5
Current STD-Related Services
|
Highlight
|
Current STD-related services and activities in the United States comprise several components, including the delivery of clinical services by health care providers, disease surveillance and information systems, training and education of health care professionals, and funding of activities and programs. Most of the components are publicly sponsored programs; but some programs, such as training and education of health professionals, are carried out by both the public and private sectors. Components such as national health surveys are directed and supported by the federal government, while others, such as disease surveillance, involve all levels of government and the private sector. Although the private sector is primarily involved in delivery of clinical services to persons with private health care insurance, this situation is rapidly changing and may have significant implications for the delivery of STD-related services.
Clinical Services
Clinical services for STDs—screening, diagnosis and treatment of STDs, patient counseling, and partner notification and treatment—are provided primarily in one of three settings:
- dedicated public STD clinics, operated by local health departments; 1
- community-based health clinics, operated by community-based health professionals or agencies that usually receive public funds; and
- private health care settings, including private physician offices, health-plan-affiliated facilities, private clinics, and private hospital emergency rooms.
The public, community-based, and private settings for STD-related care serve somewhat different, albeit overlapping, population groups, each of which has different needs related to STD prevention.
Health Care Professionals and Prevention Activities
There is a broad range of health care professionals involved in STD-related care. Most clinicians who provide STD-related care in public or private settings emphasize diagnosis and treatment and, to a lesser extent, management of sex partners rather than other approaches to STD prevention (Bowman et al., 1992). Most clinicians do not provide adequate STD risk assessment, prevention counseling, or other STD-related education, despite the fact that they may include some STD screening in their patients' medical evaluation (Lewis and Freeman, 1987; Lewis et al., 1987; Gemson et al., 1991; Bowman et al., 1992; Russell et al., 1992). In a 1986 survey of California internists, only 10 percent reported asking new patients questions that were specific enough to assess their risk of STDs (Lewis and Freeman, 1987). In a more recent national survey of primary care physicians and other health care providers (registered nurses, nurse practitioners, nurse midwives, and physician assistants), only 39 percent of physicians and 49 percent of other primary care providers reported conducting risk assessment for STDs for all or most of their new adult patients (ARHP and NANPRH, 1995). A survey of 961 physicians in the Washington, D.C., area found that only 37 percent of respondents reported regularly asking new adult patients about their sexual practices and that 60 percent did so for new adolescent patients (Boekeloo et al., 1991). Reasons typically cited for these deficits, as mentioned elsewhere in this report, include (a) health professionals' common skepticism of the efficacy of health education and behavioral interventions; (b) pressures to see large numbers of patients in a brief amount of time; (c) personal discomfort regarding taking accurate, nonjudgmental sex and STD histories, attributed to lack of training and other reasons; and (d) a widespread misconception that STDs and issues related to sexuality are too "sensitive" to discuss. The last perception is not correct; one study found that patients who were asked questions about sexual and STD histories at their initial visit to primary health care providers tended to leave those interactions with a greater sense of confidence that their providers would provide high-quality care compared to patients who did not have such histories taken (Lewis and Freeman, 1987). It has been suggested that simulated patients be used to improve clinician skills in risk assessment and counseling (Rabin et al., 1994).
Dedicated Public STD Clinics
The earliest public STD clinics were established in the 1910s, despite substantial resistance by organized medical societies (Brandt, 1985). The concept of dedicated public STD clinics is based on evidence that many persons with STDs prefer anonymous and confidential services, cannot afford to obtain care elsewhere, and are unable to obtain care from private sector health care professionals who are unable or unwilling to provide STD care. These clinics are often seen as the "safety net" for STD-related services. Historically, the stigma associated with having a disease associated with sexual intercourse has discouraged more universal use of public STD clinics and prompt health-seeking behavior for symptoms of STDs in general (Brandt, 1985). Public STD clinics and HIV programs provide the largest proportion of specialized STD-related care in the United States. Various government agencies support STD prevention activities by providing funds, setting standards, or by directly providing care. Public STD clinics usually receive a combination of federal, state, and local funds. The only federal agency that supports dedicated public STD clinics is the CDC, which primarily funds patient education, partner notification, outreach, and other prevention services rather than direct clinical services. State and local health departments also provide financial support for these clinics and programs and are often given responsibility for operating the clinics under federal policies and guidelines.
Persons Served
A recent five-center survey of more than 2,500 patients attending dedicated public STD clinics in the United States showed that users of such clinics are generally young (38 percent under 25 years of age), disproportionately of certain racial or ethnic groups (49 percent African American), and at high risk for multiple STDs (Celum et al., 1995). Approximately 15 to 20 percent of patients attending these clinics are adolescents; the median age of patients attending these clinics is approximately 23 years. The clinics generally provide care for approximately twice as many men as women. Persons who use dedicated public STD clinics tend to have a high prevalence of other health problems, including HIV infection, unintended pregnancy, and drug and alcohol use (Kassler et al., 1994; Zenilman et al., 1994; Weinstock et al., 1995). For example, in one inner-city public STD clinic, 46 percent of women attending the clinic were not using contraception and two-thirds had at least one prior pregnancy (Upchurch et al., 1987).
A significant proportion of dedicated public STD clinic patients have private insurance coverage. In the survey by Celum and others (1995) mentioned above, approximately 31 percent of male and 24 percent of female patients seen in dedicated public STD clinics had private health insurance (Figure 5-1). These data suggest that a large number of privately insured patients use public STD
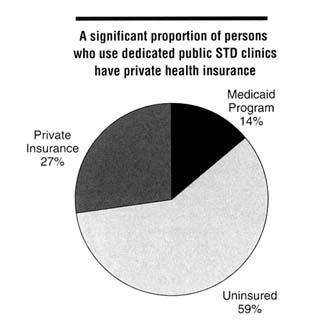
Figure 5-1
Distribution of health insurance status among persons using public STD clinics, 1995. SOURCE: Celum CL, Hook EW, Bolan GA, Spauding CD, Leon P, Henry KW, et al. Where would clients seek care for STD services under health care reform? Results of a STD client survey from five clinics. Eleventh Meeting of the International Society for STD Research, August 27-30, 1995, New Orleans, LA [abstract no. 101].
clinics without acknowledging their health insurance status. In such situations, the local health department ends up paying the cost of the services (Gary Richwald, Los Angeles County STD Program, personal communication, November 1995).
Patients may be referred to public STD clinics by health care providers who either have made a diagnosis requiring treatment or feel that the STD can be better managed by health care providers in public STD clinics. Reasons cited by clinic patients for seeking medical care included genitourinary symptoms (55-70 percent of individuals); notification of recent sexual contact with a partner diagnosed with an STD (15-20 percent); and perceived risk and desire for STD screening (approximately 20 percent) (Celum et al., 1995).
Services Provided
Publicly funded STD-related services are provided both by dedicated public STD clinics and within the context of primary care by community-based programs. Dedicated public STD clinics are located in every state, every major city,
and the majority of smaller cities and counties throughout the United States. Based on published data, the committee's interactions with other health professionals, site visits, results of site assessments conducted by the CDC, and personal experience working with dedicated public STD clinics, the quality of care, scope of services provided, and other characteristics of these clinics are quite variable. Some clinics, commonly those affiliated with academic institutions, seem to offer comprehensive, high-quality STD-related services, whereas other clinics do not provide either comprehensive or high-quality care. In addition, the scope and level of services provided by many clinics are limited by available resources. In some locations, these clinics are high-volume, full-time clinics administered by local health departments or in partnership with medical schools. In contrast, in many rural settings and smaller population centers, dedicated public STD clinics are staffed by individuals who have numerous other responsibilities; these clinics may be open only on a part-time basis, sometimes only a few days a week. Most public clinics charge only a nominal fee or have a sliding fee scale for services.
The services provided in dedicated public STD clinics emphasize diagnosis and treatment, and partner notification for a limited number of STDs (Stein, 1996). Much of this diagnostic effort focuses on gonorrhea, nongonococcal urethritis, clinically defined cervicitis, pelvic inflammatory disease, and genital ulcer disease (i.e., syphilis, chancroid, and genital herpes). These clinics often conduct STD screening for gonorrhea, syphilis, or, more recently, chlamydial infection. Voluntary HIV counseling and testing, which may be offered either in the context of an STD evaluation or as a "stand-alone" service, is offered at most, but not all, clinics.
While there has been increasing interest in, and emphasis on, counseling and health education in dedicated public STD clinics, providers receive little training in techniques and skills for conducting education or counseling (Lewis et al., 1987; Roter et al., 1990). In the fast-paced environment found in most of these clinics, there is little time allocated for, or little emphasis on, counseling (Stein, 1996). "Disease intervention specialists" are often charged with much of the counseling and health education responsibilities in these facilities, as well as with collection of partner information and partner notification. These staff, as discussed later in this chapter, typically emphasize partner notification responsibilities over patient education activities. In dedicated public STD clinics, partner notification activities are primarily focused on patients with syphilis, HIV infection, and, to a highly variable degree, gonorrhea, chlamydial infection, or pelvic inflammatory disease. Ideally, the process of interviewing index patients to obtain both the names and locations of sex partners begins with counseling and education, but it is unclear how consistently this is done. Little or no counseling is provided in dedicated STD clinics for risk reduction or management of chronic or other incurable viral STDs other than HIV infection. One study found that 28 percent of dedicated public STD clinic patients did not receive any information
regarding prevention during their clinic visit (Roter et al., 1990). Several states mandate counseling of patients, using a prescribed content outline, before to HIV testing; however, there is no method for ensuring that these regulations are followed.
Data on the specific types of STD-related services provided by local health departments through public STD clinics are limited. The Alan Guttmacher Institute, with support from the CDC, however, has recently conducted a survey to provide national estimates describing the STD-related activities of local public health agencies in the United States (Box 5-1).
|
BOX 5-1 STD-Related Services Among Local Health Agencies, 1995 A stratified, random sample of 800 local health departments that were identified as providing STD treatment were sent a questionnaire in September 1995 regarding various characteristics of their programs and policies related to STD-related clinical services. Approximately 77 percent of the eligible agencies responded. Results indicate that 50 percent of 2,888 local health departments provide treatment for STDs. Of these providers, 74 percent integrate STD-related services with HIV/AIDS-related services; 21 percent offer STD- and HIV-related services in separate programs; and 5 percent provide STD-related services but do not provide HIV screening or testing. Almost half (49 percent) of the local health departments that offer services for STDs offer both dedicated STD sessions and sessions where such services are integrated with other services, such as family planning. An additional 37 percent always integrate STD sessions with other clinic services such as family planning, and 14 percent provide only STD-related services in dedicated sessions. Only 23 percent of agencies offered services after 6 p.m. and only 5 percent had weekend hours. Regarding testing and treatment services for chlamydial infection, gonorrhea, and syphilis, a greater percentage of agencies reported treating chlamydial infection (97 percent) than testing for it at all or some sites (82 percent). The percentages of agencies testing and treating for gonorrhea and syphilis at all sites were all over 98 percent. Agencies were also asked to report what type of client history, risk assessment, and educational/counseling services they routinely provide patients making an initial STD visit (Table below). More than 90 percent of agencies reported routinely collecting information on a client's sexual, STD, and contraceptive history. A smaller proportion of agencies routinely query patients regarding any history of substance abuse (78 percent). While approximately 97 percent of STD agencies reported routinely providing educational services regarding risk factors of STDs and HIV, far smaller percentages of agencies reported routinely providing services on how to use contraceptive methods effectively or how to negotiate condom use (66-70 percent). Although more than 70 percent of health departments that provide services for STDs in integrated sessions reported routinely providing education and counseling regarding contraceptive use, less than half (47 percent) of agencies that only provide services in separate sessions provide this service. It should be noted that the survey results represent only health departments that offered treatment for STDs and that the quality and consistency of services provided were not evaluated. In addition, most agencies that reported integration of STD-related services with other services were in nonmetropolitan areas with relatively low caseloads. Distribution of Local Health Agencies Providing STD Risk Assessment and Educational and Counseling Services by Type of Service, 1995
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Complete Box on previous page. |
Effectiveness
Dedicated public STD clinics provide services to large numbers of patients at little or no cost to the patient. One of the guiding principles of these clinics is that no patient should be turned away because of cost considerations. However, it has been estimated that as many as 25 percent of those presenting for care cannot be accommodated because of inadequate clinic capacity (CDC, DSTD/HIVP, 1992). The performance of these clinics is usually evaluated on the basis of quantitative measures, such as numbers of patients seen and number of cases of
specific diseases diagnosed, rather than on quality of care measures. Based on the committee's site visits and personal experience working with dedicated public STD clinics, there is little emphasis on, and almost no reporting of, quality-related indicators such as consistency of risk-reduction counseling or numbers of patients with positive STD screening tests who are successfully treated as opposed to simply having been screened. Systems for evaluation of clinic services tend to be developed in reaction to increasing STD rates or other evidence of perceived failure. This may also be related to the clinics' emphasis on quantitative performance measures. There has been little effort to measure potential positive impact of dedicated STD clinic services on populations using their services. The CDC has not conducted routine on-site quality assessments of public STD clinics and programs since 1993. Federal oversight of quality in such clinics and programs currently consists of a yearly review of written program activities (submitted annually as a requirement for federal funding), periodic telephone and on-site technical assistance consultations, and the work of federal program consultants who are stationed in some project areas.
There are few data regarding the perceptions of care provided to STD clinic patients. In a study by Celum and others (1995), a high proportion of patients attending these clinics stated they would preferentially attend public STD clinics should they need further STD care. The most common reasons cited for preferring to use the public STD clinic were walk-in/same-day appointments, lower costs, privacy or confidentiality concerns, convenient location, and expert care (Figure 5-2). Confidentiality concerns are a primary determinant of whether adolescents seek health care for potential STDs.
Federal Role
As mentioned earlier, nonclinician public health professionals referred to as ''disease intervention specialists" (previously known as "contact tracers") have played a special role within state and local STD programs. These personnel include federal employees assigned as field staff in local programs and state and local government employees. The provision of federal field staff is referred to as "direct [federal] assistance" and "in lieu of cash," as opposed to "financial assistance," which is given to the states through the STD prevention cooperative agreements.
Historically, disease intervention specialist positions served as the entry level for all management staff within federal public health programs. Disease intervention specialists initially began at the lowest federal civil service entry levels as personnel in state or local public health field assignments, largely performing provider referral field work. Eventually, many staff were reassigned to new positions and given supervisory responsibilities within other state and local STD programs. Federal public health advisors are typically recruited back to the CDC
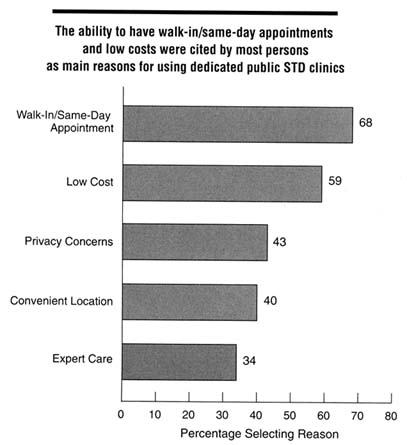
Figure 5-2
Reported main reasons for utilizing public STD clinics among clinic users in five U.S. cities, 1995. Categories are not mutually exclusive; respondents were allowed to indicate up to three reasons. SOURCE: Celum CL, Hook EW, Bolan GA, Spauding CD, Leone P, Henry KW, et al. Where would clients seek care for STD services under health care reform? Results of a STD client survey from five clinics. Eleventh Meeting of the International Society for STD Research, August 27-30, 1995; New Orleans, LA [abstract no. 101].
and to other divisions outside of the STD division and seem to have formed a useful managerial infrastructure for the agency.
Many problems existed with the system of management training for federal staff. The mixing of state, local, and federal staff often created conflict in local areas because federal salaries were higher than most local and state salaries, leading to staff resentment. Since management positions were often given to
these transient federal assignees, local staff felt that promotional opportunities were hindered by the presence of federal staff, and they questioned the loyalties of the assignees to state or local programs. Because the assignees were part of the federal cooperative agreement, many states and local areas depended, in part or fully on, federal support to maintain core program functions. This federal training program may have inhibited the development of local and state STD program capacity, because states became dependent on federal assignees to fill key service delivery and program management positions, thereby limiting the development and training of local or state staff.
As a result of these problems and the feeling that disease intervention specialist training was too narrow and not flexible enough to adapt to the future role of public health, recruitment and initial training of CDC federal nonphysician field assignees are being restructured in ways currently being defined (CDC, 1995a). CDC is initiating a transitional training program for current disease intervention specialists and has begun to reduce their total number. Thus, the total number of federal field assignees for STD prevention will be reduced by nearly half from 1996 through 2000, but remaining assignees will receive further training and new recruits will have more extensive training. Transition from a direct service delivery role to a technical assistance and local capacity-building role for federal assignees is being planned (CDC, DSTDP, 1996). The CDC is currently developing criteria for evaluating state and local government requests for replacement of federal assignees and conversion of direct assistance funds to financial assistance funds. As a result of federal downsizing, in most cases, direct assistance vacancies will not be filled on a one-to-one basis and requests for converting direct assistance to financial assistance will not result in a dollar-for-dollar conversion.
A major concern of STD program managers is that the former federal disease intervention specialists, whose number is now being reduced, have represented significant support for many STD programs and have served in key STD program management positions. Federal assignees from CDC to state and local governments have decreased in number from 1993 through 1996 as a result of the CDC downsizing program and the freeze on hiring for new positions. This resulted in closing the four training programs for new disease investigation specialists, so that the greatest decrease in federal assignees occurred in Florida, Georgia, the city of Chicago, and California, where these training programs and positions were located. These federal positions have not been replaced by reciprocal federal or state resources to hire or contract for replacement disease intervention specialists or management staff. No state or county resources are currently available to replace these positions, nor could they be used, even if available, where states are currently experiencing a hiring freeze. In essence, a major portion of the federal support that has been provided as direct assignment of disease investigation specialists is being redirected to states for other types of disease control activities (e.g., chlamydial prevention, training, and local recruitment). This may
potentially leave state and local STD programs, many in high morbidity areas such as the South, at least temporarily without the resources to conduct adequate STD surveillance and program management activities. To maintain local capacity, it will be essential for state and local governments to use both federal financial assistance and their own resources to develop local capacity as the number of federal assignees is reduced, and not simply withdraw state and local funds as federal financial assistance is received. There is no evidence that initiatives to increase funding from local sources will generate adequate resources to replace federal positions being withdrawn from local agencies.
In an effort to expand prevention efforts beyond those delivered through public STD clinics, the CDC launched the STD Accelerated Prevention Campaigns grant program for selected states and local health departments in 1994 (Noegel et al., 1993). The program seeks to (a) increase links between STD clinic activities and other health programs and community-based programs, (b) promote innovative approaches to STD prevention, (c) encourage commitment of local resources to prevention, and (d) develop cost-effective methods of prevention. Activities funded by the campaign are encouraged to focus on populations that are disproportionately impacted by STDs, including women, infants and adolescents, and certain racial and ethnic groups.
Community-Based Clinics
Many types of community-based clinics, such as family planning clinics, prenatal clinics, youth and teen clinics, homeless programs, community-based health centers, and school-based clinics, also provide STD-related services. Like dedicated public STD clinics, community-based clinics that treat STDs tend to be high-volume clinics that provide services at relatively little or no cost to the patient. STDs are not the primary focus for these clinics, but rather are dealt with in a context of providing general or specific (e.g., family planning) health care services.
Although the populations served by community-based clinics overlap substantially with public STD clinic patients, there is surprisingly little communication between these facilities. Similar to dedicated public STD clinics, community-based clinics generally serve young patients of certain ethnic and racial groups from lower socioeconomic class communities. A major difference between persons attending community-based clinics and those attending dedicated public STD clinics, however, is that some persons attending community-based clinics rely on these clinics for regular health care, that is, they attend on a scheduled basis rather than the episodic, problem-oriented basis that characterizes utilization of dedicated public STD clinics.
Even more so than for dedicated public STD clinics, the scope of STD-related clinical services in these community-based clinics is highly variable. These clinics identify proportionally more STDs through screening activities
than by evaluating patients with acute signs or symptoms of STDs. In the past, gonorrhea screening has been funded on the basis of availability of government funds. More recently, publicly funded family planning clinics have become the focus of a national initiative on preventing chlamydial infection coordinated by the CDC. Unlike dedicated public STD clinics that provide services to a disproportionate number of men, most persons who use community-based clinics are women and children. In fact, some family planning clinics will not provide services to men even if they are sex partners of infected women seen in their facility. STD-related clinical services are often provided in the context of other regular health care, and there is often little emphasis on partner notification and treatment as part of care for infected women. These clinics presumably have a strong investment and interest in issues of STD counseling and health education. Federal funds for STD diagnosis and treatment in community-based clinic settings are often restricted to specific uses.
A 1990 survey conducted by the State Family Planning Administrators collected data regarding STD-related services in 410 Title X2 family planning clinics nationwide (SFPA, 1991). The survey showed that most family planning clinics provided STD-related services to their clients, but the scope of services varied considerably. For example, 82 percent of clinics reported capability for treating gonorrhea, but only 48 percent provided treatment for syphilis. Approximately one-third of clinics reported using staff resources to contact partners for at least one STD, and approximately 60 percent of clinics provided testing for gonorrhea. Virtually all clinics provided some preventive services, and more than 80 percent reported conducting community-based education activities. Half of the clinics surveyed shared family planning and local STD program staff in integrated service settings. Family planning clinics throughout the country have implemented special programs to reach disenfranchised populations, including substance users, inmates, the homeless, disabled persons, and non-English-speaking populations (Armstrong et al., 1992; Donovan, 1996).
Data regarding STD-related services in community-based clinics such as community health centers and clinics for the homeless and migrant workers are more limited than for family planning clinics. This is a result of the failure to collect STD-service-specific data, since such services are often provided as an integral part of primary care. However, in a 1994 survey of Health Care for the Homeless programs (Section 340 of the Public Health Services Act), 68 percent of responding programs offered screening and 67 percent offered STD treatment services directly (UCLA Center for Health Policy Research, unpublished data, 1994). The remainder offered services through parent agencies, under contracts, or did not offer services.
School- and University-Based Settings
School-based health clinics in elementary and high schools and student health services on university and college campuses often provide STD-related services for their students. The number of school-based health centers has dramatically increased in recent years, from 40 in 1985 to 607 in 1994, in an effort to improve access to primary health services for children (Schlitt et al., 1995). The School Health Services and Policies Study examined school health services and HIV infection policies on a state and school district basis nationwide in 1994 (Leavy Small, et al., 1995). Approximately 60 percent of states fund school-based or -linked clinics, and 12 percent of all school districts have at least one school-based or -linked clinic. Diagnostic and treatment services for STDs were available for 16 percent of all middle and junior high schools and for 20 percent of all senior high schools. School health services are supported in part by state funds and in part by federal funds, primarily through the CDC, the Health Care Financing Administration, and the Health Resources and Services Administration (Leavy Small et al., 1995; Schlitt et al., 1995). Most school-based clinic services are provided by registered school health nurses (Igoe, 1994). For example, in one study, 31 percent of adolescents surveyed cited fear of parental discovery as their reason for not utilizing available health services at family planning clinics (Zabin et al., 1991).
Students at universities and colleges are at high risk for STDs (DeBuono et al., 1990; Reinisch et al., 1995). Recent national data on the scope and quality of STD-related clinical services among university health services are not available. Anecdotal evidence, however, suggests that university student health services at a minimum provide confidential STD diagnosis and treatment services to students (Cindy Launchbaugh, American College Health Association, personal communication, April 1996). Several universities have published descriptions of prevention activities for STDs that generally focus on HIV prevention (McLean, 1994; Turner et al, 1994; Keeling, 1995). Several have shown that specific interventions were successful in improving knowledge and promoting safer sex behavior (McLean, 1994; Turner et al., 1994).
Private Sector Settings
Many private sector health care providers, including private physician offices, health plans, private clinics, and private hospital emergency rooms, provide some STD-related services. Most cases of STDs are diagnosed by private sector health care professionals (Berg, 1990). However, very little is known about the volume, extent, disease prevalence, or spectrum of STDs encountered in private sector settings compared to either dedicated public STD clinics or community-based clinics. Accurate estimates of STD morbidity seen in private sector settings are lacking as a result of underreporting from clinicians in these settings. Nonetheless,
available data suggest that the patterns of diseases seen in these settings may be quite different from those seen in public clinics. In 1994, while public STD clinics reported 1.93 times more syphilis than all non-STD reporting sites (including private sector providers and community-based facilities), non-STD clinic sites reported 1.93 times the number of chlamydial infections (CDC, DSTDP, 1995).
In addition to the lack of disease surveillance data from private sector settings, there is limited information regarding the distribution and types of care and the costs and expenditures for STD diagnosis and management in these settings. Undoubtedly, patients with acute STD syndromes may be seen in emergency rooms as well as by private practitioners or other clinics. However, the distribution and allocation of patients among these services are unknown. Similarly, how often and how well patients are screened for STDs is unknown, although the prevailing opinion is that screening for STDs is relatively uncommon in the private sector. For example, a survey of 19 hospital-based emergency centers in Los Angeles County revealed that only 5 implemented a policy for cervical cancer screening (Marcus et al., 1990).
There are few data regarding what proportion of patients seen in private settings are given recommended therapy for STDs or whether partner notification and treatment practices are routinely conducted (Winkenwerder et al., 1993; Celum et al., 1995). One recent study provides some information regarding compliance of primary care physicians in California with the CDC recommendations regarding the management of pelvic inflammatory disease (Hessol et al., 1996). Of 553 physicians responding, 55 percent reported treating at least one case of pelvic inflammatory disease during the previous 12-month period, and of these physicians, 52 percent were either unsure of or did not follow the CDC's treatment guidelines for this STD. Partner notification is not well supported in private sector settings, probably because most private sector clinicians do not accept responsibility for partner notification; there is no reimbursement for care of sex partners; and providers may be reluctant or not trained to interview their patients regarding sexual practices.
Most private practitioners emphasize acute care and provide screening when mandated by standards of practice, but, as discussed previously, most clinicians do not routinely conduct STD risk assessment and many do not provide counseling for behavior change (Lewis and Freeman, 1987; Boekeloo et al., 1991; ARHP and NANPRH, 1995). Complicating private practitioners' management of STDs is that, although there are national treatment guidelines for STDs (CDC, 1993) and practice guidelines for STD clinics (CDC, 1991), there are no generally accepted clinical practice guidelines or standards for STD screening and risk assessment. In addition, private practitioners generally are ill-prepared to assess their patients' risks, educate and counsel them, or notify and treat their sex partners.
Managed Care Organizations and Other Health Plans
Managed care is a method of integrating the organization and financing of health care services.3 There are various approaches to managed care, including management of care under fee-for-service provider reimbursement (e.g., a preferred provider organization) and primary care case management. Prepayment places the health plan "at risk" financially because it must provide contract services in return for only the monthly premium and any nominal copayments allowed by the contract. This at-risk arrangement imposes financial incentives to control or "manage" the use of health services. Increasingly, managed care organizations are shifting some or all of this financial risk downward through capitation contracts with providers, who receive a fixed monthly sum for each member and earn their retained surplus or income by efficiently managing the clinical care. Most managed care organizations utilize a selected network of providers to help control both cost and utilization of services, thus potentially reducing access for enrolled members to STD-related services. Many plans require all services to be provided or arranged through a primary care clinician, often referred to as a "gatekeeper" or "care manager." The combination of financial incentives to control cost, including capitation, and the reliance on managing access through primary care providers places greater responsibility on managed care organizations than on traditional fee-for-service plans to ensure that beneficiaries receive comprehensive health services.
There are several types of managed care organizations, and more are evolving. Managed care organizations are usually classified by their method of providing services. Staff-model organizations employ providers on salary; group-model organizations contract with a single medical group practice; network-model organizations engage two or more medical groups; and independent practice associations (IPAs) contract with individual physicians or organized associations of independent physicians. Many managed care organizations now offer several of these options and are called mixed, or hybrid, plans. In each case, the contract can provide for payment to the provider in the form of a capitation (fixed monthly fee per member) or a discounted fee-for-service schedule. Point-of-service plans allow services from network providers at the usual copayment, but also permit
members to seek care from any provider, in which case they pay deductibles and copayments similar to traditional indemnity insurance.
The early prepaid group- and staff-model managed care organizations (e.g., Group Health Cooperative of Puget Sound, Harvard Pilgrim Health Care, Kaiser-Permanent Health Plan) are nonprofit. The greatest growth in recent years, however, has been among investor-owned for-profit plans, which are mostly independent practice associations or hybrid plans (e.g., Aetna U.S. Healthcare, United Health Care). A few managed care organizations, such as the Los Angeles County Department of Health Services' Community Health Plan and the Contra Costa County (California) Health Plan, are publicly owned and operated by local health departments. In 1994, approximately 31 percent of persons enrolled in managed care organizations were in predominantly staffor group-model organizations, and 69 percent were in network or independent practice association plans (GHAA, 1995).
For-profit managed care organizations are managed somewhat differently from most not-for-profit managed care organizations, and quite differently from publicly operated managed care organizations. Investor-owned managed care organizations are managed to generate profits and increased equity for shareholders. Not-for-profit managed care organizations must reinvest any excess revenue in the organization and provide some type of benefit for plan members. A 1986 IOM report found that not-for-profit health care organizations were more likely than for-profit ones to provide care to uninsured persons and to conduct research and educational activities (IOM, 1986). Publicly operated managed care organizations are usually formed to meet the needs of Medicaid beneficiaries and uninsured residents for whom a local health department has responsibility.
Many managed care organizations have recently formed partnerships or entered into contracts with other community health care providers. For example, several states require or encourage school-based health centers to develop agreements with managed care organizations to improve primary care services for children (Schlitt et al., 1995). Several contractual models for such agreements have been implemented (Zimmerman and Reif, 1995).
Growing Role of Managed Care
Managed care organizations have grown rapidly in the last 10 years; nationwide enrollment in managed care organizations increased from 6 million in 1976 to 51 million in 1994 (GHAA, 1995). These health plans now provide health services to more than 20 percent of all privately insured persons in the United States. In 1994, more than 60 percent of the employed and insured under 65 years of age in 14 major metropolitan areas were enrolled in a managed care organization. As more people become dependent on the services provided only through their health plan, local public health leaders have expressed concerns regarding the effects on the scope, accessibility, and quality of services traditionally provided
by, or in conjunction with, public health agencies, such as STD-related services.
One concern voiced by advocates for the poor is the rapid pace at which states are converting their Medicaid programs to prepaid managed care plans, thereby enrolling a population at higher risk for STDs into health plans with little experience managing STDs. For example, as of January 1996, 12 states have statewide managed care programs for Medicaid beneficiaries under 1115 waivers, and 47 states have more limited 1915(b) waivers from the Health Care Financing Agency (National Governors' Association, unpublished data, 1996).4 In 1994, approximately 7.8 million Medicaid beneficiaries, or 23 percent of all enrollees, were enrolled in managed care plans (The Kaiser Commission on the Future of Medicaid, 1995). The push by states to enroll Medicaid recipients into managed care plans in order to contain costs raises several concerns regarding the design and implementation of these new programs and the accountability of the new managed care plans for quality (Fisher, 1994).
Effect of Managed Care on STD Prevention
There are both opportunities and concerns associated with increased involvement of managed care organizations in the delivery of preventive and public health services (CDC, 1995b). Potential opportunities for managed care organizations to improve prevention of STDs include the following:
- The prepayment arrangement of managed care organizations provides a potential incentive to effectively prevent STDs among enrollees. They are directly responsible for providing their enrollees with comprehensive personal health services, which include care for STDs and their complications.
- The organizational structure of managed care organizations as a system for providing care increases opportunities for effectiveness. For example, group-and staff-model organizations could effectively implement guidelines for STD-related screening and case management across all clinicians in their plan. Less-structured managed care organizations, however, such as independent practice associations, would have more difficulty in consistently implementing guidelines.
- Managed care organizations, because they are responsible for delivering
- care to large numbers of plan members, may be more inclined than traditional fee-for-service providers to view their enrollees from a public health perspective, that is, to examine and address their enrollees' health needs as a community or population, rather than as individuals.
- The large size, resources, and complexity of group- and staff-model managed care organizations potentially allow them to support the services of highly trained health professionals, such as physicians trained in infectious diseases, to manage and provide STD-related services.
- The highly developed information systems of many managed care organizations can be used to monitor STD trends among their enrolled population and to assess the quality and adequacy of treatment and case management protocols. These information systems are more common among groupand staff-model organizations than among independent practice associations and network plans.
- Managed care provides opportunities for purchasers, such as employers and government programs, to hold plans accountable for specific performance standards. This is a tremendous potential opportunity to provide and improve STD-related services if purchasers take a strong interest in this issue. Information systems can also help purchasers measure the performance of managed care organizations in STD-related activities through standardized quality assessment tools, such as the Health Plan Employer Data Information Set (HEDIS),5 if such performance measures include measures of STD-related services.
The potential concerns regarding the increased role of managed care organizations in STD prevention include the following:
- STDs are not a high priority for most managed care organizations or their sponsors. Those that do not serve populations at high risk for STDs, in particular, may not have a strong interest in providing comprehensive STD-related services.
- The mission of the managed care organization is often related to whether the managed care organization is a nonprofit or for-profit organization. The mission of for-profit organizations may be in conflict with providing services that do not provide short-term cost savings to the organization.
- There is a wide spectrum of managed care organizations; consequently, there is a wide range of technical ability among such organizations in delivering services. In general, staff- and group-model managed care organizations are likely to be more effective in STD prevention than health plans that have less infrastructure. Given the limited experience of most managed care organizations in public health activities, however, even some of the best organized managed care organizations
- may not have the technical expertise to take on full responsibility for STD prevention. In addition, the types of managed care organizations that currently dominate the market typically do not have highly developed systems for ensuring quality care.
- Many managed care organizations may be reluctant to provide STD-related services that have not been shown to be cost-saving for the organization. For example, the long interval between infection and appearance of consequences of STDs may be years; managed care organizations with high turnover rates may have little incentive to emphasize STD-related services. In addition, capitated payments for services may increase the risk of cost-shifting by managed care organizations. For example, health plans may refer persons in need of STD-related services to public STD clinics to avoid assuming the costs of their care.
- Persons with STDs may prefer to receive care at public STD clinics and may not feel comfortable receiving care through a managed care organization for a variety of reasons. A recent multisite survey of STD clinic patients showed that most persons surveyed chose a public STD clinic over other providers because of the convenience of obtaining care without an appointment and lower costs (Celum et al., 1995). The lack of walk-in services among many managed care organizations may result in delays for evaluation and treatment of STDs.
- Managed care organizations may not provide services to sex partners of plan members if the partner is not a plan member. Many aspects of STD prevention, such as partner notification, screening and case finding, and community education, may involve persons who are not members of the managed care organization.
The billing and claims-processing procedures of some health plans may be a major barrier to confidential STD-related services, particularly for dependent minors. In approximately one-third of managed care organizations surveyed, the employee-beneficiary is likely to be notified of care for their dependents through a copayment bill or other means (Benson Gold and Richards, 1996). Of particular concern are billing procedures among traditional indemnity insurance plans, preferred provider organizations, and point-of-service networks, which often result in lack of confidentiality for dependents because the employee-beneficiary is usually required to be involved in the claims process.
Data regarding the impact of managed care on STD-related services are limited. One study examined the effect of managed care enrollment on the management of three ambulatory conditions (vaginitis, pelvic inflammatory disease, and urinary tract infection) among Medicaid recipients (Carey and Weis, 1990). The authors found that the presence of managed care plans did not reduce diagnostic testing or return visits for the three conditions, compared to fee-for-service providers. Many of the potential concerns mentioned above regarding expanding the role of managed care in STD prevention are similar to those associated with
managed care and other controversial health services, such as reproductive health (Delbanco and Smith, 1995) and HIV infection (Aseltyne et al., 1995).
Services and Programs of Managed Care Organizations
Studies show that most managed care organizations provide considerably more comprehensive coverage for reproductive health services than the traditional indemnity insurance plans (Benson Gold and Richards, 1996). Data regarding STD-related services provided or covered by health plans are limited.
Given the lack of published information regarding the potential role of managed care organizations in STD prevention, the committee sponsored a workshop on the role of managed care organizations in the prevention of STDs. A summary of this workshop can be found in Appendix G.
As a follow-up to the workshop, the committee conducted a limited survey of managed care organizations to collect preliminary data regarding STD-related services and programs in these organizations. Results of this survey are presented in Appendix H. Managed care organizations were selected primarily on the basis of their likelihood of serving high-risk populations (i.e., Medicaid, inner city), and therefore their increased likelihood of providing STD-related services. The committee found that 73 percent of responding managed care organizations requested information regarding previous history of STDs or sexual activity, and 65 percent requested information on sexual activity, on patient history forms. More than half (57 percent) of these organizations attempted to define high-risk groups for STDs, and approximately half reported STD prevention or clinical activities that specifically targeted adolescents. Only 26 percent of managed care organizations reported that they provided STD-related services to persons outside their plan, and just 17 percent had a specific individual in charge of STD-related activities.
Examples of programs and activities conducted by survey respondents are provided in Appendix H. Two examples of managed care organizations that have comprehensive prevention and case management programs that extend beyond the boundaries of the plan's enrollees are the Harvard Pilgrim Health Plan and Kaiser-Permanent of Southern California. At the Harvard Pilgrim Health Plan, all levels of services for members with HIV infection and AIDS have been integrated through a central multidisciplinary program. This program is closely linked to local health department services and provides extensive outreach and community services beyond the member population. In Southern California, Kaiser-Permanent has developed an effective system of STD-related services under the direction of an infectious disease specialist; the system includes monitoring of STD trends through reports from its centralized laboratory, implementation of STD treatment and case management protocols among its health professionals, and creation of a prevention and primary care program for STDs that is targeted to adolescents in the plan and in the general community. STD programs such as
those mentioned above may have a dramatic impact on reducing STDs and associated costs in managed care organizations. For example, a recent randomized control study conducted at Group Health Cooperative of Puget Sound showed that identifying, testing, and treating women at increased risk for asymptomatic chlamydial infection reduced the rate of pelvic inflammatory disease by more than 50 percent compared to women who received routine care (Scholes et al., 1996).
Government Initiatives Related to managed Care and STDs
Public health agencies, such as the CDC, have been exploring the impact of managed care on public health services. The CDC recently established a Managed Care Working Group to foster partnerships between public health agencies and managed care organizations to improve public health (CDC, 1995b). The CDC's high-priority areas for collaborative activities with managed care organizations and other health organizations include prevention effectiveness and guidelines, Medicaid and managed care, research, and capacity development in public health agencies. In addition, a CDC epidemiologist is currently assigned on detail to the American Association of Health Plans as a resource on public health issues. As mentioned previously, CDC staff have provided input regarding public health performance indicators, including STD-related indicators, for future versions of HEDIS.
The California Department of Health Services has also recently initiated the California Partnership for Adolescent Chlamydial Prevention. This is a statewide partnership, bringing together government agencies, managed care organizations, academic health centers, and professional associations to address policy issues related to STDs among adolescents. This initiative also seeks to coordinate clinical preventive services for adolescents in managed care settings with community STD prevention activities and to coordinate all categorical state STD-related programs. Other components of this initiative include a media campaign targeted towards teenagers; development of screening, counseling, and education interventions; school-based programs; and training programs for health care providers.
The Los Angeles County Department of Health Services has developed a model contract between the agency and managed care organizations that contract under the Medical (California's Medicaid program) program (County of Los Angeles, Department of Health Services, 1995). The contract, which covers a wide range of public health services, describes STD clinical services as a ''shared responsibility between County and Plan." The contract would require managed care organizations participating in Medical to "reimburse the County for services provided to Plan beneficiaries at the Medical [fee-for-service] rate." In turn, the county agrees to "make all reasonable efforts to provide medical records to the Plan relating to STD care billed to the Plan." The county's contract language
parallels language required by the California Department of Health Services for all contracts with managed care organizations serving Medical beneficiaries. Similar provisions in Medicaid contracts have also been adopted by Missouri, Oregon, and Minnesota. Although such an agreement appropriately would provide for plan reimbursement for out-of-plan STD-related services, better methods are needed to document billing of the plan for their enrollees' use of such services. The language included in the Los Angeles County model contract suggests that the health department will divulge confidential patient information to the plan and may inhibit infected persons from seeking care-an outcome that would not be in the best interests of the plan or the larger community.
National Surveillance and Information Systems
Public health surveillance is the process of collecting information regarding the frequency and distribution of disease or other health conditions among specific populations. Data are collected at local, regional, and national levels to (a) monitor trends in diseases and other health conditions, (b) identify problems that need intervention, (c) improve effectiveness of prevention or health care resources, and (d) evaluate the impact of specific interventions. The current national system for STD surveillance is a complex amalgam of different reporting systems from multiple sources.
National Notifiable Disease Reporting
The foundation of STD surveillance is the national public health notifiable disease reporting system, coordinated on a national basis by the CDC. This system is fundamentally a "passive" system, that is, reports are brought to the attention of public health officials when health care providers or laboratories submit a report of a positive laboratory test or clinical diagnosis of a reportable health condition. Active case finding is not routinely conducted. Each of the 50 states has the authority to declare certain diseases or health conditions that are to be reported by clinicians and laboratories. Syphilis and gonorrhea are reportable conditions in all 50 states; and 48 states (New York and Alaska are the exceptions) also require the reporting of chlamydial genital infection (CDC, 1995c). Other bacterial STDs, such as chancroid or lymphogranuloma venereum, are not consistently reported from state to state. Viral STDs, such as herpes simplex virus type 2 infections, are generally not well identified through this system.
In general, initial case reports generated by laboratories or providers are submitted to local public health departments and are, in turn, reported to state public health authorities. All states participate in a voluntary system whereby statewide notifiable disease data are reported to the CDC. National STD data reported by the CDC are compiled from individual reports from states, submitted without name identifiers. Not all states report uniform data to the CDC. For
example, California does not report STD morbidity data by race but includes other variables.
Limitations of the Reporting System
The passive reporting system for STDs has several major limitations. Numerous studies have indicated that reporting from public clinics, such as public STD clinics, is more complete than reporting from private providers (Anderson et al., 1994). Reported data actually may underrepresent true STD incidence by 50 percent or more because many cases diagnosed by private providers are not reported. The reporting bias toward public sector providers also skews the demographic characteristics of reported STD rates, since public providers are more likely to see poor or uninsured patients, a greater proportion of whom are from certain racial and ethnic groups (CDC, DSTD/HIVP, 1995). In a comparison of surveillance data collected by the CDC with survey data collected by population-based surveys (i.e., National Survey of Family Growth), one study found that national surveillance data may underrepresent the incidence of STDs among higher socioeconomic groups because they tend to use private sources of health care, whereas survey data may be prone to underreporting of past STDs (Anderson et al., 1994).
Analysis of national data by the CDC requires considerable effort to produce basic reports and often does not systematically assess trends within subpopulations and geographic areas. For example, the increase in heterosexual syphilis that peaked in the late 1980s was not identified in a timely way because national data were aggregated and not systematically analyzed by sex and risk behavior. This was because the decline in syphilis rates among men who have sex with men obscured the increase in syphilis among heterosexual men and women until a year or more into the epidemic.
Since surveillance data are only compiled as diagnosed cases, rather than number of cases per number of persons tested, such estimates are difficult to interpret. This is especially problematic when new diseases are added to the list of notifiable diseases or when new diagnostic technologies become available. This phenomenon occurred most recently with chlamydial genital infection. As more providers became aware of the availability of culture and nonculture diagnostic tests, screening for chlamydial infection among women increased in many health care settings. Reported statistics demonstrated dramatic increases in chlamydial genital infection that reflected increased screening rates rather than true rising disease incidence rates. Similarly, case-definition bias may also confuse interpretation of surveillance statistics. For example, women may be more likely to be tested and identified as laboratory-confirmed cases, whereas men with nongonococcal urethritis will be presumptively treated (treated after a syndromic diagnosis) and never reported as a case, since they were not confirmed by laboratory tests. Furthermore, should nongonococcal urethritis become reportable,
this will only partially reflect chlamydial infection among men because, although chlamydial is the most common cause of nongonococcal urethritis, it still accounts for less than half the cases.
Other limitations in these data may not be immediately evident. Reporting to public health agencies is sometimes not carried out in a timely way, so that delays in reporting may result. In addition, surveillance case definitions are often not uniformly followed, leading to misclassification of cases or nonreporting of true cases. This occurred in many areas when the congenital syphilis case definition changed in 1989, but local areas continued to follow the old case definition, thereby severely underestimating the true impact of syphilis on infant health.
Surveillance and Public STD Programs
Publicly funded STD programs, particularly in urban areas, have largely focused on case-finding and partner notification activities rather than development of infrastructure for data management and surveillance data analyses. In areas where syphilis rates remain high (largely in urban areas and in the South), programmatic activity by disease intervention specialists has focused on following up positive laboratory tests for syphilis with tracking and testing of sex partners. In areas of lower syphilis morbidity, this intense outreach program by disease intervention specialists was carried out for other STDs, such as gonorrhea. In all cases, efforts were largely focused on cases identified in public STD clinics, and mechanisms were not developed to uniformly assess the extent of STD diagnosis and treatment in nonpublic STD clinic settings or through private providers. In some STD programs, so little effort is being made to identify gonorrhea or chlamydial infections identified outside publicly funded STD clinics that reports sent to the local health departments from non-STD clinic providers are discarded, and little effort is made to identify broader, community-based morbidity trends. As discussed in Chapter 4, partner notification and follow-up of positive laboratory results for syphilis are labor intensive and result in a heavy emphasis on STD program indicators rather than on true population-based measures of program effectiveness.
Other Surveillance Systems
Other types of data regarding STDs are collected through a multitude of systems, some clinic-based, some local or regional, and others national in scope. Coupled with the national notifiable disease surveillance system, they provide a patchwork of information for inferring the actual scope and impact of STDs nationwide.
Rather than attempting to capture data from all data collection sites, sentinel systems are designed to capture detailed data from a few sites that are considered to be representative of the region or the country. Sentinel surveillance can address
the biases from increased screening practices or inconsistent application of case definitions, since the populations under surveillance and participating providers are well defined. Perhaps the best functioning sentinel STD surveillance system is the Gonococcal Isolate Surveillance Project sponsored by the CDC. This project collects data from 21 nationwide sites to assess patterns in gonorrhea isolate antimicrobial resistance.
Coordinated by the National Center for Health Statistics, the National Health and Nutrition Examination Survey (NHANES) collects health-related data from a randomly selected sample of the U.S. population. In addition to data regarding self-reported health behaviors, blood samples collected at the time of interview provide critical information regarding the actual prevalence of diseases through detection of serological markers. This is particularly useful for some viral STDs that are often asymptomatic, including herpes simplex virus infection, hepatitis B virus infection, and HIV infection (McQuillan et al., 1989, 1994; Johnson et al., 1993). For example, using these data, public health officials have estimated that at least one of four European American women and one in five European American men will be infected with genital herpes in their lifetime (Johnson et al., 1993).
Provider-based information systems are used to estimate the scope and frequency of treatment among private sector and other physicians and can provide useful data on STDs to supplement disease surveillance data. The National Disease and Therapeutic Index is a commercially developed provider database that systematically collects patient encounter data from a stratified sample of U.S. private practice physicians. It has been used to follow trends in the diagnosis of some nonreportable STDs, such as genital warts caused by human papillomavirus, genital herpes, vaginal infections, and nongonococcal urethritis. Another potential method for assessing STD trends is by monitoring of the sequelae of STDs. For example, the National Ambulatory Care Survey and the National Hospital Discharge Survey have been used to monitor rates of pelvic inflammatory disease. Physician consultations for infertility and cervical cancer rates within specific populations also may be used as surrogates for underlying STD trends. In addition, disease registries are helpful in documenting the occurrence of STDs in specific communities and in conducting clinical research. For example, a national disease registry collects information regarding infants exposed to herpes simplex at birth and treated with acyclovir. Although limited in scope, these registries provide useful information on treatment of STDs.
Health Behavior Surveys
A crucial but underdeveloped tool for directing and targeting STD prevention programs is the behavioral health survey. Surveys that regularly collect nationally representative information on specific STD-related risk behaviors are important in monitoring national trends and have become more prominent since
the advent of the HIV epidemic. The Behavioral Risk Factors Surveillance Survey supported by the CDC was originally developed to ascertain health behaviors related to chronic diseases such as heart disease, but has been modified to include questions regarding HIV/STD-related risk behaviors. A similar survey, the Youth Risk Behavior Surveillance System, designed to determine risk behaviors of teenagers, including sexual activity and alcohol and other drug use, has been developed by the CDC and implemented by schools nationwide. This survey is perhaps the best currently available source of information on the scope and frequency of STD-related behaviors among teenagers and is commonly used to develop effective prevention programs for adolescents (CDC, 1995d).
The National Center for Health Statistics, CDC, also sponsors health interviews with women regarding reproductive health issues. The National Survey of Family Growth provides important information regarding the self-reported prevalence of STDs and STD-related health behaviors among the general U.S. population. The National Survey of Adolescent Males is another federally funded survey that collects data on sexual behavior and contraceptive use from a nationally representative cohort of male adolescents 15-19 years old (Ku et al., 1992). Other health behavior surveys and studies that are not periodically administered have also produced important data regarding sexual behavior. For example, the National Health and Social Life Survey collected information from a probability sample of 3,432 American adults between 18 and 59 years of age in 1992 and produced the most comprehensive nationally representative data on sexual behavior among adults in the United States in many years (Laumann et al., 1994). In addition, a multiyear study, the National Adolescent Health Survey, jointly sponsored by several agencies, recently has completed data collection to examine the influence of family, peers, schools, and the community on adolescent health.
There is no evidence that participating in surveys of sexual behavior has a detrimental effect on sexual behavior. Halpern and others (1994) analyzed multiple study groups from three longitudinal studies of the effect of repeated administration of questionnaires regarding sexual behavior on male adolescents. They found no evidence that such questionnaires had an effect on sexual behavior.
Measuring and Evaluating Program Effectiveness
Data regarding federally supported projects collected by the CDC through state and local health departments consist primarily of output indicators that relate to laboratory reporting, community screening, case investigation, preventive and other clinical services, and gonorrhea, chlamydial, and HIV case detection. Other federal agencies, such as the Health Resources and Services Administration and the Health Care Financing Administration, collect information primarily related to the provision of direct health care services they support.
The committee is not aware of any nongovernmental organizations or associations that routinely collect data regarding STDs. However, the National Committee
|
BOX 5-2 Draft Description of HEDIS Measure for Chlamydial Screening Under Evaluation by the National Committee for Quality Assurance. Chlamydial Screening Chlamydial is not widely known, but it is an important health problem. It is the most common sexually transmitted bacterial disease in the United States, with an estimated 2 million new infections in women each year. It is usually a silent illness; about 70 percent of infected women have no symptoms. Left untreated, chlamydial can cause pelvic inflammatory disease, infertility, ectopic pregnancy (where the egg is implanted in the fallopian tube instead of in the womb), and chronic pelvic pain. Regular screening for the infection by testing for it during annual gynecological check-ups is often the only way to detect it so it can be treated before complications arise. Detection and treatment also help keep the person from spreading the disease. This measure estimates the percentage of women between the ages of 15 and 25 who were screened for chlamydial in the past year. This measure is being evaluated for inclusion in a future reporting set. Since sexually active women are the group of interest for chlamydial screening, a reliable method needs to be developed to distinguish women who are sexually active from those who are not. We also need to assess how reliably chlamydial screening is reported. These issues, among others, will be evaluated during the testing phase. NOTE: The above text is a direct quote from the primary source. The estimate of 2 million chlamydial infections, presumably for 1996, differs from the estimate of 2.6 million in 1994 cited previously by the committee. These estimates, however, are not necessarily inconsistent. SOURCE: NCQA, Committee on Performance Measurement. HEDIS 3.0 Draft for public comment. Washington, D.C.: National Committee for Quality Assurance, July 1996. |
for Quality Assurance, through the Committee on Performance Measurement, is currently evaluating a proposed STD-related performance measure for inclusion in subsequent versions of HEDIS. This performance measure on chlamydial screening, developed and submitted by the CDC, is presented in Box 5-2 and is in the "testing set" of HEDIS 3.0.
Another IOM committee, the Committee on Using Performance Monitoring to Improve Community Health, has evaluated the use of population-based performance monitoring to improve community health (IOM, 1996c). This committee examined capacities and processes for implementing and conducting performance monitoring. The final report, scheduled for release in 1997, will include proposed indicators for a community health profile and prototypical sets of indicators related to specific health issues.
Data Management, Utilization, and Limitations
In addition to uses associated with disease surveillance, data collected regarding STDs are primarily utilized for management of specific publicly sponsored disease control programs and for assessment of clinical performance of federally funded health care providers, such as community health centers. More specifically, with respect to the data collected on a national basis through the Division of STD Prevention at the CDC, existing management information systems are intended to support four primary service objectives in STD prevention: (1) preventing and containing early and congenital syphilis; (2) preventing and containing gonorrhea and pelvic inflammatory disease; (3) preventing and containing chlamydial infection and chlamydial-related sequelae such as pelvic inflammatory disease; and (4) preventing HIV infection through public STD clinics (Alan Friedlob, CDC, Division of STD Prevention, personal communication, February 1996).
To facilitate national data collection, the CDC provides management information software (STD*MIS) to state and local disease control programs to compile morbidity and service delivery output data. These data are intended to help state and local programs (a) track patient disease information and out-of-jurisdiction activity; (b) maintain files related to interview investigations, field investigations, morbidity, and laboratory surveillance; (c) allow electronic submission of data to the federal government; and (d) generate standard and customized reports.
However, while existing data systems may be adequate to support many of the internal management requirements outlined above, it is not clear that these data are adequate for overall program assessment. According to the Division of STD Prevention systems development staff, minimum national standards defining an adequate program for preventing STDs do not exist. STD-related services data and data collection procedures have been characterized as inflexible, relying on historic performance measures and focused on public STD clinics rather than linking data from all sites that provide STD-related services, including family planning agencies, emergency rooms, correctional institutions, substance use treatment programs, and other health care providers (Alan Friedlob, CDC, Division of STD Prevention, personal communication, February 1996). Additional deficiencies in STD-related data include lack of information on population subgroups, transmission-related behavior, services assessment, and outcomes related to specific interventions.
Information Systems Development
STD information systems development is, in concept and in application, inseparable from the larger community health information network development environment. Effective community health information network systems collect both population information and service and encounter data. This allows for (a)
integration of data across programs (e.g., tuberculosis, HIV, substance abuse); (b) efficient data-based decision-making; (c) application flexibility and systems compatibility (e.g., software independent of language or platform); (d) confidentiality safeguards; and (e) menus of indicators that are responsive to local needs. It should also be recognized that STD information systems development is occurring within a wider and rapidly changing planning environment, such as the Department of Health and Human Services proposed Performance Partnership Grants, the work of the Public Health Service's Public Health Data Policy Coordinating Committee, and the National Information Infrastructure and High Performance Computing and Communication initiatives (Braithwaite, 1995).
General issues to consider in developing and coordinating community-based information systems initiatives such as those mentioned above include:
- quality and usefulness of data;
- coordination with state- and national-level information systems;
- coordination of data collection processes, including collection of data from public and private sources;
- capacity to track outcome, process, and capacity measures in a timely manner;
- assurance of data privacy and security;
- development of standards for data elements, such as utilization of uniform diagnostic coding;
- resolution of proprietary concerns or ownership issues related to data;
- capacity to produce anonymous health data files for public health surveillance and other purposes; and
- flexibility to utilize grant funds to build and maintain information systems.
Local Data Analysis Capacity
The Division of STD Prevention at the CDC currently provides technical assistance to state and local programs implementing the division's management information software, "STD*MIS." Rather than technical assistance related to initiating or implementing information systems, it has been suggested that technical assistance is needed to develop local data analysis capacity so that data can be used to improve the effectiveness and efficiency of local STD programs (Alan Friedlob, CDC, Division of STD Prevention, personal communication, February 1996). With respect to analytic capacity, a survey of 65 STD project areas in December 1994 found that half had one half-time or less position dedicated to data analysis activities (CDC, Division of STD Prevention, unpublished data, 1995). Only 20 percent of projects surveyed had one or more full-time staff with master's or doctoral level training in epidemiology. The survey also found that 48 percent of STD programs derived epidemiological support exclusively from HIV/AIDS
programs. Finally, 44 percent of STD program managers stated that epidemiological support for their activities was inadequate.
Local capacity in data management and analysis is required if surveillance data are to be used as a program management tool. For example, the decline in rectal gonorrhea rates in many areas of the United States could be interpreted to be a result of the efficacy of HIV prevention activities among gay men. In order to make this assessment, however, local health districts need to have sufficient data management capacity both to collect data on gonorrhea incidence and STD-related behaviors, and to analyze data systematically.
A possible explanation for the lack of data analysis capacity is the dependency of information systems, such as STD*MIS, on state and local resources for maintenance and enhancement. The Council of State and Territorial Epidemiologists has reported that less than half of data resources at state and local levels is provided through federal resources (CSTE, 1995). The council further observed that almost one-half (46.4 percent) of all federal funding for state and local activities related to communicable disease data management was directed towards HIV/AIDS and that one-third of all federal funding was designated for HIV/AIDS. In addition, the council found that, with respect to data management, state and local governments were supporting 59 percent of funding for tuberculosis, 40.8 percent of funding for STDs, 44.2 percent of funding for vaccine-preventable diseases, and 86.7 percent of funding for all other communicable diseases.
Training and Education of Health Professionals
The spectrum of health care providers who are responsible for providing STD-related services includes physicians, nurse practitioners, nurses, physician assistants, and other professionals. The intensity and content of training and educational activities for health professionals vary considerably. Training may occur as part of the formal professional curricula or as part of continuing education activities.
Medical School and Medical Graduate Education
Two national committees in the United States have previously expressed concern regarding the adequacy of education in the area of STDs (Kampmeier, 1975; Work Group on Sexually Transmitted Disease, 1979). In addition, a 1980-1981 study indicated that medical school instruction regarding STDs was generally inadequate (Stamm et al., 1982). This survey of the infectious disease divisions of 122 U.S. medical schools and 15 Canadian medical schools collected information on preclinical and clinical training of medical students and clinical training of medical residents and other resident groups. Of the 127 infectious disease divisions responding to the survey, almost all offered preclinical training
consisting mostly of lectures on STD pathogens. Only 30 percent of U.S. medical schools had access to hospital-based or dedicated public STD clinics for teaching purposes. At these schools, only 30 percent of medical students received clinic-based training and the clinical experience offered to these students averaged only six hours. Thus, approximately 1 in 10 students received clinic-based STD training, and even this training was brief. Similarly, only 23 percent of internal medicine residencies in the United States offered specific clinical training in STD management and, even where available, this training only averaged 12 hours. Residents in other programs (dermatology, family practice, obstetrics and gynecology, pediatrics, and urology) were even less likely to be offered STD clinical training.
In 1991, 126 U.S. medical schools were again surveyed concerning clinical training offered medical students in the areas of STDs, including HIV infection (MacKay, 1995). The responses from 102 schools indicated that over the previous 10 years, the amount of clinical training of medical students in STDs had decreased at 6 percent of schools, remained the same at 17 percent, and increased at 77 percent of schools. Despite the increase in the number of schools that provided additional clinical training in STDs, several areas of STD-related education require improvement. For example, links to a clinic devoted specifically to STDs existed in only 42 percent of schools, and only 37 percent of schools offered students a clinical elective in STDs or HIV/AIDS. Programs for improving medical school and medical graduate training that focus on communication skills and involve a series of lectures, workshops, and role-playing have been developed (Steinberg et al., 1991; Ross and Landis, 1994).
Clinical training in dedicated public STD clinics, therefore, remains the exception rather than the rule for medical students in the United States. While infrequent encounters with patients with STDs in other ambulatory settings such as primary care, general medicine, or family medicine clinics (Berg et al., 1984) have been cited as reasons for difficulty in ensuring comprehensive training in STD management and prevention in these environments, there remains relatively little training in other aspects of STD prevention such as risk assessment or screening practices.
Primary Care Health Professional Training
Programs to train medical students, physicians, physicians assistants, nurse practitioners, nurses, and other primary care professionals in STD prevention are critical in increasing the capacity of the primary care system to address public health problems. The current system of clinical training for health care professionals is inadequate to produce effective primary care providers. For example, an IOM committee, charged to provide direction on improving primary care, came to the following conclusion regarding graduate medical education in the United States (IOM, 1996a:186-7):
Based on its public hearing and site visits, the committee shares with many medical educators and medical directors of integrated health care delivery systems concerns about traditional GME [graduate medical education], especially about the extent to which such training is preparing tomorrow's doctors for the new ways and settings in which they will be expected to function. Graduates of residency programs often lack knowledge of population-based health promotion and disease prevention, evidence-based clinical decision making, and patient interviewing skills (particularly communication and consultation skills).
The IOM committee on primary care recommended that primary care provider training be based on a common core set of clinical competencies, regardless of their disciplinary background, to be defined by a coalition of educational and professional organizations and accrediting bodies. Two specific areas of suggested emphasis were communication skills and cultural sensitivity (IOM, 1996a).
Primary care professionals (including family practitioners, internists, obstetrician/gynecologists, and pediatricians) see the majority of STDs in the United States (Berg, 1990), yet little research has been performed about STD management in primary care settings. It is likely that primary care clinicians in the private sector will be called upon to assume a greater role in STD prevention, including case management. However, because of the limited data regarding the knowledge, skills, and training needs of primary care professionals in these settings, an assessment of these issues is critical.
Primary care professionals often do not receive special training in STD risk assessment, diagnosis, and treatment. Such training, when it does exist, is often based within dedicated public STD clinics, and is not accessible to primary care professionals. Some of the federally funded STD training centers require a lengthy time commitment that limits participation by primary care providers. Further, diagnostic and management strategies and physician responsibilities appropriate for dedicated STD clinics often require modification for primary care practice (Berg, 1990).
Some primary care practices, such as community health centers in inner-city urban areas or programs for high-risk migrant populations, are the primary source of STD-related care in communities with high rates of STDs. Based on a chart review of all patient encounters in an inner-city Atlanta community health center during a four-week period in 1994, 10 percent of all patient encounters, and nearly all encounters among young men, were related to STD diagnosis and treatment (Kathleen Toomey, Georgia Department of Human Resources, Division of Public Health, unpublished data, 1996). However, patients frequently received inappropriate or inadequate treatment not consistent with published STD treatment guidelines. For example, half of all women with clinical diagnosis of pelvic inflammatory disease were treated inadequately for simple cervical infection. Clinicians in this clinic included nurse practitioners, internists, pediatricians, and family practitioners without specific training in STD clinical management. However, all staff expressed willingness to participate in training in STD diagnosis and treatment and adopted STD treatment guidelines when they were
made available. As previously mentioned, lack of physician compliance with standard practice for prevention and management of STDs has been documented (Gemson et al., 1991; Hessol et al., 1996) and additional training of clinicians in STD-related skills has been recommended (Boekeloo et al., 1991; Gemson et al., 1991; Steinberg et al., 1991; Hessol et al., 1996).
Federal Efforts in STD Training
Federal efforts to provide STD training have focused almost exclusively on training health professionals who provide services in the public sector. Since 1979, the CDC has funded 10 to 12 regional STD Prevention Training Centers. These centers have provided instruction composed of didactic lectures and clinic-based experiences to nearly 100,000 nurse practitioners, physician assistants, and physicians working in public health or family planning clinics throughout the United States. These centers are comanaged by medical schools and local or state health departments, but have not, until recently, specifically provided training to medical students or residents in training. In contrast, the AIDS Education and Training Centers funded by the Health Resources and Services Administration have focused on training primary care providers, but have not generally offered STD-related training. The Health Resources and Services Administration puts most of its sexuality-related training funds into programs related to family planning and HIV infection to the exclusion of other STDs. Generally, the National Institutes of Health's funding for STD-related training is primarily directed at training researchers, not clinicians.
To strengthen training of health professional students and trainees, the CDC plans a new initiative to support faculty positions in a limited number of medical centers to initiate clinical training of students and residents. However, the existing Regional STD Prevention Training Centers have not been adequately utilized to provide clinical training to health professional students and residents, who are often strongly motivated to obtain elective training in STDs. For example, the Seattle STD Prevention Training Centers cannot accommodate most of the medical and physician assistants and residents who seek training and have actually reduced training for these groups during the past year (King Holmes, University of Washington, personal communication, August 1996). To the committee's knowledge, no specific agency is responsible for training medical or other health professional students. Medical school curricula, which address human sexuality, do so from a perspective of sexual dysfunction and sexual ''deviance" rather than from a perspective of healthy sexuality. The CDC also supports training of disease intervention specialists, who generally coordinate HIV testing, provide patient education, and conduct partner notification for STDs, including HIV infection. This type of training support, however, is changing as described previously.
Advances in Information Technology
Advances in information technology are having a dramatic impact on how health-related information is disseminated among health professionals and have increased opportunities for their education. Telemedicine is rapidly being employed, especially in rural areas, to increase access of primary care providers to the expertise of specialists in medical centers (IOM, 1996b; Crump and Pfeil, 1995). The proliferation of the Internet has dramatically increased access to information that was previously only accessible at libraries and information reference centers. In addition, information technology has enabled many primary care professionals to learn new skills or improve current practice through distance learning activities such as televised courses. The CDC has recently initiated distance learning activities employing teleconferencing, use of the Internet, CD-ROM, and other information technologies to improve STD education.
Funding of Services
Federal, State, and Local Government Funding
Publicly funded STD programs have had several notable successes (CDC, DSTDP, 1995). Beginning in the late 1940s, and continuing through the early 1950s, aggressive national programs against syphilis succeeded in nearly eliminating early syphilis in the United States. Only 10 years after the number of cases of primary and secondary syphilis peaked at 94,957 cases in 1946, cases were reduced to 6,392 in 1956 (CDC, DSTDP, 1995). At that time, the apparent success of the program may have led to an assumption that syphilis was no longer a problem, with the result that funding was sharply reduced (CDC, DSTD/HIVP, 1995). Four years later, the number of cases of primary and secondary syphilis in the United States nearly tripled (CDC, DSTDP, 1995). In the early 1970s, and continuing through the early 1980s, national programs against gonorrhea were also initiated. Along with a number of other factors such as the decreasing proportion of the population in the age groups at greatest risk, safer sexual practices related to HIV concerns, and changes in contraceptive methods, this national program contributed to a substantial decline in gonorrhea cases. The number of reported cases dropped 59 percent from 1,013,436 cases in 1978 to 418,068 in 1994 (CDC, DSTDP, 1995).
In fiscal year 1995, the federal budget for the CDC's Division of STD Prevention, the lead federal agency for STD prevention, was $105.2 million. Of that amount, approximately $91.8 million (87 percent) was awarded in grants by the CDC to states and cities for STD prevention activities.6 The National Institute of
Allergy and Infectious Diseases and other divisions at the National Institutes of Health, which are responsible for supporting most STD-related biomedical and clinical research activities in the federal government, invested approximately $105.4 million in the same year for biomedical and clinical research in STDs. In addition, other federal agencies, such as the Health Resources and Services Administration, Office of Population Affairs, the Health Care Financing Administration (primarily through its Medicaid program), and the Indian Health Service, all directly support or provide STD-related clinical services. The amount of funds that support STD-related services in these agencies, however, is unclear because such services are provided in the context of primary care or other programs and are not allocated or accounted for separately. The proportion of federal funds that is used to support prevention activities versus other services is likewise unclear, but it is reasonable to broadly categorize funds allocated for the Division of STD Prevention at the CDC and National Institutes of Health STD-related grants as related to noncurative prevention services and to research, respectively, and funds originating from the other federal agencies as primarily used to support clinical services for STDs.
The precise amount of financial support for STD-related programs, including both curative and noncurative services at the state and local levels, is unknown because there is no matching requirement for most federal funding. State and local governments vary widely in their financial support for STD-related programs. Some jurisdictions spend several times more than they receive from the CDC, while others only provide a small proportion of the total funding for such programs in their area. Based on an informal CDC survey of state and local health departments regarding their contributions to STD program funding in 1994, the total state and local contribution to STD-related programs was approximately $125.6 million or approximately 58 percent of combined state, local, and federal funding (CDC, Division of STD Prevention, unpublished data, April 1994). State and local contributions, as a percentage of combined state, local, and federal funding in the respective area, ranged from 0 percent ($0) to 90 percent ($22.7 million). These estimates are sensitive to variability in how STD program funding is categorized in state and local government budgets. However, in order to provide a rough estimate of public investment in STD prevention (including STD treatment),7 it is reasonable to use the estimated state and local contribution in 1994 ($125.6 million) and the actual CDC contribution to state and local STD programs in federal fiscal year 1995 ($91.8 million).
Given the assumptions mentioned above, the total national public investment in STD prevention in fiscal year 1995 was approximately $230.8 million, and approximately $105.4 million was invested in biomedical and clinical research in STDs. Comparing these estimates to the estimated total costs of selected STDs, excluding AIDS ($9.954 billion), the total cost associated with STDs in the United States in 1994 was approximately 43 times the total national public investment in STD prevention and 94 times the total national investment in STD-related research (Figure 5-3). Again, it should be noted that the estimate of public investment in STD prevention and research does not include all publicly funded prevention or research programs that are related to STDs or STD-related programs funded by the private sector.8 Similarly, the estimate of total costs for STDs does not include costs for all STDs. Even if the true public investment in STD prevention and research is several times higher than that estimated by the committee, the public investment would still be extremely low compared to the total costs of STDs in the United States.
Categorical Funding
Funding for state and local health departments comes from the CDC cooperative agreements and state and local governments. The legislative authority for the prevention of STDs in the United States stems from Section 318 of the Public Health Service Act, which authorizes the Department of Health and Human Services to make grants to and assist states, their political subdivisions, and public and nonprofit private entities for STD prevention research, demonstrations, public information and education programs, and training, education, and clinical skill improvement of health care providers. The Department of Health and Human Services is also authorized to make grants to states and their political subdivisions to carry out prevention programs. A list of prevention programs funded by the CDC's Division of STD Prevention in fiscal year 1996 is presented in Table 5-1.
The Preventive Health Amendments of 1992 modified Section 318 and authorized the CDC to make grants and provide assistance for activities to reduce STDs that can cause infertility in women. These amendments also authorized grants for the purpose of conducting research to improve the delivery of STD-related infertility prevention services.
Reimbursement for STD-related services in the private sector comes from
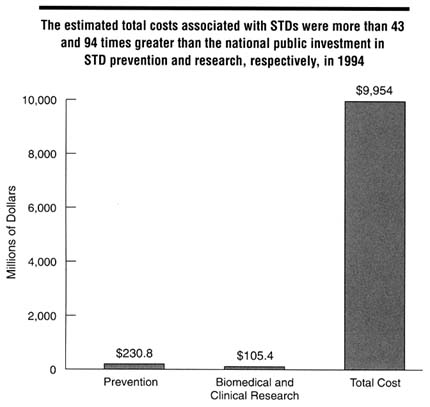
Figure 5-3
Comparison of estimated annual direct and indirect costs for selected STDs and their complications in 1994 versus national public investment in STD prevention and research in federal fiscal year 1995. NOTE: The estimate for investment in STD prevention provided here represents public funding for all interventions, behavioral, curative, or otherwise, that are needed to reduce the spread of infection in a population. SOURCES: Total cost of illness estimate was calculated by the IOM Committee on Prevention and Control of STDs; estimate of federal, state, and local investment in STD prevention was based on unpublished data from the CDC, Division of STD Prevention, 1996; and estimate of national investment in research was based on unpublished data from the National Institutes of Health, 1996.
third party reimbursement, such as private health insurance and Medicaid. STD-related care provided in community-based health facilities, such as family planning clinics and community health centers, receive federal and other support. Local health departments receive reimbursement for services provided by public STD clinics to persons with private insurance only to the extent allowed by law or under written contract.
TABLE 5-1 STD Prevention Programs Funded by the Division of STD Prevention, Centers for Disease Control and Prevention (CDC), Fiscal Year 1996
|
Program |
Funding Level ($) |
|
Preventive Health Services |
|
|
STD Accelerated Prevention Campaign |
69,954,310 |
|
Prevention of Infertility Caused by STDs |
9,798,309 |
|
Intra-agency Agreement: Office of Population Affairs and the CDC |
1,700,000 |
|
The National AIDS, STD, and National Immunization Program Hotline |
715,390 |
|
Intra-agency Agreement: Indian Health Service (IHS) and the CDC |
325,000 |
|
Human Resources Development |
|
|
STD/HIV Prevention Training Centers |
4,008,547 |
|
Public Health Graduate Training Certification Program |
2,965,623 |
|
Sexually Transmitted Diseases Faculty Expansion Program |
648,070 |
|
Association of Schools of Public Health (ASPH) |
400,000 |
|
Association of Teachers of Preventive Medicine (ATPM) |
22,500 |
|
Extramural Research and Demonstrations |
|
|
STD Accelerated Prevention Campaign Enhanced Projects |
1,463,510 |
|
Research and Evaluation Issues in Prevention of Infertility Due to STDs |
1,275,000 |
|
STD Accelerated Prevention Campaign: Enhanced Projects for STD Prevention in High-Risk Youth |
1,025,188 |
|
Innovations in Syphilis Prevention in the United States: Reconsidering the Epidemiology and Involving Communities |
1,000,000 |
|
Development and Feasibility Testing of Interventions to Increase Health-Seeking Behaviors in, and Health Care for, Populations at High Risk for Gonorrhea |
750,000 |
|
STD APC Enhanced Projects: Jail STD Prevalence Monitoring |
125,000 |
|
Total Program Funding |
96,176,447 |
|
SOURCE: CDC, Division of STD Prevention. Unpublished data, October 1996. |
|
Block Grant Proposals
Current proposals from the Department of Health and Human Services would consolidate many federal categorical programs into block grants that each state would allocate among competing health needs and among local public and private sector agencies. A Senate legislative proposal would consolidate and replace 12 CDC categorical programs with 1 or 2 block grants. The Department of Health and Human Services advocates consolidating these CDC programs into three new public health "Performance Partnership Grants," including one for HIV/AIDS, STD, and TB. Performance Partnership Grants are essentially block grants that
require states to set health objectives in an interactive process with the Department of Health and Human Services, providing some federal oversight but few constraints on state policies, programs, or funding.
Arguments for Block Grants
Proponents of block grants argue that categorical funding has forced programmatic rigidity and excess administrative costs on local programs. Categorical funding, proponents of block grants argue, imposes a bureaucratic straitjacket on public health and safety-net programs, forcing local programs and services into one-size-fits-all national models, ignoring local conditions and slowing innovation. In addition to restraining innovation and modifications to meet local needs, grant applications for federal funding are time-consuming, imposing substantial administrative costs on local agencies. State allocation of block grant funds may simplify the application process.
Categorical funding also encourages narrowly defined programs even when it is logical to merge staff and services. For example, many states have kept HIV and STD prevention programs completely separate, although most observers acknowledge that it is logical to coordinate programs to prevent HIV infection and other STDs because they share common modes of transmission and risk groups and many common interventions. By measuring accountability in terms of the number of persons who receive a service or educational program, federal categorical funding encourages state and local agencies to keep programs separate.
Some observers believe that block grants will free local communities from the rigidity and administrative burdens of categorical funding. States, they argue, will allocate funds based on locally and professionally determined health and social needs and will be responsive to state and local conditions. Whereas categorical programs subordinate local needs to uniform federal requirements imposed by distant bureaucrats, block grant supporters believe state officials will allocate federal and state moneys guided by the technical assessments of state health agencies, the judgments of the public health professionals, and the views of local communities.
Arguments Against Block Grants
Opponents of consolidating STD funding into a block grant along with other public health programs believe that STDs will suffer in competition with less controversial public health problems or other state priorities. In the real world of allocating budgets and setting priorities, they are concerned that elected state officials will make funding decisions based more on political considerations than on assessments by public health professionals and agencies. STD programs traditionally have weak political constituencies and will suffer in competition with
programs that have powerful constituencies. As discussed in Chapter 3, advocacy for STD funding has been traditionally weak because many patients infected with STDs are unaware of the infection, and those who are rarely want to disclose their infection in public, let alone organize public support for STD funding.
Opponents of block grants are particularly concerned that socially conservative interest groups will prevail in lobbying against STD programs at the state level. Allowing states to set funding allocations would also increase the already wide variability in STD programs among the states, because some states may seriously neglect STD programs. In addition, consolidating STD funding into a block grant may also result in the dissolution of the relatively weak constituency groups fighting for STD funding. The Coalition to Fight STDs, an alliance of more than 40 groups organized by the American Social Health Association, monitors public sector efforts against STDs and advocates at the national level for funding for STD prevention, treatment, and research. STD coalitions at the state or local level are rare, although they are emerging in some states, such as North Carolina, to improve STD funding.
Lastly, opponents suggest that past experience with other block grants may portend the fate of STD programs in a consolidated state grants program. For example, in 1981, the categorical Lead-Based Paint Poisoning prevention program was folded into the newly created Preventive Health and Health Services block grant, and the Urban Rodent Control program was folded into the new Maternal and Child Health block grant. These efforts lost funds in virtually all states after the federal categorical programs that funded these services were folded into the block grants. Both of these programs were widely viewed as federal "big-city" programs that found little support in state legislatures dominated by rural representation. Programs that had state support before the advent of federal categorical programs fared better than those that previously had little or no state funding (U.S. General Accounting Office, 1984; Peterson et al., 1986; Elling and Robins, 1991).
Conclusions
Current STD prevention services comprise several disjointed components, including provision of clinical services, disease surveillance and information collection activities, training and education of health care professionals, and funding of activities and programs. Although these components are largely publicly sponsored programs, they involve all levels of government and the private sector.
Dedicated public STD clinics have been instrumental in public efforts against STDs since they were established several decades ago. The quality and effectiveness of services delivered in these settings, however, are extremely variable and clearly need significant improvement. Until universal health care coverage is implemented in the United States, the function of public clinics as providers of
care to the uninsured will need to be preserved. Unlike dedicated public STD clinics, community-based clinics and private health plans provide STD-related services in the context of primary care. However, the scope and quality of services provided in these environments are unknown. It is evident that clinical preventive services in both public and private health care settings need to be expanded and improved. Risk assessment and counseling to effect behavior change remain underutilized by primary care professionals, in part because the providers are poorly trained in their use.
Data regarding the scope and quality of STD-related services among managed care organizations and other health plans are limited, but data collected by the committee and other information suggest that, with several notable exceptions, even managed care organizations that serve high-risk populations are not providing comprehensive services to infected persons and their partners in a consistent manner. Most managed care organizations and other health plans do not currently give STDs sufficiently high priority. One of the more notable potential advantages of increasing the role of managed care organizations in providing STD-related services is the opportunity to increase accountability, particularly with the support of employers and other purchasers of health care. The traditional role of dedicated public STD clinics and some of the functions of public health agencies will likely change given the national trend towards managed care, especially the increasing enrollment of Medicaid beneficiaries into managed care plans. This change in the health care delivery environment is both an opportunity to improve services and a cause for concern that the "safety net" for essential public health services will be eroded.
Surveillance and information systems provide the basis for public health decision-making and function as the backbone for an effective system of STD prevention. An extensive system of data collection has been developed based on passive surveillance (with biases and incomplete data), sentinel surveys, and population-based surveys. However, specific improvements in these components are needed. Improvements are also needed in the current system for training and educating health professionals to deliver high-quality STD-related clinical services.
Even in an era of shrinking federal and state budgets, the current investment in STD prevention is extremely low when compared to the enormous economic consequences of these diseases. Proposals to consolidate federal funding for STD programs to the states in the form of block grants have serious flaws, given the lack of adequate accountability. The current system of categorical funding, however, needs to be substantially improved.
References
Anderson JE, McCormick L, Fichtner R. Factors associated with self-reported STDs: data from a national survey. Sex Transm Dis 1994;21:303-8.
Armstrong K, Samost L, Bencivengo M. Integrating family planning services into drug treatment programs. In: Epidemiologic trends in drug abuse. Proceedings Community Epidemiology Work Group. Rockville, MD: National Institute on Drug Abuse, June 1992:548-59.
Aseltyne WJ, Cloutier M, Smith MD. HIV disease and managed care: an overview. J Acquire Immune Defic Syndr Hum Retrovirol 1995;8[Suppl 1]:S11-22.
ARHP, NANPRH (Association of Reproductive Health Professionals and National Association of Nurse Practitioners in Reproductive Health). STD counseling practices and needs survey: a national survey of primary care physicians and other health care providers. Conducted by Schulman, Ronca, and Bucuvalas, Inc. , Silver Spring, MD. June 28, 1995.
Benson Gold R, Richards CL. Improving the fit. Reproductive health services in managed care settings. New York: Alan Guttmacher Institute, 1996.
Berg AO. The primary care physician and sexually transmitted diseases control. In: Holmes KK, Mårdh PA, Sparling PF, Wiesner PJ, Cates W Jr, Lemon SM, et al., eds. Sexually transmitted diseases. 2nd ed. New York: McGraw-Hill, Inc., 1990:1095-8.
Berg AO, Heidrich FE, Fihn SD, Bergman JJ, Wood RW, Stamm WE, et al. Establishing the cause of genitourinary symptoms in women in a family practice. Comparison of clinical examination and comprehensive microbiology. JAMA 1984;251:620-5.
Boekeloo BO, Marx ES, Kral AH, Coughlin SC, Bowman M, Rabin DL. Frequency and thoroughness of STD/HIV risk assessment by physicians in a high-risk metropolitan area. Am J Public Health 1991;81:1645-8.
Bowman MA, Russell NK, Boekeloo BO, Rafi IZ, Rabin DL. The effect of educational preparation on physician performance with a sexually transmitted disease-simulated patient. Arch Intern Med 1992;152:1823-8.
Braithwaite WR. Health information systems. Presentation delivered at the Conference on Strengthening Public Health Infrastructure, July 28, 1995, Chicago.
Brandt AM. No magic bullet: a social history of venereal disease in the United States since 1980. New York: Oxford University Press, Inc., 1985.
Carey TS, Weis K. Diagnostic testing and return visits for acute problems in prepaid, case-managed Medicaid plans compared to fee-for-service. Arch Intern Med 1990;150:2369-72.
CDC (Centers for Disease Control and Prevention). Sexually transmitted diseases. Clinical practice guidelines. Department of Health and Human Services, Public Health Service. Atlanta: Centers for Disease Control, May 1991.
CDC. 1993 Sexually transmitted diseases treatment guidelines. MMWR 1993;42(No. RR-14):56-66.
CDC. The future role of CDC field assignees. Internal memo from Dr. David Satcher, Director of CDC, May 25, 1995a.
CDC. Prevention and managed care: opportunities for managed care organizations, purchasers of health care, and public health agencies . MMWR 1995b;44:(No. RR-14):1-12.
CDC. Summary of notifiable disease reporting, United States, 1994. MMWR 1995c;43:1-80.
CDC. Youth risk behavior surveillance-United States, 1993. MMWR 1995d;44:(No. SS-1).
CDC, DSTD/HIVP (Division of STD/HIV Prevention). Annual report 1991. U.S. Department of Health and Human Services, Public Health Service. Atlanta: Centers for Disease Control, 1992.
CDC, DSTD/HIVP. Annual report 1994. U.S. Department of Health and Human Services, Public Health Service. Atlanta: Centers for Disease Control and Prevention, 1995.
CDC, DSTDP (Division of STD Prevention). Sexually transmitted disease surveillance 1994. U.S. Department of Health and Human Services, Public Health Service. Atlanta: Centers for Disease Control and Prevention, 1995.
CDC, DSTDP. Guidance for requesting direct and financial assistance personnel. Draft document, October 1996.
Celum CL, Hook EW, Bolan GA, Spauding CD, Leone P, Henry KW, et al. Where would clients seek care for STD services under health care reform? Results of a STD client survey from five clinics. Eleventh Meeting of the International Society for STD Research, August 27-30, 1995; New Orleans, LA [abstract no. 101].
CSTE (Council of State and Territorial Epidemiologists). Blueprint for a national health surveillance system for the 21st century. Washington, D.C.: CSTE, May 1995.
County of Los Angeles Department of Health Services. Draft proposal of the agreement between the County of Los Angeles and plan. March 29, 1995.
Crump WJ, Pfeil T. A telemedicine primer. An introduction to the technology and an overview of the literature. Arch Fam Med 1995;4:796-803; discussion 804.
DeBuono BA, Zinner SH, Daamen M, McCormack WM. Sexual behavior of college women in 1975, 1986, and 1989. N Engl J Med 1990;322:821-5.
Delbanco S, Smith MD. Reproductive health and managed care-an overview. West J Med 1995;163[3 Suppl]:1-6.
Donovan P. Taking family planning services to hard-to-reach populations. Fam Plann Perspect 1996;28:120-6.
Elling RS, Robins LS. The politics of federalism and intergovernmental relations in health. In: Litman TJ, Robins LS, eds. Health politics and policy. Albany, NY: Delmar Publishers, 1991:200-5.
Fisher RS. Medicaid managed care: the next generation? Acad Med 1994;69:317-22.
Gemson DH, Colombotos J, Elinson J, Fordyce EJ, Hynes M, Stoneburner R. Acquired immunodeficiency syndrome prevention. Knowledge, attitudes, and practices of primary care physicians. Arch Intern Med 1991;151:1102-8.
GHAA (Group Health Association of America [currently American Association of Health Plans]). 1995 National directory of HMOs. Washington, D.C.: Group Health Association of America, 1995.
Halpern CT, Udry JR, Suchindran C. Effects of repeated questionnaire administration in longitudinal studies of adolescent males' sexual behavior. Arch Sex Behav 1994;23:41-57.
Hessol NA, Priddy FH, Bolan G, Baumrind N, Vittinghoff E, Reingold AL, et al. Management of pelvic inflammatory disease by primary care physicians: a comparison with Centers for Disease Control and Prevention Guidelines. Sex Transm Dis 1996;23;157-63.
Igoe, JB. A closer look: a preliminary report of some of the findings from the national survey of school nurses and school nurse supervisors . Denver: University of Colorado Health Sciences Center, Office of School Health, 1994.
IOM (Institute of Medicine). For-profit enterprise in health care. Gray BH, ed. Washington, D.C.: National Academy Press, 1986.
IOM. Primary care: America's health in a new era. Donaldson MS, Yordy KD, Lohr KN, Vanselow NA, eds. Washington, DC: National Academy Press, 1996a.
IOM. Telemedicine. A guide to assessing telecommunications in health care. Field MJ, ed. Washington, DC: National Academy Press, 1996b.
IOM. Using performance monitoring to improve community health: exploring the issues. Workshop summary. Durch JS, ed. Washington, D.C.: National Academy Press, 1996c.
Johnson R, Lee F, Hadgu A, McQuillan G, Aral S, Keesling S, et al. U.S. genital herpes trends during the first decade of AIDS-prevalences increased in young whites and elevated in blacks. Proceedings of the Tenth Meeting of the International Society for STD Research, August 29-September 1, 1993, Helsinki [abstract no. 22].
The Kaiser Commission on the Future of Medicaid. Medicaid and managed care: lessons from the literature. Menlo Park, CA: The Henry J. Kaiser Family Foundation , 1995.
Kampmeier RH. The national commission on venereal disease. J Am Vener Dis Assoc 1975;1:99-104.
Kassler WJ, Zenilman JM, Erickson B, Fox R, Peterman TA, Hook EW 3rd. Seroconversion in patients attending sexually transmitted disease clinics. AIDS 1994;8:351-5.
Keeling RP. College health responds to HIV; hard lessons and a rich heritage. J Am Coll Health 1995;43:271-2.
Ku L, Sonenstein FL, Pleck JH. Patterns of HIV risk and preventive behaviors among teenage men. Public Health Rep 1992;107:131-8.
Laumann EO, Gagnon JH, Michaels RT, Michael S. The social organization of sexuality: sexual practices in the United States. Chicago, IL: University of Chicago Press, 1994.
Leavy Small M, Smith Majer L, Allensworth DD, Farquhar BD, Kann L, Pateman BC. School health services. J Sch Health 1995;65:319-26.
Lewis CE, Freeman HE. The sexual history-taking and counseling practices of primary care physicians. West J Med 1987;147:165-7.
Lewis CE, Freeman HE, Corey CR. AIDS-related competence of California's primary care physicians. Am J Public Health 1987;77:795-9.
MacKay HT, Toomey KE, Schmid GP. Survey of clinical training in STD and HIV/AIDS in the United States. Proceedings of the IDSA Annual Meeting; September 16-18, 1995, San Francisco [abstract no. 281].
Marcus AC, Crane LA, Kaplan CP, Goodman KJ, Savage E, Gunning J. Screening for cervical cancer in emergency centers and sexually transmitted disease clinics. Obstet Gynecol 1990;75:453-5.
McLean DA. A model for HIV risk reduction and prevention among African American college students. J Am Coll Health 1994;42:220-3.
McQuillan GM, Khare M, Ezzati Rice TM, Karon JM, Schable CA, Murphy RS. The seroepidemiology of human immunodeficiency virus in the United States household population: NHANES III, 1988-1991. J Acquire Immune Defic Syndr 1994;7:1195-201.
McQuillan GM, Townsend TR, Fields HA, Carroll M, Leahy M, Polk BF. Seroepidemiology of hepatitis B virus infection in the United States. 1976 to 1980. Am J Med 1989;87:5S-10S.
NCQA (National Committee for Quality Assurance), Committee on Performance Measurement. HEDIS 3.0 Draft for Public Comment, Washington, D.C.: National Committee for Quality Assurance, July 1996.
Noegel R, Kirby J, Schrader M, Wasserheit J. Sexually transmitted disease accelerated prevention campaigns. Opportunities to expand prevention efforts in the United States. Sex Transm Dis 1993;20:118-9.
Peterson GE, Bovbjerg RR, Davis BA, Davis WG, Durman EC, Gullo TA. The Reagan block grants: what have we learned? Washington, D.C.: The Urban Institute, 1986.
Rabin DL, Boekeloo BO, Marx ES, Bowman MA, Russell NK, Gonzalez Willis A. Improving office-based physicians' prevention practices for sexually transmitted diseases. Ann Intern Med 1994;121:513-9.
Reinisch JM, Hill CA, Sanders S, Ziemba-Davis M. High-risk sexual behavior at a Midwestern university: A confirmatory study. Fam Plann Perspect 1995;27:79-82.
Ross PE, Landis SE. Development and evaluation of a sexual history-taking curriculum for first- and second-year family practice residents. Fam Med 1994;26:293-8.
Roter DL, Knowles N, Somerfield M, Baldwin J. Routine communication in sexually transmitted disease clinics: an observational study. Am J Public Health 1990;80:605-6.
Russell NK, Boekeloo BO, Rafi IZ, Rabin DL. Unannounced simulated patients' observations of physician STD/HIV prevention practices. Am J Prev Med 1992;8:235-40.
Schlitt JJ, Rickett KD, Montgomery LL, Lear JG. State initiatives to support school-based health centers: a national survey . J Adolesc Health 1995;17:68-76.
Scholes D, Stergachis A, Heidrich FE, Andrilla H, Holmes KK, Stamm WE. Prevention of pelvic inflammatory disease by screening for cervical chlamydial infection. New Engl J Med 1996;334:1362-6.
SFPA (State Family Planning Administrators). Family planning clinic provision of STD and HIV services: national questionnaire findings. Seattle, WA, February 1991.
Stamm WE, Kaetz SK, Holmes KK. Clinical training in venereology in the United States and Canada. JAMA 1982;248:2020-4.
Stein ZA. Editorial: family planning, sexually transmitted diseases, and prevention of AIDS-divided we fail? Am J Public Health 1996;86:783-4.
Steinberg JK, Wellman J, Melrod J. A proposal for strengthening medical school training in STD prevention techniques. Public Health Rep 1991;106:196-202.
Turner JC, Garrison CZ, Korpita E, Waller J, Addy C, Hill WR, et al. Promoting responsible sexual behavior through a college freshman seminar. AIDS Educ Prev 1994;6:266-77.
Upchurch DM, Farmer MY, Glasser D, Hook EW. Contraceptive needs and practices among women attending an inner-city STD clinic. Am J Public Health 1987;77:1427-30.
U.S. General Accounting Office. Maternal and child health block grant: program changes emerging under state administration. Washington, D.C.: General Accounting Office, 1984.
Weinstock HS, Sidhu J, Gwinn M, Karon J, Petersen LR. Trends in HIV seroprevalence among persons attending sexually transmitted disease clinics in the United States, 1988-1992. J Acquire Immune Defic Syndr Hum Retrovirol 1995;9:514-22.
Winkenwerder W, Levy B, Eisenberg JM, Williams SV, Young MJ, Hershey JC. Variation in physicians' decision-making thresholds in management of a sexually transmitted disease. J Gen Intern Med 1993;8:369-73.
Work Group on Sexually Transmitted Disease. Conference on preventing disease/promoting health-objectives for the nation: sexually transmissible diseases . Sex Transm Dis 1979;6:273-7.
Zabin LS, Stark HA, Emerson MR. Reasons for delay in contraceptive clinic utilization. Adolescent clinic and nonclinic populations compared. J Adolesc Health 1991;12:225-32.
Zenilman JM, Hook EW 3rd, Shepherd M, Smith P, Rompalo AM, Celentano DD. Alcohol and other substance use in STD clinic patients: relationships with STDs and prevalent HIV infection. Sex Transm Dis 1994;21:220-5.
Zimmerman DJ, Reif CJ. School-based health centers and managed care health plans: partners in primary care. J Public Health Manage Pract 1995;1:33-9.













































