4
Catalytic Extraction Process Technology
Process Description
Catalytic Extraction ProcessingTM (CEPTM) is a proprietary technology patented by its developer, Molten Metal Technology, Inc., and licensed to M4 Environmental L.P. for specified U.S. governmental applications. 1 M4 Environmental L.P. joined with several other firms to prepare the submission on CEP in response to the Army request for information on alternative technologies.2 Hereafter in this chapter, M4 Environmental LP. and its supporting firms will be referred to as the technology proponent company (TPC). In addition to processing of HD and VX, the submission included processing of the steel ton containers and all dunnage generated in the course of demilitarization operations at the two sites. Destruction of HD and VX by CEP is accomplished in a series of unit operations after the ton containers have been opened and the contents transferred to interim storage tanks.
CEP has been designated by the U.S. Environmental Protection Agency (EPA) as a nonincineration technology. The distinction between incineration (or combustion) and CEP is based upon reaction mechanisms as well as end products. Combustion, which occurs by means of a series of gaseous, reactive intermediates (free radicals), requires high temperature, intimate mixing, adequate residence time, and excess oxygen to achieve high destruction efficiency. CEP, by contrast, is conducted mainly within a molten metal bath at high temperature and low oxygen potential. The products of combustion are in high oxidation states (e.g., CO 2, H2O), whereas products of CEP are in reduced states (e.g., CO, H2).
Technology Overview
A CEP reactor, which is called a catalytic processing unit (CPU), contains a bath of molten metal, typically iron or nickel. For treating chemical warfare agents, the TPC has decided that two CPUs are required. Each CPU is a steel pressure vessel containing a molten metal bath and an optional slag or flux cover. In CEP, these reactors are typically operated in the temperature range of 1425°C to 1650°C (2600°F to 3000ºF). The vessel is lined with refractory materials selected to provide thermal insulation and resistance to corrosion, erosion, and penetration by components of the bath. An electric induction coil, embedded within the refractory lining surrounding the metal bath, provides the energy to melt the metal charge and maintain the temperature of the bath during processing. The CPU headspace, which is several times the height of the molten metal bath, provides physical space to allow disengagement of the offgas from the molten metal and slag. One or more tapping ports through the vessel sidewall allow recovery of metal and slag phases with minimal interruption of operation. One CPU is fitted with a side chamber that can be heated by its own induction coil to melt ton containers. The molten metal flows from the side chamber into the main bath of the CPU. The TPC plans to feed dunnage, placed in steel containers, directly into the metal bath.
The feed material and the cofeeds of oxygen and methane can be injected into the molten metal bath either through a lance entering the top of the bath or through one or more bottom-entering tuyeres. (The TPC has used top-entering lances in numerous bench-scale CPUs.) A tuyere consists of three concentric metal tubes cast into a removable refractory block that is bolted into the bottom of the CPU. The TPC proposes using the tuyere injection of liquid agent and cofeed gases for chemical demilitarization.
Feed material, which may be liquid, gas, finely divided entrained solids, or a pumpable slurry, is metered, mixed, and pumped through the central tube of the
tuyere at moderately high pressure, less than 10 atmospheres. Oxygen, in stoichiometric proportion to convert all carbon in the feed and the methane cofeed to carbon monoxide, is metered into the next annulus at high velocity to induce turbulence, mixing with the feed stream, and formation of a jet that rapidly breaks up into small bubbles. A small amount of methane is fed through the outer annulus to cool the tuyere.
An inert gas is injected automatically into each of the feed lines as needed to make up the difference between the total flow required in each line and the set-point flow of each feed component (agent, oxygen, and methane). During startup and shutdown, the inert gas alone is pumped through all feed lines to prevent molten metal from entering and plugging the tuyere.
According to the TPC's description of the process, when feed material is injected into the bath along with oxygen and methane, the molecular entities in the feed material are decomposed by catalysis into their component elements. These elements dissolve in the metal and form intermediates by bonding chemically with the metal. By appropriate selection of process conditions, the dissolved elements with high solubility in the metal (e.g., carbon, sulfur, and phosphorus) can either be retained in the metal bath up to their saturation limit or induced to react with less soluble elements (e.g., hydrogen, oxygen, and chlorine) to form gaseous products—principally H2, CO, HCl, and H2S with minor amounts of H2O, and CO2. These gaseous products then form bubbles, which ascend and exit the bath. According to the TPC, because CEP is carried out at low oxygen potential and decomposes feed molecules to elements regardless of their starting molecular structure, the process provides neither pathways nor precursors for the formation of oxides of nitrogen or sulfur or the formation of dioxins and furans.
The TPC has reported that it expects the process residuals from treating VX or HD, the ton containers, and dunnage to be ferrous alloys, aqueous hydrochloric acid, elemental sulfur, and a synthesis gas. The TPC also has reported that markets for the alloys, hydrochloric acid, and sulfur have been identified. The synthesis gas is combusted, along with natural gas, in an on-site gas turbine generator to provide electricity used in the process. A small amount of slag or ceramic (less than 5 percent of total solid product mass) is also produced and must be disposed of as waste. The panel agrees with the TPC that this slag is likely to pass the U.S. Environmental Protection Agency Toxic Characteristic Leaching Procedure (TCLP) test. (Unless it is delisted, however, it could still be classified as hazardous waste because it is derived from agent.)
Chemical Demilitarization Process
According to the submitted design, chemical demilitarization operations are to be conducted in a central processing building of approximately 13,000 square feet. The building is partitioned into distinct areas by function (Figure 4-1). Precautionary safety measures confine agent to small areas, reduce the possibility of cross contamination, and reduce requirements for heating, ventilation, and air conditioning (HVAC); high efficiency particulate-arresting filters; carbon filters; and agent monitoring equipment.
Ton containers are opened in area 100 and, if necessary for interim storage, cleaned to 3X condition. Dunnage from daily operations is compacted and packaged in small metal containers in the same area. The equipment and techniques used to handle ton containers, including the punch-and-drain process, vacuum transfer of agent and decontamination liquids to interim storage tanks, safe airlock passage, cascaded HVAC, double-containment envelopes, and low pressure injection are based on the equipment and techniques used in the baseline system facilities at Johnston Atoll in the Pacific Ocean and at Tooele, Utah. The only significant change is the addition of an aspirated, self-cleaning gland surrounding the punch to mitigate spillage of agent when the container is penetrated.
The two CPUs, designated CPU-1 and CPU-2, are located in area 200. The gas handling train (GHT) and facilities for product recovery are located in Area 300. Area 500 is devoted to product gas utilization; products of CEP are stored in area 700; utilities are located in area 800; and area 1000 houses the emergency relief system. The CPUs and the equipment in the product recovery areas are of modular design, which will allow the TPC to use the same CPUs and product recovery equipment at the Aberdeen site to process HD and, afterward, at the Newport site to process VX.
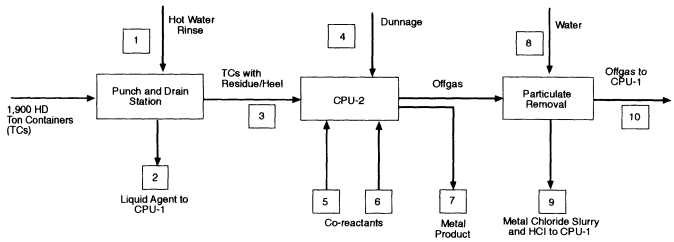
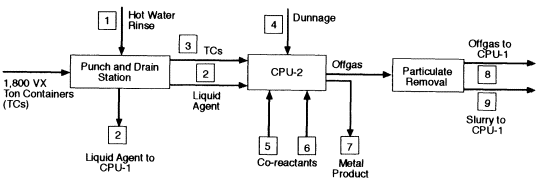
For processing either agent, CPU-2 contains molten iron and processes all ton containers and dunnage. Emptied ton containers are fed by horizontal indexing conveyors and coordinated, double-door, cascade-ventilated airlocks to the premelting side chamber of CPU-2. The steel ton containers melt, and the organics, including all remaining gels, solids, and surface agent residuals, are pyrolyzed. Pyrolysis products and molten
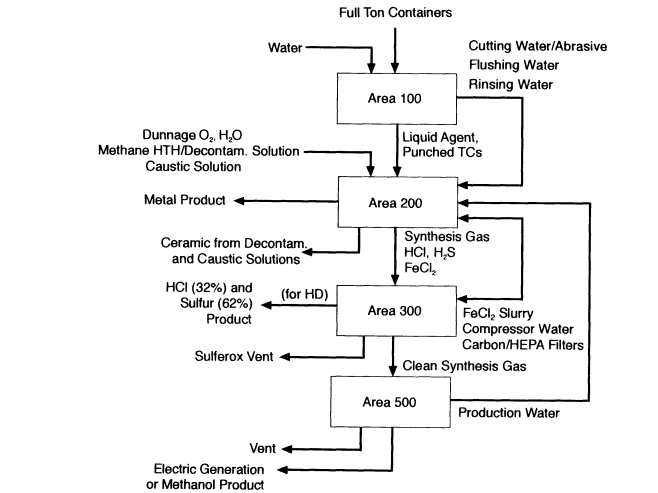
Primary agent and residue process flows for a chemical demilitarization CEP facility. Area 700 (product storage), Area 800 (utilities), and Area 1000 (emergency relief system) are not shown. Source: M4 Environmental L.P., 1996b.
metal then enter CPU-2 through a side chute above the level of the molten bath. The TPC states that dunnage canisters will be fed directly into CPU-2. If the ton containers are melted as they are emptied, at the proposed processing rate of VX (169 kg/hour) they will add about 725 kg of metal to the bath every 5 hours. This quantity of metal will increase the bath height about 8 cm, necessitating tapping the bath at approximately 10-hour intervals to maintain an optimum level. The metal tap, which will probably be located at the desired bath height, will be opened by heating it to melt the metallic or slag plug. The tap will be closed by cooling it to solidify a metal or slag plug.
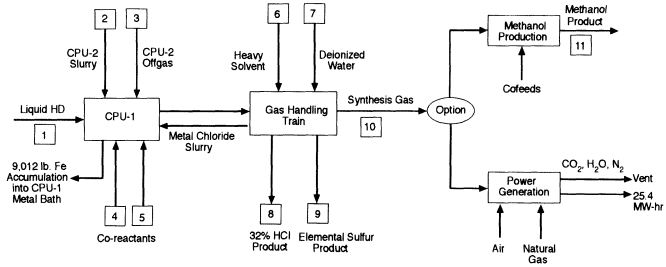
Different strategies are required for processing HD (Figure 4-2) and VX (Figure 4-3). In the HD strategy, liquid agent is injected by tuyere into CPU-1, which uses a molten nickel bath to reduce the formation and carryover of metal chlorides. Chlorine is released from the bath as HCl. Sulfur from the HD accumulates in the bath to a concentration of about 27 percent, a concentration at which sulfur is released from the bath as HS. The offgas from CPU-2, which originates from processing the ton containers, any residue in them, and dunnage is quenched with water, pressurized, and injected into CPU-1 to ensure complete reaction of any products of incomplete conversion. Product gases from CPU-1 are quenched with water, filtered, and scrubbed with water to recover aqueous HCl. At this point, the offgas consists primarily of H2S, CO, and H2. The H2S is subsequently converted to elemental sulfur using the commercial SulFeroxTM process. The remaining gases, principally H2 and CO, form the synthesis gas, which is pressurized and stored in one of three
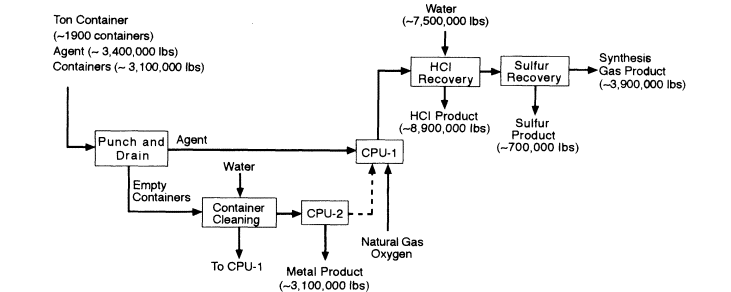
High level block diagram for the destruction of HD by CEP. Source: M4 Environmental L.P., 1996b.
tanks with a capacity of 4 m3 each. After a filled tank has been analyzed for agent and other toxic, the gas is combusted in a gas turbine electric generator.
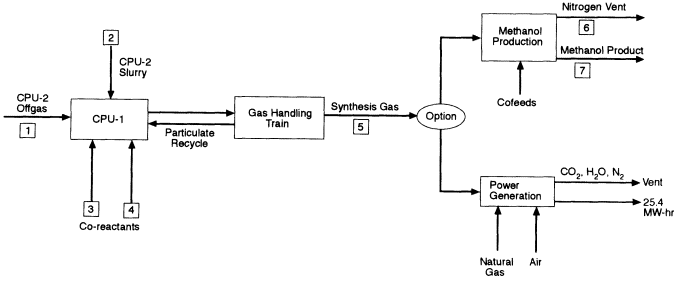
In the VX strategy, CPU-2 is the primary reactor for processing agent. Both sulfur and phosphorus from the VX are held in solution in the molten iron and recovered as an Fe-S-P alloy when CPU-2 is tapped to control the bath level. The offgas from CPU-2 is conditioned as described above for HD and injected into CPU-1, which in this case contains an iron bath and functions as a polishing reactor to ensure the destruction of remaining agent or other organics. The offgas from CPU-1 is quenched with water and filtered to yield the synthesis gas of CO and H2. Trace amounts of HCN in the product gas are decomposed by catalysis to H2, N2, and carbon. The VX strategy uses the same approach as the HD strategy for storing and analyzing the synthesis gas prior to combustion.
In both treatment strategies, aqueous cleaning and decontamination solutions, including particulates and condensates recovered as water-base slurries from cooling and cleaning the CPU offgases, will probably be injected into CPU-2 for destruction, so that all slag-forming components are kept in the same CPU. Slag formed by the interaction of debris entering with the emptied ton containers, lime-based decontamination solutions, and dunnage can be removed in the same way molten metal is removed.
Should the need arise, the facility design includes the capability of opening a ton container with a high pressure water-jet containing abrasive particles. A water spray then removes the gels, residues, and remaining agent, and calcium-based decontamination solution is used to clean the container to 3X condition. The resulting finely divided aqueous slurry can be removed from the cleaning area by aspiration, transported by vacuum pumping to temporary storage, and injected into one of the CPUs for processing to the same residuals as other cleaning solutions and slurries. The use of a water-jet, of course, would require suitable enclosure and capture/treatment of effluent from the spray operation.
If a situation arises in which liquids or gases from vessels, piping, or either CPU are vented by means of pressure relief devices, the facility design includes standby equipment to quench the vented material and absorb acid gases. Any residual agent or HS is combusted in a standby boiler prior to releasing the gaseous residual to the atmosphere.
Scientific Principles
The TPC and the developer of CEP describe the molten metal bath as a dissociation catalyst for molecular entities in feed materials, a solvent for elemental
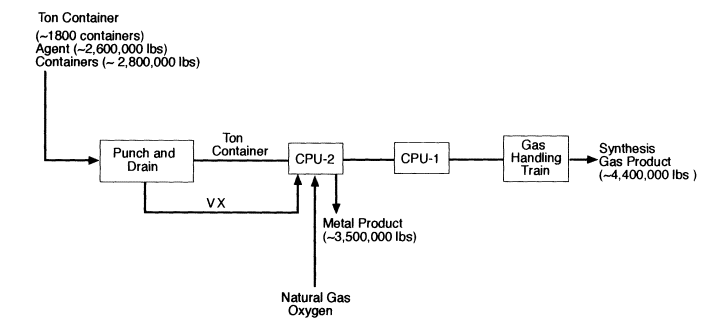
Figure 4-3
High level block diagram for the destruction of VX by CEP. Source: M4 Environmental L.P., 1996b.
fragments, and a medium for product synthesis. The TPC divides the process conceptually into stages comprising catalytic dissociation of the feed, formation of elemental intermediates with the solvent metal, product synthesis by interaction of elemental intermediates, and partitioning of products among metal, slag, and gas phases. A recent publication by technologists who work for the developer of CEP states, "the CEP unit is not acting as a thermal treatment device in that temperature is not the primary means to change the physical and chemical composition of the feed material . . ." (Nagel et al., 1996, p. 2158).
The above description does not address initial thermal and gas-phase reactions in the overall sequence of events between the introduction of feeds and the release of final products. Although bench-scale tests of the process have demonstrated that the process can destroy agent as required by the Army, analysis by the AltTech Panel indicates that the actual conditions are probably more complex than this description implies. The panel's review indicates that a complete description of the scientific principles underlying CEP requires discussion of several additional phenomena, including gas-phase reactions among agent, oxygen, and methane in the inlet jet immediately following tuyere injection; interactions of these gases and intermediate products with metal vapor inside bubbles; and boundary reactions between bubble components and the surrounding metal. Accordingly, the following discussion attempts to provide a more detailed description of the probable scientific principles and further develops details of the probable processes involved.
The TPC notes that the submitted design reflects many years of experience in the steel industry with injecting gases into molten steel baths by the use of similar tuyere inlets. However, experience in the steel industry relates primarily to the injection of gases for the purpose of changing the composition of the bath. The escape of a small surplus of these gases from the bath surface is of little concern other than as an economic loss. Thus, there is no long-established precedent from industrial experience for the complete reaction of injected gases with a molten metal bath to the very low-level of residuals required for agent destruction. The panel is not aware of industrial experience with injecting liquids into a molten metal bath.
Dissociation and Reaction of Tuyere-Injected Materials
In the CEP, a liquid agent or other feed to be destroyed, inert carrier gas, oxygen in stoichiometric proportion to oxidize all carbon in feeds and cofeeds to CO,
and methane are injected by tuyere at moderately high pressure (less than 10 atmospheres) and high velocity into the molten metal bath. The injected materials form a jet that extends several tuyere diameters into the bath. The high velocity of the oxygen gas stream causes turbulence and contributes to entrainment of metal vapor and droplets within the jet. These effects of the initial momentum quickly dissipate, and the jet breaks into bubbles that rise through the molten metal because of their buoyancy. Subdivision of larger bubbles increases the total surface-contact area and increases the collision frequency between gas molecules and the molten metal. As the bubbles rise to the surface, they continue to change in size for several reasons. They tend to increase in size as the ferrostatic head decreases; they tend to decrease as gaseous intermediates are absorbed into the molten metal; and they tend to increase as product gases released from the molten metal migrate back into them. Some very small bubbles may also form through the nucleation of gases produced in the molten metal and then grow as they agglomerate with other bubbles or accumulate more gas released from metal.
Radiant heat transfer from the hot metal to the aspirated liquid droplets and gas bubbles is extraordinarily rapid at the high temperature of the bath because the rate of radiant heat transfer is proportional to the fourth power of the absolute temperature. For example, a hypothetical sphere 100 µm in diameter will receive energy at 1600°C at the rate of 5 × 10-3 calories per second, which is sufficient to vaporize a like volume of liquid agent and heat the resultant vapor, as multiple 100-µm bubbles, to 1000°C in less than 50 milliseconds. The panel's judgment is that partial degradation of agent and gas-phase reaction between agent or agent fragments and oxygen is very likely under these circumstances. A significant fraction of the feed probably undergoes partial oxidation, and the products of partial oxidation then interact with the molten metal to form intermediates. The panel also concludes that oxidation is probably not complete and should not be termed combustion, even though reactions proceed stepwise by molecular collisions among gas-phase intermediates.
Increasing the effective pressure of the bubbles increases the gas density and therefore the collision frequency between bubble contents and the molten metal. Thus, increasing the operating pressure of the CPU or increasing the bath depth increases the rates of reactions in the bubbles. The TPC has ascertained that the processing rate for a given reactor increases significantly with an increase in operating pressure.
An important issue is whether there is opportunity for back reactions to form complex organic compounds from intermediates. The assumption that the opportunity is negligible is important to the TPC's statement that no detectable recombinant dioxins or furans are produced. However, it is possible and thermodynamically feasible to produce HCN in the conditions of the CEP bath when processing VX. In the original submission from the TPC, the inert gas was specified to be nitrogen. The TPC has subsequently considered using argon for this purge/make-up gas. For processing HD at least, using argon instead of nitrogen would resolve the issue of HCN formation by removing any source of nitrogen. Although the extent of HCN production can be controlled to very small concentrations, the fact that it does occur indicates that the claim that no detectable recombinant dioxins or furans (i.e., complex compounds) are produced does not apply to simple compounds like HCN.
Dissolution kinetics are also important to the formation of intermediates. For example, hydrogen is sparingly soluble in molten iron, and when organic compounds containing hydrogen are injected into molten iron, hydrogen gas evolves from the bath while the carbon dissolves in the metal. It is also reasonable to expect that the initial bubbles formed by the break-up of the jet contain H2. (If nitrogen were used as the inert make-up gas, N2 would also be a significant component of the initial bubbles.)
Catalysis by the Bath and the Formation of Intermediates
There is ample evidence in the peer-reviewed literature to support the TPC's position that the molten metal bath serves as a true catalyst by decreasing the activation energy for dissociation of organic molecules, participating in the formation of intermediates, and increasing the efficiency of product formation without itself undergoing change (Satterfield, 1991). Given the formation of intermediates, their relative solubilities in the metal are another factor to consider, particularly for the VX strategy, in which some elements are to be retained in the bath while others exit as offgas.
The panel estimated the solubility of VX components in the bath and the time required to saturate the bath under processing conditions of 1600°C and the proposed feed rate (Table 4-1). Columns 2 and 3 list the saturation solubility (in parts per million by weight) and the total weight of elements in the bath, based on a reasonable
TABLE 4-1 Calculated Solubility of VX and Cofeed Elements in Iron at 1600°C and Time to Saturate the Iron Bath at Processing Conditions
|
|
Solubility in Bath |
|
|
|
|
Element |
ppm |
kga |
Feed Rate kg/hb |
Time to Saturate Bath h |
|
C |
54,000c |
442 |
87.4 |
5.05 |
|
H2 |
25d |
0.20 |
17.8 |
0.011 |
|
P2 |
110,000c |
892 |
19.6 |
45.5 |
|
O2 |
1,290c |
10.6 |
130.7 |
0.081 |
|
S2 |
110,000c |
892 |
20.3 |
43.9 |
|
N2 |
88d |
0.14 |
8.9 |
0.016 |
|
Notes a Bath assumed to contain 8,163 kg iron; contribution of dissolved elements was not considered. b Feed rates: 169 kg/h VX agent; 110 kg/h oxygen; and 5 kg/h methane. c From Massalski, 1986, pages 842 (C), 1746 (P), and 1762 (S). d From Rao, 1985, pages 438 (H2) and 463 (N2). |
||||
assumption of the partial pressures of the gases derived from the feeds. Column 5 lists the time required to saturate the bath at the elemental feed rate given in column 4, which is derived from the molecular composition of the feed and cofeeds and their feed rates. These values are only computational estimates; numerous simplifying assumptions were needed, and interactions among bath components were ignored. However, the calculations do illustrate the following points.
Bath Saturation Point for Retained Elements. Because the solubilities in molten iron of carbon, phosphorus, and sulfur are significant, amounting to 5.4, 11, and 11 wt pct, respectively, considerable time is required to saturate the bath with these elements. The TPC's strategy for VX calls for controlling the release of phosphorus and sulfur gases (preventing breakthrough) by keeping the bath below saturation. The strategy is to remove alloyed bath metal at intervals by tapping, while adding molten iron by processing ton containers. Once the bath reaches saturation for phosphorus or sulfur, the ton containers must be processed at a rate sufficient to supply enough new iron to alloy all the phosphorus and sulfur in the agent feed. The calculated values in column 2 of the table indicate that the amount of iron in a ton container, 636 kg, will dissolve only about 69 kg of sulfur and a similar quantity of phosphorus. The 682 kg of VX within a ton container contains about 82 kg of sulfur and 79 kg of phosphorus. Although these calculations are based on numerous simplifying assumptions, they indicate that synchronizing the addition of iron to the bath with the agent feed rate will be critical in avoiding the breakthrough of sulfur and phosphorus into the offgas. In particular, these computations indicate that the TPC's suggestion of stockpiling ton containers for treatment at a later date while processing VX is not an option unless there is a significant alternative iron feed.
Hydrogen and Nitrogen. The solubilities of hydrogen and nitrogen in molten iron are extremely low, and Table 4-1 suggests that the bath will become saturated with these elements in less than 1 minute. Although the bath, when in continuous operation for processing VX, is likely to be saturated with hydrogen and nitrogen, the kinetics indicate that significant proportions of hydrogen and nitrogen in the feed may not pass through metallic intermediates but may form gas bubbles directly. Supersaturation of the bath as a whole with these and other sparingly soluble elements is likely because the feed materials are introduced into the bath at the bottom, where the ferrostatic head is greatest.
Oxygen. The solubility of oxygen in molten iron is much greater than hydrogen or nitrogen but far less than carbon, sulfur, or phosphorus. The calculated time of less than 5 minutes for the bath to become saturated reflects the high feed rate. The solubility of oxygen favors the formation of an iron-oxygen intermediate.
These calculations indicate all components in the feeds and cofeeds are soluble enough to support the TPC's description of the formation of elemental intermediates. Given the formation of elemental intermediates, product synthesis can occur by chemical reaction among those intermediates.
Partitioning of Products among Metal, Slag, and Gas Phases
To some extent, the process residuals from CEP can be customized by adding appropriate cofeeds or controlling operating conditions. As noted above, the design specifies that oxygen cofeed is provided in stoichiometric proportion to convert carbon in the feed material and the methane cofeed to CO at the desired carbon concentration and temperature of the bath. The oxygen stoichiometry determines the ratio of CO to CO2 in the product gas, and this ratio is monitored as a process control on the oxygen feed rate. Hydrogen appears as H2 in the product gas because the oxygen potential in the bath is less than the potential required to form significant amounts of H 2O. Similarly, SO2 and NOx formation are thermodynamically unfavorable.
For processing HD, sulfur can be recovered in the gas-phase by allowing sulfur in the bath to increase to a saturation concentration above which the formation of H2S from H2 and the Fe-S intermediate is thermodynamically favored. Or, sulfur can be recovered as an alloy element by tapping bath metal from the CPU before the saturation concentration is reached, as the TPC proposes to do for processing VX. The chemistry of phosphorus, although more complicated, is similar in that phosphorus can be obtained as an iron alloy by tapping the metal before the saturation concentration is reached. The panel notes, however, that although CEP has been performed extensively with iron baths containing carbon, sulfur, and chlorine, to the panel knowledge it has not been performed with iron baths containing phosphorus in addition to carbon and sulfur.
Metals such as aluminum, calcium, and silicon that form oxides that are more stable than CO at the operating temperature will be oxidized and will accumulate in the slag phase (as Al2O3, CaO, and SiO2, respectively). Cofeeds may be required to ensure the slag is sufficiently fluid. For example, silica and lime are appropriate cofeeds if the feed material contains appreciable aluminum or alumina. Metals whose oxides are less stable than CO will either accumulate in the molten metal (Co, Cr, Cu, Ni, Mn) or exit the bath as vapor (Cd, Pb, Zn).
Iron is the preferred bath metal for processing VX. However, if iron were used to process HD, there would be substantial formation and carryover of FeCl2 vapor, which would form a dust in the downstream systems, requiring a more extensive dust removal strategy than the particle filters included in the current design. The use of a nickel bath for processing HD reduces this problem because NiCl2 is less stable than HCl and does not form to a significant extent. Nearly all of the chlorine from the HD forms HCl and is recovered in the aqueous scrubber. Under the same processing conditions, a nickel bath will become saturated with sulfur in about the same time as an iron bath of equal mass and will become saturated with carbon in less than half the time of an iron bath.
Process Modeling
The most important consideration to the panel, in light of the short residence time of bubbles in the bath, is whether agent or significant fragments of agent can avoid decomposition by remaining in or migrating to a bubble and passing unreacted through the bath. An analysis of the probability and consequences of the requisite reactions at the molecular level would involve complicated computations dependent on numerous assumptions. Instead, it is customary in such circumstances to use engineering models that work from both basic principles and experimental data to provide an approximation adequate for design purposes. The TPC has done extensive experimentation and modeling to understand bubble formation, break-up dynamics, and the operating limits of CEP performance. The models used by the TPC indicate that the process depends heavily on three factors: (1) bubble size, with the critical largest-bubble diameter being on the order of a fraction of an inch (the actual size is proprietary); (2) residence time, with the typical single-path residence time being a fraction of a second (actual time is proprietary); and (3) an energy dissipation term that reflects the degree to which metal vapor and droplets inside the bubbles increase the gas-metal contact.
Although these models were developed and used by the TPC, the panel did not review or evaluate them in detail for this report. Rather, the panel has relied upon the TPC's representations that the model results correlate well with the very high DRE (destruction removal efficiency) values that were achieved in the experimental and commercial-scale demonstration reactors to which the models were applied. The TPC has stated that it intends to use a residence time that provides a design safety factor of at least 10 to assure the destruction of VX or HD agent to at least the required six 9's DRE (99.9999 percent).
Conclusions on the Underlying Science
The TPC's explanation of CEP performance is based upon accepted free energy principles.3 The panel believes the engineering design models used to design the system have been based upon solid scientific data. The panel did not, however, review these models in detail.
The TPC' s original submission did not include equipment for holding the synthesis gas until analysis had ensured the complete destruction of agent or other toxic components prior to combusting the gas in a gas turbine or using it in some other way. However, in response to the concerns of communities near the storage sites, the TPC has subsequently changed the design to include three 4-m3 storage tanks, in parallel, in the synthesis gas line prior to the gas turbine. Each tank has the capacity to store 15 minutes of anticipated output of synthesis gas pressurized to 20 atmospheres, gauge (300 psig). This storage capacity allows the synthesis gas to be analyzed before it is used as a fuel and the emissions are released to the atmosphere.
The proposed design for a chemical demilitarization facility is undergoing continuous development as the TPC accumulates operating experience in other applications. The opinion of the AltTech Panel is that the process is adequately understood and satisfactorily engineered at this time to process either HD or VX successfully and safely, when operated properly, to meet the required six 9's DRE.
Technology Status
The information available to the panel on CEP operational units is summarized in Table 4-2. As of early 1996, the TPC reported more than 15,000 hours of molten metal test experience with its reactors. Much of this experience was in tests on the 10 to 15 bench-scale units at the TPC's Fall River site. The nominal bath size of these units is 4 to 9 kg.
Fall River Demonstration Unit
The Fall River Demonstration Unit (Demo Unit) is the largest operational CPU. As of April 1996, the longest period of continuous, commercial-scale operation in this unit while processing liquid or gaseous organics was 120 hours, during which 1,680 kg of feed was processed. The associated on-stream factor was between 50 and 80 percent, depending on experimental requirements.4 The TPC plans to use an on-stream factor of about 82 percent for the CPUs for destroying HD and VX at Aberdeen and Newport.
The TPC also reports that the Demo Unit was used to demonstrate the long-term operability, reliability, and product performance of CEP as a contractual milestone prior to an agreement with a major chemical manufacturer to build a commercial facility. The 93-hour test included a switch-over from injecting solid feed material (biosludge) to injecting heavily chlorinated liquid organic material (RCRA waste F024). The TPC reports that the results of this test surpassed more than 40 performance criteria (for environmental protection, product quality, reliability, operability, feed injection, etc.) established by the customer, Hoechst Celanese. The reported test results included an on-stream factor up to 90 percent, mass balance closures at 100 percent, and feed injection rates that met commercial-operation requirements. The TPC reported that steady-state operational requirements were met and surpassed (validated by on-site customer evaluations), as demonstrated by the steady-state production of high-quality synthesis gas that met the customer's on-site recycling requirements.
TABLE 4-2 Status of CEP Units from Bench Scale to Commercial Scale a
Oak Ridge Facilities
The Quantum-CEP reactor units at the TPC's Oak Ridge site are referred to as RPUs (radioactive processing units). Members of the AltTech Panel observed the bench-scale units at Oak Ridge in operation during site visits.
The SEG/Quantum-CEP units are located at a separate site in Oak Ridge and are designed for batch-mode commercial operations. Each campaign will consist of a 36-hour startup, 3 to 5 days of injection of radioactive ion-exchange resins, and a 36-hour shutdown, for a total campaign duration of 6 to 8 days. During the panel's site visit in March 1996, the SEG facility at Oak Ridge was still in scale-up activities using nonradioactive resins, prior to commercial operation. As of May 1996, the facility was reported to have processed more than 27,000 kg of ion-exchange resins. The TPC reported that a peak throughput rate of 150 percent of design had been achieved and that equipment upgrades were being made.
Agent Testing
Battelle/Columbus Laboratory (a member of the team that prepared the TPC submissions) has tested agent destruction in a bench-scale CEP unit. The TPC has issued a news release reporting a "destruction percentage" of eight 9's (99.999999 percent) for processing HD and VX (M4 Environmental L.P., 1996a). From the AltTech Panel's preliminary review of the full report on these tests, the panel concludes that the tests demonstrated that the CEP technology can destroy agent to at least the six 9's DRE required by the Army. Further implications of the test results for a full-scale operation are discussed below in the section on Scale-Up.
Summary of Technology Status
The development of the various subsystems required for a chemical demilitarization facility has been demonstrated by successfully injecting feed materials, generating process products, and achieving high on-stream factors at developmental facilities.
A wide range of materials has been processed, including polystyrene with graphite, ion-exchange resins, acetone, industrial biosolid waste, chlorotoluene with heavy organics, chlorobenzene, fuel oil with chlorotoluene, dimethyl acetamide with heavy organics, benzonitrile, diazinon, diazinon with sulfur, and surplus metal components. These materials have been in various physical forms, including liquids, slurries, fine solids, and bulk solids. Various feed-addition systems, including configurations with a top-entering lance or a bottom-entering tuyere, have been studied. Successful tuyere injections of liquids, slurries, and fine solids have been demonstrated in which the injection rates and the reactor design were optimized for steady-state operations. Injection rates comparable with commercial levels have been demonstrated at both the demonstration-scale and advanced processing units.
Bulk additions of metal components, scrap metals, and wood have been demonstrated at feed rates comparable to commercial- scale and with successful conversion of materials. The TPC's design for processing bulk solids uses two reactors. The receiving unit includes a premelting chamber for melting and volatilization. The second unit is used to polish the offgas from the first unit.
Panel Summary of Technology Status
As of May 1996, the TPC has accumulated considerable test experience with CEP technology, as described above, and is gaining commercial experience. However, the TPE does not yet have extended, continuous commercial experience with CPUs of commercial size.
Process Operation
Process Description
The TPC provided the following process diagrams, which will be referred to in this and subsequent sections as needed:
- Block flow diagram for CEP facility (Figure 4-4)
- CEP process flow diagram for VX feed injection system into CPU-2 with premelting chamber for ton containers (Figure 4-5)
- CEP process flow diagram for VX CPU-2 offgas treatment (Figure 4-6)
- CEP process flow diagram for VX CPU-1 gas handling train (Figure 4-7)
- CEP process flow diagram for VX relief system (Figure 4-8)
- CPU block diagram and material balances for HD treatment (Figure 4-9)
- CPU block diagram and material balances for VX treatment (Figure 4-10)
- CEP heat and material balances for VX gas handling (Table 4-3)
Agent Detoxification
Residual Agent
Based on tests using HD, VX, and agent surrogates as CEP feed materials, the TPC anticipates a DRE for each agent in excess of six 9's (99.9999 percent). If, as the result of equipment failure, operator error, or some other circumstance, residual agent remains in the synthesis gas emerging from the gas handling train (see Figure 4-7), it can be detected in the hold-up tanks before the gas is released to the energy recovery system for combustion. If analysis of a tank detects the presence of agent above the six 9's DRE limit, the contents can
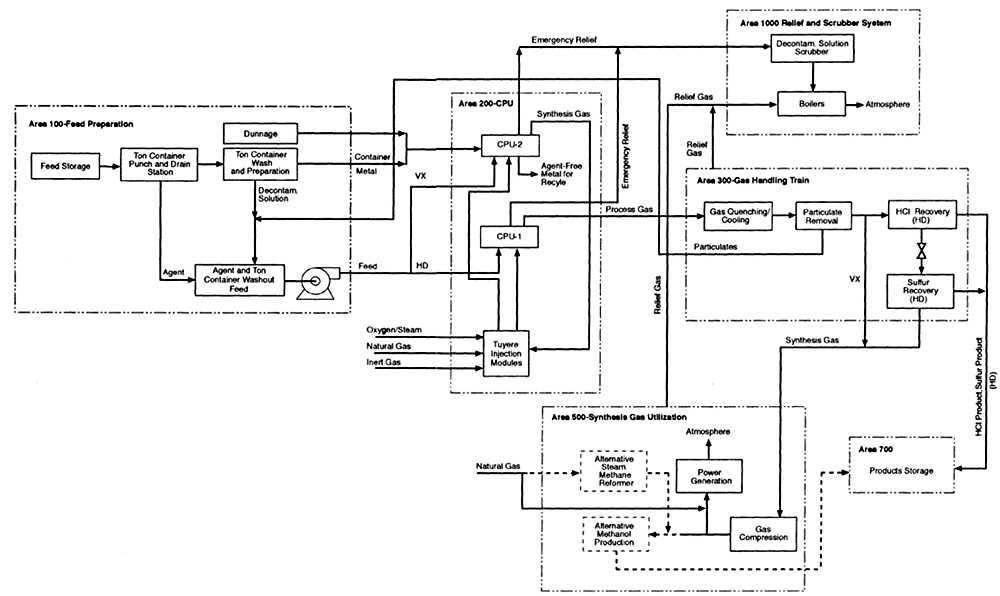
Figure 4-4
Block flow diagram for CEP facility. Source: M4 Environmental L.P., 1996b.
be recycled to the appropriate CPU for retreatment. Neither the TPC nor the panel expects that agent or other off-specification gases will be emitted from the process.
In a case requiring venting gases from the CPUs, piping, or other vessels by way of the pressure relief system (Figure 4-8), the on line caustic scrubbers would further destroy any agent that might potentially
enter the relief header downstream of the reactors. (The exact level of destruction is not known, but it would be more like a 3X condition than a 5X condition, if agent did in fact exit the CPU.) Only under unusual circumstances would the relief system be exercised. If it is, the only residuals would be the scrubber liquor wastes, which would not contain agent above the 3X level.
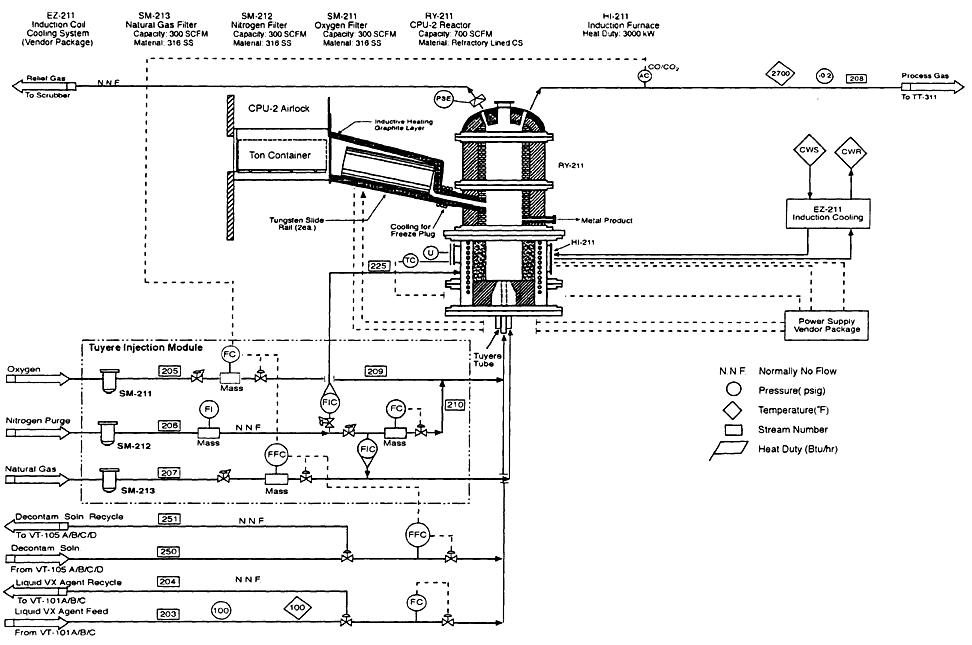
Figure 4-5
CEP process flow diagram for VX feed injection system into CPU-2, with premelting chamber for on containers. Source: M4 Environmental L.P., 1996b.
Reversibility of Reactions to Reform Agent
None of the process reactions is reversible to the extent that agent could be reformed. The formation of chemical warfare agents as unintended by-products in the product stream from CEP treatment of HD or VX is not possible under the proposed operating conditions. The reaction paths and conditions required for the production of HD or VX from species in the product gas stream will not be present in an operating CEP plant.
Toxicity of Process Residuals
The solid, liquid, and gaseous residuals from the process are discussed below in the section on Residual Streams. The process as designed does not produce
residuals with toxicities that are known to be hazardous to human health or the environment.
Cleaning Out Ton Containers
It is not necessary to remove all residual agent from the ton containers prior to their destruction by CEP. The procedure presented by the TPC ensures detoxification to the Army 5X standard because the containers are melted, a treatment at more severe conditions than the conditions required by the 5X standard. Analysis of ton containers prior to processing is not necessary, provided they are not stored prior to CEP treatment. (Interim storage of emptied containers would require cleaning to the 3X standard.) The molten metal and slag phases from CPU-2 will be cast into ingot or slag molds, as
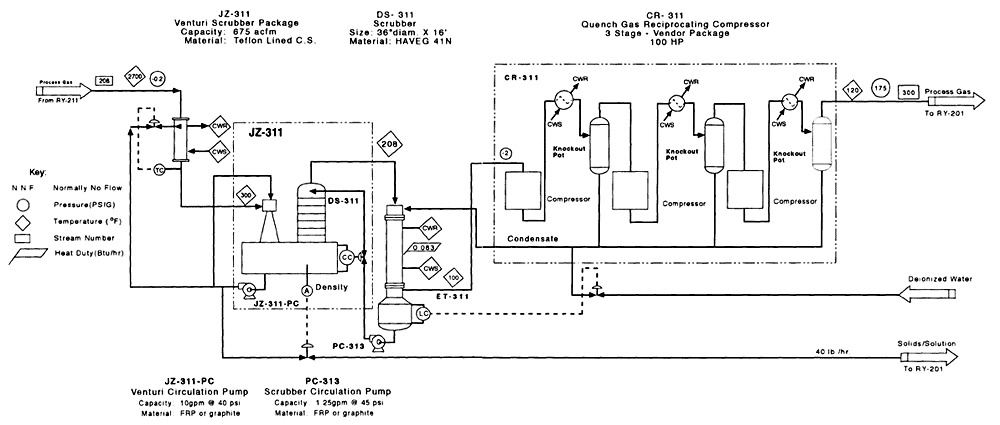
Figure 4-6
CEP process flow diagram for VX CPU-2 offgas treatment. Source: M4 Environmental L.P., 1996b
appropriate. The metals will be offered for sale, and the slags will be committed to an appropriate landfill, as determined by TCLP testing.
Operational Modes
Substantial time is required to heat the CEP system, including the CPUs and the gas handling trains, to operating temperature or to cool the system from operating to ambient temperature. Therefore, it is preferable to operate a CEP facility continuously, 24 hours per day, for extended periods. The units can be kept in a shutdown-but-ready mode if electrical power to the induction coils keeps the bath near operating temperature and if the tuyeres are kept open by maintaining flows of inert gas through the feed lines in place of the agent, oxygen, and methane feeds.
Startup and Shutdown
As explained above, it is preferable to operate a CEP facility continuously, 24 hours a day. Startup and shutdown typically cause the greatest wear on the process equipment. Although operating the system for only 8 hours a day is technically possible, it is not a reasonable approach. Startup of the CPUs requires:
- opening the vessel and filling it with a weighed quantity of iron or nickel spheres (or other metal shapes)
- installing the gas-fired headspace heater
- starting the systems for handling offgas from each CPU
- starting inert gas flow through the tuyeres to keep them open and cool as the bath metal heats and melts
-
- preheating the CPUs with a gas-fired heater through the critical metal melting stage
- inserting additional metal, if required to adjust metal level
- stopping and removing the preheater and closing the reactor vessel
- turning on electrical heaters to gradually heat the downstream equipment for the gas handling systems (to avoid too rapid heating of the HavegTM or other special materials in the HCl recovery area)
- switching from inert gas feed to feed streams of methane, oxygen, and finally agent
Shutdown to a hot standby mode requires gradual substitution of an inert gas for agent, oxygen, and methane to keep the tuyeres open; readjustment of the electrical power to keep the baths molten; and maintaining the gas handling trains for both CPUs at operating temperature. Restart from hot standby is the reverse of this shutdown procedure.
Moving to a cold shutdown from a hot standby mode requires that the metal and slag be drained and that the CPUs be allowed to cool. Failure to drain the units would require breaking out the solidified metal and replacing the refractory.
CPU-2 Operation
The configuration and operation of the CPUs are similar except that CPU-2 has a side chamber to melt the ton containers. Emptied ton containers, which may contain agent residues, enter this premelting chamber on CPU-2 by means of horizontal indexing conveyors and coordinated double-door, cascade-ventilated airlocks. The chamber is purged with inert gas, and the chamber induction coil is activated to heat the
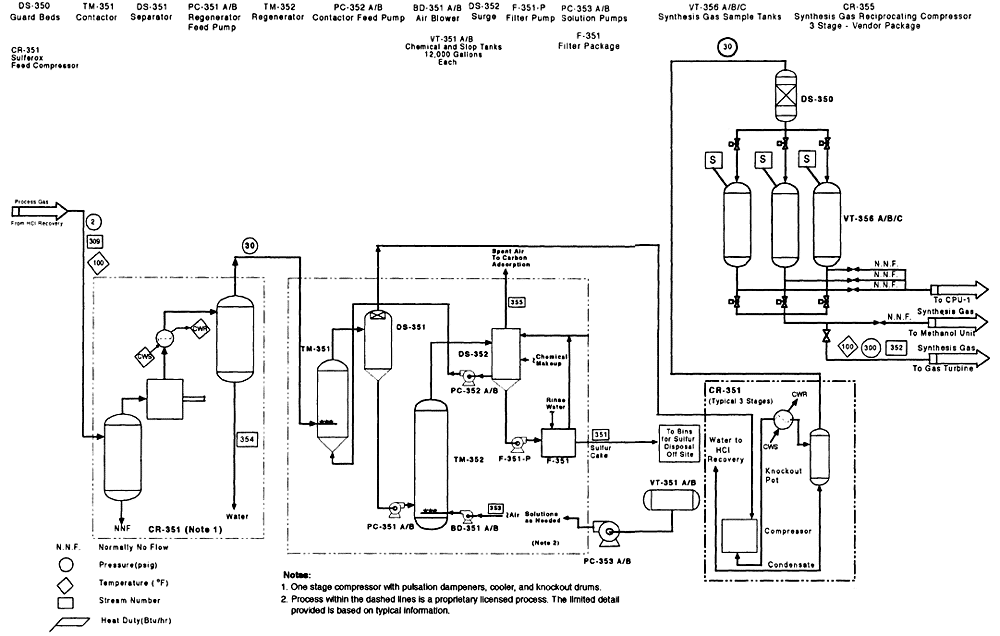
Figure 4-7
CEP process flow diagram for VX CPU-1 gas handling train. Source: M4 Environmental L.P., 1996c.
chamber and melt the metal. Visual observation through a viewport determines when melting is complete.
Molten metal is tapped from CPU-2 at intervals, as needed to maintain appropriate bath depth and remove Fe-S-P-C alloy (in the case of VX processing). The bath is tapped by opening a proprietary-design tapping nozzle on the side of the bath. The tap is opened by heating to melt the solidified metal plug. The molten alloy flows out into a mold. When the desired amount of alloy has been removed, the heating is replaced by cooling to solidify the molten alloy in the tap to form a
metal plug. Ceramic slag is similarly tapped at intervals, as required.
Feed Streams
This section discusses only the feed streams into the facility and not the internal process streams.
Agent
The design flow rate for chemical agent is set to achieve destruction of the stockpile at each site in a nominal one year period. The HD design flow rate is 204 kg/h to CPU-1. The VX rate is 169 kg/h to CPU-2.
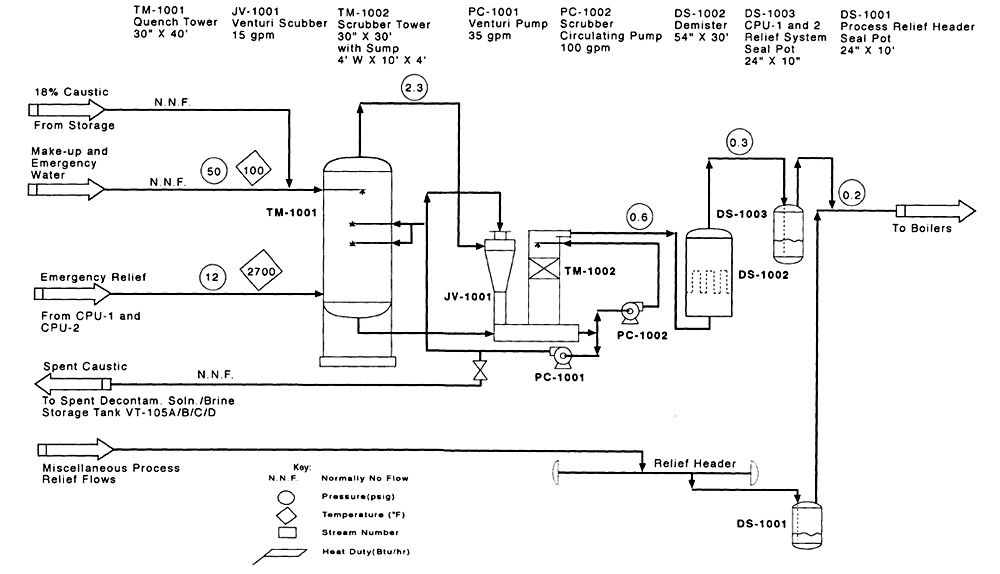
Figure 4-8
CEP process flow diagram for VX relief system. Source: M4 Environmental L.P., 1996b.
Metal
At cold startup, each of the CPUs is loaded with iron (or nickel for the HD CPU-1). For HD processing, there is no additional metal feed stream (other than metal from ton containers and dunnage canisters) unless the units are drained for maintenance or repair and then restarted. The same is true for VX processing, provided the addition of ton containers can be synchronized with the agent feed rate, as explained in the section on Catalysis by the Bath and the Formation of Intermediates.
Gases
Oxygen is used to oxidize the carbon in the agent and the methane to CO. An inert gas is injected automatically
as needed into each feed line to make up the difference between the flow rate of the feed material and the desired total pressure in that line. The flow rates for these feeds are shown in Figures 4-9 and 4-10.
Gas Storage Units
Oxygen will be supplied from an off-site vendor. The on-site storage area will have standard oxygen safety systems. The TPC plans to use pipeline natural gas as the methane source, with no on-site storage.
Decontamination Solution
The TPC submissions do not specify the required quantity of decontamination solution, but it should be less than the amount required in the baseline system because CEP does not require decontamination of ton
|
Component |
Stream 1 Hot Water |
Stream 2 Liquid HD |
Stream 3 TCs/Resid. |
Stream 4 Dunnage |
Stream 5 Oxygen |
Stream 6 Methane |
Stream 7 Metal Prod. |
Stream 8 Water |
Stream 9 Slurry |
Stream 10 Offgas |
|
C |
|
990,945 |
78,109 |
18,515 |
|
5,412 |
|
|
|
|
|
H |
|
165,699 |
7,888 |
1,904 |
|
1,804 |
|
|
|
|
|
Cl |
|
1,426,311 |
45,826 |
|
|
|
|
|
|
|
|
S |
|
666,045 |
50,426 |
|
|
|
50,426 |
|
|
|
|
O2 |
|
|
|
6,031 |
129,893 |
|
|
|
|
|
|
P |
|
|
1,216 |
|
|
|
1,216 |
|
|
|
|
Fe |
|
|
3,012,355 |
|
|
|
3,003,331 |
|
|
|
|
Mn |
|
|
15,200 |
|
|
|
15,200 |
|
|
|
|
FeCl2 |
|
|
|
|
|
|
|
|
20,481 |
|
|
H2O |
158,500 |
158,500 |
|
|
|
|
|
96,945 |
96,965 |
|
|
CO |
|
|
|
|
|
|
|
|
|
237,960 |
|
H2 |
|
|
|
|
|
|
|
|
|
10,597 |
|
HCl |
|
|
|
|
|
|
|
|
35,348 |
|
|
Total (lb) |
158,500 |
3,407,500 |
3,211,000 |
26,450 |
129,893 |
7,216 |
3,070,173 |
96,945 |
152,298 |
248,557 |
Figure 4-9a
CPU block diagram and material balances for HD treatment. Adapted from M4 Environmental L.P., 1996b.
containers. Decontamination solution would be used primarily to decontaminate the punch-and-drain equipment and, work area. Standard storage and mixing facilities for the decontamination solution will be used.
To avoid introducing sodium into the CPU-2 bath, the TPC prefers, according to its submissions, calcium-based decontamination solutions instead of the Army standard sodium-based solutions. Although there is experience in the use of calcium-based decontamination solutions, their effectiveness and acceptability to the Army have not been established.
Pretreatment Requirements
Cleaning the ton containers is not necessary in this process. If the Army requires precleaning of the ton containers for temporary storage, the high pressure water-jet cleaning system will require a small amount of water (on the order of a few gallons per ton container) and iron abrasives. The drainage from the cleaning system will be pumped to temporary storage and ultimately processed in CPU-2.
Residual Streams
This section covers the residual streams coming out of the chemical demilitarization facility. It does not describe internal process product streams.
Mass Balance
The mass balances provided by the TPC for residuals from each agent are shown in Figures 4-5, 4-9, and 4-10
|
Component |
Stream 1 Liquid HD |
Stream 2 Slurry |
Stream 3 CPU-2 Gas |
Stream 4 O2 |
Stream 5 Methane |
Stream 6 Solvent |
Stream 7 D.I. Water |
Stream 8 Aq. HCl |
Stream 9 Sulfur |
Stream 10 Synthesis Gas |
Stream 11 MeOH |
|
C |
990,945 |
|
|
|
76,151 |
393,349 |
|
|
|
|
|
|
H |
165,699 |
|
|
|
25,562 |
71,054 |
|
|
|
|
|
|
Cl |
1,426,311 |
|
|
|
|
|
|
|
|
|
|
|
S |
666,045 |
|
|
|
|
|
|
|
666,357 |
|
|
|
O2 |
|
|
|
1,718,615 |
|
|
|
|
|
|
|
|
Fe |
|||||||||||
|
FeCl2 |
|
20,481 |
|||||||||
|
|
|||||||||||
|
H2O |
158,500 |
96,945 |
|
|
|
|
3,217,224 |
3,217,224 |
|
|
|
|
CO |
|
|
237,960 |
|
|
|
|
|
|
3,643,882 |
|
|
H2 |
|
|
10,597 |
|
|
|
|
|
|
218,739 |
|
|
HCl |
|
35,348 |
|
|
|
|
|
1,513,992 |
|
|
|
|
H2S |
|
|
|
|
|
|
|
|
|
|
|
|
MeOH |
|
|
|
|
|
|
|
|
|
|
3,793,514 |
|
Total |
3,407,500 |
152,744 |
248,557 |
1,718,615 |
101,713 |
464,403 |
3,217,224 |
4,731,216 |
666,357 |
3,862,621 |
3,793,514 |
Figure 4-9b
CPU block diagram and material balances for HD treatment. Adapted from M4 Environmental L.P., 1996b.
and Table 4-3. There are no residuals from Area 100, the feed handling and punch-and-drain systems. All feed materials are eventually sent to Area 200, the CPU area, for processing. The residuals from Area 200 are the metal and slag phases that are tapped from the CPUs. The offgas from CPU-2 is fed to CPU-1. The offgas from CPU-1 goes to Area 300 for processing in the gas handling train.
Solids
HD and VX processing will produce about 1,360 and 1,590 metric tons per year, respectively, of metallis products. The TPC proposes to sell this material.
The only solid-waste residual will be approximately 62 metric tons per year of ceramic slag from processing decontamination solutions and dunnage. The ceramic slag will be placed in drums and shipped to a permitted hazardous waste landfill. The TPC reports having had initial discussions with several commercial disposal firms regarding disposal of this material, as well as pursuing possibilities for marketing it. If sodium-based decontamination solution is used at the facility, the sodium will appear in the ceramic slag and alter its properties, including its solubility and strength.
H2S in the offgas from processing HD will be converted to elemental sulfur and offered to the market.
|
Component |
Stream 1 Hot Water |
Stream 2 Liquid VX |
Stream 3 TCs |
Stream 4 Dunnage |
Stream 5 O2 |
Stream 6 Methane |
Stream 7 Metal Prod. |
Stream 8 Offgas |
Stream 9 Slurry |
|
C |
|
1,309,889 |
28,800 |
18,515 |
|
60,166 |
|
|
|
|
H |
|
260,154 |
|
1,904 |
|
20,055 |
|
|
|
|
O |
|
332,568 |
|
6,031 |
1,443,991 |
|
|
|
|
|
P |
|
316,476 |
1,152 |
|
|
|
317,628 |
|
|
|
S |
|
319,158 |
1,440 |
|
|
|
320,598 |
|
|
|
N |
|
142,146 |
|
|
|
|
|
142,146 |
|
|
Fe |
|
1,341 |
2,834,208 |
|
|
|
2,835,549 |
|
|
|
Mn |
|
|
14,400 |
|
|
|
14,400 |
|
|
|
Ni,Cu |
|
268 |
|
|
|
|
268 |
|
|
|
H2O |
118,800 |
118,800 |
|
|
|
|
|
|
144,000 |
|
CO |
|
|
|
|
|
|
|
3,305,466 |
|
|
H2 |
|
|
|
|
|
|
|
295,407 |
|
|
Particulates |
|
|
|
|
|
|
|
|
144,000 |
|
Total (lb) |
118,800 |
2,800,800 |
2,880,000 |
26,450 |
1,443,991 |
80,221 |
3,488,443 |
3,743,019 |
288,000 |
Figure 4-10a
CPU block diagram and material balances for VX treatment. Adapted from M4 Environmental L.P., 1996b.
Liquids
There are no continuous aqueous residual streams that will require disposal. Internal aqueous process streams, including spent decontamination solution, scrubbing liquors from the relief-system vent-gas, and spent liquors from the HCl and sulfur recovery processes, can be fed to the CPUs. The HCl from HD processing will be recovered as an aqueous solution that can be offered to the market.
Gases
The offgas from processing HD will include H2, CO, HCl, H2S, and trace components. The TPC anticipates that the offgas from processing VX will contain the same gases, except that HCl and H2S will be present in trace quantities, at most. The panel expects that there will probably also be trace amounts of HCN. The HCl and H2S from HD will be recovered as aqueous HCl solution and elemental sulfur, respectively.
The gases remaining after scrubbing, referred to by the TPC as synthesis gas or syngas (see Figure 4-9), will be burned along with natural gas in a gas turbine generator to supply in-plant electricity needs, subject to permit approval. The TPC projects that the effluent gas released to the atmosphere from the gas turbine will have the composition shown in Table 4-4. If combustion of the synthesis gas is not allowed, the TPC has stated that it will provide a methanol recovery module, which will recover hydrogen, carbon, and oxygen as liquid
|
Component |
Stream 1 CPU-1 OG |
Stream 2 CPU-2 Slurry |
Stream 3 O2 |
Stream 4 Methane |
Stream 5 Synthesis Gas |
Stream 6 N2 Vent |
Stream 7 MeOH |
|
C |
|
|
|
96,003 |
|
|
|
|
H |
|
|
|
32,226 |
|
|
|
|
O |
|
|
norm. 0 |
|
|
|
|
|
CO |
3,873,868 |
|
|
|
4,097,759 |
|
|
|
H2 |
339,434 |
|
|
|
387,772 |
|
|
|
N2 |
142,146 |
|
|
|
142,146 |
142,146 |
|
|
H2O |
|
144,000 |
|
|
|
|
|
|
Particulates |
|
144,000 |
|
|
|
|
|
|
MeOH |
|
|
|
|
|
|
4,161,641 |
|
Total |
4,355,448 |
288,000 |
norm. 0 |
128,229 |
4,627,677 |
142,146 |
4,161,641 |
Figure 4-10b
CPU block diagram and material balances for VX treatment. Adapted from M4 Environmental L.P., 1996b.
methanol. The panel has not analyzed the fate of trace gaseous components if methanol recovery is substituted for synthesis gas combustion.
There are also minor air emissions from the chelate regeneration equipment in the sulfur recovery system. This vent stream passes through an activated carbon filter before being released to the atmosphere. During startup of the CPUs, intermittent combustion gases are produced by the headspace heater, which burns natural gas.
Nonprocess Wastes
Dunnage from daily operations will consist of PPE (personal protective equipment) including demilitarization protective ensembles, undergarments, suits, gloves, and boots that are no longer usable; rags used in maintenance and decontamination operations; and laboratory waste. The dunnage will be compacted, packaged into small metal containers, and fed to CPU-2 for destruction. The materials in the dunnage contribute to the ceramic slag and the offgas components, described above.
Off-Site Shipping and Processing Options
The CEP technology as submitted by the TPC to the Army is a "total solution" approach to chemical demilitarization. It includes methods for processing ton containers, decontamination solutions, and dunnage, as well as for destructive processing of chemical agents. Most
Table 4-3 CEP Heat and Material Balances for VX Gas Handling
|
Stream Number |
229 |
301 |
302 |
310 |
312 |
313 |
352 |
|
Description |
Reactor offgas from CPU-1 |
Quench offgas |
Process gas feed to JV-301 |
Quench water to reactor offgas |
Particulate slurry |
Make-up water to DS-305 |
Synethesis gas to gas turbine |
|
Phase |
Vapor |
Vapor |
Vapor |
Liquid |
Slurry |
Liquid |
Vapor |
|
Mass flow (lb./h) |
|
|
|
|
|
|
|
|
H2 |
53.9 |
53.9 |
53.9 |
0.0 |
0.0 |
0.0 |
53.9 |
|
CO |
569.1 |
569.1 |
569.1 |
0.0 |
0.0 |
0.0 |
569.1 |
|
H2S |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
|
N2 |
19.7 |
19.7 |
19.7 |
0.0 |
0.0 |
0.0 |
19.7 |
|
Water |
0.0 |
353.3 |
768.1 |
768.1 |
0.0 |
19.0 |
19.0 |
|
CH3OH |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
0.0 |
|
Particulates |
20 |
20 |
0.0 |
0.0 |
20 |
0.0 |
0.0 |
|
Solvent |
0.0 |
0.0 |
0.0 |
0.0 |
20 |
0.0 |
0.0 |
|
Total mass flow (lb./h) |
662.7 |
1,016.1 |
1,410.8 |
768.1 |
40 |
19.0 |
661.7 |
|
Mole flow (lb. mole/h) |
47.7 |
67.4 |
90.4 |
42.6 |
|
1.1 |
48.8 |
|
Volume flow (ft.3/h) |
54,520 |
40,378 |
24,723 |
12.0 |
12 |
0.30 |
6,557.0 |
|
Temperature (°F) |
2,700 |
1,199 |
300 |
100 |
100 |
1,000 |
100 |
|
Pressure (psia) |
29.7 |
29.7 |
29.7 |
46.7 |
20.7 |
35 |
44.7 |
|
Vapor fraction |
1 |
1 |
1 |
0 |
0 |
0.0 |
1 |
|
Enthalpy (Btu x 106/h) |
-0.028 |
-2.431 |
-5.253 |
-5.225 |
|
-0.129 |
-1.068 |
|
Source: Adapted from M4 Environmental, L.P., 1996b. |
|||||||
TABLE 4-4 Expected Composition of CEP Gas Streams prior to and after Combustion in a Gas Turbine Generator
|
|
Gas Stream to Generator |
|
|
|
Constituent |
HD Offgas |
VX Offgas |
Generator Exhaust Gas |
|
CO |
12.4% |
|
12.4% |
|
19.5 ppmv |
|
|
|
|
H2 |
9.7% |
|
15.3% |
|
|
none |
|
|
|
HCl |
<0.5 ppmv |
|
none |
|
<0.5 ppmv |
|
|
|
|
H2S |
<0.03 ppmv |
|
none |
|
<0.03 ppmv |
|
|
|
|
SOx |
none detectableb |
none detectableb |
0.039 ppmvc |
|
N2a |
1.17% |
1.53% |
none |
|
NOx |
none detectableb |
none detectable |
<130 ppmv |
|
HCN |
none detectableb |
none detectableb |
not stated |
|
Trace Organics |
none detectable |
none detectable |
9.7 ppmve |
|
a TPC states that most of the nitrogen shown is typical of the natural gas combusted with the synthesis gas. TPC states that no nitrogen is introduced in the HD process and nitrogen from VX processing is approximately 0.36% prior to natural gas injection. b TPC used the following lower detection limits: SOx = 1 ppm; NO and NO2 = 3 ppm; HCN = 0.01 ppm; trace organics = 0.1 ng 2, 3, 7, 8 TEQ/Nm3. c TPC based this value on typical sulfur concentration in natural gas. d Expressed as NOx corrected to 15 percent oxygen. TPC stated that, if required, this amount could be reduced to 42 ppmv by water injection. e TPC based value on unburned hydrocarbons from the natural gas cofuel to the turbine generator. Source: M4 Environmental L.P., 1996b. |
|||
of these feed materials are converted to useful products, including iron-based alloy, synthesis gas for power generation, aqueous HCl, and elemental sulfur.
Instrumentation and Control
The CEP design includes a distributed control system (DCS) for overall monitoring and control of material processing and related support systems. The control architecture for the CEP chemical demilitarization facility is an integrated DCS that provides executive control of the monitoring and process intervention required for safe and efficient operation in processing chemical agents. Two fully operational control systems will be installed. One actively controls and monitors the process; the second remains on active standby, monitoring the process and serving as a redundant system that can take over control operations if the primary system malfunctions or some other internal problem arises. The facility includes a local area network with an independent bus for control and communications.
Process instrumentation and controls are located throughout the central building and support areas for monitoring and controlling parameters such as tank and bath levels, flow rates, pressure, pH, temperature, motor current, weight, volume, and valve position. The sensor instrumentation for monitoring process parameters includes detectors, signal conditioning, transmitters, and other devices as required. Continuous, real-time control is provided for critical processes. The DCS interfaces with the process monitoring and control instrumentation through input/output devices, which are located throughout the facility to reduce the amount of cabling, the number of connections, and the number of cell penetrations. Ground-bus connections isolate the grounds for the instrumentation and control circuits from power grounds. Additional analytical instrumentation is used to monitor for agent releases in the central building.
Most of the systems and equipment to be controlled are located in various work cells of the central building. These in-cell systems have hermetic feed throughs for wall-penetration assemblies that provide interfaces for equipment, components, or input/output devices.
Monitoring and control systems that perform safety functions are hard-wired and sufficiently redundant to meet the criteria for avoiding single-point failures. They are powered by an uninterruptible power supply consisting of batteries, with chargers and inverters to allow use of power from backup generators. The design basis for these systems includes protection against natural events (e.g., earthquakes or severe storms) and worst-case environmental conditions. Systems are designed with fail-safe circuits to meet these requirements. Each redundant system required to perform safety functions is physically and electrically separated from its counterpart and from nonsafety-related circuits and components.
Part of the TPC's stated control strategy is to perform an analysis of the entire system during the detailed design phase to define the critical control systems that will be hard-wired. The hard-wired systems will include all safety systems and all systems necessary to ensure the safety of workers and the public and to protect the environment.
Operations will be directed and monitored from a master control room adjacent to the central building. The control room is isolated from areas that could become contaminated with agent. Video surveillance provides visual monitoring of the entire process, end to end.
The process monitoring and controlling requirements for the feeds to the CPU reactors include gas mass-flow controllers for the oxygen, inert gas, and natural gas streams and liquid flow controllers for agent and for solutions used to clean ton containers. Agent assays of the ton container contents will be performed by taking a grab sample from each container and analyzing it via GC/MS (gas chromatography followed by mass spectrometry) with a lower detection limit less than 0.1 µg/ml (100 ppb).
Key parameters for controlling the CPUs are bath temperature, bath composition, bath level, and containment monitoring. Monitoring and control for each of these are described below.
Bath Temperature Control
By varying the power to the induction coil, the bath temperature control system maintains the molten metal bath at a stable operating temperature (±28°C) at least 110°C above the liquidus temperature of the bath (temperature at which the bath metal is entirely molten). Based on the preliminary design submitted, the operating temperature of the CPU-1 bath for processing HD (nickel bath) is likely to be about 1425°C. The iron baths will operate at about 1500°C to 1650°C.
Two temperature-sensing systems are used for monitoring: an infrared lightpipe and thermocouples embedded in the CPU refractory material. The primary temperature sensor is the infrared lightpipe, which provides a continuous, non-invasive method for sensing bath temperature. The lightpipe, which transmits infrared radiation directly from the bath to a dual-wavelength pyrometer, provides fast response and, precise measurements, and requires minimum calibration.
The redundant system for controlling the bath temperature uses thermocouples embedded in the refractory wall combined with a proprietary, on line control model that predicts the metal bath temperature during operation. The method is nonintrusive and robust for CEP processing conditions.
In addition to control of bath temperature, headspace temperature is kept high enough to avoid solidification of molten metal on surfaces.
Bath Composition Control
Control of the bath composition is necessary to obtain the required agent DRE, to produce offgas with the desired composition, and to maintain the structural integrity of the containment system. The carbon concentration in the bath is controlled by varying the oxygen flow rate and monitoring the composition of the offgas, specifically the ratio of CO to CO2. The model used to infer carbon concentration from the composition of the offgas has been validated with actual measurements of bath carbon.
A contingency method of modeling the bath composition is based on the material balance for feed and product streams to and from the bath. The TPC has routinely estimated bath carbon concentration in its large Demo Unit CPU by using a feed-forward model and an offgas composition model. The basis of each model is a general steady-state carbon balance on the reactor. In the feed-forward model, composition is estimated using partitioning and thermodynamic models. Analysis data on offgas composition provide estimates for the second model. The results from these models are combined with feedback control based on the CO/CO2 ratio to ensure an appropriate bath carbon concentration.
For VX processing, sulfur and phosphorus are controlled by adding iron from ton containers and tapping
Fe-S-P-C alloy from the bath, but monitoring procedures were not discussed.
Monitoring Bath Level
There is a bath-level monitoring system for each CPU. Each CPU is fitted with a side-mounted lightpipe that senses the bath temperature directly and provides an indirect indication of the bath height when compared with the bath temperature provided by the thermocouples in the refractory lining. In addition, a microwave level switch is used as a sensing system for maximum bath level.
Monitoring Containment
The CPU design provides for two linings of refractory to serve as the primary and secondary containment for the molten metal. The inner lining, called the working lining, is the primary containment. The outer lining is designed primarily as an insulating layer to lower the temperature at the outer steel vessel, but it also serves as a backup containment, capable of holding the bath long enough for the molten metal to be drained if the working lining is breached. In addition, portions of the outer steel vessel are water cooled, which cools the adjacent refractory enough to freeze a layer of slag on the surface of the working lining, thereby prolonging its life.
The two systems for monitoring the integrity of the primary containment are embedded thermocouples and grid assemblies. These redundant monitoring systems give the operators an indication of normal refractory wear and warn of molten metal encroachment to the secondary containment. During normal operations, the primary monitoring system is the thermocouples embedded in the refractory. The temperature differences among thermocouples indirectly measure refractory wear from the temperature gradient across the working lining, which is directly proportional to thickness of the refractory.
The secondary level of monitoring the refractory containment consists of detection grids incorporated in the primary lining. Contact with molten metal opens a grid and provides a reliable indication of either localized or uniform deterioration of the working lining. Complete coverage of the refractory lining with grids, together with the embedded thermocouples, provides continuous monitoring of the refractory, thereby allowing sufficient time for a normal system shutdown in the event of excessive deterioration.
These containment monitoring systems have performed reliably in the units at the TPC's Fall River facility (see Table 4-2).
Monitoring Residual Streams
Solids
Metal ingots and ceramic slag can be analyzed by the EPA's TCLP test to verify compliance. Verifying that the metal ingots and ceramic slag do not contain agent within their internal matrices is difficult because any technique used to extract samples for analysis is also likely to destroy agent. However, this internal verification is probably not necessary because the conditions under which the ceramics and metal ingots are produced exceed the Army's definition of a 5X material (which is considered agent free).
Gases
The TPC plans to install a continuous emission monitoring system to monitor gas effluent streams for O2, CO2, CO, NOx, H2, HCl, and H2S. Similar monitoring systems have been proven and used extensively at the operating demilitarization facilities. The TPC states that it will review and incorporate lessons learned from these sites prior to specifying the final type of detector to be used for the emission monitoring system.
Provision for retaining synthesis gas for analysis prior to release for combustion has been added to the original design, as described above in Conclusions on the Underlying Science.
The depot area air monitoring system (DAAMS) and "mini" continuous air monitoring system (MINICAMS) used by the Army are sufficient for monitoring for agent inside the CEP facility and at the site perimeter. Gas chromatographs, mass spectrometers, and the continuous emission monitoring system are capable of analyzing the feed, internal process, and residual streams to meet regulatory and operational requirements.
Monitoring Synthesis Gas prior to Combustion
The TPC plans to choose among one of three analysis systems during the next stage of design. One is the automatic continuous air monitoring system (ACAMS),
which is the standard Army monitoring system to existing agent destruction facilities. The second is the MINICAMS, which is also used to monitor for agent at existing Army facilities. The third system is the TAGA 6000E (trace atmospheric gas analyzer), which has been tested at the Army Chemical Agent Munitions Disposal System. The ACAMS and MINICAMS use gas chromatography with flame photometric detectors and have response times of 3 to 5 minutes for the agent detection levels required. The TAGA 6000E has a response time of 15 seconds. The TPC plans to install several sensors for each of the three retention tanks, with a "voting logic" system to reduce the number of false positives. If the system logic determines that agent is present in a tank, the tank contents would be recycled to CPU-1 for reprocessing. The TPC's description makes no reference to testing the retained gas for constituents other than agent.
A preliminary analysis by the panel suggests that this three-tank design may not be adequate; at least one more tank may be required. At 20 atmospheres, gauge, each 4-m3 tank holds 60 kg of synthesis gas. If a tank is found to be contaminated (call it Tank A), the contents must be fed back through CPU-1, along with cofeeds of oxygen and methane. The minimum mass to be "reprocessed" is thereby increased to about 64 kg, all of which reappears as offgas from CPU-1, assuming the bath is saturated with C, O, and H. The gas in the next tank to be filled (call it Tank B) must be presumed to be contaminated until that tank is filled and testing shows it is clean. If Tank B is contaminated, it cannot be used to hold the surge from Tank A. This leaves only the third tank (Tank C) to hold the 64 kg of gas from reprocessing Tank A. Tank C will be full before Tanks A and B are emptied. The fourth tank must be empty and ready to handle the overflow from reprocessing Tank A. When the fourth tank is full, Tank A can be refilled to handle overflow from Tank B, if it is contaminated.
Air in the Containment Building
In the submitted design, air inside the secondary containment building will be monitored using a variety of instruments to provide both real-time and time-weighted-average agent monitoring. A detailed agent monitoring plan for a CEP demilitarization facility would be developed initially as part of the detailed design process before pilot-testing. The plan would be refined as the facility is constructed and commissioned. The general strategy for safety and environmental agent monitoring is much the same as the strategy used at the Johnston Atoll Chemical Agent Disposal System (JACADS) and Tooele Chemical Agent Disposal Facility (TOCDF), although the TPC states that less of the plant would require monitoring by virtue of the inherent safety features of CEP. In the central building, each enclosed room would be monitored by a near-real-time instrument and a DAAMS. The detection range and alarm level will be based on the hazard category (protective clothing level) for each room.
Near real-time monitoring could be provided by either the ACAMS or the MINICAMS, These instruments would be used to monitor for agent throughout the demilitarization facility at the following statutory levels: MPL (maximum permissible limit, a very high level), HLE (high level exposure), TWA (time-weighted-average, a low-level), and IDLH (immediately dangerous to life and health). According to the TPC, the MINICAMS provides additional flexibility in software functionality and the future availability of the ACAMS is uncertain, so the TPC currently considers the MINICAMS as the monitor of choice for near real-time monitoring. DAAMS, which is used at operating Army facilities, will be used to monitor the perimeter for very low-levels of agent and, in the event of a MINICAMS or ACAMS alarm, to obtain longer-term samples to confirm whether agent was present.
Stability, Reliability, and Robustness
Stability
Stability of CEP is discussed under the topics of out-of-control operations, stored energy, and catastrophic failures.
Out-of-Control Operation. The large mass of the metal bath provides commensurately large thermal inertia, which prevents a significant temperature excursion in the event of perturbations in the feed rate of agent or cofeeds. The bath mass provides a margin of safety for bath composition and feed rate and allows the CPU to operate over a relatively wide range of conditions.
Stored Energy. According to the TPC, the total stored energy of each iron bath is approximately 4 x 106 kJ. The nickel bath used for HD processing has two-thirds the mass of an iron bath and about 2.6 x 106 kJ of stored energy.
Catastrophic Failures. There are no identified process mechanisms, such as uncontrolled reactions, under normal operating conditions that could lead to a catastrophic failure of the facility. However, catastrophic accidents can always occur if the equipment fails—a break in a tuyere or tapping nozzle, for example—or if there is operator error, such as inserting an undrained ton container into the CPU-2 melting unit. In response to questions from the panel, the TPC has added several levels of operational controls to the design to prevent an accidental insertion of an undrained container.
An extended failure of electrical power would require a cold shutdown of the CPUs, with related problems whose severity would depend on the reliability of emergency standby power to open taps and drain the molten baths before they solidified (see Startup and Shutdown, above).
Reliability
Performance Record
The CPUs closely resemble the induction furnaces used in melting metal, as well as the TPC's several demonstration CPUs. Materials of construction were selected in light of process conditions and process-fluid characteristics. Allowances for stress and wear are incorporated to ensure adequate life and performance throughout the operational period.
The basic CPU design has been tested under severe conditions. Most of the front-end equipment is either the same as equipment in the Army baseline incineration system or closely resembles that equipment and is likely to be as reliable.
The offgas recovery units are based on proven commercial design but require some special features for processing the offgas from chemical agent destruction.
Backup Systems
In the event of an equipment failure in the oxygen supply, methane cooling gas supply, or offgas treatment, the system can stop the agent feed almost instantaneously. The CPUs can be held at hot standby condition indefinitely.
If the site has a single line of access to the electric power grid, an uninterruptible battery power system with a response time of a few milliseconds can maintain critical safety and control services until backup power can be brought on line. Essential services for a no-feed, hot standby condition can be provided by the gas-powered turbine generator used to recover energy from the synthesis gas. If a turbine generator is not installed, a diesel generator capable of a 10-minute response from cold start can be used to provide power for standby services.
Robustness
The CPUs can operate over a range of operating conditions. The thermal inertia of the bath is large enough that, with a loss of power, the bath takes approximately 2 hours to freeze. Responses to upsets and control mechanisms have already been described.
Materials of Construction
Systems and Materials
The block flow diagram for the facility in Figure 4-4 shows the layout and interconnects for process operations. The conceptual design for the facility was performed by competent engineering firms that are participants in the team that prepared the submissions. These firms have experience in designing chemical processing units and nuclear power plants, many of which have been in operation for years and have documented safety records. System design and material selection appear to be based upon sound engineering practice.
An inquiry from the panel led to one change in material selection from the original submission. The initial design specified tungsten for the slide rails inside the premelting chamber of CPU-2 to support the ton container during melting. The TPC changed the material to a refractory oxide after a question from the panel about the substantial solubility of tungsten in iron at the melting point of iron.
Materials Specifications
According to the TPC, the design follows the published specifications of the American Society of Mechanical Engineers (ASME) for piping materials, valve bodies and trims, shell-side and tube-side materials for
heat exchangers, and impeller materials for pumps. The corrosion allowances and specifications for piping and components, including special materials requirements such as stress relief, also use the ASME recommendations for specific components.
Welding Specifications
Most of the piping, vessels, and other equipment in a CEP facility contain welds. Where equipment is welded to piping, the equipment is generally flanged and bolted to the welded piping spools. Structural steel used to support the piping and equipment is also typically welded. According to the design for an agent destruction facility, agent transfer lines from the storage tanks to the CPUs are double-walled piping; the annular space between the walls is monitored for low-level agent vapor, as an early indicator of a leak in the inner wall. Special stress relief requirements, welding processes, filler metals, and gas shielding conform to standard welding specifications. These extensive specifications are normally tailored to the requirements of a project during the detailed engineering phase. The design states that welding procedures will follow the current ASME codes and applicable Military Standard, MIL-STD-1261C(MR).
Stress Relief
In the design generally, stress relief, where required, is based on details of the material, thickness, or service. Materials that often require stress relief regardless of thickness are martensitic steels containing 1 to 12 percent chromium. Carbon steel often requires stress relief above a certain thickness, per the applicable codes. For instance, ASME Section VIII for vessels requires stress relief when carbon steel is thicker than 1.5 inches (3.8 cm), and ASME Standard B31.3 for piping requires stress relief when carbon steel is thicker than 0.75 inches (1.9 cm).
Stress relief for service generally applies when the material would be susceptible to stress corrosion cracking, such as when carbon steel is in contact with caustic or amine solutions or when stainless steel is in contact with chloride or sulfide solutions. Operating temperature is often an important variable in determining if stress corrosion cracking may occur. For the solutions listed above, the temperature range of concern is from 38°C to 66°C. In the TPC's design, these solutions listed above are either at room temperature or an appropriate lining is specified.
Weld Inspection
According to the submitted design, the minimum amount of weld inspection will be to an appropriate industry code, typically ASME Section VIII for vessels, ASME/ANSI B31.3 for piping, and AWS D1.1 for structural steel. (ANSI refers to codes approved by the American National Standards Institute; AWS refers to codes approved by the American Welding Society.) This degree of inspection requires spot radiography and hydrotesting for the majority of welds of equipment and piping. For the double-walled agent transfer line, large vapor lines, and refractory-lined piping and equipment, hydrotesting will not be practical, so 100 percent radiographic testing will be performed. The TPC states that a reputable third party will conduct the weld inspections and evaluate results. The TPC will furnish welding specifications with the detailed design to provide information on inspection methods and criteria. Weld inspections will be conducted in accordance with paragraphs 5.1.4 through 5.1.4.4 (magnetic particle inspection, radiographic inspection, dye penetrant inspection, and ultrasonic inspection) of MIL-STD-1261C (MR). A report will be issued in accordance with Data Item Description DI-THJM-81194.
Environmental Chemistry and Conditions
Nominal Internal Environmental Conditions
The CEP processing conditions described here are based on the submitted design, which is preliminary and subject to revision during further design and development. For processing HD, the nominal chemical environment in CPU-2, where ton containers and dunnage are processed, is a molten iron phase containing a controlled concentration of carbon and a gas-phase consisting of H2, CO, H2S, and HCl. Table 4-5 gives the nominal composition for elements other than carbon. The nominal composition of the metal phase in CPU-1 for HD processing is nickel containing about 2 percent carbon. Temperatures in both CPUs are in the range of 1425°C to 1650°C, at an absolute pressure of about 2 atmospheres in CPU-1 and I atmosphere in CPU-2.
TABLE 4-5 Nominal Composition of CPU-2 Metal Phase (weight percent)
|
Element |
HD |
VX |
|
Sulfur |
1.64 |
9.11 |
|
Phosphorus |
0.04 |
9.19 |
|
Iron |
97.82 |
81.28 |
|
Manganese |
0.50 |
0.41 |
|
Nickel, copper |
0.008 |
0.008 |
For processing VX, the bath in CPU-1 is iron with carbon controlled in the range of 1 to 2 percent. The nominal composition of the metal phase in CPU-2 has higher concentrations of sulfur and phosphorus than in the CPU-2 bath for HD (Table 4-5).
For processing either agent, the chemical and physical environment of the quench, absorber, and compressor between the two CPUs is the gas-phase from CPU-2. This gas consists mainly of CO, H2, and H2S. In HD processing, some HCl will be present from residuals in the ton containers and from spent process solutions. Temperatures in this area range from about 1500°C exiting CPU-2 to 38°C at the suction of the compressor; absolute pressures range from 1 atmosphere as the gas leaves CPU-2 to about 10 atmospheres at the discharge of the compressor. The temperatures for quenching and cleaning CPU-2 offgas range from 260°C for the offgas at the inlet to the absorber to 38°C after the cooler and about 66°C in the bottom of the absorber.
The gas handling train operates at low pressure, about 1 atmosphere, gauge. For HD processing, the offgas from CPU-1 will be scrubbed in the HCl recovery section to absorb HCl gas in water and recover it as HCl solution. H2S in the offgas is converted to elemental sulfur. For VX processing, the HCl and sulfur recovery systems are not required because VX does not contain chlorine, and the sulfur is retained in the iron bath of CPU-2. The offgas is scrubbed with water, compressed, stored for analysis, and sent to the gas utilization unit (e.g., gas turbine or methanol recovery). Typical flow rates in the gas handling train during HD destruction are shown in Table 4-6.
Nominal External Environments
Design for exterior environments generally depends on whether the equipment is inside or outside a building, whether heat is being transferred, or whether protection of personnel or equipment is required. In the CEP design as submitted, the environment inside the central building will be protected from weather and maintained at a comfortable temperature. Atmospheric contaminants
TABLE 4-6 Flow Rates in the Gas Handling Train for HD Processing
|
Gas Handling Service or Equipment |
Flow Rate |
|
Reactor offgas |
750 acfm (354 l/s) |
|
HCl product |
2.5 gpm (9.5 l/min.) |
|
Quench water to reactor offgas |
1.4 gpm (5.3 l/min.) |
|
Primary HCl recovery column overhead |
250 acfm (118 l/s) |
|
Recycle liquid to primary column |
1.6 gpm (6 l/min.) |
|
Primary column pump-around |
7.3 gpm (28 l/min.) |
|
Makeup water to secondary column |
1.2 gpm (4.5 l/min.) |
|
Offgas to sulfur recovery |
220 acfm (104 l/s) |
are not expected to be a controlling condition for the design at either site because the piping, equipment, and structures are protected from the weather. Equipment and piping will be insulated either for heat conservation or for protection of personnel (maximum surface temperature 66°C) and equipment. Heat conservation requirements, which will be determined during detailed design, will be based on the cost of heat loss or on the need to provide a stable internal temperature to prevent undesirable swings in process controls. Insulation and heat tracing will be used to prevent freezing in areas where the ambient temperature could fall below freezing and the contents of the piping and equipment could freeze.
The TPC has stated that, for the design of the pilot-test facility, the exterior environments for the piping and components—temperature extremes, relative humidity, atmospheric contamination, and leached chemicals—will be approximated by ambient conditions for the nearest city for which data are available. For the final design, the TPC plans to use conditions at the sites. These conditions enter into the specifications and design basis of various items of equipment as well as the structural facilities. For example, the ambient wetand dry-bulb temperatures are used to set the design cooling water temperature and to specify the capacity of the cooling tower. The rainfall, snowfall, and wind velocity are important to the design of all buildings, other outdoor structures, and surface drainage. The seismic zone will be determined during detailed engineering and taken into account in the design of structures.
Ambient air composition is important if the small amounts of certain substances, such as carbon dioxide and ammonia, that may be present in air are significant to the process. For CEP these components have no significant impact on the design as long as they are not present in concentrations harmful to humans. Air is used in CEP for combustion air to the gas turbine generator, startup burners, and the relief-system boiler; for blowing (oxidative regeneration of) the SulFerox solution; and for evaporative-cooling of water in the cooling tower. None of these uses is sensitive to minor impurities.
Crevices, Surface, and Bottom Deposits
The TPC states that its construction practice is to minimize all crevices, deposits, sources of galvanic corrosion and other design features that can increase corrosive conditions. The detailed design will be reviewed for this purpose by materials specialists on the TPC team. Corrosion in crevices can occur in aqueous electrolytic services. In this design, most of these services are being handled with Haveg, impregnated graphite, or plastic-lined carbon steel, which prevents of crevice corrosion. The industry codes and the TPC's standard practice is to use butt welding for all piping instead of socket welding. Galvanic couples will be avoided in electrolytic services except where the area ratios are such that corrosion is expected to be minimal. (For example, alloy valve trim is specified in carbon steel piping but galvanic corrosion is minimized because the surface area of the trim is much smaller than the area of the carbon steel valve-body and piping.) If underdeposit corrosion is a risk, either larger corrosion allowances will be specified on the bottom head or boot or upgraded alloys, coating, or lining will be specified. The TPC plans to assess the risk and take adequate design precautions based on past experiences with similar services.
Heat Transfer Surfaces, Heat Fluxes, and Crevice Geometries at Tube Supports
Reactor Vessel Shell. Heat flux in the CEP design is limited to that which will produce an external metal temperature of approximately 150°C. This heat flux is in the range of 200 to 500 Btu/h/ft2 (2,300 to 5,700 kJ/h/m2) of external surface.
Reactor Containment. The entire reactor is lined with several overlapping courses of refractory brick. Where the bricks meet, some molten metal, slag, or gas can penetrate between them, but this penetration is stopped by the next layer. Molten material freezes as the temperature drops through the refractory, sealing the interstices from further penetration.
Reactor Internals. The bath refractory is surrounded by an induction coil that heats the bath metal. The coil is internally water cooled. This technology is in widespread use in the steel industry.
Reactor Offgas Piping. Hot offgas in the gas handling train is transferred in a jacketed pipe, which is designed to be cooled with water to maintain the pipe temperature within the maximum temperature limit for carbon steel. Insulation is provided to protect personnel.
Heat Flux in Crevice Geometries. Crevices are particularly prone to corrosion when the heat flux in the vicinity of the crevices creates an enduring temperature differential at the crevice surfaces. For example, tube-to-tube-sheet joints in heat exchangers are prone to corrosion, particularly the crevice in the back of the tube-sheet. In most designs, the tubes are not rolled to the full width of the tube-sheet, which results in this crevice. Corrosion at this crevice is a concern especially with stainless steel tubes. Because there are no heaters or fired furnaces in the present design, no problems of this type are anticipated and no special requirements have been specified for tube rolling. In HCl environments where corrosion would be expected to be severe, the design specifies graphite block exchangers that do not use tube-sheets or other constructions with crevices.
An important crevice that does exist in this system is the joint between the headspace refractory and the refractory containment of the metal bath. The panel learned that, during the early stage of testing the Demo Unit at Fall River, molten metal leaked out through this joint into the annular space that contains the induction coil and burned out the rubber hoses that supply cooling water to the coil. The TPC subsequently developed a proprietary means of sealing this joint that prevents such leakage. The leak did not create a safety hazard but did require a complete shutdown and replacement of the induction furnace.
Startup and Shutdown Procedures
Startup and shutdown procedures have already been described (in Startup and Shutdown in the Process Operations section). Detailed startup procedures, including hot and cold restart specifically for an agent destruction facility, will be developed in the detailed design phase, based on the existing general CEP operating manuals.
Deoxygenating and Heating Rate on Startup
The CPU is deoxygenated as part of normal startup. The procedure for deoxygenating on startup is to pass an inert gas through the CPUs and the downstream piping and equipment until oxygen levels, as determined by analysis, are well below the lower flammable limits of the expected offgas composition. One way to ensure that dead spaces are purged is to open all vents, drains, and bypasses with the inert gas flowing. A variation is to pressurize the system with inert gas and then vent down to atmospheric pressure, with pressurizing and venting repeated several times. Still another variation is to evacuate the system and then break the vacuum with the inert gas, with several repetitions. The TPC plans to decide which procedure to use in this facility during the detailed design phase and will incorporate it into the operating instructions.
The only critical equipment items sensitive to temperature change rate are the refractory lining of the CPUs and the special materials in the HCl recovery system, such as Haveg and graphite. A reasonable rate of temperature change for these items is 110°C/h. Heating rates will be specified in the operating instructions for the CPUs and for other equipment containing ceramic, graphite, or plastics such as Haveg.
Design Life of the Process Equipment
The process equipment is sized to process the entire inventory of HD at Aberdeen in 300 operating days and then to be relocated to Newport to process the entire inventory of VX in 300 operating days. A preoperational period will be required to check out the equipment and controls and to train the operators. Therefore, the panel expects that the required operating life of the process equipment is less than 3 years, which is well within the normal design life of chemical processing equipment (generally 10 to 20 years). The TPC has stated that no attempt will be made to reduce quality and corrosion allowances because of the shorter life expectancy of this facility.
Certain parts of a plant of this kind may require replacement during a normal operational period. Examples are the refractory lining of the CPUs and parts of the HCl recovery section. Refractory life depends on many variables, such as temperatures, changes in temperature, compressive stresses, the corrosive action of slags, actions of different molten metal solutions, and actions of gases. In CEP reactors, changes in temperature are both gradual and controlled, thereby reducing the stress on the refractory linings. Injection forces are mediated by directing the jets from the tuyeres toward the center of the bath. Refractory life is therefore expected to be long enough for the relatively short duration of each agent campaign.
Qualification and Testing of Materials of Construction
The design states that selection of materials of construction will be based on equipment operating conditions and on corrosion and mechanical testing. Materials selection for the punch-and-drain system will be based on the baseline system and lessons learned from existing facilities that process agent. Refractory for the CPU linings will be selected on the basis of testing experience at the TPC's research facilities. The panel believes the refractory can be maintained to accommodate the projected one year agent processing campaigns at each site; replacing the refractory will probably not be necessary.
Materials selection for the gas handling section will be based on the experience of the TPC partners with similar applications, in consultation with experts in the manufacture of chlorinated chemicals, and on corrosion testing of material coupons at the TPCs research facilities. This experience indicates that, with proper maintenance and operating procedures, these materials rarely fail within the first 10 years in service. The expected operating life of this facility of less than 3 years is therefore well within the anticipated usable life of the materials.
Potential Failure Modes for Materials and Components
This section describes only the experience and analytical work related to understanding the failure modes of materials and components in a CEP system. The TPC's general approach to identifying failure modes and hazards in CEP technology and in the design for an agent destruction facility is described below in the Failure and Hazards Analysis section under Operation and Maintenance.
Several systems in the design of the facility use materials and components designed for intrinsically safe modes of operation. First, the molten metal bath quickly dissociates the chemical agent, and this dissociation greatly reduces the chances of contamination downstream. Second, the tuyere line diameter and pressure are designed to limit the agent flow rate to a safe maximum. As a consequence, a valve failure, even in full-open mode, cannot cause a hazardous condition. Third, the reactor has three internal containment (two refractory linings and the steel vessel) and two external containment (the CPU module and the enclosed central building) to reduce the potential for an off-site release.
The TPC states that, in addition to the hazard studies discussed below, the failure modes of the CPUs are understood from the TPC's nearly four years of experience at the Fall River facility. The principal failure modes affect reliability and economical performance but not safety. Careful design and operation are needed to avoid plugging the tuyere (which would prevent agent feed and cause downtime), excessive wear on the refractory (which would reduce on-stream time), loss of coolant to the induction furnace (which would cause downtime), and inadequate control of the process (which could lead to solidifying or skulling of metal or ceramic phase on the walls of the CPU and thus reduce on-stream time).
The failure modes in the gas handling train that are of some concern are loss of coolant in the offgas precooler (which could damage downstream equipment), solidifying of molten carryover from the CPU in the piping to the first quench, and corrosion in the offgas handling equipment.
Monitoring and Inspection
Monitoring methods for the bath temperature, composition, and containment, as incorporated in the CEP design, are described above in Process Instrumentation and Control. Offgas from CPU-1 will be cooled by water quenching. The temperature of the gas quench outlet will be measured and the flow rate of quench water adjusted to maintain the set-point temperature.
Inspection Frequency, Locations, and Observations
The TPC plans to base the frequency of inspection for the monitoring system on its general industry experience with corrosion and the Army's experience with corrosion at other agent destruction facilities. For example, probes for the continuous emission monitoring system last only a few days in high temperature, acidic environments, so they will be monitored on a daily schedule of preventive maintenance. The schedule for other monitoring locations with lower corrosion rates will be weekly or monthly.
The agent monitoring system itself will be used to warn of leaks in agent piping, fittings, valves, and pumps. All equipment used to deliver agent to the CPU
will have double containment walls. The space between the primary and secondary containment walls will be monitored with DAAMS tubes, which will enable maintenance personnel to identify and repair leaking valves, fittings, etc., in the primary containment before the leak allows agent to escape the second containment.
The TPC plans to develop a maintenance control document as part of the detailed design phase. This document will include equipment maintenance schedules; parts lists for routine maintenance; lubrication requirements for each item of equipment; and maintenance procedure summaries specifying the frequency, purpose, references, prerequisites, and listings of all tasks and reviews. The documents will also include an instrument index and spares list, as well as preventive maintenance procedures for instruments, and will serve as a source book for miscellaneous maintenance items required for startup. Software will be used to record maintenance schedules and provide daily reminders and reports.
Operations and Maintenance
Operational Safeguards
All important variables such as temperatures, pressures, flow rates, and levels are measured, recorded, and alarmed throughout the system. Critical controls are provided with automatic alternatives if there is a safety risk or the possibility of damage to equipment. In areas of the plant that handle agent, the interstitial space in double-walled piping and equipment will be continuously monitored for agent, as a means of detecting leaks in the primary containment.
In the gas handling train, the quench water source has assured backup water sources, such as the firewater system. The backup water source ensures that hot offgas from the CPUs is cooled to prevent damage to the gas handling train.
The entire system is designed for operation via remote instrumentation, controls, and video cameras from a control center separate from the central building. The architecture of the DCS uses a centrally integrated executive protocol, which includes an emergency process-shutdown that is hard-wired and completely independent of the control computers and requires no human intervention.
The plant design adheres to approved safety principles for operations involving hazardous chemicals, including the following:
- All operations are designed to keep agent and agent-contaminated fluids inside the ton container, storage tank, or process piping at all times. Agent and agent-contaminated fluids are transferred from the collection point to nearby storage tanks by vacuum pumping techniques.
- The capacity and number of storage tanks for agent and agent-contaminated fluids are set to the minimum needed for the design throughput. Each tank is contained within a separate cell, and all cells are located together in the same area.
- Pumps for pressurizing the agent feed are located as close to the reactor as possible to minimize the length of piping that conveys pressurized agent to the CPU. The pump pressure is as low as possible consistent with maintaining reliable feed conditions under all operating conditions.
- Liquid agent and agent-contaminated fluids are transferred only through double-wall piping. The annulus is purged continuously with inert gas and monitored to detect the presence of agent.
- Pipes and ducts are welded and fully inspected. Bolted and sealed connections are used only where they are essential.
- In the event of a transfer-pump failure, agent or agent-contaminated fluid in the piping drains back into the source tank.
- All agent-involved pipes are sized and routed to allow unimpeded flow and minimize the chance of contamination traps.
- All components involved in pumping, storage, or piping of agent are mounted to be readily accessible for corrective maintenance and area housekeeping by personnel wearing appropriate safety gear.
- The areas around the CPUs are designed for convenient and secure access and are maintained at ambient temperature, to permit immediate emergency response via multiple routes for personnel in full protective clothing.
- The central building is partitioned in such a way that air monitors placed throughout the process areas can detect and verify agent leaks quickly and effectively.
Failure and Hazards Analysis
The TPC has performed several hazard and operability studies of CEP technology for the demonstration and
commercial facilities described above. In addition, the TPC contracted with a third party to perform a hazard analysis specifically to support its submission for the chemical demilitarization program (M4 Environmental L.P., 1996e). This analysis, which used a failure modes and effects analysis (FMEA) approach, identified 1,129 failure events. Of these, 17 unique events for both facility sites were assigned a risk assessment code of 2, indicating that the risk was not acceptable. None of these code 2 risks involved exposure to chemical agent, and only one involved personal injury. The remaining 16 involved only a possible loss of processing capability because of damage to critical components in the gas handling train.
The TPC plans to conduct additional safety and hazards reviews during the design, engineering, and facility commissioning phases of development. The TPC states that, for these reviews, it will use methodologies and techniques developed by E.I. DuPont de Nemours and Company, Imperial Chemical Industries, and the Chemical Process Safety Institute that meet or exceed the requirements specified in the Occupational Safety and Health Administration regulations, Process Safety Management of Highly Hazardous Chemicals (29 CFR 1910.119).
The TPC also plans to implement a comprehensive health and safety program to establish best practices for ensuring safety. These practices include emergency response plans, plans for communicating information on chemical and radiological hazards, ALARA (as low as reasonably achievable) review procedures, safety training requirements, procedures for change management, and standard industrial safeguards. The TPC intends to document all operational procedures and practices, incident investigation reports, and compliance audits.
Maintenance
Routine Maintenance Requirements
For the feed preparation systems, feed systems, and balance of the plant (Areas 100 and 900), most of the routine maintenance after startup involves checking and adjusting for wear and tear of mechanisms and stops and replacing pressure seals and glands to prevent leakage of fluids and gases. Critical elements of the feed preparation equipment such as the punch tools, the probes for extracting liquid agent, and the water-jet cutting nozzles and cleaning heads need frequent replacement because they have high rates of wear.
Because operations at the two sites will be of short duration (about one year each) and the number of process cycles to be completed is fairly low (1,700 ton containers at each site, plus miscellaneous discrete items), the wear on the process equipment should be within acceptable limits.
An important aspect of routine maintenance will be calibration of instruments such as the ACAMS (automatic continuous air monitoring system) or MINICAMS. Because both of these instruments are gas chromatographs, they require a significant level of routine calibration and maintenance. The experience of one of the TPC partners in working with the instrumentation at the Tooele Chemical Disposal Facility gives the TPC team experience in setting up and operating a calibration and maintenance program for these and other agent monitoring instruments.
Maintenance Manuals and Procedures
The TPC provides maintenance manuals and operating procedures for all its operating CEP units. Because the CEP facility for chemical demilitarization is still in the conceptual design phase, no facility-specific manuals or procedures have been developed yet. The TPC plans to develop a project maintenance manual covering preventive maintenance, lubrication, scheduled checks and inspections, cold test plans, and integrated test plans for startup. The manual will be prepared as the detailed design nears completion and will contain detailed procedures, checklists, and valve line-ups.
Documented Record of Performance
The feed preparation systems, feed systems, and most of the balance-of-plant systems (Areas 100 and 900) use equipment that is the same as or similar to equipment used in the Army baseline incineration system. Records of performance probably exist for this equipment, and one can reasonably assume that similar levels of operation and maintenance will apply when the equipment is used in the proposed CEP system.
Downtime Experience
Based on the TPC's experience to date, the TPC has allowed for approximately 60 days of maintenance and
300 days of continuous operation per operating year for each site (Aberdeen and Newport).
Utility Requirements
Table 4-7 summarizes the TPC's stated utility requirements for a CEP agent destruction facility. The numbers in the table represent steady-state processing of agent at the design rate (upper bound) of one ton container (liquid agent to CPU-1, empty container to CPU-2) approximately every 4 hours.
The principal utility requirements are natural gas and electric power. Note that the total electric power load of 1,510 kW shown in the table is a net load and includes a load-reducing contribution of 3,525 kW from cogeneration. Of the 33.35 x 106 Btu/hr (9,767 kW equivalent) of natural gas required at steady-state operation, 30.6 x 106 Btu/hr (8,962 kW equivalent), or 92 percent, is used for cogenerating electric power. The energy contribution to cogeneration from the synthesis gas is estimated at about 2 x 106 Btu/hr (586 kW equivalent).
For electric power, the maximum operating load of about 7,500 kW (not shown in Table 4-7) occurs when starting up the two CPUs together and lasts a maximum of 2 days. During CPU startup, there is also additional demand for natural gas to fuel the headspace heaters.
The water requirement is minor, consisting of makeup for a small offgas scrubber, makeup for a small cooling tower, and use by personnel. The total average requirement is estimated at 10 gallons (38 liters) per minute.
Scale-Up Requirements
The discussion of scale-up requirements for CEP is divided into issues related to scaling up the equipment and issues related to how processes are likely to perform when carried out at a larger scale.
Equipment Scale-Up
Front End and Back End Equipment
The development of all process operations and equipment at the front-end of the process, as well as the back end of the plant, is well advanced. The same or similar equipment is used either in the Army's baseline program or in industry at the scale required for an agent destruction facility. For example, the punch-and-drain equipment for ton containers has operated successfully at the JACADS chemical demilitarization facility.
CPU Equipment
The state of development of the CPU and related equipment is described above in the Technology Status section. The Demo Unit is a commercial-scale reactor with a metal bath size of 2,700 kg. The three iron CPUs in the CEP conceptual design submitted to the Army are about 8,200 kg each; the nickel bath is about 5,350 kg. Based on these preliminary estimates of nominal bath size, a scale-up of approximately 3:1 from the largest CPU in operation is required. In the judgment of the panel, the TPC has sufficient experience and understanding of CEP technology to perform the scale-up of bath size successfully.
The TPC has told the panel that it plans to use multiple tuyeres in each of the CPUs. Basic oxygen furnaces in the steel industry use many more tuyeres than are under consideration for this process. (At a meeting with the panel in January 1996, a TPC representative said that 16 to 20 tuyeres per furnace is common in the steel industry.) The TPC is continuing to validate the use of multiple tuyeres in an agent destruction CPU, and confirmation on an appropriate number of tuyeres will be part of a final engineering design.
The design concepts for the premelting chamber to melt ton containers and for the system for feeding dunnage (in steel canisters) into the CPU-2 bath do not, to the panel's knowledge, have similarly close industrial counterparts. The TPC has conducted a demonstration program to test the processing of scrap metal, as a surrogate for some solid-waste feed streams of interest to the U.S. Department of Energy. However, the premelting chamber as suggested for the chemical demilitarization facility will require extensive development and demonstration. The TPC's reported experience to date includes a demonstration test in which six marine-location markers supplied by the Department of Defense were enclosed in cylindrical steel containers 0.8 m long and 9 cm in diameter. The containers were fed one by one into a molten metal bath through a gland in the top of the CPU. This test lends some credence to the submitted method for processing dunnage by loading it into cylindrical steel canisters 1 m long by 30 cm in diameter and feeding the canisters into CPU-2.
TABLE 4-7 Summary of Utility Requirements for a CEP Facility
|
|
|
|
|
|
|
|
|
Fuels |
|
Chemical |
|
|
|
Unit |
Description |
Plant Air (scfm) |
Instrument Air (scfm) |
Breathing Air (scfm) |
Nitrogen (scfm) |
Oxygen (scfm) |
natural Gas (Btu x 106h) |
|
Fuel Oil (Btu x 106h) |
Type |
Gallon |
Water Make-U (gal./ min.) |
|
100 |
Feed preparation |
120 @ 200 psig |
10 @ 90 psig |
100 @ 90 psig |
40 @ 50 psig |
|
|
|
|
10% HTH decontam. soln. |
10 to 40 per ton container |
0.5 |
|
|
|
+ 30 @ 90 psig |
|
|
|
|
|
|
|
|
= 5 to 20 gal./h |
|
|
200 |
Catalytic processing |
20 @ 90 psig |
20 @ 90 psig |
100 @ 90 psig |
150 @ 200 psiga |
50 @ 200 psig |
0.6 |
|
|
|
|
|
|
300 |
Gas handling train |
20 @ 90 psig |
10 @ 90 psig |
50 @ 90 psig |
|
|
|
|
|
10% HTH decontam. soln. |
600a |
|
|
500 |
Power generation |
10 @ 90 psig |
10 @ 90 psig |
|
|
|
30.6 |
|
|
|
|
|
|
700 |
Product storage |
20 @ 90 psig |
20 @ 90 psig |
|
|
|
|
|
|
|
|
|
|
900 |
Infrastructure |
100 @ 90 psig |
5 @ 90 psig |
50 @ 90 psig |
10 @ 50 psig |
|
2 |
1.2b |
|
10% HTH decontam. soln. |
600a |
9.5 |
|
1000 |
Relief system |
10 @ 90 psig |
75 @ 90 psig |
150 @ 90 psig |
|
|
0.15 |
|
10%HTH decontam. soln. |
|
600a |
|
|
Totals |
|
120 @ 200 psig + 210 @ 90 psig |
75 @ 90 psig |
300 @ 90 psig |
150 @ 200 psig + 50 @ 50 psig |
50 @ 200 psig |
33.35 |
1.2b |
|
|
|
10.0 |
|
|
|
Steamc |
Boiler Feed Water |
Condensate |
|
|
|
|
|
|
|
|
|
Unit |
Description |
High Pressure (lb./h) |
Low Pressure (lb./h) |
High Pressure (lb./h) |
Low Pressure (lb./h) |
High Pressure (lb./h) |
Low Pressure (lb./h) |
Electrice Power (KW) |
Coolingd Water (gal./ min.) |
Demineralized Water (gal./min.) |
Domestic Water (gal./ min.) |
Sanitary Sewage (gal./ min.) |
|
100 |
Feed preparation |
|
20 @ 15 psig |
|
0.05 |
|
|
|
|
|
|
|
|
200 |
Catalytic processing |
|
|
|
|
|
|
4,000 |
750 |
|
|
|
|
300 |
Gas handling train |
|
|
|
|
|
|
225 |
210 |
5 |
|
|
|
500 |
Power generation |
(1,720) @ 435 psig |
|
4.2 |
|
|
|
(3,525) |
151 |
|
|
|
|
700 |
Product storage |
|
|
|
|
|
|
5 |
|
|
|
|
|
900 |
Infrastructure |
|
780 @ 15 psig |
|
1.95 |
|
0.5 |
800 |
379 |
|
5 |
5 |
|
1000 |
Relief system |
|
|
|
|
|
|
5 |
|
|
|
|
|
Totals |
|
(1,720) @ 435 psig |
800 @ 15 psig |
4.2 |
2 |
0 |
0.5 |
1,510 |
1,500 |
5 |
5 |
5 |
|
a For intermittent use. b Additional fuel oil will be required for a short period, to power diesel generators in case of an electrical power outage. c For steam ( ) indicates quantity produced. d Cooling water supplied at 80ºF and returns at 100ºF. e Connected electric load is 6000 KW. essential load is 3500 KW, UPS load is 150 KW. |
||||||||||||
Performance Scale-Up
Front End and Back End Performance
All the processes in areas 100 and 900 have been demonstrated in the Army baseline system with live agent at scales similar to the scale for an operational CEP facility, except for the optional high pressure water-jet systems for cutting open and cleaning ton containers. The panel expects the water-jet systems will work as proposed because they are commercial systems that have worked well on similar materials under extremely harsh conditions over long periods of time.
CPU Performance
The TPC has done extensive experimentation and modeling of CPU performance to understand bubble formation, breakup dynamics, and the operating limits of molten metal baths. As described in the Process Modeling section, this modeling work has identified three key factors in CPU performance to be bubble size, residence time, and energy dissipation by gas-metal mixing and gas-metal contact within gas bubbles. The TPC states that the modeling results correlate well with DRE values achieved in actual tests. The design for the full-scale baths is stated to provide a residence time with at least a tenfold safety factor over the residence time required to meet the requirement of at least six 9's (99.9999 percent) DRE.
Testing Agent Surrogates in CEP
The TPC tested destruction of an HD surrogate, half-mustard gas (HMG, 2-chloroethyl ethyl sulfide). The result was a DRE of at least nine 9's for conversion of HMG to synthesis gas, HCl, Fe-S alloy, and H2S. The DRE calculation was limited by the amount of agent processed and the lower detection limit of the analytical method.
In another test, diazinon, which is structurally similar to VX, was reported to have been converted to synthesis gas, with the phosphorus and sulfur from the diazinon retained in the metal phase as an Fe-S-P alloy. Analysis of the offgas was conducted in accordance with EPA method TO-14. By this method, no C2 or higher hydrocarbons were detected at the lower detection limits, which are in the part-per-billion range. Third party analyses confirmed that no hazardous organic constituents were present in the ceramic or metal alloy products, which also passed the TCLP test for RCRA metals. The TPC states that the results verify that these solid products are nontoxic and potentially marketable.
The AltTech Panel agrees with the TPC's interpretation of these tests as showing that the technology can destroy agent. The AltTech Panel sees no reason to expect the qualitative aspects of these test results to be different when the process is scaled up. The major conversion products and the partitioning between gaseous and condensed-phase are expected to be the same. The panel also believes the tests provide a strong preliminary indication that the residuals from a carefully designed CEP process to destroy chemical agents are likely to be nontoxic and safe for release to the environment or to commercial use, as the TPC anticipates.
However, the panel cautions that the particular quantitative results obtained in these tests on surrogates, such as a particular DRE value or the nondetection of trace products at parts-per-billion concentrations in residuals, should not be directly extrapolated to full-scale operation unless information on certain key scaling parameters is provided. In the case of CEP test results, an important scaling parameter is one that the panel has named the specific processing rate, which for convenience can be defined as the amount of agent (in kilograms) processed per hour, per unit size of the bath (measured, for example, in 1,000 kg of molten metal). The closer the specific processing rate of a test is to the specific processing rate projected for a full-scale operation, the more confidence one can place in extrapolating quantitative test results. In the case of the tests on agent surrogates, the panel did not receive data from which specific processing rates could be calculated. Therefore, the quantitative results obtained under full-scale operation could be better or worse than these bench-scale test results with agent surrogates.
Testing Actual Agent in CEP
As noted in the Agent Testing section of Technology Status, the TPC has tested actual HD and VX agent in a bench-scale CPU at Battelle/Columbus Laboratories. The panel received the full report on these tests in early June 1996. The report states the agent destruction efficiency of the bench unit as eight 9's (99.999999 percent) for HD and VX. Based on the panel's preliminary review of the report, it appears to be more accurate to
call this result a DRE because the offgas passed through at least one filter before it was tested.
The panel obtained sufficient data on the tests on actual agents to calculate specific processing rates for comparison with the rates for the full-scale system (Table 4-8). (The latter were computed from the design feed rates of agent and the nominal bath size.) Of several bath compositions tested for each agent, the panel used the results from the bath composition closest to that of the full-scale bath under steady-state operation. The bench-scale tests used a single top-entering lance to feed agent into the bath, whereas the design for a full-scale facility has bottom-entering tuyeres.
As the table shows, these bench-scale tests of agent destruction were run at significantly lower specific processing rates than the rates the TPC has designed for a full-scale facility. In the panel's judgment, with the admonition stated above about extrapolating quantitative results from small-scale tests to performance of a full-scale operating facility, the implicit scaling factor in the specific processing rate for VX of 2.6:1 is within acceptable engineering practice. In making this judgment, the panel has taken into account the TPC's stated design safety margin of 10:1 in bath residence time and the reported test result of eight 9's DRE, which implies a performance margin beyond the required six 9's DRE. The panel cautions that the implicit scaling factor in the specific processing rate for HD of 5.4:1 leads to even greater uncertainty in extrapolating the bench-scale DRE to full-scale performance.
The panel believes that the TPC understands the complexity of scaling quantitative performance measures such as DRE from bench-scale tests to full-scale operations. However, the panel would prefer DRE data for VX and especially for HD from bench-scale tests conducted at specific processing rates closer to the rates for the full-scale design.
Unit Operations
This section summarizes the unit operations in CEP treatment of chemical agents for the Aberdeen and Newport sites, including unit operations required to treat secondary process streams and residuals prior to disposal. A unit operation is a combination of equipment that accomplishes one specific step in a process. Table 4-9 lists the unit operations for CEP by process area.
Process Safety
Process safety risk factors for a CEP agent destruction facility can be divided into two categories: factors related to handling agent prior to its introduction into the CPUs and factors related to the molten bath technology.
The risk factors inherent in the handling of agent prior to entry into the CPUs include storage risk, transportation risk, and the risk from the punch-and-drain operation. These risk factors are common to all the agent destruction technologies reviewed in this report, but they can be exacerbated or ameliorated by aspects of a specific technology. For example, how quickly a facility using the technology can reach operational status or the rate at which the agent can be processed with that technology can alter the storage risk by changing the length of time that the agent must be stored. The CEP technology is well advanced, and the design calls for processing the agent at each site in one year. Both of these technology-specific features help in reducing storage risk. As another example, the capability in the CEP design for treating emptied ton containers to the equivalent of 5X condition by melting and processing them immediately reduces the risk from handling the containers. The process safety risk factors inherent in CEP include issues associated with high temperature molten
TABLE 4-8 Specific Processing Rates of Bench Tests Relative to Full-Scale Design Rates
|
|
|
Specific Processing Rate (kg agent/hour/1,000 kg bath metal) |
|
|
|
Agent Tested |
Bath Composition |
Bench Test |
Full-Scale (design) |
Scaling Factor (full-scale/bench) |
|
HD |
Ni + 2% C |
7 |
38 |
5.4 |
|
VX |
Fe + 7% P+ 7% S + C |
8 |
21 |
2.6 |
TABLE 4-9 CEP Unit Operations by Process Area
|
Area 100, Container and Dunnage |
Feed storage (ton containers) |
|
Transportation and Handling |
Punch-and-drain station Ton container wash and preparation Dunnage handling and preparation Liquid (agent and container-washout) storage and feed |
|
Area 200, CPUs |
CPU-1 Premelting chamber to CPU-2 CPU-2 CPU-2 offgas quench, scrub, particulate removal, and compressor |
|
Area 300, Gas Handling Train |
Gas quench and particulate removal HCl recovery Sulfur recovery |
|
Area 500, Synthesis Gas Utilization |
Gas compression and retention/analysis Power generation Steam-methane reformer (option for methanol recovery)a Methanol production (option for methanol recovery)a |
|
Area 700, Products Storage |
Sulfur product storage HCl product storage Methanol product storage (option for methanol recovery)a |
|
Area 800, Utilities |
Inert gas storage and feed Oxygen storage and feed Natural gas feed Air-plant air and instrument air Water-plant, potable, cooling, boiler feed, and chilled Steam-generation and condensate handling Electricity Diesel power backup |
|
Area 1000, Relief and Scrubber System |
Scrubber (decontamination solution) Boilers |
|
a These unit operations are only present if synthesis gas is converted to methanol instead of being burned to generate power. Under the methanol option, the power generation unit process would not be installed. |
|
baths such as the integrity of the refractory confinement, the proximity of the molten bath to water cooling coils (raising the possibility of steam explosions), the behavior of the tuyeres, and the instrumentation for monitoring the refractory confinement. In the panel's judgment, none of these factors presents an insurmountable impediment to the safety of the process. Many of the risk factors have already been addressed by the TPC in the hazard analysis it conducted for design of a chemical demilitarization facility (discussed above under Failure and Hazards Analysis) or on the basis of the TPC's research and operational experience with CEP.
The panel was satisfied that the TPC had adequately addressed several issues the panel had raised during site visits regarding integrity of the refractory. The panel found no evidence of scenarios involving a loss of electrical power, loss of cooling, failures of pumps or valves, breaks in agent lines from inadvertent over-pressurization, or inadvertent temperature transients that would lead to off-site releases of agent or toxic process products. Pessimistic scenarios for a coincident loss of normal power, loss of backup power, and loss of cooling result in the solidification of the molten metal bath in place without significant release to the atmosphere.
Based on the panel's preliminary and qualitative evaluation, the most significant off-site risk appears to be associated with risk factors inherent in handling agent prior to the CEP process. In particular, the principal risk factors appear to involve mishaps during the punch-and-drain operation or damage from airplane crashes or other external events to holding tanks where agent is stored before being fed to the main reactor. The subsections on process safety below address the risk factors specific to CEP technology. However, the panel believes that none of these factors seriously challenges the safety of the facility.
Safety Issues Related to Off Site Releases
The following issues should be addressed fully and clearly in a final CEP process design.
Integrity of the Refractory. The work by the TPC on the integrity of the refractory must be included in the safety documentation for a final CEP design. The TPC has done much work to avoid gas-jet impingement on the refractory lining of the CPU and to select refractory materials for the lining that resist gas permeation, thermal degradation, corrosion, erosion, and penetration by components of the molten metal and slag.
Integrity of the Agent-Bearing Components. This issue was explored briefly by the panel, and no significant issues were uncovered. However, because certain parts of the design are still preliminary, the panel encourages the TPC to pursue its stated plans for continuing, comprehensive safety and hazard analyses as part of the development process. Particularly important is further exploration of scenarios involving failures of piping or components. (Failure could be caused by thermal attack by molten material, system overpressure, subtle system interactions, or other causes.)
Cooling Offgas Piping. Scenarios involving a failure to cool the offgas piping should be explored. This is probably not an issue, but at the time of the panel's review, the consequences of such scenarios were not clear.
Buildup of Combustible Gases. The TPC's design as submitted prevents a buildup of combustible gases in the vicinity of the system by maintaining a high ventilation rate. Assurances should be made that combustible gas buildup cannot occur and that the high ventilation rate does not compromise the design capability to contain leakage of agent.
Worker Safety Issues
There are a number of worker safety issues associated with high temperature molten baths, high temperature corrosives in the scrubbers, and secondary containment (concerning both inadvertent leaks and maintenance activities). These risk factors need to be addressed in the final operational design, and realistic emergency responses need to be spelled out.
Specific Characteristics that Reduce Risk Inherent in the Design
Because of the natural temperature gradient in the CPU refractory material, the molten material will solidify before it gets very far into the refractory. This self-sealing feature helps keep the molten metal away from the water-filled induction coils and thus reduces the possibility of a steam explosion.
A loss of electrical power, of cooling water to the heat exchanger, or of the cooling for pumps could result in the molten metal solidifying in place. Although solidification would be an operational problem if it were to occur, it is not a safety issue.
Schedule
Figure 4-11 is the latest schedule submitted to the AltTech Panel from the TPC for the major activities and milestones in a chemical demilitarization program to use CEP technology at the Aberdeen and Newport sites. Table 4-10 is the panel's analysis, based on the TPC schedule, of activities on the critical path to completion of the program, their duration, and the cumulative time from start of the program to the end of that activity. An important aspect of the TPC's concept as submitted to the Army is that the same CEP equipment would be installed first at Aberdeen for HD destruction, then moved to Newport and installed there for VX destruction. Advantages and disadvantages of this approach are discussed below.
Another key aspect of the design is that the TPC's preferred approach, after a go-ahead from the Army to begin work, is to move directly to design of a facility
Figure 4-11
CEP program schedule and phasing concept. Source: M4 Environmental L.P., 1996d.
with full-scale CPUs for the next stage of development. A facility at that scale is more conventionally referred to as a demonstration plant than a pilot plant. To indicate how the schedule relates to the Defense Acquisition Board's decision to proceed with pilot-scale development, the panel will refer to this next stage as pilot/ demonstration. The facility for this pilot/demonstration phase at each site will be equipped with enough gas handling capability to ensure protection of human health and the environment, but the full gas handling train will not be installed until full-scale operation.
The TPC foresees no scale-up effort required to move from pilot-testing to full-scale processing. The panel cautions, however, that although use of full-scale equipment at the pilot/demonstration stage means that no equipment scale-up will be required, whether performance scale-up is needed depends on how closely the final stages of pilot-testing resemble the process conditions for full-scale, continuous operation. The pilot/demonstration activities will entail a good deal of work, including systemization with agent surrogates, preoperational surveys, an operational readiness evaluation, and similar requirements prior to full-scale operation. Provided that the TPC continues testing and develops an adequate design basis prior to construction of the pilot/demonstration facility (that is, resolves remaining issues such as demonstrating the premelting chamber, scaling the bath to the larger size required, resolving the number and placement of tuyeres, and demonstrating process performance at the design
TABLE 4-10 Critical Activities in the Program Schedule
|
Activity |
Duration (months) |
Cumulative (months) |
|
1. Prepare and obtain regulatory permits, etc., for Aberdeen |
15 |
15 |
|
2. Aberdeen construction (site prep. and installation) |
12.2 |
27.2 |
|
3. Aberdeen pilot/demonstration (startup, test, and system modifications) |
8 |
35.2 |
|
4. Aberdeen full-scale HD operations |
12 |
47.2 |
|
5. Newport construction (site prep. and installation)a |
14.25 |
60.45 |
|
6. Newport pilot/demonstration (startup, test, and system modifications) |
8.25 |
68.7 |
|
7. Newport full-scale VX operation |
13 |
81.7 |
|
a Newport construction overlaps one month with Aberdeen full-scale operation. |
||
specific processing rates), the panel believes that 8 months can suffice for performance scale-up and required startup activities.
The full-scale operation at each site is designed to be continuous, 24 hours per day, at the agent feed rates specified above in the Feed Streams section. The scrubbed offgas is either combusted with natural gas in a gas turbine generator to produce electricity for the plant or converted to methanol. At this stage, process residuals would be placed on the commercial market. The design as submitted is not clear about how process residuals would be handled during the earlier pilot/ demonstration stage.
The TPC has stated that the submitted design provides sufficient throughput to allow all agent, ton containers, and dunnage to be destroyed in 12 months from the start of full-scale operation at Aberdeen and in 13 months from the start of full-scale operation at Newport (M4 Environmental L.P., 1996d). Assuming that construction at Aberdeen can be approved by January 30, 1998, the TPC anticipates that the program for both sites will be completed before the end of 2003, more than a year before the Army deadline of December 31, 2004. The AltTech Panel believes that the TPC's goal of completing the destruction of each stockpile in 12 to 13 months after commencing full-scale operation is achievable, if the throughput rates assumed in the submission can be sustained for the duration of the operation.
In the panel's judgment, the time allotted for pilot/demonstration activities at Newport is essential. The VX configuration uses the same equipment but a different set of processing parameters and constraints, as well as handling a different agent and a different partitioning of chemical elements to product phases.
After processing HD at Aberdeen has been completed, the CEP systems will be decontaminated, decommissioned, and relocated to Newport for processing VX. The TPC believes this plan for reusing equipment is a cost-effective and time-saving solution for destroying agent stockpiles at multiple sites. The panel agrees that there are advantages to sequential operations but cautions that there are also risks to the schedule. A significant delay in the Aberdeen schedule could delay the agent destruction schedule at Newport. In fact, any delay in one of the activities along the critical path can delay subsequent activities.
For example, the submitted schedule reflects early and vigorous efforts to complete the required reviews and secure necessary approvals. The TPC estimates that a permit for construction of a plant producing atmospheric emissions can be obtained in Maryland within 15 months of project start. The panel notes that this relatively short time for permitting may depend on the TPC acquiring a recycle waiver from RCRA permitting requirements. If the permitting process takes longer and construction is delayed, the schedule does have about
15 months of slippage time at the end to still meet the Army deadline.
The panel notes in passing that the time shown in Figure 4-11 for decontamination and decommissioning is probably only the time required to decontaminate and decommission the CEP systems. (The schedule refers to the activity as phase 1 of decontamination and decommissioning.) Additional time will probably be required for decontaminating and decommissioning the central building and the associated infrastructure.















































