1
Introduction
The Call for Disposal
The United States has maintained a stockpile of highly toxic chemical agents and munitions for more than half a century. Chemical agents are extremely hazardous, which is why they have been used in weapons. The manufacture of chemical agents and munitions and their subsequent stockpiling were undertaken in the belief that they had value as deterrents to the use of similar materials against U.S. forces. Today, other deterrents are considered more appropriate. In an attempt to avoid the worldwide risk posed by chemical warfare, the United States is entering into an agreement with many other nations to rid the world of all chemical weapons and munitions. Even apart from this agreement, the United States can no longer justify the continuing risk and expense of storing them. Consequently, there is ample incentive for the United States to dispose of its chemical agents and munitions as soon as this can be done safely.
In 1985, Public Law 99-145 mandated an "expedited" effort to dispose of M55 rockets because these particularly hazardous munitions have the potential for self-ignition during storage. The M55 rockets are loaded with chemical agent, a fuse, an explosive designed to disperse the agent (a burster), and ignition-ready rocket propellant. This mandate soon expanded into the Army Chemical Stockpile Disposal Program (CSDP), whose mission was to eliminate the entire stockpile of unitary 1 chemical weapons. The CSDP developed the current baseline incineration system for this purpose. In 1992, after setting several intermediate goals and dates, Congress enacted Public Law 102-484, which directed the Army to dispose of the entire unitary chemical warfare agent and munitions stockpile by December 31, 2004.
Description of the Stockpile
Agents
The principal unitary chemical agents in the U.S. stockpile are the two nerve agents (GB and VX)2 and three related forms of blister, or mustard, agent (H, HD, and HT). These agents are stored and exist largely as liquids: nerve agent VX, a high-boiling point liquid that will adhere to surfaces for days or weeks; nerve agent GB (sarin), a liquid that has a volatility similar to water and therefore evaporates relatively quickly; and a blister agent (mustard) that evaporates slowly. These agents are stored in a variety of munitions and containers. The stockpile consists of 30,600 tons of unitary agents (U.S. Army, 1996h).
Nerve agents are organophosphonate compounds; that is, they contain phosphorus double-bonded to an oxygen atom and single-bonded to a carbon atom. They are highly toxic and lethal in both liquid and vapor forms. They can kill in a matter of minutes by interfering with respiratory and nervous system functions. In pure form, nerve agents are practically colorless and odorless. GB evaporates at about the same rate as water and is relatively nonpersistent in the environment. VX evaporates much more slowly and can persist for a long time under average weather conditions.
Bis(2-chloroethyl)sulfide is the principal active ingredient in blister agents, or mustard.3 Mustard has a
TABLE 1-1 Physical Properties of Chemical Warfare Agents
|
Agent Characteristic |
VX (Nerve Agent) |
HD (Blister Agent) |
|
Chemical formula |
C11H26NO2PS |
(ClCH2CH2)2S |
|
Molecular weight |
267.38 |
159.08 |
|
Boiling point, °C |
298 |
217 |
|
Freezing point, °C |
< -51 |
14.45 |
|
Vapor pressure, mm Hg |
0.0007 @ 25°C |
0.072 @ 20°C |
|
Volatility, mg/m3 |
10.5 @ 25°C |
75 @ 0°C (solid) |
|
|
|
610 @ 20°C (liquid) |
|
Surface tension, dynes/cm |
32.0 @ 20°C |
43.2 @ 20°C |
|
Viscosity, cS |
12.256 @ 20°C |
3.95 @ 20°C |
|
Liquid density g/cm3 at 20°C |
1.0083 |
1.2685 |
|
Solubility, g/100 g of distilled water |
5 @ 25°C; best solvents are dilute mineral acids |
0.92 @ 22°C; soluble in acetone, CCl4, CH3Cl, tetrachloroethane, ethyl benzoate, ether |
|
Heat of combustion |
|
|
|
Btu/lb |
15,000 |
8,100 |
|
(cal/g) |
(8.33) |
(4.5) |
|
Source: NRC, 1993. |
||
garlic-like odor and is hazardous on contact and as a vapor. Because it is practically insoluble in water, mustard is very persistent in the environment. Table 1-1 lists some of the physical properties of VX and HD.
Containers and Munitions
Unitary chemical agents are stored in spray tanks, bulk-storage (ton) containers,4 and a variety of munitions including land mines, M55 rockets, bombs, and artillery and mortar projectiles. Some munitions contain no explosives or propellant, whereas others contain some combination of fuse, booster, burster, and propellant. These components are referred to collectively as energetics. They incorporate a variety of chemical compounds that must also be eliminated as part of the CSDP.
Geographical Distribution
The unitary chemical stockpile is located at eight continental U.S. storage sites (see Figure 1-1) and at Johnston Atoll in the Pacific Ocean about 700 miles southwest of Hawaii. Table 1-2 gives the composition of the stockpile at each continental U.S. site by type of container or munition and by type of agent.
As specified in the study panel's statement of task, only the two sites at Aberdeen Proving Ground, Maryland, (Aberdeen site) and at the Newport Chemical
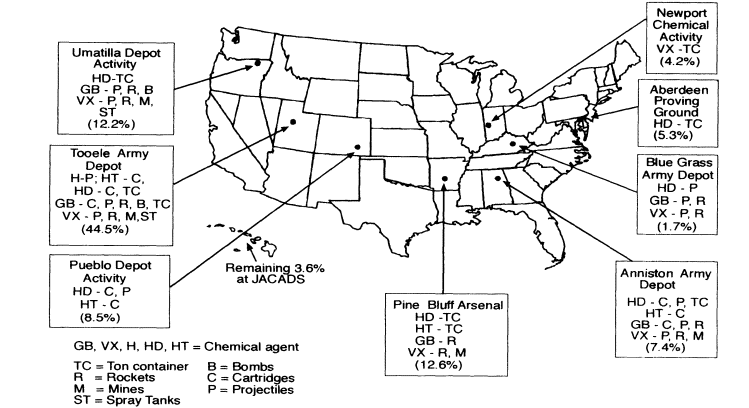
Figure 1-1
Types of agent and munitions and percentage of total agent stockpile at each storage site. Derived from OTA, 1992; NRC, 1996.
Activity, Indiana, (Newport site) are considered in this report. The unitary agent stockpile at the Aberdeen site consists entirely of HD (1,625 tons in 1,818 ton containers), and the stockpile at the Newport site consists entirely of VX (1,269 tons in 1,689 ton containers) (U.S. Army, 1996h). Because munitions containing agent and energetics are not present, the process requirements for disposing of only ton containers of agent are less demanding than the processing requirements for the more complex stockpiles at other sites.
The VX nerve agent stored at the Newport site is 90.5 to 94.8 percent pure. It was formulated with 1 to 3 percent diisopropyl carbodiimide as a stabilizer to protect it against decomposition by traces of water. During the 30 to 40 years that the VX has been in storage, some of the stabilizer has hydrolyzed, but most of the nerve agent has not been affected. Traces of a toxic compound, called "pyro," 5 are present from VX hydrolysis. An impurity, called "bis," which is formed during VX manufacture, hydrolyzes to give EA-2192, which is also highly toxic. In a recent survey conducted by the Army, gas chromatographic analysis of the materials in containers of VX (32 containers were randomly selected and sampled) revealed the presence of the compounds shown in Table 1-3 (U.S. Army, 1996f). Other components, such as bis, have also been detected in some samples by 31PNMR (phosphorus 31 nuclear magnetic resonance spectroscopy).
The HD agent stored at the Aberdeen site was distilled when produced, but it also contains several impurities formed either during manufacture or from decomposition of the HD during storage. The Army estimates that each ton container of HD contains about 14 pounds of "land-banned" chemical impurities (chemicals subject to strict hazardous waste regulations, including limitations on landfill disposal). These strictly regulated impurities include 1,2-dichloroethane, trichloroethylene, tetrachloroethylene, 1,1,2,2-tetrachloroethane, and hexachloroethane. There are also about 30 pounds of dithiane per container and varying amounts of chloroethyl sulfides other than HD. In a
TABLE 1-2 Chemical Munitions Stored in the Continental United States
|
Chemical Munitions (Agent) |
APG |
ANAD |
BGAD |
NECA |
PBA |
PUDA |
TEADa |
UMDA |
|
Mustard agent (H, HD, or HT) |
|
|
|
|
|
|
|
|
|
105-mm projectile (HD) |
|
X |
|
|
|
X |
|
|
|
155-mm projectile (H, HD) |
|
X |
X |
|
|
X |
X |
|
|
4.2-in. mortar (HD, HT) |
|
X |
|
|
|
X |
X |
|
|
Ton container (HD) |
X |
X |
|
|
X |
|
X |
X |
|
Ton container (HT) |
|
|
|
|
X |
|
|
|
|
Agent GB |
|
|
|
|
|
|
|
|
|
105-mm projectile |
|
X |
|
|
|
|
|
|
|
155-mm projectile |
|
X |
X |
|
|
|
X |
|
|
8-in. projectile |
|
X |
X |
|
|
|
X |
X |
|
M55 rocket |
|
X |
X |
|
X |
|
|
X |
|
500-lb bomb |
|
|
|
|
|
|
X |
X |
|
750-lb bomb |
|
|
|
|
|
|
X |
X |
|
Weteye bomb |
|
|
|
|
|
|
X |
X |
|
Ton container |
|
Xb |
|
|
|
|
X |
|
|
Agent VX |
|
|
|
|
|
|
|
|
|
155-mm projectile |
|
X |
X |
|
|
|
X |
X |
|
8-in. projectile |
|
|
|
|
|
|
X |
X |
|
M55 rocket |
|
X |
X |
|
X |
|
X |
X |
|
M23 land mine |
|
X |
|
|
X |
|
X |
X |
|
Spray tank |
|
|
|
|
|
|
X |
X |
|
Ton container |
|
|
|
X |
|
|
X |
|
|
Miscellaneous |
|
|
|
|
|
|
|
|
|
Ton containers (L) |
|
|
|
|
|
|
X |
|
|
Ton containers (GA) |
|
|
|
|
|
|
X |
|
|
a Small quantities of Lewisite and tabun (GA) are stored in ton containers at TEAD. b Small quantities of agent drained as part of the Drill and Transfer System assessment for the M55 rockets. NOTE: APG, Aberdeen Proving Ground, Maryland; ANAD, Anniston Army Depot, Alabama; BGAD, Blue Grass Army Depot, Kentucky; NECA, Newport Chemical Activity, Indiana; PBA, Pine Bluff Arsenal, Arkansas; PUDA, Pueblo Depot Activity, Colorado; TEAD, Tooele Army Depot, Utah; and UMDA, Umatilla Depot Activity, Oregon SOURCE: Adapted from NRC, 1996 and U.S. Army, 1996h. |
||||||||
recent survey conducted by the Army, analysis of the materials in 27 randomly selected and sampled containers of HD reveals the compounds shown in Table 1-4 (U.S. Army, 1996g). In addition to these impurities, which are dissolved in the much larger quantity of HD in the container, all containers tested recently at Aberdeen appear to contain solid or semisolid deposits, called a "heel." The quantities and composition of the heel vary from container to container, but it appears to consist largely of sulfonium and iron salts with adsorbed HD. The heel solids appear to dissolve readily in hot water (U.S. Army, 1996b). The relatively high freezing point of HD (14.45°C) and the outside storage of ton containers at Aberdeen will require facilities to thaw HD during cold weather, prior to processing. This requirement is independent of the destruction technology evaluated and is based on the required processing rates and the maximum amount of agent that can be present in a destruction facility at one time.
Role of the National Research Council
The National Research Council (NRC) Committee on Demilitarizing Chemical Munitions and Agents was formed in August 1983 to review the status of the
TABLE 1-3 Composition of VX from Ton Containers Stored at Newport (based on gas chromatography analysis)
|
Compound |
Average (weight percent) |
|
VX |
93.71 |
|
Dimethyl ketone (acetone) |
0.01 |
|
Diisopropylamine |
0.14 |
|
N,N-Diisopropylmethylamine |
0.01 |
|
Diisopropyl Carbodiimide (stabilizer) |
1.74 |
|
N,N-Diisopropylethylamine |
0.01 |
|
O-Ethyl methylphosphonate |
0.20 |
|
1,3-Diisopropylurea |
0.03 |
|
Diethyl methylphosphonate |
0.06 |
|
2-(Diisopropylamino) ethane thiol |
0.89 |
|
O,O-Diethyl methylphosphonate |
0.21 |
|
O,S-Diethyl methylphosphonothioate |
0.07 |
|
2-(Diisopropylamino)ethyl ethyl sulfide |
0.13 |
|
Diethyl dimethylpyrophosphonate (pyro) |
0.99 |
|
O,O-Diethyl dimethylpyrophosphonothioate |
0.23 |
|
O-(2-Diisopropylaminoethyl) |
|
|
O-ethylmethylphosphonate |
0.26 |
|
1,2-bis(ethyl methylphosphonothiolo)ethane |
0.62 |
|
Unknowns, plus trace metals |
0.69 |
|
Total |
100.00 |
|
Source: U.S. Army, 1996g. |
|
stockpile and technologies for disposal. That committee reviewed a range of technologies and, in its final report in 1984, endorsed incineration as an adequate technology for the safe disposal of chemical agents and munitions (NRC, 1984). The committee also concluded that the stockpile was well maintained and posed no imminent danger but expressed concern about future storage risk due to the potential for an increased rate of stockpile deterioration.
In 1987, at the request of the Undersecretary of the Army, the Committee on Review and Evaluation of the Army Chemical Stockpile Disposal Program (referred to as the Stockpile Committee) was established under the aegis of the NRC Board on Army Science and Technology to provide the Army with technical advice and counsel on specific aspects of the disposal program. Under this charter, the Army has requested and received from the Stockpile Committee 15 reports that evaluated stages of progress and specific aspects of the program.
In March 1991, as a result of growing public concerns about and opposition to the baseline incineration system and the rising cost of the CSDP, the Stockpile Committee suggested, and the Army agreed, that a new study of alternatives to incineration for the destruction of the stockpile should be undertaken.
In January 1992, the NRC, at the request of the office of the Assistant Secretary of the Army for Installations, Logistics and Environment, established the Committee on Alternative Chemical Demilitarization Technologies (Alternatives Committee) to develop a comprehensive list of alternative technologies and to review their capabilities and potential as agent and munitions disposal technologies. In June 1993, this committee published its report, Alternative Technologies for the Destruction of Chemical Agents and Munitions (NRC, 1993).
The Stockpile Committee, working with the report of the Alternatives Committee and with its own knowledge of the baseline system and disposal requirements, formulated recommendations regarding the investigation of potential alternatives to incineration. This work was reported in February 1994 in Recommendations for the Disposal of Chemical Agents and Munitions (NRC, 1994b). The Stockpile Committee concluded that the baseline system is adequate for disposal of the stockpile and that the storage risk will persist until disposal of all stockpile materials is complete. The report recommended that the CSDP proceed expeditiously and with technology that minimizes total risk to the public at each site.
The Stockpile Committee also found, after examination of all the technologies brought to its attention by the Alternatives Committee and others, that four neutralization-based systems offered the most promise for agent destruction (NRC, 1994b). In view of the increasing total risk associated with delays in the disposal program, and recognizing that public opposition might delay the program for a number of reasons, including opposition to incineration, the committee stated that alternative technologies should be developed promptly. The committee also recommended that the Army continue to
TABLE 1-4 Composition of HD from Ton Containers Stored at Aberdeen
|
Compound |
Average (weight percent) |
|
HD |
90.20 |
|
2 methyl 1-propene |
0.021 |
|
thiirane |
0.017 |
|
2-chlorobutane |
0.002 |
|
1,2-dichoroethane |
0.350 |
|
1,4-oxathiane |
0.070 |
|
1,4-dithiane |
1.476 |
|
trichloroethylene |
0.001 |
|
1,2,5-trithiepane |
0.086 |
|
tetrachloroethylene |
0.132 |
|
1,1,2,2-tetrachloroethane |
0.037 |
|
2-chloroethyl 3-chloropropyl sulfide |
1.092 |
|
bis(2-chloropropyl) sulfide |
0.366 |
|
C6H12C12S isomers |
0.548 |
|
2-chloroethyl 4-chlorobutyl sulfide |
1.136 |
|
bis(2-chloroethyl disulfide) |
0.643 |
|
2-chloroethyl (2-chloroethoxy) ethyl sulfide |
0.054 |
|
Q, 1,2-bis(2-chloroethylthio) ethane |
2.639 |
|
bis(2-chloroethyl) trisulfide |
0.072 |
|
hexachloroethane |
0.152 |
|
Unknown |
0.015 |
|
Copper as CuCl2 |
0.003 |
|
Iron as FeCl2 |
0.888 |
|
Total |
100.00 |
|
Source: U.S. Army, 1996g. |
|
monitor other research programs and developments involving potential alternatives.
In April 1994, the Army produced its own report, U.S. Army's Alternative Demilitarization Technology Report for Congress (U.S. Army, 1994). The Army accepted the Stockpile Committee's recommendation to pursue neutralization-based technologies but limited the Army's research and development to two alternatives: (1) stand-alone neutralization, and (2) neutralization followed by biodegradation. The Army also agreed to monitor additional developments in alternative disposal technologies.
One aspect of the Army's work on neutralization alternatives was to prepare detailed assessment criteria for decisions on proceeding with the development of neutralization technologies. The Army released its draft report of these criteria in April 1995 as Assessment Criteria to Aid in the Selection of Alternative Technologies for Chemical Demilitarization (U.S. Army, 1995a, hereafter cited as the Army Criteria Report). The Army also asked the Stockpile Committee to evaluate these draft criteria, which it did in Evaluation of the Army's Draft Assessment Criteria to Aid in the Selection of Alternative Technologies for Chemical Demilitarization (NRC, 1995, hereafter cited as the NRC Criteria Report Evaluation). Both of these reports were particularly pertinent to the present study.
Scope and Organization of the Study
Since these earlier reports, the Army believes that research developments have sufficiently enhanced the database on the performance of some alternative technologies to warrant reexamination of specific alternatives for use at certain sites. In the summer of 1995, the Assistant Secretary of the Army for Research, Development and Acquisition informally explored with the NRC Stockpile Committee the possibility of examining alternative chemical disposal technologies. Following numerous discussions between the Army and the NRC, a decision was made to conduct a new NRC study to reexamine the status of a limited number of alternative chemical disposal technologies to be selected by the Army (including the two neutralization-based processes on which the Army was currently conducting research) for possible use in the CSDP.
In August 1995, the Army advertised in the Commerce Business Daily (Appendix A) for alternative disposal technologies other than the two already being evaluated by the Army. The purpose of this announcement was to determine whether any other technologies were capable, within the CSDP schedule, of meeting chemical demilitarization requirements for the two sites where agent is stored only in bulk (the Aberdeen and Newport sites). The announcement requested information from industry on non-incineration technologies that were sufficiently developed to meet the needs of the CSDP. Following a preliminary 30-day screening review, the Army in November 1995 selected three technologies for review and evaluation by the NRC—gas-phase reduction, molten metal catalytic
extraction, and electrochemical oxidation—in addition to the two processes, neutralization and neutralization followed by biodegradation, that were already being developed by the Army.
In parallel with the Army selection process, the NRC formed the Panel on Review and Evaluation of Alternative Chemical Disposal Technologies (AltTech Panel). The AltTech Panel held its first meeting prior to the announcement of the Army's selection. Anticipating the broad types of technologies that might be selected by the Army, the panel developed a project plan and preliminary report outline, based on its knowledge of the Stockpile Committee reports and activities. The NRC added three members to the panel after submissions were received for the three technologies to be reviewed. The new members were added to supplement the expertise already on the panel and to provide coverage for the specific technologies to be evaluated.
From November 1995 to June 1996, the panel conducted in-depth reviews and evaluations of the five selected technologies. The entire panel met six times; designated panel teams conducted 14 site visits to study the technologies; and panel members met with regulators, citizens advisory commissions (CACs), and local citizens in Maryland and Indiana. The panel's activities are delineated in the following statement of task.
At the request of the Assistant Secretary of the Army for Research, Development and Acquisition, the National Research Council will carry out a review of alternative chemical agent disposal technologies. To conduct this review, a Panel on Review and Evaluation of Alternative Chemical Disposal Technologies under the auspices of the Board on Army Science and Technology will examine no more than three alternative technologies (to the baseline incineration system), as well as neutralization and neutralization followed by biodegradation for the disposal of chemical agent at Aberdeen, Maryland (mustard agent) and Newport, Indiana (nerve agent) only. The panel will meet, as appropriate, to:
- establish criteria to assess and evaluate selected alternative technologies;
- conduct site visits as appropriate to assess firsthand the viability and maturity of technologies being reviewed;
- conduct site visits to possible locations where alternative technologies may be employed and to hold open meetings there to solicit CAC views on the alternative technologies under consideration;
- assess technical aspects, strengths and weaknesses, and advantages and disadvantages of each technology;
- consider the option of shipping treated effluents (agent free) to off-site appropriately permitted disposal facilities; and
- make recommendations regarding which, if any, of these technologies merit full evaluation and presentation to the Defense Acquisition Board6 as candidates for pilot plant demonstration by the Army.
Initially the Army also asked the panel to examine technologies to be used solely for the treatment of neutralization hydrolysate. (Hydrolysate is the aqueous solution of products from the neutralization step.) These technologies were not "stand-alone" technologies (like those selected by the Army for consideration for total on-site agent-treatment) but were polishing steps to be taken after neutralization. The panel felt the limited time available would not allow for a complete investigation leading to specific recommendations in this report regarding these technologies. However, consistent with earlier Stockpile Committee report recommendations and based on information provided by the Army, the AltTech Panel is aware that the Army continues to examine technologies for this purpose and supports these efforts.
In conducting this review, the panel recognized that, although it had been charged with evaluating technologies, each of the technologies under evaluation was being developed and submitted for consideration by a specific company. (Hereafter, these companies are referred to as technology proponent companies, or TPCs.) Consequently, the present engineering status of each technology is company-dependent, and the panel's evaluations must, by necessity, depend on the TPCs for information. However, the panel's evaluations apply only to the application of each technology, as submitted for the panel's consideration, to agent destruction at the bulk-storage sites, not to the general capabilities of the TPC or to other applications of the technology.
The panel's interactions with the TPCs during the course of this study clearly showed that technology development had continued after the October submissions responding to the announcement in the Commerce Business Daily. The panel realized that these technologies will continue to evolve, but to conduct a review within the time provided, the panel requested that all TPCs submit "final" designs by April 4, 1996. Hence
the technology assessments and evaluations in this report reflect the status of each technology as of that date.
The Army required that the TPCs perform supervised tests to obtain data on how the technology performed in destroying actual chemical agent. The tests were conducted by an Army-approved laboratory at the TPCs' expense. The test data were not available to the NRC panel for review until late June, which did not allow enough time for the panel to conduct an assessment of the reported by-products produced during the tests. Consequently, the tests were used by the panel simply to make a yes-or-no determination as to whether the technology can destroy agent.
In addition, all TPCs were required to give the Army projected cost and implementation schedules by March 17, 1996. The cost data were not provided to the panel and their consideration is outside the scope of this study.
Public Law 102-484 identifies safety as a critical factor in the selection of a technology for the alternative technology program. Process safety risk encompasses risk to the health and safety of workers and the public, as well as risk to the environment. The panel insisted, and the Army agreed, that, consistent with the varying depth and scope of available technical information on the proposed alternative technologies and the need to provide timely support to the Defense Acquisition Board's decision-making process and the NRC panel review, the Army would request preliminary risk assessments of the technologies by an independent contractor (MitreTek Systems, Inc.).
The scope of work for this risk assessment required that the contractor provide a preliminary assessment of the potential process safety risks associated with implementing the baseline incineration system as compared with each of the five alternative disposal technologies at the Aberdeen and Newport sites. Significant discriminators of process safety risk among the baseline system and the alternative technologies were to be identified and evaluated. Discrimination was to be based on safety and health risks to workers, safety and health risks to the public, environmental risks, and storage risks. Risks to plant equipment and operations were not to be considered directly. The contractor was required to present results of the preliminary risk assessment in a draft report by April 15, 1996, and to provide a final report by May 31, 1996.
The contractor's analysis was constrained by two factors. (1) Because the technical information and design maturity of the proposed alternative technologies are at present limited in comparison with the baseline system, assessments of certain aspects of risk were limited and qualitative in nature. (2) The time available to perform the analysis precluded detailed analysis of even the limited information available on the alternative technologies.
The AltTech Panel's risk assessment expert participated in some of the contractor's efforts to gather data, performed an independent risk evaluation of the five technologies, and reviewed the contractor's report. These activities enabled the panel to assess, on a qualitative basis, the process safety risks for each alternative technology. The independent risk evaluation focused on characteristics inherent in each technology that had the potential to lead to accidental release and only briefly addressed accident scenarios caused by combinations of system failures (pipes, pumps, valves, power systems, and cooling systems). The hazards of transporting ton containers from storage to the processing area and of the punch-and-drain operations to remove the agent from containers are common to all the technologies being evaluated. However, the mode of feeding agent into the process may be somewhat more hazardous for some technologies than for others (the differences are discussed in Chapters 4 through 8).
At this point, however, no comprehensive, quantitative risk assessment has been performed on any of the alternative technologies.
The Commerce Business Daily announcement and the Army's criteria for selecting potential alternative technologies required that TPCs demonstrate the feasibility of using their technology to conduct all the activities required to process agent on-site,7 consistent with the objectives and capabilities of the baseline incineration system. However, since the time of the announcement, the CSDP has continued to explore ways to increase cost-effectiveness. Off-site shipping, for example, is already being used for limited quantities of various process wastes, including empty, cleaned ton containers; used decontamination fluids; and hydrolysate from tests of the neutralization technology. Offsite shipping on a larger scale may significantly improve cost-effectiveness. Consequently, the statement of task for the AltTech Panel was amended to direct that the panel examine the option of shipping process wastes off-site for final treatment. This option is discussed in Chapter 3.
Report Organization
The AltTech Panel divided its evaluation into three phases: organization, data gathering, and report preparation. Because time was limited, the principal data-gathering efforts could not exceed six months and each phase had to be carefully planned. The organization of this report reflects these efforts.
Before site visits were undertaken, the panel extracted relevant evaluation factors from the Army Criteria Report and the NRC Criteria Report Evaluation and developed its own framework for evaluation. This framework became the basis for a questionnaire sent to the TPCs and the Army well before the panel's site visits. Chapter 2 discusses the evaluation factors, and Chapter 3 describes the framework for gathering information.
Chapters 4 through 8 contain specific technology assessments based on the information gathered by the panel. Chapter 4 assesses the catalytic extraction process (molten metal); Chapter 5, electrochemical oxidation; Chapter 6, gas-phase reduction; Chapter 7, neutralization of HD; and Chapter 8, neutralization of VX.
The regulatory process and the opinions of the public and other stakeholders can have a dramatic effect on the implementation schedule. Because delays extend the time of exposure to stockpile storage risk, they can increase overall risk. To assess these effects, the panel held meetings with regulators in Maryland and Indiana and conducted public forums, where concerned citizens were encouraged to voice their opinions of the alternatives under consideration. Chapter 9 discusses this aspect of the study.
Chapter 10 presents the panel's comparison of the alternative technologies based on the criteria developed in Chapter 2. Chapter 11 contains the major findings and recommendations that the panel distilled from the technology assessments and from comparing the technologies.
Because of time constraints on preparing this report and because agent test data were not available until very late in the process, the panel was not able to analyze these test data in-depth. Also, the panel had time for only a preliminary review of the MitreTek Systems risk assessment report. Both issues are discussed further in Chapter 2.









