (>400 ppm-y) and the group with a cumulative exposure at 200-400 ppm-y. In the group exposed at 40-200 ppm-y, the SMR was 322 (95% confidence interval 36-1165). Using such observations to predict a benzene concentration to which crew members could be continuously exposed for 180 d with no more than a 0.01% increase in leukemia is not simple. At the end of this section, there is a chronological summary in Table 2-4 of risk assessments that have been published since the late 1970s for benzene inhalation exposure. The concentrations calculated in this document and shown in Table 2-4 are for continuous 0.5-y exposures and are typically determined from the long-term intermittent exposure predictions provided by the original risk assessors. The values are not the 95% confidence limits; for ease of comparison, only mean or maximum likelihood values are calculated.
Since the mid-1970s, when the threshold limit value (TLV) for benzene was lowered from 25 ppm to 10 ppm (ACGIH, 1991), thought concerning the leukemogenic properties of benzene has evolved. In 1979, the U.S. Environmental Protection Agency (EPA) estimated that
the risk of dying from leukemia as a result of a 70-y exposure to benzene at 1 ppm was 0.024 (EPA, 1979). This value was based on the geometric mean of three risk estimates that varied from 0.014 to 0.046 (Aksoy et al., 1974; Infante et al., 1977; Ott et al., 1978). However, these risk estimates were based on epidemiological studies that had substantial shortcomings. In 1986, the National Research Council's Committee on Toxicology used the EPA values to estimate that a 0.004-ppm lifetime exposure would impart a risk of 0.01% (NRC, 1986). Similarly, the EPA conclusions can be used to calculate a 180-d (0.5-y) exposure concentration (C) that would impart a 0.01% risk:
C (0.5 y) = 1 ppm x 70/0.5 x 0.0001/0.024 = 0.6 ppm.
This value is not the upper confidence limit of a 0.01% risk; it is the most likely value estimated from the cited studies.
In the mid-1980s, the preliminary epidemiological investigations of the rubber workers in Ohio (Infante et al., 1977) were reexamined (Rinsky et al., 1981) and updated (Rinsky et al., 1987). These data have been interpreted in several ways, and several shortcomings have been indicated. The interpretations will be described below; however, no attempt will be made to refine the original author's conclusions.
One of the major shortcomings of the positive epidemiological findings is that worker exposures to benzene must be estimated; hence, the leukemia risk varies depending on variations in the exposure estimates. In 1985, the 5-y occupational exposure (about 1 y of continuous exposure) risk of exposure to benzene at 1 ppm was estimated to range from 0.5/1000 to 2/1000. The range is due to uncertainty about the concentrations of benzene in the exposures of workers in the cohort (Infante and White, 1985). The Ohio cohort and a Michigan cohort (Ott et al., 1978) gave essentially the same risk estimate when the one-hit linear model was applied to the leukemia incidence. Using the geometric average of the range (1/1000), the concentration yielding a risk of 0.01% after a continuous exposure for 0.5 y was calculated as follows:
C (0.5 y) = 1 ppm x 0.0001/0.001 x 1/0.5 = 0.2 ppm.
This value does not take into account statistical uncertainty from the incidence of leukemia or possibly inappropriate modeling.
The question of the link between benzene and cancer was subjected to cancer modeling in which the increase in cancers relative to the background incidence was estimated. One model, based on the concept that both initiation and promotion mechanisms must be considered (the linear nonthreshold model does not model epigenetic cancer mechanisms), predicted that 2 ppb would yield an incidence of 0.00001 for all cancers based on extrapolation from the most sensitive orally dosed rodents (Albert, 1988). This estimate assumes that a concentration that doubled malignant tumors in exposed animals would double the natural incidence of all cancers in humans (0.02). If this calculation is applied to a working lifetime of 45 y (10 y continuous exposure), then the half-year concentration can be calculated for a 0.0001 risk as follows:
C (0.5 y) = 0.002 ppm x 10/0.5 x 0.0001/0.00001 = 0.4 ppm.
This is not an upper limit, and it applies, in principle, to all benzene-induced cancers.
In a somewhat similar relative-risk approach, epidemiological data were used to predict the odds ratio of benzene-exposed workers dying of leukemia relative to unexposed workers dying of leukemia (Rinsky et al., 1987). Using a linear low-dose extrapolation, the ratio was estimated to be 1.05 (range 1.01 to 1.09) for lifetime industrial exposure to benzene at 0.1 ppm. Because the odds of males dying of myelogenous leukemia is about 0.002, a 1/20 increase in this value is 0.0001. The half-year continuous-exposure concentration would be calculated as follows:
C (0.5 y) = 0.1 ppm x 0.0001/0.0001 x 10/0.5 = 2.0 ppm.
This number is the most likely value; however, the concentration would be nearly halved if the upper 95% limit of the odds ratio (1.09) were used.
At about the same time as the above analyses were published, the use of a linear low-dose extrapolation was questioned (Grilli et al., 1987). The authors concluded from animal metabolism data that the tumor risk from exposures at 10 ppm seemed to be 2-3 times the risk that would be predicted from incidence data on exposures at 100 ppm. This increase stems from a flattening of the dose-response curve above about
40 ppm, where the high-affinity, low-capacity metabolic activation pathways start to become saturated.
Using a weighted average from four epidemiological studies, Austin et al. (1988) concluded that a 30-y lifetime occupational exposure (6.6 continuous years) to benzene at 1 ppm would impart an excess risk of death from leukemia of 0.005. Again the authors used a linear extrapolation to lower concentrations from conclusions reached at much higher concentrations. The prediction for the condition of 0.5 y of continuous exposure is as follows:
C (0.5 y) = 1 ppm x 6.6/0.5 x 0.0001/0.005 = 0.3 ppm.
Again, this value is not based on the upper-confidence interval of the risk; if it were, the value would be about 0.1 ppm. It includes only deaths from leukemia, even though the analysis by Rinsky et al. (1987) suggested that multiple myeloma might also be an outcome of benzene exposure.
In an analysis of data on hematopoietic neoplasms in C57BL mice (Snyder et al., 1980), Beliles and Totman (1989) concluded that the maximum likelihood estimate for neoplasm risk in humans was 0.014 for lifetime occupational exposure at 10 ppm. This result was derived by noting that the mice were exposed experimentally to benzene for 15% of their lifetimes and humans working for 45 y (10 y continuous exposure) in a benzene-contaminated environment would be exposed for 14% of their lifetimes. Species extrapolation was done by allometric equations based on surface area or metabolic-rate analyses. It was found that body-weight ratios to the exponent 0.74 provided the best fit to mouse and human data. The half-year risk calculation from this estimate was as follows:
C (0.5 y) = 10 ppm x 10/0.5 x 0.0001/0.014 = 1.4 ppm.
The actual production of myelogenous leukemia in mice by benzene inhalation has not been achieved by any investigator (Farris et al., 1993).
Using both positive and negative epidemiological data and a linear model, Swaen and Meijers (1989) estimated five to six excess deaths from nonlymphatic leukemia per 1000 deaths in workers exposed at 300 ppm-y of benzene (10 ppm x 30 y work exposure, or 6.6 y continuous
exposure). The positive data were from Rinsky et al. (1987) and the negative data were from several massive studies of 34,000-38,000 workers presumably exposed to low concentrations of benzene (and many other chemicals). The authors of the latter studies used the unproven assumption that the low-concentration exposures averaged 50 ppm-y. From their analysis of the positive epidemiological data, the half-year concentration is as follows:
C (0.5 y) = 10 ppm x 6.6/0.5 x 0.0001/0.006 = 2.2 ppm.
This result is similar to the value derived from the original work by Rinsky.
Recognizing the wide range of risk estimates for benzene, Byrd and Barfield (1989) asked whether this uncertainty was due to differences in data, methods, or concept. They concluded that methodological differences contributed most to the uncertainty. As an example, Parodi et al. (1989) argued that benzene lacks promoting potential below 10 ppm; hence, a sublinear response at low-concentration exposures might be predicted relative to exposures in the range of 10-30 ppm. However, they noted that a leveling-off appears in the range of 50-100 ppm, resulting in a sigmoidal dose-response curve.
Brett et al. (1989) examined various assumptions about the concentrations of benzene that the Ohio rubber workers were exposed to and the matching to control populations. Variations in control matching with Rinsky's exposure assumptions gave predicted increases in leukemia death rates in the range of 4.2-6.4/1000 after 45 ppm-y of exposure (equivalent to 10 y continuous exposure at 1 ppm), whereas the exposure assumptions of Crump and Allen (1984) gave a range of leukemia death rates of 0.5-1.6/1000 for the same variations in control matching. Clearly, the exposure assumptions have a larger effect on the estimates of leukemia risk than the control-matching choices. The authors' preference, using conditional logistic regression, gave leukemia death rates of 1.2-1.6/1000 workers (45 ppm-y of exposure) based on the three control-matching options (Brett et al., 1989). Using the average of this range, gives the following half-year continuous exposure limit:
C (0.5 y) = 1 ppm x 10/0.5 x 0.0001/0.0014 = 1.4 ppm.
This value falls between the low-concentration exposure group (<0.6 ppm) and the high-concentration exposure group (>2 ppm).
In 1990, the ACGIH proposed a TLV of 0.1 ppm for benzene (ACGIH, 1991) and that value has been defended on scientific grounds (Infante, 1992). In particular, inhalation exposures at 1 ppm have been noted to induce cytogenic effects in animals and humans. In addition, Infante asserts that the Michigan cohort has provided evidence that low cumulative exposures of benzene can increase the risk of leukemia. Using the TLV (45 working y = 10 y continuous exposure) as a starting point without any quantitative specification of leukemia risk, the half-year concentration can be calculated as follows:
C (0.5 y) = 0. x 10/0.5 = 2 ppm.
This value is at the high end of the allowable concentrations and suggests that the proposed TLV is not especially conservative.
Factors affecting the outcome of benzene risk estimates have been considered by a panel whose general conclusions have been published (Voytek and Thorslund, 1991). The panel preferred the epidemiological data over the animal data for risk assessment; however, the animal data were considered a valuable adjunct to the human exposure results. It was not clear whether updating (for new leukemia deaths) and combining epidemiological studies would provide a more powerful basis for risk assessment. The panel felt that it was important to focus on specific disease entities (e.g., myelogenous leukemia) rather than on more general categories (e.g., leukemia). The linear quadratic model was preferred over linear models based on leukemia and bone-cancer risks from radiation exposure, provided the former was found to be a "sensitive" model at low concentrations. A preference for absolute risk over relative risk was expressed by the panel.
Review of the risk assessments described above shows that they predict a half-year exposure concentration range of 0.2-2.2 ppm to cause a 0.01% increase in the risk of death due to leukemia. Yardley-Jones et al. (1991) concluded, not surprisingly, that without the three highest exposure cases in the Rinsky study, the model was statistically insignificant in predicting leukemia risks at low concentrations. Furthermore, in the parts-per-billion range, the largest degree of uncertainty is due to
dose-response extrapolation rather than to uncertainties in worker exposures in the parts-per-billion range (Snyder et al., 1993a). Moreover, factoring in the four multiple myeloma cases (one expected) found by Rinsky should be considered. The problem is that three-fourths of the cases occurred in the lowest-concentration exposure group (< 40 ppm-y) with a very long latency period. It is difficult to understand why the multiple myeloma cases occurred in the lowest-concentration exposure group with such a preference if the myelomas were caused by exposure to benzene. It was decided not to include these neoplasms in the present risk analysis.
The average of the nine estimates, which are not independent of each other, is 1.2 ppm. As a conservative measure, the lowest of the concentrations expected to impart a risk of no more than 0.01% was selected—that is 0.2 ppm. Higher radiation exposures during spaceflight are inevitable when compared with earth environments. The radiomimetic properties of benzene are well known; hence, it would be prudent to consider the effects as interacting, probably in an additive way. The degree of radiation protection that will be provided to crews is unknown; hence, a reduction by a factor of 3 in benzene's leukemogenic allowable concentration was selected somewhat arbitrarily. Therefore, the AC to protect against excess leukemia for a continuous exposure of 180 d is 0.07 ppm. Using linear extrapolation to shorter times, the ACs for 30, 7, and 1 d were found to be 0.4 ppm, 1.7 ppm, and 12 ppm, respectively. An estimate for 1 h was not considered suitable because the value would be well into the dangerous range based on other toxic effects.
TABLE 2-4 Concentrations Predicted to Increase Leukemia Risk 0.01% in 6 Months of Continuous Exposure
|
Year |
Predicted Concentration ppm |
Basis of the Original Estimate and Comments |
Reference |
|
1979 |
0.6 |
EPA estimates from geometric mean of three epidemiology studies, each with definite shortcomings. |
EPA, 1979 |
|
1985 |
0.2 |
Risk of 5-y occupational exposure at 1 ppm was 0.001, based on Ohio and Dow cohorts. Actual worker exposures were not measured. |
Infante and White, 1989 |
|
1987 |
2.0 |
From an odds ratio (1.05) of 0.1-ppm-exposed workers dying of myelogenous leukemia compared with unexposed workers. |
Rinsky et al., 1987 |
|
1988 |
0.4 |
Considers all tumors in orally dosed rodents, based on the assumption that exposure causing a doubling of tumors in rodents would double natural incidence in humans. Mechanisms are considered in model. |
Albert, 1988 |
|
1988` |
0.3 |
Weighted average of four epidemiology studies with linear extrapolation to low doses. |
Austin et al., 1988 |
|
1989, 1980 |
1.4 |
Allometric extrapolation of mouse hematopoietic neoplasm data to humans. |
Beliles and Totman, 1989; Snyder et al., 1980 |
|
1989 |
2.2 |
Estimates from Cronkite et al. (1989) data. Compares well with risks suggested by negative epidemiology studies. |
Swaen and Meijers, 1989 |
|
1989 |
1.4 |
Author's preference in exposure assumptions and control groups applied to Rinsky's epidemiology data using conditional logistic regression. |
Brett et al., 1989 |
|
1989, 1992 |
2.0 |
Using TLV as a starting basis without specific statement of risk from exposure. |
ACGIH, 1991; Infante, 1992 |
Summary of Toxic Effects
The analysis of toxic effects induced by benzene generally followed guidelines provided to NASA by the NRC Committee on Toxicology (NRC, 1992). A summary of the analysis has been provided in Table 2-5. Important deviations from past practices were the following: (1) A species extrapolation factor of 3 was used rather than 10 for effects caused by metabolites of benzene. (2) For the first time, a spaceflight factor was applied to an immunotoxicant because of the immune-modulating effects of spaceflight. (3) A radiation uncertainty factor was applied because of the leukemogenic properties of benzene and the relatively high radiation exposure of astronauts. (4) Finally, the present analysis of leukemogenic effects deviated from the NRC-recommended linearized multistage model because of uncertainty involving the human epidemiology database and variations in low-dose extrapolation methods used by investigators.
TABLE 2-5 End Points and Acceptable Concentrations
|
|
|
|
Uncertainty Factors |
|
||||||
|
End Point |
Exposure Data |
Species and Reference |
Time |
Species |
Spaceflight |
Acceptable Concentrations, ppm |
||||
|
1 h |
24 h |
7d |
30 d |
180d |
||||||
|
Nervous system toxicity, loss of hind-limb grip strength |
NOAEL at 300 ppm, 10 x 6 h |
MUS (Dempster et al., 1984) |
1 |
10 |
1 |
30 |
30 |
30 |
30 |
30 |
|
Hematotoxicity |
|
|||||||||
|
Anemia |
NOAEL at 300 ppm, 2 x 6 h |
MUS (Dempster et al., 1984) |
1 |
3 |
3 |
33 |
16 |
—a |
— |
— |
|
Hemotoxic effects |
NOAEL at 10 ppm, 50 x 6h |
MUS (Green et al., 1981b) |
1 or HRb |
3 |
3 |
— |
— |
1.1 |
0.5 |
— |
|
|
NOAEL at 10 ppm, 8 w continuous |
MUS (Toft et al., 1982) |
HR |
3 |
3 |
— |
— |
— |
— |
0.3 |
|
Immuntoxicity |
|
|||||||||
|
Decrease in peripheral lymphocytes |
NOAEL at 100 ppm, 6 h |
MUS (Dempster et al., 1984) |
1 or HR |
3 |
3 |
11 |
3 |
— |
— |
— |
|
Resistance to bacterial infection and reduced spleenic lymphocyte count |
NOAEL at 10 ppm, 12 x 6 h |
MUS (Rosenthal and Snyder, 1986) |
1 or HR |
3 |
3 |
— |
— |
0.5 |
0.1 |
— |
|
Decrease in peripheral lymphocytes |
NOAEL at 9.6 ppm, 50 x 6 h |
MUS (Green et al., 1981b) |
1 or HR |
3 |
3 |
— |
— |
1.1 |
0.4 |
0.07 |
|
Leukemia |
Lowest of 0.01% risk estimates at 0.2 ppm, 180 d continuous |
Varieties |
— |
— |
3 rad |
— |
12 |
1.7 |
0.4 |
0.07 |
|
SMAC |
|
10 |
3 |
0.5 |
0.1 |
0.07 |
||||
|
a Extrapolation to these exposure durations produces unacceptable uncertainty in the values. b HR = Haber's rule. |
||||||||||
Recommendations
Benzene ranks among the most well-studied chemicals known to cause toxic effects in humans. Nonetheless, during our efforts to set exposure limits for astronauts, important limitations in the database became apparent; these limitations should be targets of further research on benzene's toxicity. The acceptable concentration for protection from neurotoxicity induced by short-term benzene exposures was based on rodent data. That was because the reported effects in humans appeared to be based on impressions from industrial experience rather than on specific human data. Well-controlled short-term human exposures to assess neurotoxicity (e.g., performance decrements) are needed to place the acceptable concentrations for such effects on a more reliable foundation. Ethical constraints could limit the scope of studies involving controlled human exposures to benzene.
Continuation of scientific investigations into the role of various enzymes and cofactors in the activation of benzene to its myelotoxic products should continue (Snyder et al., 1993b). Discovery of new potentially toxic metabolites, such as 6-hydroxy-trans, trans-2,4-hexadienoic acid, will further elucidate the mechanism of benzene toxicity (Kline et al., 1993). Refinement of toxicokinetic models will lead to better definition of research aims and facilitate comparative toxicity study (Woodruff and Bois, 1993). Taken together, such scientific investigations will result in an improved definition of time and concentration dynamics, particularly in the areas of continuous-vs.-intermittent exposure and low-concentration extrapolation.
The uncertainty factors selected to compensate for ''target-organ'' effects common to both benzene and spaceflight were arbitrarily assigned. Experiments designed to assess the interaction of chemical toxicity and spaceflight-induced changes would be valuable in evaluating the accuracy of such selections. For example, rodent experiments involving concomitant exposure to benzene and spaceflight (or a model of spaceflight) could be designed to show whether spaceflight modulates benzene's hematotoxicity or immunotoxicity. Findings in such experiments would improve the risk assessment and circumvent the need to make arbitrary choices in uncertainty factors.
References
ACGIH. 1991. 1991-1992 Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices. American Conference of Governmental Industrial Hygienists, Cincinnati, Ohio.
Aksoy, M. 1978. Benzene and leukaemia. Lancet i:441.
Aksoy, M. 1980. Different types of malignancies due to occupational exposure to benzene: A review of recent observations in Turkey. Environ. Res. 23:181-190.
Aksoy, M., and S. Erdem. 1978. Followup study on the mortality and the development of leukemia in 44 pancytopenic patients with chronic benzene exposure. Blood 52:285-292.
Aksoy, M., K. Dincol, T. Akgun, S. Erdem, and G. Dincol. 1971. Haematological effects of chronic benzene poisoning in 217 workers. Br. J. Ind. Med. 28:296-302.
Aksoy, M., K. Dincol, S. Erdem, T. Akgun, and G. Dincol. 1972. Details of blood changes in 32 patients with pancytopenia associated with long-term exposure to benzene. Br. J. Ind. Med. 29:56-64.
Aksoy, M., S. Erdem, and G. Dincol. 1974. Leukemia in shoe-workers exposed chronically to benzene. Blood 44:837-841.
Aksoy, M., S. Erdem, G. Erdogan, and G. Dincol. 1976. Combination of genetic factors and chronic exposure to benzene in the aetology of leukemia. Hum. Hered. 25:149-153.
Albert, R.E. 1988. Quantitated carcinogen risks assessment and carcinogen exposure standards. In Risk Assessment and Risk Management of Industrial and Environmental Chemicals, C.R. Cothern, M.A. Mehlman, and W.L. Marcus, eds. Princeton, N.J.: Princeton Scientific.
ATSDR. 1989. Toxicological Profile for Benzene. ATSDR/TP-88/ 03. Agency for Toxic Substances and Disease Registry, Washington, D.C.
Au, W.W., V.M.S. Ramanujam, J.B. Ward Jr., and M.S. Legator. 1991. Chromosome aberrations in lymphocytes of mice after subacute low-level inhalation exposure to benzene. Mutat. Res. 260: 219-224.
Austin, H., E. Delzell, and P. Cole. 1988. Benzene and leukemia. A
review of the literature and a risk assessment. Am. J. Epidemiol. 127:419-439.
Baarson, K., C.A. Snyder, and R.E. Albert. 1984. Repeated exposures of C57B1 mice to 10 ppm inhaled benzene markedly depressed erythropoietic colony formation. Toxicol. Lett. 20:337-342.
Barlow, S.M., and F.M. Sullivan. 1982. Pp. 83-103 in Reproductive Hazards of Industrial Chemicals. London: Academic.
Beliles, R.P., and L.C. Totman. 1989. Pharmacologically based risk assessment of workplace exposure to benzene. Regul. Toxicol. Pharmacol. 9:186-195.
Blank, I.H., and D.J. McAuliffe. 1985. Penetration of benzene through human skin. J. Invest. Dermatol. 85:522-526.
Bois, F.Y., T.J. Woodruff, and R.C. Spear. 1991. Comparison of three physiologically based pharmacokinetic models of benzene disposition. Toxicol. Appl. Pharmacol. 110:79-88.
Bond, G.G., E.A. McLaren, C.L. Baldwin, and R.R. Cook. 1986. An update of mortality among chemical workers exposed to benzene. Br. J. Ind. Med. 43:685-691.
Brett, S.M., J.V. Rodricks, and V.M. Chinchilli. 1989. Review and update of leukemia risk potentially associated with occupational exposure to benzene. Environ. Health Perspect. 82:267-281.
Byrd, D.M., and E.T. Barfield. 1989. Uncertainty in the estimation of benzene risks: Application of an uncertainty taxonomy to risk assessments based on an epidemiology study of rubber hydrochloride workers. Environ. Health Perspectives 82:282-287.
Carpenter, C., C. Shaffer, C. Weil, and H. Smyth. 1944. Studies on the inhalation of 1:3 butatiene; with comparison of its narcotic effect with benzol, toluol, and styrene and a note on the elimination of styrene by the human. J. Ind. Hyg. Toxicol. 26:69-78.
Chang, I.W. 1972. Study on the threshold limit value of benzene and early diagnosis of benzene poisoning. J. Cathol. Med. Coll. 23:429-434.
Choy, W.N., J.T. MacGregor, M.D. Shelby, and R.R. Maronpot. 1985. Induction of micronuclei by benzene in B6C3F1 mice: Retrospective analysis of peripheral blood smears from the NTP carcinogenesis bioassay. Mutat. Res. 143:55-59.
Clarke, M.G., A. Yardley-Jones, A.C. MacLean, and B.J. Dean.
1984. Chromosome analysis from peripheral blood lymphocytes of workers after an acute exposure to benzene. Br. J. Ind. Med. 41: 249-253.
Contreras, C.M., T. Gonzalez-Estrada, D. Zarabozo, and A. Fernandez-Guardiola. 1979. Petit mal and grand mal seizures produced by toluene or benzene intoxication in the cat. Electroenceph. Clin. Neurophysiol. 46:290-301.
Cornish, H.H., and R.C. Ryan. 1965. Metabolism of benzene in non-fasted, fasted, and aryl-hydroxylase inhibited rats. Toxicol. Appl. Pharmacol. 7:767-771.
Cronkite, E.P., J.E. Bullis, T. Inoue, and R.T. Drew. 1984. Benzene inhalation produces leukemia in mice. Toxicol. Appl. Pharmacol. 75:358-361.
Cronkite, E.P., R.T. Drew, T. Inoue, and J.E. Bullis. 1985. Benzene hematotoxicity and leukemogenesis. Am. J. Ind. Med. 7:447-456.
Cronkite, E.P., R.T. Drew, T. Inoue, Y. Hirabayashi, and J.E. Bullis. 1989. Hematotoxicity and carcinogenicity of inhaled benzene. Environ. Health Perspect. 82:97-108.
Crump, K.C., and B.C. Allen. 1984. Quantitative Estimates of the Risk of Leukemia from Occupational Exposure to Benzene. Paper prepared for the Occupational Safety and Health Administration, Washington, D.C.
Dean, B.J. 1978. Genetic toxicology of benzene, toluene, xylenes and phenols. Mutat. Res. 47:75-97.
Dean, B.J. 1985. Recent findings on the genetic toxicology of benzene, toluene, xylenes and phenols. Mutat. Res. 154:153-181.
Deichmann, W.B., W.E. MacDonald, and E. Bernal. 1963. The hemopoietic toxicity of benzene vapors. Toxicol. Appl. Pharmacol. 5:210-224.
Dempster, A.M., H.L. Evans, and C.A. Snyder. 1984. The temporal relationship between behavioral and hematological effects of inhaled benzene. Toxicol. Appl. Pharmacol. 76:195-203.
Dowty, B.J., J.L. Laseter, and J. Storer. 1976. The transplacental migration and accumulation in blood of volatile organic constituents. Pediatr. Res. 10:696-701.
Drew, R.T., and J.R. Fouts. 1974. The lack of effects of pretreatment with phenobarbital and chlorpromazine on the acute toxicity of benzene in rats. Toxicol. Appl. Pharmacol. 27:183-193.
Driscoll, K.E., and C.A. Snyder. 1984. The effects of ethanol ingestion and repeated benzene exposures on benzene pharmacokinetics. Toxicol. Appl. Pharmacol. 73:525-532.
Duvoir, M.R., A. Fabre, and L. Derobert. 1946. [The significance of benzene in the bone marrow in the course of benzene blood diseases.] Arch. Mal. Prof. 7:77-79.
EPA. 1979. Carcinogenic Assessment Groups Final Report of Population Risk to Ambient Benzene Exposures. EPA/450/S-80-004. U.S. Environmental Protection Agency , Washington, D.C.
EPA. 1989. Health Effects Assessment for Benzene. EPA/600/8-89/ 086. U.S. Environmental Protection Agency, Washington, D.C.
Erexson, G.L., J.L. Wilmer, and A.D. Kligerman. 1985. Sister chromatid exchange induction in human lymphocytes exposed to benzene and its metabolites in vitro. Cancer Res. 45:2471-2477.
Evans, H.L., A.M. Dempster, and C.A. Snyder. 1981. Behavioral changes in mice following benzene inhalation. Neurobehav. Toxicol. Teratol. 3:481-485.
Farris, G.M., J. Everitt, R. Irons, and J. Popp. 1993. Carcinogenicity of inhaled benzene in CBA mice. Fundam. Appl. Toxicol. 20:503-507.
Flury, F. 1928. Modern industrial poisonings in pharmacological and toxicological respect. Arch. Exp. Pathol. Pharmakol. 138:65-82.
Forni, A., E. Pacifico, and A. Limonta. 1971a. Chromosome studies in workers exposed to benzene or toluene or both. Arch. Environ. Health 22:373-378.
Forni, A.M., A. Cappellini, E. Pacifico, and E.C. Vigliani. 1971b. Chromosome changes and their evolution in subjects with past exposure to benzene. Arch. Environ. Health 23:385-391.
Funes-Cravioto, F., C. Zapata-Gayon, and B. Kolmodin-Hedman. 1977. Chromosome aberrations and sister chromatid exchange in workers in chemical laboratories and a rotoprinting factory and in children of women laboratory workers. Lancet ii: 322-325.
Gerarde, H.W. 1960. Pp. 97-108 in Toxicology and Biochemistry of Aromatic Hydrocarbons, E. Browning, ed. New York: Elsevier.
Gerarde, H.W. 1962. The aromatic hydrocarbons. Pp. 1219-1225 in Patty's Industrial Hygiene and Toxicology, Vol. 2, 2nd Rev. Ed., D.W. Fassett and D.D. Irish, eds. New York: Interscience.
Ghantous H., and B.R.G. Danielsson. 1986. Placental transfer and
distribution of toluene, xylene and benzene, and their metabolites during gestation in mice. Biol. Res. Pregnancy Perinatol. 7:98-105.
Gill, D.P., and A.E. Ahmed. 1981. Covalent binding of [14C]benzene to cellular organelles and bone marrow nucleic acids. Biochem. Pharmacol. 30:1127-1131.
Gill, D.P., V.K. Jenkins, R.R. Kempen, and S. Ellis. 1980. The importance of pluripotential stem cells in benzene toxicity. Toxicology 16:163-171.
Gofmekler, V.A. 1968. Embryotropic action of benzene and formaldehyde inhalation. Hyg. Sanit. (USSR) 331(3):327-332.
Goldstein, B.D. 1977. Hematotoxicity in humans. J. Toxicol. Environ. Health Suppl. 2:69-105.
Goldstein, B.D., C.A. Snyder, S. Laskin, I. Bromberg, R.E. Albert, and N. Nelson. 1982. Myelogenous leukemia in rodents inhaling benzene. Toxicol. Lett. 13:169-173.
Goldwater, L.J. 1941. Disturbances in the blood following exposure to benzol. J. Lab. Clin. Med. 26:957-973.
Gonasun, L., C. Witmer, J. Kocsis, and R. Snyder. 1973. Benzene metabolism in mouse liver microsomes. Toxicol. Appl. Pharmacol. 26:398-406.
Green, J.D., B.K.J. Leong, and S. Laskin. 1978. Inhaled benzene fetotoxicity in rats. Toxicol. Appl. Pharmacol. 46:9-18.
Green, J.D., C.A. Snyder, J. LoBue, B.D. Goldstein, and R.E. Albert. 1981a. Acute and chronic dose/response effects of inhaled benzene on multipotential hematopoietic stem (CFU-S) and granulocyte/ macrophage progenitor (GM-CFU-C) cells in CD-1 mice. Toxicol. Appl. Pharmacol. 58:492-503.
Green, J.D., C.A. Snyder, J. LoBue, B.D. Goldstein, and R.E. Albert. 1981b. Acute and chronic dose/response effect of benzene inhalation on the peripheral blood, bone marrow, and spleen cells of CD-1 male mice. Toxicol. Appl. Pharmacol. 59:204-214.
Greenlee, W.F., E.A. Gross, and R.D. Irons. 1981. Relationship between benzene toxicity and the disposition of 14C-labelled benzene metabolites in the rat. Chem. Biol. Interact. 33:285-299.
Grilli, S., W. Lutz, and S. Parodi. 1987. Possible implications from results of animal studies in human risk estimations for benzene: Nonlinear dose-response relationship due to saturation of metabolism. J. Cancer Res. Clin Oncol. 113:349-358.
Guberan, E., and P. Kocher. 1971. Pronostic lointain de l'intoxication benzolique chronique: Controle d'une population 10 ans apres l'exposition. Schweiz Med. Worchenscher 101:1789-1790.
Harigaya, K., M.E. Miller, E.P. Cronkite, and R.T. Drew. 1981. The detection of in vivo hematotoxicity of benzene by in vitro liquid bone marrow cultures. Toxicol. Appl. Pharmacol. 60:346-353.
Hirokawa, T., and K. Nomiyama. 1962. Studies on the poisoning by benzene and its homologues: Oxidation rate of benzene in the rat liver homogenate. Med. J. Shinshu Univ. 7:29-39.
Henderson, R.F., P.J. Sabourin, M.A. Medinsky, L.S. Birnbaum, and G.L. Lucier. 1992. Benzene dosimetry in experimental animals: Relevance for risk assessment. Pp. 93-105 in Relevance of Animal Studies to the Evaluation of Human Cancer Risk. New York: Wiley-Liss.
Huff, J.E., W. Eastin, J. Roycroft, S.L. Eustis, and J.K. Haseman. 1988. Carcinogenesis studies of benzene, methyl benzene, and dimethyl benzenes. Ann. N.Y. Acad. Sci. 534:427-440.
IARC. 1982. International Agency for Research on Cancer. Monographs on the evaluation of the carcinogenic risk of chemicals to humans. Some industrial chemicals and dyestuffs. IARC Monogr. Eval. Carcinog. Risk Chem. Man. 29:93-148.
Ikeda, M., and H. Ohtsuji. 1971. Phenobarbital-induced protection against toxicity of toluene and benzene in the rat. Toxicol. Appl. Pharmacol. 20:30-43.
Infante, P.F. 1978. Leukemia among workers exposed to benzene. Tox. Rep. Biol. Med. 37:153-161.
Infante, P.F. 1992. Benzene and leukemia: The 0.1 ppm ACGIH proposed threshold limit value for benzene. Appl. Occup. Environ. Hyg. 7:253-262.
Infante, P.F., and M.C. White. 1985. Projections of leukemia risk associated with occupational exposure to benzene. Am. J. Ind. Med. 7:403-413.
Infante, P.F., R.A. Rinsky, J.K. Waggoner, and R.J. Young. 1977. Leukemia in benzene workers. Lancet. ii: 76-78.
Inoue, O.K., K. Seiji, and M. Kasahira. 1986. Quantitative relation of urinary phenol levels to breath zone benzene concentrations: A factory survey. Br. J. Ind. Med. 43:692-697.
Irons, R.D., and D.A. Neptun. 1980. Effects of the principal hy-
droxy-metabolites of benzene on microtubule polymerization. Arch. Toxicol. 45:297-305.
Irons, R.D., D. Wierda, and R.W. Pfeifer. 1982. The immunotoxicity of benzene and its metabolites. Pp. 37-50 in Carcinogenicity and Toxicity of Benzene, M.A. Mehlman, ed. Princeton, N.J.: Princeton Scientific.
James, J.T., T.F. Limero, H.J. Leano, J.F. Boyd, and P.A. Covington. 1994. Volatile organic contaminants found in the habitable environment of the space shuttle: STS-26 to STS-55. Aviat. Space Environ. Med. 65:851-857.
Kalf, G.F., T. Rushmore, and R. Snyder. 1982. Benzene inhibits RNA synthesis in mitochondria from liver and bone marrow. Chem. Biol. Interact. 42:353-370.
Kalf, G.F., G.B. Post, and R. Snyder. 1987. Solvent toxicology: Recent advances in the toxicology of benzene, the glycol ethers, and carbon tetrachloride. Annu. Rev. Pharmacol. Toxicol. 27:399-427.
Keller, K.A., and C.A. Snyder. 1986. Mice exposed in utero to low concentrations of benzene exhibit enduring changes in their colony forming hematopoietic cells. Toxicology 42:171-181.
Kline, S., J. Forbes-Robertson, V. Lee-Grotz, B.D. Goldstein, and G. Witz. 1993. Identification of 6-hydroxy-trans, trans-2,4-hexadienoic acid, a novel ring-opened urinary metabolite of benzene. Environ. Health Perspect. 101:310-312.
Lange, A., R. Smolik, W. Zatonski, and J. Szymanska. 1973. Serum immunoglobulin levels in workers exposed to benzene, toluene and xylene. Int. Arch. Arbeitsmed. 31:37-44.
Leong, B.K. 1977. Experimental benzene intoxication. J. Toxicol. Environ. Health Suppl. 2:45-61.
Lesnyak, A.T., G. Sonnenfeld, M.P. Rykova, D.O. Meshkov, A. Mastro, and I. Konstantinova. 1993. Immune changes in test animals during space flight. J. Leuk. Biol. 54:214-226.
Li, G.L., N. Yin, and T. Watanabe. 1986. Benzene-specific increase in leukocyte alkaline phosphatase activity in rats exposed to vapors of various organic solvents. J. Toxicol. Environ. Health. 19:581-589.
Longacre, S., J. Kocsis, and R. Snyder. 1981. Influence of strain differences in mice on the metabolism and toxicity of benzene. Toxicol. Appl. Pharmacol. 60:398-409.
Luke, C.A., R.R. Tice, and R.T. Drew. 1988a. The effect of expo-
sure regimen and duration on benzene-induced bone marrow damage in mice. II. Strain comparisons involving B6C3F1, C57B1/6 and DBA/2 male mice. Mutat. Res. 203:273-295.
Luke, C.A., R.R. Tice, and R.T. Drew. 1988b. The effect of exposure regimen and duration on benzene-induced bone marrow damage in mice. I. Sex comparison in DBA/2 mice. Mutat. Res. 203:251-271.
Lutz, W.K., and C.H. Schlatter. 1977. Mechanism of the carcinogenic action of benzene: Irreversible binding to rat liver DNA. Chem. Biol. Interact. 18:241-245.
Marcus, W.L. 1987. Chemical of current interest-Benzene. Toxicol. Ind. Health 3:205-266.
Magos, G.A., M. Lorenzana-Jimenez, and H. Vidrio. 1990. Toluene and benzene inhalation influences on ventricular arrhythmias in the rat. Neurotoxicol. Teratol. 12:119-124.
Maltoni, C., A. Ciliberti, G. Cotti, B. Conti, and F. Belpoggi. 1989. Benzene, an experimental multipotential carcinogen: Results of the long-term bioassays performed at the Bologna Institute of Oncology. Environ. Health Perspect. 82:109-124.
Medinsky, M.A., P.J. Sabourin, G. Lucier, L.S. Birnbaum, and R.F. Henderson. 1989. A physiological model for simulation of benzene metabolism by rats and mice. Toxicol. Appl. Pharmacol. 99:193-206.
Michon, S. 1965. Disturbances of menstruation in women working in an atmosphere polluted with aromatic hydrocarbons. Pol. Tig. Lek. 20:1648-1649.
Morimoto, K. 1983. Induction of sister chromatid exchanges and cell cycle division delays in human lymphocytes by microsomal activation of benzene. Cancer Res. 43:1330-1334.
Morimoto, K., and S. Wolff. 1980. Increase of sister chromatid exchanges and cell cycle perturbations of cell division kinetics in human lymphocytes by benzene metabolites. Cancer Res. 40:1189-1193.
Mukhametova, I.M., and M.A. Vozovaya. 1972. Reproductive power and the incidence of gynecological disorders in female workers exposed to the combined effect of benzene and chlorinated hydrocarbons. Gig. Tr. Prof. Zabol. 16:6-9.
Murray, F.J., J.A. John, L.W. Rampy, R.A. Kuna, and B.A. Schwetz. 1979. Embryotoxicity of inhaled benzene in mice and rabbits. Am. Ind. Hyg. Assoc. J. 40:993-998.
Nahum, L.H., and H.E. Hoff. 1934. The mechanism of sudden death in experimental acute benzol poisoning. J. Pharmacol. Exp. Ther. 50:336-345.
NIOSH. 1974. Criteria for a Recommended Standard . . . Occupational Exposure to Benzene. National Institute for Occupational Safe ty and Health, Department of Health, Education and Welfare. NIOSH-74-137. National Institute for Occupational Safety and Health, Cincinnati, Ohio.
NIOSH. 1977. Revised Recommendation for an Occupational Exposure Standard for Benzene. National Institute for Occupational Safety and Health, Cincinnati, Ohio.
Nomiyama, K., and H. Nomiyama. 1974a. Respiratory retention, uptake and excretion of organic solvents in man. Benzene, toluene, n-hexane, trichloroethylene, acetone, ethyl acetate and ethyl alcohol. Int. Arch. Arbeitsmed. 32:75-83.
Nomiyama, K., and H. Nomiyama. 1974b. Respiratory elimination of organic solvents in man. Benzene, toluene, n-hexane, trichloroethylene, acetone, ethyl acetate and ethyl alcohol. Int. Arch. Arbeitsmed. 32:85-91.
Novikov, Y.V. 1956. Effects of small benzene concentrations on higher nervous activity of animals in chronic experiments. Gig. Sanit. 21:20.
NRC. 1986. Emergency and Continuous Exposure Guidance Levels for Selected Airborne Contaminants. Benzene and Ethylene Oxide, Vol. 6. Washington, D.C.: National Academy Press.
NRC. 1992. Guidelines for Developing Spacecraft Maximum Allowable Concentrations for Space Station Contaminants. Washington, D.C.: National Academy Press.
Oehme, F. 1969. Ph.D. thesis. Comparative Study of the Biotransformation and Excretion of Phenol. Columbia: University of Missouri. 207 pp.
Ott, M.G., J.C. Townsend, W.A. Fishbeck, and R.A. Langner. 1978. Mortality among workers occupationally exposed to benzene. Arch. Environ. Health 33:3-10.
Parke, D.V., and R.T. Williams. 1953. Studies in detoxication. 49. The metabolism of benzene containing [14C]benzene. J. Biochem. 54:231-238.
Parodi, S., W.K. Lutz, A. Colacci, M. Mazzullo, M. Taningher, and S. Grilli. 1989. Results of animal studies suggest a nonlinear dose-response relationships for benzene effects. Environ. Health Perspect. 82:171-176.
Pfeifer, R., and R. Irons. 1981. Inhibition of lecithin-stimulated lymphocyte agglutination and mitogenesis by hydroquinone: Reactivity with intracellular sulfhydryl groups. Exp. Mol. Pathol. 35:189-198.
Picciano, D. 1979. Cytogenetic study of workers exposed to benzene. Environ. Res. 19:33-38.
Pushkina, N.N., V.A. Gofmekler, and G.N. Klevtsova. 1968. Changes in content of ascorbic acid and nucleic acids produced by benzene and formaldehyde. Bull. Exp. Biol. Med. (USSR) 66:868-870.
Rickert, D.E., T.S. Baker, and J.S. Bus. 1979. Benzene disposition in the rat after exposure by inhalation. Toxicol. Appl. Pharmacol. 49:417-423.
Rinsky, R.A., R.J. Young, and A.B. Smith. 1981. Leukemia in benzene workers . Am. J. Ind. Med. 2:217-245.
Rinsky, R.A., B. Alexander, and M.D. Smith. 1987. Benzene and leukemia: An epidemiological risk assessment. N. Engl. J. Med. 316:1044-1050.
Rosenthal, G.J., and C.A. Snyder. 1985. Modulation of the immune response to Listeria monocytogenes by benzene inhalation. Toxicol. Appl. Pharmacol. 80:502-510.
Rosenthal, G.J., and C.A. Snyder. 1986. Altered T-cell responses in C57B1 mice following sub-chronic benzene inhalation. Toxicologist 61:68.
Rozen, M.G., and C.A. Snyder. 1985. Protracted exposure of C57B1/6 mice to 300 ppm benzene depresses B- and T-lymphocyte numbers and mitogen responses. Evidence for thymic and bone marrow proliferation in response to the exposures. Toxicology 371(2): 13-26.
Rozen, M.G., C.A. Snyder, and R.E. Albert. 1984. Depressions in B- and T-lymphocyte mitogen-induced blastogenesis in mice exposed
to low concentrations of benzene. Toxicol. Lett. 20:343-349.
Sabourin, P.J., B.T. Chen, G. Lucier, L.S. Birnbaum, E. Fisher, and R.F. Henderson. 1987. Effect of dose on the absorption and excretion of [14C]benzene administered orally or by inhalation in rats and mice. Toxicol. Appl. Pharmacol. 87:325-336.
Sabourin, P.J., J.D. Sun, J.T. MacGregor, C.M. Wehr, L.S. Birnbaum, G. Lucier, and R.F. Henderson. 1990. Effect of repeated benzene inhalation exposures on benzene metabolism, binding to hemoglobin, and induction of micronuclei. Toxicol. Appl. Pharmacol. 103:452-462.
Sabourin, P.J., B.A. Muggenburg, R.C. Couch, D. Lefler, G. Lucier, L.S. Birnbaum, and R.F. Henderson. 1992. Metabolism of [14C]-benzene by cynomolgus monkeys and chimpanzees. Toxicol. Appl. Pharmacol. 114:277-284.
Sammett, D., E.W. Lee, J.J. Kocsis, and R. Snyder. 1979. Partial hepatectomy reduces both metabolism and toxicity of benzene. J. Toxicol. Environ. Health 5:785-792.
Sarto, F.S., I. Cominato, A.M. Pinton, P.G. Brovedani, E. Merler, M. Peruzzi, V. Bianchi, and A.G. Levis. 1984. A cytogenetic study on workers exposed to low concentrations of benzene. Carcinogenesis 5:827-832.
Sandmeyer, E.E. 1981. Aromatic hydrocarbons. Pp. 3253-3283 in Patty's Industrial Hygiene and Toxicology, Vol. 2B, 3rd Rev. Ed., G.D. Clayton and F.E. Clayton, eds. New York: John Wiley & Sons.
Sato, A., and T. Nakajima. 1979. Partition coefficients of some aromatic hydrocarbons and ketones in water, blood, and oil. Toxicol. Appl. Pharmacol. 48:49.
Sato, A., and T. Nakajima. 1985. Enhanced metabolism of volatile hydrocarbons in rat liver following food deprivation, restricted carbohydrate intake, and administration of ethanol, phenobarbital, polychlorinated biphenyl, and 3-methylcholanthrene: A comparative study. Xenobiotica 15:67-75.
Sato, A., Y. Fujiwara, and T. Nakajima. 1974. Solubility of benzene, toluene, and m-xylene in various body fluids and tissues of rabbits. Sangyo Igaku 16:30.
Sato, A., T. Nakajima, and Y. Fujiwara. 1975. Kinetic studies on sex difference in susceptibility to chronic benzene intoxication-With
special reference to body fat content. Br. J. Ind. Med. 32:321-328.
Schrenk, H.H., W.P. Yant, and S.J. Pearce. 1941. Absorption, distribution and elimination of benzene by body tissues and fluids of dogs exposed to benzene vapor. J. Ind. Hyg. Toxicol. 23:20-34.
Seidel, H.J., E. Barthel, and D. Zinser. 1989a. The hematopoietic stem cell compartments in mice during and after long-term inhalation of three doses of benzene. Exp. Hematol. 17:300-303.
Seidel, H.J., G. Beyvers, M. Pape, and E. Barthel. 1989b. The influence of benzene on the erythroid cell system in mice. Exp. Hematol. 17:760-764.
Seidel, H.J., R. Bader, L. Weber, and E. Barthel. 1990. The influence of ethanol on the stem cell toxicity of benzene in mice. Toxicol. Appl. Pharmacol. 105:13-18.
Sherwood, R.J. 1972. Benzene: The interpretation of monitoring results. Ann. Occup. Hyg. 15:409-421.
Smyth, H.F., C.P. Carpenter, C.S. Weil, U.C. Pozzani, and J.A. Striegel. 1962. Range-finding toxicity data: List VI. J. Ind. Hyg. Assoc. 231:95-107.
Snyder, C.A. 1987. Benzene. In Ethyl Browning's Toxicity and Metabolism of Industrial Solvents. Vol. 1: Hydrocarbons, 2nd Ed. R. Snyder, ed. New York: Elsevier Science.
Snyder, C.A, B.D. Goldstein, and A.R. Sellakumar. 1978. Hematotoxicity of inhaled benzene to Sprague-Dawley rats and AKR mice at 300 ppm. J. Toxicol. Environ. Health 4:605-618.
Snyder, C.A., B.D. Goldstein, A.R. Sellakumar, I. Bromberg, S. Laskin, and R.E. Albert. 1980. The inhalation toxicology of benzene: Incidence of hematopoietic neoplasms and hematotoxicity in AKR/J and C57B1/6J mice. Toxicol. Appl. Pharmacol. 54:323-331.
Snyder, C.A., J.D. Green, J. LoBue, B.D. Goldstein, C.D. Valle, and R.E. Albert. 1981. Protracted benzene exposure causes a proliferation of myeloblasts and/or promyelocytes in CD-1 mice. Bull. Environ. Contam. Toxicol. 27:17-22.
Snyder, C.A., B.D. Goldstein, A.R. Sellakumar, and R.E. Albert. 1984. Evidence for hematoxicity and tumorigenesis in rats exposed to 100 ppm benzene. Am. J. Ind. Med. 5:429-434.
Snyder, R., E.W. Lee, J.J. Kocsis, and C.M. Witmer. 1977. Bone marrow depressant and leukemogenic actions of benzene. Life Sci. 21:1709-1722.
Snyder, R., S.L. Longacre, C.M. Witmer, and J.J. Kocsis. 1981. Biochemical toxicology of benzene. Pp. 123-153 in Reviews in Biochemical Toxicology 3, E. Hodgson, J.R. Bend, and R.M. Philpot, eds. Amsterdam: Elsevier-North Holland.
Snyder, R., G. Witz, and B.D. Goldstein. 1993a. The toxicity of benzene. Environ. Health Perspect. 100:293-306.
Snyder, R., T. Chepiga, C.S. Yang, H. Thomas, K. Platt, and F. Oesch. 1993b. Benzene metabolism by reconstituted cytochromes P450 2B1 and 2E1 and its modulation by cytochrome b microsomal epoxide hydrolase, and glutathione transferases: Evidence for an important role of microsomal epoxide hydrolase in the formation of hydroquinone. Toxicol. Appl. Pharmacol. 122:172-181.
Srbova, J., J. Teisinger, and S. Skramovsky. 1950. Absorption and elimination of inhaled benzene in man. Arch. Ind. Hyg. Occup. Med. 2:1-8.
Stommel, P., G. Muller, W. Stucker, C. Verkoyen, S. Schobel, and K. Norpoth. 1989. Determination of S-phenylmercapturic acid in the urine-An improvement in the biological monitoring of benzene exposure. Carcinogenesis 10:279-282.
Stoner, R.D., R.T. Drew, and D.M. Bernstein. 1981. Benzene inhalation effect upon tetanus antitoxin responses and leukemogenesis in mice. Pp. 445-461 in Coal Conversion and the Environment, D.D Mahlum, R.H. Gray, and W.D. Felix, eds. Technical Information Center, U.S. Department of Energy, Oak Ridge, Tenn.
Sun, J.D., M.A. Medinsky, L.S. Birnbaum, G. Lucier, and R.F. Henderson. 1990. Benzene hemoglobin adducts in mice and rats: Characterization of formation and physiological modeling. Fundam. Appl. Toxicol. 15:468-475.
Swaen, G.W.H., and Meijers, J.M.M. 1989. Risk assessment of leukaemia and occupational exposure to benzene . Br. J. Ind. Med. 46: 826-830.
Taylor, G.R. 1993. Immune changes during short-duration missions. J. Leukocyte Biol. 54:202-208.
Teisinger, J., V. Bergerova-Fiserova, and J. Kudrna. 1952. The metabolism of benzene in man. Pracov. Lek. 4:175-188.
Tice, R.R., D.L. Costa, and R.T. Drew. 1980. Cytogenetic effects of inhaled benzene in murine bone marrow: Induction of sister chromatid exchanges, chromosomal aberrations and cellular proliferation
inhibition in DBA/2 mice. Proc. Natl. Acad. Sci. USA 77:2148-2152.
Toft, K., T. Olofsson, A. Tunek, and M. Berlin. 1982. Toxic effects on mouse bone marrow caused by inhalation of benzene. Arch. Toxicol. 51:295-302.
Tough, I.M., and W.M. Court Brown. 1965. Chromosome aberrations and exposure to ambient benzene. Lancet i:684.
Travis, C.C., J.L. Quillen, and A.D. Arms. 1990. Pharmacokinetics of benzene . Toxicol. Appl. Pharmacol. 102:400-420.
Ungvary, G., and E. Tatrai. 1985. On the embryotoxic effects of benzene and its alkyl derivatives in mice, rats and rabbits. Arch. Toxicol. Suppl. 8:425-430.
Uyeki, E.M., A.E. Ashkar, D.W. Shoeman, and T.U. Bisel. 1977. Acute toxicity of benzene inhalation to hemopoietic precursor cells. Toxicol. Appl. Pharmacol. 40:49-57.
Valle-Paul, C., and C.A. Snyder. 1986. Effect of benzene exposure on bone marrow precursor cells of splenectomized mice. Toxicologist 61:285.
Van Raalte, H.G.S., and P. Grasso. 1982. Hematological, myelotoxic, clastogenic, carcinogenic, and leukemogenic effects of benzene. Regul. Toxicol. Pharmacol. 2:153-176.
Vara, P., and O. Kinnunen. 1946. Benzene toxicity as a gynecologic problem. Acta Obstet. Gynecol. Scand. 26:433-452.
Vigliani, E.C. 1976. Leukemia associated with benzene exposure. Ann. N.Y. Acad. Sci. 271:143-151.
Vigliani, E.C., and A. Forni. 1976. Benzene and leukemia. Environ. Res. 11:122-127.
Von Oettingen, W.F. 1940. Public Health Bulletin No. 255, U.S. Public Health Service, Washington, D.C.
Voytek, P.E., and T.W. Thorslund. 1991. Benzene risk assessment: Status of quantifying the leukemogenic risk associated with low dose inhalation of benzene. Risk Anal. 11:355-357.
Ward, C.O., R.A. Kuna, N.K. Snyder, R.D. Alsaker, W.B. Coate, and P.H. Craig. 1985. Subchronic inhalation toxicity of benzene in rats and mice. Am. J. Ind. Med. 7:457-473.
Ward, J.B., Jr., M.M. Ammenheuser, V.M.S. Ramanujam, D.L. Morris, E.B. Whorton, Jr., and M.S. Legator. 1992. The mutagenic effects of low level sub-acute inhalation exposure to benzene in CD-1 mice. Mutat. Res. 268:49-57.
Watanabe, T., A. Endo, Y. Kato, S. Shima, T. Watanabe, and M. Ikeda. 1980. Cytogenetics and cytokinetics of cultured lymphocytes from benzene-exposed workers. Int. Arch. Occup. Environ. Health 46:31-41.
White, M.C., P.F. Infante, and B. Walker. 1980. Occupational exposure to benzene: A review of the carcinogenic and related health effects following the United States Supreme Court decision. Am. J. Ind. Med. 1:233-243.
Wierda, D., and R. Irons. 1982. Hydroquinone and catechol reduce the frequency of progenitor B lymphocytes in mouse spleen and bone marrow. Immunopharmacology 4:41-54.
Wilson, R.H. 1942. Benzene poisoning in industry. J. Lab. Clin. Med. 27:1517-1521.
Winek, C.L., and W.D. Collom. 1971. Benzene and toluene fatalities. J. Occup. Med. 131:259-261.
Winek, C.L., W.D. Collom, and C.H. Wecht. 1967. Fatal benzene exposure by glue sniffing. Lancet i:683.
Wolf, M.A., V.K. Rowe, D.D. McCollister, R.L. Hollingsworth, and F. Oyen. 1956. Toxicological studies of certain alkylated benzenes and benzene. AMA Arch. Ind. Health 14:387-398.
Woodruff, T.J., and F.Y. Bois. 1993. Optimization issues in physiological toxicokinetic modeling: A case study with benzene. Toxicol. Lett. 69:181-196.
Woodruff, T., F.Y. Bois, J. Parker, D. Auslander, S. Selvin, M. Smith, and R. Spear. 1989. Design and analysis of a model of benzene toxicokinetics in mammals. Pp. 254-255 in Proceedings of the 11th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, IEEE, Seattle.
Yardley-Jones, A., D. Anderson, and D.V. Parke. 1991. The toxicity of benzene and its metabolism and molecular pathology in human risk assessment. Br. J. Ind. Med. 48:437-444.


































































