7
Neutralization Technology for Mustard Agent HD
The NRC Stockpile Committee recommended that the Army accelerate research and development on neutralization-based technologies for the destruction of chemical agents, particularly for use at sites where bulk agents are stored (NRC, 1994b). Neutralization1 employs process conditions that are specific for each type of agent. Thus, a neutralization process for destroying a specific agent or class of agents would not be suitable for treating a wide range of other wastes (e.g., commercial hazardous wastes).
The virtue of neutralization is that it detoxifies HD agent rapidly at low temperature and low pressure. Batch or semibatch processing allows retention of the products from neutralization until testing can verify destruction of the chemical agent. Bench-scale testing indicates that most of the processing equipment for a neutralization process is commercially available. The ability to use equipment already being used in the chemical industry should minimize the time and cost of construction and process startup. The use of standard equipment should also enhance the reliability and ease of maintenance of the facility.
The U.S. Army, like the defense ministries of other nations, has evaluated many different approaches to the neutralization of HD (NRC, 1993; Yang et al., 1992). Intensive testing in the past two years has led to selection of direct hydrolysis with hot water followed by biodegradation of the hydrolysis product as the best candidate for scale-up to a pilot plant demonstration (U.S. Army, 1996b). Within the Army, the Alternative Technologies Program was established to pursue the testing and development of neutralization alternatives. With respect to the AltTech Panel's evaluation of alternative technologies, this Army program office, much like the companies have whose technologies are described in Chapters 4 through 6, have functioned as the technology proponent. For the remainder of this chapter, the Army Alternative Technologies Program will be referenced as the TPC (technology proponent company).
Proposed pilot-scale testing at the Aberdeen site would consist of a single process train, which subsequently would be replicated to scale-up to the full-scale destruction facility for that site. Thus, successful pilot-scale testing would directly provide the technical basis for constructing and operating a full-scale facility at Aberdeen for disposal of the HD agent stored there.
Background to Process Configurations
Although neutralization of HD detoxifies the agent, the resulting hydrolysate requires further treatment prior to final disposal. Treatment of the hydrolysate must destroy both thiodiglycol, which is the major residual in the hydrolysate, and chlorinated volatile organic compounds (VOCs), which originate as impurities in the HD (see Chapter 1). Management of hydrolysate from HD neutralization may be either on-site, through additional treatment following the neutralization process, or off-site, by shipping the hydrolysate to a permitted waste-management facility—a RCRA TSDF (treatment, storage or disposal facility). On-site treatment requires substantially more complex processing than does the neutralization process alone. The primary process considered for on-site treatment of hydrolysate is biodegradation. Aqueous effluent from an on-site biodegradation process potentially could be discharged to the existing federally owned treatment works (FOTW) at Aberdeen or recycled as process water.2
The processing options are further complicated by the possibilities for treating VOCs and recycling water. Separating and treating VOCs prior to on-site biological treatment of the hydrolysate is necessary because the process configuration selected for on-site biodegradation in sequencing batch bioreactors (SBRs) would cause the VOCs to be air-stripped from the hydrolysate and subsequently adsorbed onto the activated carbon filters, rather than being biodegraded. This outcome would result in an unacceptably high rate of use of activated carbon. The Army has proposed using photochemical oxidation to destroy VOCs during on-site treatment of hydrolysate. The primary process considered for off-site management of hydrolysate is shipping it to an off-site TSDF that includes biodegradation as a process step. VOCs in the hydrolysate would also be treated at the TSDF.
Selection of the specific process sequence for use at the Aberdeen site requires consideration of Army programmatic requirements, requirements for shipment to commercial wastewater management facilities or discharge to a FOTW, and regulatory constraints (i.e., permitting requirements). Currently, the policy of the program office for the CSDP (Chemical Stockpile Disposal Program) requires that no liquid effluents be discharged from an agent destruction facility. This policy would have to be modified to make possible either off-site management of the hydrolysate or use of biodegradation followed by discharge of the effluent to a FOTW. In recognition of these policy limitations, the TPC has developed a process configuration of neutralization followed by biodegradation that requires neither shipment of hydrolysate for off-site treatment nor discharge of effluents to a FOTW. However, in the interests of process simplicity and cost-effectiveness, the TPC also has developed several simplified process configurations that may be implemented if CSDP policy is revised. The primary process options are (1) discharging liquids from the process or not and (2) on-site or off-site biodegradation of the hydrolysate.
There are four primary neutralization process configurations under consideration by the Army. Configuration 1 (Figure 7-1 ) is neutralization followed by biodegradation with on-site water recycling and photochemical
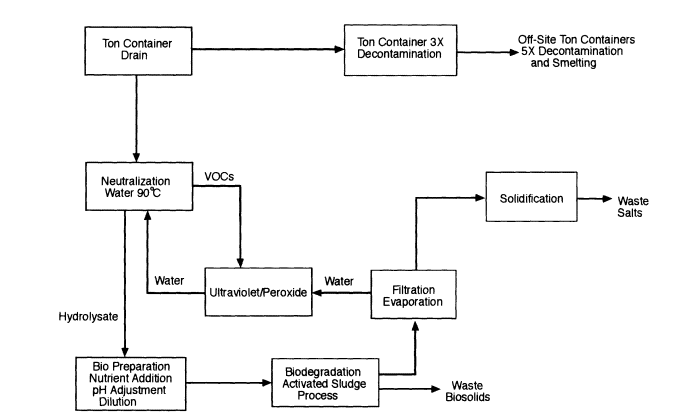
Figure 7-1
Process Configuration 1: Neutralization followed by on-site biodegradation, including water recycling and photochemical oxidation of VOCs.
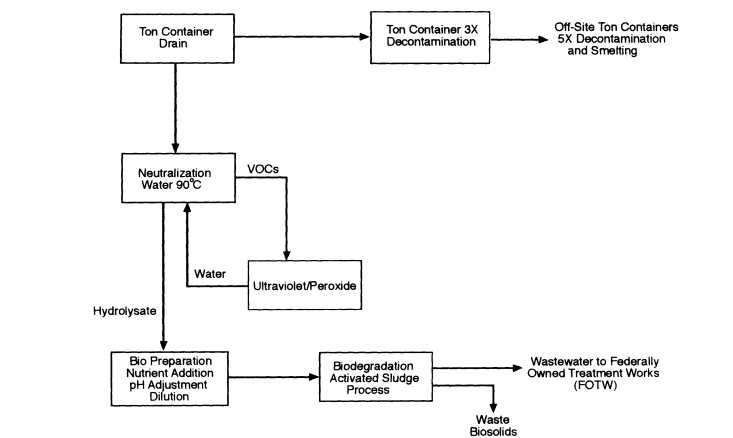
Figure 7-2
Process Configuration 2: Neutralization followed by on-site biodegradation. VOCs are treated by photochemical oxidation. Biodegradation process effluent is discharged to a FOTW.
oxidation to destroy VOCs. This configuration meets the current CSDP policy of discharging no liquid process effluents and fulfills treaty requirements under the CWC (Chemical Weapons Convention).
Configuration 2 (Figure 7-2) is neutralization followed by biodegradation, with process effluents discharged to a FOTW. Photochemical oxidation is used to destroy VOCs to FOTW standards, but water recycling is not used. This configuration fulfills CWC requirements (i.e., it destroys "scheduled precursors") while neutralization effluents are under Army control. The primary difference between configuration 1 and configuration 2 is how process water is managed. In configuration 1, process water is recycled; excess water is lost by evaporation in the cooling tower for water recycle and in the air discharged from biological treatment. In configuration 2, process water is used once (that is, it is not recycled) and then discharged in the aqueous process residual stream to a FOTW.
Configuration 3 (Figure 7-3) is neutralization followed by biodegradation, with process effluents discharged to a FOTW, but VOCs are separated from the hydrolysate and shipped to a TSDF for subsequent treatment and disposal. This configuration also meets CWC requirements by destroying scheduled precursors while neutralization effluents are under Army control, but the process is simplified by eliminating the photochemical oxidation step retained in configuration 2.
Configuration 4 (Figure 7-4) is neutralization followed by shipping the hydrolysate to a TSDF. VOCs in the hydrolysate would be treated at the receiving TSDF in accordance with permit requirements. This is the simplest configuration but requires acceptance of the hydrolysate by a TSDF. Accepting the hydrolysate would subject the TSDF to inspection under the verification requirements of the CWC because destruction of a scheduled precursor (thiodiglycol) would occur at the commercial facility. Configurations 2, 3, and 4 would all require modification of CSDP policy and could have different regulatory permitting requirements.
In a data submission to the panel late in the panel's review process, the TPC chose configuration 2 as its
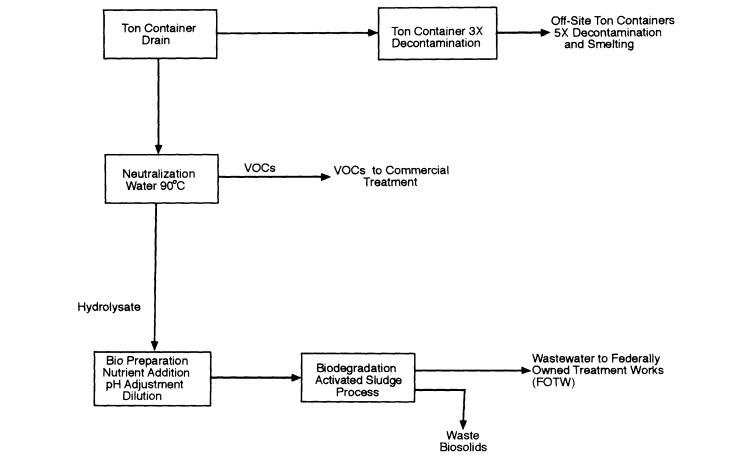
Figure 7-3
Process Configuration 3: Neutralization followed by on-site biodegradation. VOCs are shipped to an off-site TSDF. Biodegradation process effluent is discharged to a FOTW.
preferred candidate for development (U.S. Army, 1996b). However, to provide a complete evaluation of the TPCs technology in this chapter, configuration 1 of the proposed neutralization process is described and discussed in the most detail. Configurations 2, 3 and 4 are simplifications achieved by eliminating specific components of configuration 1 without requiring major modifications of other process steps. These simplified configurations are discussed by comparing by process flow diagrams and mass balances in the appropriate sections of this chapter. The neutralization technology submitted by the TPC for destroying VX nerve agent is discussed in Chapter 8.
Process Description
Figure 7-1 shows the several steps involved in the configuration 1 process for neutralization of HD (U.S. Army, 1996b). The ton container, which contains the agent, is drained into an agent holding tank. The HD then is neutralized by vigorous mixing in water at 90°C (194°F) using a 4 wt pct feed of HD. At the beginning of a reaction batch, the reactor initially contains most of the required hot water. HD is added to the reactor over a period of one hour to minimize the quantity of unreacted agent in the reactor at any given time. The reaction completely destroys the HD and is 90 percent selective to formation of thiodiglycol. During the neutralization reaction, 2 wt pct of hydrochloric acid is produced, resulting in acidic reaction conditions.
Once the reaction is complete, sodium hydroxide (prepared as an 18 wt pct solution) is added to adjust the pH to 12. The dilute processing of HD and the addition of sodium hydroxide after completion of the neutralization reaction are designed to minimize the production of unwanted by-products during reaction. Laboratory testing has indicated that either increased loading of HD (up to about 10 wt pct) during neutralization or adding sodium hydroxide to the hot water prior to introduction
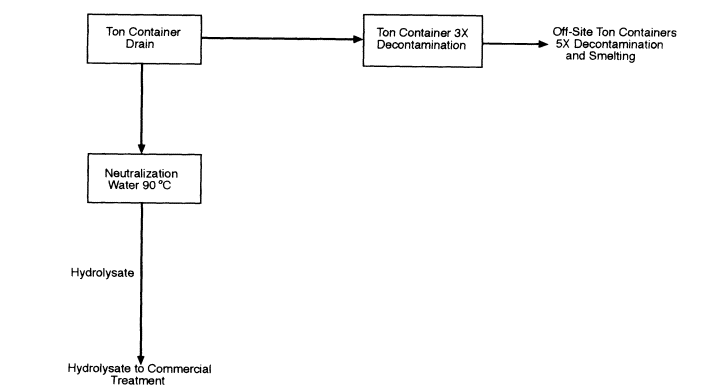
Figure 7-4
Process Configuration 4: Neutralization followed by off-site biodegradation of the hydrolysate at a TSDF. VOCs remain in the hydrolysate.
of HD (rather than after the neutralization reaction is completed) results in lower yields of thiodiglycol and increased concentrations of reaction by-products in the hydrolysate (U.S. Army, 1996b).
The solution resulting from the addition of sodium hydroxide also contains minor amounts of organic impurities that were present in the stored agent and metal salts from corrosion of the storage container or the processing equipment used in the manufacture of the HD (see Chapter 1 for details on impurities in HD). After testing to ensure agent destruction to less than 200 ppb HD,3 the hydrolysate is transferred from the toxic control cubicle for further treatment.
In configuration 4, at this point, the hydrolysate would be shipped to a TSDF. In configurations 1, 2, and 3, the hydrolysate is partially evaporated to remove VOCs such as chlorinated ethylenes. The resulting aqueous condensate contains approximately 0.2 wt pct VOCs. The VOCs are then either shipped to a TSDF (configuration 3) or passed through a photochemical oxidation unit in which hydrogen peroxide is added and the solution is irradiated with ultraviolet light to destroy the organic compounds (configurations 1 and 2).
In configurations 1, 2, and 3, the aqueous solution from the evaporator bottom is adjusted to neutral pH and fed to SBRs which reduce the dissolved organic carbon content by 90 percent and destroy more than 99 percent of the thiodiglycol. This level of destruction meets the CWC definition for destruction of scheduled precursors. The vapor stream from the SBRs is filtered through activated carbon to control odors and remove traces of organic contaminants; it is then released to the atmosphere. The biomass (a thick slurry of solid organic material in water) from the SBRs is fed to an aerobic digester to reduce the volume, then dewatered in a filter press and disposed of as a hazardous waste undercurrent Maryland requirements, even though the dewatered sludge does not contain hazardous concentrations of any constituents. Delisting of this material may allow it to
be disposed of as a nonhazardous solid waste.4 The liquid effluent from the SBRs is filtered and either recycled through the evaporator (configuration 1) or discharged to a FOTW (configurations 2 and 3).
The bottom stream from the evaporator consists of salt brines that are mixed with solidifiers (e.g., cement) and packaged for off-site disposal in a landfill. The evaporator distillate (overhead stream), which is predominantly water containing with low-levels of organic impurities, is recycled to the neutralization operation for use in diluting the next batch of HD.
After HD is drained from a ton container, the empty container is flushed with hot water, cut in half, and cleaned with hot, high pressure water and steam. The cleaned container is then monitored to ensure adequate decontamination from agent and sent to Rock Island Arsenal, Illinois, to be melted.5 The liquid effluent from this cleaning process is used in the neutralization process to replace part of the required process water.
The vapors from the ton container cleanout process, neutralization reactors, and hydrolysate holding tanks are all passed through a single caustic scrubber. Then they are reheated to reduce the relative humidity of the gas; filtered through activated carbon beds, which serve as guard beds to ensure there is no release of toxic organic vapors; filtered through the plant-ventilation activated carbon filter beds; and finally discharged to the atmosphere.
Scientific Principles
The neutralization process proposed for the disposal of HD is, in principle, a simple hydrolysis, that is, a reaction with water to form thiodiglycol (bis(2-hydroxyethyl) sulfide) and hydrochloric acid:
S(CH2CH2Cl)2 + H2O ↔ S(CH2 CH2OH)2 + 2HCl
Even though HD is only slightly soluble in water, the C-Cl bonds, which are essential to mustard's toxicity, react readily in hot water to produce the relatively innocuous thiodiglycol. Pure agent reacts with neutral or acidic water predominantly as shown in the equation above, although the detailed reaction mechanism, as presented in Figure 7-5, is complex (U.S. Army, 1996b; Yang, 1995). The reaction is carried out in hot water, with the final hydrolysate being a dilute aqueous solution (e.g., less than 10 wt pct hydrolyzed agent) to minimize the production of unwanted by-products such as sulfonium ions (R3S+ where R is an organic constituent). The hydrolysis reactions are exothermic, releasing about 15 kilocalories per mole of HD in the neutral-to-acidic hydrolysis (U.S. Army, 1996b).
Under alkaline conditions, much the same chemistry occurs, but it is accompanied by side reactions that give rise to many minor products, some of which are undesirable. Therefore, sodium hydroxide is not added until after the initial reaction, when it is used to neutralize the hydrochloric acid formed in the hydrolysate from the reaction of mustard with water and to react any remaining sulfonium ions. As implied by the equation, the hydrolysis is, in principle, reversible. But the reformation of mustard agent is prevented by adding sodium hydroxide to make the hydrolysate alkaline.
Munitions-grade mustard agent contains several impurities that are formed during manufacture. Even the distilled agent (HD) stored at the Aberdeen Proving Grounds is only 85 to 95 percent pure (U.S. Army, 1996b). Several significant impurities—dithiane and chlorinated ethanes—do not react extensively with water under standard hydrolysis conditions, and they remain in the hydrolysate.
On the basis of extensive laboratory and bench-scale testing (Irvine et al., in press), biodegradation has been selected by the TPC as a preferred treatment for the hydrolysate (U.S. Army, 1996b). Microorganisms in sewage sludge can adapt to using thiodiglycol as a primary energy and carbon source. Biodegradation of thiodiglycol requires adjusting the pH of the hydrolysate to neutral by adding sodium bicarbonate buffer. Aqueous ammonia is added as a nitrogen source, phosphoric acid as a phosphorus source, and mineral salts as trace nutrients. The bacteria oxidize thiodiglycol efficiently to carbon dioxide, water, and sulfate with high efficiency, as expressed in the idealized equation:
S(CH2CH2OH)2 + O2 → CO2 + H2O + H2SO4 + biomass
During actual operation, approximately 0.8 g of cell mass (dry weight basis) is produced for every 1 g of organic carbon removed from solution. Excess bacterial
|
4 |
Delisting is a regulatory process by which a solid waste that has been classified as a hazardous waste based on its origin is demonstrated not to be hazardous. The delisted waste may then be disposed of at waste management facilities designated for nonhazardous wastes. |
|
5 |
The required level of decontamination is specified as a 3X condition (see Capacity to Detoxify Agent in Chapter 2). |
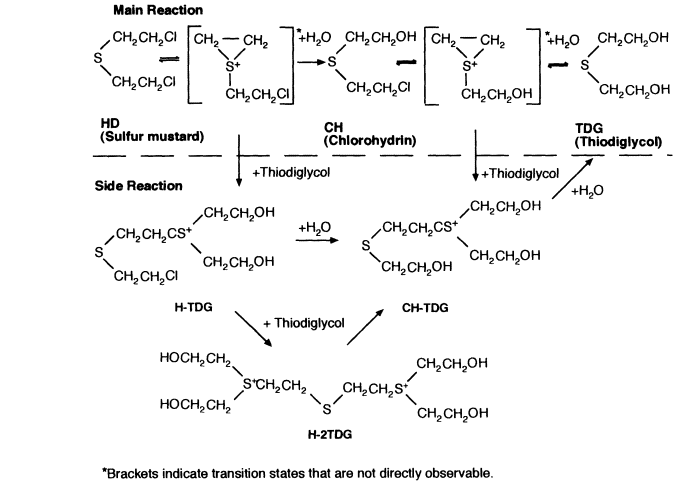
Figure 7-5
Chemical reactions during the hydrolysis of HD. Thiodiglycol is the primary product, and side reactions are minimized by maintaining HD as a dilute component in water during reaction. Sulfonium ions are indicated by S+. Source: U.S. Army, 1996b.
cell mass is separated from the biodegradation process effluent. This biomass is further oxidized (degraded) through aerobic digestion, dried, and disposed of at a commercial TSDF. Fortunately, dithiane and the least volatile chlorinated ethane, hexachloroethane, are oxidized along with the thiodiglycol (U.S. Army, 1996b).
The TPC has proposed photochemical oxidation as a polishing step to destroy VOCs that were present in the HD and remained in the hydrolysate after neutralization. Because of their volatility relative to water, VOCs can be removed from the hydrolysate by a stripper unit. Volatilized VOCs and water vapor are then condensed, and the resulting condensate is photochemically oxidized by adding hydrogen peroxide in the presence of ultraviolet light. Under these conditions, the VOCs are both directly degraded by photolytic dissociation and oxidized by HO radicals, which are formed by the photochemical dissociation of hydrogen peroxide (Solarchem Environmental Systems, 1996). The products of the photochemical oxidation are simple organic compounds (aliphatic organic acids such as acetic acid), chlorides, and carbon dioxide. The panel's evaluation of the specific photochemical oxidation process proposed by the TPC was limited to reviewing the treatability study provided by the TPC and applying the prior experience of panel members with photochemical oxidation processes.
Overall, the neutralization, biodegradation, and photochemical oxidation operations yield a relatively simple set of final products: carbon dioxide, water, chloride and sulfate salts, metal salts (originally present in the HD), and biomass.
Technology Status
Hydrolysis of HD
Alkaline hydrolysis has been used extensively to detoxify mustard agents and the G family of nerve agents. (Application to nerve agents is discussed in Chapter 8.) Munitions-grade mustard agents have been hydrolyzed with methanolic NaOH on a pilot-scale in Poland (Koch and Wertejuk, 1995). This procedure was effective for liquid agent and for solids that remained in the storage containers. The methanol solutions of hydrolyzed agent were incinerated. In Canada in the mid-1970s, mustard agent was hydrolyzed on a production-scale (8-ton batches) with hot lime water prior to incineration (NRC, 1993, p. 63). Recent laboratory evaluations of the alkaline hydrolysis of HD by the U.S. Army have not shown any particular advantages in using lime instead of the more soluble sodium hydroxide (Harvey et al., 1994). Lime most likely was used in the Canadian hydrolysis process to avoid high concentrations of sodium in the hydrolysate during subsequent incineration, because sodium attacks common refractories.
Simple hydrolysis of mustard with hot water is not as well documented as alkaline hydrolysis, but it has apparently been used in France (Harvey, 1995) and is the basis for the long-used method of steam cleaning and decontaminating storage containers. Although hydrolysis with water under neutral or acidic conditions is slowed by the limited solubility of the agent, the reaction proceeds well at low concentrations (1 to 10 percent agent in water). With vigorous agitation and temperatures of 75°C to 90°C, the reaction is essentially complete in one hour, as was demonstrated in a laboratory-scale experiment witnessed by panel members.
The laboratory-scale tests were initially conducted in small glassware but were later scaled up to 1-liter flasks to permit hydrolysis of about 10 g of HD at a concentration of 1.3 wt pct or 67 g at 8.6 wt pct. Tests at this scale were used to optimize conditions for tests in bench-scale reactors. Experiments in 2-liter Mettler reactors (up to 150 g of HD per test at 9.3 wt pct) yielded precise thermodynamic data that guided larger tests and design studies (U.S. Army, 1996b).
Subsequent tests in a 114-liter reactor (U. S. Army, 1996b) provided valuable operating experience and basic engineering data on a scale that can be easily extrapolated for designing the pilot- and production-scale reactors. In addition, these experiments produced substantial volumes of hydrolysate for bench-scale biodegradation studies. In a typical run, 7.22 kg of munitions-grade HD was hydrolyzed to produce 88 liters of hydrolysate. When the 114-liter reactor was converted for VX hydrolysis studies (see Chapter 8), HD hydrolysate for ongoing research on biodegradation was supplied by a larger reactor fabricated from a 55-gallon (208-liter) drum. The stainless steel drum was lined with polypropylene and fitted with an efficient stirrer coated with KynarÔ resin. Operations with this reactor provided about 130 liters per run of hydrolysate, which derived from HD concentrations in the reaction mixture of either 5.7 kg HD at 3.8 wt pct or 13 kg at 8.6 wt pct. In addition to producing hydrolysate for the biodegradation studies, the runs in this reactor provided useful experimental data on the rate of HD disappearance under conditions similar to conditions expected in full-scale operations (Harvey et al., 1996).
These large scale tests showed that the reaction proceeds cleanly with thiodiglycol as the primary reaction product. The residual HD concentrations in the hydrolysate from these experiments dropped below 200 ppb in less than 20 minutes, and the toxicity was dramatically reduced (see Agent Detoxification) (Harvey et al., 1996). Processing at bench-scale also has demonstrated the successful destruction of HD present in the heel removed from a ton container.6 Thus, the neutralization process has been demonstrated to work well for the distilled HD stored at the Aberdeen site. Overall, 161 kg of HD were destroyed in the laboratory and bench-scale studies at Aberdeen (Novad, 1996).
In the past, incineration has been the principal process used for disposing of wastes from the alkaline hydrolysis of mustard agents, as described above for the Polish and Canadian methods. To facilitate public acceptance of neutralization-based technology, the TPC has studied the biodegradation of thiodiglycol and other organic compounds obtained from the neutralization of HD. Previous attempts to use microorganisms to destroy HD itself failed because the agent is toxic to all life forms tested. However, various bacterial cultures readily oxidize thiodiglycol (Zulty et al., 1994).
Biodegradation of Hydrolysate
Laboratory-Scale Tests
The biological treatment of hydrolysate has been extensively tested at laboratory-scale using SBRs with a 1- to 12-liter working volume (Irvine et al., in press). The primary objectives of the laboratory testing were to determine treatment efficiency, the quality of effluent from the process, the optimum operating conditions, and the effects of HD impurities (e.g., ton container heel and iron floc). More than 500 days of continuous bioreactor operation were completed during laboratory-scale testing. Individual bioreactors successfully operated continuously for duration of 130 to 159 days. Unacclimated and acclimated mixed bacterial cultures were evaluated for treating hydrolysate produced from the neutralization of 1.27 to 9.5 wt pct HD. The feed to the SBRs was diluted to the equivalent of 1.27 wt pct HD for all tests. Good results were obtained with mixed cultures such as those obtained from the Back River municipal waste treatment plant in Baltimore, Maryland. Thiodiglycol removal was greater than 99 percent in almost all cases, and the mean TOC (total organic carbon) removal ranged from 86 percent to greater than 92 percent, depending on the specific operating conditions. The effect of the concentration at which HD was hydrolyzed was minimal for hydrolysis concentrations up to 8.6 wt pct HD. An operating regime of 10 days hydraulic residence time (HRT), 15 days solids residence time (SRT), and hydrolysate organic carbon loading of 0.08 to 0.1 g TOC/g MLSS-day7 (mixed liquors suspended solids) was repeatably demonstrated to remove greater than 99 percent of the thiodiglycol and greater than 90 percent of the TOC. The SBRs were operated continuously for several months and demonstrated stable operation at temperatures between 8°C and 35°C. No significant detrimental effect was observed as a consequence of the iron floc in the hydrolysate feed. Hydrolysate toxicity was reduced by a factor of 50 based on Microtox assays.
Bench-Scale Tests
Based on the laboratory findings described above, the biodegradation of hydrolysate was demonstrated on an 80-liter scale (bench-scale) in a SBR (U.S. Army, 1996e). Three 80-liter test cases for biodegradation of hydrolysate were conducted, with each case in continuous operation for more than 30 days. Unacclimated biomass from the Back River treatment plant in Baltimore was used as the seed population for each test. In each case, the feed was diluted to about 99 percent water for biotreatment regardless of the starting concentration of HD in the preceding hydrolysis step. The reactor was operated successfully with hydrolysate from 3.81 wt pct HD, a 10-day HRT, and 24-day SRT. Thiodiglycol removal was greater than 99 percent and mean chemical oxygen demand (COD) removal was greater then 90 percent. Operation with hydrolysate from 8.49 wt pct HD (13.5-day HRT, 56-day SRT) resulted in thiodiglycol removal of greater than 99 percent and mean COD removal of 88 percent. Operation with hydrolysate from 7.9 wt pct HD was unsuccessful in that poor biomass settling resulted in a gradual decline in the efficiency of biodegradation. However, the cause of the poor settling characteristics is not known. Startup operation of the SBR was not replicated in these tests, which were run after an initial startup period for the SBR.
Successful operation was defined as meeting removal targets consistently for at least 3 HRTs or 1 SRT, whichever was longer. The results suggest that the operations can succeed with hydrolysate from about 4 wt pct HD and, potentially, with hydrolysate from up to 8.6 wt pct HD. The panel suggests further bench-scale testing of SBRs with at least 3 SRTs at design HRT conditions and up to 8.6 wt pct HD hydrolysate to define the most beneficial operating regimes. If biodegradation of hydrolysate from 8.6 wt pct HD can be successfully demonstrated, schedule and cost may be reduced; for example, either smaller neutralization equipment or fewer HD neutralization batches would be needed.
Overall, the results of these tests are encouraging. Bioassay testing of the aqueous effluent from the biodegradation process indicated that the effluent had low toxicity (see section below on Agent Detoxification and Consistency of Standards). Final disposal must be decided upon for the products of the biodegradation, which consist of the biomass sludge from bacterial growth and an aqueous solution of sodium chloride, sodium sulfate, and low-levels of organics. Full-scale operation of SBRs for treating industrial wastewater has been demonstrated extensively; they have been in commercial operation for several years (Irvine and Ketchum, 1988; Brenner et al., 1992). Reduction of the biomass sludge by aerobic digestion and dewatering are standard
processes in municipal and industrial wastewater treatment facilities (Metcalf & Eddy, Inc., 1979).
Off-Site Biodegradation Options
Off-site biodegradation (configuration 4) would most reasonably occur at a commercial TSDF that receives wastewater from many sources and includes multiple processing steps. HD hydrolysate would therefore be a small contribution to the total loading at the TSDF. A survey of TSDFs carried out by the TPC indicated that several potentially could process HD hydrolysate (U.S. Army, 1996c).
The panel anticipates that thiodiglycol would be biodegraded at a TSDF, after microbial acclimation, as efficiently as in the SBR tests described above. Biological treatment at a TSDF that receives wastewater from a variety of sources also may be more tolerant than the SBRs of the side products that arise from neutralization at higher HD concentrations (e.g., 8 to 10 wt pct HD). Laboratory-scale (5 to 12 liter) testing carried out by the Army (U.S. Army, 1996i) and a contractor (SBR Technologies, 1996) have successfully treated hydrolysate produced from HD concentrations of up to 9.5 wt pct. Unsuccessful operation of a bench-scale (80 liter) test of biodegradation of hydrolysate produced from an HD concentration of 7.9 wt pct was hypothesized to be the result of an increased concentration of sulfonium ions in the hydrolysate, produced by the higher HD concentration during neutralization. In a large TSDF that receives wastewater from multiple sources, the increased sulfonium ion concentrations would be diluted below the level that could upset treatment. In addition, the diversity of the microbial population would be greater at a TSDF, making the process more resilient.
Results from testing the fate of VOCs during normal process operations at one candidate TSDF indicate that approximately 40 percent of the VOCs would evaporate to the atmosphere, 5 percent would be adsorbed onto activated carbon and biomass, and 25 percent would be biodegraded (O'Brien and Teather, 1995; Douglass, 1996). The anticipated concentration of VOCs in the HD hydrolysate (approximately 250 mg/liter) at the design disposal rate (approximately 130,000 kg/day) also appears to be significantly less than the maximum allowable intake of candidate off-site treatment facilities under current permit restrictions. However, treatment effectiveness at a TSDF must be demonstrated through detailed treatability studies. Preliminary treatability studies based on respirometry with unacclimated biomass yielded an 84 percent biodegradation of the thiodiglycol present in the hydrolysate (U.S. Army 1996e). The panel anticipates that improved removal efficiency similar to that achieved with the laboratory SBRs (greater than 90 percent biodegradation of thiodiglycol) can be achieved with acclimated biomass. Once treatability has been demonstrated, off-site biodegradation has the potential to greatly simplify the process requirements, construction, operations, and decommissioning required at the Aberdeen site.
It is possible that off-site biodegradation at a TSDF may be successful at higher weight percentages of HD in the neutralization process (e.g., 8.6 wt pct), even if on-site biodegradation is not favorable under those conditions. If off-site treatment of the hydrolysate from higher HD loading is successful, the neutralization step could proceed faster, accelerating the schedule and reducing costs.
Treatment of VOCs
For on-site biodegradation, VOCs present in the hydrolysate must first be separated and treated to prevent them from being air stripped during biodegradation. Air stripping would result in unacceptably high rates of use of activated carbon to remove the VOCs from the biodegradation offgas stream. In configurations 1 and 2, the TPC proposes to use a combination of ultraviolet light and hydrogen peroxide to destroy VOCs that would be subject to regulatory constraints if they were released to water or a landfill (land-ban chemicals). The VOCs would be distilled from the hydrolysate and diluted with a large volume of water, which in configuration 1 would be the main reuse for recycled water. The ultraviolet/peroxide technology can only be used on low concentrations (parts per million range) of organic compounds because higher concentrations decrease the photon efficiency and increase unwanted side reactions. Preliminary laboratory-scale tests using simulated VOC distillates have shown that this ultraviolet/peroxide treatment can destroy initial concentrations of 48 to 114 ppm of chlorinated hydrocarbons to less than 1 ppm (Solarchem Environmental Systems, 1996). However, two of the three tests were performed on samples lacking the other organic materials, such as dithiane, that might co-distill with the chlorinated hydrocarbons from the HD hydrolysate. If these organic compounds are present in the solution, the requirements for electrical power and hydrogen peroxide in the VOC
treatment process will increase significantly. This possibility seems to have occurred in a third laboratory test, in which the VOC simulants were diluted with evaporated bioreactor effluent rather than with water. Further testing is required to validate the ultraviolet/peroxide process as a cost-effective means of destroying the VOCs from an HD neutralization process.
An attractive alternative to the photochemical oxidation process for destruction of VOCs may be to ship the VOC stripper condensate to a commercial TSDF for disposal, as in configuration 3. Off-site treatment of the VOCs would have the benefit of simplifying the process requirements and operations at the Aberdeen site. Similar mixtures of chlorinated VOCs are commonly treated at many TSDFs. Evaluation of shipping of the VOCs to a TSDF also warrants a reconsideration of the design of the VOC stripper unit. The current design incorporates a falling-film evaporator to achieve VOC separation. This design is fully compatible with the water recycling in configuration 1, but it may result in more water in the condensate than necessary and increased disposal costs. Alternative design configurations and operating temperatures to optimize stripper efficiency should be evaluated if configuration 3 is selected for pilot-testing.
Operational Requirements and Considerations
Process Operations
Drainage, Clean-Out, Packaging, and Off-Site Shipment of Ton Containers
The ton container punch-and-drain system, which is common to all configurations, will be essentially identical to the well-proven JACADS system. There will be one ton container cleanout area, which is designed to operate 24 hours per day and drain 28 ton containers per week. The ton containers are moved into the toxic handling area, laid horizontally, and punched to create a 6-cm hole in the upper side. A tube is inserted into each container, and the liquid agent is pumped out into a 900-liter (240-gallon) holding tank. Residual agent and contaminated solids are removed by a high pressure wash system with water at approximately 88°C. An abrasive is added if necessary to remove sludge. The containers are then cut in half by the equivalent of a large pipe cutter and washed with hot water (88°C), steam cleaned (177°C), air dried, and sent to a sampling area where an ACAMS (automatic continuous air monitoring system) tests for residual HD vapor over the metal.
Similar washing systems are commonly used in industry but have not been used with high levels of agent contamination. If decontamination to the 3X level is verified, the metal is packaged for shipment off-site to the Rock Island Arsenal for melting operations previously demonstrated at that site (U.S. Army, 1996b). Melting ton containers after thermal treatment to a 5X condition is not necessary for disposal but is standard practice to facilitate recycling the metal.
Process gases from the ton container draining and decontamination area are passed sequentially through a condenser, a caustic scrubber, a reheater (to reduce the relative humidity of the gas), activated carbon filters for the process, and the carbon filters for facility ventilation. They are then discharged to the atmosphere. Condensate from the condenser is recycled for use as rinse water for the ton container cleanout. The use of hot water for the ton container cleanout process should result directly in the destruction of a large fraction of the residual HD via the same hydrolysis reaction that occurs in the neutralization operation for the drained HD. Hydrolysis in the caustic scrubber should destroy any residual HD present in the process gases prior to filtering through activated carbon. Thus, the activated carbon filters serve as redundant safeguards to ensure the removal of all HD from the process gases prior to release to the atmosphere. The scrubber and process-specific carbon filters are designed to operate at a slight negative pressure (about -0.3 atm gauge) to avoid potential leaks from process piping. The scrubber and filter for treating process gas from the ton container cleanout area are the same units used to treat vent gases from the bulk HD neutralization process.
Although shields are used to contain splashes of decontamination fluid and metal cuttings, the spread of agent contamination will probably be somewhat greater than now occurs with the Army baseline system, where there is no water wash. The spray systems use recirculation as much as possible. All of the cleaning liquids are ultimately consolidated and transferred to the primary agent-neutralization reactor for complete destruction of the agent.
No major difficulties are foreseen with this operation, although somewhat greater contamination of the equipment and surrounding areas may be expected from the high pressure spray systems. It will be difficult to predict the rates of use of water and decontamination solution, the requirements for their subsequent interim storage, and their dilution effect on the neutralization
system. Filters provided to protect pumps must be cleaned out, decontaminated, and disposed of off-site. In addition, the operating conditions for treatment of ton container cleanout and decontamination solutions, either separately or with agent, have not been defined, although similar materials from laboratory and bench-scale testing have been handled without difficulty.
The sodium content of the spent caustic solution used to decontaminate the work areas is not expected to be an issue for either the neutralization or biotreatment processes. It will simply replace some of the sodium hydroxide required by the agent neutralization process. The effects of the spent caustic solution on reaction pH can be balanced by controlling the rate of substituting spent solution for HD in the feed to the hydrolysis reactor.
Current analytical methods used in the Army's baseline technology should be adequate to monitor these front-end operations.
Agent Storage System
The drained agent is pumped to an interim holding and surge tank system, analogous to the proven Army system (NRC, 1994e). The capacity can easily be adjusted to account for local external-hazard risks. In the current design, gases vented from these tanks pass directly through an activated carbon filter bed located in the storage area and then are discharged to the room ventilation system. In the panel's judgment, this design does not provide the multiple layers of protection inherent in the scrubber and two-stage activated carbon filtering used for vent gases from all other tanks that might contain agent. Processing vent gases from the agent holding tanks through the common scrubber and activated carbon filtration system is an alternative that should be evaluated.
Neutralization Systems
In the TPC's design, there are three neutralization process lines in separate toxic containment areas; each process line includes two neutralization reactors designed to work in parallel. Neutralization is planned to continue 24 hours per day, 7 days per week. The liquid HD from the agent holding tank is pumped along with hot water through a static mixer, which is intended to disperse the agent in the water as droplets roughly 60 m in diameter. The aqueous dispersion of agent is pumped to a well-stirred 8.7-m3 (2,300-gallon) Kynar-lined reactor partially filled with hot water. The agent concentration in the reactor is tentatively designed to be approximately 4 wt pct. The agent feed rate is controlled to maintain an excess of water which prevents the formation of sulfonium salts that slow the completion of neutralization and give rise to additional by-products. If neutralization is incomplete because of a process upset in the agent feed system, experience in the bench-scale reactors indicates that neutralization can be completed by extending the heating time in the neutralization reactor.
As the agent reacts with water, the neutralization reaction produces hydrochloric acid, which will lower the pH until the mixture is highly acidic (about pH 2). The reactors and related equipment must therefore be capable of withstanding highly acidic conditions.
Adding the agent and neutralizing it takes about one hour, during which time the exothermic reaction releases heat to the reaction mixture. The heat is removed by reactor cooling and a heat exchanger in the recirculation loop. Data have also been presented concerning the anticipated heating of the main reactor vessel by the slightly exothermic reactions.
During the first phase of a batch run, hot water is added to the reactor, and displaced gases are vented. While liquid agent is being added to the reactor, no venting occurs and the pressure increases by up to 1 atm from the compression of nitrogen in the headspace of the reactor. Because the reactor is operated at up to 1 atm gauge pressure, venting is only required during filling with hot water at the beginning of a reaction batch. Thus, only residual VOCs remaining in the empty reactor from the previous batch are vented during the filling process. Any residual agent in the gas stream is neutralized in the caustic scrubber.
After approximately 1 hour, 18 percent sodium hydroxide solution is added to bring the pH to about 12, which neutralizes the acid, completes the neutralization process, and prevents the re-formation of agent. Thus, the reactor and related equipment must be able to withstand alkaline as well as acidic conditions.
The neutralized hydrolysate is transferred to a 20-m3 (5,300 gallon) storage tank and analyzed for residual agent prior to subsequent processing (e.g., biodegradation). Residual agent must be less than 200 ppb, which represents a destruction efficiency greater than 99.9995 percent. If agent is detected in the hydrolysate at greater than 200 ppb, the hydrolysate is returned to the reactor for further processing. The overhead gases from the hydrolysate tanks are sent to the scrubber and two-stage-activated carbon filter system prior to release to the
atmosphere.8 In configurations 1 and 2, the resulting hydrolysate is purged of VOCs and subjected to biodegradation. In configuration 1 only, VOCs are also solidified before final disposal.
VOC Stripping
Several VOCs remain in the neutralized hydrolysate. Because these are air stripped and not treated in the bioreactors, they are first removed in a waste VOC evaporator, then condensed in an overhead condenser and stored for subsequent treatment or disposal.
In configurations I and 2, the VOCs are destroyed by photochemical oxidation. Since this reaction must be carried out in dilute solution to allow adequate penetration of ultraviolet light through the mixture, the VOC condensates are added to the full water recycle stream and the entire stream is processed. This treatment oxidizes residual organic compounds in the recycled process water from biodegradation. Tests by a vendor have demonstrated at laboratory-scale the technical feasibility of using a photochemical oxidation system in this way. Scale-up of the results has been extrapolated to a set of nine ultraviolet reactors and appropriate hydrogen peroxide feed systems (Solarchem Environmental Systems, 1996).
Biological Treatment of Hydrolysate
Biodegradation of the organic constituents of the hydrolysate (primarily thiodiglycol) can be carried out either on-site in coordination with HD neutralization or off-site at a commercial TSDF. Off-site biodegradation at the Aberdeen FOTW is not practical because the FOTW is not designed to treat organic constituents at the concentrations present in the hydrolysate to neutralize the sulfuric acid generated during the biodegradation of thiodiglycol.9
The TPC has selected biodegradation in SBRs (sequencing batch bioreactors) as the most robust design for on-site biological treatment (configurations 1, 2, and 3). SBRs have been used for full-scale treatment of a variety of industrial wastewater streams for several years (Irvine and Ketchum, 1988). An SBR is a large tank that contains piping for the injection of air, feedstock (the hydrolysate), and inorganic nutrients; a manifold for the withdrawal of settled sludge; a floating intake on an articulated arm, which is used to withdraw clear supernatant liquid; and a circulating pump to agitate the contents of the reactor.
SBRs are semibatch biological reactors that operate in several different states during a complete reaction cycle. During startup, a bacterial culture that has either been adapted to grow on thiodiglycol or that comes from a wastewater treatment facility is added to the reactor tank. During the first step in the reaction cycle, the hydrolysate diluted with additional water and supplemented with inorganic nutrients (Wolin salts) and sodium bicarbonate (for pH control) is pumped into the reactor tank. The filling process is carried out in the presence of air over a period of about five hours. Mixing and the addition of air initiates microbial oxidation of the thiodiglycol and other organic compounds in the hydrolysate. The major products are water, carbon dioxide, and bacterial cell mass; traces of methane may be evolved. During this aerobic phase, the sulfur in the thiodiglycol is oxidized to sulfate. The air injection is continued for about 17 hours, at which point greater than 99 percent of the thiodiglycol and 90 percent of the TOC in the hydrolysate have been oxidized. Next, the air injection and circulation are stopped, and the solids in the reactor are allowed to settle. The clear liquid at the top is decanted, and some of the settled sludge is pumped out through the manifold in the bottom of the tank. The residual liquid and sludge are left in the tank for a fresh cycle of reactions. The cycle of filling, reacting, and decanting is repeated every 24 hours. The residence time of the liquid contents in the SBR is about 10 days; the residence time of the solids is about 15 days.
The relatively clear liquid decanted from each SBR is sent to a water recycling facility. The sludge withdrawn from the bottom of the SBR is sent to a pair of aerobic digesters for further biotreatment. Polymers to facilitate dewatering are added to the digested sludge in a drum thickener, after which the sludge is dewatered in a filter press. Water exuded from the press is either sent for recycling (configuration 1) or discharged (configuration 2). The solid residue from the filter press (filter cake) is disposed of accordance with standard disposal practices for dewatered sludges from biological wastewater treatment. In this case, the filter cake may be disposed of at a
TSDF (most likely in a landfill) or could be delisted and disposed of as a nonhazardous solid waste. In the past, the hydrolysate from bench-scale neutralization and the aqueous effluent from bench-scale bioreactors used to treat hydrolysate have been delisted.
Water Recycling
The process uses water for decontamination, the neutralization process, and dilution of the bioreactor feed. Much of this water leaves the process in the effluent from the bioreactors. If the effluents are discharged to a FOTW (configurations 2 and 3), there is no need for water recycling capability. However, if only the solids can be sent off-site to a hazardous waste facility, water must be recycled to prevent an accumulation requiring off-site disposal. Water recycling is not required for configuration 4 because the hydrolysate is shipped to a TSDF immediately after the neutralization of agent.
To recycle water, configuration 1 uses a conventional thickener and filter press to separate the water from the biomass solids, a conventional sand filter with solids rejection by a clarifier to return residual solids to the bioreactors, and a conventional evaporator and mechanical vapor-recompression water-purification system. Salts recovered from the evaporator are to be solidified and stabilized prior to disposal at a TSDF (most likely in a landfill). Solidifying the salt stream is not an attractive option because it requires a large quantity of solidification agents. In addition, cement-based solidification processes have not been effective in reducing the long-term leaching of monovalent cations (e.g., Na+) and halogen anions (e.g., Cl-) (Kosson et al., 1995).
Agent Detoxification and Consistency of Standards
Laboratory and bench-scale tests have shown that the primary neutralization process can destroy the chemical agent to less than 200 ppb in the hydrolysate (see Technology Status section). The Army has proposed that residual HD in the hydrolysate must be less than 200 ppb before it can be transferred out of the toxic containment area. Preliminary analysis by the panel indicates that this standard appears to be inconsistent with the 3X standard for decontaminating solid materials and the airborne exposure limit for sulfur mustard.10 Furthermore, no clear toxicologic or regulatory basis has been presented for the proposed release standard. Thus, the toxicologic and regulatory basis for the release of liquids that could contain agent or are derived from agent needs to be reevaluated. The consistency of standards for liquids with other related release standards such as the airborne exposure limit, the 8-hour time-weighted average, and the 3X standard should be considered in the reevaluation.
In conclusion, the Army needs to establish standards applicable to the transportation and disposal of the neutralization hydrolysate. This reevaluation may result in either less stringent or more stringent requirements. However, the reevaluation is unlikely to seriously constrain off-site disposal options.
The biodegradation process after neutralization effectively removes the thiodiglycol so that back-reaction to produce new mustard is not possible. Further, the Army has shown that after biological treatment in the SBRs, the toxicity of the hydrolysate has been substantially reduced; the remaining low toxicity (to aquatic organisms) is primarily associated with the inorganic salt content (sodium chloride and sodium sulfate) of the SBR effluents. Table 7-1 shows the results from tests of the acute aquatic toxicity of SBR feed and effluent from laboratory and bench-scale operations. EC50 represents the solution concentration at which a negative response was obtained in 50 percent of the test population. Test populations included Photobacterium phosphoreum (a bioluminescent marine bacterium used in the Microtox assay), brine shrimp, Daphnia magna (water fleas), sheepshead minnows, and fathead minnows. These
TABLE 7-1 Aquatic Toxicity of Bioreactor Feed and Effluent from Laboratory and Bench-Scale SBR Testinga
|
Test Population |
Test Duration |
SBR Feed (diluted hydrolysate)b |
SBR effluent (laboratory-scale)b |
SBR Effluent (bench-scale)b |
|
Photobacterium phosphoreum |
5 min. |
3.9% |
95.4% |
44.3% |
|
Brine shrimp |
24 h. |
91.0% |
n.t. |
n.t. |
|
Daphnia magna |
48 h. |
5.4% |
27.0% |
23.5% |
|
Sheepshead Minnows |
48 h. |
84.2% |
82.1% |
87.8% |
|
|
96 h. |
70.0% |
77.2% |
82.5% |
|
Fathead Minnows |
48 h. |
n.a. |
n.a. |
57.3% |
|
|
96 h. |
n.a. |
n.a. |
40.0% |
|
n.t. = nontoxic when not diluted. n.a = not analyzed. a All results presented as EC50 values (the concentration that induces a response in 50 percent of the test organisms) in volume/volume percentages. b Concentration of inorganic salts in all three test materials was 2 wt%. Source: Haley, 1996 |
||||
results indicate that SBR effluent is a good candidate for discharge to the FOTW at Aberdeen. The TPC anticipates additional tests of toxicity in support of regulatory permitting, if the process is selected for pilot-testing.
Process Flow Diagrams and Overall Mass and Energy Balances
Process flow diagrams with corresponding overall process mass and energy balances for configurations 1 through 4 are presented in Appendix G. Individual unit operations and inputs for each configuration are summarized in Table 7-2. The number of separate unit operations required to complete processing for each configuration can be used as an indication of overall process complexity. Configuration 4 requires only three unit operations to complete processing to a point suitable for disposal at a commercial TSDF, whereas the other configurations require six or more unit operations.
Table 7-3 summarizes the waste streams and quantities for each configuration. The panel considers it advantageous to minimize the total amount of waste that requires subsequent off-site treatment and disposal. Each configuration produces the following waste quantities for disposal, in addition to the decontaminated (3X) ton containers. Configuration 1 produces 9 kg of solid waste per kg of agent destroyed. Configurations 2 and 3 produce 1 kg of solid waste and 88 kg of wastewater for discharge to a FOTW per kg of agent destroyed. The wastewater is anticipated to contain approximately 0.03 kg of residual organic contaminants and 1.6 kg of salts per kilogram of agent destroyed. In addition, configuration 3 produces 2.4 kg of aqueous waste containing VOCs for disposal at a TSDF. Configuration 4 produces less than 0.01 kg of solid waste and 29 kg of hydrolysate (aqueous) per kg of agent destroyed for disposal at a TSDF. Thus, configuration 4 results in the least quantity of solid wastes that must be shipped off-site for disposal. Of the three configurations with the option for off-site disposal of liquid residuals, configuration 4 produces the least aqueous waste to be treated and discharged. Configuration 1 results in the lowest overall mass of waste (solid and liquid) that must be shipped off-site but requires the most complex on-site processing.
All configurations require air for ventilation and drying during the ton container cleanout process. In addition, on-site biodegradation requires air to supply oxygen for biodegradation of the hydrolysate, biomass digestion, and sand filtration backwashing (configurations 1, 2, and 3). Biodegradation processes result in the release of carbon dioxide from the microbial oxidation of organic constituents in the hydrolysate (primarily thiodiglycol). Release of organic and inorganic
TABLE 7-2 Summary of Unit Operations and Inputs Required for Each Process Configuration
|
|
|
Configuration |
|
|||
|
Unit Operation |
Inputs |
1 |
2 |
3 |
4 |
Notes |
|
Ton container (TC) drain and cleanout |
TCs, agent, water, steam, air |
Ö |
Ö |
Ö |
Ö |
Required for all alternative technologies |
|
Neutralization reaction |
agent, water, sodium hydroxide |
Ö |
Ö |
Ö |
Ö |
|
|
Vent gas scrubber |
TC cleanout and neutralization vent gas, water, sodium hydroxide |
Ö |
Ö |
Ö |
Ö |
Vent gas cleanup from TC cleanout will be required for all alternative technologies |
|
VOC separation |
hydrolysate (with VOCs) |
Ö |
Ö |
Ö |
|
VOCs either treated by photochemical oxidation(configs. 1 and 2) or shipped to TSDF (config. 3) |
|
Biodegradation (SBRs) |
hydrolysate, water, air, sodium carbonate, nutrients |
Ö |
Ö |
Ö |
|
Dilution required to biodegrade thiodiglycol in hydrolysate |
|
Biomass digester and filter press |
biomass from SBRs, air, conditioning chemicals |
Ö |
Ö |
√ |
|
|
|
Water recycle evaporator |
effluent water from biodegradation |
√ |
|
|
|
|
|
Photochemical oxidation |
sodium hydroxide, hydrogen peroxide |
Ö |
Ö |
|
|
Requires dilution water for config. 2 |
|
Residual salt solidification/ stabilization |
concentrated salts from water recycle |
√ |
|
|
|
|
|
Number of unit operations |
|
9 |
7 |
6 |
3 |
|
|
√ = required unit operation. |
||||||
contaminants through atmospheric emissions is anticipated to be negligible because of scrubbing and multistage activated carbon filtration prior to release.
On-site energy requirements for each process configuration are presented in Table 7-4. These values do not include the energy required for office and laboratory operations or for normal building ventilation. Configuration 1 requires much more energy (356 MJ per kg of agent destroyed) than the other configurations because of the evaporator required to recycle water. Configuration 4 requires significantly less energy (49 MJ per kg of agent destroyed) than the other configurations. The on-site energy needs were used by the panel as a measure of process complexity rather than a metric for discrimination because the energy requirements can easily be met by existing power sources. The energy requirements associated with the many off-site disposal options were not considered by the panel and are judged to be of little or no significance in discriminating among the configurations.
An evaluation of process complexity in conjunction with the above mass and energy balances indicates that
TABLE 7-3 Summary of Waste Streams and Quantities for Each Process Configuration
|
|
|
Waste Quantity (kg/1000 kg agent destroyed) |
|||||
|
|
|
Configuration |
|||||
|
Waste Stream |
Disposal Method |
1 |
2 |
3 |
4 |
||
|
Air Emissions |
|
|
|
|
|
||
|
Ton container cleanout and neutralization |
scrub and filter through carbon before atmospheric discharge |
1,651 |
1,651 |
1,651 |
1,651 |
||
|
Biodegradation and VOC separation |
filter through carbon before atmospheric discharge |
71,339 |
71,339 |
71,339 |
n.a. |
||
|
Liquid Wastes |
|
|
|
|
|
||
|
Hydrolysate |
to TSDF (biodegradation) |
n.a. |
n.a. |
n.a. |
29,468 |
||
|
VOCs (aqueous solution) |
to TSDF (incineration) |
n.a. |
n.a. |
2,352 |
n.a. |
||
|
Biodegradation aqueous effluent |
to FOTW |
n.a. |
87,947 |
87,947 |
n.a. |
||
|
Solid Wastes |
|
|
|
|
|
||
|
Ton containers, valves, plugs, and metal cuttings |
to Rock Island Arsenal for smelting |
|
|
|
|
||
|
Solidified salts |
to TSDF (landfill) |
8,754 |
n.a. |
n.a. |
n.a. |
||
|
Biomass filter cake |
to TSDF (landfill) |
972 |
972 |
972 |
n.a. |
||
|
Activated carbon |
to TSDF (landfill) |
6 |
6 |
6 |
2 |
||
|
n.a. = not applicable. |
|||||||
configuration 4 (neutralization followed by off-site biodegradation) is the most advantageous. If biodegradation must be carried out on-site, configuration 3 (neutralization and on-site biological treatment, with discharge of effluent to a FOTW and shipment of VOCs to a TSDF) is the most advantageous configuration.
Operational Modes
The punch-and-drain operation and the subsequent decontamination and packaging of ton containers for off-site shipping is essentially a batch process. This process is planned for 24-hour-per-day operation. Although it could be conducted on an 8-hour-per-day-basis, the current restriction on storing no more than 1.9 m3 (500 gallon) of agent inside a facility will probably require close coupling of the punch-and-drain operation with the subsequent neutralization reaction.
The primary neutralization process is a semibatch process that requires approximately four hours to complete a batch, including the testing time to verify complete agent destruction. To meet the overall schedule requirements, it is best to operate this portion of the plant 24 hours per day. However, it is technically feasible to process batches only 8 hours per day. The process could also be redesigned as a continuous, rather than semibatch, process. But this change would require more complicated process controls and would have a higher probability of failures and upsets. A continuous process
TABLE 7-4 Summary of Energy Requirements for Each Process Configuration
|
|
Energy Required, by Configuration (MJ/kg agent destroyed) |
|
|||
|
Energy Input |
1 |
2 |
3 |
4 |
Notes |
|
Steam (for heating) |
300 |
37 |
37 |
28 |
Config. 1 requires substantial steam heat for the evaporator to recycle water |
|
Electricity |
|
|
|
|
|
|
Motors, fans, etc. |
42 |
40 |
33 |
12 |
Substantial power input is required for fans to aerate bioreactors (configs. 1, 2, and 3) |
|
For cooling |
14 |
14 |
14 |
9 |
Required primarily for condensers |
|
Total Energy Input |
356 |
91 |
84 |
49 |
|
would have the added disadvantage of making it more difficult to confirm complete agent destruction prior to release of material from the toxic control area.
The bioreactors operate semicontinuously; that is, there are cyclic additions of feed from an intermediate surge tank. Although the bioreactors require little nighttime monitoring, they must operate 24 hours per day. The water recycling facility can be easily started and stopped, assuming surge capacity is provided, and could be operated either 8 or 24 hours per day. The optional solidification plant for stabilizing the effluent from biodegradation is operated intermittently during daylight hours.
Emergency Startup and Shutdown
Because the total process consists of a sequence of batch or semibatch operations with holding capacity between them, there are separate startup and shutdown procedures for each operation.
Ton Container Handling. In cold weather, the ton containers must be preheated to melt the mustard. Melting HD with ambient room temperature would probably take several days and would therefore require a substantial inventory in storage. Thus, for thawing ton containers in cold weather, procedures must be developed that are consistent with the limitations on the total agent inventory in the processing facility (currently established as the 1.9-m3 limit for the agent storage tank).
Punching and draining full containers; removing sludge, and decontaminating, cutting, testing, and shipping empty containers are sequential steps in a mechanical handling system with no significant surge capability between them. Thus, the whole sequence must be started up and maintained as a mechanical production line. Shutdown requires stopping the feed and allowing the in-process containers to continue to completion, followed by decontamination of the work area as necessary. These front-end procedures, which are common to all systems for treating ton containers regardless of the HD destruction technology, reflect extensive Army experience at JACADS (NRC, 1994a).
Neutralization. The primary neutralization process can be stopped in an emergency by stopping the feed or chemicals. Under these conditions, the reactions continue until the reactants are depleted. Only a very small quantity of agent is present in the reactor at any time because of the slow feed rate and the rapid reaction in the reactor.
VOC Stripper and Oxidation System. These systems can be started or stopped by initiating or stopping feed from intermediate storage tanks.
Bioreactors. The SBRs can be filled and started up using either the hydrolysate or a surrogate material to establish an equilibrium composition and distribution of the microorganisms, and to stabilize functioning support systems such as the various nutrient feed streams. Tests have shown that no further acclimatization of the microbial population
is required prior to introducing the hydrolysate for biodegradation.
Although the bioreactors must continue to operate to maintain their active microbial populations, several SBRs will be operating in different phases, providing flexibility in operational modes. Further, the feed to each SBR can be shut off for a short time without significantly harming the microbial population. Alternative feeds can be provided to maintain the population during more extended shutdowns.
Sludge Dewatering. This independent system has feed and waste storage containers so that it can be independently started and stopped. The feed flocculation tank would ordinarily be drained prior to shutdown. If an extended shutdown is expected, the filter press would be cleaned.
Water Recycling. Startup requires turning on the system and internally recycling water until the desired water quality is achieved. The system can then be operated in standby recycling mode until effluent water to be recycled is added or until recycled water is needed. The system can be simply shutdown. The VOC photochemical oxidation system can be shutdown with or without continued water circulation by turning off the power to the ultraviolet lamps and stopping the hydrogen peroxide feed.
Waste Solidification. This batch operation is similar to concrete mixing and pouring, with the usual need for cleaning out the system periodically to avoid the buildup of solids that would impede the flow of the slurry.
Reagents and Feed Streams
Decontamination and storage container processing will require caustic solutions prepared by dilution on-site of commercial, 50 wt pct NaOH solutions. Large volumes of process water are required for these operations. Neutralization also requires dilute NaOH solutions and large volumes of water. Much of the water may come from the ton container processing area and from water recycled from the bioreactor effluents (in configuration 1).
The biodegradation operations require sodium bicarbonate to control pH, as well as aqueous ammonia and mineral nutrients for the growth of the microorganisms. For startup of the SBRs, significant quantities of biomass are needed. This can be obtained from a local wastewater treatment facility. Various solids-conditioning chemicals are needed to facilitate operation of the filter press.
The waste solidification operation, if included, requires Portland cement, lime, some additives, and water. Water recycling, if incorporated in the overall process, may require ferric chloride as a flocculating agent and polymeric water conditioners. If the photochemical oxidation process is used to destroy VOCs, hydrogen peroxide must be supplied.
Process Stability, Reliability, and Robustness
Neutralization
The system will use standard industrial components that have been used extensively in conventional applications. Although feed rates are important, most of the process phenomena occur relatively slowly, so response time should not be critical.
The hydrolysis and acid neutralization reactions are mildly exothermic (420 kJ/kg and 700 kJ/kg agent, respectively). The heat of reaction is removed by cooling coils and by evaporation of water with subsequent condensation from the offgas in a reflux condenser cooled by chilled water. Failure of the cooling system would cause a temperature excursion estimated to heat a full batch of agent and water from 90°C (1 atm gauge) to 108°C (1.4 atm gauge). The design pressure for the neutralization system is 6.8 atm gauge. There should be no catastrophic thermal excursions.
The stored thermal energy in each neutralization reactor is 420,000 kJ at 90°C. The maximum agent present in each reactor is approximately 275 kg of HD, diluted to 4 wt pct in water. The actual quantity of agent present in the reactor will be much less because of the slow agent feed rate and rapid reaction when the agent is added to the large volume of hot water present in the reactor at the beginning of each process cycle.
The system is operated nominally at 1 atm gauge pressure. This headspace pressure is used to minimize loss of VOCs when the reactor is being filled and headspace gases are displaced. The principal headspace gas is nitrogen.
There is a possibility of excessive heat being generated if agent were to be inadvertently introduced into concentrated caustic solution. Although this situation is
unlikely, if it occurred, it would be detected quickly by the amount of heat released. Reliability (e.g., on line availability) of the integrated process for the treatment of agent can not be directly assessed without pilot-scale performance data. However, the reliability of equipment components (e.g., pumps, valves, and reactors) and subsystems can be assessed on the basis of their performance at JACADS and in other industrial chemical processing environments. The design assumes continuous operation (24 hours per day) while the facility is on line, with 6 hours per day allowed for slack time (when agent feed or other throughput operations are halted for maintenance, testing, etc.), but the remainder of facility is still on line. The design schedule also allows for 30 percent downtime (facility off line) on an annual basis (U.S. Army, 1996b).
This level of availability has been designed into the process through the incorporation of multiple neutralization processing lines and the installation of redundant components (pumps, valves, etc.) in critical flow paths within the area where toxic materials are handled or processed. The design basis for the on-site biodegradation process is treatment of the hydrolysate from 3,175 kg of HD per day. This requirement can be met with a downtime of one month for two of the three SBRs during the planned 12-month operating interval. The on line availability of biodegradation increases for off-site processing because an off-site TSDF would have to be continuously available to handle other feed stream.
Biotreatment
The bioreactors have proven to be stable when properly operated. Possible improper operations include an upset in a feed (air, nutrients, or hydrolysate) or improper mixing. Because several SBRs are used in parallel, a failed reactor could be readily restarted with biomass from another operating SBR.
Waste Solidification
This operation is very similar to mixing concrete. It is only mildly exothermic, and no stability problems are likely. Standard industrial components with proven reliability are used in this operation. A typical problem might be the accumulation of hardened cement, which could inhibit the flow of solids. Handling solids is usually the operation most subject to problems in any processing plant.
Water Recycling
There are no inherent instability problems in this operation. The sand filter and evaporator are common operational equipment, usually very reliable and robust. The photochemical oxidation system uses lamps and peroxide injection systems that are proven and reliable. Polymerization of residual organic contaminants during oxidation, resulting in opaque deposits on illumination surfaces, may require periodic cleaning of these surfaces. The lamps also will require periodic monitoring and replacement.
Materials of Construction
When agent is first hydrolyzed, the solution in the reaction vessel becomes acidic. This solution is moving at a relatively high velocity because it is being vigorously mixed. Depending on how much oxygen is available, corrosion of the reactor and components at the vapor-liquid interface may be accelerated. The extent of these combined effects is uncertain because sulfur and chloride in the solution may also influence the corrosion rate. The possibility of accelerated corrosion resulting from the combination of these conditions should be considered. In addition, the contents of the neutralization reactor go from a low pH near the end of hot-water hydrolysis to an alkaline pH from the addition of sodium hydroxide just prior to discharge to the bioreactor system. This pH change dictates that the reactors be made from a versatile metal or one lined with glass or a plastic like Kynar, but these are not unusual materials of construction. Most other systems are standard or have already been developed for the baseline technology.
The TPC has initiated an extensive program for testing materials of construction, including metallic and nonmetallic materials to be used in vessels, piping, seals, gaskets, and other components. Specific testing for metallic materials includes weight loss (immersion) testing, U-bend stress corrosion cracking tests, and electrochemical testing using solutions representative of the process environments. Nonmetallic materials are being tested with standardized immersion testing procedures. Additional information for selecting materials comes
from the experience gained during agent destruction operations at JACADS.
Operations and Maintenance
Operational Experience
All of the unit operations included in the overall process have been used extensively in commercial operations with related, proven equipment. In addition, the TPC has acquired a substantial body of experience from bench-scale testing at Aberdeen. There has also been significant prior experience with a similar method of agent neutralization, albeit with agent GB.
There has also been significant prior industry experience with the design, construction, and operation of SBRs for industrial waste disposal. However, there has been no prior commercial experience with this specific grouping of unit operations or with the use of the unit operations for agent destruction. The TPC and the panel anticipate no unusual problems with process integration.
Maintenance
Maintenance requirements for all systems but the bioreactors are similar to requirements for the baseline technology systems. Maintenance requirements for SBRs have been established through industrial experience. The panel foresees no unusual problems. Maintenance manuals and documented procedures are not yet available for this combination of unit operations.
The lifetimes of major equipment items should all exceed the duration of plant operations, although some small items, such as small pumps or instrumentation, may require replacement. The downtime for replacing conventional components will probably be governed by preventive safety precautions rather than by actual failure of the equipment. Critical items (e.g., pumps) have redundant systems installed as part of the process design.
Worker safety practices to prevent exposure to chemical agent will be similar to practices used at JACADS. Additional safety precautions will be required for handling hydrogen peroxide, if photochemical oxidation is part of the process. The handling of caustic solutions should be similar to the handling of decontamination solutions at JACADS.
Utility Requirements
Heating, Ventilation, and Air Conditioning System. Normal building heating will be required for cold weather operations. In addition, the ton containers require heating to melt frozen HD in the winter. Although ton container heaters will be provided, the cold containers may require extra heating for the container storage area. Steam heating will be required for various small-scale operations. Steam will be provided by four boilers, each rated at 32,000 kJ/hour at 10 atm gauge and 185°C.
Electrical Systems. The systems will be standard for the chemical industry with a 3,400 kVA load.
Plumbing and Piping System. These systems will be standard for the chemical industry with the exception of floor drains in the ton container and neutralization-process areas to drain spent decontamination fluids.
Fire Protection Design Requirements. The requirements will be normal for the chemical industry. Special hazards include hydrogen peroxide.
Other Systems. Chilled water is needed for the reactor cooling coils, the condensers, and other cooling systems. (Power for the refrigeration systems is included in the electrical systems load.) Compressed air is supplied by approximately three 186-kW (250 hp) compressors (power required is included in the electrical systems load).
Scale-Up Requirements
Bench Scale to Pilot Plant
The following key unit operations are required for configuration 1: (1) ton container processing, (2) reagent preparation and storage, (3) agent neutralization, (4) biodegradation of neutralization hydrolysate, (5) biosolids digestion and filtration, (6) evaporation, (7) photochemical oxidation, and (8) solidification (optional). The critical new unit operations are agent neutralization, biodegradation including the SBRs, and biosolids digestion and filtration. Scale-up is considerably simplified if one of the simpler configurations is selected-especially configuration 4, which requires only the first three unit operations.
Agent neutralization tests have been conducted in 110-liter (30 gallon) batch reactors. This test size will
be extrapolated to 8.7 m3 (2,300 gallon) reactors for pilot-testing. The principal variables to be extrapolated are likely to be mixing and heat exchange. No catalysis is involved. Heat exchange is not likely to be a problem if adequate mixing takes place.
Mixing is of concern since the neutralization process involves intermediate reactions and requires an adequate ratio of water to agent to avoid the production of sulfonium salts. Mass transfer studies are currently under way by the TPC, including designing impeller baffles and eliminating dead spots.
Bioprocessing of the hydrolysate has also been limited to bench-scale reactors, typically 80-liter volumes. However, similar scale-up is usual in the development of biotreatments for many waste materials and is not expected to be a problem.
Pilot Plant to Full-Scale Facility
The TPC plans to develop the full-scale facility using the pilot plant neutralization reactors as one module. The full-scale plant will essentially require the addition of two more modules. Thus scale-up should be straightforward, apart from matters such as scheduling.
The SBRs for biodegradation of the neutralization hydrolysate will also be built by adding modules like the ones used for pilot- tests. Other systems may or may not be modules, but they are relatively standard unit operations that should not be difficult to scale.
As with the other technologies, process safety risk factors for the neutralization process can be divided into factors inherent in handling agent prior to its introduction to the neutralization process and factors related to the neutralization technology itself. The risk factors inherent in handling agent prior to neutralization are the same as the risks for the other technologies; they include storage risk, transportation risk, and risks associated with punching, draining, and cleaning ton containers. Storage risk can vary among the configurations because they may require different processing schedules. A simpler set of operations, such as configuration 4, can increase process reliability (e.g., less downtime) and increase the intrinsic safety of the process (less training needed, fewer things to go wrong).
The process-safety risk factors inherent in the neutralization process seem to be minor. The process operates in a batch mode at low pressure and low temperature. The purity of the neutralization products and scenarios for loss of cooling have been explored. It is difficult to envision safety being threatened except by an external factor of some sort.
Based on the panel's preliminary and qualitative evaluation, the most significant off-site risk appears to be associated with handling agent prior to neutralization. In particular, the principal risk factors appear to be mishaps in the punch-and-drain operation or damage to agent holding tanks from an airplane crash or other external event.
Process Safety
Two scale-up considerations are involved, namely, scale-up from the existing bench-scale operation to the pilot level and scale-up from the pilot level to the full-scale operation. The TPC currently plans to conduct pilot-testing in a single reactor line of a multireactor production facility. This approach should significantly reduce the difficulties and time involved in the second scale-up.
Worker Safety Issues
There are some worker safety issues associated with handling ton containers and handling associated chemicals used in the process. Specific concerns include manual handling of agent-filled ton containers (both injury from manual manipulation of large, heavy objects and the potential for agent exposure in case of a leak), concentrated sodium hydroxide, and hydrogen peroxide. These risk factors need to be addressed in the final operational design.
Specific Characteristics that Reduce Inherent Risk of Design
The system operates in a batch mode at near atmospheric pressure and low temperature. The process proceeds extremely slowly. Temperature transients, if they occur, appear to be very mild. Safety risks from the destruction process appear to be minimal.
Schedule
The proposed schedule is based on constructing a full-scale, multitrain facility at the Aberdeen site (U.S.
Army, 1996b). There would be three two-reactor trains, one of which would be used initially as a pilot facility to demonstrate the effectiveness of the neutralization and biodegradation processes. This approach would facilitate scale-up to full-scale. It should also eliminate delays associated with previous plans to pilot-test the technology at the Chemical Agent Munitions Disposal System facility in Utah before construction of a production-scale facility at Aberdeen. Consequently, the TPC forecasts that destruction of the HD stored in bulk at Aberdeen can be completed by August 1, 2003. The TPC projects that plant closure would occur late in 2004.
The pilot plant design reached the design level required for a RCRA permit in April 1996 and should be at the 90 percent stage by the end of November 1996. Modifications to the design that simplify the process are not likely to cause delays. If a decision to pilot this technology at Aberdeen is made in October 1996, permit applications would be submitted in January 1997. The Army's plan allows one year for permit acquisition, which seems reasonable considering the generally favorable reception of neutralization technology by the Maryland Citizens Advisory Commission (see Chapter 9).
During the latter part of the permit acquisition period, a contract for construction of the facility would be let and orders would be placed for equipment with long lead times for delivery. Construction would begin in June 1998 and be completed about October 30, 2000. Initial systemization of a single production line would take about nine months. Pilot operations in this reactor train would be carried out until February 1, 2002, at which time systemization of the other reactor trains would start and a low-rate production operation would begin. Full-scale operations beginning in August 2002 would continue for about nine months. The production schedule assumes treatment of six ton-containers per day, with the facility operating on a two-shift basis. Plant decommissioning and decontamination is estimated to require one year.























