17
Physical Performance at High Altitudes
Robert B. Schoene1
INTRODUCTION
Upon ascent to high altitudes, work capacity decreases (Cymerman et al., 1989; Pugh et al., 1964; Sutton et al., 1988; West et al., 1983) (Figure 17-1). After acclimatization at moderate altitudes (3,000 m [9,843 ft]), much of the loss in exercise performance is regained (Buskirk et al., 1967); whereas, at higher altitudes (3,000 to 8,000 m [9,843 to 26,247 ft]), no matter how long the acclimatization period, full recovery of aerobic function is never achieved (Cymerman et al., 1989; Pugh, 1964; Sutton et al., 1988; West et al., 1983). In fact, at extreme altitudes, deterioration occurs that may be complicated by psychological and cognitive impairment (Hornbein et al., 1989). Even at moderate altitudes, factors that impair exercise may be confounded by concomitant maladaptation or altitude illness (see Anand and Chandrashekhar, Chapter 18 in this volume).
The purpose of this chapter is to review the adaptations that optimize the transport of oxygen from the air to the cell in an environment where the barometric
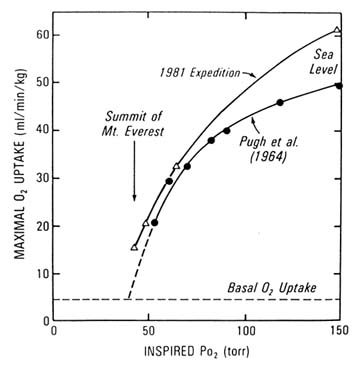
FIGURE 17-1 Data taken from two expeditions to high altitudes showing the decrease in maximal oxygen consumption that occurs with a decrease in inspired partial pressure of oxygen as one ascends to high altitudes. SOURCE: West et al. (1983), used with permission.
pressure is lower and thus the availability of oxygen is less. Additionally, limits to these adaptations will be discussed, and suggestions for areas of further research will be proposed.
ADAPTATIONS AND LIMITS
Ventilation
Minute ventilation (the quantity of air inspired in 1 minute) increases on ascent to high altitudes (Boycott and Haldane, 1908; Dempsey and Forster, 1982; Houston and Riley, 1947; Rahn and Otis, 1949). This response is mediated by stimulation of the carotid body by the low partial pressure of oxygen in the blood (Lahiri et al., 1981; Vizek et al., 1987; Weil, 1986) and
is measured by the increase in ventilation, the hypoxic ventilatory response (HVR) (Weil et al., 1970). For instance, at 6,100 m (20,013 ft), ventilation for a given metabolic rate is almost four times that at sea level (Sutton et al., 1988; West et al., 1983) (Figure 17-2). Extraordinary degrees of hyperventilation have been measured on the summit of Mount Everest, which demonstrates the very high levels of ventilation that can be achieved (West et al., 1983) (Figure 17-3). There is inherent individual variation in this response (Weil et al., 1970), but people who adapt normally at moderate and higher altitudes experience an ongoing augmentation of breathing for 10 to 14 days such that oxygenation of the blood continues to improve with acclimatization (Weil, 1986).
A wealth of data shows that individuals with blunted ventilatory responses are predisposed to altitude illnesses (Hackett and Rennie, 1979; Hackett et al., 1981, 1982, 1988). Almost everyone, however, if they allow enough time for adaptation, will experience an adequate degree of ventilatory augmentation such that altitude illnesses are not experienced. At extreme altitudes, a brisk HVR and subsequent ventilatory response at rest and during exercise convey an advantage in physical performance (Schoene et al., 1984). Although HVR, which is a marker for the overall ventilatory response, can be measured, it is not necessary to screen individuals for tolerance to high altitudes.
Three limitations are inherent in the ventilatory response at high altitude: (1) constraints in lung mechanics, (2) degree of an individual's chemosensitivity and subsequent ventilation, and (3) dyspnea. The first probably is not a
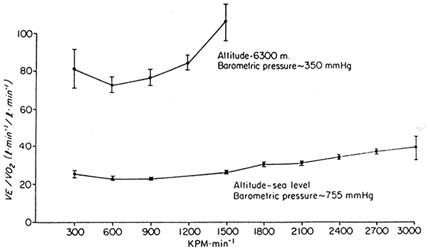
FIGURE 17-2 Ventilatory equivalents (VE/![]() o2) for oxygen consumption during exercise in the same individuals at sea level (bottom line) and 6,300 m (20,669 ft) after 6 weeks of acclimatization. KPM, kilopound meters. SOURCE: Schoene, et al. (1984), used with permission.
o2) for oxygen consumption during exercise in the same individuals at sea level (bottom line) and 6,300 m (20,669 ft) after 6 weeks of acclimatization. KPM, kilopound meters. SOURCE: Schoene, et al. (1984), used with permission.
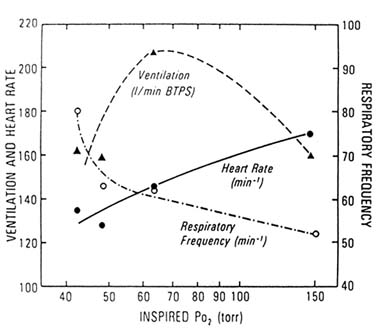
FIGURE 17-3 Respiratory frequency, heart rate, and ventilation as a function of the partial pressure of inspired oxygen. Minute ventilation during maximal exercise increases as the partial pressure of inspired oxygen decreases until an extreme altitude is reached such that a very low work rate and subsequent decrease in ventilatory demand are achieved. Additionally, maximal heart rate decreases in acclimatized individuals. BTPS, Body Temperature Pressure Saturated. SOURCE: West et al. (1983), used with permission.
major factor (Coates et al., 1979); the second has been discussed in the previous paragraph; and the third has not been studied.
Gas Exchange
At sea level, transfer of oxygen from the air to the blood takes place with little or no restriction because perfusion to the lung is well matched to ventilation, and the driving pressure for oxygen from air to blood is high, thus allowing adequate time for equilibration of oxygen from the air to the blood. At high altitude, however, a diffusion limitation exists at the air-blood interface such that the alveolar-arterial oxygen difference is accentuated and is further exaggerated with exercise (Gale et al., 1985; Wagner et al., 1986, 1987; West et al., 1962) (Figure 17-4).
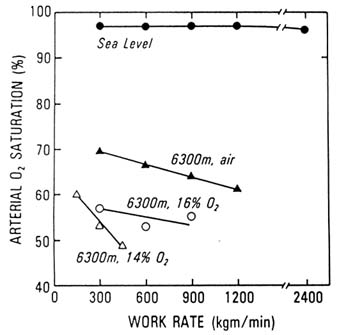
FIGURE 17-4 Arterial oxygen saturation (%) during exercise at sea level and 6,300 m (20,669 ft) with simulated higher altitudes (8,000 and 8,800 m [26,247 and 28,871 ft]). A diffusion limitation for oxygen exists at high and not low altitudes and is accentuated with each increase in altitude.
SOURCE: West et al. (1983), used with permission.
A growing body of research has helped to elucidate why this diffusion limitation exists. Not only is there less oxygen available in the ambient air, but there is also smaller gradient in oxygen at the alveolar-capillary barrier than occurs at low altitudes. The higher one goes, the greater is the drop (Wagner et al., 1987; West et al., 1983). The hypoxic pulmonary vasoconstrictive response (HPVR) results in improved matching of blood flow to ventilation. Nevertheless, the diffusion limitation persists because of (1) a lower driving pressure for oxygen from air to blood, (2) the decreased transit that results from a higher cardiac output (especially with exercise), and (3) the fact that the acquisition of oxygen by hemoglobin takes place on the steep part of the oxygen-hemoglobin dissociation curve (Gale et al., 1985; Wagner et al., 1986, 1987; West et al., 1983).
Cardiac Response
Cardiac output increases upon acute ascent to high altitudes but then decreases to a level that is comparable to sea-level values for any given
metabolic rate (Astrand et al., 1958; Hartley et al., 1967; Klausen, 1966; Pugh, 1964). Myocardial function is well preserved, even at extreme altitudes and in spite of high pulmonary vascular resistance from HPVR and exercise (Groves et al., 1987; Reeves et al., 1987). Upon first ascent, the heart rate at set workloads is higher than at sea level. With adaptation, these heart rates return toward sea level values. Maximum heart rates at very high altitudes, with acclimatization, however, are lower than sea level maximum values (Reeves et al., 1987; West et al., 1983) (Figure 17-3). After much speculation, most physiologists agree that the lower maximum rate is not secondary to hypoxic depression (maximum heart rate) or primary myocardial dysfunction but is merely an appropriate response to the metabolic rate, which is limited by other factors (Reeves et al., 1987). The heart, therefore, appears to function well in the high-altitude environment.
Hematologic Response
Oxygen delivery to the tissues is improved by the hematologic response at high altitudes. This response occurs in two ways: (1) erythropoiesis, which results in greater carrying capacity of the blood to transport oxygen to the tissues (Abbrecht et al., 1972; Erslev, 1953; Faura et al., 1969); and (2) at very high altitudes, a profound respiratory alkalosis contributes to a leftward shift in the oxygen-hemoglobin dissociation curve which optimizes loading of oxygen to the blood at the lung but probably does not impair unloading at the tissues where other factors may facilitate unloading (Winslow et al., 1984). The erythropoietic response is mediated by a rapid increase in erythropoietin but is not manifest by a higher blood hemoglobin for at least 7 to 14 days.
Limitations to this response lie in either an impairment to erythropoiesis from nutritional deficiencies or in an overresponse, resulting in polycythemia, higher blood viscosity, a decrease in perfusion, and subsequent lower oxygen delivery to the peripheral tissues (Aggio et al., 1972; Humphrey et al., 1979; Messmer, 1975; Winslow et al., 1987). The polycythemia may be accentuated by the diuresis that occurs at high altitudes and the tendency of individuals not to take enough fluid, especially with prolonged sojourns. Thus, excessive hyperviscosity may be minimized by ensuring adequate fluid ingestion.
Peripheral Tissues
Utilization of oxygen at the tissue level requires an adequate availability of oxygen to the cells and mitochondria and sufficient substrate for oxidative metabolism. Appropriate responses, therefore, entail: (1) improving blood flow by increasing capillary density, (2) maintaining adequate fuel, and (3) increasing mitochondrial density. What actually happens is not very clear.
Substrate availability (availability of free fatty acids and glycogen) remains good, even at extreme altitudes (Green et al., 1989). An apparent increase in capillary and mitochondrial density with prolonged stay at high altitudes may in fact be merely a reflection of muscle cell atrophy, such that morphometric measurements of capillary and mitochondrial densities may be misleading (Banchero, 1982; Green et al., 1989).
Other limitations may be imposed by diffusion limitation of oxygen from the blood to the tissues. Data at very high simulated altitudes in a hypobaric chamber in Operation Everest II suggest that the low driving pressure for oxygen in the periphery imposes a predictable limitation that is greater the higher one goes (Wagner et al., 1987). This finding is controversial because it is not clear what the minimal level of oxygen is that will allow diffusion of oxygen from hemoglobin, across the cell membrane into the cytoplasm, and into the mitochondria (Hochachka et al., 1983). Some researchers feel there is adequate oxygen available for this process even in extreme hypoxemia, and recent data using nuclear magnetic resonance (NMR) spectroscopy show that in acute hypoxia in the exercising human forearm, phosphocreatine regeneration is not impaired (Conley et al., 1994). More work is necessary to elucidate this critical issue.
Nervous System
The data about oxygen delivery from the air to the tissues do not explain fully why exercise is decreased at high altitudes, so one must turn to the central and peripheral nervous system to look further. It is conceivable that hypoxia may suppress the brain, and the subsequent neural output to the peripheral nervous system and/or hypoxia itself may actually inhibit neuromuscular transmission of impulses. Some data exist that help in part to elucidate this question.
Exposure to extreme altitudes results in at least transient neuropsychometric dysfunction (Hornbein et al., 1989; Regard et al., 1989). Climbers on Mount Everest, as well as subjects on a simulated ascent to Mount Everest in a hypobaric chamber (Operation Everest II), experienced word aphasia (difficulty in speaking) and apraxia (difficulty in manual dexterity), as well as neurological impairment in tasks such as memory recall and rapid finger tapping. In the climbing group, the finger-tapping function remained decreased a year later while the other tests had returned to normal. The subjects from the chamber experiment were not studied again. Interestingly, there was a correlation between the subjects' HVRs and neuropsychometric dysfunction, such that those who had higher HVRs, subsequent higher oxygen saturations at rest, less profound desaturation with exercise, and better physical performance at altitude had more impairment (Hornbein et al., 1989). The authors speculated that those subjects who had greater hyperventilation and therefore
hypocapnia (the fall in carbon dioxide levels that results from hyperventilation) also had more cerebral vasoconstriction and lower oxygen delivery to the brain. This phenomenon may also occur during sleep since more periodic breathing and oxygen desaturation has been documented in those with higher HVRs (Lahiri et al., 1983; White et al., 1987).
Other studies have investigated some of the same variables in climbers with repeated exposure to extreme altitudes (Clark et al., 1983; Jason et al., 1989; Regard et al., 1989). Some of the same impairments are present, but they do not correlate with length of time spent above 8,000 m (26,247 ft), and the retrospective nature of the studies make interpretation of the data tenuous. Impairment in memory was documented in Himalayan climbers (Cavaletti, 1987; Regard et al., 1989), but other investigators have not duplicated these findings (Clark et al., 1983; Jason et al., 1989). Interpretation of these studies must be tempered with the thought that severe environmental hypoxia may be necessary to induce these changes and that there may be individual variability in susceptibility to hypoxia.
Neuromuscular function results from messages from the central nervous system to the peripheral nervous system. In the peripheral nervous system, successful contraction requires transmission of the signal from central sources along the lower motor neuron across the neuromuscular junction. Using both electrically stimulated and voluntary muscle contractions of the ankle flexors, investigators from Operation Everest II, the simulated ascent of Mount Everest, found evidence of central fatigue (evidence of decreased neurologic signals from the central nervous system) as well as decreased transmission along the nerve fiber but preservation of function at the neuromuscular junction (Garner et al., 1990).
More recent data, collected before and after a 1-month residence at less-extreme altitudes (5,000 m [16,404 ft]), showed preservation of peripheral nervous system function but modest inhibition in parameters of central nervous system output (Kayser et al., 1994). The authors thought that these data could explain the decrease in exercise performance and the lower lactate accumulations that have been documented repeatedly at high altitudes. The investigation of nervous system function is still a fertile area and may hold the essential information for the understanding of physical performance at high altitudes.
THE FUTURE
A great deal of work has been done in order to understand the decrease in performance at high altitudes. A better understanding of the transport of oxygen from the hypoxic environment to the cell has emerged, but there are several glaring omissions in this puzzle, which provide areas for future work.
First, most of the exercise studies have been performed with maximum-exercise protocols. There are good reasons for this approach: a wealth of
research over many years at all altitudes, reproducibility, and ease of administration. Although the information gathered from these studies is invaluable, humans at high altitudes perform for hours, days, or months at submaximal levels, which involves a complex interaction of oxygen transport, adequate nutrition, and cerebral and psychologic function. Studies to elucidate the relationship among these factors are obviously more difficult to perform and interpret, but attention must be focused in these areas.
Second, the last topic discussed, that of central and peripheral nervous system integrity, still holds a number of mysteries that need unraveling.
Third, the delivery of oxygen from the blood to the cell and mitochondria is not fully understood, and the proposed mechanism is controversial at best. Further work is necessary in this area as well.
The most important step, however, is the interpretation and integration of these data with the goal of practical application. Fortunately, most investigators in the field are aware of this need, and important advances have been made to facilitate the sojourner's journey to high mountain regions of the world in a healthy and safe manner.
REFERENCES
Abbrecht, P.H., and J.K. Little 1972 Plasma erythropoietin in men and mice during acclimatization to different altitudes. J. Appl. Physiol. 32:54–58.
Aggio, M.C., M.J. Montano, M.T. Bruzzo, and N. Guisto 1972 Possible inefficiency of polycythemia on intolerance to high altitude. Acta Physiol. Latinoam. 22:123–128.
Astrand, P.O., and I. Astrand 1958 Heart rate during muscular work in man exposed to prolonged hypoxia. J. Appl. Physiol. 13:75–80.
Banchero, N. 1982 Capillary density of skeletal muscles in dogs exposed to simulated altitude. Proc. Soc. Exp. Biol. Med. 148:435–439.
Boycott, A.E., and J.S. Haldane 1908 The effects of low atmospheric pressure on respiration. J. Physiol. 37:355–377.
Buskirk, E.R., J. Kollias, R.F. Akers, E.K. Prokop, and E.P. Reategui 1967 Maximal performance at altitude and return from altitude in conditioned runners. J. Appl. Physiol. 23:259–266.
Cavaletti, G., R. Moroony, P. Garavaglia, and G. Tredici 1987 Brain damage after high altitude climbs without oxygen. Lancet 1:101.
Clark, C.F., R.K. Heaton, and A.N. Wiens 1983 Neuropsychological functioning after prolonged high altitude exposure in mountaineering. Aviat. Space Environ. Med. 54:202–207.
Coates, G., G. Gray, A. Mansell, C. Nahmias, A. Powles, J. Sutton, and C. Webber 1979 Changes in lung volume, lung density, and distribution of ventilation during hypobaric decompression. J. Appl. Physiol. 46:752–755.
Conley, K.E., R.B. Schoene, M.L. Bly, and M. Kushmerick 1994 Metabolic recovery from exercise: No flow or diffusion limitations [abstract]. Fed. Am. Soc. Exp. Biol. J. 8(4):A288.
Cymerman, A., J.T. Reeves, J.R. Sutton, P.B. Rock, B.M. Groves, M.K. Malconian, P.M. Young, P.D. Wagner, and C.S. Houston 1989 Operation Everest II: Maximal oxygen uptake at extreme altitudes. J. Appl. Physiol. 66:2446–2453.
Dempsey, J.A., and H.V. Forster 1982 Mediation of ventilatory adaptations. Physiol. Rev. 62:262–347.
Erslev, A.J. 1953 Humoral regulation of red cell production. Blood 8:349–357.
Faura, J., J. Ramos, C. Reynarfarje, E. English, P. Finne, and C. Finch 1969 Effect of altitude on erythropoiesis. Blood 33:668–676.
Gale, G.E., J.R. Torre-Bueno, R.E. Moon, H.A. Saltzman, and P.D. Wagner 1985 Ventilation-perfusion inequality in normal humans during exercise at sea level and simulated altitude. J. Appl. Physiol. 58:978–988.
Garner, S.H., J.R. Sutton, R.L. Burse, A.J. McComas, A. Cymerman, and C.S. Houston 1990 Operation Everest II: Neuromuscular performance under conditions of extreme simulated altitude. J. Appl. Physiol. 68:1167–1172.
Green, H.J., J.R. Sutton, A. Cymerman, P.M. Young, and C.S. Houston 1989 Operation Everest II: Adaptations in skeletal muscle. J. Appl. Physiol. 66:2454–2461.
Groves, B.M., J.T. Reeves, J.R. Sutton, P.D. Wagner, A. Cymerman, M.K. Malconian, P.B. Rock, P.M. Young, and C.S. Houston 1987 Operation Everest II: Elevated high altitude pulmonary resistance unresponsive to oxygen. J. Appl. Physiol. 63:521–530.
Hackett, P.H., and I.D. Rennie 1979 Rales, peripheral edema, retinal hemorrhage, and acute mountain sickness. Am. J. Med. 67:214–218.
Hackett, P.H., I.D. Rennie, R.F. Grover, and J.T. Reeves 1981 Acute mountain sickness and the edemas of high altitude: Common pathogenesis? Respir. Physiol. 46:383–390.
Hackett, P.H., I.D. Rennie, S.E. Hofmeister, R.F. Grover, E.B. Grover, and J.T. Reeves 1982 Fluid retention and relative hypoventilation in acute mountain sickness. Respiration 43:321–329.
Hackett, P.H., R.C. Roach, R.B. Schoene, G.L. Harrison, and W.J. Mills, Jr. 1988 Abnormal control of ventilation in high altitude pulmonary edema. J. Appl. Physiol. 64:1268–1272.
Hartley, L.H., J.K. Alexander, M. Modelski, and R.F. Grover 1967 Subnormal cardiac output at rest and during exercise and residence at 3,100 meters altitude. J. Appl. Physiol. 23:839–848.
Hochachka, P.W., C. Stanley, J. Merkt, and J. Sumar-Kalinowski 1983 Metabolic meaning of elevated levels of oxidative enzymes in high altitude adapted animals: An interpretive hypothesis. Respir. Physiol. 52:303–313.
Hornbein, T.F., B.D. Townes, R.B. Schoene, J.R. Sutton, and C.S. Houston 1989 The cost on the central nervous system of climbing to extremely high altitude. N. Engl. J. Med. 321:1714–1719.
Houston, C.S., and R.L. Riley 1947 Respiratory and circulatory changes during acclimatization to high altitude . Am. J. Physiol. 149:565–588.
Humphrey, P.R.D., J. Marshall, R.W. Ross-Russell, G. Wetherley-Mein, G.H. DuBoulay, T.C. Pearson, L. Symon, and E. Zulakha 1979 Cerebral blood flow and viscosity in relative polycythemia. Lancet 1 (October):873–877.
Jason, J.W., E.M. Parjurkova, and R.G. Lee 1989 High altitude mountaineering and brain function: Neuropsychological testing of members of a Mount Everest expedition. Aviat. Space Environ. Med. 60:170–173.
Kayser, B., M. Narici, T. Binzoni, B. Grassi, and P. Cerretelli 1994 Fatigue and exhaustion in chronic hypobaric hypoxia: Influence of exercising muscle mass. J. Appl. Physiol. 76:634–640.
Klausen, K. 1966 Cardiac output in man at rest and working during and after acclimatization to 3,800 meters. J. Appl. Physiol. 21:609–616.
Lahiri, S., N.H. Edelman, N.S. Cherniack, and A.P. Fishman 1981 Role of carotid chemoreflex in respiratory acclimatization to hypoxemia in goats and sheep. J. Appl. Physiol. 46:367–382.
Lahiri, S., K.H. Merrit, and M.G. Sherpa 1983 Dependence of high altitude sleep apnea on ventilatory sensitivity to hypoxia. Respir. Physiol. 52:281–301.
Messmer, K. 1983 Hemodilution. Surg. Clin. North Am. 55:659–678.
Pugh, L.G. 1964 Cardiac output in muscular exercise at 5,800 meters (19,000 ft). J. Appl. Physiol. 19:441–447.
Pugh, L.G., M.B. Gill, S. Lahiri, J.S. Milledge, M.P. Ward, and J.B. West 1964 Muscular exercise at great altitude. J. Appl. Physiol. 19:431–440.
Rahn, H., and A.B Otis 1949 Man's respiratory response during and after acclimatization to high altitude. Am. J. Physiol. 157:445–482.
Reeves, J.T., B.M. Groves, J.R. Sutton, P.D. Wagner, A. Cymerman, M.K. Malconian, P.B. Rock, P.M. Young, and C.S. Houston 1987 Operation Everest II: Preservation of cardiac functions at extreme altitude. J. Appl. Physiol. 63:531–539.
Regard, M., O. Oelz, P. Brugger, D. Biol, and T. Landis 1989 Persistent cognitive impairment in climbers after repeated exposure to extreme altitude. Neurology 39:210–213.
Schoene, R.B., S. Lahiri, P.H. Hackett, R.M. Peters, Jr., J.S. Milledge, C.J. Pizzo, F.H. Sarnquist, S.J. Boyer, D.J. Graber, K.M. Marct, and J.B. West 1984 Relationship of hypoxic ventilatory response to exercise performance on Mt. Everest. J. Appl. Physiol. 56:1478–1483.
Sutton, J.R., J.T. Reeves, P.D. Wagner, B.M. Groves, A. Cymerman, M.K. Malconian, P.B. Rock, P.M. Young, S.D. Walter, and C.S. Houston 1988 Operation Everest II: Oxygen transport during exercise at extreme simulated altitude. J. Appl. Physiol. 64:1309–1321.
Vizek, M., C.K. Pickett, J.V. Weil 1987 Increased carotid body hypoxic sensitivity during acclimatization to hypobaric hypoxia. J. Appl. Physiol. 63:2403–2410.
Wagner, P.D., G.E. Gale, R.E. Moon, J.R. Torre-Bueno, B.W. Stolp, and H.A. Saltzman 1986 Pulmonary gas exchange in humans exercising at sea level and simulated altitude. J. Appl. Physiol. 61:260–270.
Wagner, P.D., J.R. Sutton, J.T. Reeves, A. Cymerman, B.M. Groves, and M.K. Malconian 1987 Operation Everest II: Pulmonary gas exchange during a simulated ascent of Mt. Everest. J. Appl. Physiol. 63:2348–2359.
Weil, J.V. 1986 Ventilatory control at high altitude. Pp. 703–727 in Handbook of Physiology. The Respiratory System II, Control of Breathing, Part II, N.S. Cherniack and J.G. Widdicombe, eds. Bethesda, Md.: American Physiological Society.
Weil, J.V., E. Byrne-Quinn, I.E. Sodal, W.O. Friesen, B. Underhill, and G.F. Filley 1970 Hypoxic ventilatory drive in normal man. J. Clin. Invest. 49:1061–1072.
West, J.B., S. Lahiri, M.B. Gill, J.S. Milledge, L.G. Pugh, and M.P. Ward 1962 Arterial oxygen saturation during exercise at high altitude. J. Appl. Physiol. 17:617–621.
West, J.B., S.J. Boyer, D.J. Graber, P.H. Hackett, K.H. Maret, J.S. Milledge, R.M. Peters, Jr., C.J. Pizzo, M. Samaja, F.H. Sarnquist, R.B. Schoene, and R.M. Winslow 1983 Maximal exercise at extreme altitudes on Mount Everest. J. Appl. Physiol. 55:688–698.
White, D.P., K. Gleeson, C.K. Pickett, A.M. Rannels, A. Cymerman, and J.V. Weil 1987 Altitude acclimatization: Influence on periodic breathing and chemoresponsiveness during sleep. J. Appl. Physiol. 63:401–412.
Winslow, R.N., M. Samaja, and J.B. West 1984 Red cell function at extreme altitude on Mt. Everest. J. Appl. Physiol. 56:109–116.
Winslow, R.M., C. Monge, E.G. Brown, H.G. Kline, F. Sarnquist, and N.J. Winslow 1985 The effect of hemodilution on O2 transport in high altitude polycythemia. J. Appl. Physiol. 59:1495–1502.












